Abstract
Insulin-like growth factors (IGFs) are expressed in many tissues and control cell differentiation, proliferation, and apoptosis. In teeth, the temporo-spatial pattern of expression IGFs and their receptors has not been fully characterized. The purpose of this study was to obtain a comprehensive profile of their expression throughout the life cycle of ameloblasts, using the continuously erupting rat incisor model. Upper incisors of young male rats were fixed by perfusion, decalcified, and embedded in paraffin. Sections were processed for in situ hybridization and immunohistochemistry. mRNA and protein expression profiles IGF-I, IGF-II, IGF-IR, and IGF-IIR mRNA were essentially identical. At the apical loop of the incisor, very strong signals were seen in the outer enamel epithelium while the inner enamel epithelium showed a moderate reaction. In the region of ameloblasts facing pulp, inner enamel epithelium cells were still moderately reactive while signals over the outer enamel epithelium were slightly reduced. In the region of ameloblasts facing dentin and the initial portion of the secretory zone, signals in ameloblasts were weak while those over the outer enamel epithelium were strong. In the region of postsecretory transition, signals in both ameloblasts and papillary layer cells gradually increased. In maturation proper, signals in ameloblasts appeared as alternating bands of strong and weak reactivities, which corresponded to the regions of ruffle-ended and smooth-ended ameloblasts, respectively. Papillary layer cells also showed alternations in signal intensity that matched those in ameloblasts. These results suggest that the IGF family may act as an autocrine/paracrine system that influences not only cell differentiation but also the physiological activity of ameloblasts.
T
The specific receptors IGF-IR and IGF-IIR are structurally unrelated (reviewed in Foulstone et al. 2005). It has been demonstrated that IGF-IR binds IGF-I with 15- to 20-fold higher affinity than IGF-II (Germain-Lee et al. 1992). IGF-IIR binds IGF-II but barely recognizes IGF-I (Nissley et al. 1991; Kornfeld 1992). IGF-I has characteristics similar to insulin and also interacts with the insulin receptor (Daughaday and Rotwein 1989). IGF-IIR also serves as a cation-independent mannose-6-phosphate receptor (MacDonald et al. 1988; Tong et al. 1988) involved in lysosomal enzyme targeting and has a role in IGF-II turnover (Oka et al. 1985; Kiess et al. 1987; Nolan et al. 1990).
The IGF system plays a role in the formation of the mandible and teeth (reviewed in Werner and Katz 2004) and participates in the regulation of bone metabolism (Baker et al. 1993; Mohan and Baylink 1999; Zhang et al. 2002; Mohan et al. 2003). It has been proposed that IGF-I functions as an autocrine/paracrine regulator of tooth development (Joseph et al. 1996). IGFs are believed to influence ameloblast differentiation and the production of various enamel matrix constituents (Joseph et al. 1993, 1994a, 1996, 1997; Takahashi et al. 1998; Caton et al. 2005).
Previous reports on the in vivo distribution of IGF ligands in teeth dealt with IGF-I and its receptor and focused on differentiation of the ameloblast and apoptosis (Joseph et al. 1994a, 1997, 1999). The developmental sequence of appearance of both IGF-I and IGF-II was investigated by RT-PCR in mouse molars (Takahashi et al. 1998; Caton et al. 2005). The expression of IGF-I coincides with the expression of amelogenin, ameloblastin, and enamelin at the late bell and secretory stage (Takahashi et al. 1998; Caton et al. 2005). The IGF-II receptor was immunodetected in the rat incisor (Al Kawas et al. 1996) but was not found using RT-PCR in mouse molars (Caton et al. 2005). When the molar teeth were cultured with IGF-I and heparin, induction of cytological differentiation was observed. Addition of exogenous IGF-I and IGF-II to molars maintained in culture resulted in an increase in tooth volume. This suggested that IGF-I promoted differentiation and development of ameloblasts and odontoblasts (Caton et al. 2005). Immunolocalization of IGF-IIR in ameloblasts showed an alternating distribution pattern corresponding to the ameloblast modulation (Josephsen and Fejerskov 1977; Smith et al. 1987) in the maturation zone (Al Kawas et al. 1996). It remains obscure how the IGF system acts as an autocrine/paracrine system and influences cell differentiation and the physiological activity of ameloblasts. The fact that both IGF-I and IGF-II and their receptors exhibit similar expression profiles further suggests some coordinated interaction and that they, indeed, work in synchrony not only with themselves but most likely with other regulating molecules.
There are no studies addressing tissue and cell distribution of IGF-II in teeth or describing the relationship of IGFs with respect to ameloblast modulation during maturation. The purpose of this work was, therefore, to carry out a systematic study and to obtain a comprehensive profile of the expression of both IGF-I and IGF-II and their receptors throughout the life cycle of ameloblasts, both at the mRNA and protein level. A detailed mapping of the temporo-spatial expression pattern is important because, although not determinant, it is indicative of function. The continuously erupting rat incisor (reviewed in Warshawsky and Smith 1974; Leblond and Warshawsky 1979) was used for our mapping studies.
Materials and Methods
RT-PCR
Male Wistar rats weighing 100 g were sacrificed by cervical dislocation. Incisors from three mandibles were carefully dissected out, and their enamel organs in the region of maturation were scraped off the enamel surface using a scalpel blade. Total RNA was extracted using a Total RNA Miniprep Kit (Stratagene; La Jolla, CA). cDNA was synthesized with oligo (dT) primer and random hexamers of the total RNA using a Ready-To-Go You-Prime First-Strand Beads kit according to the manufacturer's protocol (Amersham-Pharmacia Biotech; Piscataway, NJ). The primer sets used for RT-PCR are listed in Table 1. A primer set amplifying glyceraldehyde-3-phosphate dehydrogenase (GAPDH; Clontech, Palo Alto, CA) was used as a control.
