Abstract
The endothelin (ET) receptor system is markedly involved in the regulation of renal function under both physiological and pathophysiological conditions. The present study determined the detailed cellular localization of both ET receptor subtypes, ETA and ETB, in the vascular and tubular system of the rat kidney by immunofluorescence microscopy. In the vascular system we observed both ETA and ETB receptors in the media of interlobular arteries and afferent and efferent arterioles. In interlobar and arcuate arteries, only ETA receptors were present on vascular smooth muscle cells. ETB receptor immunoreactivity was sparse on endothelial cells of renal arteries, whereas there was strong labeling of peritubular and glomerular capillaries as well as vasa recta endothelium. ETA receptors were evident on glomerular mesangial cells and pericytes of descending vasa recta bundles. In the renal tubular system, ETB receptors were located in epithelial cells of proximal tubules and inner medullary collecting ducts, whereas ETA receptors were found in distal tubules and cortical collecting ducts. Distribution of ETA and ETB receptors in the vascular and tubular system of the rat kidney reported in the present study supports the concept that both ET receptor subtypes cooperate in mediating renal cortical vasoconstriction but exert differential and partially antagonistic effects on renal medullary function.
E
ET-1 acts through two seven-transmembrane G-protein-coupled receptors, the ETA and the ETB receptor. Renal effects of ET-1 have been ascribed to both receptor subtypes by the use of non-selective and selective ET receptor agonists and antagonists. In the rat, proliferative effects of ET-1 on mesangial cells are mediated by the ETA receptor subtype (Fukuda et al. 1996), and stimulation of ETB receptors inhibits sodium and water reabsorption (Watanabe et al. 1991; Gariepy et al. 2000). ETA as well as ETB receptors are involved in the regulation of renal vascular resistance. Whereas stimulation of ETA receptors leads to vasoconstriction of preglomerular arteries and efferent arterioles (Lanese and Conger 1993), the ETB receptor is able to mediate both vasodilation and vasoconstriction. ETB receptor stimulation has been coupled to the release of nitric oxide (NO) and prostacyclin (Hyslop and de Nucci 1992), leading to vasodilation. In addition, presence of ETB receptors on vascular smooth muscle cells has been observed in different vascular beds (Wendel-Wellner et al. 2002; Wendel et al. 2005), and the ETB receptor subtype was considered to be the main receptor mediating ET-1-induced renal vasoconstriction in the rat (Wellings et al. 1994; Endlich et al. 1996).
Many studies demonstrate in vivo and in vitro binding of radioactively labeled ET-1 (125I-ET-1). Concurringly, these studies identified the glomeruli, longitudinal stripes traversing the medulla, as well as the outer part of the inner medulla as major sites of 125I-ET-1 binding, Interglomerular regions showed only sparse labeling, and the outer stripe of the outer medulla was completely negative (Davenport et al. 1989; Kohzuki et al. 1989). However, data are inconsistent concerning the determination of cellular binding sites by high-resolution light and electron microscopy (Furuya et al. 1992; Dean et al. 1994,1996; Yukimura et al. 1996). Furthermore, the design of these autoradiographic studies did not allow discrimination between receptor subtypes. Few autoradiographic studies have addressed this issue by use of unlabeled ET-receptor-subtype-specific ligands. Using 125I-ET-1 binding in the presence of excess of unlabeled ETA or ETB receptor ligands, Yukimura et al. (1996), as well as Dean et al. (1996), reported that the ETB receptor agonist sarafotoxin S6c abolishes 125I-ET-1 binding almost completely with exception of binding to the inner medulla, which also could not be abolished by a combination of ETA and ETB receptor antagonists.
Direct evidence for the cellular distribution of ET receptor subtypes at the level of the receptor protein is limited. No data for the ETA receptor are available, and only two studies used immunohistochemistry (IHC) for the ETB receptor (Hagiwara et al. 1993; Yamamoto and Uemura 1998). Whereas Yamamoto and Uemura (1998) observed strong ETB receptor immunoreactivity of renal proximal tubules and only fine immunoreactive puncta over glomerular capillaries, Hagiwara et al. (1993) reported strong ETB receptor immunoreactivity of glomerular and peritubular capillaries, as well as vasa recta. The latter results are in accordance with previous binding studies indicating highest densities of 125I-ET-1 binding to glomerular and peritubular capillaries.
On this basis, we studied the detailed cellular distribution of both ET receptor subtypes in the rat kidney by IHC using ETA- and ETB-receptor-specific antisera. To identify the cellular distribution of ET receptor immunoreactivity, IHC double labeling was carried out with specific markers for vascular endothelial cells and defined segments of the renal tubular system.
Materials and Methods
Animals
Ten adult Wistar rats of both sexes were used. Rats were killed by chloroform inhalation, and the kidneys were excised and snap frozen. Care and handling of animals conformed to the guiding principles in the Care and Use of Animals as approved by the Council of the American Physiologic Society.
Antibodies
Primary antibodies used were rabbit anti-ETA receptor antibody and control peptide for the ETA receptor antibody as well as rabbit anti-aquaporin-2 antibody (Alomone Labs; Jerusalem, Israel). Rabbit anti-ETB receptor antibody α-CLK23 (Cramer et al. 1997) and its corresponding control peptide were a gift from Dr. C. Schroeder (Mainz, Germany). Mouse anti-RECA-1, a panendothelial cell marker (Duijvestijn et al. 1992), was purchased from Serotec (Oxford, UK), mouse-anti-calbindin D28K was from Sigma (Deisenhofen, Germany), and mouse anti-N+/H+ exchanger isoform NHE3 clone 2B9 was from Chemicon (Hofheim, Germany). Mouse anti-desmin clone DE-R-11 was from Biozol (Eching, Germany). Secondary antibodies used were Cy3-conjugated donkey anti-rabbit-IgG, Cy3-conjugated goat anti-mouse IgG, fluorescein isothiocyanate (FITC)-conjugated goat anti-rabbit-IgG, and FITC-conjugated goat anti-mouse-IgG (all from Dianova; Munich, Germany).
