Abstract
Cholinergic neurons in the dorsal motor nucleus of the vagus (DMNV) are particularly vulnerable to laryngeal nerve damage, possibly because they lack fibroblast growth factor-1 (FGF1). To test this hypothesis, we investigated the localization of FGF1 in cholinergic neurons innervating the rat larynx by immunohistochemistry using central-type antibodies to choline acetyltransferase (cChAT) and peripheral type (pChAT) antibodies, as well as tracer experiments. In the DMNV, only 9% of cChAT-positive neurons contained FGF1, and 71% of FGF1-positive neurons colocalized with cChAT. In the nucleus ambiguus, 100% of cChAT-positive neurons were FGF1 positive. In the intralaryngeal ganglia, all ganglionic neurons contained both pChAT and FGF1. In the nodose ganglia, 66% of pChAT-positive neurons were also positive for FGF1, and 90% of FGF1-positive ganglionic cells displayed pChAT immunoreactivity. Neuronal tracing using cholera toxin B subunit (CTb) demonstrated that cholinergic neurons sending their axons from the DMNV and nucleus ambiguus to the superior laryngeal nerve were FGF1 negative and FGF1 positive, respectively. In the nodose ganglia, some FGF1-positive cells were labeled with CTb. The results indicate that for innervation of the rat larynx, FGF1 is localized to motor neurons, postganglionic parasympathetic neurons, and sensory neurons, but expression is very low in preganglionic parasympathetic cholinergic neurons.
Keywords
F
The trophic effect of FGF1 on cholinergic neurons has been shown in various physiological studies. Administration of FGF1 reduces degeneration of nucleus basalis magnocellularis cholinergic neurons after cortical ablation (Figueiredo et al. 1993) and reduces degeneration of septal cholinergic neurons in aging (Sasaki et al. 1999). FGF1 also prevents cell death in motor neurons after axonal injury (Cuevas et al. 1995; Laird et al. 1995; Jacques et al. 1999). Pathological studies have shown that loss of FGF1 precedes death of motor neurons in patients with amyotrophic lateral sclerosis (Kage et al. 2001). Cholinergic neurons in the oculomotor nucleus expressing high levels of FGF1 (Elde et al. 1991) are relatively spared in amyotrophic lateral sclerosis. These data suggest that FGF1 plays a trophic role in cholinergic neurons and that cholinergic neurons lacking FGF1 may be more severely affected by injury or disease.
The laryngeal nerve recovers poorly after injury. For reasons that remain unclear, cholinergic neurons in the dorsal motor nucleus of the vagus (DMNV) are particularly vulnerable to laryngeal nerve damage (Lewis et al. 1972; Navaratnam et al. 1998). We and other authors have demonstrated that cholinergic neurons in the DMNV express relatively low levels of FGF1 (Elde et al. 1991; Stock et al. 1992; Toyoda et al. 2006). FGF1 is believed to be released by cellular injury and to exert a trophic effect on damaged neurons (Eckenstein et al. 1991; Ishikawa et al. 1992). Therefore, lack of FGF1 in cholinergic neurons sending their axons to the larynx may underlie poor recovery of the laryngeal nerve after axonal damage. To test this hypothesis, we used immunohistochemistry (IHC) and neuronal tracer experiments to investigate the localization of FGF1 in cholinergic neurons in the DMNV, nucleus ambiguus, nodose ganglion, and intralaryngeal ganglia.
Materials and Methods
Tissue Preparation for IHC
This study was performed in accordance with the Public Health Service Policy on Humane Care and Use of Laboratory Animals, the NIH Guide for the Care and Use of Laboratory Animals, and the Animal Welfare Act (7 U.S.C. et seq.). The animal use protocol was approved by the Institutional Animal Care and Use Committee (IACUC) of the Shiga University of Medical Science. Five male Wistar rats weighing 200 to 250 g were used for IHC. Under deep anesthesia with sodium pentobarbital (80 mg/kg), animals were transcardially perfused with 10 mM PBS followed by ice-cold fixative consisting of 4% paraformaldehyde (PFA) in 0.1 M phosphate buffer (PB; pH 7.4). The medulla oblongata, nodose ganglia, and larynx were then removed from each animal. Specimens were post-fixed for 24 hr in the same fixative as used in perfusion and for cryoprotection were then immersed for 24 hr in 0.1 M PB containing 15% sucrose and 0.1% sodium azide. A cryostat was used to cut the larynx and medulla oblongata into 20-μm sections, and the nodose ganglion was cut into 12-μm sections. Laryngeal sections were placed directly on gelatinchrome-coated glass slides, while the other sections remained free floating for processing. Prior to staining, these sections were kept for at least 4 days at 4C in 0.1 M PBS containing 0.3% Triton X-100 (PBST; pH 7.4) to increase the penetration of antibodies into the tissues.
FGF1 monoclonal antibody was raised against human recombinant FGF1 and reacted with both human and rat FGF1 (Imamura et al. 1994). In this study, specificity of the FGF1 monoclonal antibody was examined using Western blot analysis and an immunoabsorption test.
Western Blot Analysis and Immunoabsorption Test
For Western blots, two male Wistar rats weighing 250 g were perfused with 10 mM PBS, pH 7.4, under deep anesthesia with sodium pentobarbital (80 mg/kg). The medulla oblongata was dissected out and homogenized in five volumes of ice-cold 50 mM Tris-HCl (pH 7.4) containing 0.5% Triton X-100 and Complete Mini protease inhibitor mixture tablets (1 tablet/10 ml; Roche Diagnostics, Mannheim, Germany). The homogenates were centrifuged at 12,000 × g for 20 min at 4C, and the supernatants were collected as crude protein fractions. Protein concentration was assayed using the method of Lowry et al. (1951). Fifty μg of the crude extracted protein, 50 ng of human recombinant FGF1 (140 amino acid form, 15.8 kDa; Wako Pure Chemicals, osaka, Japan), and prestained Precision protein standards (Bio-Rad; Hercules, CA) were electrophoresed on a 15% SDS-PAGE gel under reducing conditions and then transferred to a polyvinylidene difluoride membrane (Immobilon-P; Millipore, Tokyo, Japan). The membrane was incubated for 1 hr with 5% skim milk powder in 25 mM Tris-buffered saline (TBS; pH 7.4) at room temperature and further incubated overnight with the monoclonal antibody against FGF1 (1 μg/ml) in 25 mM TBS containing 1% skim milk powder at room temperature. After washing with 25 mM TBS containing 0.1% Tween-20 (Bio-Rad), the membrane was incubated for 2 hr with a peroxidase-coupled F(ab')2 anti-mouse IgG (diluted 1:50; Histofine, Nichirei Corp., Tokyo, Japan). Peroxidase labeling was detected by chemiluminescence using the SuperSignal West Pico Chemiluminescent Substrate (Pierce; Rockford, IL).
