Abstract
Liver growth factor (LGF) is a mitogen for liver cells that shows biological activity in extrahepatic sites and may be useful for neuroregenerative therapies. The aim of this work was to investigate the effects of the intrastriatal (IS) infusion of LGF in the 6-hydroxydopamine rat model of Parkinson's disease. Tyrosine hydroxylase-positive innervation was significantly increased in the dopamine-denervated striatum of rats receiving intrastriatal LGF infusions (160 ng/day/rat × 15 days) as compared with a vehicle-infused group. There was no evidence of dopaminergic neurogenesis in the striatum or substantia nigra in any experimental group at the times studied. However, in those animals undergoing IS-LGF infusion for 48 hr, we found a significant increase in both microglial proliferation and in the number of microglial cells that acquired the ameboid morphology. This is characteristic of activated microglia/macrophages that has been reported to play an important role in dopamine terminal sprouting. In summary, our study shows that IS infusion of LGF stimulates the outgrowth of tyrosine hydroxylase-positive terminals in the striatum of 6-hydroxydopamine-treated rats. As apomorphine-induced rotational behavior was also reduced in these animals, we propose LGF as a novel factor that, when delivered to the striatum, may be useful in the treatment of Parkinson's disease.
Keywords
P
Liver growth factor (LGF), a hepatic mitogen with both in vivo and in vitro activity, was purified by our group some years ago (Diaz-Gil et al. 1986). LGF and hepatic growth factor (HGF) are two completely different entities in terms of molecular weight, chemical structure, activity in hepatocytes, and even the active concentration in brain (LGF, 160 ng/day/rat, see Materials and Methods; HGF, 1.4-4.2 μg/day/rat) (Date et al. 2004). Following an in-depth chemical and immunological study, we demonstrated that LGF is an albumin-bilirubin complex (Diaz-Gil et al. 1987, 1988, 1989). In a model of CCl4-induced cirrhosis in rats, LGF injection decreased total liver collagen; restored serum enzymes, partial architectural integrity, and necrotic tissue; reduced ascites; and improved hemodynamics (Diaz-Gil et al. 1999). The mitogenic activity of LGF in rat liver is mediated by upregulation of tumor necrosis factor-alpha (TNF-α) mRNA expression, as injection of anti-TNF-α antibody substantially decreased LGF activity (measured by stimulation of liver DNA synthesis and increase in proliferating cell nuclear antigen (PCNA)-positive hepatocytes). The first targets of LGF in liver are portal vein endothelial cells (increased TNF-α expression as measured by immuno-histochemistry). Additionally, at 5 pg/ml, LGF stimulated TNF-α secretion in endothelial cells in culture but did not stimulate ICAM-1 or VCAM-1 upregulation (Diaz-Gil et al. 2003). Considering the aforementioned activity of LGF in endothelial cells and the increasing number of reports that relate endothelial stimulation to neurogenesis (Palmer et al. 2000; Jin et al. 2002; Shen et al. 2004), neuroprotection (Sun et al. 2003; Yasuhara et al. 2004), and nerve regeneration (Sondell et al. 1999; Hobson et al. 2000), we wondered whether LGF could be equally active in a known model of PD in rats.
In the present study we report that intrastriatal (IS) infusion of LGF stimulated the outgrowth of DA terminals in the striatum of unilaterally 6-hydroxydopamine (6-OHDA)-lesioned rats, as measured by tyrosine hydroxylase (TH) immunostaining. Moreover, Is-LGF partially reduced apomorphine-induced rotational behavior in these animals. The role of activated microglia/macrophages as mediators of LGF-induced DA fiber sprouting is discussed.
Materials and Methods
LGF Purification
LGF was purified from rat serum following a previously reported procedure (Diaz-Gil et al. 1994). Purity, that is, the absence of other growth factors and/or contaminants in the LGF preparation, was also assessed according to standard criteria (Diaz-Gil et al. 1986, 1987, 1988, 1994). All LGF preparations showed a single band in SDS-PAGE electrophoresis. LGF preparations were lyophilized and kept at 4C until use, at which time aliquots were dissolved in saline for pump perfusion.
