Abstract
Two epithelial cell types cover the alveolar surface of the lung. Type II alveolar epithelial cells produce surfactant and, during development or following wounding, give rise to type I cells that are involved in gas exchange and alveolar fluid homeostasis. In culture, freshly isolated alveolar type II cells assume a more squamous (type I-like) appearance within 4 days after plating. They assemble numerous focal adhesions that associate with the actin cytoskeleton at the cell margins. These alveolar epithelial cells lose expression of type II cell markers including SP-C and after 4 days in culture express the type I cell marker T1α. Those cells that express T1α also deposit fibers of laminin-311 in their matrix. The latter appears to be related to their development of a type I phenotype because freshly isolated, primary type I cells also assemble laminin-311-rich fibers in vitro. A β1 integrin antibody antagonist inhibits the assembly of laminin-311 matrix fibers. Moreover, the formation of laminin fibers is dependent on the activity of the small GTPases and is perturbed by ML-7, a myosin light chain kinase inhibitor. In summary, our data indicate that assembly of laminin-311 fibers by lung epithelial cells is integrin and actin cytoskeleton dependent, and that these fibers are characteristic of type I alveolar cells.
T
Type I and type II cells line a common basement membrane (BM) composed of collagen type Iv, perlecan, entactin/nidogen, and several laminin isoforms. Laminins are a large family of heterotrimeric matrix proteins composed of α, β, and γ subunits (Tunggal et al. 2000; Aumailley et al. 2005). There are at least five α subunits, three β subunits, and three γ subunits, allowing as many as 45 different isoforms of laminin to be assembled, of which 15 have been identified (Tunggal et al. 2000; Aumailley et al. 2005). Laminins play a structural role in tissue organization and appear to stabilize epithelial cell interaction with the BM (McGowan and Marinkovich 2000). However, there is now emerging data that laminins also play an important role in morphogenesis and tissue remodeling via stimulation of various signaling cascades (see, for example, Boudreau and Bissell 1998; Goldfinger et al. 1999; Belkin and Stepp 2000; Muschler et al. 2002).
In the lung, laminin subunits show tissue-specific and developmentally regulated patterns of expression (Virtanen et al. 1996; Miner et al. 1997; Pierce et al. 1998; Nguyen et al. 2002a). For example, laminin α1 expression is restricted to early human lung morphogenesis, whereas laminin α3 and α5 expression are continuous from early lung development throughout adulthood (Virtanen et al. 1996; Pierce et al. 1998; Miosge et al. 2002; Nguyen et al. 2002b). Indeed, some authors have speculated that alveolar cells may express at least five laminin isoforms (Pierce et al. 1998). The complexity of expression of laminins in developing and adult lung BM implies that laminins play important functions in the airway. In support of this idea, studies using transgenic mice indicate that the α5 laminin subunit is involved in lung septation and visceral pleura BM formation, although what functions other laminins perform in the developing and developed lung remain unclear (Nguyen et al. 2000). Moreover, our recent work indicates that α3 subunit-containing laminins transduce mechanosignals via dystroglycan in alveolar cells maintained in vitro (Jones et al. 2005).
In this study we have analyzed the ability of primary alveolar epithelial cells to assemble a fibrillar laminin-311 (previously designated laminin-6)-rich matrix (Aumailley et al. 2005; Jones et al. 2005). Our data indicate that type I-like cells secrete and organize laminin-311 in complex networks in their extracellular matrix. Furthermore, we show here that laminin-311 fibers are assembled in a β1 integrin-dependent manner, and that this assembly requires activity of the small GTPases and the actin cytoskeleton.
Materials and Methods
Cell Culture
Alveolar epithelial type II cells were isolated from pathogenfree male Sprague Dawley rats (200-225 g), as previously described (Dobbs 1990). Briefly, the lungs were perfused via the pulmonary artery, lavaged, and digested with elastase (3 U/ml) (Worthington Biochemical; Lakewood, NJ). Alveolar epithelial cells were purified by differential adherence to IgG-pretreated dishes, and cell viability was assessed by trypan blue exclusion (>95%).
Alveolar epithelial type I cells were also isolated from Sprague Dawley rats using a procedure detailed by others (Chen et al. 2004). In brief, excised lungs from male Sprague Dawley rats were perfused via the pulmonary artery and subsequently lavaged to remove contaminating macrophages. Cells were disaggregated by enzymatic digestion using 16 U/ml elastase (Worthington Biochemical). Lung tissue was dissected from the airways, minced to 1-mm3 fragments, and filtered through layers of cotton gauze and 150-μm nylon mesh (Sefar America; Depew, NY). After centrifugation, cells were incubated on bacteriological Petri dishes coated with rat IgG (Sigma-Aldrich; St Louis, MO) and lightly panned to collect non-adherent cells. Type I cells were isolated from the remaining cells by positive immunoselection using a polyclonal antibody against T1α, followed by an incubation with rat anti-mouse IgG magnetic beads prior to magnetic selection (Dynal Biotech; Invitrogen, Carlsbad, CA). Type I cells were released from the magnetic beads using a DNAase I-releasing buffer supplied by the manufacturer.
Animal studies conformed to National Institutes of Health Guidelines for the Care and Use of Laboratory Animals (1985).
Type I and type II cells were resuspended in DMEM (Cellgro; Herndon, VA) containing 10% FBS (Hyclone; Logan, UT) with 2 mM glutamine, 100 U/ml penicillin, 0.25 μg/ml amphotericin B, and 100 μg/ml streptomycin (Gibco; Carlsbad, CA). Approximately 6 × 104 cells were plated per 35-mm tissue culture dish. Cells were incubated in a humidified atmosphere of 5% CO2/95% air at 37C. The day of isolation and plating is designated culture day 0.
