Abstract
The role of endothelium in the progression of atheromasic disease has already been demonstrated. Endothelin-1 (ET-1) is released from endothelial cells during acute and chronic vascular damage and it appears to be the strongest vasoconstrictor agent known.
The aim of this study is to investigate the amount of endothelial damage in patients with unstable angina (UA), as defined by serum levels of ET-1, to verify a possible correlation with increased ischaemic damage by evaluation of serum N-terminal pro-brain natriuretic peptide (NT-proBNP) and interleukin 8 (IL-8) levels.
Serum levels of ET-1, IL-8 and NT-proBNP obtained from 10 patients affected by low-risk UA were compared to those belonging to eight healthy subjects. In order to compare the laboratory data pertaining to the two populations, a Student’s t-test and a Mann-Whitney U test were performed.
Levels of ET-1, IL-8 and NT-proBNP in samples of peripheral blood of patients affected by UA were significantly elevated, compared with those of the control group. The linear correlation analysis demonstrated a positive and significant correlation between levels of ET-1 and IL-8, between levels of ET-1 and NT-proBNP, and between levels of IL-8 and NT-proBNP in subjects affected by UA.
Early elevated levels of ET-1, IL-8 and NT-proBNP in patients with UA show a coexistence between ischaemic insults and endothelial damages. A positive and significant linear correlation between levels of ET-1 and IL-8, between levels of ET-1 and NT-proBNP, and between levels of IL-8 and NT-proBNP confirms that an increased ischaemic insult is correlated to inflammation signs and endothelium damage signs.
In patients with UA, ischaemia is always associated with a systemic immuno-mediated activity induced by acute endothelial damage. We suggest early administration of ET-1-selective receptor blockers and anti-inflammatory drugs.
Introduction
The role of the endothelium in the progression of atheromasic disease has already been widely demonstrated. However, the erosion, fissuration or rupture of a coronary plaque – with the subsequent thrombosis and formation of an unstable plaque – may play a fundamental role in the pathogenesis of the acute coronary syndrome (ACS).1–3
This mechanism has also been clearly demonstrated thanks to the results of many randomised trials that analysed recurrent use of anticoagulant and anti-platelet drugs in patients suffering from unstable angina (UA).
Not only does endothelium represent a mechanical barrier, but also an important tissue, with the ability to influence the inflammatory cascade responsible for plaque formation and instability.4–10
Endothelin 1 (ET-1) is released from endothelial cells 11 during acute and chronic vascular damage and it represents the strongest vasoconstrictor known.12,13 Its action is due to a particular linkage with specific receptors: Endothelin ETA and ETB receptors. 14 Many clinical trials have tried to define the clinical meaning of ET-1 serum levels in UA, but unfortunately their results were often inconclusive and controversial.15–17 Zuk-Popiolek et al. previously demonstrated that ET-1 blood concentration in the coronary sinus and peripheral vessels is elevated during UA. 18 A possible mechanism involving ET-1 could be based on an autocrine and paracrine response. In fact ET-1 provide a steroid and mineral-corticoid secreting action via ETA and ETB receptors expressed in adrenal glands and endocrine components of the gonads.19–21 Further, the activation of the ETB receptor mediates the secretagogue effect of ET-1 on aldosterone. 22 Moreover it was previously demonstrated that aldosterone stimulates angiotensin II Type-1 (AT1) receptor and cause vasoconstriction by increasing oxidative stress in coronary microcirculation. 23
Not only does interleukin 8 (IL-8) represent an important pro-inflammatory cytokine, but also an important α-chemokine. It is released from many inflammatory cells like lymphocytes, monocytes, mast-cells and it seems that IL-8 is stored inside endothelial cells.24–26
IL-8 enhances the adhesion and migration of granulocytes and monocytes from the vascular lumen to the inflammation site. High levels of IL-8 precede the IL-6 peak in acute myocardial infarction, but there are many evidences regarding its poor specificity as a risk factor during ACS. 27
The N-terminal pro-brain natriuretic peptide (NT-proBNP) is a 76 amino acid-molecule which is co-secreted in equimolar entity with the brain natriuretic peptide (BNP) during hypoxia and/or ventricular muscular cells stretching. 28 There is strong evidence about its role as a novel marker of acute myocardial ischaemia during UA.29–33 High levels of NT-proBNP represent a strong risk factor for re-infarction or death for cardiovascular causes in patients with myocardial infarction. 34 Because of its relatively poor specificity and high sensitivity, the use of this marker only represents a support to diagnose an acute myocardial ischaemia.