Animals and 6-OHDA Lesion Surgery
A total of 90 female Sprague Dawley rats weighing 220-250 g at the beginning of the experiments were used. Care and use of animals was carried out according to the European Union Council Directive (86/609/EEC). Animals were housed in a temperature-controlled environment with 12 hr light/dark cycles and access to food and water ad libitum. Under fluothane anesthesia, rats received two stereotaxic injections of 6-OHDA, one in left SN (pars compacta) and the other in the medial forebrain bundle (Ungersted 1971). Using a 10-μl Hamilton syringe, 4.0 μl of 6-OHDA (3.6 μg/μl in 0.2 mg/ml 1-ascorbate-saline) was injected into the left mesostriatal pathway at AP −5.3, ML +2.1, DV −7.8 (in mm with respect to bregma and dura, tooth bar −3.0 mm) and at AP −4.3; ML +1.4; DV −8.7, according to the stereotaxic atlas of Paxinos and Watson (1997). The injection rate was 1 μl/min, and the cannula was left in place for an additional 5 min before being slowly withdrawn.
To minimize variability due to the degradation of the toxin, the 6-OHDA solutions were freshly prepared, kept on ice, and protected from exposure to light.
Rotational Behavior
Apomorphine-induced rotational behavior was studied to determine whether the creation of the 6-OHDA lesion had been successful. Eight days postlesion, rats were placed in plastic rotometer bowls and connected to a mechanical counter. Counting of rotations began 5 min after the SC injection of apomorphine (0.5 mg/kg diluted in 0.9% saline), and rotational asymmetry was monitored over 15 min. Rats rotating at least 100 turns in 15 min were selected for intracerebral infusions; after infusion began, rotation was monitored once a week until the end of the study period.
Factor Infusion
Rats were stereotaxically implanted with a 28-gauge infusion cannula (brain infusion kit; Alzet, Cupertino, CA), connected to an osmotic minipump (model 2002; Alzet) via 5 cm of catheter tubing, for delivery of LGF, basic fibroblast growth factor (bFGF), and vehicle.
Pumps were designed for infusion over 15 days at a rate of 0.5 μl/hr. The solution consisted of rat albumin at 100 μg/ml in 0.9% saline in vehicle groups. LGF or bFGF, at a dose of 160 ng/day/rat, was added to the vehicle solution. The minipumps were filled with 200 μl of the corresponding solution and incubated overnight in normal saline at 37C. The 4.5-mm cannula attached to the minipump was stereotaxically implanted into the left striatum (AP +0.5; ML +3.5; DV −4.5) or left ventricle (AP −0.8, ML +1.4 DV −4.5) using bregma as a reference, and DV coordinates were calculated from the surface of the skull. The tooth bar was set at −3.0 mm. The minipump was placed into a subcutaneous pocket slightly posterior to the scapula. Six weeks after the unilateral 6-OHDA lesion, animals were divided according to the infused brain area and infusion solution as follows: IS-LGF infusion (n=14), IS-bFGF infusion (n=5), and IS-vehicle (n=11); ICV-LGF infusion (n=10), ICV bFGF infusion (n=5); and ICV-vehicle (n=9). Animals in the lesion-control group (n=9) did not undergo minipump implantation and were sacrificed 6 weeks postlesion.
To determine whether the intracerebral infusion of LGF increased the proliferation of new neurons in the striatum and the SN, animals were injected daily for 2 weeks with the mitotic marker bromodeoxyuridine (BrdU) (50 mg/kg IP), starting 7 days after the beginning of infusion.
Tissue Processing
At different time points after the creation of the 6-OHDA lesion (6 weeks postlesion, 48 hr after the start of infusion, and 2 weeks after the end of the infusion period), under deep anesthesia animals were perfused via ascending aorta with 50 ml of heparanized saline (5 U of heparin/ml of 0.9% NaCl) followed by 250 ml of 4% paraformaldehyde in 0.1 M phosphate buffer (PB), pH 7.4. Brains were removed, postfixed in the same solution for 24 hr at 4C, transferred to sequential washes in 10%, 20%, and 30% sucrose in PB, and frozen on dry ice before sectioning into 20-μm-thick coronal sections on a cryostat.
Antibodies and Immunochemicals
The following antibodies and final dilutions were used: mouse anti-TH (1:500; Chemicon International, Temecula, CA), rabbit anti-proliferating cell nuclear antigen (PCNA, 1:75; Santa Cruz Biotechnology, Santa Cruz, CA), rabbit anti-β-tubulin III (1:2000; BabCO, Richmond, CA), mouse anti-vimentin (1:200; Amersham Biosciences, Europe GmbH, Freiburg, Germany), mouse anti-nestin (clone Rat 401, 1:20; Developmental Studies Hybridoma Bank, University of Iowa, Ames, IA), peroxidase-labeled isolectin IB4 (Sigma Chemical Co; St Louis, MO), biotinylated goat anti-mouse IgG (Zymed Laboratories; South San Francisco, CA), streptavidin-biotin-peroxidase complex (DakoCytomation; Carpinteria, CA), diaminobenzidine (DAB) + substratechromogen system (DakoCytomation), Alexa Fluor-568 goat anti-mouse IgG and Alexa Fluor-488 goat anti-rabbit IgG (both from Molecular Probes; Eugene, OR).