Western Blotting
For Western blotting, kidneys from two rats were homogenized in 0.1 M HEPES buffer containing 250 mM sucrose (Merck; Darmstadt, Germany), 5 mM EDTA, 0.5% CHAPS, 2 μM pepstatin A, and 2 μM leupeptin (all from Sigma). In one preparation, the EDTA concentration in the homogenization buffer was increased to 50 mM to inhibit proteolytic degradation of the ETB receptor molecule. After centrifugation at 2500 × g and 4C for 10 min, the supernatant was collected and centrifuged at 100,000 × g and 4C for 1 hr. Protein content of the pellet representing the membrane fraction was determined using the Micro BCA Protein Assay Reagent Kit (Perbio Science; Bonn, Germany). Twenty-μg protein samples were then separated by SDS-PAGE on a gradient gel (10-15% acrylamide) and transferred to a polyvinylidene fluoride membrane (PVDF; Roth, Karlsruhe, Germany). Blots were blocked with 5% dry milk powder (Roth) in Tris-buffered saline (TBS), pH 7.4, for 1 hr. Primary antibodies were diluted in TBS containing 0.05% Tween (TBST; Serva, Heidelberg, Germany) and 2.5% dry milk powder. Blots were incubated with either the ETA receptor antibody (1:800) or the ETB receptor antibody (1:7500) at 4C overnight. Blots were then washed twice with TBST and incubated with horseradish peroxidase (HRP)-conjugated anti-rabbit immunoglobulin (1:5000; New England BioLabs, Frankfurt, Germany) for 1 hr at room temperature. Enhanced chemiluminescence was performed with the ECL Western blot detection kit (Amersham; Freiburg, Germany) according to the manufacturer's instructions. Blots were exposed to hyperfilm (Amersham) for 30 min.
For determination of molecular mass, a broad-range biotinylated SDS-PAGE standard (BioRad; Munich, Germany) was used and visualized using anti-biotin HRP-linked antibody (1:2000; New England BioLabs) and enhanced chemiluminescence as described above.
Immunofluorescence
Six-μm-thick sections of rat kidneys were fixed either by acetone for 10 min at −20C or by Cytofix/Cytoperm buffer (BD Biosciences; Heidelberg, Germany) for 10 min at 4C. After blocking with 10% normal porcine serum in PBS for 30 min, sections were incubated with primary antibodies overnight at room temperature. In the double-labeling protocol, sections were incubated with either ETA (1:800) or ETB receptor (1:1000) antiserum together with either anticalbindin DK28 (1:400), anti-RECA-1 (1:40), anti-NHE3 (1:800), or anti-desmin (1:400). In the single-labeling protocol, consecutive sections were labeled with anti-ETA or anti-ETB receptor antiserum and anti-aquaporin-2 (1:800). After rinsing twice in PBS, pH 7.4, sections were incubated with fluorochrome-conjugated secondary antibodies for 1 hr at room temperature. After rinsing twice in PBS, sections were coverslipped with carbonate-buffered glycerol, pH 8.6, and evaluated by epifluorescence with an Olympus BX 20 microscope (Olympus; Hamburg, Germany).
Preabsorption
Specificity of ETA and ETB receptor immunoreactivity was controlled by preabsorbing the antisera with their corresponding peptides used for immunization at a concentration of 100 μg/ml for 14 hr at 4C.
Results
Western Blot
In Western blots of rat kidney membrane preparations, the ETA receptor antibody recognized a single band of ~54 kDa. For the ETB receptor, we observed three bands of ~34, 52, and 60 kDa (Figure 1).
Immunofluorescence
In large intrarenal (interlobar and arcuate) arteries and in veins, ETA receptors were detected on vascular smooth muscle cells (Figures 2A and 2B), whereas in the media of interlobular arteries as well as afferent and efferent arterioles both ETA and ETB receptors were present (Figures 2C-2F). Weak ETB receptor immuno-reactivity was observed in vascular endothelial cells of preglomerular arteries (Figure 2E), whereas peritubular capillary endothelial cells in the renal cortex displayed strong ETB receptor immunoreactivity (Figure 2F). In the glomeruli, both ET receptor subtypes were detected (Figure 3). Whereas both ETA and ETB receptors were present on mesangial cells, only ETB receptor immunoreactivity colocalized with RECA-1 on glomerular endothelial cells.