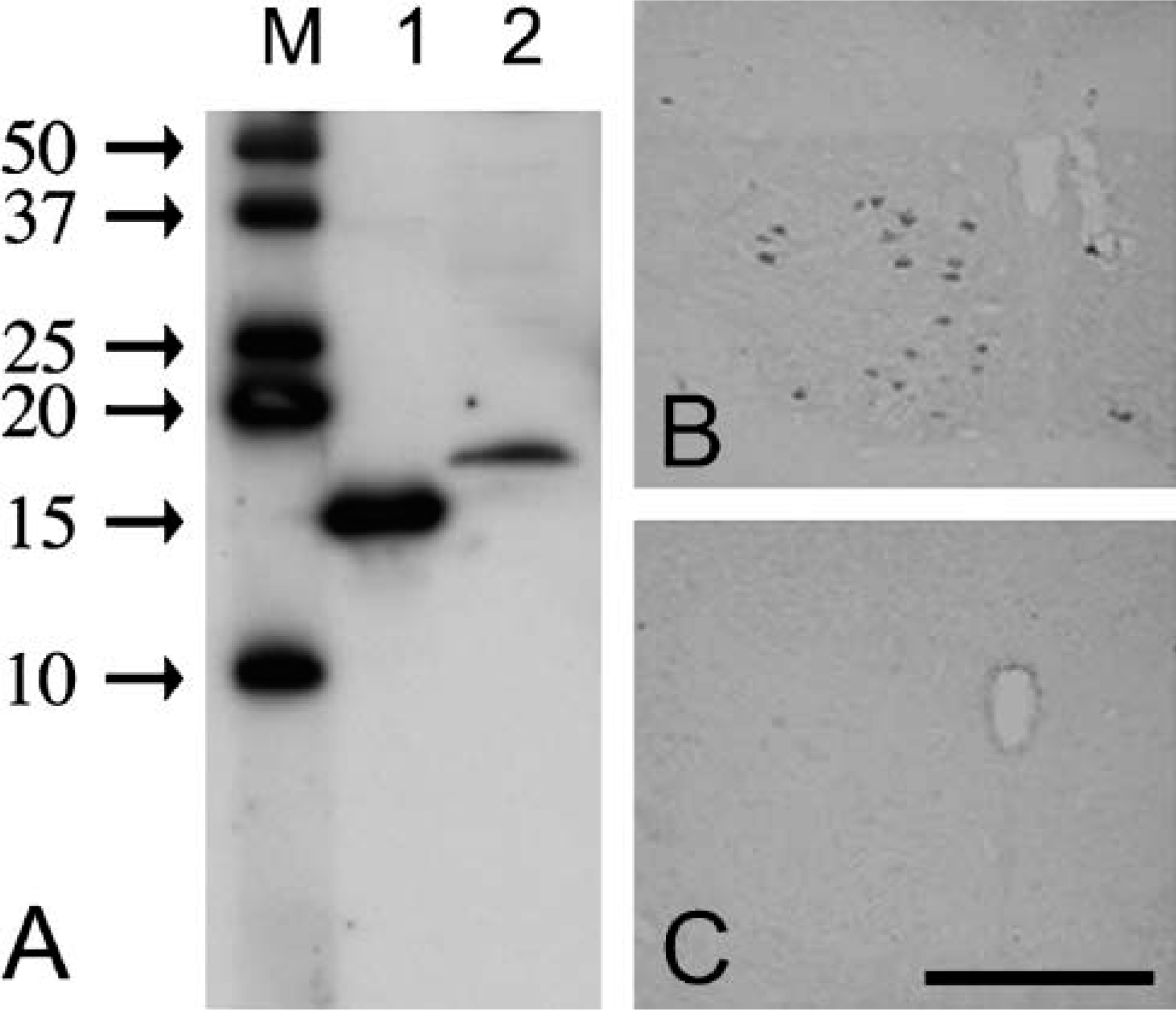
Western blot analysis using the mouse anti-FGF1 antibody (
For the immunoabsorption test, the FGF1 antibody at the same dilution as used for IHC was preincubated overnight at 4C in 0.5 ml of PBST with or without 10 μ/ml of the human recombinant FGF1. To remove the FGF1/IgG complex, 0.5 ml of heparin-Sepharose beads (Amersham Pharmacia Biosciences; Piscataway, NJ) was added to the solutions. After incubating for 1 hr at room temperature, each suspension was centrifuged for 20 min at 15,000 × g at 4C. Supernatants were collected and used to immunostain the sections as described below.
