Abstract
Recently there have been reports that identify two transient receptor potential channels in cell–matrix junctions known as focal adhesions. These are the calcium channel TRP canonical 7 and the calcium-activated monovalent ion channel, TRP melastatin (TRPM) 4. Here, we report on the occurrence of TRPM4 in focal adhesions of fibroblasts. Of three commercial antibodies recognizing this channel, only one yielded focal adhesion staining, while the other two did not. The epitope recognized by the focal adhesion-localizing antibody was mapped to the extreme C-terminus of the TRPM4 protein. The other two antibodies bind to N-terminal regions of the TRPM4 proteins. Deletion of the TRPM4 gene by CRISPR/cas9 techniques confirmed that this channel is a bona fide focal adhesion component, while expression of full-length TRPM4 proteins suggested that processing may occur to yield a form that localizes to focal adhesions. Given the reports that this channel may influence migratory behavior of cells and is linked to cardiovascular disease, TRPM4 functions in adhesion should be explored in greater depth. (J Histochem Cytochem 71: 495–508, 2023)
Introduction
Ion channels are ubiquitous regulators of the intracellular environment, key to many processes such as cytoskeletal assembly and stability, differentiation, signal transmission, survival, and apoptosis. The transient receptor potential (TRP) channels take many forms and comprise a number of subclasses. In mammals, these are TRP canonical (TRPC), TRP vanilloid, TRP melastatin (TRPM), and TRP ankyrin with the more distantly related TRP mucolipin and TRP polycystins.1–3 All the TRP channels comprise six transmembrane domains with intracellular N- and C-termini and a pore region that is located between the fifth and sixth transmembrane domains. In addition, TRPC but not TRPM channels have three or more ankyrin repeats at the N-terminus which have roles in protein–protein interactions. 3 Two TRPMs also differ from others in containing a C-terminal kinase domain (TRPM6 and TRPM7). 4 This family of ion channels has a long evolutionary history and are present in yeast through to mammals. TRPs are gated by physical and chemical stimuli, including stretch, changes in temperature, touch, light, pH, and endogenous or exogenous ligands. 5
In humans, there are 28 TRP channels, many of which have not been extensively studied. However, in the past few years, two channels, TRPC7 and TRPM4, have been localized to focal adhesions.6,7 These are organelles characteristic of cell–extracellular matrix interaction and have functions in signaling to the cytoskeleton, but also in cell survival.8,9 They are formed by many cell types in vitro, e.g., fibroblasts, endothelial and epithelial cells, and smooth muscle cells, but infrequently by leukocytes. In vivo equivalent structures have been identified, 10 notably in response to shear stress (endothelial cells) 11 or wound healing (myofibroblasts), 12 but more stable analogous structures may be muscle costameres, myotendinous junctions, and dense bodies of smooth muscle.
Several proteomic studies in the recent past have identified a substantial focal adhesion proteome,13–15 but a careful analysis suggests a core proteome of around 60 proteins, mostly actin-associated proteins such as talin, vinculin and zyxin, integrins, and focal adhesion kinase (FAK). 15 However, no ion channels have been identified by proteomics although there may be technical reasons why these multiple membrane-spanning proteins have eluded detection. Certainly, their presence might be expected where mechanosensitivity and dynamics are important, e.g., turnover associated with cell migration or invasion.
TRPM4 is a calcium-activated monovalent ion channel, 16 and its presence alongside the calcium channel TRPC7 is suggestive of a correlated mode of action. Here, we explore the presence of TRPM4 in focal adhesions but have found that it is likely that only a modified form of the channel is present in adhesions. However, it is widespread, and our data suggest that it is not required for focal adhesion assembly, since after successful gene deletion, adhesions still form and cell spreading and cytoskeletal architecture are apparently unperturbed.
Materials and Methods
Antibodies and Reagents
Primary antibodies for immunocytochemistry (ICC) and western blotting (WB) were as follows. Anti-β-tubulin (mouse, TUB2.1, Sigma-Aldrich, Merck Life Science A/S, Søborg, Denmark, T4026, WB: 1:750–1:1000), anti-green fluorescent protein (GFP; chicken, IgY Fraction, Aves Labs, Inc., Davis, CA, GFP-1020, WB: 1:1000), anti-vinculin (mouse, hVIN-1, Sigma-Aldrich, V9131, WB: 1:1000, IF: 1:200), and anti-TRPC7 (rabbit, Sigma-Aldrich, HPA031126, ICC: 1:100). Rabbit antibodies against TRPM1 (Acc-042), TRPM2 (Acc-043), TRPM3 (Acc-050), TRPM4 (Acc-044), TRPM5 (Acc-045), TRPM6 (Acc-046), TRPM7 (Acc-047), andTRPM8 (Acc-049) were all from Alomone Labs, Jerusalem, Israel and used at 1:100 (ICC) or 1:200 (WB). Other antibodies against TRPM4 were from OriGene, Rockville, MD, (mouse 10H5/TA500381, WB: 1:700, ICC: 1:100) and Santa Cruz Biotechnology, Inc., Heidelberg, Germany, (goat, sc-27540; WB: 1:200, ICC: 1:100), Secondary Alexa Fluor 488-nm, 568-nm, or 647-nm-conjugated antibodies—goat antirabbit IgG, goat antimouse IgG, goat antirat IgG, donkey antigoat IgG, and donkey-antirabbit IgG—were from Molecular Probes (Life Technologies, Paisley, UK, ICC: 1:200). Alexafluor 645-nm-conjugated phalloidin was from Molecular Probes (Life Technologies, 1:200). Secondary horseradish peroxidase (HRP)-conjugated antibodies were antichicken IgY H&L (goat, Abcam, Cambridge, UK, ab6877, WB: 1:3000), antigoat IgG (rabbit, Dako Denmark A/S, Glostrup, Denmark, P0449, WB: 1:3000), and antimouse IgG (goat, Dako, P0477, WB: 1:3000). Lysate from HEK293T cells transiently transfected with Myc/FLAG-tagged human TRPM4 (accession numbers NM_107636, NM_060106) was from Origene (LC413636).
Cell Culture
Mouse embryo fibroblasts (MEFs) and rat embryo fibroblasts cells were cultured in α modification of Eagle’s Medium (Invitrogen-Life Technologies) supplemented with 2-mM glutamine (Invitrogen) and 10% and 5%, respectively, dialyzed, sterile-filtered FCS. Human embryo kidney and Michigan Cancer Foundation-7 (MCF-7) cells were cultured in Dulbecco’s modified Eagle’s Media (Life Technologies) containing 10% FCS. All cell lines were routinely screened for mycoplasma.