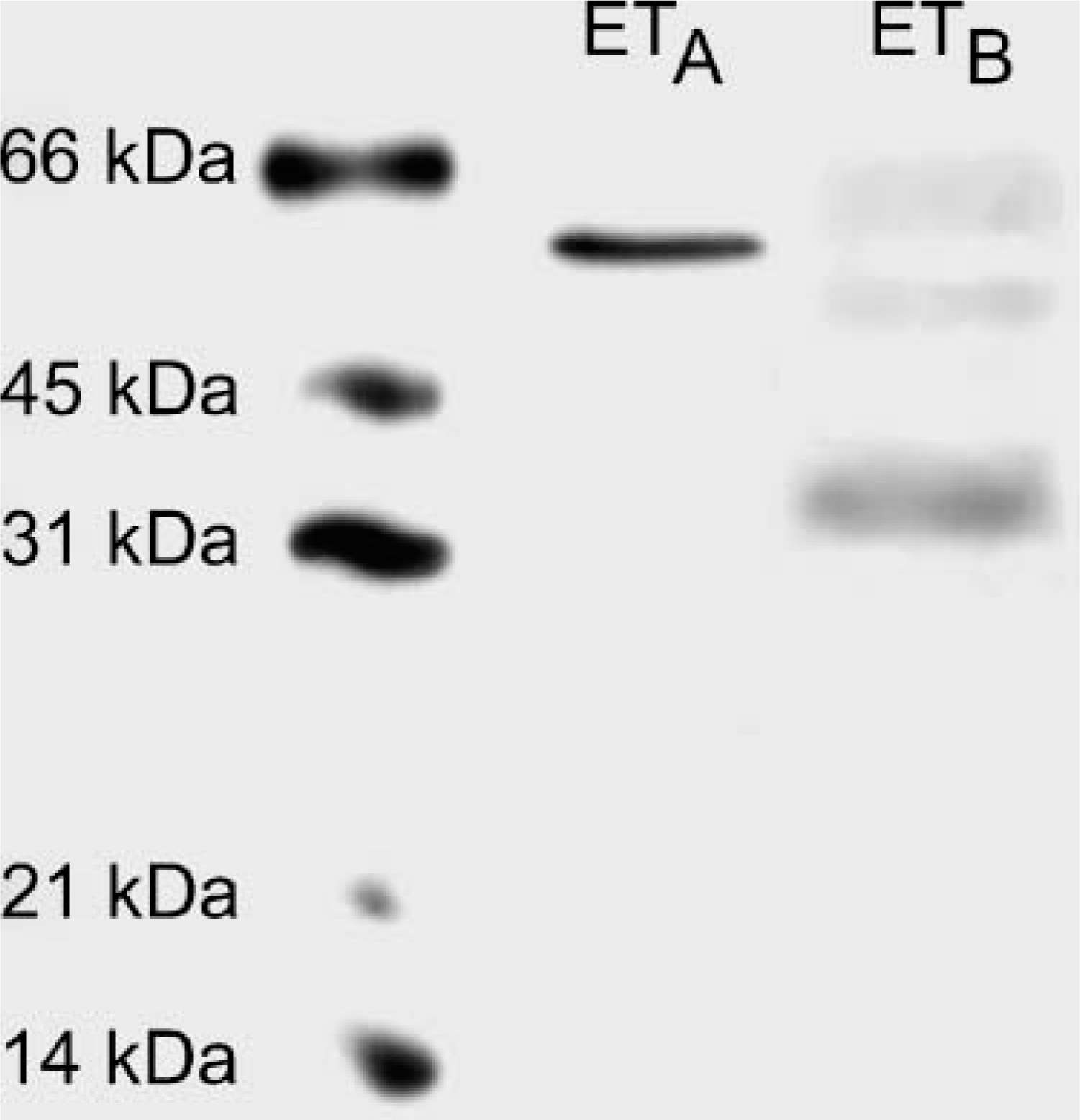
Western blot. Rat kidney membrane fractions were isolated, loaded on a SDS-polyacrylamide gel (20 μg protein/lane), and blotted. Incubation with the ETA receptor antibody resulted in a single band of ~54 kDa, whereas the ETB receptor antibody recognized three bands of ~34, 52, and 60 kDa.
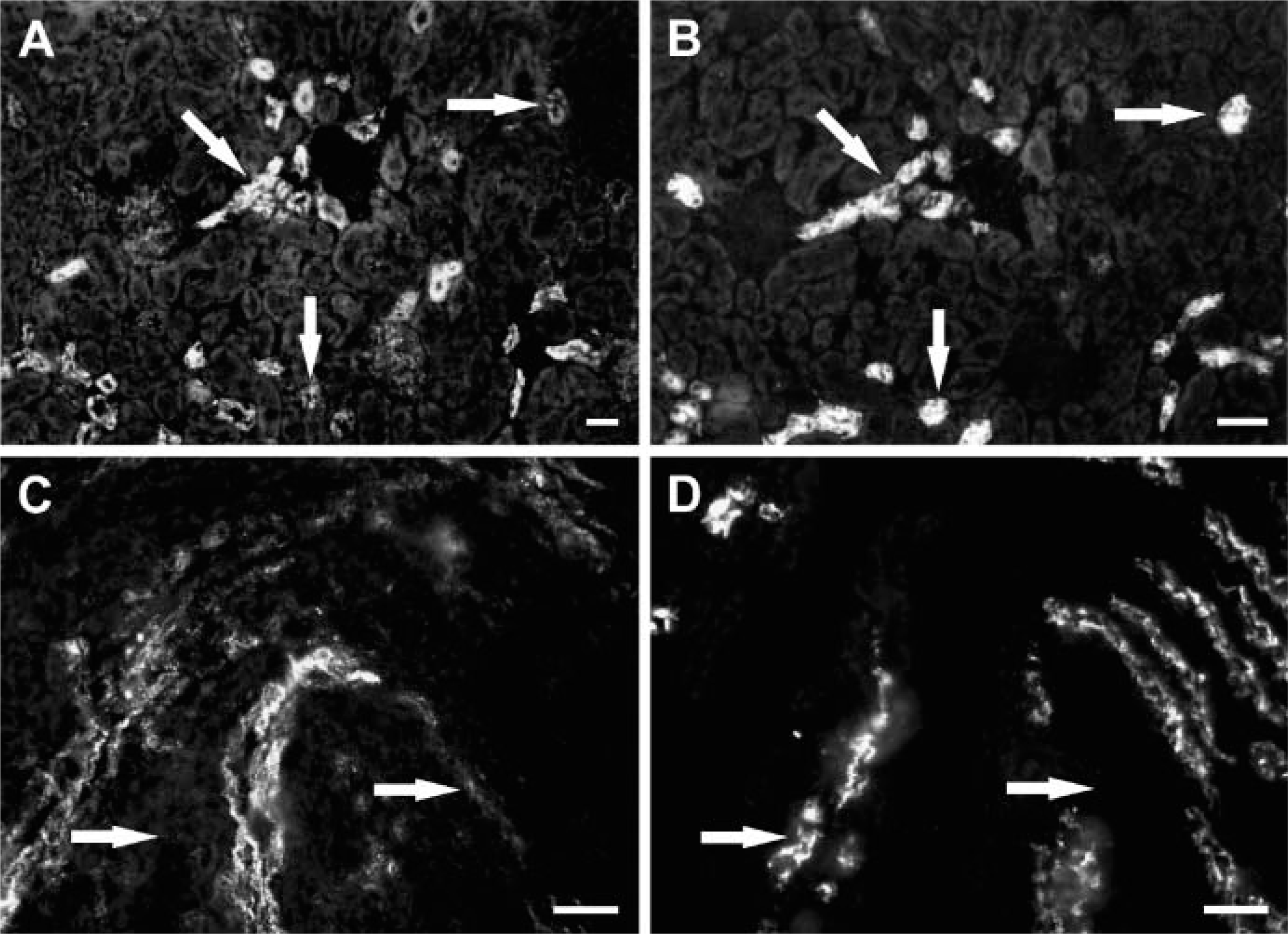
Segments of the rat renal tubulus system were identified by labeling with specific markers. Collecting ducts were identified by aquaporin-2 immunoreactivity. In serial sections, we observed ETA receptor immunoreactivity of epithelial cells in cortical but not in outer medullary collecting ducts (Figure 4). In the distal tubulus identified by calbindin D28K immunoreactivity, partial colocalization of calbindin with the ETA but not with the ETB receptor subtype was observed (not shown). Proximal tubules were identified by NHE3 immunoreactivity in the renal cortex. This antibody also specifically labels the thick ascending and thin limbs of Henle (Biemesderfer et al. 1997). By double-labeling immunofluorescence, ETB but not ETA receptor immunoreactivity colocalized with NHE3 in proximal tubules, whereas in the loop of Henle neither ETA nor ETB receptor immunoreactivity was detected (Figure 5).