Immunohistochemistry
Sections were mounted on silane-coated slides, treated with sodium acetate 10 mM, pH 6.0, at 95C for 5 min, and preincubated with 5% normal goat serum (NGS) in Tris-buffered saline (0.15 M NaCl and 0.1 M Tris HCl, pH 7.4)/0.1% Triton-X 100 for 30 min. Primary antibodies were applied for 24 hr at room temperature. Anti-nestin antibody was visualized by means of an immunoperoxidase procedure, using a biotinylated anti-mouse secondary antibody, followed by incubation in streptavidin-biotin-peroxidase complex. The reaction was detected with 3,3-DAB. Sections were dehydrated in ascending ethanol concentrations, cleared in xylene, and coverslipped with Depex (Serva Feinbiochemica GmbH; Heidelberg, Germany).
Anti-PCNA, -TH, -BrdU, and -vimentin antibodies were visualized by means of an immunofluorescence procedure, using appropriate fluorochrome-conjugated secondary antibodies directed against the species in which the primary antibody was raised. The slides were coverslipped in a medium containing p-phenylenediamine and bisbenzimide (Hoechst 33342; Sigma) for detection of nuclei.
Immunohistochemical Detection of Isolectin IB4
One series of sections was processed sequentially for immunohistochemical detection of PCNA, followed by microglia/macrophage visualization. After immunofluorescent visualization of PCNA, sections were again blocked with 5% NGS and incubated with peroxidase-conjugated isolectin IB4 for 2 hr. Finally, the reaction product was detected with DAB chromogen.
Morphometric Analysis
For quantitative estimation of TH-immunoreactive fibers and PCNA-positive or isolectin IB4-positive cells in the striatum, a computerized stereological analysis system was used (CAST-GRID; Olympus, Ballerup, Denmark). In each animal, measurements were performed in several coronal sections from two rostrocaudal levels through the striatum, the second level being centered at the infusion site, i.e., AP + 1.2; AP + 0.5 relative to bregma, according to the atlas used (Paxinos and Watson 1997). For quantitative estimation of TH-immunoreactive fibers, the cross-sectional area of the striatum was outlined and measured in each section, and the area occupied by TH-positive fibers was expressed as a percentage of the total striatal cross-sectional area. PCNA-positive and isolectin IB4-positive cells were expressed as positive cells/mm2. The number of TH-positive neurons in the SN was assessed by counting, using a x20 objective, all TH-positive neurons per section, in two sections 120 μm apart, at the level of bregma −5.3.
Statistical Analysis
The results represent the mean ± SEM from 5 to 10 individual animals. Statistical analyses were performed using ANOVA followed by the Bonferroni multiple comparison test, and the difference was considered significant when p≤0.05.
Results
Effects of LGF on TH-positive Innervation
To evaluate the degree of degeneration of the nigrostriatal DA projection prior to LGF infusion, a lesion-control group (n=9) was sacrificed 6 weeks postlesion. Immunohistochemical analysis of coronal sections at two different levels of the striatum demonstrated that striata from unilaterally 6-OHDA-lesioned rats contained no TH-positive neurons, and only 5.5 ± 1.9% of the structure corresponding to the ventral-most region of the striatum showed TH-positive innervation. IS infusion of LGF for 15 days in 6-OHDA-lesioned rats increased TH-positive innervation in the ventromedial and central regions of the striatum and around the site of the infusion cannula (Figures 1A-1F). The effect seen at the striatal level corresponding to the infusion site (Figure 2A) in the IS-LGF group differed significantly from that found in the vehicle and lesion-control animals and was not observed in animals receiving IS infusions of bFGF (Figure 2A). On the other hand, ICV infusion of LGF slightly increased TH-positive innervation in the striatum, but similar changes were observed in vehicle and ICV-bFGF-treated animals (Figure 2B).