Immunofluorescence Microscopy
Immunostaining was performed on cells grown on glass coverslips overnight. The cells were washed twice with phosphate-buffered saline (PBS) and fixed with PBS containing 4% paraformaldehyde for 15 min, permeabilized with PBS containing 0.1% or 0.5% TritonX-100 for 10 min, and quenched with NH4Cl for 20 min. Cells transfected with cDNA encoding enhanced greeen fluorescent protein only (EGFP) were not quenched. Coverslips were washed with PBS between each step and three times for 10 min with PBS after quenching. Blocking was achieved with 5% bovine serum albumin (BSA) in PBS for 20 min. After blocking, the coverslips were incubated with primary antibodies diluted in PBS containing 1% BSA for 1 hr at room temperature, washed extensively with PBS, then incubated with fluorescence-labeled secondary antibody diluted in PBS with 1% BSA for 1 hr at room temperature in the dark. Coverslips were washed with PBS and mounted in ProLong Gold antifade reagent (Invitrogen). Slides were analyzed on a Zeiss Axioplan-2 (Zeiss, Oberkochen, Germany) microscope using Apochromat 63X/1.4 oil objective or a Zeiss LSM5 confocal microscope. Images were processed using Adobe Photoshop. For focal adhesion counting after double staining for TRPC7 and TRPM4 (with phalloidin to detect microfilament bundles), confluent MEF monolayers on coverslips were scratch wounded and cultured for 2 or 4 hr before fixation and processing. Table 1 contains data from a total of 361 adhesions.
Presence of TRPC7 and/or TRPM4 in Focal Adhesions of Motile Murine Embryo Fibroblasts.

Abbreviations: TRPC, TRP canonical; TRPM, TRP melastatin.
Micrographs of cultures at ≤4 hr after scratch wounding of confluent monolayers were analyzed, and the data are expressed as percentages.
Plasmid Isolation and Transfection
TRPM4-EGFP cDNAs 17 were isolated using a High Pure Plasmid Isolation Kit and protocol (Roche, Basel, Switzerland, 11 754 785 001) from DH5α bacteria grown in lysogeny broth (LB-medium) with 50 µg/ml kanamycin at 37C overnight. A NanoDrop 2000 spectrophotometer (Thermo Fisher, Braunschweig, Germany) was used to define the concentration and purity of extracted plasmid DNA.
Reverse transfection with plasmid DNA was performed using Lipofectamine LTX (Thermo Fisher). Two microliters of plus reagent was mixed with 50 μl of Optimem medium (Thermo Fisher) or serum-free medium, and 500 ng of plasmid DNA was diluted in the mixture. Meanwhile, 1 μl of Lipofectamine LTX reagent was mixed with 50 μl of Optimem medium or serum-free medium. Both mixtures were incubated for 10 min at room temperature. The two solutions were mixed and incubated for 30 min at room temperature in 24-well plates to which 24 × 103 cells were then added per well. After 4 hr of incubation, the medium was replaced with a medium containing serum. Alternately, cells were seeded in 24-well plates in a normal culture medium overnight, and the cDNA/transfection agent mixture was added to each well after changing to serum-free medium. Cultures were incubated for 48 hr under normal culture conditions before processing for ICC or WB.
CRISPR/Cas9 Deletion of TRPM4 in Cultured Fibroblasts
Two different strains of MEFs were prepared where the TRPM4 gene was deleted by CRISPR/Cas9 methodology. The sgRNA sequences were GGGCCAGTGTGGGCAACCTC (TRPM4-1) and GGTAACTACGGCTGCCCCAA (TRPM4-2). The cutting sites targeted were chr7:45327864-45327886 (TRPM4-1) and chr7:45327811-45327833. In both cases, it was hypothesized that all known isoforms of TRPM4 would be deleted. The vector system was used as described. 18 Derived clones for each knockout were sequenced using the following primers: TRPM4-1 forward CACCGGGGCCAGTGTGGGCAACCTC and reverse AAACGAGGTTGCCCACACTGGCCCC and TRPM4-2 forward CACCGGGTAACTACGGCTCCCCAA and reverse AAACTTGGGGCAGCCGTAGTTACCC.
Western Blotting
Lysates from subconfluent monolayer cultures were washed 3× with PBS and then solubilized in Laemmli buffer at 37C (50 mM Tris HCl, pH 6.8, containing 2% sodium dodecylsulfate, 10% glycerol, 5% β-mercaptoethanol, and 0.05% bromophenol blue). Samples were sheared by passage through 25-g needles 10 times, then resolved by 7.5% or 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE), and transferred to polyvinylidene difluoride (PVDF) membranes as previously described. 19 Membranes were blocked in 5% skimmed milk in tris-buffered saline (TBS) and incubated with antibodies as described previously. 19 Secondary antibodies were HRP-conjugated, and the reaction product from chemiluminescence (Biological Industries, Haemek, Israel) was captured on Hyperfilm ECL (GE Healthcare, Amersham, UK Code 28906837).
Peptide Synthesis
The human TRPM4b sequence (Q8TD43) without the six putative transmembrane domains was synthesized as an overlapping series of peptides, with two or four amino acids offset, on cellulose membranes using a Multipep automated peptide synthesizer (INTAVIS Bioanalytical Instruments AG, Koeln, Germany) as previously described. 20 The TRPM4b blocking peptide (LSRSALLPPGGPPPPDLPGSKD; peptide 1) and two scrambled control peptides (PAPSGLPSPGPKSDDGLRLPLP and PSGPDSGSPLPKDLPGALPRPL; peptides 2 and 3m respectively) were synthesized to a purity of 93–95% by Genscript Corp (Piscataway, New Jersey).
Epitope Mapping and TRPM4b Blocking Experiment
The human TRPM4b peptide arrays were overlaid with anti-TRPM4b (Origene 10H5, #TA500381) (1:20,000) in 1% casein overnight at 4C and washed three times for 5 min in TBS with 0.1% Tween-20 (TBST) before incubation with antimouse IgG conjugated with HRP (#NA931; GE Healthcare) for 1 hr at room temperature. After further three washes in TBST-T, the signal was developed with ECL Prime (RPN 2232; GE Healthcare). For the TRPM4b blocking experiment, the TRPM4b antibody (1:20,000) was preincubated with or without the TRPM4b blocking peptide (5 µM) or the scrambled control peptides (5 µM) overnight at 4C, before immunoblotting for 2 hr at room temperature. Experiments were repeated three times.
In other experiments, MCF-7 and MEF lysates were resolved by 7.5% SDS-PAGE and the peptides preincubated with the primary antibody (10 µM, 20–30 min) before incubation with the membranes. Similarly, indirect immunofluorescence microscopy of MEF cells included a peptide adsorption step (10 µM) with the 10H5 antibody before the mixture was applied to coverslips.
Results
Primary murine fibroblasts were stained for transient receptor potential melastatin 4 (TRPM4) with a panel of three commercially available antibodies (Figs. 1 and 2). Of these, only one (10H5/TA500381) clearly delineated focal adhesions, as demonstrated by their characteristic appearance but also double staining for the focal adhesion component, zyxin (Fig. 2A). The other two antibodies (Acc-044 and sc-27540) did not stain focal adhesions. Instead, these antibodies yielded a punctate or reticular staining pattern (Fig. 2B). It was noted that the staining patterns of these two antibodies were dissimilar to each other. The 10H5 antibody was then used in double staining of rat and MEFs, for TRPM4 and TRPC7. While both channels were focal adhesion components (Fig. 3), there were subtle differences in their location. Where both channel proteins were present in the same adhesion, the TRPC7 was consistently located more distally than the TRPM4. In many cases, there was not a region of overlap. Moreover, in some adhesions, only one of the two channels was present, suggestive of different roles in these organelles. A quantitative analysis of motile fibroblast adhesions is shown in Table 1. The data suggest that only TRPC7 was located in central adhesions, while both channels were located at peripheral adhesions. Most adhesions contained one channel, <20% had both.