In descending vasa recta (DVR) bundles and in medullary sections, both ETA and ETB receptor immunoreactivity were observed (Figure 5). ETB receptors colocalized with the panendothelial cell marker RECA-1 in DVRs. In the inner medulla, ETB receptors were present on capillary endothelial cells identified by RECA-1 and were also detected on epithelial cells of inner medullary collecting ducts (IMCDs). ETA receptor immunoreactivity of IMCDs was faint. In DVR bundles, ETA receptor immunoreactivity was distinct from RECA-1 but showed colocalization with desmin on pericytes of DVRs.
Specificity Control of ET A and ETB Receptor Immunoreactivities
Both ETA and ETB receptor immunoreactivity were completely abolished by preabsorbing the antisera with the corresponding antigen used for immunization (Figure 6).
Discussion
In the present study we report the detailed distribution of ETA and ETB receptor proteins in the vascular and tubular system of the rat kidney. We used specific antibodies against ETA and ETB receptors, which were characterized by Western blotting of rat kidneys. For both antibodies, bands of characteristic molecular sizes were obtained. The ETA receptor antibody recognized a single band of ~54 kDa. For the ETB receptor antibody, we obtained bands of ~34, 52, and 60 kDa. In a previous study we used the same antibody in rat lungs and obtained bands of ~52 and 34 kDa (Wendel et al. 2004) in accordance with the report by Kozuka et al. (1991). In the present study we observed an additional band of ~60 kDa. This observation is in line with the findings of Cramer et al. (1997) who characterized the antiserum in transfected cells