Newly generated dopamine neurons may contribute to the observed increase in striatal TH-positive innervation. Double immunostaining showed very few β-tubulin III-positive/BrdU-positive cells in the ipsilateral and contralateral striatum of vehicle and LGF-infused animals (data not shown). However, the IS-LGF and IS-vehicle groups and the ICV-LGF-infused animals showed no TH-positive neurons in the striatum. In the SN, a few DA neurons remained in the 6-OHDA-lesioned side as compared with the contralateral SN (Figures 1G and 1H). IS infusion of LGF did not affect the total number of TH-positive neurons in the SN when compared with vehicle or lesion-control animals (Figure 2C). In addition, although a few BrdU-positive nuclei were observed in the SN, none of the TH-positive neurons incorporated BrdU in either the ipsilateral or contralateral side of the structure. Similarly, no TH-positive/BrdU-positive neurons were observed in the SN of ICV-LGF-infused animals.
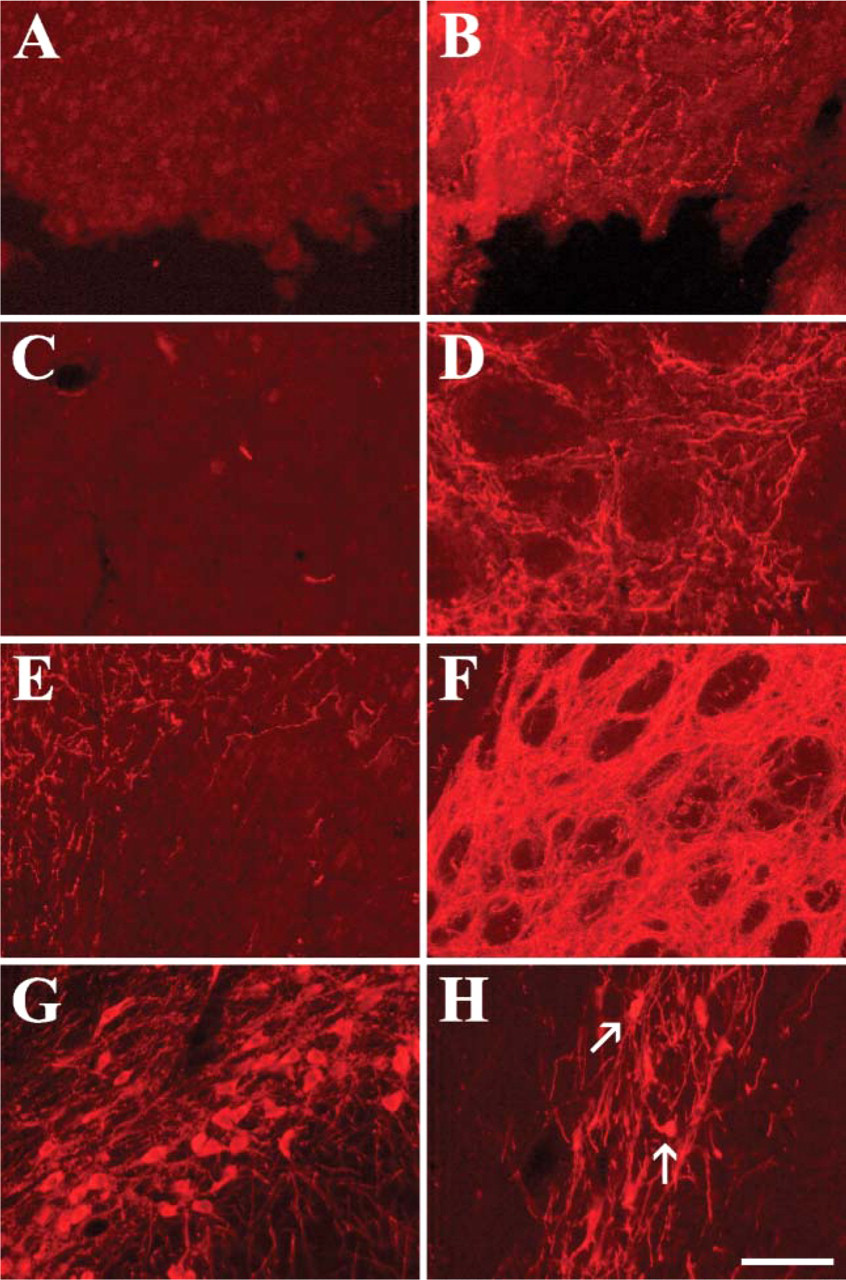
Intrastriatal (IS) infusion of the liver growth factor (LGF) induces tyrosine hydroxylase (TH) protein expression in the striatum of 6-hydroxydopamine (6-OHDA)-lesioned rats. Six weeks after creation of unilateral 6-OHDA lesions in the substantia nigra (SN), minipumps containing the vehicle solution
IS-LGF Infusion Stimulated Proliferation of Microglia in the Striatum
Enhanced vascularization (Casper et al. 2002) and/or glial proliferation (Silverman et al. 1999; Yasuhara et al. 2004) have been related to the survival and growth of dopamine neurons promoted by vascular endothelial growth factor (VEGF). To determine whether LGF promoted proliferation in 6-OHDA-lesioned rats, immunohistochemical detection of PCNA was performed after 48 hr of IS-LGF infusion. LGF produced a significant 1.6-fold increase in the number of proliferating (PCNA-positive) cells in the striatum ipsilateral to the infusion (Figure 3A). LGF also promoted a significant increase in the total number of cells expressing isolectin IB4 (Figure 3A), a vital marker of microglia and macrophages (Streit 1990; Boya et al. 1991). Moreover, a greater percentage of these cells showed the ameboid morphology of activated microglia and macrophages in IS-LGF-treated animals (Figures 3B and 3E). Interestingly, >60% of the isolectin IB4-positive cells double labeled with PCNA in the central and ventral regions of the striatum (Figures 3D and 3E).