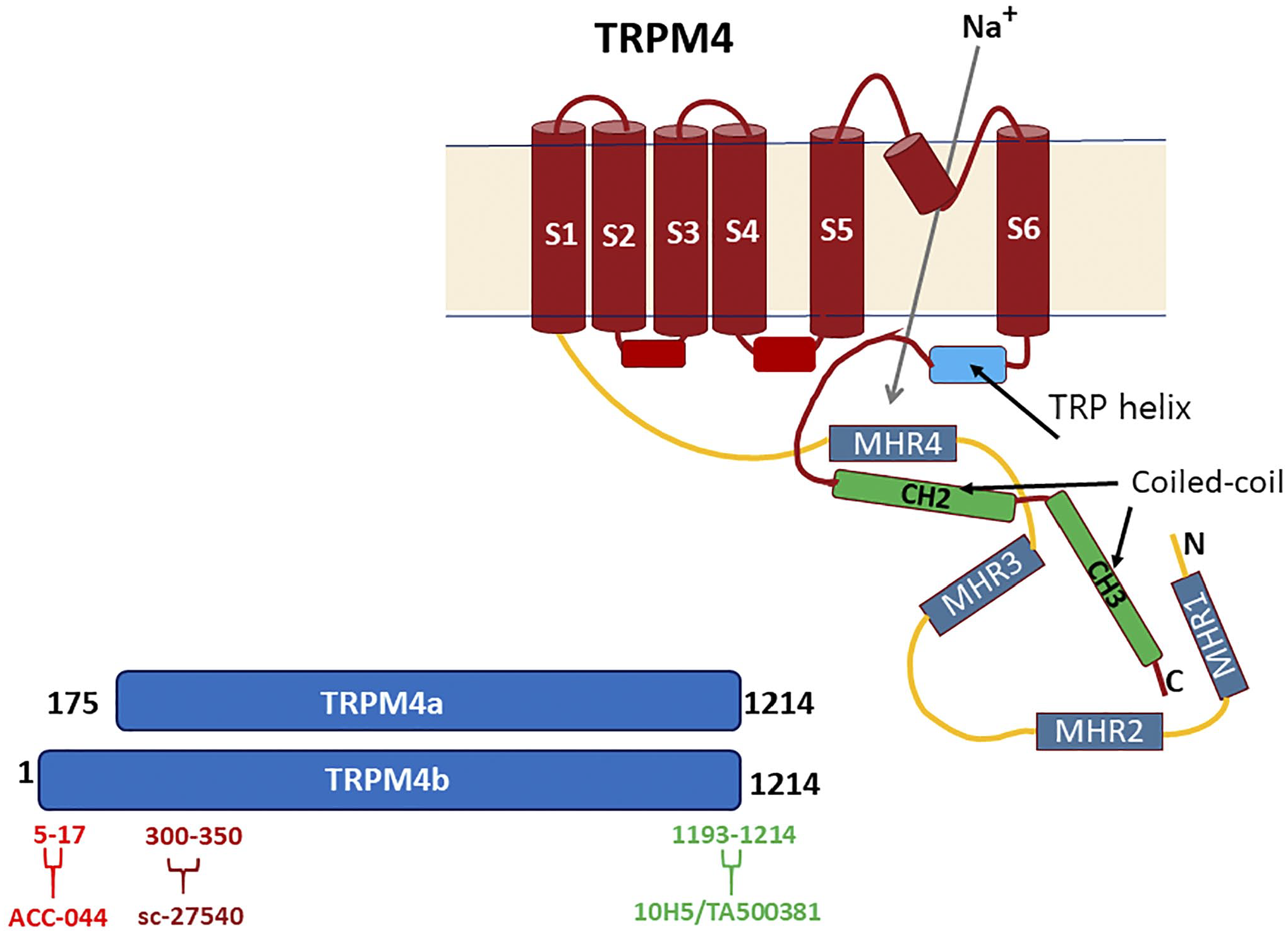
Diagrammatic representation of the TRPM4b structure, based approximately on the published structure. 21 S1–S6 represent the six transmembrane domains, while MHR1–4 are TRPM homology regions. Lower left, the sites of reactivity of the three antibodies used in this study are shown, based on human sequences. That for the 10H5 antibody was elucidated in this study. The Acc-044 antibody has specificity for TRPM4b but not for TRPM4a.
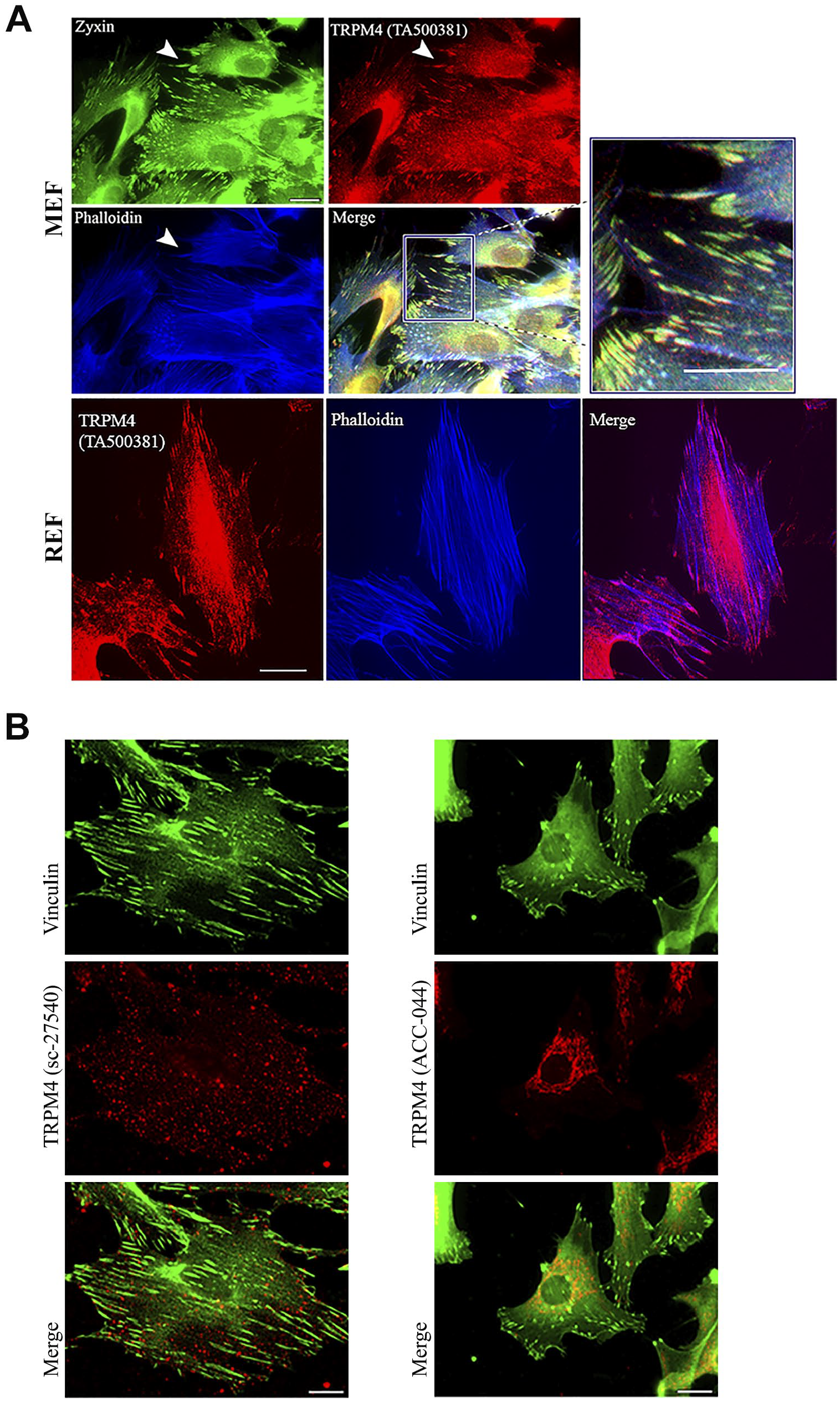
(A) TRPM4 detected by the 10H5 (TA500381) antibody is a focal adhesion component. Murine embryo fibroblasts (MEFs) were triple stained for TRPM4 (red), the focal adhesion marker zyxin (green), and F-actin (phalloidin, blue). A merged image with high power inset is shown. Arrowheads mark TRPM4-positive focal adhesions. Rat embryo fibroblast (REF) focal adhesions were also positive for TRPM4, the F-actin bundles detected by phalloidin (blue). A merged image is also shown. Scale bar = 10 µm. (B) The TRPM4b-specific antibody ACC-044 and TRPM4a- and TRPM4b-specific antibody sc-27540 do not localize to focal adhesions, demarcated by vinculin in murine fibroblasts. Scale bar = 10 µm.