Literature suggests that its use be implemented with clinical, laboratory and anamnestic data. 35
The aim of the present study is to investigate if endothelial damage (indicated by ET-1 serum levels) during UA promotes an early inflammatory reaction (indicated by IL-8 serum levels) and a subsequent early evolution to myocardial ischaemia denoted by NT-proBNP levels.
Materials and methods
We studied 10 patients (6 men, 4 women; age range, 40–61 years) with a diagnosis of low-risk UA, in accordance with AHA/ACC criteria and ESC guidelines for UA and non-ST elevation myocardial infarction (NSTEMI), 36 and eight healthy subjects (5 men, 3 women; age range, 38–60 years) as control subjects. All enrolled patients signed an informed consent form as a standard procedure, as stated by the Helsinki declaration. This study was approved by the local ethical committee.
Patients underwent serial quantitation of serum cardiac Troponin I (cTnI), creatine kinase-MB (CK-MB) and myoglobin (Myo) levels immediately after their Emergency Department (ED) acceptance, and the laboratory exams were repeated after 6 and 12 h.
A standard 12-leads electrocardiogram (EKG) was performed in all patients under investigation. Low-risk UA patients with two consecutive samples negative for myocardial enzymes level elevation underwent a treadmill stress test according to the Bruce protocol. Only patients whose stress test had a positive outcome were included in the study reference group and a coronary angiography was performed on them. Ethical principles would not allow us to make the control group undergo invasive procedures. Statin-treated patients were excluded from the study to avoid the positive pleiotropic effects of the statins on endothelial cells.
Blood samples were collected from a brachial vein immediately after admission in the ED in a mean time of 60 min from the beginning of chest pain, in order to detect the levels of ET-1, IL-8 and NT-proBNP in the serum. Said serum was prepared by allowing the blood to clot, followed by centrifugation at 2000 rpm for 10 min.
The serum was stored at −80°C after a mean time of 25 min from the collection of the sample. Serum ET-1 concentration was measured by an enzyme-linked immunosorbent assay (ELISA) test using specific antibodies of a commercial kit constituted by reagents and 96- microplates aligned in strips (Assay Designs, Inc., Ann Arbor, MI, USA).
The procedure for ET-1 detection by ELISA required these steps:
dilute assay buffer and wash buffer with deionised water
prepare ET-1 standards at different concentrations: 100, 50, 25, 12.5, 6.25, 3.125, 1.563 and 0.781 pg/mL
prepare the ET-1 antibody (Ab) by diluting 10 µL of the supplied Ab concentrate with 1 mL of Ab diluent for every mL of 1X needed
remove excess microplates strips
100 µL of assay buffer was added into the well
100 µL of the ET-1 standards (from number 1 to number 8 dilutions) and 100 µL of the serum samples were added respectively to each well. The microplates were covered and incubated for 1 h at room temperature
at the end of incubation the microplates were empty, washed with wash buffer and added 100 µL of diluted antibody to ET-1 into each well except the blank. The microplates were covered and incubated for 30 min at room temperature
at the end of incubation the microplates were empty, washed to remove excess HRP labelled antibody
100 µL of TMB substrate solution was added to each well and incubated for 30 min
100 µL of stop solution was added to each well to stop the substrate reaction
the solution was read at 450 nm (procedure from Assay Designs, Inc., Ann Arbor, MI, USA).