Adenovirus
Adenoviruses containing the N19 dominant negative mutant of RhoA and the N17 dominant negative mutants of Rac1 and Cdc42 were gifts of Dr. Anne Ridley, University College (London, UK). Adenovirus encoding dystroglycan silencing RNA and control silencing RNA were described and characterized by us elsewhere (Jones et al. 2005). Cells were infected with the virus as previously detailed at a multiplicity of infection of 1:10 at 2 days after plating (Jones et al. 2005). The following day, the medium was replaced and assays were performed 24 hr later.
Treatment with Cytoskeletal Inhibitors
Myosin light chain kinase (MLCK) inhibitor ML-7 was purchased from Calbiochem-Novabiochem Corp. (San Diego, CA). Alveolar epithelial cells were treated with ML-7, 2.5 μM, diluted into medium from a stock of 40 mM water at 2 days after plating so as to allow the cells to attach and spread prior to treatment. Control cells were treated with the corresponding amount of vehicle (water).
Antibodies and Other Reagents
The mouse monoclonal antibody (MAb) CM6 against the laminin α3 subunit was described previously (Baker et al. 1996). A hamster MAb (Ha2/5) that inhibits the function of rat β1 integrin was purchased from BD Pharmingen (Franklin Lakes, NJ). A rabbit antiserum against the β1 laminin subunit and mouse MAb against the γ1 laminin subunit (MAB1920) were obtained from Dr. Peter Yurchenco (Department of Pathology, Robert Wood Johnson Medical School, Piscataway, NJ and Chemicon International; Temecula, CA), respectively. MAb against T1α (RT140) was the generous gift of Dr. Leland Dobbs, University of California, San Francisco (Dobbs et al. 1988; Gonzalez and Dobbs 1998). A polyclonal antiserum against surfactant protein C was purchased from Chemicon International (Temecula). A rabbit antibody against phosphorylated focal adhesion kinase was obtained from Biosource (Camarillo, CA). The mouse MAb 8D5 against β-dystroglycan was purchased from Novocastra Labs (Newcastle, UK). MAb against actin (MAB 3128, clone 10A5) was purchased from Chemicon International. Control IgGs and IgMs and fluorescein, rhodamine, and horseradish peroxidase-conjugated secondary antibodies were purchased from Jackson ImmunoResearch Laboratories (West Grove, PA). Alexa 488-conjugated phalloidin was purchased from Molecular Probes (Eugene, OR).
SDS-PAGE and Immunoblotting
Cultured alveolar epithelial cells were solubilized in sample buffer consisting of 8 M urea, 1% SDS in 10 mM Tris-HCl, pH 6.8, 15% β-mercaptoethanol. Proteins were separated by SDS-PAGE, transferred to nitrocellulose, and processed for immunoblotting as previously described (Harlow and Lane 1988).
Immunofluorescence Microscopy
Cells on YIGSR peptide-coated glass coverslips were extracted for 2 min in −20C acetone and air dried. Alternatively, cells were fixed in 3.7% formaldehyde for 5 min, extracted in 0.5% Triton X-100/PBS at 4C for 8 min in situ, and then washed thoroughly in PBS. Appropriate single or mixes of primary antibodies and conjugated chemicals were overlaid onto the fixed cells, and the preparations were incubated at 37C for 60 min. Cells on coverslips were washed in three changes of PBS and then overlaid with secondary antibodies, placed at 37C for an additional 60 min, washed extensively, and then mounted on slides. Controls included use of non-relevant immunoglobulins in place of primary antibodies followed by incubation in conjugated secondary antibodies to assess nonspecific antibody binding. All preparations were viewed on a Zeiss laser scan confocal microscope (LSM 510; Carl Zeiss, Thornwood, NY). Microscope images were exported as TIFF files, and figures were generated using Adobe Photoshop software (San Jose, CA).
Results
Phenotype of Primary Alveolar Cells that Assemble Laminin-311 Fibers
Isolation of type II alveolar cells from the lungs of both rats and mice is a well-documented and standard procedure (Dobbs 1990). Following plating, primary type II cells initially appear rounded but within 2 days flatten onto their substrate (Figure 1-Figure 3). Coincident with flattening, the cells show an extensive actin cytoskeleton network, assemble numerous focal contacts (adhesions) where the cells abut the substratum, and elaborate a complex fibrillar matrix rich in laminin-311 composed of the α3, β1, and γ1 subunits (Aumailley et al. 2005; Jones et al. 2005) (Figure 1A). Only staining of the α3 laminin subunit is shown. Toward the edges of cells, focal contact proteins detected with an antibody against focal adhesion kinase show some co-distribution with laminin fibers, indicating that cells likely adhere intermittently along the length of the fibrous matrix (Figure 1C).