RT-PCR was performed using a PCR amplification kit (Amersham-Pharmacia Biotech). The reaction was carried out with denaturation at 94C for 5 min, followed by cycles of denaturation at 94C for 30 sec, primer annealing for 30 sec, and product enlongation at 74C for 30 sec. Final elongation was performed at 74C for 5 min. Annealing temperatures and number of cycles are shown in Table 1. PCR products were electrophoresed on 4.5% polyacrylamide gels. Gels were stained with ethidium bromide (IBI/Shelton Scientific; Shelton, CT) and photographed under ultraviolet light (Figure 1).
Tissue Preparation
Twenty male Wistar rats weighing 40 g were anesthetized with 10 g/kg body weight of sodium pentobarbital (Nembutal; Abbott, North Chicago, IL) and perfused via the left ventricle with 4% paraformaldehyde buffered with 0.025 M sodium phosphate, pH 7.4. The maxillae were dissected out and immersed in the same fixative for 2 hr at 4C. They were demineralized with 0.1 M EDTA (pH 7.4) for 3 weeks at 4C, dehydrated in graded ethanols, cleared with xylene, and processed for embedding in paraffin. Sections were cut at 5-μm thickness along the long axis of the incisors.
All animal procedures were approved by the Institutional Ethical Committee for Animal Experiments and properly carried out according to the Guidelines for Animal Experimentation at Nihon University School of Dentistry.
In Situ Hybridization
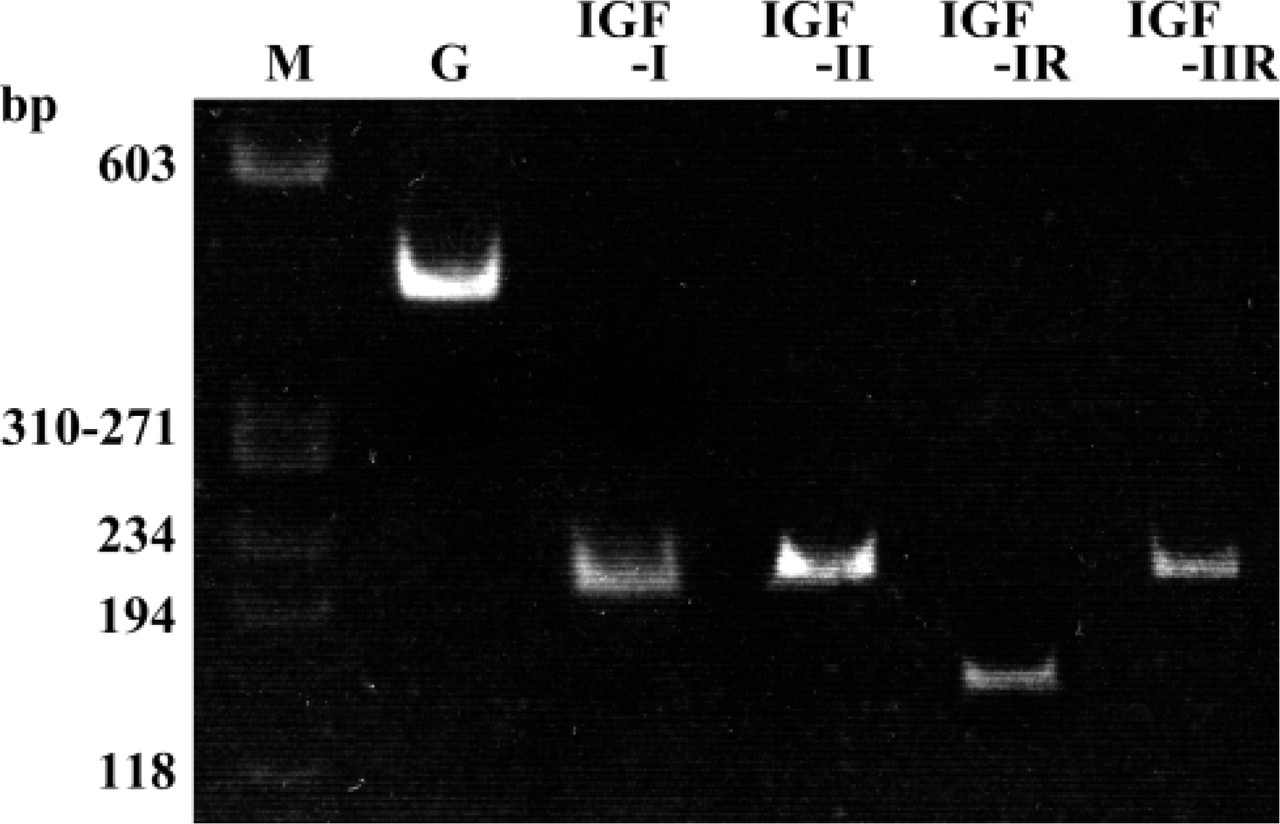
RNA Probes. RT-PCR products were inserted into pBlue-script II SK(+) (Stratagene) and sequenced. The plasmids were linearized using the restriction enzymes HindIII or EcoRI (Takara; Shiga, Japan) and transcribed with T3 or T7 RNA polymerase (Roche Diagnostics; Mannheim, Germany) to generate antisense and sense probes, which were labeled with digoxigenin (DIG-labeling kit; Roche Diagnostics). For control experiments, sense probes were used. Positive controls were performed on sections taken from pancreas, liver, and colon of the Wistar rats weighing 40 g (Figure 2).