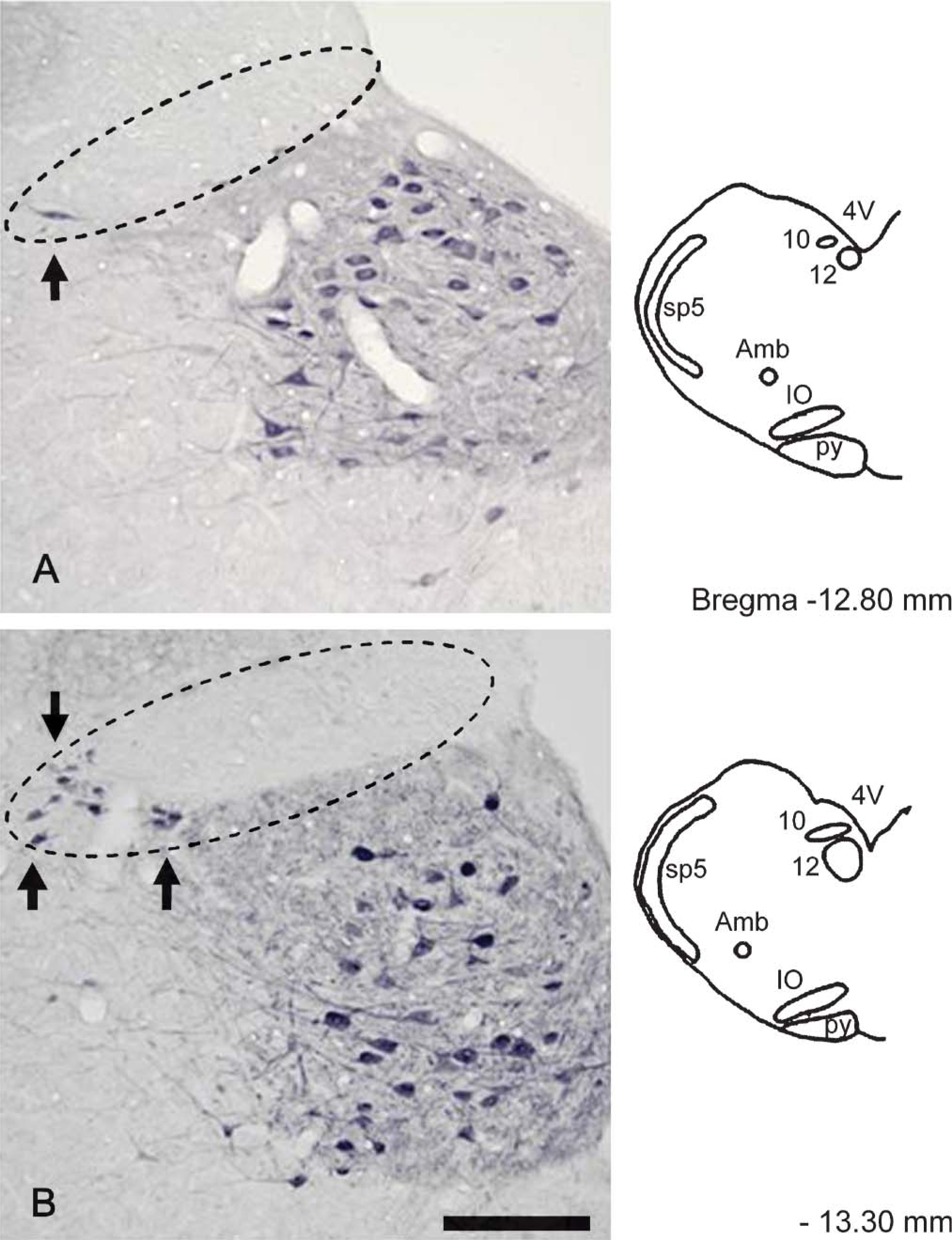
Fibroblast growth factor-1 (FGF1) immunoreactivity in the dorsal motor nucleus of the vagus (DMNV) at-12.80 mm (
FGF1 IHC
Prior to staining, endogenou peroxidase was eliminated by incubating the sections in PBST containing 0.5% hydrogen peroxide for 30 min at room temperature. After several washes with PBST, sections were incubated for 2 days at 4C with the mouse monoclonal antibody to FGF1 diluted to 1 μg/ml. Following incubation with the primary antibody, sections were incubated with biotinylated anti-mouse IgG (diluted 1:1000; Vector Laboratories, Burlingame, CA) for 1 hr at room temperature and with avidin-biotinylated peroxidase complex (diluted 1:4000; Vector Laboratories) for 1 hr at room temperature. PBST was used for diluting the immunohistochemicals and for washing the sections between steps. A purple color was developed with 0.02% 3,3′diaminobenzidine, 0.3% nickel ammonium sulfate, and 0.0045% hydrogen peroxide in 50 mM Tris-HCl buffer (pH 7.6). Free-floating sections were mounted on gelatin-chrome-coated glass slides and air dried.
Double Immunofluorescence for FGF1 and Choline Acetyltransferase
To visualize cholinergic neurons, we used antibodies against choline acetyltransferase (ChAT) of central type (cChAT) or peripheral type (pChAT) (Tooyama and Kimura 2000). Most cChAT antibodies do not label peripheral cholinergic neurons, but recent studies have shown the usefulness of pChAT antibody for labeling them (Nakanishi et al. 1999; Nakajima et al. 2000; Chiocchetti et al. 2003; Yasuhara et al. 2003; Brehmer et al. 2004; Chiocchetti et al. 2004; Yasuhara et al. 2004; okano et al. 2005; Yuan et al. 2005).
For simultaneous visualization of FGF1 and ChAT, a double-immunofluorescence method was used. Sections were incubated in primary antibodies for 2 days at 4C with a mixture of the mouse anti-FGF1 antibody (1 μg/ml) and goat anti-cChAT antibody (AB-144p, diluted 1:1000; Chemicon, Temecula, CA) or rabbit anti-pChAT antibody (diluted 1:10,000) (Tooyama and Kimura, 2000). Sections were then incubated for 4 hr at room temperature in a mixture of Alexa 488-conjugated anti-mouse IgG (1:500; Molecular Probes, Eugene, OR) and Alexa 594-conjugated anti-goat IgG or anti-rabbit IgG (1:500; Molecular Probes). PBST was used to dilute the antibodies and wash the sections between steps. Free-floating sections were mounted on gelatin-coated glass slides and examined under a confocal laser-scanning microscope (Bio-Rad).
Quantification of Labeled Neurons
Four male Wistar rats were used for quantitative analysis of FGF1-positive and/or ChAT-positive neurons. The brain stem from each rat was cut into serial 20-μm sections using a cryostat. For the DMNV, 4 sections from each rat (total 16 sections) were selected at levels −12.80 mm, −13.30 mm, −13.80 mm, and −14.30 mm from the Bregma. After double immunostaining, these sections were mounted on gelatincoated glass slides and examined under the confocal laser-scanning microscope. The numbers of FGF1-positive and/or cChAT-positive cells in the DMNV were counted by eye on saved images. For the nucleus ambiguus, we used a total of 12 sections at levels −12.80 mm, −13.30 mm, and −13.80 mm from the Bregma. Nodose ganglia from four rats were cut into 20-μm sections using a cryostat. Four sections from each rat (total 16 sections) were used for quantification as above. For the intralaryngeal ganglia, we analyzed 15 ganglia in sections obtained from four rats. In all regions, only neurons with visible nuclei were counted.