It is also well known that, when maintained in vitro, type II cells undergo differentiation to a type I-like cell (see, for example, Chen et al. 2004; Olsen et al. 2005). Thus, we assessed whether the loss of type II phenotype and gain of expression of type I alveolar cell markers correlated with assembly of a laminin-311-rich matrix. To do so, we double stained our cultured, alveolar cells using an antibody against surfactant protein C (SP-C), a marker of type II cells, or a MAb against T1α, a 40-kDa apical membrane, type I cell marker, in combination with antibodies against subunits of laminin-311 (Figure 2 and Figure 3) (Dobbs et al. 1988; Gonzalez and Dobbs 1998). Only α3 laminin subunit localization is shown. At 2 days after isolation and plating, ∼30-40% of the primary epithelial cells express SP-C protein, which localizes in a punctate fashion throughout the cytoplasm (Figure 2A). These cells show minimal staining with our laminin-311 probes, although some cytoplasmic localization is observed (Figure 2B). After 4 days of isolation, few if any of the cells are recognized by the SP-C probe, although these same cells show extensive arrays of laminin-311 fibers (Figures 2C and 2D). In contrast, the T1α antibody shows minimal staining of the cultured primary alveolar cells at 2 days after plating when there is also little staining by laminin-311 probes (Figures 3A and 3B). However, the antibody does generate some background nuclear staining. After 4 days of isolation, at least 80% of the alveolar cells exhibit robust cytoplasmic T1α antibody staining (Figure 3C). Moreover, the T1α positively stained cells assemble laminin-311 fibers along the surface where they adhere to their substrate (Figure 3D). Consistent with these immunofluorescence data, Western immunoblotting indicates that SP-C expression decreases dramatically in the cultured primary cells 2 days after plating, whereas T1α levels show the inverse (Figure 4).
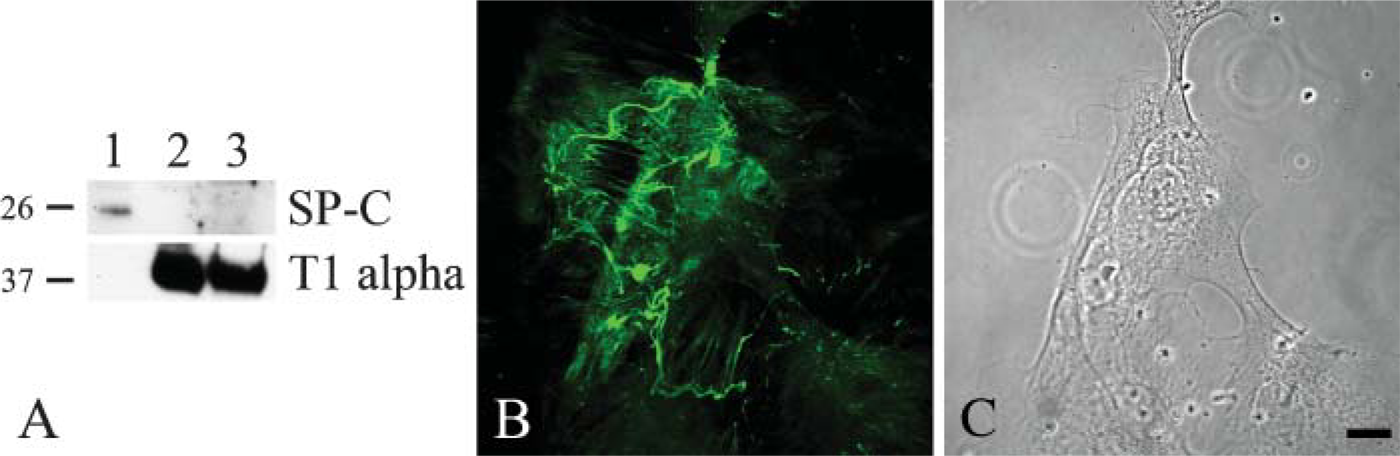
The above data indicate that alveolar cells displaying type I characteristics are those that elaborate a laminin-311 matrix. To provide additional support for this notion, type I alveolar epithelial cells were isolated and selected from rat lungs by antibody capture, following procedures detailed by others (Chen et al. 2004). These cells were positive for the type I markers T1α (and aquaporin 5 and the receptor for advanced glycation end products; not shown) but negative at all times for surfactant protein at days 0, 1, and 2 after plating (Borok et al. 1998a,b; Fehrenbach et al. 1998; Dahlin et al. 2004; Shirasawa et al. 2004) (Figure 5A; compare with blots in Figure 4). Moreover, at 4 days after plating the cells assemble a fibrillar laminin-311-rich matrix as determined by confocal immunofluorescence microscopy using antibodies against all three subunits of laminin-311 (Figures 5B and 5C). Only the results using the α3 laminin subunit probe are shown.