Primer sequences and PCR conditions
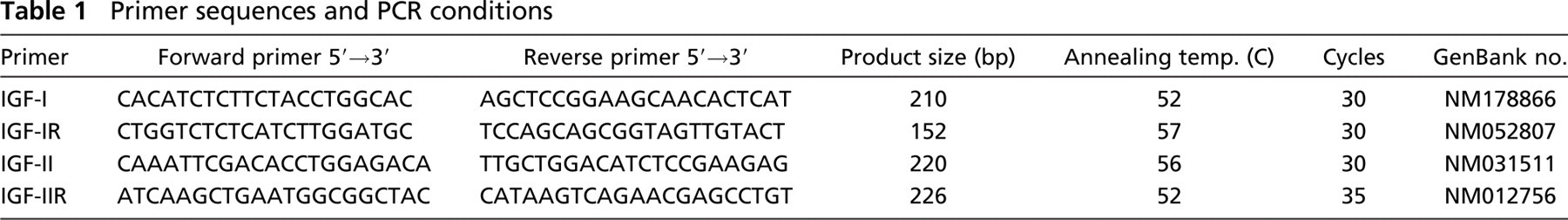
Electrophoresis of products after RT-PCR amplification with IGF-I, IGF-II, IGF-IR, and IGF-IIR from enamel organ of maturation of rat incisors and GAPDH (G) specific primer sets. The bands of IGF-I (210 bp), IGF-II (220 bp), IGF-IR (152 bp), IGF-IIR (226 bp), and GAPDH (452 bp) are detected. M, PhiX174 DNA-Hae III.
Hybridization. Deparaffinized tissue sections were postfixed in 4% paraformaldehyde for 10 min and dipped in 0.1 M triethanolamine containing 0.25% acetic anhydride also for 10 min. Hybridization was performed for 12 hr at 48C using the denatured antisense or sense probes in hybridization solution (50% formamide, 10 mM Tris-HCl buffer, 1 mM EDTA, 1X Denhardt's solution, 0.25% sodium dodecyl sulfate, 10% polyethylene glycol 6000, 50 μg/ml Escherichia coli t-RNA). After hybridization, the sections were treated with RNase A (Roche Diagnostics) (5.0 μg/ml, 30 min, 37C) and extensively washed with salted sodium citrate buffer.
Detections
5-β-Bromo-4-chloro-3-indolylphosphate and Nitroblue Tetrazolium Method. Washed, hybridized sections were reacted with anti-digoxigenin antibody conjugated with alkaline phosphatase (Roche Diagnostics) (1:100 diluted in 1% chicken ovalbumin in PBS) for 12 hr at 4C, washed with PBS, and treated with 5-β-bromo-4-chloro-3-indolylphosphate nitroblue tetrazolium (NBT/BCIP) solution (Roche Diagnostics) according to the manufacturer's instructions.
Tyramide Signal Amplification Method. Washed sections were reacted with anti-digoxigenin antibody conjugated with peroxidase (Roche Diagnostics) (1:100 diluted in 1% chicken ovalbumin in PBS) for 12 hr at 4C. They were washed with PBS and exposed to biotin-labeled tyramide (Perkin-Elmer Life Sciences; Boston, MA) diluted 1:50 in amplification diluent (Perkin-Elmer Life Sciences) for 20 min at 37C, followed by peroxidase-labeled streptavidin (Perkin-Elmer Life Sciences) diluted 1:100 in PBS for 30 min at 37C. Reactions were revealed by incubating sections for 20 min at room temperature with a peroxidase substrate (0.05% 3,3′-diaminobenzidine tetrahydrochloride, DAB; Sigma-Aldrich, St Louis, MO) in 0.05 M Tris-HCl, pH 7.6, supplemented with 0.001% H2O2.
Immunohistochemistry
Antibodies. The following polyclonal antibodies were used: goat anti-mouse IGF-I (Santa Cruz Biotechnology; Santa Cruz, CA and Sigma-Aldrich), goat anti-human IGF-II (Santa Cruz Biotechnology and Sigma-Aldrich), rabbit anti-human IGF-IR (Santa Cruz Biotechnology), goat anti-human IGF-IR (Sigma-Aldrich), and goat anti-mouse IGF-IIR (Santa Cruz Biotechnology).
Immunohistochemical Reactions. Dewaxed and hydrated paraffin sections were pretreated with 0.3% H2O2 in methanol to inhibit endogenous peroxidase activity, followed by blocking with 10% normal rabbit serum (for goat anti-IGF-I, -IGF-II, -IGF-IR, and -IGF-IIR) or 10% normal goat serum (for rabbit anti-IGF-IR). Sections were incubated with the primary antibodies, diluted 1:25 with 1% chicken ovalbumin in PBS for 12 hr at 4C, and washed with PBS followed by biotinylated secondary antibodies (Nichirei; Tokyo, Japan), rabbit anti-goat antibodies (for goat anti-IGF-I, -IGF-II, -IGF-IR, and -IGF-IIR) or goat anti-rabbit antibodies (for rabbit anti-IGF-IR). Immunoreactive sites were detected with the Vectastain Elite ABC kit (Vector Laboratories; Burlingame, CA) and visualized by treating with 0.05% DAB.