Serum IL-8 levels were measured using a ‘capture and competitive immunoassay kit’ (Assay Designs, Inc., Ann Arbor, MI, USA). Serum NT-proBNP levels were obtained via electrochemical lighting procedures (Elecsys 2010 Roche Diagnostics, Risch, Switzerland).
Statistical analysis
A statistical descriptive analysis was performed in order to obtain results expressed as median values and ranges. Because of reduced numbers of samples for each variable and group, a Kolmogorov-Smirnov one sample data test was performed in order to confirm or not the presence of values. Fisher’s exact test was already applied as a comparative test for the analysis of nominal variables because of the low number of data. Analysed parameters were compared using Student’s t-test for unpaired data and the matching was confirmed using a Mann-Whitney non-parametrical U test.
Intra-group correlations were computed using Pearson and Spearman test for data correlations accepting a P value <0.05 as significant.
Results
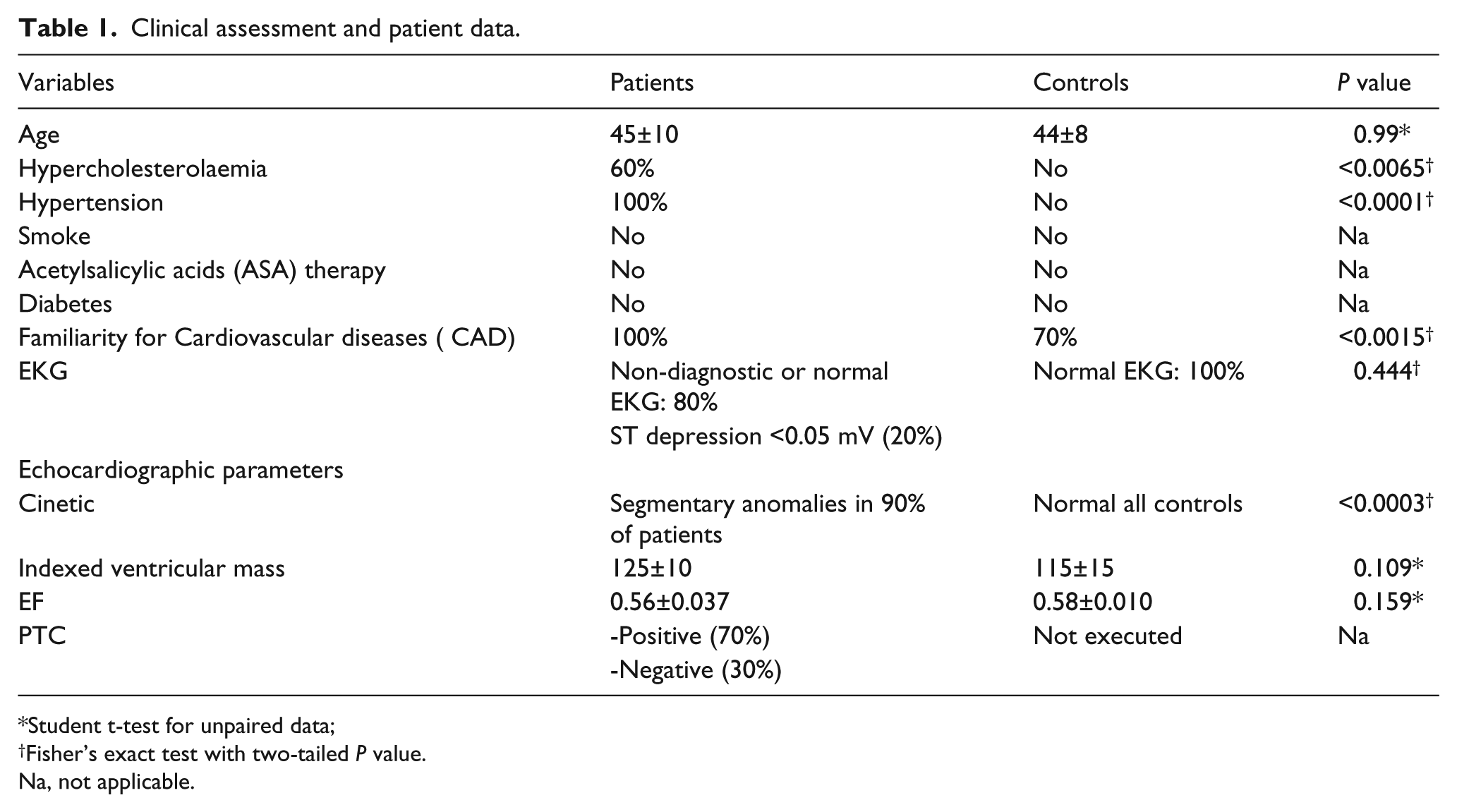
Most UA patients suffered from hypercholesterolemia and hypertension as well.