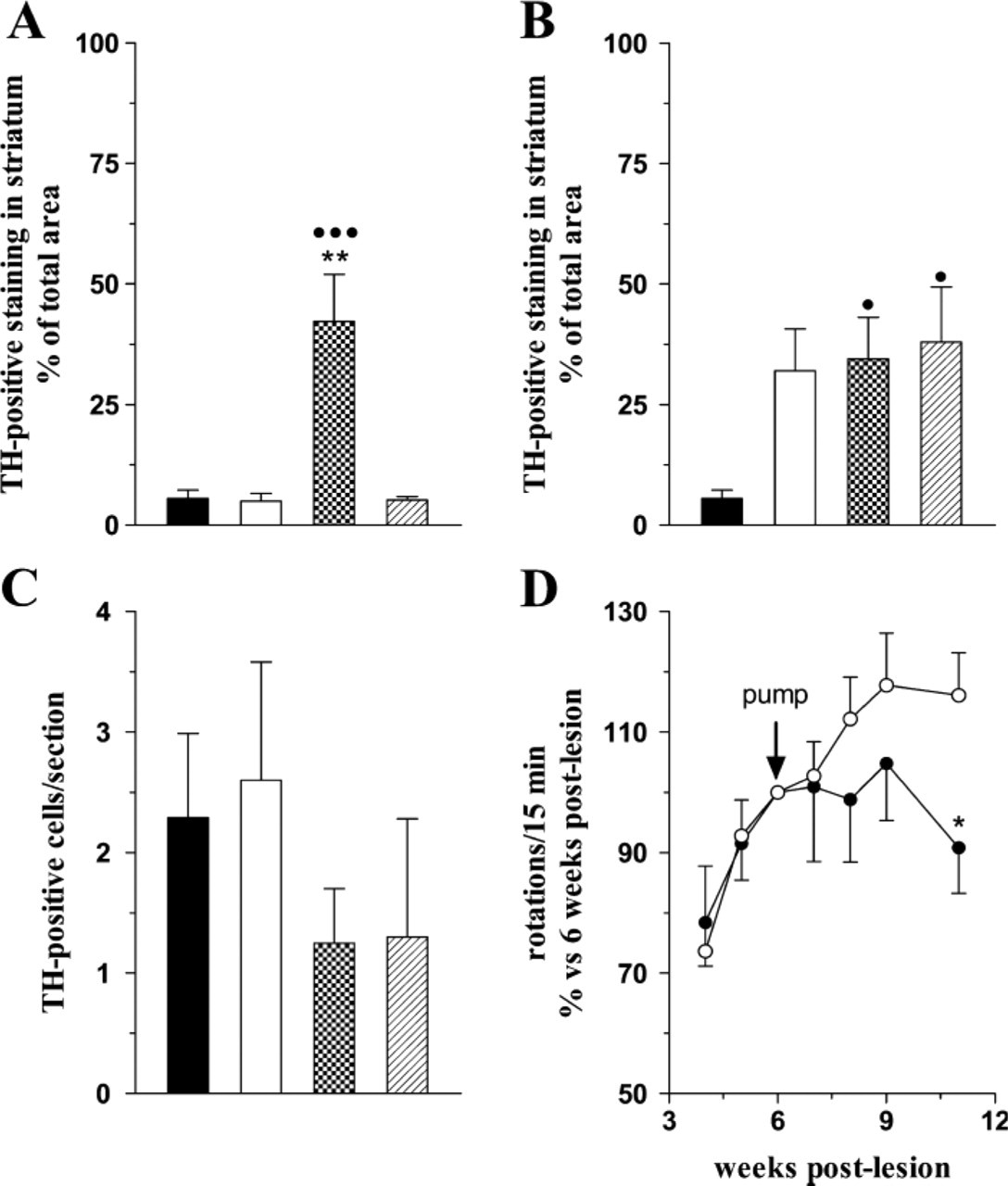
Effects of intracerebral infusion of the LGF on TH protein expression and apomorphine-induced rotations. Minipumps containing the vehicle solution (white bars), 160 ng/day/rat LGF (dot bars), or 160 ng/day/rat bFGF (line bars) were placed in the striatum (IS) or ventricle (ICV) of rats 6 weeks after creation of unilateral 6-hydroxydopamine lesions in the SN (lesion-control group, black bars), as described in Materials and Methods. One month later, animals were sacrificed and TH immunostaining was performed in the striatum
Other cell types were also proliferating around the infusion site in the striatum. Some PCNA-positive cells colabeled with nestin, a neurofilament expressed by reactive astrocytes (Figure 3C), and a few were vimentin positive (data not shown). In addition, PCNA-positive cells were located within the blood vessel walls and showed the typical flat morphology of endothelial cells (Figure 3C, Insert). However, no significant differences were observed between the IS-vehicle- and the IS-LGF-infused animals in the number of these cell types.
