Abstract
Syncytiotrophoblast and invasive extravillous trophoblast arise from a common stem cell, namely villous cytotrophoblast, but have very different characteristics. The study of the differentiation process relies on the availability of suitable markers for these different cell types of developing placenta. In this work, we have produced monoclonal antibodies that are specific to human villous cytotrophoblast. Monoclonal antibody (MAb) MG2 was specific to villous cytotrophoblast across gestation, and recognizes hepatocyte growth factor activator inhibitor type 1. MAb MD10 stained villous cytotrophoblast across gestation and also some endothelial cells, particularly in the second or third trimester. MAb MD10 recognizes human integrin α6β4. As a test for specificity, the novel MAbs were also used for staining of frozen tissue from human colon carcinoma. The results show that the two antibodies can be used as tools to study human villous cytotrophoblasts and also human tumors. The MG2 antibody seems most specific and promising for the study of various aspects of human villous cytotrophoblast.
V
Before 1999, various cytokeratins were used as markers for cells of trophoblast subpopulations (Daya and Sabet 1991; Mühlhauser et al. 1995; Pröll et al. 1997). However, there are a few reports of cytokeratins being expressed also in stroma of villi (Khong et al. 1986; Beham et al. 1988; Blaschitz et al. 1997). Studies on fibroblast cells from the mesenchymal part of first-trimester villi showed the expression of cytokeratins 8 and 18, and suggested that cytokeratin 7 would be the most specific marker for human trophoblast (Haighn et al. 1999). These findings were later confirmed by Blaschitz et al. (2000). Subsequently, the MAb AC133-2 against CD133 was suggested as a marker for all subtypes of trophoblast, similar to cytokeratin 7 (Pötgens et al. 2001).
In recent years, workshops arranged by the International Federation of Placenta Associations have gathered to discuss common problems in placental research. The issue of the lack of markers that discriminate between syncytiotrophoblast and cytotrophoblast has repeatedly been raised in the workshops (Frank et al. 2000,2001; King et al. 2000; Shiverick et al. 2001). Recently, a method for the isolation of pure villous cytotrophoblast cells based on positive immunoselection with a cytotrophoblast-specific antibody, MAb C76-18, was described (Pötgens et al. 2003).
The purpose of this work was to produce MAbs that are specific to human cytotrophoblast. Such antibodies would be useful tools in the study of the differentiation of trophoblast subpopulations from villous cytotrophoblast. We describe here the production and characterization of two new monoclonal antibodies to villous cytotrophoblast that will be valuable in further studies of these cells.
Materials and Method
Production of Monoclonal Antibodies
Twelve BALB/c mice were immunized with a cell suspension of human placenta from the first trimester (7-13 weeks gestation). Human first-trimester placenta was obtained from the Department of Obstetrics and Gynecology, Helsinki University Hospital after elective termination of pregnancy. Both human and animal studies were examined and approved by local ethics committees. The cytotrophoblast cells from first-trimester human placenta were isolated by the method described by Fisher et al. (1990). Briefly, placenta was washed in PBS, and the chorionic villi were separated. The syncytiotrophoblast layer was removed by enzymatic degradation and discarded. Then the cytotrophoblast layer was dissociated from villi by a second enzymatic degradation. The cytotrophoblast cells were further purified by gradient centrifugation in a Percoll gradient as described by Kliman et al. (1986). The results of the isolation procedure were monitored by indirect immunostaining with anti-human cytokeratin 7 monoclonal antibody (clone OVTL/12/30; Dako, Glostrup, Denmark). The mice received four to six intraperitoneal immunizations at 2-week intervals. The final booster was given intravenously 4 days before fusion. The spleen cells were harvested and fused to X63-Ag8.653 myeloma cells at a ratio of 1:10 in the presence of 50% polyethylene glycol 1500 (Boehringer Mannheim; Mannheim, Germany) according to standard protocols.
Hybridoma supernatants were screened by indirect immunohistochemical staining of cryostat sections from first-trimester human placenta. Selected hybridoma cells were cloned, expanded, and stored in liquid nitrogen. The immunoglobulin heavy-chain and light-chain classes were determined using the IsoStrip kit (Boehringer-Mannheim) according to the manufacturer's instructions.
Immunohistochemistry
Human first-trimester placenta was obtained after elective termination of pregnancy. Term placenta was obtained immediately after spontaneous vaginal delivery. Small pieces of villous tissue were snap frozen in liquid nitrogen and stored at −80C. Cryostat sections were fixed in acetone for 10 min at 4C and immunostained using the Histostain-Plus kit (Zymed; South San Francisco, CA). The cell culture supernatant containing the antibodies was diluted 1:10 in PBS and applied for 1 h at room temperature. The sections were counterstained for 3 min with Mayer's haematoxylin, rinsed in distilled water, and mounted in GVA mount (Zymed).
Paraffin blocks were obtained from the tissue archives of the Department of Pathology, University of Helsinki. The specimens were fixed in 10% formalin and embedded in paraffin. Four-μm-thick paraffin sections were cut, mounted on 3-aminopropyl-triethoxy-silane (Sigma; St. Louis, Mo) -coated slides and dried for 12 h at 37C. The sections were deparaffinized by immersion in xylene for 5 min and rehydrated through a series of alcohols. Three different methods of antigen retrieval were tried. Microwave oven pretreatment was done in sodium citrate buffer, pH 6.0, or 1 mM EDTA in water, pH 8-9. The other antigen retrieval methods used were 5 min incubation in SDS solution (1% SDS in PBS) and 25 min incubation in trypsin solution (0.% trypsin in PBS). The indirect immunostaining was performed in the same way as for the cryosections.
As controls for staining, frozen sections from human tissue were obtained from the Department of Pathology. The staining procedure was as above using FITC-conjugated goat anti-mouse secondary antibodies (diluted 1:200) for detection of the proteins using a Leica fluorescence microscope.