Controls consisted of incubating sections with (a) antigen-adsorbed antibody, (b) normal non-immune serum, (c) PBS instead of primary antibody, or (d) directly with secondary antibodies.
Results
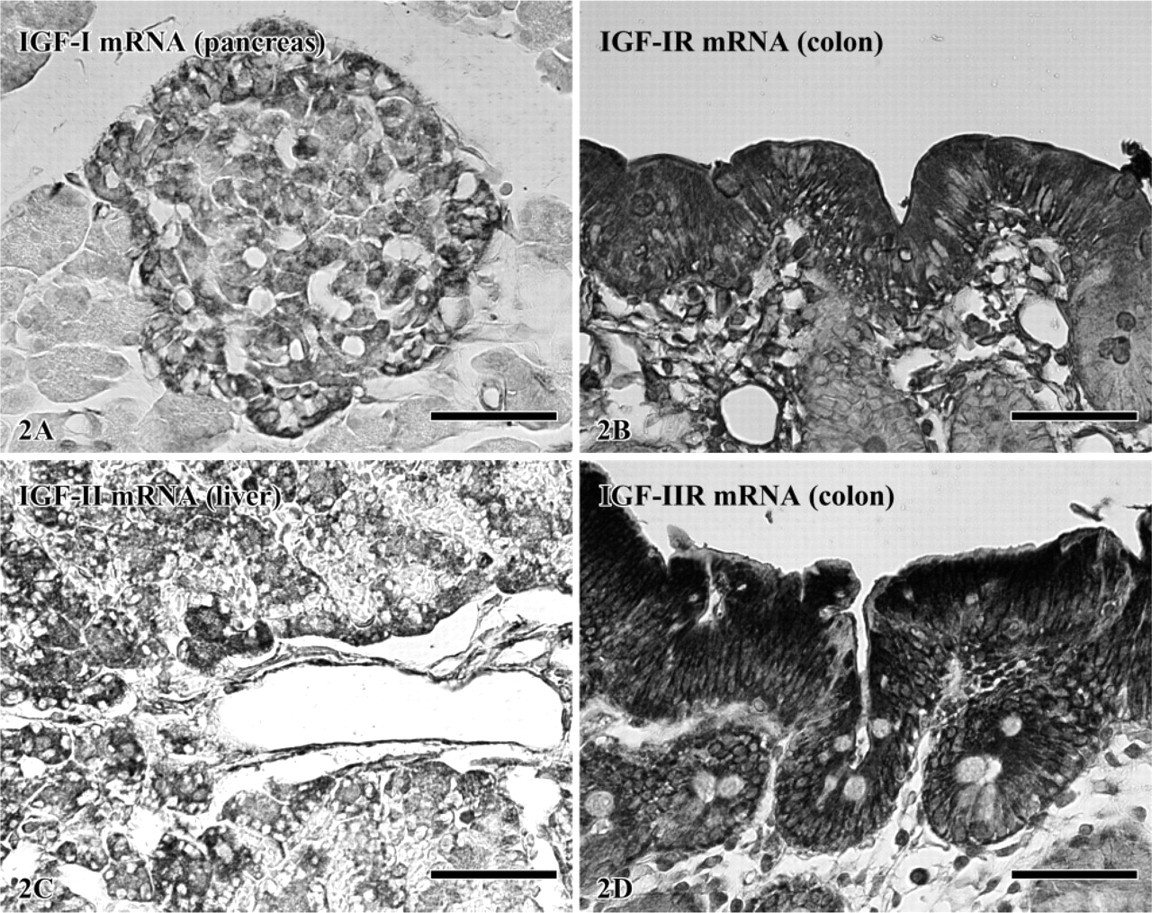
The classification of Warshawsky and Smith (1974) was used to subdivide the zones of amelogenesis. Groups of ruffle-ended ameloblasts (RAs) and smooth-ended ameloblasts (SAs) across the maturation zone were denoted as described by Nanci et al. (1987).
RT-PCR confirmed the expression of IGF-I, IGF- II, IGF- IR, and IGF-IIR mRNAs in the maturation stage enamel organ (Figure 1).
Expression of IGF-I, IGF-II, IGF-IR, and IGF-IIR mRNA
Control hybridizations using sense RNA strands resulted in no signals in ameloblasts or in any other cells (data not shown). For positive control, IGF-I mRNA was detected in islet cells of pancreas (Figure 2), IGF-IR mRNA in mucosal epithelial cells of the colon (Figure 2), IGF-II mRNA in hepatocytes (Figure 2), and IGF-IIR mRNA in mucosal epithelial cells of the colon (Figure 2D).
The expression pattern of IGF-I, IGF-II, IGF-IR, and IGF-IIR mRNA was essentially identical. Although detections by NBT/BCIP and tyramide signal amplification (TSA) methods showed the same distributions, results with TSA were generally the strongest.