IS-LGF Infusion Partially Restored Motor Function
To determine whether intracerebral administration of LGF could restore the motor function of rats with unilateral 6-OHDA lesions, apomorphine-induced rotations were evaluated once a week before, during, and after the infusion period. As demonstrated in Figure 2D, 4 weeks after LGF infusion had begun, the IS-LGF group showed a slight but significant reduction in apomorphine-induced rotational behavior as compared with IS-vehicle animals. In contrast, none of the ICV-infused animals showed significant changes in apomorphine-induced rotations when compared with lesion-control animals (data not shown).
Discussion
In the present study we show for the first time that IS infusion of LGF significantly increases TH-positive innervation in the striatum of unilaterally 6-OHDA-lesioned rats. Moreover, IS-LGF treatment partially reduces apomorphine-induced rotational behavior in these animals. We also suggest that LGF stimulates the sprouting of dopamine terminals through the activation of microglia/macrophages in the striatum.
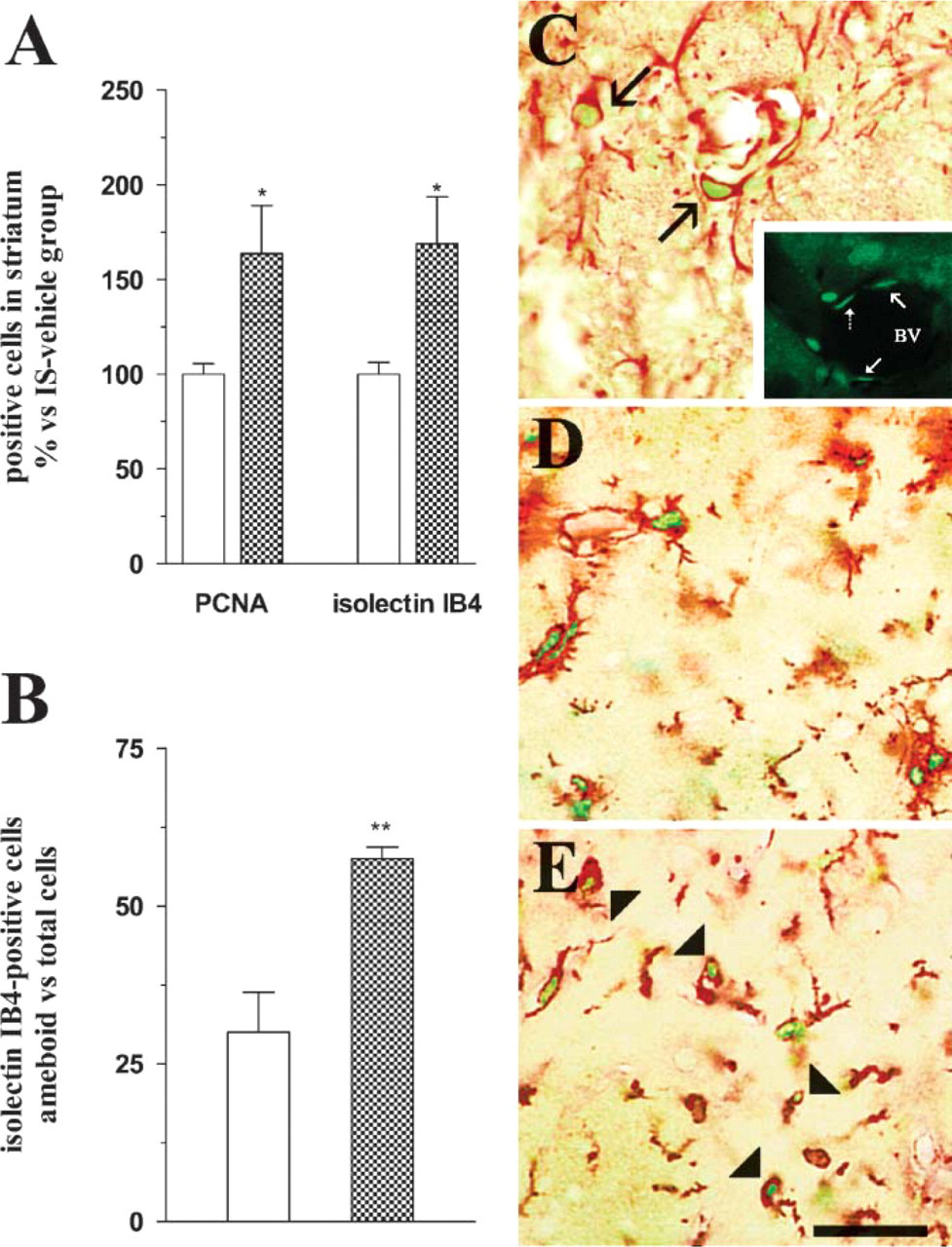
IS infusion of the LGF promotes the proliferation and activation of microglia in the striatum of 6-hydroxydopamine (6-OHDA)-lesioned rats. Six weeks after the creation of unilateral 6-OHDA lesions in the SN, minipumps containing the vehicle solution (white bars) or 160 ng/day/rat LGF (dot bars) were delivered to the striatum, as described in Materials and Methods. Forty-eight hr later, animals were sacrificed and striata were processed for immunohistochemical analysis. IS infusion of LGF significantly increased the percentage of proliferating (PCNA-positive) cells in the striatum ipsilateral to the 6-OHDA lesion
Many studies have reported that intracerebral administration of trophic factors increases neurogenesis in undamaged (Craig et al. 1996; Kuhn et al. 1997; Zigova et al. 1998; Pencea et al. 2001; Jin et al. 2002) and damaged (Fallon et al. 2000; Sun et al. 2003; Cooper and Isacson 2004) adult brains. Moreover, neurogenesis has recently been reported in the SN of adult 6-OHDA-denervated and naïve