Cholera Toxin B Injection
Use of cholera toxin B subunit (CTb) as a neuronal tracer was based on the methods of Hisa et al. (1991). Under anesthesia with sodium pentobarbital (40 mg/kg), 2 μl of CTb (List Biological Laboratories; Campbell, CA) was injected using a Hamilton syringe, over a period of 1 min, into the right superior laryngeal nerve in three rats. After injection, the syringe was kept in the nerve for one additional minute. After a survival period of 5 days, the animals were deeply anesthetized and perfused with 10 mM PBS followed by ice-cold fixative consisting of 4% PFA in 0.1 M PB (pH 7.4). The medulla oblongata and nodose ganglia were removed and processed for IHC.
Double Immunofluorescence for FGF1 and CTb
For simultaneous visualization of FGF1 and CTb, we employed a double-immunofluorescence method using the mouse anti-FGF1 antibody and goat anti-choleragenoid antibody. Sections were incubated for 3 days at 4C with a mixture of the mouse anti-FGF1 antibody (1 μg/ml) and goat anti-choleragenoid antibody (diluted 1:100,000; List Biological Laboratories). After washing with PBST several times, sections were incubated for 4 hr at room temperature with a mixture of Alexa 488-conjugated anti-mouse IgG (1:500; Molecular Probes) and Alexa 594-conjugated anti-goat IgG (1:500; Molecular Probes). After washing with PBST, free-floating sections were mounted on gelatin-chrome-coated glass slides and examined under the confocal laser-scanning microscope.
Results
Characterization of FGF1 Antibody