Positive controls. Expressions of IGF-I, IGF-IR, IGF-II, and IGF-IIR mRNA. (
At the apical loop of the incisor, very strong signals were seen in cells of the outer enamel epithelium while cells of the inner enamel epithelium showed a moderate reaction (Figures 3A and 3B). In the region of ameloblasts facing pulp, inner enamel epithelium cells (preameloblasts) were still moderately reactive while signals over the outer enamel epithelium were slightly reduced and stellate reticulum was weak. In the region of ameloblasts facing dentin and the early portion of the secretory zone, signals associated with differentiating ameloblasts were weak while those over the outer enamel epithelium, stellate reticulum, and stratum intermedium were strong (Figures 3A and 3C). However, toward the end of the secretory zone, signals over the latter cell layers decreased to moderate levels. In the maturation zone, signals in both ameloblasts and papillary layer cells gradually increased from weak to moderate as the cells progressed through the region of postsecretory transition (Figure 3). In maturation proper, hybridization signals in ameloblasts appeared as alternating bands of strong and weak reactivities, which corresponded to the regions of RAs and SAs, respectively (Figure 3A, Figure 4, and Figure 5). The most incisal portion of each RA band and each SA band always exhibited stronger signals than those of the band itself (Figure 4). Papillary layer cells also showed alternations in signal intensity that matched those over ameloblasts but with slightly weaker intensities. In addition, some SA cells occasionally expressed strong signals (Figure 3A, Figure 4, and Figure 5). Expression of IGFs and their receptors are summarized in Table 2.
Immunolocalization of IGF-I, IGF-II, IGF-IR, and IGF-IIR
Immunolabeling patterns and relative staining densities for IGF-I, IGF-II, IGF-IR, and IGF-IIR were identical to the mRNA expressions patterns (Figure 6). No labeling was observed in negative control sections (not shown).
Expression of IGF-I mRNA (5-β-bromo-4-chloro-3-indolylphosphate nitroblue tetrazolium detection). (
Discussion
We have shown that both IGF-I and IGF-II and their corresponding receptors are expressed throughout amelogenesis in the rat incisor, and that both the transcript (mRNA) and the translated protein distributions correlate. Although the expression pattern of IGF-I throughout enamel formation has been previously reported (Joseph et al. 1993, 1994a, 1996), this is the first comprehensive description for IGF-II. The level of expression of these two ligands and their receptors varies across the incisor. In addition, our analyses have shown, for both the ligands and receptors, an alternating pattern of expression in the maturation zone that overlays the modulation cycle of ameloblasts. These data suggest that the IGF system likely participates in more than one process during amelogenesis.
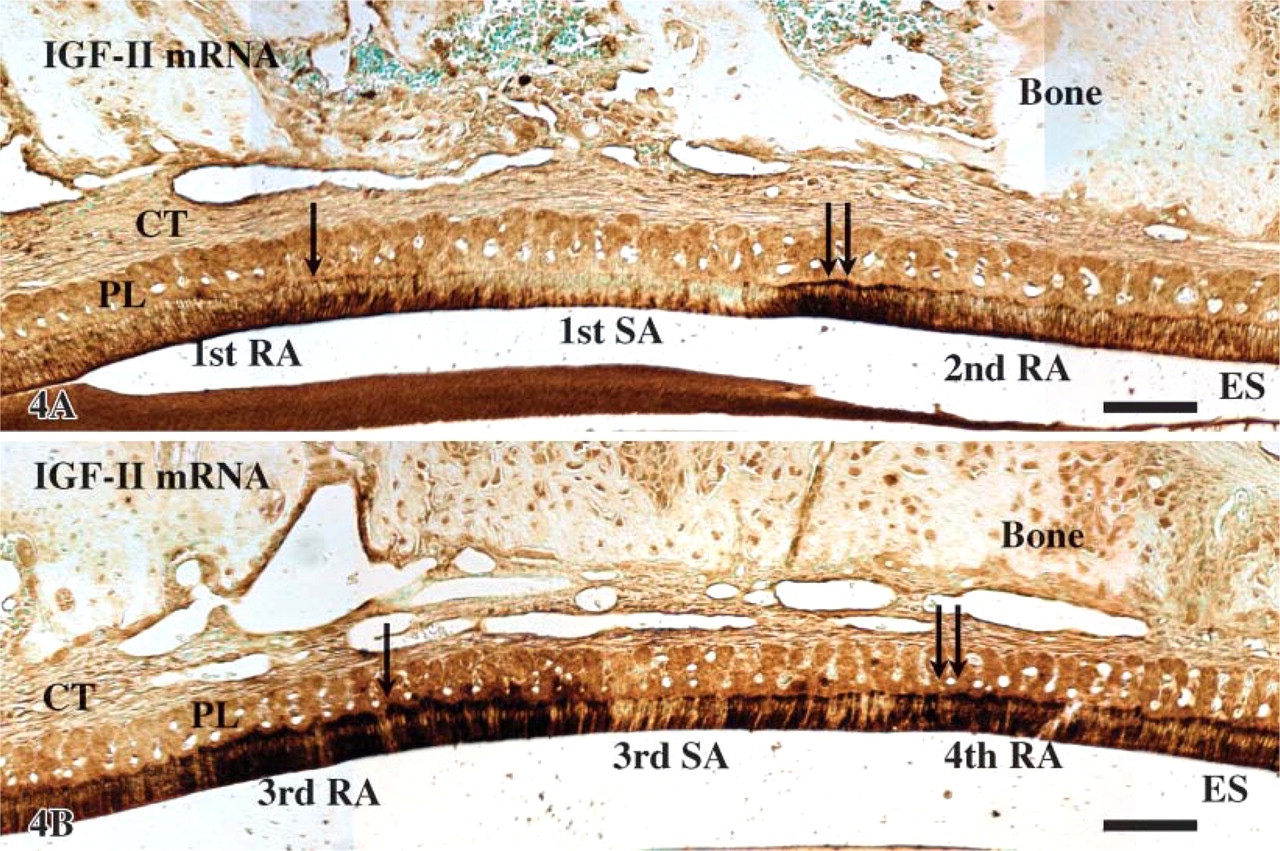
Expression of IGF-II mRNA [tyramide signal amplification (TSA) detection] in the transition from 1st RA to 2nd RA (
In positive control, IGF-I mRNA was detected in islet cells of pancreas as reported by Petrik et al. (1998), IGF-IR mRNA was detected in mucosal epithelial cells of the colon (Heinz-Erian et al. 1991), IGF-II mRNA was detected in hepatocytes (Beck et al. 1988), and IGF-IIR mRNA was detected in mucosal epithelial cells of the colon (Heinz-Erian et al. 1991).