expressing the human ETB receptor and in isolated kidney epithelial cells. In this study, bands of ~41 and 45 kDa were obtained in cells overexpressing the ETB receptor protein, whereas in preparations of renal epithelial cells an additional band of higher molecular mass was present. No bands were observed when preimmune serum was used. Most probably, the band with higher molecular mass in renal epithelial cells reflects cell type-specific posttranslational modification of the ETB receptor molecule.
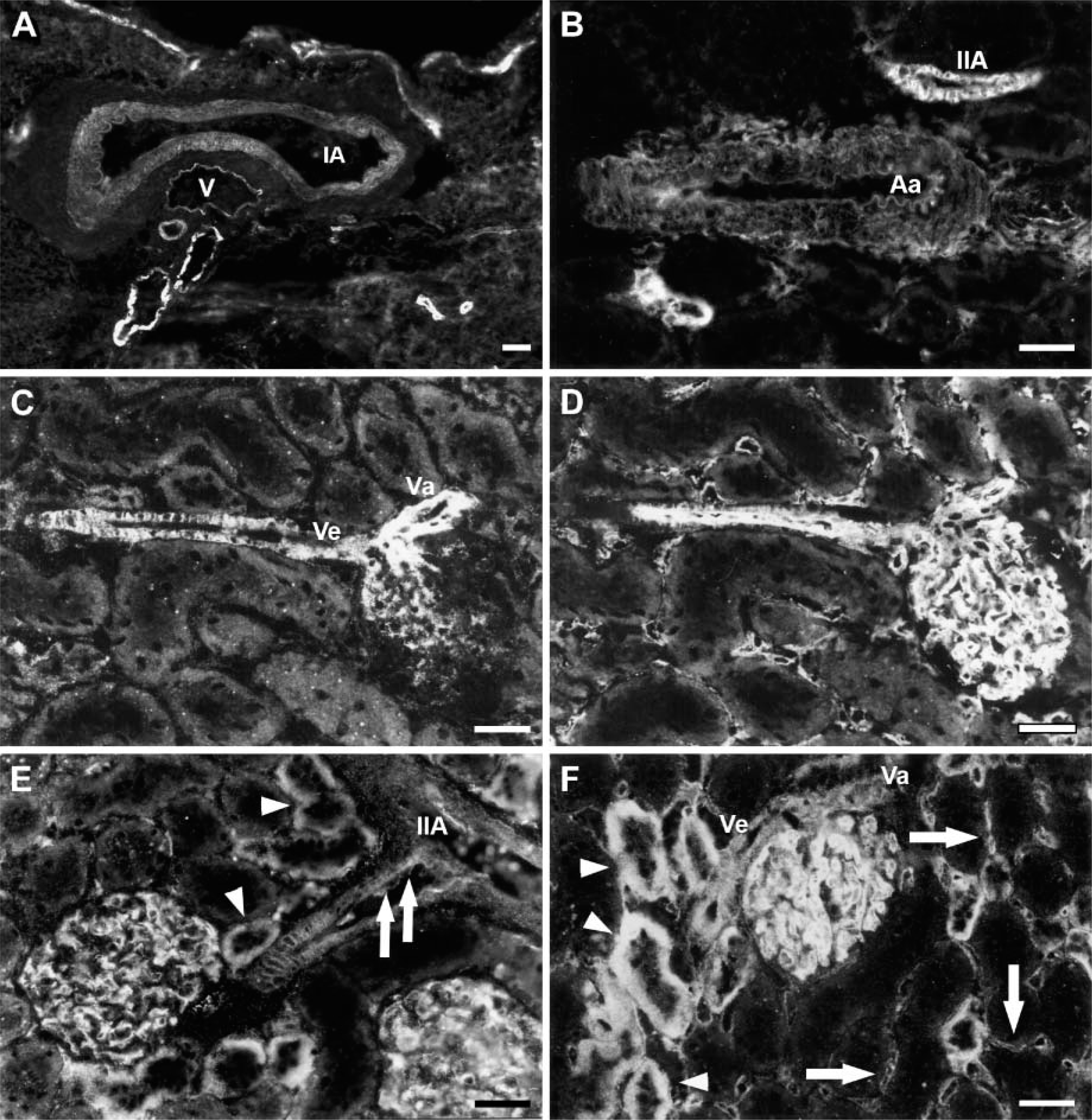
ETA and ETB receptor immunoreactivity of renal cortical vessels and glomeruli. (
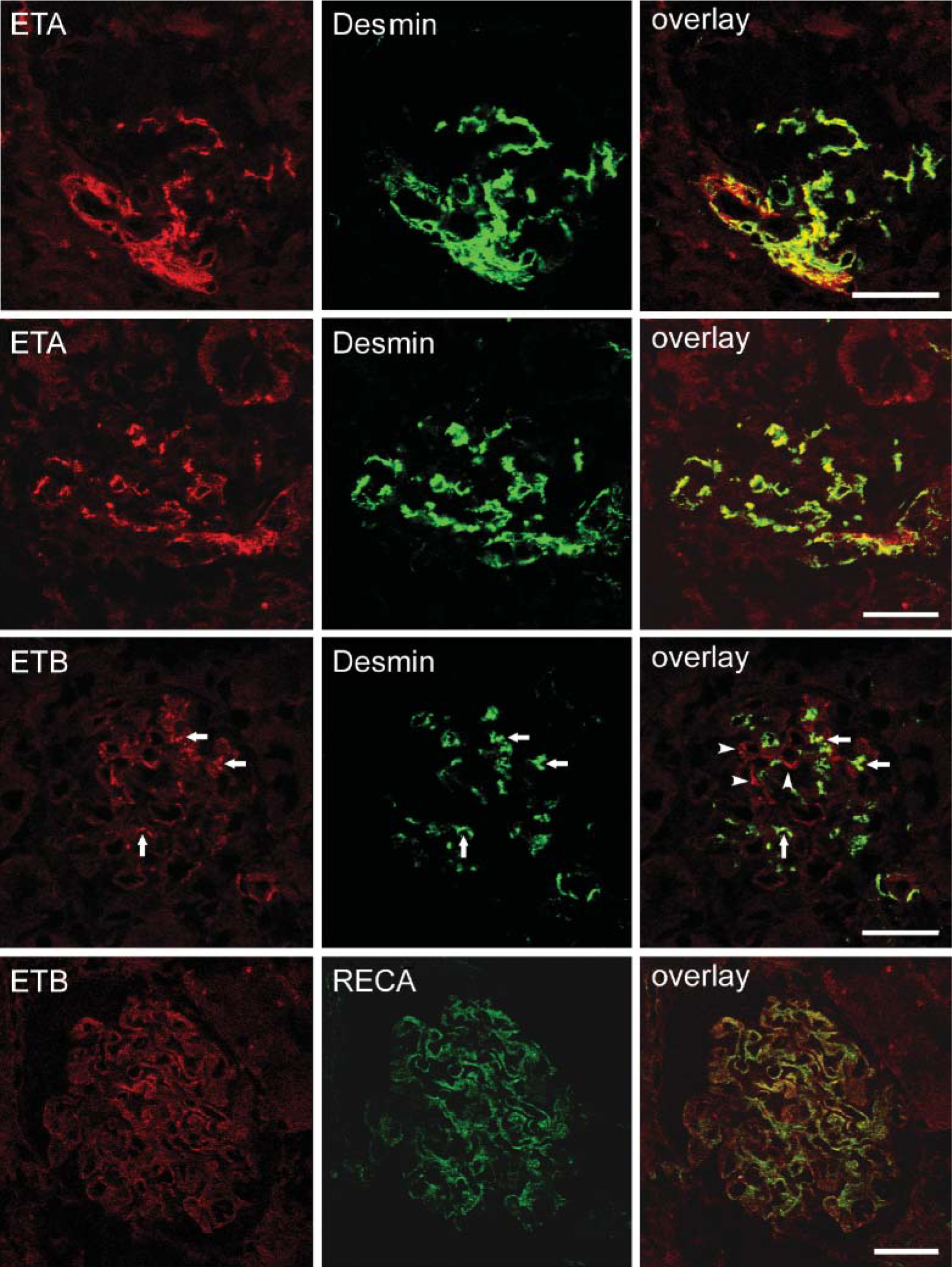
Double-labeling immunofluorescence and confocal laser scanning microscopy for ET receptors (left column), mesangial and endothelial cells identified by anti-desmin, and anti-RECA-1 antibodies (middle column) in rat glomeruli. In the right column, the corresponding overlay pictures are shown. ETA receptor immunoreactivity colocalizes with desmin on extra-and intraglomerular mesangial cells as evidenced by yellow color in the overlay pictures. For the ETB receptor subtype, partial colocalization with desmin (arrows) but also distinct ETB receptor immunoreactivity (arrowheads in the overlay picture) can be seen. Broad colocalization of ETB receptors with the endothelial cell marker RECA-1 is observed on glomerular capillary endothelial cells.
By immunofluorescence microscopy, we further confirmed the specificity of the antisera by preabsorption. Both ETA and ETB receptor immunoreactivity were completely abolished when the antiserum was preabsorbed with the peptide used for immunization.