Western blot analysis showed that the monoclonal anti-FGF1 antibody (Figure 1A) detected a 15.8-kDa band representing the recombinant FGF1 (Figure 1A, Lane 1) and a single band with a molecular mass of ~16.5 kDa in the rat medulla oblongata (Figure 1A, Lane 2). In agreement with a previous report (Stock et al. 1992), the antibody clearly labeled the cytoplasm of some neurons (Figure 1B). This labeling was abolished using the antibody preabsorbed with 10 μg/ml of FGF1 (Figure 1C).
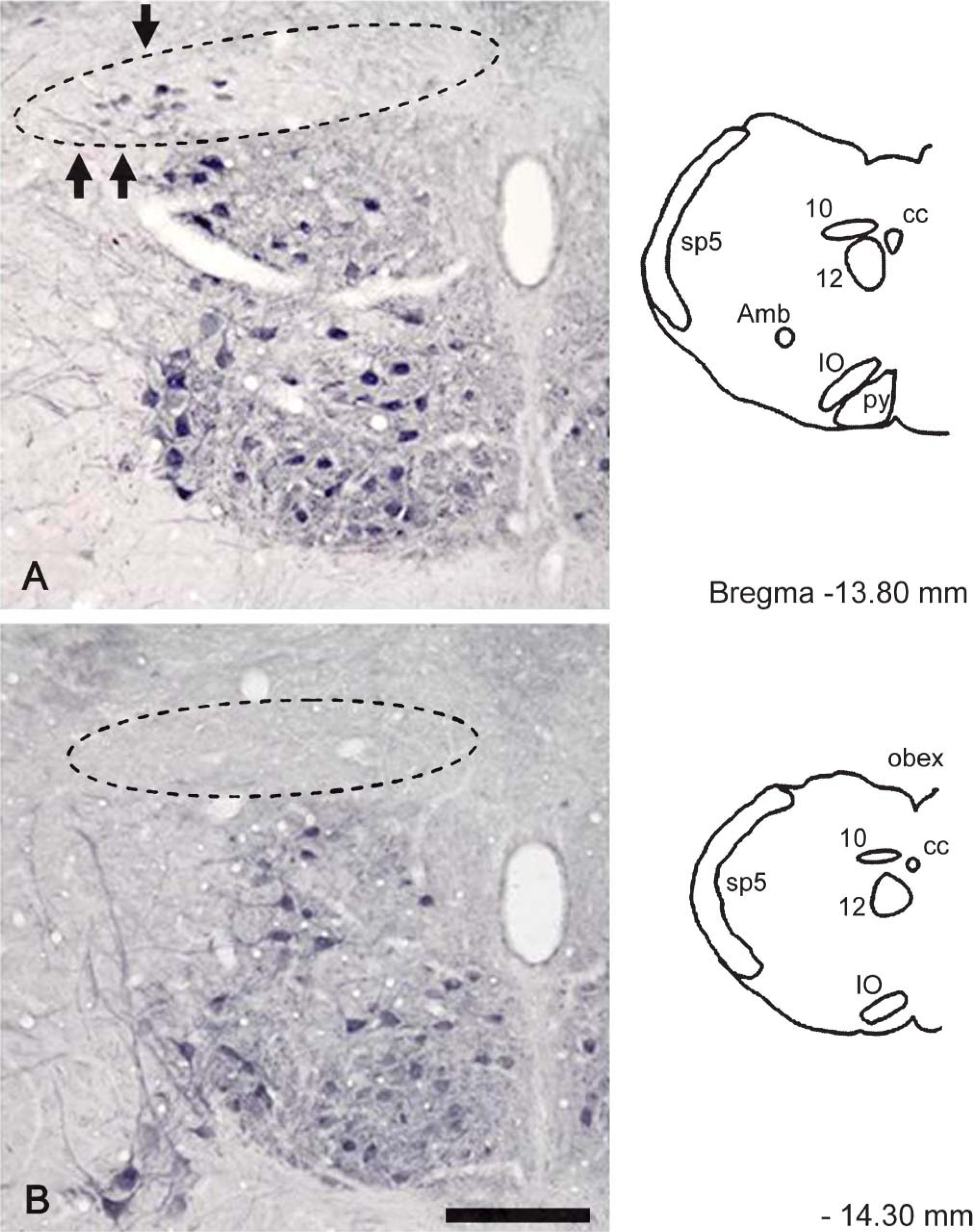
FGF1 immunoreactivity in the DMNV at −13.80 mm (
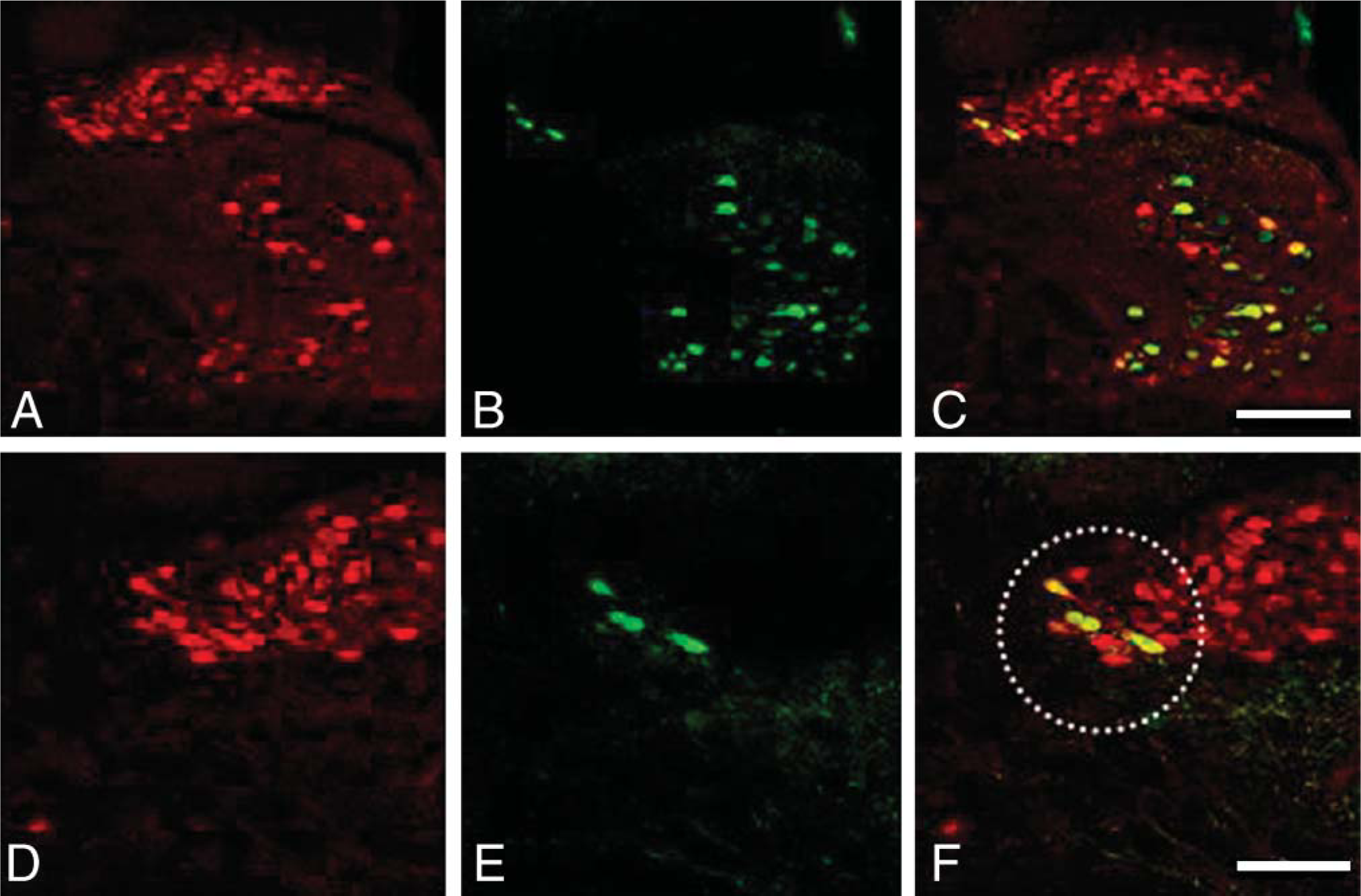
Double immunostaining for central-type antibodies to choline acetyltransferase (cChAT) (red) and FGF1 (green) in the DMNV and hypoglossal nucleus. (
Localization of FGF1 in Cholinergic Neurons in the DMNV and Nucleus Ambiguus
Figure 2 and Figure 3 demonstrate the distribution of FGF1-positive neurons in the rat DMNV. At −12.80 mm from the Bregma, a few FGF1-positive neurons were seen in the lateral part of the DMNV (Figure 2A). At the −13.30 mm and −13.80 mm levels, some FGF1-positive neurons were distributed in the lateral part of the DMNV (Figure 2B and Figure 3A). No positive neurons were seen at the −14.30 mm level. Few positive neurons were seen in the medial part of the DMNV at any level.
Figure 4 shows a typical example of double immunostaining for FGF1 (green) and cChAT (red) at −13.30 mm from the Bregma. As indicated above, a small number of cChAT neurons in the lateral part of the DMNV were positive for FGF1 (Figure 4F), and a large number of cChAT-positive neurons were negative for FGF1 (Figure 4). Quantitatively, only 9% of cChAT-positive neurons were positive for FGF1 in the DMNV, and 71% of FGF1-positive neurons were positive for cChAT (Table 1).