In previous reports, the strongest mRNA signals for IGF-I were found in secretory zone ameloblasts while maturation zone ameloblasts showed much weaker signals (Joseph et al. 1996). In these two zones there was no significant difference in levels of IGF-I and its receptor as detected by immunohistochemistry (Joseph et al. 1993, 1994a). There are no reports on either the message or protein expression of IGF-II across the stages of amelogenesis, but IGF-IIR was revealed by immunolabeling to be more or less equally expressed in secretory and maturation zone ameloblasts (Al Kawas et al. 1996). Surprisingly, mRNA for IGF-IIR was not detected in mouse molars (Caton et al. 2005). Our results reveal some important differences with respect to the previous reports. First, RT-PCR, ISH, and immunolabeling all confirm the presence IGF-II receptors in the enamel organ of the rat incisor. Second, our results differ significantly from the relative intensities of the signals reported for IGF-I and its receptor and IGF-IIR. Third, we have found both message and protein signals in postsecretory transition ameloblasts for IGF-I. The reasons for these differences are not clear but may include technical as well as toothmodel considerations.
It is well known that IGFs influence cell division and differentiation (Conover 2000; Tumber et al. 2000; Werner and Katz 2004; Foulstone et al. 2005). The presence of both IGF-I and IGF-II and their receptors in the apical loop region where cell renewal occurs and in the early part of the presecretory stage where ameloblast differentiation takes place is consistent with the concept that both IGFs are involved in these activities during tooth development.
The focus of previous tooth studies has largely been on the regulatory influence of IGFs on matrix formation (Joseph et al. 1993, 1994b; Takahashi et al. 1998). With the onset of enamel secretion, a strong hybridization signal for IGF-I mRNA was found in secretory ameloblasts (Joseph et al. 1996). In organ culture of mouse molars, exogenous administration of insulin, IGF-I, and IGF-II