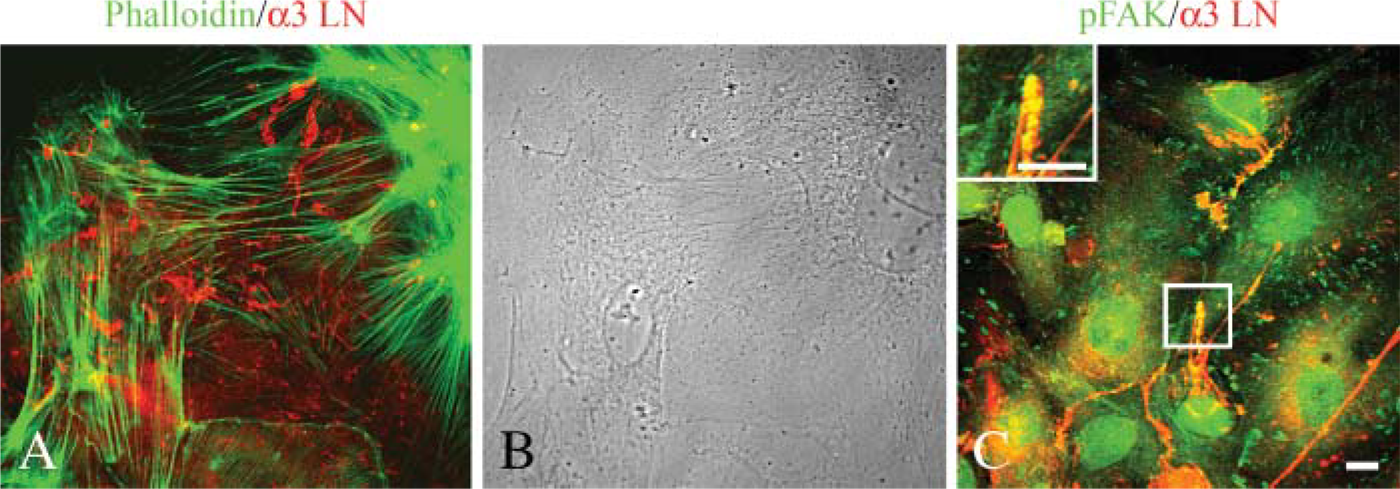
Localization of laminin-311 fibers, actin microfilaments, and focal contacts in cultures of primary type II alveolar epithelial cells. Primary type II alveolar cells were prepared for double labeling using antibodies against α3 laminin subunit (red,
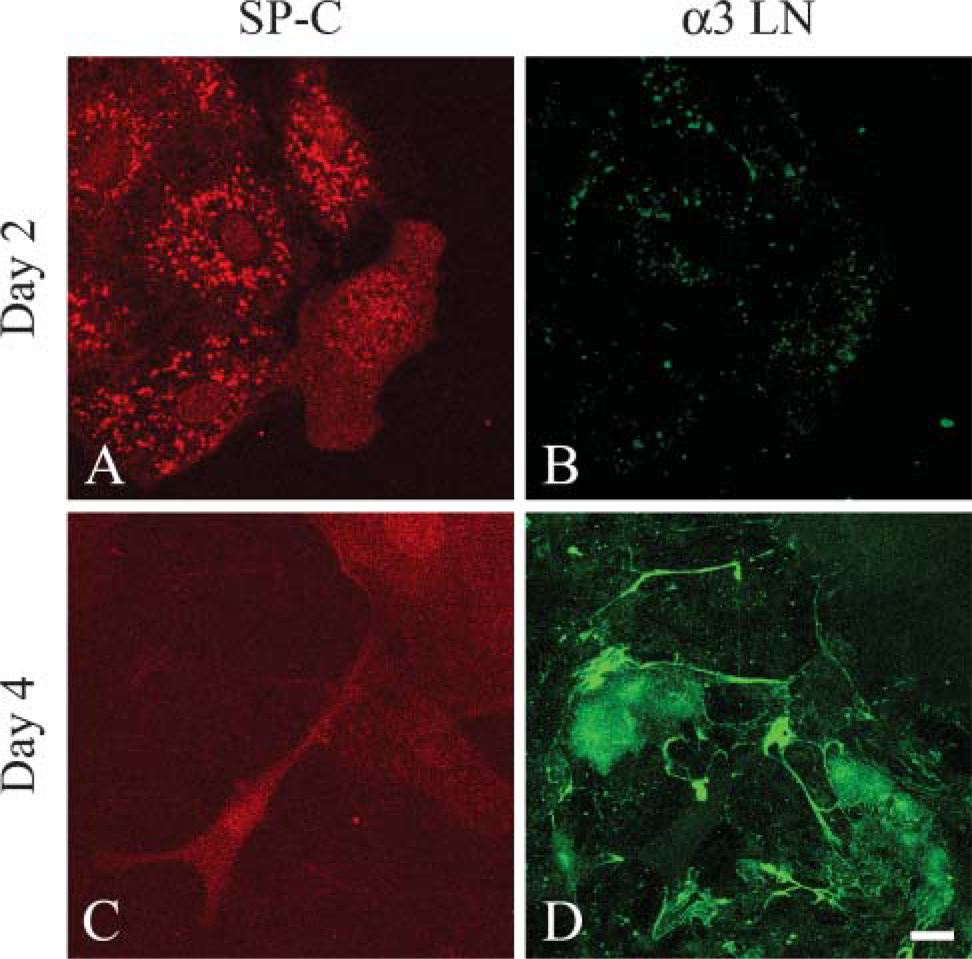
Type II alveolar cells lose expression of SP-C and at the same time assemble laminin-311 fibers when maintained in vitro. Primary type II alveolar cells were prepared for double-label confocal immunofluorescence microscopy using antibodies against SP-C (
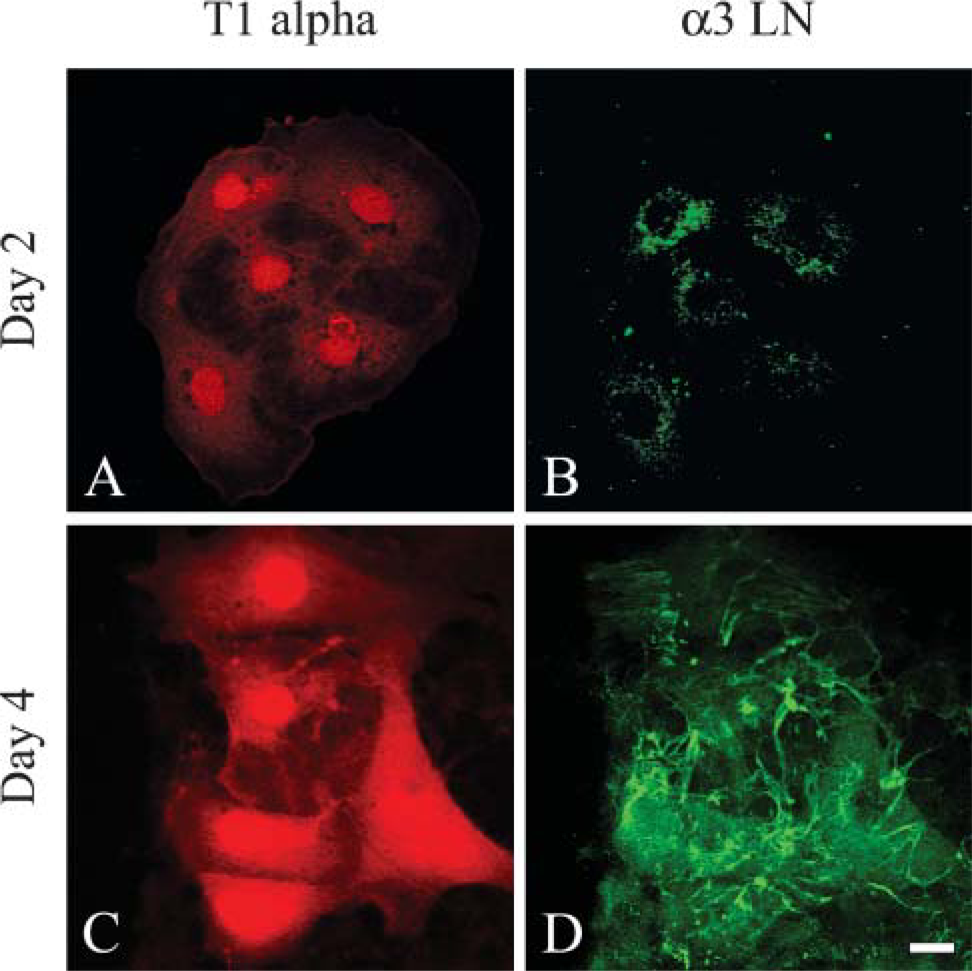
Alveolar cells expressing T1α assemble an extensive fibrillar matrix rich in laminin-311 in vitro. Primary type II alveolar cells were prepared for double labeling using antibodies against T1α (
Assembly of Laminin-311-rich Fibers Is Regulated by β1 Integrin, the Small GTPases, and Myosin
Organization of laminin into fibers in the matrix of the type I-like alveolar cells is unusual because most laminins assemble into sheets rather than fibers (Colognato et al. 1999). Indeed, laminin-311-rich fibers bear superficial resemblance to fibronectin fibrils found in the matrix of many different types of cells. These fibrils have been shown to assemble in a cytoskeleton- and matrix receptor-dependent fashion (Wierzbicka-Patynowski and Schwarzbauer 2003). We therefore tested the possibility that laminin-311 fiber assembly follows the same