In the glomeruli, both ETA and ETB receptor immunoreactivity could be detected and were present on distinct cell types. Double-labeling immunofluorescence with the panendothelial cell marker RECA-1 identified endothelial cells of the glomerulus possessing ETB but not ETA receptors, whereas both ET receptors were present on mesangial cells. ETB receptor immunoreactivity of glomerular capillaries has previously been described (Hagiwara et al. 1993; Yamamoto and Uemura 1998), whereas the presence of ETB receptors on mesangial cells could only be observed in culture (Owada et al. 1994). ETA-receptor-mediated mitogenic effects of ET-1 on mesangial cells are well known and correlate with protective effects of ETA receptor antagonists on the development of glomerulosclerosis in animal studies (Simonson et al. 1989; Wang et al. 1994; Ebihara et al. 1997; Dhein et al. 2000).
Single-labeling immunofluorescence of consecutive cryosections for the ETA receptor (
In the rat kidney vascular system, we found ETA receptors on smooth muscle cells of all preglomerular arteries and efferent arterioles. These findings are in accordance with the results of functional studies (Lanese and Conger 1993; Wu et al. 1997; Cavarape et al. 1998) and with the detection of ETA receptor mRNA in all renal arteries (Hocher et al. 1996). We can also confirm a weak signal for the ETB receptor in arterial endothelial cells. Although neither Hocher et al. (1996) nor Yamamoto and Uemura (1998) could detect ETB receptors on vascular smooth muscle cells by in situ hybridization and IHC, respectively, in our study ETB receptor immunoreactivity of smooth muscle cells was obvious in interlobular arteries as well as in afferent and efferent arterioles. These findings agree with previous functional studies demonstrating vasoconstriction of afferent and efferent arterioles by ET-1 being sensitive to ETA and ETB receptor antagonists (Endlich et al. 1996; Hercule and Oyekan 2000a) and with the observation of ET-3-mediated decreases in renal cortical blood flow (Hercule and Oyekan 2000b). In the medullary circulation we observed both ETA and ETB receptors in DVR. This is in line with several autoradiographic studies demonstrating 125I-ET-1 labeling of longitudinal stripes traversing the medulla (Davenport et al. 1989; Kohzuki et al. 1989) and with the IHC findings of Hagiwara et al. (1993) who reported ETB receptor immunoreactivity of DVR. In our study, ETA and ETB receptors showed differential cellular distribution in DVR bundles. Whereas ETB receptor immunoreactivity showed broad colocalization with the endothelial cell marker RECA-1, ETA receptor immunoreactivity differed from RECA-1 labeling and was found on pericytes adjacent to the endothelial cells. These observations are in line with the finding that ET-1 and ET-2 constricted isolated microperfused DVR (Pallone and Silldorff 2001), whereas infusion of ET-3 resulted in an increase in medullary perfusion. The latter, however, was converted into vasoconstriction upon cyclooxygenase blockade (Hercule and Oyekan 2000b), implying that ETB receptors in the medullary circulation are coupled to the generation of vasodilator cyclooxygenase products. Regulation of renal vascular tone by ET-1 has recently been demonstrated to be intimately linked to the cytochrome P450 (CYP450) system. CYP450-derived 20-hydroxyeicosatetraenoic acid (20-HETE) markedly contributes to ET-1-induced vasoconstriction of renal vessels (Imig et al. 1996; Oyekan et al. 1997; Hercule and Oyekan 2000a), and inhibition of 20-HETE production was shown to dampen ET-1-induced vasoconstriction. In the rat preglomerular arteriole, both ETA and ETB receptors can mediate vasoconstriction through liberation of 20-HETE (Hercule and Oyekan 2000a); however, in the renal medulla there is an antagonism between ETA-receptor-dependent vasoconstriction mediated by 20-HETE and NO-mediated vasodilation in response to ETB receptor stimulation (Hercule and Oyekan 2000b). NO is well known to suppress ET-1 gene expression (Boulanger and Luscher 1990; Kourembanas et al. 1993) and also interferes with the CYP450-system. NO inhibits several CYP450 enzymes (Khatsenko et al. 1993; Wink et al. 1993; Alonso-Galicia et al. 1997) attenuating CYP450-dependent production of arachidonic acid metabolites. Our observation that, on DVR bundles, ETA and ETB receptors are present on pericytes and endothelial cells, respectively, suggests that NO released by stimulation of endothelial ETB receptors exerts a tonic inhibitory effect on ETA-receptor-dependent pericyte contraction mediated by 20-HETE. This functional antagonism of ET-1 actions mediated by ETA-and ETB-receptor-dependent signaling pathways is essential for maintenance of medullary perfusion under pathophysiological conditions characterized by elevated levels of ET-1. A study recently published by Vassileva et al. (2003) demonstrated selective ETB-receptor-mediated medullary vasodilation in response to big-ET-1 in rats fed a high-salt diet. This model also explains the detrimental effects of ETB receptor blockade and NO synthase (NOS) inhibition. Blockade of ETB receptors aggravated functional impairment in models of ischemic acute renal failure (Forbes et al. 2001) and led to salt-sensitive hypertension (Gariepy et al. 2000) as did genetic ETB receptor deficiency (Matsumura et al. 2000). The natriuretic and diuretic effect of big-ET-1 was significantly reduced after pretreatment with the NOS-inhibitor L-NAME, implicating that NO mediates this effect (Hoffman et al. 2000). In vitro studies demonstrated expression of mRNA for the ETB receptor subtype (Terada et al. 1992) as well as the three NOS isoforms in cultured IMCDs (Mohaupt et al. 1994; Ye et al. 2003). ET-1 also inhibited vasopressin action and stimulated prostaglandin E2 accumulation (Edwards et al. 1993; Kohan et al. 1993) and eNOS gene expression (Ye et al. 2003). From these findings it was concluded that there is a local ET/ETB receptor system in IMCD cells, which upon stimulation leads to an increase in NO generation and an inhibition of sodium reabsorption. Our observation that ETB receptors are present on IMCDs in vivo is in line with these functional data.