increased the synthesis of amelogenin, ameloblastin, and enamelin (Takahashi et al. 1998; Caton et al. 2005) and the volume of the enamel layer produced (Takahashi et al. 1998). Together these results suggest that IGFs increase enamel formation by regulating the expression of enamel matrix genes (Takahashi et al. 1998). In our study, secretory stage ameloblasts showed the weakest signals for IGFs and their receptors, both at the message and protein level. The observed higher densities at other regions therefore likely relate to activities other than matrix protein production.
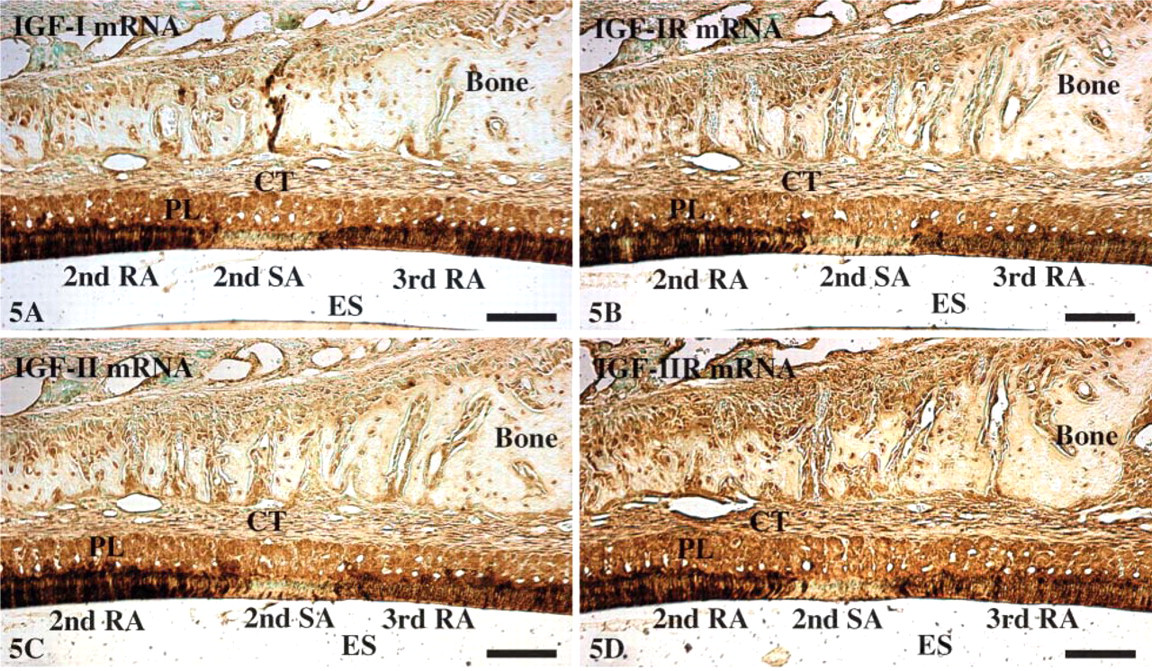
Expressions of IGF-I (
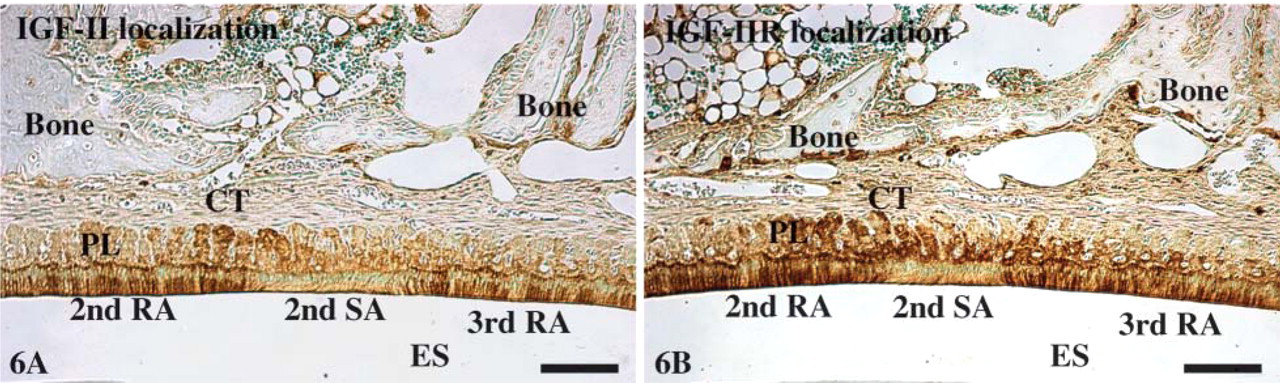
Localizations of IGF-II (
About 25% of cells die during postsecretory transition whereas another 25% do so during maturation proper (Smith and Warshawsky 1977; Smith 1979). IGF-IR blocks apoptosis in embryonic cells and other cell types (Resnicoff et al. 1995; D'Ambrosio et al. 1997; Kulik et al. 1997; Valentinis and Baserga 2001). Joseph et al. (1993,1994a) showed that mRNA expression (Joseph et al. 1996) and immunoreactivity for IGF-I was weak or absent in postsecretory ameloblasts, and that these cells showed no immunoreactivity for IGF-IR (Joseph et al. 1994a, 1997). Based on these results, Joseph et al. (1994a, 1997, 1999) suggested that IGF-IR expression is required to block the default apoptotic pathway in ameloblasts, and that its paucity during postsecretory transition permits cell death to occur. However, this interpretation does not take into account the fact that half the apoptotic activity occurs in maturation proper where ameloblasts were shown to express IGF-IR (Joseph et al. 1994a, 1997; Al Kawas et al. 1996). The intense immunoreactivity for IGF-IIR observed during maturation was related to the lysosomal activity of ameloblasts and their involvement in matrix removal (Al Kawas et al. 1996).
Expression of IGFs and receptors
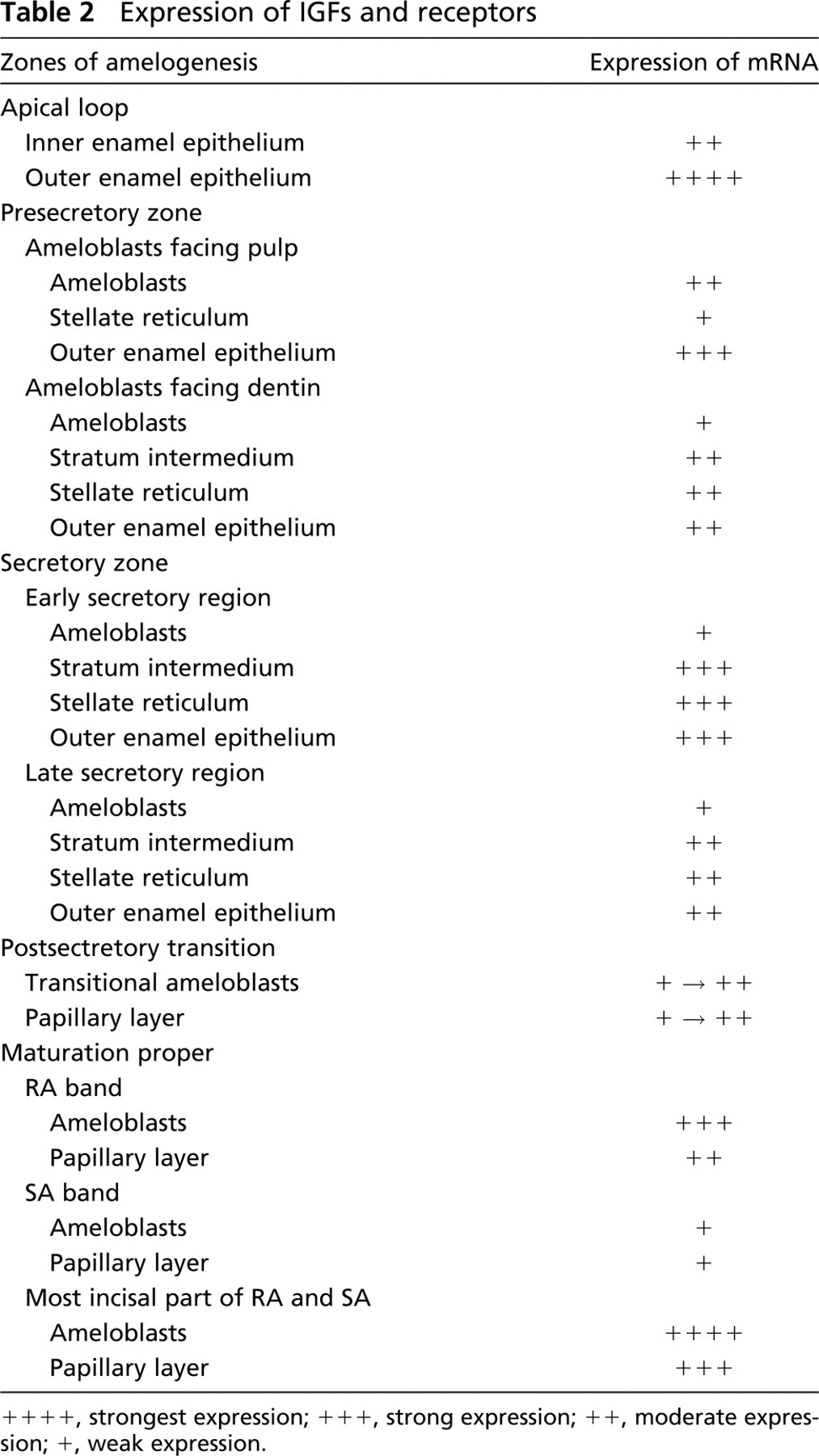
++++, strongest expression; +++, strong expression; ++, moderate expression; +, weak expression.