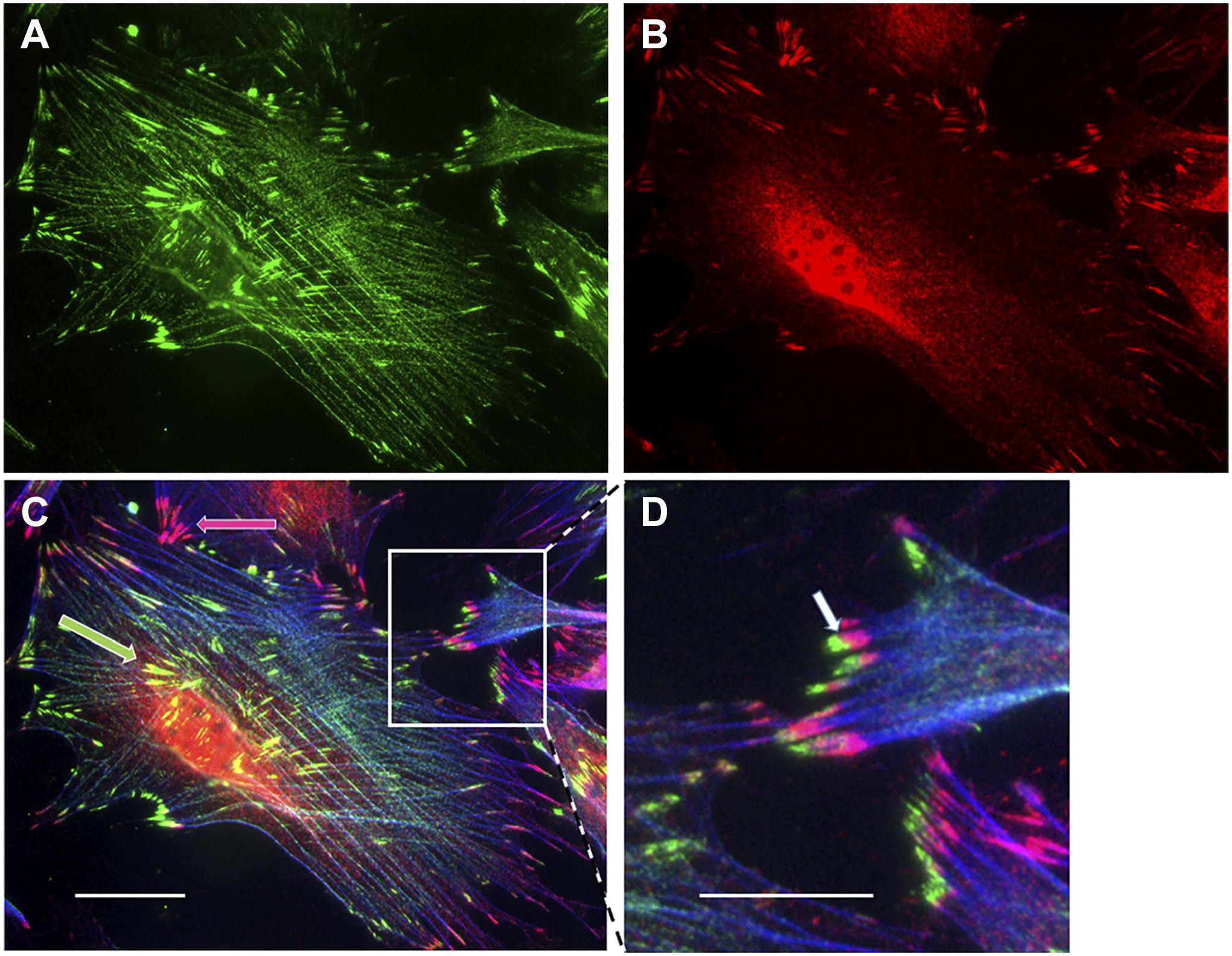
Murine fibroblasts double stained for TRPC7 (A; green) and TRPM4 (B; red). A triple exposure with phalloidin-stained microfilament bundles is shown in (C). Both channels are localized to focal adhesions, but partially in discrete populations (C; green and red arrows). Where both are present in the same adhesion, theTRPC7 is more distal (D; inset, white arrow). Scale bars = 10 µm and 5 µm in inset.
To investigate whether the 10H5 antibody might cross-react with other TRPM channels, a panel of antibodies against all the TRPM proteins was used to determine if any were focal adhesion components (Fig. 4). However, none resulted in focal adhesion staining, indicating that TRPM4 might be the only member of the family with this distinctive localization.