Patients, who were referred to our clinic as outpatients, were included in the study if they were not being treated with statins. Table 1 represents the clinical, EKG, echocardiography and coronarography findings. No significant difference in age was observed between UA patients and controls. A significantly statistical difference was observed in the presence of hypertension and hypercholesterolaemia in UA patients in comparison with healthy patients (Table 1). Normal or non-diagnostic aspecific EKG abnormalities were observed in 80% of UA patients and ST segment depressions, as a marker of positive EKG abnormalities was observed in only 20% of patients (no statistically significant differences via Fisher’s exact test). Kinetic echocardiographic abnormalities were only observed in UA patients (P <0.0001 according to Chi-square analysis) with non-statistical differences in ejection fraction (EF) performance and left ventricle (LV) index-mass.
Clinical assessment and patient data.
Student t-test for unpaired data;
Fisher’s exact test with two-tailed P value.
Na, not applicable.
Because of the ethical protocol applied, percutaneous transluminal coronary (PTC) was not performed in healthy subjects.
Severe coronary artery obstruction was observed in 70% of UA patients (no distinction was made between patients with disease affecting one, two, three vessels or even the left main artery) while 30% of UA patient demonstrated no presence of coronary obstructive lesions (Table 1).
There are statistically significant differences between ET-1 peripheral blood mean levels in patients affected by UA compared with the control group. Moreover IL-8 and NT-proBNP mean values in UA patients are significantly elevated in comparison to controls (Table 2).
Statistic descriptive analysis of ET-1, IL-8 and N-ProBNP.
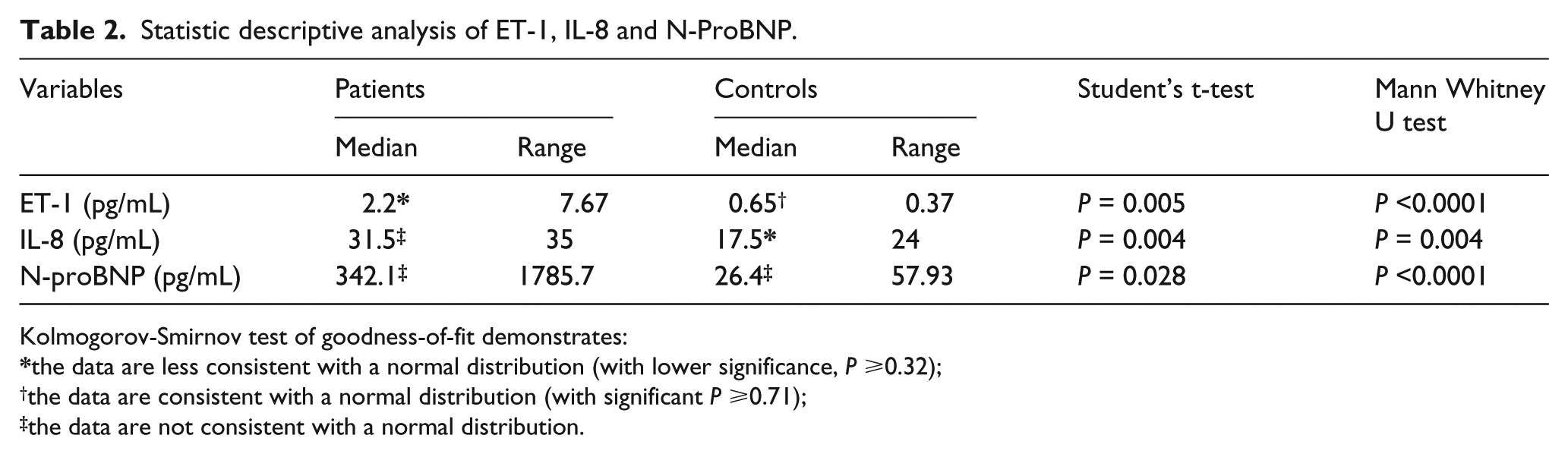
Kolmogorov-Smirnov test of goodness-of-fit demonstrates:
the data are less consistent with a normal distribution (with lower significance, P ⩾0.32);
the data are consistent with a normal distribution (with significant P ⩾0.71);
the data are not consistent with a normal distribution.
The linear correlation analysis via Spearman and Pearson methods demonstrates a significantly positive statistical correlation between ET-1 and IL-8, between ET-1 and NT-proBNP, and between IL-8 and NT-proBNP (P <0.001) (Figure 1).

Linear correlation between ET1, IL8 and NproBNP in patients affected by unstable Angina (A) and controls (C).
Discussion