Number of neurons doubly stained for ChAT and FGF1 as percentages of total ChAT-positive neurons (upper) and total FGF1-positive neurons (lower)

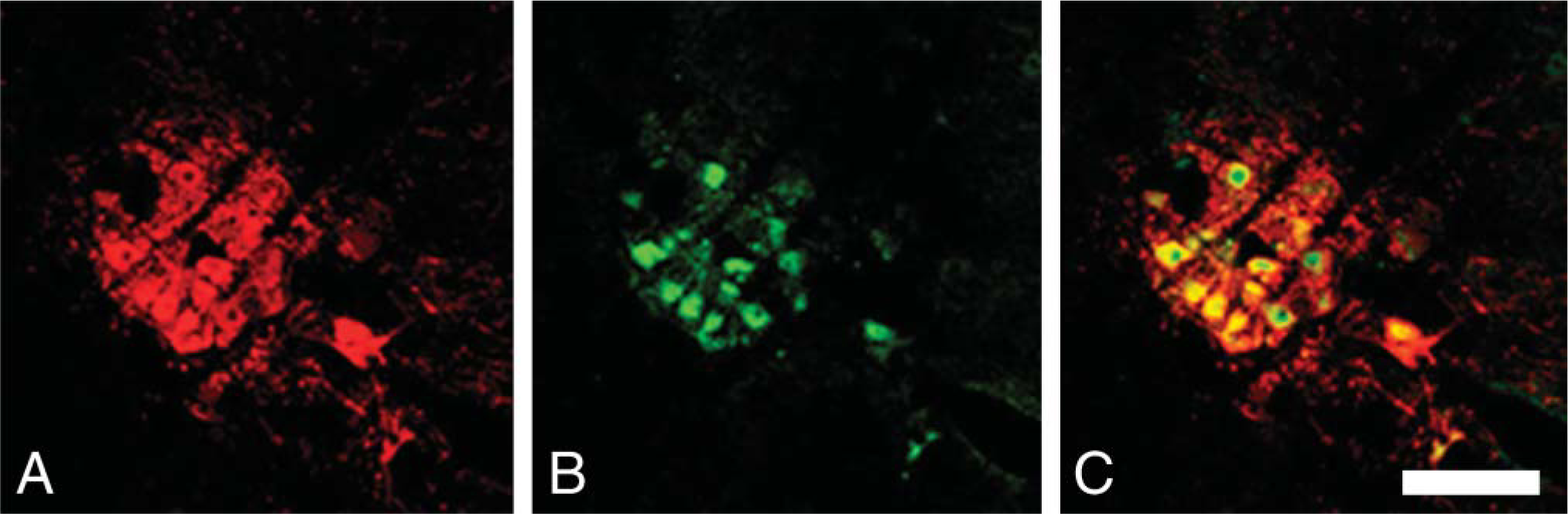
Double immunostaining for cChAT (red) and FGF1 (green) in the nucleus ambiguus. (
In the nucleus ambiguus, all cChAT-positive neurons also contained FGF1 (Figures 5A-5C; Table 1).
Localization of FGF1 in the Intralaryngeal and Nodose Ganglia
In the larynx, all ganglionic cells in the intralaryngeal ganglia were clearly labeled by pChAT (Figure 6A). All pChAT-positive ganglionic cells also contained FGF1 (Figures 6B and 6C; Table 1). Some of the neurons in the nodose ganglion were intensely labeled by pChAT antibody (Figure 7B). Many of the pChAT-positive neurons in the nodose ganglion also contained FGF1-immunoreactivity (Figures 7A and 7C). Quantitatively, 66% of pChAT-positive ganglionic cells contained FGF1, and 90% of FGF1-positive neurons were also positive for pChAT (Table 1).
Neuronal Tracing Experiments
Tracing experiments with CTb were carried out to determine whether the cholinergic neurons in the DMNV and nucleus ambiguus that send their axons to the larynx contain FGF1. Figure 8 shows a typical example of double immunostaining for FGF1 (green) and CTb (red). In the DMNV, CTb-labeled neurons did not contain FGF1 (Figures 8A-8C). In the nucleus ambiguus, all CTb-labeled neurons were also positive for FGF1 (Figures 8D-8F). Some FGF1-positive neurons in the nodose ganglion were also labeled with CTb (Figures 8G-8I).
Discussion
Characterization of FGF1 Antibody
When examined by Western blot analysis, the FGF1 monoclonal antibody recognized the 15.8-kDa human recombinant FGF1 (140 amino acid form) (Gimenez-Gallego et al. 1986). In the rat medulla oblongata homogenate, the antibody detected a single band with a molecular mass of 16.5 kDa corresponding to the molecular mass of a native form of FGF1 (Thomas et al. 1984; Elde et al. 1991; Tooyama et al. 1991). Although the 140 amino acid form of FGF1 was first purified from bovine brain (Gimenez-Gallego et al. 1986), a native form of FGF1 is believed to have an extended N-terminal (Jaye et al. 1986).