Western Blot Analysis
Chorionic villous (CV) tissue from first-trimester placenta was frozen in liquid nitrogen and homogenized by mechanical disruption. The homogenized tissue was reconstituted in Laemmli buffer [0.25 M Tris/HCl, pH 6.8, 20% (v/v) glycerol, 4% (w/v) SDS, 1% (v/v) β-mercaptoethanol, and 0.002% (w/v) bromophenol blue] and boiled for 2 min. Cultured cells were washed three times with PBS and immediately scraped off on ice. The cells were reconstituted in Laemmli buffer.
After separation by SDS-PAGE on 12.5% resolving gels (Laemmli 1970), samples were electrotransferred onto nitrocellulose membrane (Hybond-C Pure; Amersham Pharmacia Biotech, Uppsala, Sweden). The remaining binding sites were blocked with 5% defatted milk powder or 3% bovine serum albumin (BSA) in 10 mM Tris-HCl, 0.15 M NaCl, 0.05% Tween-20, pH 8.0, (TBST) overnight. After washing in TBST, the membranes were incubated for 1 h with primary antibody. The primary antibody was MAb-producing cell culture supernatant diluted 1:5 in 3% BSA in TBST, or anti-hepatocyte growth factor activator inhibitor type 1 (HAI-1) antibodies MAb 1N7 and MAb C76-18, which were diluted 1 mg/ml in 1% BSA in TBST. After washing in TBST, the membranes were incubated for 1 h with peroxidase-labeled anti-mouse IgG alkaline phosphatase conjugate (Sigma). The membranes were washed, and the specific bands were detected by colored alkaline phosphatase (AP) substrate consisting of nitro blue tetrazolium and 5-bromo-4-chloro-3-indolyl-phosphate (Promega; Madison, WI), which were mixed in AP buffer (0.1 M Tris-HCl, pH 9.5, 0.1 M NaCl, 5 mM MgCl2) according to the manufacturer's instructions. Alternatively, the specific bands were detected on photographic film with an enhanced chemiluminescence system according to the manufacturer's instructions (Amersham Pharmacia Biotech).
Immunostaining of Cells Cultured from CV Biopsies and Choriocarcinoma Cell Lines
Cell cultures were started from CV samples derived at 10 to 12 weeks of gestation. CV cultures were obtained from the Department of Obstetrics and Gynecology, Helsinki University Hospital. The cells were grown in Chang D medium (Irvine Scientific, Santa Ana, CA). Cells were grown for 20 h on glass coverslips to confluence. Coverslip-attached cells were then fixed in acetone for 10 min at −20C and rinsed in PBS. The primary antibody was layered on fixed cells and incubated for 30 min in a humid chamber. Immunostaining was continued using the Histostain-Plus kit (Zymed). The sections were counterstained for 3 min with Mayer's hematoxylin, rinsed in distilled water, and mounted in GVA mount (Zymed). BeWo, JAR, and JEG cells were cultured on glass coverslips for 20 h and stained as described above.
Immunopurification of the Antigens for MAb MD10 and MG2
MAbs MD10 and MG2 were purified and concentrated from cell culture supernatant by HiTrap Protein A affinity columns (Amersham Pharmacia Biotech). Purified MAbs were cross-linked to Protein G Sepharose (Amersham Pharmacia Biotech) with dimethylpimelimidate (Gersten and Marchalonis 1978).
JEG cells, grown to subconfluency, were lysed in cold NET buffer (20 mM Tris-HCl, pH 8.0, 400 mM NaCl, 1 mM EDTA, 1% Triton-X-100) for 30 min on ice. The lysates were centrifuged for 35 min at 16,000 × g. The supernatant was used in the further purification steps and the pellets were discarded.
The antigens were immunoprecipitated from the supernatant. One ml of supernatant was preadsorbed with Protein G Sepharose without antibodies for 30 min. Then the supernatant was recovered and incubated with MAb-protein G Sepharose. The Sepharose was washed, and the antigen was dissociated with 1% SDS in water for 40 min at 40C. The immunoprecipitated proteins were separated by SDS-PAGE in a 10% gel and stained with Coomassie Brilliant Blue.
In-gel Digestion of Proteins and Mass Spectrometric Analysis
Coomassie Brilliant Blue-stained protein bands were cut out of gels and destained, and in-gel digestion was performed using previously described methods (Shevchenko et al. 1996). Proteins were reduced and alkylated with iodoacetamide before digestion with trypsin (Sequencing Grade-Modified Trypsin, Promega) overnight at 37C. The peptides were extracted once with 25 mM ammoniumbicarbonate and twice with 5% formic acid, and the extracts were pooled. Before matrix-assisted laser desorption/ionization time-of-flight (MALDI-TOF) mass spectrometric analysis, the peptide mixture was desalted using Millipore μ-C18 ZipTip (Millipore Corporation; Billerica, MA). Mass mapping of the peptides generated was performed with a Biflex MALDI-TOF mass spectrometer (Bruker Daltonics; Bremen, Germany) in a positive ion reflector mode using α-ano-4-hydroxycinnamic acid as the matrix. The MALDI spectra were internally calibrated with the standard peptides angiotensin II and adrenocorticotropin-18-39. Database searches were carried out using the ProFound database (http://prowl.rockefeller.edu/cgi-bin/ProFound).
Expression of a Plasmid Encoding Human HAI-1
The plasmid containing the cDNA for the whole coding region of HAI-1 was linearized by XmnI and transfected into CHO cells using Lipofectamine Reagent (Life Technologies/Finnzymes; Espoo, Finland) according to the manufacturer's instructions. After transfection, the cells were cultured in Ham's F-12 medium containing Geniticin (Life Technologies/Finnzymes). Geniticin-resistant colonies were cultured and processed for immunoblot analysis.