rats (Zhao et al. 2003). Newly generated DA neurons may account for the increase in DA innervation as measured by TH-positive immunostaining, observed after the infusion of LGF into the striatum of 6-OHDA-lesioned rats. Double immunostaining showed a few β-tubulin III-positive/BrdU-positive cells, but striata from the vehicle- and the LGF-infused rats did not present TH-positive neurons. Consequently, no new TH-positive neurons were observed in the striatum of these animals after 2 weeks of LGF infusion. In addition, no new DA neurons were found in the lesioned SN of any experimental group, indicating the absence of neurogenesis in that region.
The delivery of LGF to the striatum may protect nigral DA neurons from cell death, as has been reported for glial-derived neurotrophic factor (GDNF) and VEGF in 6-OHDA-lesioned rats (Kirik et al. 2001; Ogunshola et al. 2002; Yasuhara et al. 2004). In the case of VEGF, the latter authors demonstrated a significant preservation of both TH-positive neurons in the SN and TH-positive fibers in the striatum (Yasuhara et al. 2004). Although we cannot exclude a possible protective effect of LGF on DA neurons, our results strongly suggest that IS-LGF promotes the sprouting of DA terminals in the striatum. After the 6-OHDA lesions, a few DA neurons remained in the damaged lateral SN of the lesion-control group, and vehicle- and IS-LGF-infused animals showed approximately the same number of TH-positive neurons. On the other hand, in lesion-control- and IS-vehicle-infused animals, only 5% of the cross-sectional area of the damaged striatum showed TH-positive innervation, whereas the IS-LGF group exhibited TH-immunoreactivity in about 50% of the cross-sectional surface area. We have not analyzed dopamine metabolism, but the fact that IS-LGF animals showed a slight but significant reduction in apomorphine-induced rotational behavior suggests that LGF partially restores dopamine levels in the striatum. However, long-term experiments should be performed to determine whether IS-LGF is able to completely restore dopamine metabolism and motor behavior in 6-OHDA-lesioned rats.