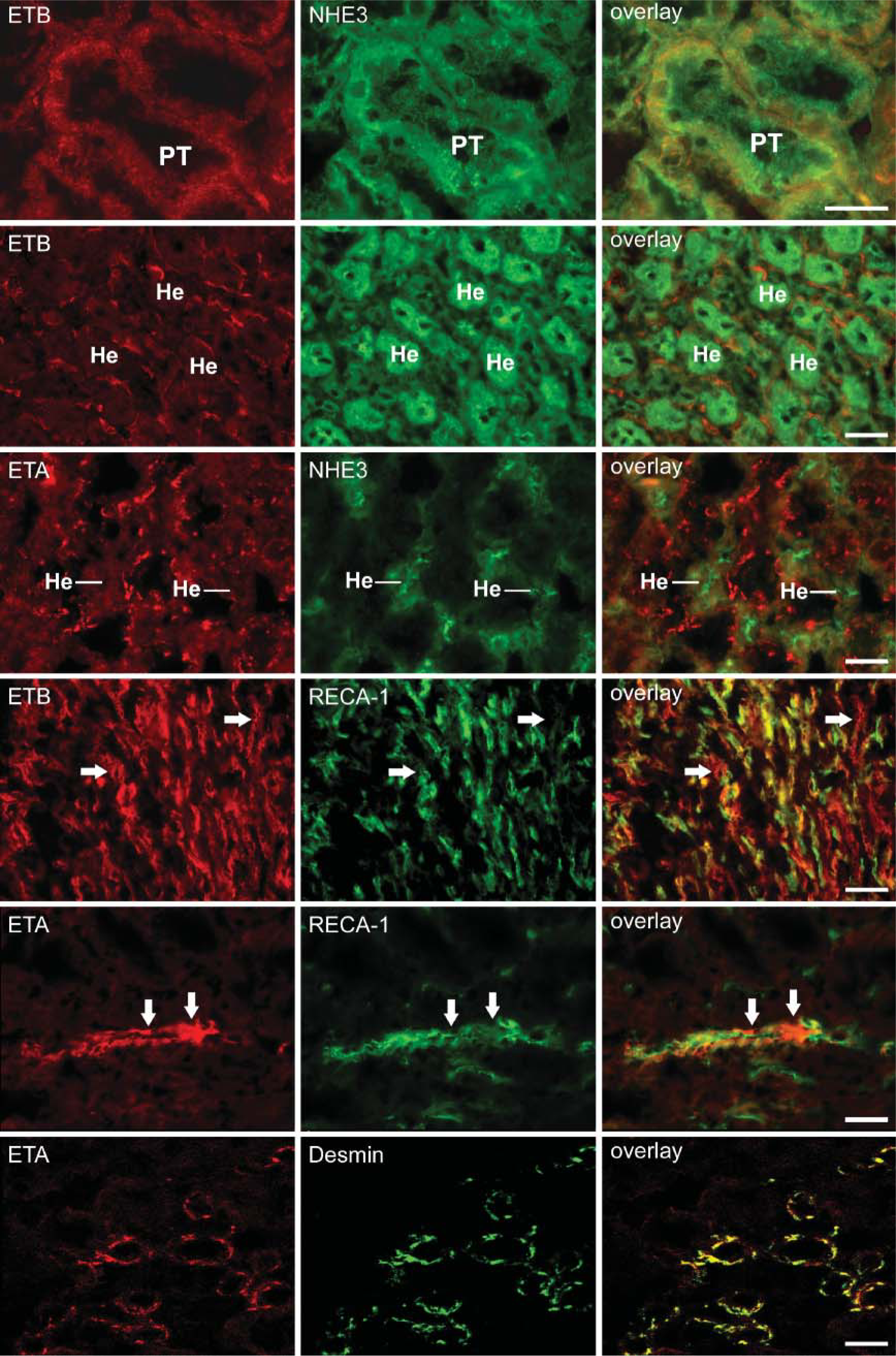
Double-labeling immunofluorescence for ET receptors (left column) and cell-type-specific markers NHE3, RECA-1, and desmin (middle column) in rat kidneys. ETB receptor immunoreactivity is evident in proximal tubules (indicated as PT) but not in the limb of Henle (indicated as He), both identified by NHE3 immunoreactivity. ETA receptor immunoreactivity also does not colocalize with NHE3 immunoreactivity in the limb of Henle. Double-labeling immunofluorescence for ETB receptors and RECA-1 shows broad colocalization in the inner medulla where labeling of inner medullary collecting duct epithelial cells is also evident (indicated by arrows). In descending vasa recta (DVR) bundles, ETA receptor immunoreactivity (indicated by arrows) is distinct from RECA-1 immunoreactivity but colocalizes with desmin on DVR pericytes (yellow color in the overlay picture). Bar = 50 μm.