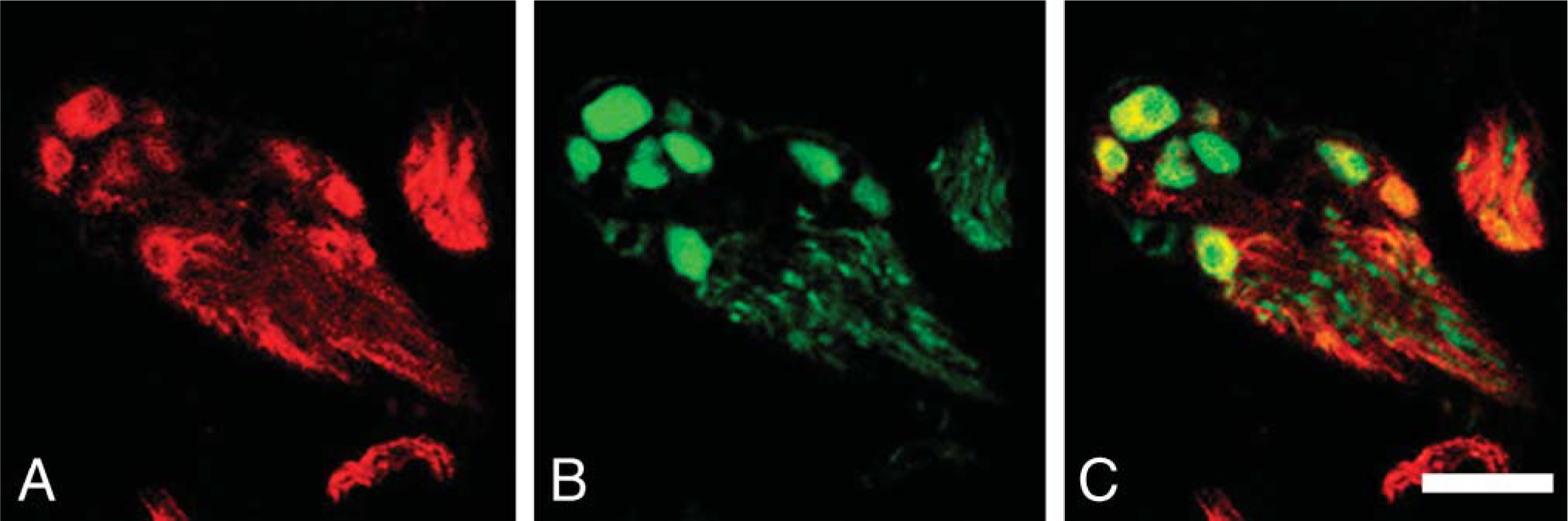
Double immunostaining for peripheral type (pChAT) antibodies (red) and FGF1 (green) in the intralaryngeal ganglion. (
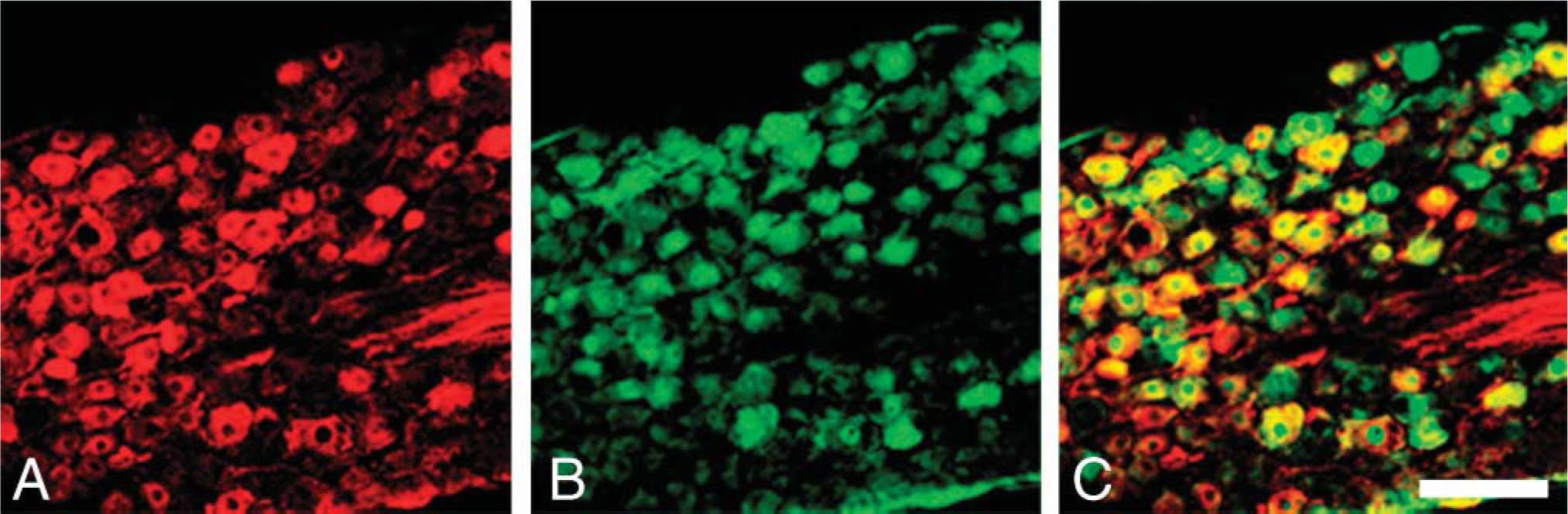
Double immunostaining for pChAT (red) and FGF1 (green) in the nodose ganglion. (
For the immunoabsorption test, we used heparin-Sepharose beads to remove the complex of FGF1 and FGF1 antibody because FGF1 in the solution often reacts with heparan sulfate in tissues. Labeling was abolished using the antibody preabsorbed with 10 μg/ml of FGF1. These results indicate that the monoclonal anti-FGF1 antibody specifically labels FGF1 in rat tissues.
We examined the effects of fixation on FGF1 labeling. FGF1 labeling was greatly reduced by using a fixative containing glutaraldehyde (data not shown). However, there was no apparent difference in FGF1 labeling between 1 and 3 days fixation in 4% PFA, with the exception that the intensity of FGF1 in cell bodies increased with the shorter fixation times. Although neuronal cell bodies, proximal processes, and some thick bundles were clearly labeled by the FGF1 antibody, labeled nerve terminals were not observed under any of the fixation conditions. The reasons for the absence of labeled terminals remain unclear, but we cannot rule out the presence of FGF1 in terminals at levels below the sensitivity of the immunocytochemical protocol. However, previous studies have also shown that FGF1 immunoreactivity is predominant in the cytoplasm of cell bodies (Stock et al. 1992; Kage et al. 2001). Some isoforms of FGF1 receptors exist in the perinuclear region of the cell body (Prudovsky et al. 1996). In addition, FGF1 contains nuclear localization signals and plays a role in nuclear functioning (Imamura et al. 1990; Wesche et al. 2005). The predominance of FGF1 in cell bodies may therefore result from interaction with these receptors in the perinuclear region.
Co-localization of FGF1 in Cholinergic Neurons
The present study demonstrated localization of FGF1 in cholinergic neurons in the DMNV, nucleus ambiguus, and nodose ganglion as well as in the intralaryngeal ganglia. Extent of colocalization of FGF1 with ChAT varied among regions. In the nucleus ambiguus, all cholinergic neurons expressed FGF1. This is in agreement with previous findings of high levels of FGF1 expression in motor neurons of rat cranial nuclei including the nucleus ambiguus (Elde et al. 1991; Stock et al. 1992). In human spinal cord, 96% of ChAT-positive motor neurons are also positive for FGF1 (Kage et al. 2001). These results indicate that FGF1 plays an important role in motor neurons.