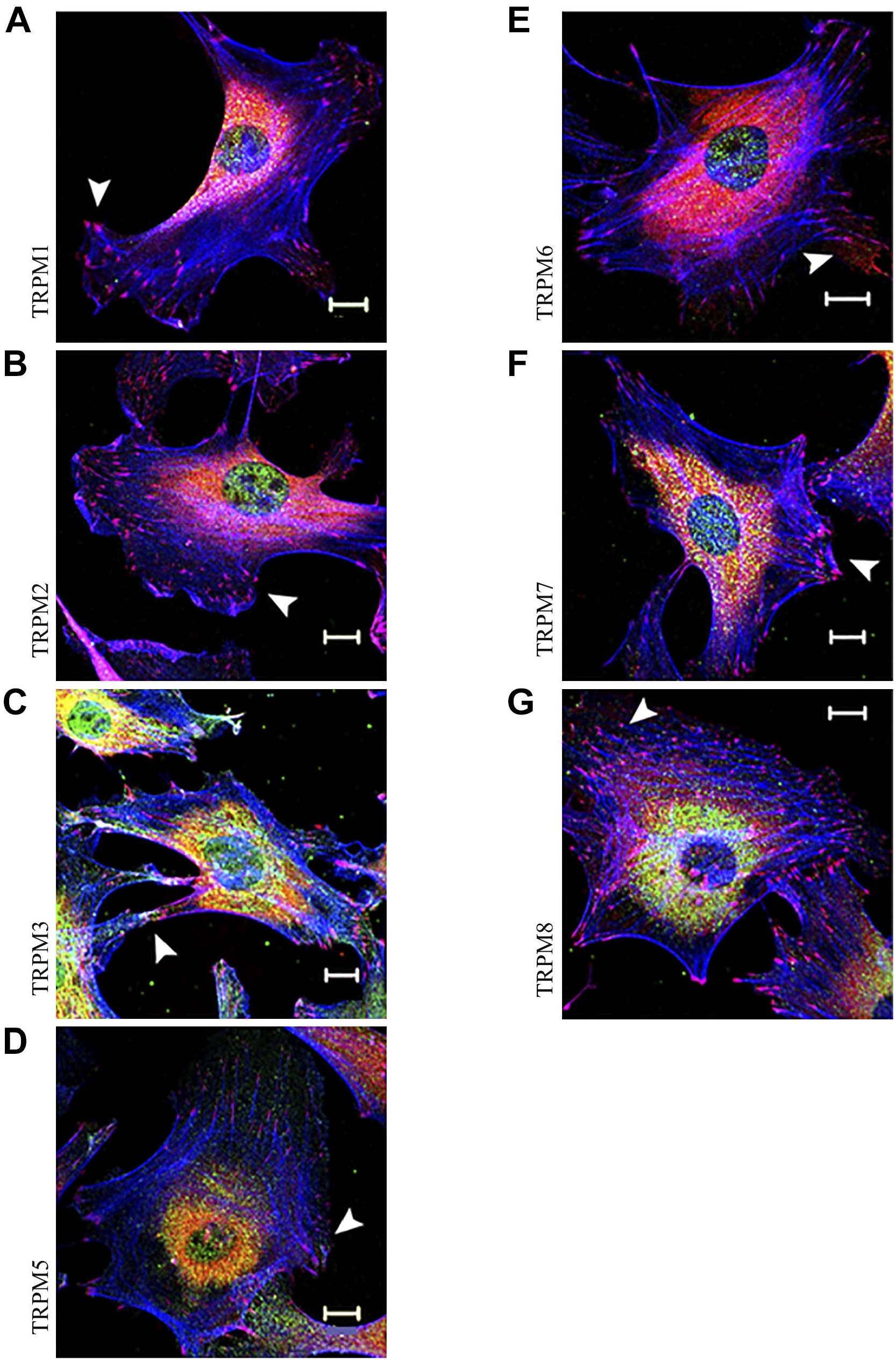
TRPM proteins, other than TRPM4, do not localize to focal adhesions. Rat embryo fibroblasts were triple stained for vinculin (red; arrowheads), F-actin (blue), and TRPM channel (green) as indicated. Scale bars = 10 µm.
In order to understand the relationships between antibodies and channel isoforms, it was necessary to epitope map the 10H5 antibody, which was not known. A series of overlapping peptides corresponding to the human TRPM4b protein, but excluding the putative transmembrane domains, were synthesized in duplicates on membranes and probed with the 10H5 antibody. Only one region, a peptide corresponding to the C-terminal 22 amino acids (residues 1193–1214), was recognized by the antibody, but reactivity was very robust (Fig. 5A). Further peptides were synthesized, corresponding to the reactive 22 amino acids (peptide 1) and two scrambled versions (peptides 2 and 3). Further dot blot experiments (Fig. 5B) showed that the synthetic peptide corresponding to the C-terminal sequence of human TRPM4b (peptide 1) could block reactivity to two overlapping peptides on the blots, while a scrambled peptide did not. In WB assays, the reactive peptide could block reactivity of 10H5 antibody toward the murine fibroblast TRPM4 in cell lysates while the scrambled peptides had no impact (Fig. 5C). A comparison of human and rat TRPM4b C-terminal sequences are shown in Fig. 5D, along with the dissimilar sequence of human TRPM5. Experiments were extended to indirect immunofluorescence microscopy of fibroblasts. The native peptide 1 could block focal adhesion staining in fibroblasts by the 10H5 antibody while focal adhesions were detected by the 10H5 antibody in the presence of the scrambled peptides 2 and 3 (Fig. 5E).
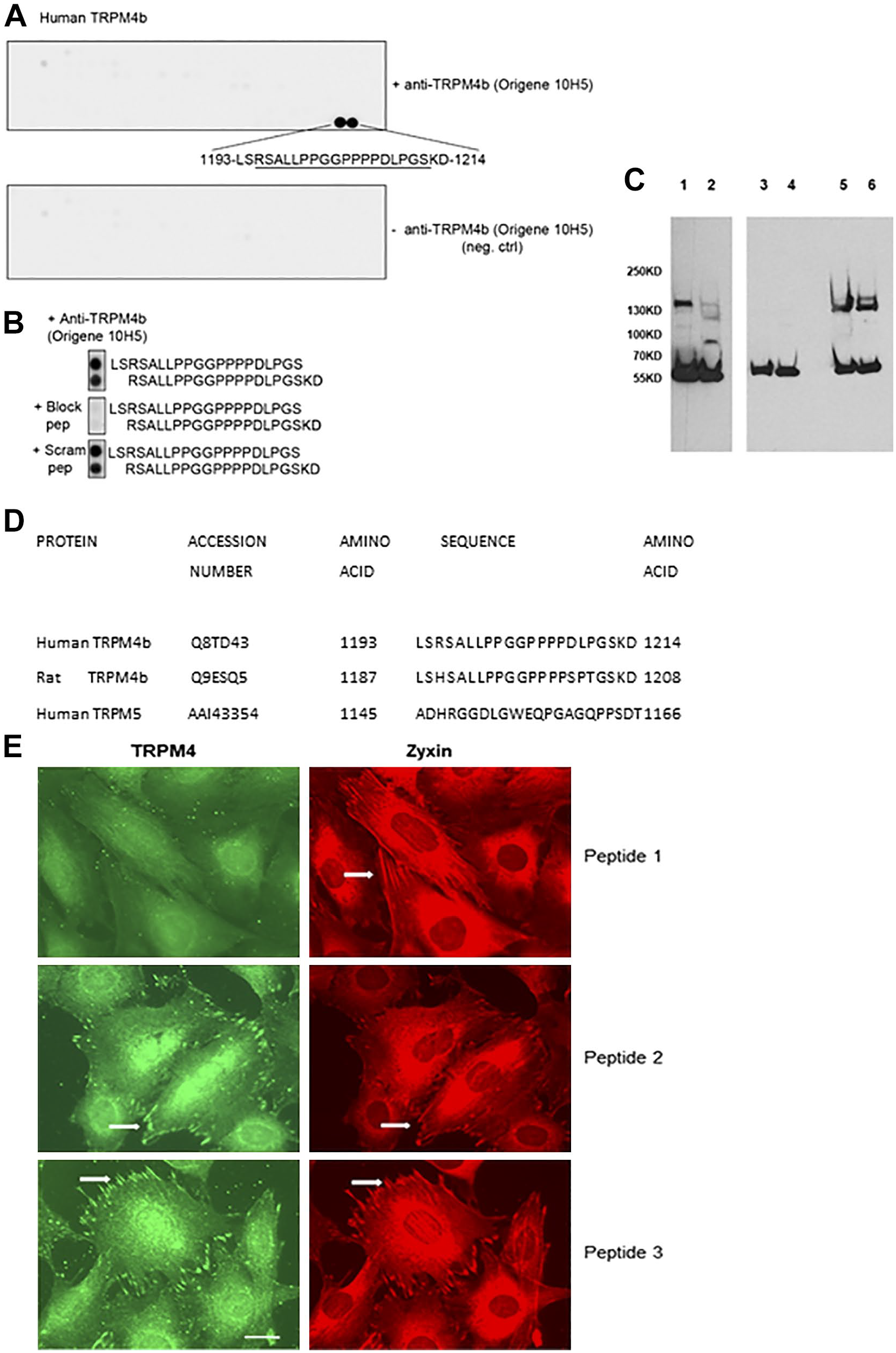
Epitope mapping of the 10H5 monoclonal antibody. (A) An overlapping series of synthetic peptides corresponding to the extracellular and intracellular domains of TRPM4b were synthesized in duplicate. Primary antibody was omitted in the lower control blot. Two single peptides were positive corresponding to amino acids 1193–1214. (B) Further controls included blocking the antibody by premixing with a 22-residue synthetic peptide corresponding to the native TRPM4b C-terminus (block pep; peptide 1) or a scrambled version of the peptide (peptide 2). (C) Western blotting of MCF-7 (lanes 1, 3, and 5) and murine embryo fibroblast lysates (lanes 2, 4, and 6) with the 10H5 antibody. In lanes 3 and 4, the antibody was premixed with the human TRPM4b peptide (peptide 1; see A) or a scrambled version in lanes 5 and 6 (peptide 2). Peptide 1 completely blocked the antibody, while the scrambled version, peptide 2, did not. The polypeptide at 58 kD is β-tubulin detected by a specific antibody. (D) Amino acid sequences of the C-termini of human and rat TRPM4b and human TRPM5. Peptide 1 corresponds exactly to the human TRPM4b sequence shown, while peptides 2 and 3 are scrambled versions (Materials and Methods). (E) Indirect immunofluorescence microscopy of murine fibroblasts stained for TRPM4 (10H5 antibody) and zyxin focal adhesion marker. Synthetic peptides corresponding to the C-terminal region (peptide 1; see A) or two scrambled versions (peptides 2 and 3) were mixed with the antibody prior to application. Only peptide 1 blocked recognition of focal adhesions (examples are arrowed). Scale bar = 10 µm. Abbreviations: MCF,Michigan Cancer Foundation.