Results
Monoclonal Antibodies
From the eight fusions carried out, approximately 200 hybridomas were screened by indirect immunostaining of cryosections from first-trimester placenta. of these hybridomas, 21 were positive to placenta. Three hybridomas producing monoclonal antibodies to cytotrophoblast were recovered, but one of them ceased antibody production. The two MAbs that survived, MD10 and MG2, were both isolated from the same mouse.
MAbs MD10 and MG2: Isotype and Molecular Mass
Antibodies MD10 and MG2 were both of IgG1 kappa isotype. MAb MD10 did not give any bands in immunoblotting (Figure 1A). The antibody probably recognizes a native conformation of the antigen, which is not observed in immunoblots. The molecular mass of MG2 antigen was approximately 68 kDa by Western blot analysis of placenta extract (Figure 1). Immunoblot analysis of choriocarcinoma cell line extracts gave bands at approximately 65 kDa (Figure 1). Some weak unspecific background is seen in this immunoblot.
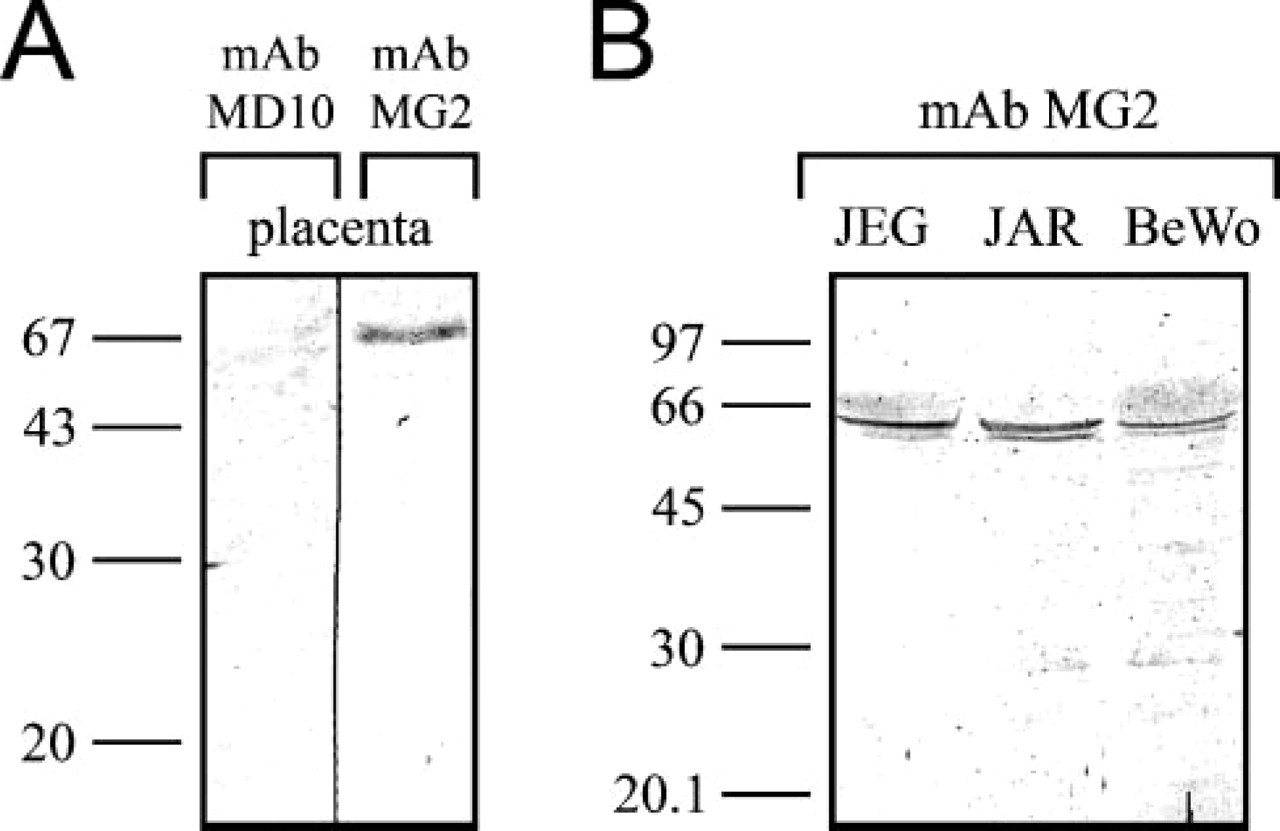
Western blot analyses using MAbs generated in this study. (
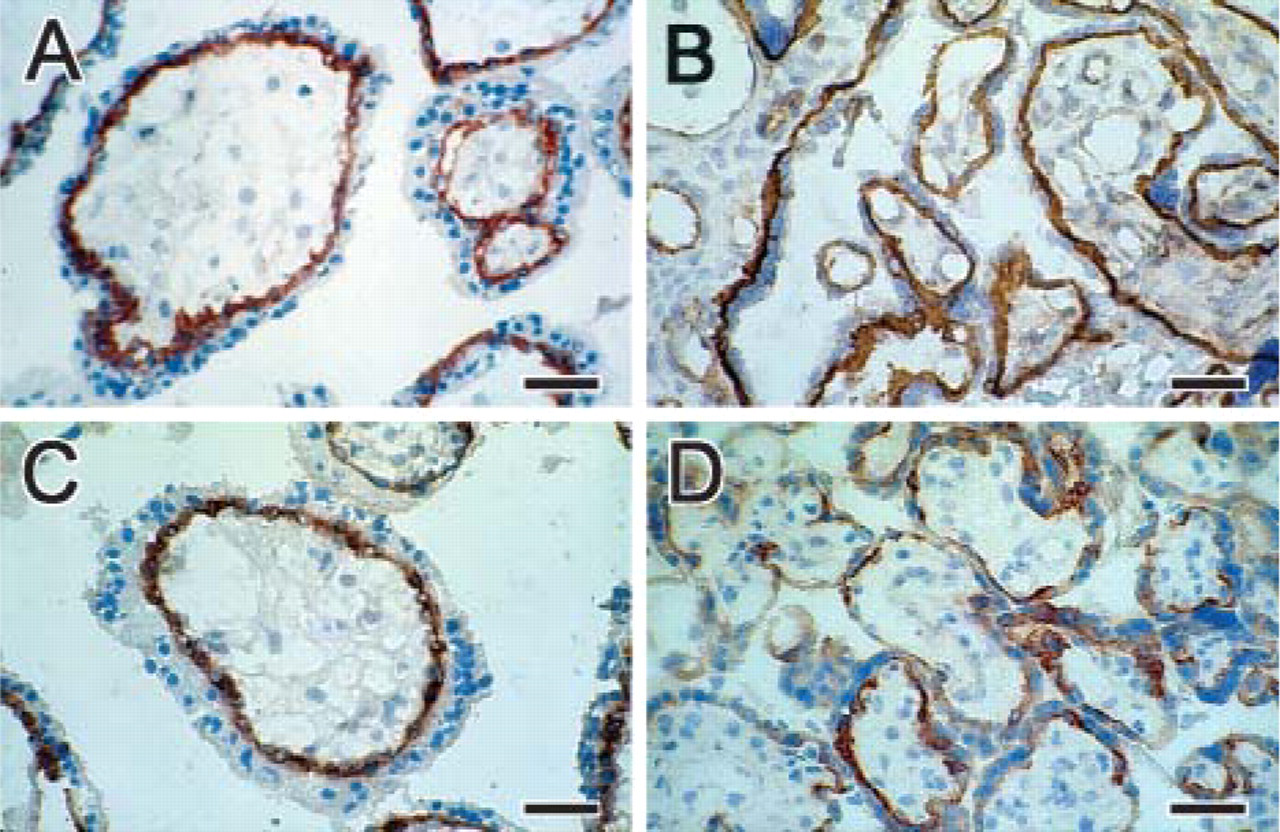
Immunohistochemistry of human placenta at first trimester (
Immunohistochemistry and Immunostaining of Cell Cultures