mechanism. There are two good candidate receptors that could play a role. The first are integrins, components of focal contacts, which show association with laminin-311 (Figure 1B). The second candidate is dystroglycan, which we previously showed to be in a functional complex with laminin-311 (Jones et al. 2005).
We used small-hairpin (sh) RNA technology to knockdown dystroglycan protein expression in alveolar cells or incubated alveolar cells with an antibody antagonist against β1 integrin and assayed the ability of the treated cells to assemble laminin-311 fibers (Figure 6). In each instance, cells were either infected with virus encoding control or dystroglycan shRNA, or antibody was added to the medium of cells at 2 days after plating and cells were processed for immunofluorescence microscopy using antibodies against laminin-311 subunits 2 days later. We confirmed the efficiency of our shRNA treatment by performing Western blotting analyses indicating that dystroglycan shRNA, but not control shRNA, induces a >90% knockdown in dystroglycan protein expression (Figure 6) (Jones et al. 2005). Cells infected with control shRNA or incubated in control isotype-matched immunoglobulin assemble normal arrays of laminin-311 fibers (Figure 6B and not shown). In addition, cells lacking expression of dystroglycan also assemble laminin-311 fibers (Figure 6A). In sharp contrast, cells incubated in the function-blocking β1 antibody fail to do so (Figure 6D).