Two major isoforms of human TRPM4 have been demonstrated previously (Fig. 1). 4 To further elucidate the localization of TRPM4, full-length cDNAs encoding GFP-tagged rat TRPM4a and TRPM4b were expressed in fibroblasts (Fig. 6). While both were readily detected, as judged by robust GFP expression, neither protein localized to focal adhesions detected by vinculin staining (Fig. 6A). A GFP control localized prominently to nuclei. Control staining experiments revealed that the sc-27540 antibody co-localized with both GFP-rTRPM4a and GFP-rTRPM4b (Fig. 6B) while the Acc-044 antibody yielded almost complete co-localization with the GFP-rTRPM4b protein but not the GFP-rTRPM4a protein (Fig. 6C). WB confirmed that the Acc-044 antibody recognized a protein of the same mass as the GFP-rTRPM4b, but not the GFP-rTRPM4a (Fig. 6D). Anti-GFP was used as a positive control to detect the fusion proteins, which also confirmed that both proteins were expressed, each having a mass consistent with that predicted from the DNA sequence, and suggested a lack of degradation (Fig. 6). These data are consistent with the known specificity of these antibodies (Fig. 1). GFP expressed from the empty vector in rat fibroblasts was recognized by the anti-GFP as a ≈25-kD protein, but not by any of the TRPM4 antibodies (Fig. 6D). Unfortunately, the sc-27540 did not function in western blots, perhaps indicating that it recognized a native but not denatured protein.
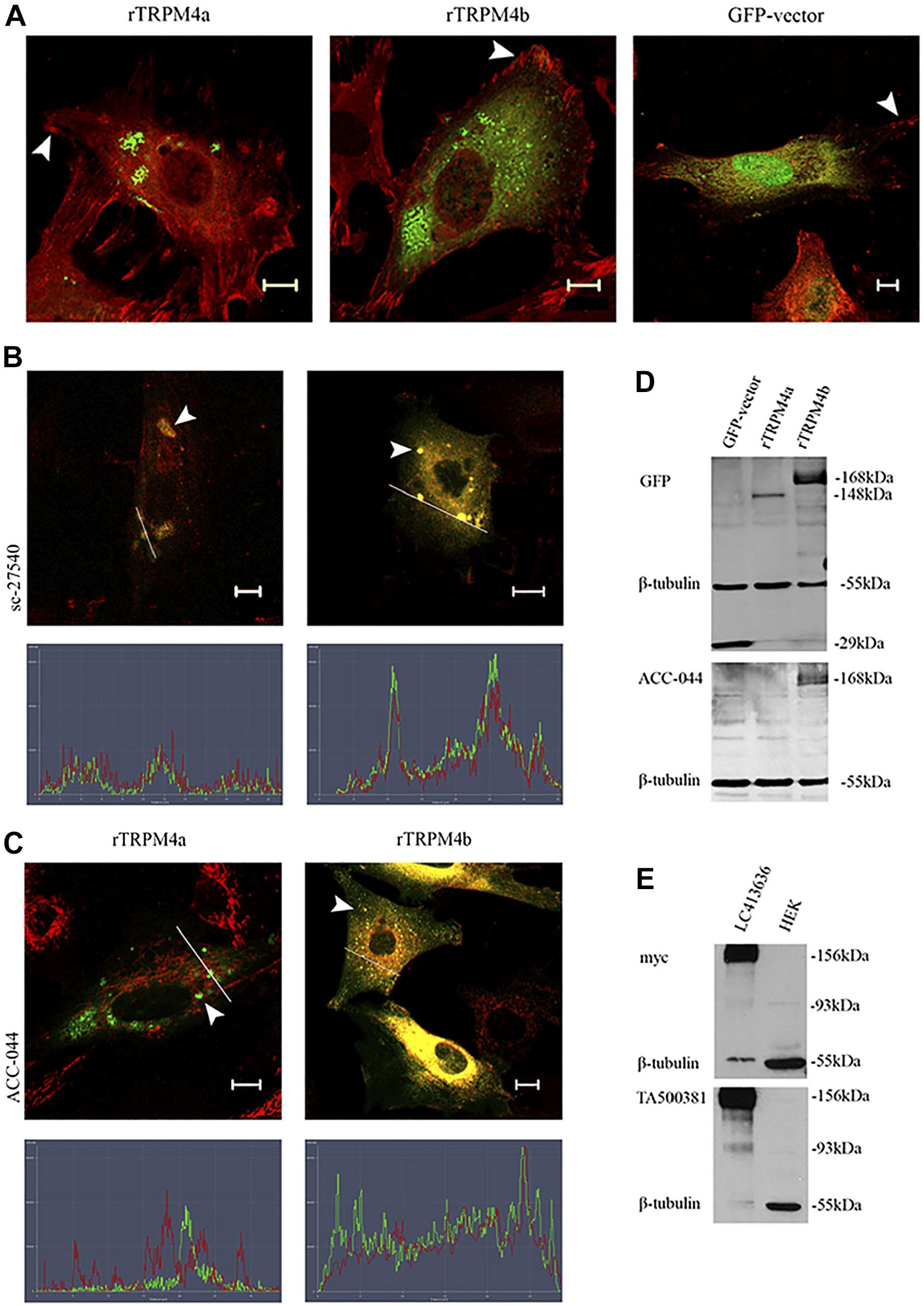
Recombinant expression of TRPM4 does not lead to focal adhesion localization. (A) GFP-TRPM4a (left) and GFP-TRPM4b (center) of rat origin do not co-localize to rat embryo fibroblast focal adhesions (zyxin, red). GFP alone (right panel) localizes strongly to nuclei. Confocal microscopy images. (B) Both GFP-TRPM4a (left) and GFP-TRPM4b (right) are recognized by the sc-27540 antibody (red), analyzed by confocal microscopy and line scanning. (C) GFP-TRPM4b (right) but not GFP-TRPM4a (left) is recognized by the Acc-044 antibody (red) as judged by confocal microscopy with line scanning. (D) The top left panel shows that anti-GFP identifies GFP alone (lane 1), GFP-TRPM4a (lane 2), and GFP-TRPM4b (lane 3) in rat embryo fibroblast lysates. The lower left panel is an identical western blot probed with the Acc-044 antibody, which recognizes TRPM4b only, consistent with its known specificity. (E) Western blots show commercial control HEK lysates (right lanes) and HEK expressing myc-hTRPM4b (left lanes) probed with anti-myc (upper blot) and 10H5 (TA50038; lower blot). This demonstrates that the 10H5 antibody does react with full-length myc-tagged humanTRPM4b. Abbreviations: HEK, Human Embryonic Kidney.