MAbs MD10 and MG2 revealed strong staining of villous cytotrophoblast in cryostat sections from first-trimester and term placenta (Figure 2). In some cases, MAb MD10 gave an even stronger staining than MG2 in first-trimester placenta. Also, the staining of term placenta differed: the MAb MD10 stained the whole cell layer connected to the basal lamina as well as some endothelium, whereas MAb MG2 stained separate cells (villous cytotrophoblast) connected to the basal lamina but did not stain the endothelium (Figures 2B and 2D). In paraffin sections, we were unable to reveal staining using these antibodies despite testing various antigen retrieval methods. Extravillous trophoblast cells in cell islands and cell columns were stained using MAb MD10 and MAb MG2 only when in contact with or in close proximity to the basement membrane. Extravillous trophoblast cells in basal plate were not stained (Figure 3).
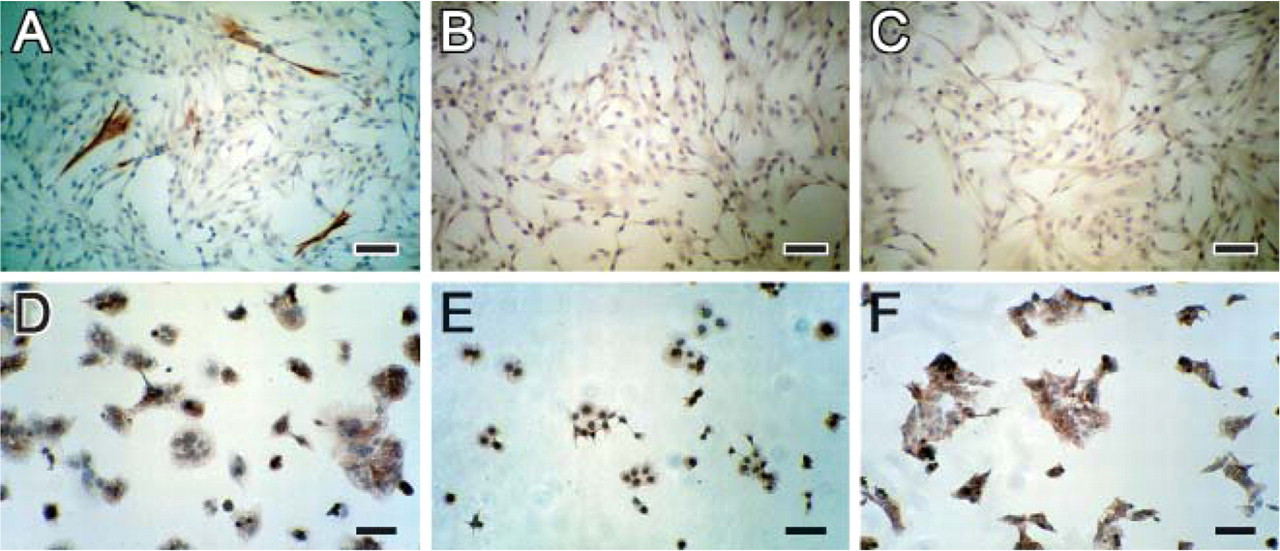
Neither of these MAbs stained any cells cultured from CV samples (Figures 4B and 4C). In these cultures, cells of trophoblastic origin were identified by using anti-cytokeratin 7 antibodies (Figure 4A). MAb MD10 and MAb MG2 stained BeWo, JAR, and JEG cells similarly (Figures 4D-4F; figures showing staining by MAb MG2).