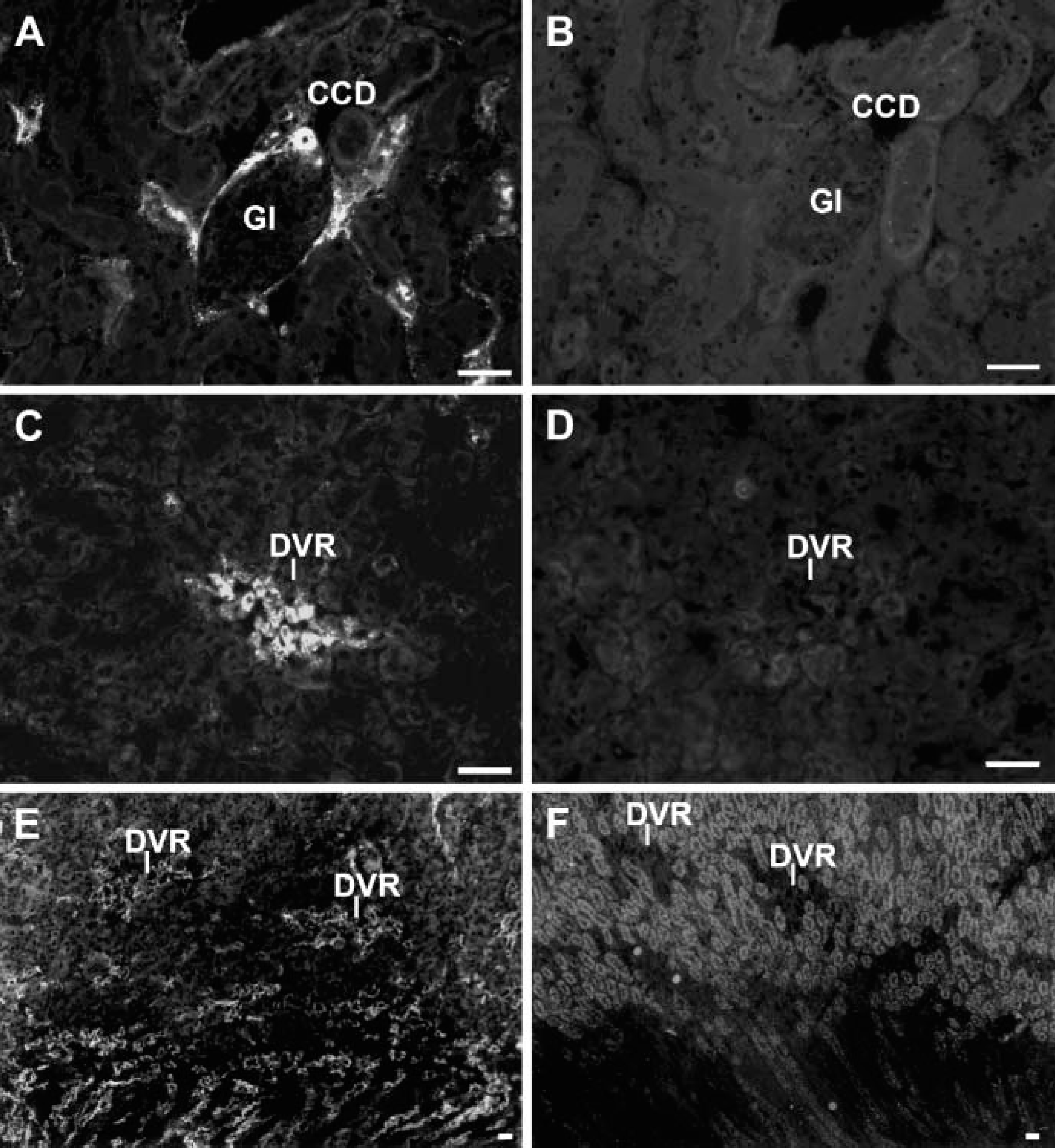
Preabsorption of ETA and ETB receptor immunoreactivity. (
In contrast to functional data showing ET-1-induced inhibition of chloride flux via ETB-receptor-mediated NO release in isolated thick ascending limbs of Henle (Bailly 2000; Plato et al. 2000), we were not able to detect ETB receptor immunoreactivity in this nephron segment. Our findings are also partially discrepant to the observations of Hocher et al. (1996) and Terada et al. (1992) who reported ETB receptor mRNA to be expressed not only in IMCDs but also in cortical collecting ducts and the loop of Henle by in situ hybridization and RT-PCR, respectively. However, previous immunohistological studies also did not confirm these observations. From these discrepant findings one might conclude that there may be large differences in the level of ETB receptor gene expression and protein content resulting in ETB receptor protein levels too low to be detected by IHC or immunofluorescence techniques. Receptor autoradiographic studies also demonstrated only sparse silver grains over basal infoldings of medullary collecting duct cells and the thin limb of Henle (Yukimura et al. 1996), whereas dense silver grains were located over peritubular spaces (Furuya et al. 1992; Dean et al. 1996; Yukimura et al. 1996).
In addition to the presence of ETB receptors in IMCD we observed ETB receptor immunoreactivity in proximal tubules of the rat kidney. Again, results from binding studies are controversial. Low 125I-ET-1 binding to proximal tubules was observed by Dean et al. (1994) and Kohzuki et al. (1989) but not confirmed by the studies of Furuya et al. (1992) and Yukimura et al. (1996). However, previous IHC studies reported that tubular ETB receptor immunoreactivity was mainly restricted to proximal tubules (Hagiwara et al. 1993; Yamamoto and Uemura 1998). These observations correlate with ETB-receptor-induced increases in NHE3 activity reported by Laghmani et al. (2001).
We also observed ETA receptor immunoreactivity of renal tubules, which was mainly found in cortical collecting ducts and parts of the distal tubulus. In the inner medulla, labeling of IMCD epithelium was faint. Previously, ET-1 actions have been observed in cortical collecting ducts and distal tubules (Korbmacher et al. 1993; Kurokawa et al. 1993; Tomita et al. 1993). However, studies by Hocher et al. (1996) and Terada et al. (1992) did not demonstrate ETA receptor mRNA in rat cortical tubules.
Despite a number of efforts in characterizing the distribution of ETA and ETB receptors in the rat kidney vascular and tubular system by means of receptor autoradiography, in situ hybridization, RT-PCR, and IHC, significant discrepancies remain in the reported cellular distributions of ET receptor subtypes. Our study, together with previously reported data on ET receptor localization and ET-1 actions, affirms the concept that ETA receptors are mainly located on contractile cells (vascular smooth muscle and mesangial cells as well as pericytes of DVR bundles) mediating cortical and medullary vasoconstriction, whereas ETB receptors are differentially distributed. In renal cortical vessels, ETB receptors are located on pre- and postglomerular arterioles contributing to renal vasoconstriction, whereas in the renal medulla, ETB receptor engagement in DVR and IMCD leads to simultaneous medullary vasodilation and inhibition of sodium reabsoprtion via ETB-receptor-mediated release of NO and cyclooxygenase products.
Footnotes
Acknowledgements
The authors thank Dr. C. Schroeder, Mainz, Germany for providing the ETB receptor antibody and Prof. R. Snipes, Giessen, Germany for critically reviewing the manuscript. The technical assistance of Karola Michael, Tamara Papadakis, and Martin Bodenbenner is gratefully acknowledged.