The 10H5 antibody that stained focal adhesions did not recognize full-length GFP-rTRPM4 fusion protein in western blots, but this was consistent with the ICC (not shown). However, we also tested cell lysates from wild-type human embryonic kidney (HEK) cells and HEK cells over-expressing a myc-tagged human TRPM4b (Fig. 6E). The 10H5 antibody detected a polypeptide of predicted mass of TRPM4b only in cells expressing the myc-hTRPM4b but not in untransfected cells. A 93kD protein (lacking myc) was also detected, but again not in untransfected cells. The myc-TRPM4b fusion protein was confirmed by WB with a myc-specific antibody (Fig. 6E). All these data suggested that both full-length and a truncated form of TRPM4 were recognized by the 10H5 antibody.
There remained the possibility that in addition to recognizing the TRPM4b protein, 10H5 might also recognize an additional component that is localized to focal adhesions. To address this directly, we carried out CRISPR/Cas9 deletion of TRPM4 in murine fibroblasts (Fig. 7). The guide RNAs were designed in such a way that all known isoforms of the protein should be absent. WB with both the 10H5 and Acc044 antibodies showed that the TRPM4 protein was present in wild-type MEFs but absent after CRISPR/Cas9-mediated deletion (Fig. 7A). Moreover, staining of focal adhesions by antibody 10H5 was abrogated in deleted but not control cells, strongly arguing that TRPM4 is a genuine focal adhesion component. Moreover, deletion of the channel did not impact either the presence of TRPC7 or other focal adhesion components (Fig. 7B). All the data together suggest that TRPM4 is not required for focal adhesion assembly.
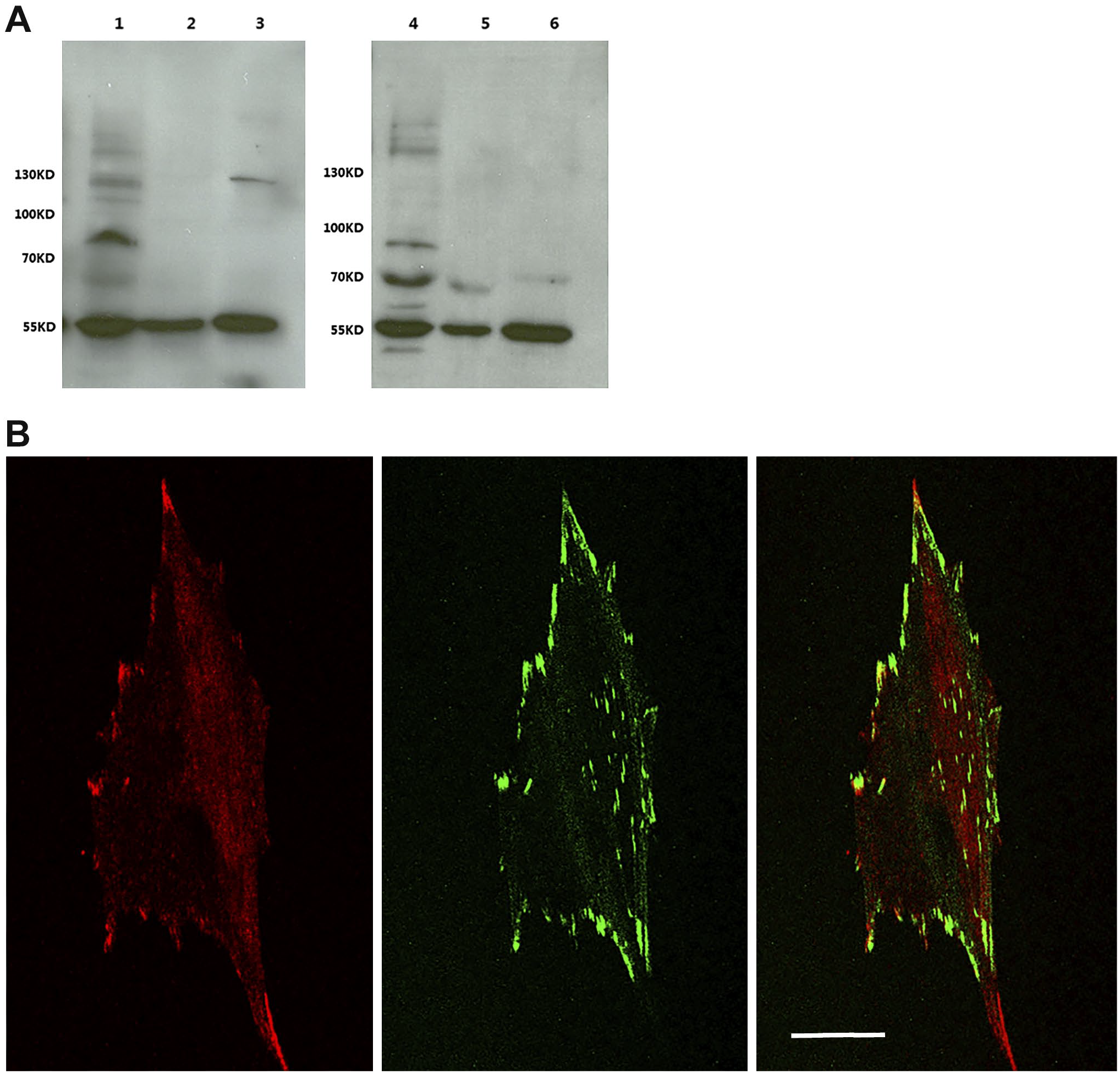
Murine fibroblasts treated with a CRISPR-Cas9 technique to deplete TRPM4 isoforms reveal that even in the absence of the channel, focal adhesions still form. (A) Lane 1 is control cells and lanes 2 and 3 are lysates from the use of two different guide RNAs (TRPM4-1 and TRPM4-2, respectively). The left blot is probed with the 10H5 antibody, while the right blot is probed with the Acc-044 antibody. The polypeptide at 58 kD is β-tubulin present in all lanes. (B) Immunofluorescence image of a TRPM4-depleted fibroblast (TRPM4-2) stained for TRPM4 (red) and zyxin (green). A merged image is shown in the right panel. Although highly TRPM4-depleted, focal adhesions still formed, indicating that the ion channel is not required for focal adhesion assembly. Bar = 10 µm.