Immunoprecipitation of Antigens Recognized by MAb MD10 and MAb MG2 and Mass Spectrometry Analyses
Immunoprecipitation by MAb MD10 from JEG lysate gave three major specific protein bands in SDS-PAGE (Figure 5, Lane D), and these were further identified by cutting bands out of the gel, in-gel digestion with trypsin, and analysis of peptides by MALDI-TOF mass spectrometry. The two upper bands were identified as human integrin β4, which exists in two subtypes with sizes characteristic of these bands. The other bands present are most probably the precursor of the protein (40 and 27 mass matches, respectively). The lower band was identified as human integrin α6 (28 mass matches). From these data, the protein was unambiguously identified as human integrin α6β4 heterodimer.
MAb MG2 gave a diffuse band (Figure 5, Lane B), which was analyzed using an approach similar to that described above. Masses of fifteen tryptic peptides resulting from this band matched into the sequence of HAI-1. Immunoprecipitation without antibodies did not reveal any of the above-mentioned protein bands in the SDS-PAGE (Figure 5, Lane C). Likewise, digestion of the gel pieces did not reveal any of the peptides specific for the proteins.
Comparison of MAb MG2 with Other Anti-HAI-1 Antibodies in Western Blot Analysis
Two HAI-1-specific antibodies, MAb 1N7 and MAb C76-18, very strongly recognized the protein that was immunoprecipitated with MAb MG2 (Figure 6, Lanes C and D). A lysate of CHO cells transfected with human HAI-1 cDNA served as the positive control. A strong double band was seen with MAb 1N7 and a weaker single band with MAb C76-18 (Figure 6, Lanes B and E). Untransfected CHO cell lysate and immunoprecipitation without any antibody served as negative controls. Faint bands were observed around 66 kDa with MAb 1N7 (Figure 6, Lanes A and H) and none with MAb C76-18 (Figure 6, Lanes F and G).
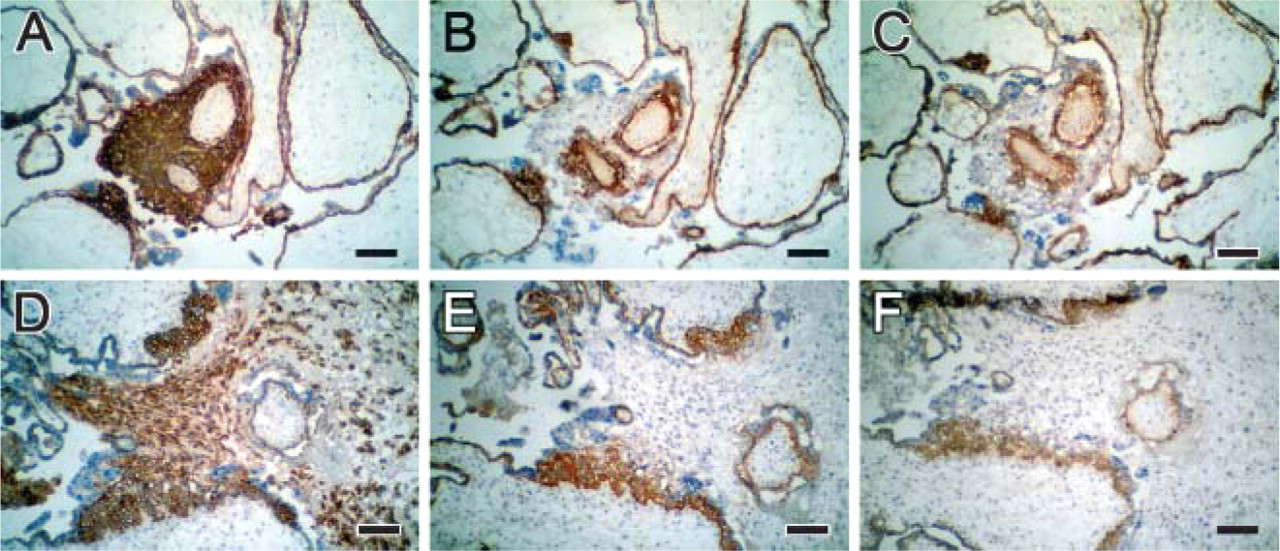
Immunohistochemistry of human placenta with extravillous cytotrophoblast in cell islands (
(
Immunostaining of Human Tumor Cells Using MD10 and MG2 Antibodies
To show specificity, frozen tissue from human tissues other than placenta were used for staining, employing the novel MAbs. In invasive human colon carcinoma, mAB MD10 stained cells in the tumor tissue but not in the neighboring stroma (Figures 7A and 7B). This is in keeping with the expression of α6β4 integrin in invasive and metastatic carcinomas, where the high levels correlates with disease progression (Chung et al. 2004). Using the MG2 antibody, we observed also an intense staining of HAI-1 in the colon carcinoma cells in typical cell clusters, whereas the adjacent stroma was negative (Figures 7C and 7D). A similar increase in HAI-1 was recently reported in colorectal carcinoma cells, and the expression was associated with the invasion front of the tumor (Nagaike et al. 2004). Apart from tumors, staining of human tissues with MAb MG2 revealed the same distribution of HAI-1 as reported earlier (Kataoka et al. 2000), including positive staining of columnar epithelium at mucosal surfaces and negative staining of hepatocytes (data not shown).