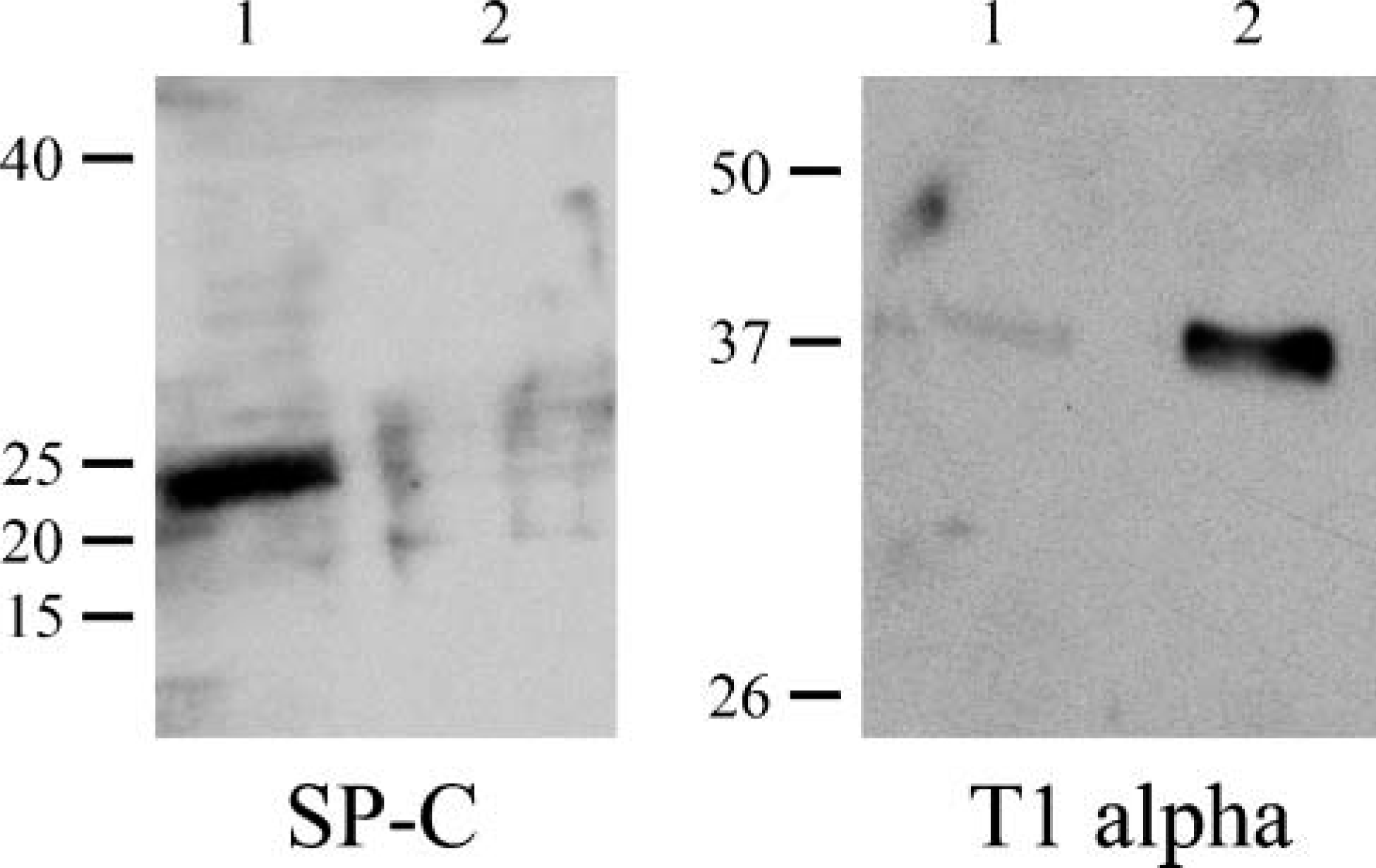
Western immunoblotting analyses reveal changes in expression level of SP-C and T1α by primary alveolar type II cells as a function of time in culture. Extracts of primary type II alveolar cells at 2 (Lane 1) and 4 days (Lane 2) after placement of the cells in culture were processed for SDS-PAGE/immunoblotting using antibodies against either SP-C or T1α as indicated. The same amount of protein was loaded into each lane. Molecular weight standards are indicated at the left of each set of panels.
Type I alveolar cells assemble an extensive fibrillar matrix rich in laminin-311 in vitro. (
Previous work indicates that β1 integrin signaling via the small GTPases regulates assembly of certain laminin matrices (deHart and Jones 2004). To assess whether laminin-311 fibers are also assembled via a mechanism involving the small GTPases, alveolar cells at 2 days after plating were infected with control adenovirus or adenovirus encoding dominant-negative c-myc-tagged Rac1, RhoA, and Cdc42. Two days later the adenovirus-infected cell populations were processed for immunofluorescence microscopy using antibody probes against laminin-311 subunits in combination with the c-myc antibody 9E10 (Figures 7A and 7B). There is an inhibition in fiber assembly of the cells expressing the dominant negative constructs (Figure 7B). Only an image of cells expressing dominant-negative RhoA is shown. Remarkably, in all instances, laminin-311 subunits appear as large aggregates randomly arranged in the extracellular matrix of infected cells as determined by confocal microscopy (Figure 7B). Moreover, we also evaluated whether fiber assembly was dependent on the activity of MLCK. As in the case of the alveolar cells expressing the dominant negative small GTPases, laminin-311 subunits fail to assemble into fibers but aggregate in a non-fibrillar fashion along the substrate-attached surface of the cells treated with ML-7, an inhibitor of MLCK (Figures 7C and 7D). Drug treatment, but not vehicle alone, also induces a shape change in the cells, which appear more elongated and less flat than their non-drug-treated counterparts (compare cells in Figure 7D with those in Figure 1).