Discussion
The TRPM4 cation channel has attracted considerable interest since it has involvement in some forms of cardiac arrthymia. 22 Our studies on ion channels that regulate cell–matrix adhesion were complicated considerably by the finding that only one of three commercial antibodies against TRPM4 stained focal adhesions of fibroblasts. Moreover, this single antibody, 10H5/TA500381, did not appear to recognize GFP-tagged full-length rat TRPM4a or TRPM4b by ICC or WB. However, it did recognize full-length myc-tagged human TRPM4b expressed in HEK cells in western blots and a polypeptide corresponding to full-length TRPM4 in MCF-7 human mammary cells. The reason for these apparently contradictory results is unclear, but it should be noted that the GFP-TRPM4a and GFP-TRPM4b were of rat origin, and the myc-TRPM4 was human. While the 10H5 antibody could detect endogenous TRPM4 in rat fibroblasts both by ICC and WB, recognition of rat TRPM4 was not as robust as it is for human TRPM4. The alignment of the key C-terminal peptides that are recognized by the 10H5 antibody shows a few amino acid differences between rat and human TRPM4b, which may explain these results (Fig. 5C).
It is also conceivable that N-terminal tagging of the TRPM4 protein with GFP sterically hinders its movement into focal adhesions. In this regard, cryo-electron microscopy data revealed that the N-terminal region of the MHR1 domain (Fig. 1) interacts with MHR3 of a neighboring molecule in the mature homotetramer. 23 The presence of GFP at the N-terminus may inhibit TRPM4 oligomerization. Moreover, the binding of the N-terminus-directed Acc-044 antibody may also be compromised in the homotetramer. The sc-27540 antibody putatively binds in the MHR2 region. Therefore, it is possible that homomeric and/or heteromeric molecular interactions of the MHR motifs may prevent access to the two antibodies that do not stain focal adhesions. In addition, recent data suggest that TRPM4 interacts with microtubule end proteins EB1 and EB2 through an SxIP motif very close to the N-terminus, which is required for modulation of focal adhesion dynamics. 24 This tetrameric peptide lies within the region recognized by the ACC-044 antibody. Once again, it is possible that EB1/2 binding to TRPM4 blocks access to the ACC-044 antibody. The mechanisms by which this channel and TRPC7 localize to focal adhesions remain to be established.
The structure of TRPM4 also indicates that the C-terminal peptide recognized by the 10H5 antibody lies immediately distal to a coiled-coil domain that is involved in homo-tetramerization. Therefore, in such a structure, four copies of the C-terminal domain lie in immediate proximity and may be required for 10H5 binding. Moreover, in the homotetramer of TRPM4, these C-terminal domains are exposed at the (cytoplasmic) base of the complex and therefore potentially accessible to antibody. 22
In humans, three TRPM4 isoforms derived from alternate splicing have been identified, but in mice, TRPM4b was identified and also other isoforms not correlating with TRPM4a or TRPM4c. 21 Therefore, it appears that multiple isoforms may exist, even without the added possibility of post-translational protein processing. In this regard, there may be short TRPM4 isoforms expressed that would not be subject to silencing in our CRISPR knockouts. This may explain the weak peripheral staining seen in Fig. 7B. Given that focal adhesion proteomics has not identified any TRP channel,13,15 it will be a challenge to characterize the TRPM4 polypeptide(s) in focal adhesions that are identified by 10H5 ICC.
There can be concerns that antibodies, whether monoclonal or particularly polyclonal, cross-react with other proteins. TRPM4 has around 50% homology with TRPM5, but our studies with a panel of antibodies indicated that none of the other TRPM proteins can be detected in focal adhesions. Moreover, the C-terminal sequence of human TRPM5 does not resemble that of TRPM4 (Fig. 5C). In addition, the results of CRISPR/Cas9 deletion of TRPM4 and peptide adsorption experiments support that a TRPM4 isoform is a focal adhesion component.
The single previous study identifying TRPM4 in focal adhesions 7 used the same 10H5 antibody as here. These authors, using channel inhibitor and small interfering RNA data, conclude that the channel is involved in regulating the number, size, and turnover of focal adhesions 7 through a RacGTPase-mediated pathway. It is well known that RhoGTPase family members, including Rac1, are involved in the regulation of actin cytoskeleton and cell migration. 25 It was previously shown that mast cells derived from TRPM4−/− mice exhibited defective migration, thought to result from altered calcium regulation. 26 This channel is a modulator of membrane potential, and therefore a possible driver of Ca2+ entry. 4 In that regard, it is interesting that the other focal adhesion ion channel identified to date is the calcium channel TRPC7. This is, at least in part, regulated by protein kinase C, and we have shown that the kinase can be regulated in turn by the cell surface proteoglycan, syndecan-4. 6 It has been reported that TRPC3, a close homolog of TRPC7, can form heteromers with TRPM4. 27 The co-localization of TRPC7 and TRPM4 in focal adhesions has not been studied previously. However, analysis of our fibroblasts lacking TRPM4 shows that at least the presence of TRPC7 in focal adhesions does not depend on TRPM4. Furthermore, we noted differences in the localization of the two channels, which only partially overlapped. Most adhesions contained one of the two channels, with TRPM4 restricted to peripheral adhesions and lacking from central adhesions. Whether these two channels can form heteromeric complexes with each other is not resolved, but our data suggest that such complexes are not required for focal adhesion localization. We speculate that the presence or absence of these channels in focal adhesions may be a reflection of their status, i.e., newly formed, maturing, or turning over.28–30 These interesting possibilities can be explored in the future.
Since TRPM4 is characteristic of some focal adhesion populations, its role remains to be understood. More generally, the presence of TRP channels in focal adhesions raises interesting questions regarding how the local ionic environment, in what is a specialized microdomain, influences kinase activities, e.g., FAK, Src, p130Cas, phosphatidylinositol kinases, and protein kinase C31–34 but also actin cytoskeletal organization. It has recently been suggested that TRPM4 can regulate Ca2+/calpain-mediated cleavage of FAK. 35 Given these data, TRP channels may regulate focal adhesion size and turnover, two important factors in the regulation of cell motility.
Footnotes
Acknowledgements
We thank Professor Jae-Yong Park (Gyeongsang National University College of Medicine, Jinju, South Korea) for the GFP-rat TRPM4 cDNA constructs.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors have contributed to this article as follows: experimental work (IM, CRC, HABM, and JRC), essential advice and input to the CRISPR experiments (CB), and all authors contributed to the preparation and editing of the manuscript and approved the submitted version. This article was authored by Ioli Mitsou during her studies at the University of Copenhagen. The findings and opinions expressed in this work belong solely to the author and do not reflect the findings and opinions of, nor is this work attributable to the author’s employment at, Agilent Technologies, Inc.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