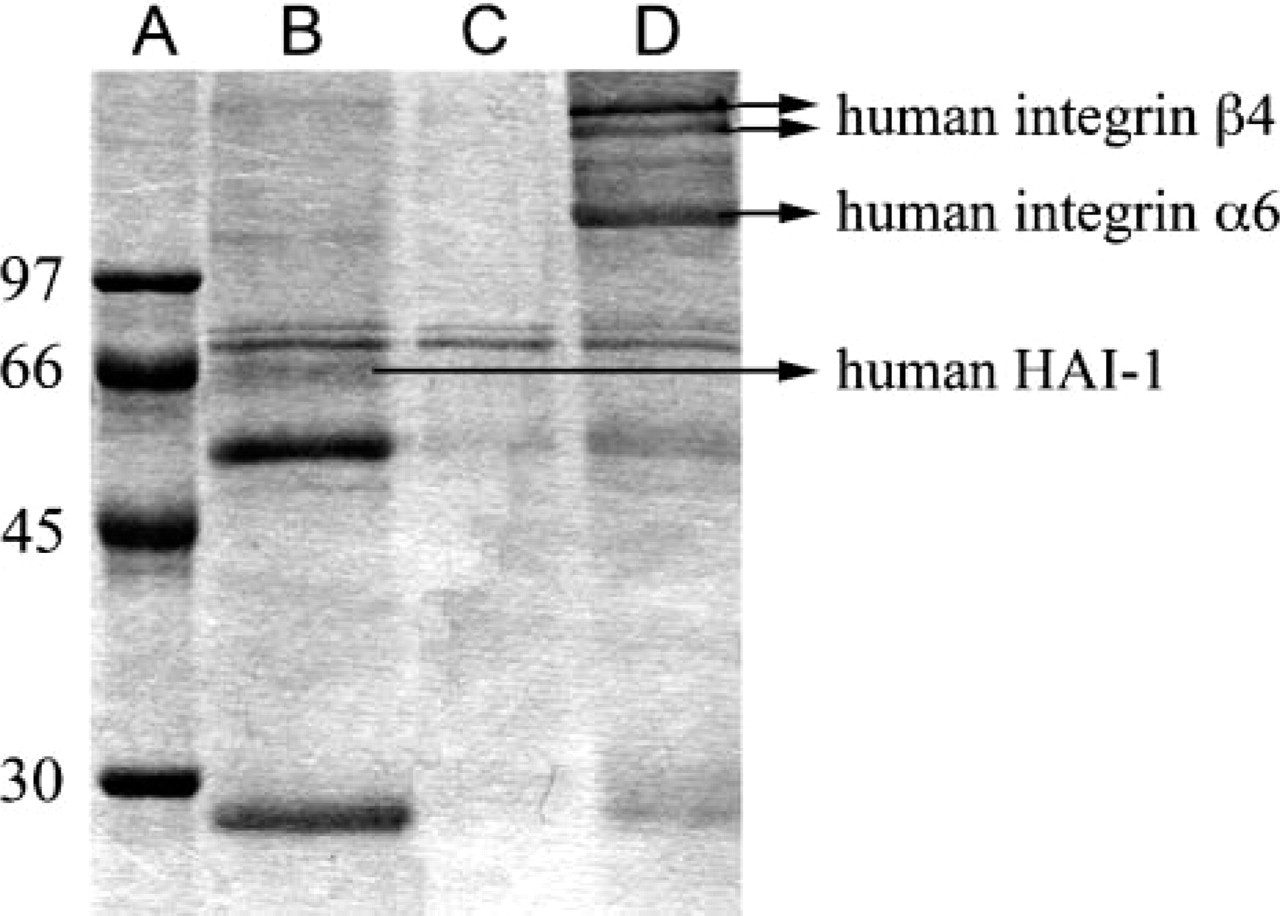
SDS-PAGE of antigens immunoprecipitated from JEG lysate and molecules identified in specific bands by mass spectrometry. Lane A: molecular mass marker; Lane B: proteins immunoprecipitated with MAb MG2; Lane C: negative control; Lane D: proteins immunoprecipitated with MAb MD10. Note the presence of hepatocyte growth factor activator inhibitor type 1 (HAI-1) in Lane B and integrin species in Lane D. The 55-kDa and 25-kDa bands in Lanes B and D represent immunoglobulin heavy and light chains.
Discussion
In this study, we have produced two monoclonal antibodies that recognize villous cytotrophoblast in human placenta. MAb MG2 was specific to villous cytotrophoblast across gestation, as was MAb MD10, but the latter also showed some endothelial staining, particularly in older placenta, where capillaries are more prominent.