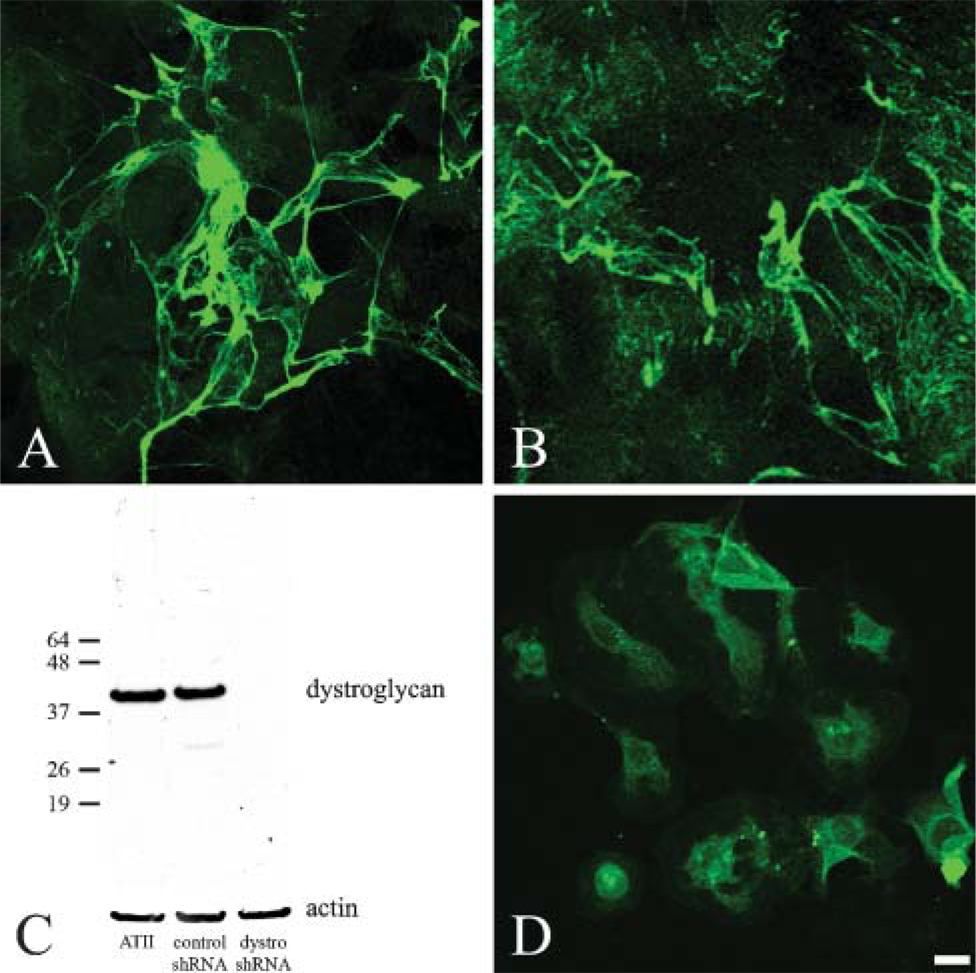
Type II alveolar type cells treated with a β1 integrin antagonist fail to assemble laminin-311 fibers in vitro. Type II alveolar cells were infected with adenovirus encoding either dystroglycan small-hairpin (sh) RNA (
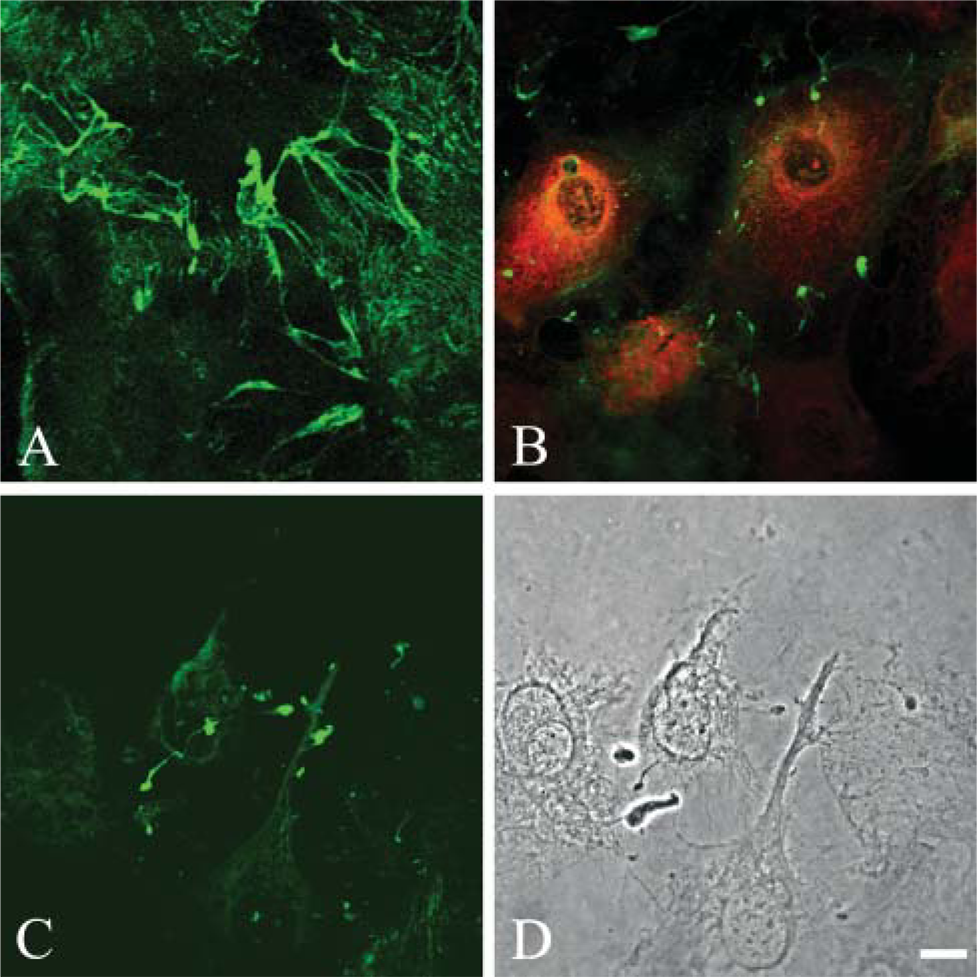
Primary alveolar type cells infected with adenovirus encoding dominant-negative RhoA or treated with the myosin light chain kinase inhibitor ML-7 fail to assemble laminin-311 fibers in vitro. Alveolar type II cells, 2 days after plating, were infected with control adenovirus (
Discussion
Defining the precise molecular composition and structure of the alveolar BM is essential for determining how its components contribute to the homeostasis of alveolar tissue architecture and how BMs are involved in the etiology of various lung diseases. The latter include Goodpasture's syndrome where the integrity of the alveolar BM is perturbed, asthma, and chronic obstructive lung disease where the airway BM may become abnormally thickened (Jeffery 2001; Hudson et al. 2003). In intact lung tissue, epithelial cells that line the internal surface of the alveoli and endothelial cells that line the extensive capillaries within the lung sit on either side of a common BM, and presumably both contribute to its composition and assembly. This clearly complicates any analyses of alveolar BM structure in intact tissue. Thus, we have focused our studies on cultured primary alveolar cells. Although our previous work and that of others indicate that alveolar cells secrete a laminin-311 matrix, in this work we attempted to define the phenotype of the alveolar epithelial cell that deposits laminin-311 and to determine how alveolar cells organize laminin-311 into a fibrillar matrix (Miner et al. 1997; Miosge et al. 2002; Jones et al. 2005).