Many authors have studied inflammation during ACS in order to stratify the risk for patients during the evolution of the cardiac disease. In order to identify patients with ACS with a higher risk of death or cardiovascular major events, many inflammatory markers have been proposed as risk stratification factors. However, at the moment, none of them has been proved to show the sensitivity and specificity needed to become a credible biochemical stratification marker.
The activity level of the inflammation cascade and the entity of ischaemic cardiac damage have still not been associated with a laboratory sign because of the difficulty to introduce an ischaemic biochemical marker with good statistical sensitivity and specificity.
NT-proBNP was recently proposed as a sensible marker of ischaemia in patients without volume overload cardiac diseases (heart failure and pulmonary hypertension). 37 This approach justifies the choice of UA patients with a low risk.
An early elevation of IL-8 and ET-1 serum levels in UA patients demonstrates, if compared to controls, both an acute endothelial damage and an early inflammatory cascade activation. NT-proBNP mean values elevation represents a clear confirm of ischaemia during ACS as previously demonstrated. 38
Significantly positive statistical linear correlations between serum levels of ET-1 and IL-8, between serum levels of ET-1 and NT-proBNP, and between serum levels of IL-8 and NT-proBNP demonstrate that endothelial damage entity, inflammatory cytokine levels and ischaemia are closely correlated in UA patients.
Interestingly, this correlation also suggests that inflammation, cardiac ischaemia and endothelial reactivity all descend from a common primum movens: the endothelium.
Øynebråten et al. recently demonstrated that IL-8 is especially stored inside endothelial cells and there are strong evidences that ET-1 promotes in vitro releasing of IL-8 from endothelial cells via autocrine and paracrine secretion. 39
Possible arteriolar spasm and microcirculation involvement in the district of partially occluded epicardial arteries confirm the presence of two negative angiographic results. These can be explained with the hypothetical vasoconstricting action of ET-1 on microvessels.
Finally, the authors suggest the hypothetical implementation of data from multicentre studies to confirm this comprehensive global vision of the entire cardiac ischaemic phenomenon during acute coronary syndrome (ACS).
In order to introduce a possible therapy in the future, the authors suggest the use of selective endothelin 1 type A receptor blockers in the early phase of UA.
Footnotes
Acknowledgements
The authors wish to thank Dr. Antonio Giovinazzi for his careful English revision of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