In contrast with motor neurons, only 9% of cholinergic neurons in the DMNV were immunoreactive for FGF1. These FGF1-positive neurons were distributed mainly in the lateral part of the DMNV. Tracer experiments demonstrated that CTb-positive neurons in the DMNV were a separate population from the FGF1-positive neurons. These results indicate that preganglionic cholinergic neurons sending their axons to the larynx do not contain FGF1. The findings are consistent with a previous study showing that the lateral part of the DMNV contains many preganglionic cells sending their fibers to the ileum and colon (Satomi et al. 1978).
Low expression of FGF1 in preganglionic cells in the DMNV suggests that FGF1 may not be involved in the physiological functioning of parasympathetic preganglionic neurons. However, when the vagal nerve is injured, administration of FGF1 into the injured axon enhances the survival of neurons in the DMNV (Jacques et al. 1999). A previous in situ hybridization study demonstrated that neurons in both the DMNV and nucleus ambiguus expressed high levels of FGF receptor 1 (FGFR1) (Wanaka et al. 1990). Together these findings suggest that FGF1 supplementation may be useful for treatment of vagal nerve injury, via action on FGFR1.
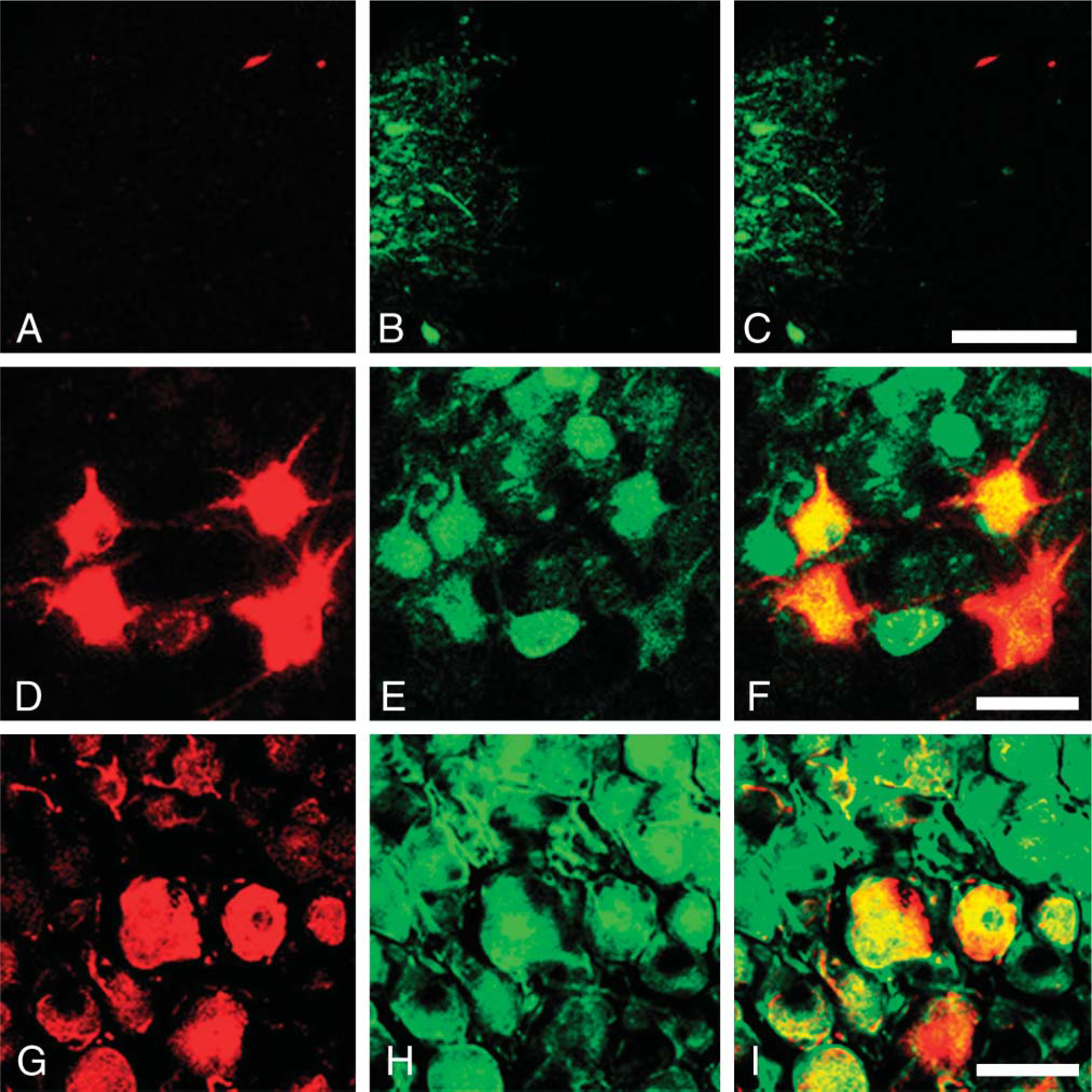
Double immunostaining for cholera toxin B subunit (CTb) (red) and FGF1 (green) in the DMNV (
In the present study we demonstrated that 100% of the pChAT-positive neurons in the intralaryngeal ganglia contained FGF1. pChAT-positive neurons in the intralaryngeal ganglia are parasympathetic neurons (Nakanishi et al. 1999). In the nodose ganglia, some neurons were positive for pChAT. The present study demonstrated that 66% of pChAT-positive neurons contained FGF1. It is unclear whether pChAT-positive neurons in the nodose ganglion were cholinergic. However, some FGF1-positive neurons in the nodose ganglia were labeled with CTb, suggesting that at least some of the sensory neurons innervating the rat larynx contain FGF1. Further studies will be needed to determine whether or not these sensory neurons are cholinergic.
In conclusion, we have demonstrated by IHC and tracing experiments that FGF1 is localized to cholinergic neurons in the nucleus ambiguus, intralaryngeal ganglia, and nodose ganglion, but FGF1 expression is low in cholinergic neurons sending their axons from the DMNV to the larynx. Localization patterns suggest that for innervation of the larynx, FGF1 may have a function in motor neurons, postganglionic parasympathetic neurons, and sensory neurons, but not in preganglionic parasympathetic neurons.