We used a standard procedure to isolate alveolar type II cells from the lungs of rats (Dobbs 1990). These cells exhibit type II cell phenotype immediately upon isolation but, over time, downregulate expression of markers of type II cells including SP-C. During the same period they begin to express type I cell markers such as T1α protein (Dobbs et al. 1988). These results are consistent with reports of others (Borok et al. 1998a; Gonzalez and Dobbs 1998; Isakson et al. 2001; Olsen et al. 2005). Concomitant with adoption of a type I phenotype in vitro, alveolar epithelial cells assemble an unusual fibrous laminin network rich in laminin-311 (Jones et al. 2005). Thus, we propose that in the intact lung the cell type responsible for depositing and assembling laminin-311 is the type I cell. In support of this notion, we also show that freshly isolated rat alveolar type I cells also assemble a laminin-311 fibrillar matrix. Defining precise differences in laminin composition in the BM underlying type I and type II cells in intact lung is highly problematic because of the complex architecture of the tissue. Thus, we cannot rule out the possibility that type II cells in vivo also assemble laminin-311 fibers. However, based on our cell culture studies and our finding that a variety of immortalized type II cells (including A549 cells) fail to secrete laminin-311 (unpublished data), we believe this to be unlikely. Rather, we propose that the BM of the alveolar sacs shows distinct differences in its laminin composition with BM underlying type I cells enriched in laminin-311 fibrils. Indeed, this hypothesis is consistent with previous work showing that the composition and structure of the BM under type I and type II cell types is not the same (Brody et al. 1982). Specifically, others have reported that the BM underlying type II cells is often discontinuous, whereas the BM beneath type I cells is not (Brody et al. 1982). Moreover, BMs under type I vs type II cells exhibit differences in their sulfate ester composition (Brody et al. 1982).
The importance of the laminin-311 fibers assembled by type I-like cells is emphasized by our previous study where we identified laminin-311 as part of the ‘chain’ of matrix molecules that transmits mechanical forces initiated by stretching the substrate to which the cells adhere (Jones et al. 2005). Dystroglycan mediates the transmission of mechanosignals at the surface of alveolar epithelial cells; integrin β1 is not involved (Jones et al. 2005). In sharp contrast, a β1 integrin antagonist inhibits the assembly of laminin-311 fibers in alveolar epithelial cells whereas knockdown of dystroglycan does not. Moreover, inhibition of the small GTPases also inhibits assembly of laminin-311 fibers in the matrix of our alveolar epithelial cells. Indeed, the assembly of laminin-311 fibers in the matrix of alveolar epithelial cells involves the same molecules that regulate assembly of laminin-5 matrix in keratinocytes (deHart et al. 2003; deHart and Jones 2004). In the latter instance, laminin-5 matrix assembly is perturbed by antagonists of various integrins and the small GTPases (deHart and Jones 2004). It is also myosin dependent (deHart et al. 2003). Indeed, our studies support a model by which β1 integrin ‘unfolds’ laminin-311 from aggregates of protein into a fibrous organization in the matrix of alveolar cells. We hypothesize that integrins do so by being shuttled over the basal surface of alveolar cells in an actin-dependent manner. There is considerable support for this model because integrins are known to be dynamic in the plane of the membrane (Smilenov et al. 1999; Zamir et al. 2000; Tsuruta et al. 2002). Moreover, we have also shown that the dynamics of integrins are regulated by myosin motors (Tsuruta et al. 2002).
In summary, cultured primary alveolar cells exhibiting a type I cell phenotype assemble a fibrillar laminin matrix in an integrin, small GTPase, and cytoskeleton-dependent manner. Because type I cells line ∼95% of the internal surface of the alveoli of the lungs, laminin-311 fibers are likely to play a major role in maintaining alveolar tissue architecture, in addition to their function in relaying mechanical signals in the form of stretch (Jones et al. 2005).
Footnotes
Acknowledgements
This work was supported by National Institutes of Health (NIH) Grant PO1 HL-071643.
We are most grateful to Drs. Leland Dobbs, Peter Yurchenco, and Anne Ridley for generous gifts of antibodies and reagents. We also thank Dr. Sznajder for helpful discussion.