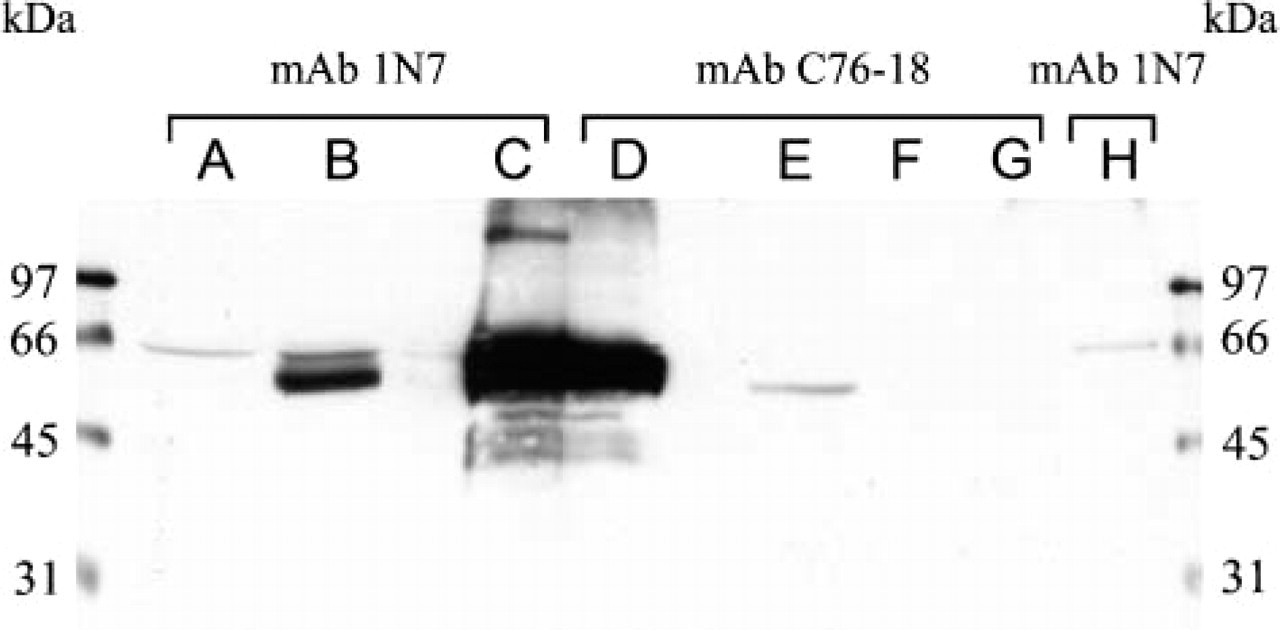
Western blots of cell lysates and protein samples were probed with two anti-HAI-1 antibodies, MAb 1N7 (Lanes A, B, C, and H) and MAb C76-18 (Lanes D, E, F, and G). Lanes A and F: CHO lysate as a negative control; Lanes B and E: HAI-1-transfected CHO lysate as a positive control; Lanes C and D: protein sample immunoprecipitated with MAb MG2 from JEG lysate; Lanes F and G: negative control for immunoprecipitation without antibodies using JEG lysate. The antigen purified by MAb MG2 is strongly recognized by the anti-HAI-1 antibodies MAb 1N7 and MAb C76-18 (Lanes C and D), which also recognize HAI-1 expressed in CHO cells.
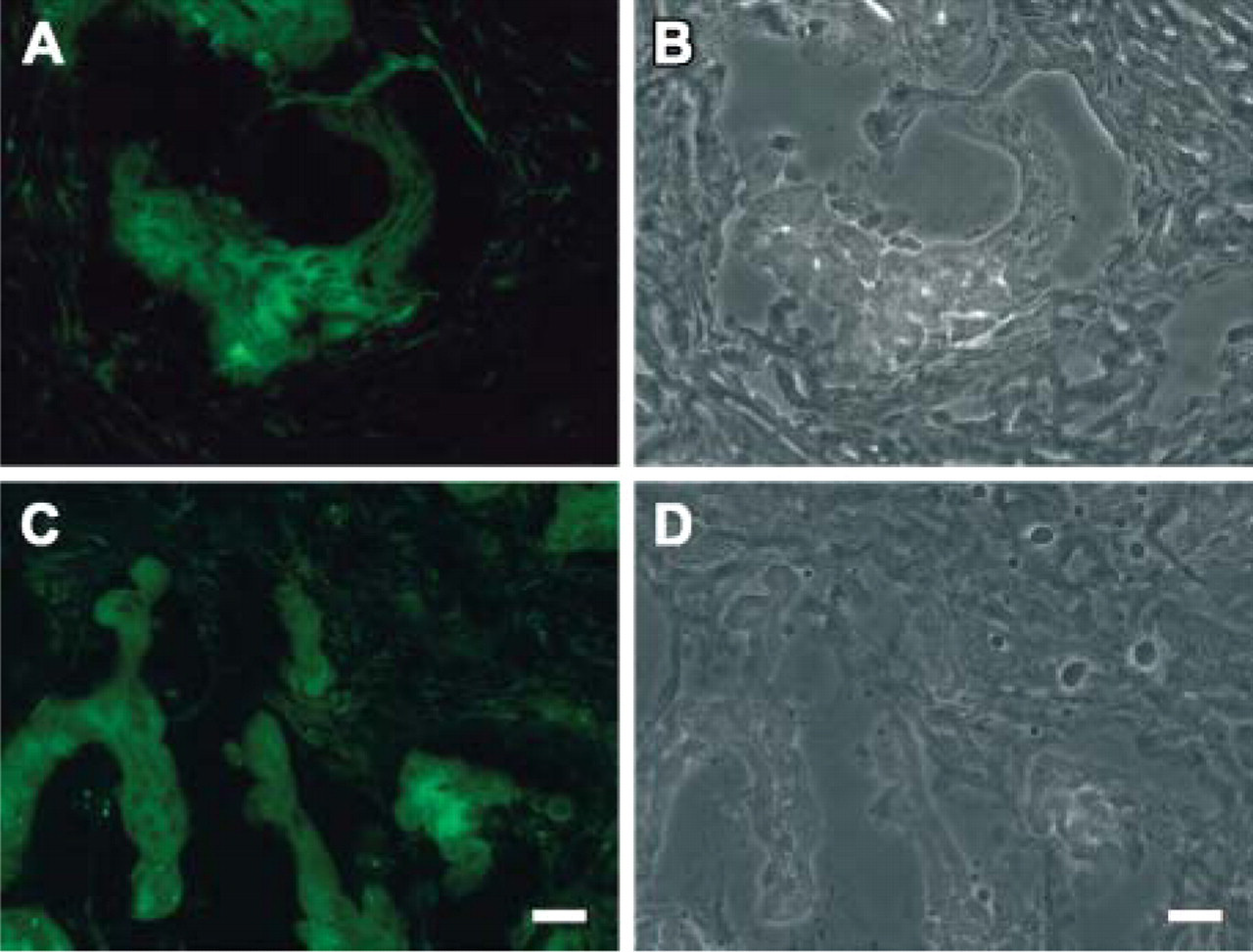
Immunohistochemistry of frozen tissue from human colon carcinoma using (
MAb MD10 was found to immunoprecipitate human integrin α6β4. Previous data on expression of human integrin α6β4 in placentas support our data (Korhonen et al. 1991; Damsky et al. 1992; Aplin 1993). The integrin β4 subunit exists in complex with a range of other integrin subunits, whereas the α6 subunit seems capable of forming complex in similar or identical form with β4 and β1 (Hemler et al. 1989). The complete primary structures of the α6 and β4 subunits have been described from cDNA clones of pancreatic carcinoma cell lines (Tamura et al. 1990).