Our data show no major change in expression of IGF-I and IGF-II and their corresponding receptors from secretion to postsecretory transition and a gradual increase from weak to moderate signals from post-secretory transition to maturation proper. These observations are not consistent with a relationship to the onset of apoptosis. The weak signal observed at the beginning of postsecretory transition may be related to secretory activity because ameloblasts at this stage in the rat incisor still synthesize matrix proteins, albeit at a much reduced rate (Smith 1984; Nanci et al. 1987, 1998; Inage et al. 1996). With regard to the relatively similar immunolabeling signal for IGF-IIR across amelogenesis, the finding is surprising because the lysosomal pool of ameloblasts rapidly expands in preparation for the secretory stage and then gradually enlarges through secretory and into the maturation zone (Smith 1984). In addition, it is now generally agreed that matrix removal during maturation occurs primarily through an extracellular-mediated process (reviewed in Smith 1998). In our opinion, the gradual increase from secretion to maturation is more significant as it may signify the ushering of another, not yet defined, function associated with maturation proper.
In maturation proper, ameloblasts undergo repeated changes in the morphology of their apical extremity and in function, a process called modulation (Josephsen and Fejerskov 1977). Smith et al. (1987) studied the dynamics of this process using fluorochromes. They found that new modulation waves arise near the region of postsecretory transition and progress along the ameloblast layer toward the incisal tip of the tooth, in the rat incisor, at a rate of one modulation every 8.5 hr. Regulation of the modulation process is still not understood and, according to the present results, may implicate the IGF system.
Whereas previous studies have shown some mRNA and immunolabeling signals for IGF-I in maturation stage ameloblasts (Joseph et al. 1993, 1994a, 1996), our results reveal very strong mRNA expression and immunolabelings for both IGF-I and IGF-II and their receptors in these cells. The signals were not constant throughout the maturation zone but exhibited a banding pattern that overlaid with the ameloblast modulation, a finding that has also been previously reported for IGF-IIR (Al Kawas et al. 1996). A differential expression between RAs and SAs has also been reported for calbindins (Berdal et al. 1991) and Ca2+,Mg2+-ATPase (Inage et al. 1979), RAs generally showing more immunoreactivity. Although such variations in protein expressions may be directly linked to regulation of the modulation cycle, it may also be that they simply reflect variations in the overall physiological activity of RAs and SAs. The observation that the strongest reactions correlate with transition points between RAs to SAs and SAs back to RAs is, however, noteworthy. RAs and the underlying papillary layer cells were dramatically more reactive than SAs and associated papillary layer cells. Such a differential labeling in papillary layer cells was not reported in the study of Al Kawas et al. (1996). Indeed, our results showing that the papillary layer signals match those of the ameloblasts would be more consistent with the proposal that maturation ameloblasts and papillary layer cells likely act as a functional unit (Al Kawas et al. 1996; Smith et al. 2005).
In conclusion, mRNA and protein signals for IGF-I and II and their receptors have been demonstrated in the enamel organ across amelogenesis. In ameloblasts, the most intense signals were observed in the pre-secretory and postsecretory transition and maturation proper ameloblasts. Although not providing a direct causal relationship, our data together with those from previous reports lend strong support to the possibility that these molecules may act as an autocrine/paracrine system that influences not only cell differentiation but also the physiological activity of ameloblasts. The fact that both IGF-I and IGF-II and their receptors exhibit similar expression profiles further suggests some coordinated interaction and that they, indeed, work in synchrony not only with themselves but most likely with other regulating molecules.
Footnotes
Acknowledgements
This work was supported in part by grants from the Ministry of Education, Culture, Sports, Science and Technology to promote multi-disciplinary research projects (to TY and TI), Sato Fund, Nihon University School of Dentistry (to TI), Uemura Fund, Nihon University School of Dentistry (to TI), Dental Research Center, Nihon University School of Dentistry (to TI), and Grant-in-Aid for Scientific Research (to TI).