The MD10 antibody recognizes specifically the α6 subunit of human integrin α6β4, inasmuch as it failed to precipitate other integrins. Moreover, MD10 did stain endothelial cells in villous capillaries, supporting the view that the antibody is specific to the α6 subunit, because this chain is expressed in capillaries, whereas the β4 subunit is not (Sonnenberg and Linders 1990). There are few commercially available antibodies to integrin α6β4 that are suitable for immunohistochemistry. The MD10 antibody is specific and has very high affinity. It strongly stains cytotrophoblasts in placental tissue sections and in cell culture supernatants diluted 1:100.
MAb MG2 immunoprecipitated human HAI-1 protein. The previously reported molecular mass, 66 kDa for HAI-1, is very close to the 67 kDa we observed (Shimomura et al. 1999). The anti-HAI-1 antibodies strongly recognize the antigen, which was purified with MAb MG2 (Figure 6). This shows that the antigen of MAb MG2 is HAI-1. In the choriocarcinoma cell lines, the molecular masses were a bit lower, approximately 65 kDa, but there is a variation in the size of HAI-1 in tissues and cell lines, possibly due to differences in degree of glycosylation (Kataoka et al. 1999).
HAI-1 has recently been identified from the conditioned medium of the human MKN45 stomach carcinoma cell line on the basis of its ability to inhibit hepatocyte growth factor activator protein (Shimomura et al. 1997). HAI-1 has also been purified from human milk in a complex with matriptase, a trypsin-like serine protease (Lin et al. 1999). This molecule has been localized to villous cytotrophoblast of human placenta, which is in accordance with our results (Kataoka et al. 2000).
The biological role of HAI-1 is not fully understood. HAI-1 exists as a transmembrane protein, but at least in culture, cells produce several soluble forms that appear to be the cleavage products of the transmembrane protein (Shimomura et al. 1999). In cell culture, HAI-1 has also been shown to act as a selective binder of hepatocyte growth factor activator protein. Because the binding is reversible, HAI-1 can create a local reservoir of this protein that can then be released for its action (Kataoka et al. 2000). The N-terminal Kunitz domain of HAI-1 is a potent inhibitor of hepatocyte growth factor and matriptase (Denda et al. 2002).
HAI-1 is widely expressed in the simple columnar epithelium of the ducts, tubules, and mucosal surface of various organs, and the expression is upregulated by tissue injury (Kataoka et al. 1999). HAI-1 is found in normal tissue, as well as in malignant epithelial cells, suggesting a role in invasion and metastasis (Oberst et al. 2001). Recently, HAI-1 has more often been referred to in connection with cancer cells (e.g., Parr et al. 2004; Jin et al. 2005), and overexpression of HAI-1 was shown to occur at the invasion front of colorectal carcinoma cells (Nagaike et al. 2004). We were able to confirm this using our MG2 antibody in human colon carcinoma. As shown here, the observed expression of HAI-1 in conjunction with α6β4 integrin in the colon carcinoma cells suggests a high invasive capacity for these tumor cells. The significance and the colocalization of these proteins in other tumor cells will be addressed in more detail in the future.
As shown by targeted mutation of the HAI-1 gene, HAI-1 appears to be crucial during embryonic development, with death of HAI-1-/- mice at E10.5 (Tanaka et al. 2005). Further analysis of these mice revealed that the lack of functional HAI-1 caused impairment in the formation of the placental labyrinth layer. The exact role of HAI-1 in placental physiology and in cytotrophoblasts needs to be studied further. Available markers for villous cytotrophoblast are currently limited, and the antibodies generated in this study are therefore very useful for the study of these cells in different systems. The MG2 antibody can have wide applications in immunohistochemistry and immunoblotting, in cell staining of isolated cytotrophoblasts, in characterization of cell lines, and in the immunopurification of cytotrophoblasts. At the present time, there are commercially available monoclonal antibodies to HAI-1, mainly for Western blot and ELISA applications.
MAbs MD10 and MG2 stained cryosections, but so far, did not work well in paraffin sections. The reason may be that the antigen is recognized in a more native conformation, which may open up the possibility of the isolation of living cells by antibody labeling and cell sorting. We have demonstrated fetal trophoblastic cells in blood samples from pregnant mothers at 12-15 weeks of gestation using the antibody MG2 (Kilpivaara et al. 2002). Our MD10 and MG2 antibodies are included in an EU Network Center of Excellence Study for the identification and isolation of fetal cells in maternal blood for prenatal diagnosis (Special NonInvasive Advances in Foetal and Neonatal Evaluation Network, Project No. 503243).
It is evidently difficult to find new molecules on trophoblast cells. We immunized 16 animals, did 8 fusions, and observed 21 placenta-positives ones, of which only 3 were specific to villous cytotrophoblast and only 2 survived. In recent years, few markers of trophoblasts have been reported, and cytokeratin 7 is the only one in wider use. It is also hard to find markers that would differentiate between trophoblast subpopulations. It is therefore interesting, as shown here, that the MAb MG2 is specific for villous cytotrophoblast among the trophoblast subpopulations.
Footnotes
Acknowledgements
This work was supported by the Finnish Technology Agency, the Finnish Society of Sciences and Letters, Magnus Ehrnrooth's Trust, and the Minerva Foundation. O.K.H. was supported by the Viikki Graduate School in Biosciences, and J.M.A. was supported by Finska Läkaresällskapet. We thank Dr. Laura Korhonen for help with the figures.