Under our experimental conditions, IS-bFGF neither improved TH-positive innervation nor reduced apomorphine-induced rotational behavior. Other studies showed that IS-bFGF increases TH expression and activity in the striatum of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-lesioned mice (Otto and Unsicker 1990, 1993) and restores motor behavior and dopamine metabolite levels in MPTP hemiparkinsonian monkeys (Fontan et al. 2002). Perhaps bFGF is ineffective when administered once the toxin-induced lesion is totally established, as has been reported in MPTP-lesioned mice (Otto and Unsicker 1990, 1993). Moreover, the use of a different model of PD and/or differences in the administration of the factor (daily infusions vs FGF-soaked gel foam or weekly injections) might explain the lack of action of IS-bFGF in our study.
The ICV-vehicle group, as well as the ICV-LGF and ICV-bFGF animals, presented TH-positive fibers in ~30% of the striatal cross-sectional area. Most of the ICV-infused animals exhibited a slight enlargement of the infused lateral ventricles, a sign of hydrocephalus. Other studies in hydrocephalic rat brain have reported increased levels of growth-associated protein-43, a condition that appears to be permissive for axonal regeneration (Zhang and Del Bigio 1998). On the other hand, hydrocephalus affects the ependyma (Kiefer et al. 1998; Nojima et al. 1998), and ependymal cells express secretory proteins involved in several processes, such as the guidance of axonal growth cones (Bruni 1998).
An important issue was to determine the target cell and the molecular effectors that mediate LGF-induced dopamine fiber outgrowth in 6-OHDA-lesioned rats. As our results show, IS infusion of LGF significantly increased the number of PCNA-positive cells. About 60% of these cells were also positive for isolectin IB4 and some were positive for vimentin. Proliferation is part of the activation response of microglia, and vimentin expression serves as a marker for activated states of these cells (Graeber et al. 1988). Microglial activation also results in a morphological transformation from a ramified to an ameboid shape (Vilhardt 2005). The striatum of IS-LGF-infused animals also showed an increased number of isolectin IB4-positive cells with an ameboid, macrophage-like morphology. Activated microglia have been associated with the pathogenesis of several neurodegenerative diseases including PD (Teismann and Schulz 2004). Additionally, these cells may play a key role in the development and regeneration of the central nervous system through the release of trophic factors and extracellular matrix molecules (Polazzi and Contestabile 2002; Streit 2002). In this respect, the available evidence suggests that the secretion of GDNF and brain-derived neurotrophic factor by activated microglia and/or macrophages induces the sprouting of DA fibers in the injured striatum (Batchelor et al. 1999, 2002). Further experiments are warranted to determine whether LGF-activated microglia are able to synthesize and release these neurotrophins in 6-OHDA-lesioned rats.
LGF activity is mediated by the upregulation of TNFα mRNA and protein expression in rat liver (Diaz-Gil et al. 2003). Because microglia proliferate and undergo morphological changes characteristic of activation when stimulated with TNFα (Benveniste 1992; Benveniste and Benos 1995; Kauppinen and Swanson 2005), we may argue that this cytokine is responsible for the activation of microglial cells observed in 6-OHDA-lesioned rats. As occurs in liver (Diaz-Gil et al. 2003), TNFα could be produced by endothelial cells, the stimulation of which has been related to the survival and growth of DA neurons (Silverman et al. 1999; Casper et al. 2002; Yasuhara et al. 2004). Another possibility is that LGF directly stimulates microglia to produce TNFα. Microglia originate from the invasion of monocytes in early development (Ling and Wong 1993), and LGF induces TNFα release in human monocytes (Diaz Gil JJ, unpublished data). Moreover, activated microglia are able to synthesize and release TNFα (Chabot et al. 1997; Wang et al. 2003; Suzuki et al. 2004).
In summary, our study shows that IS infusion of LGF stimulates the sprouting of TH-positive terminals in the striatum of DA-denervated rats. DA sprouting seems to be mediated by microglia, which may be activated by the cytokine TNFα. Because apomorphine-induced rotational behavior was also reduced in these animals, we propose LGF as a novel factor that may be useful in the treatment of PD when delivered to the denervated striatum.
Footnotes
Acknowledgments
This work was funded by the Fondo de Investigaciones Sanitarias (FISS PI02/0853 and FISS PI03/0648) and Fundación MAPFRE MEDICINA. R.A. was the recipient of a Fundación MAPFRE MEDICINA fellowship.
We are grateful to Dr. Justo García de Yébenes (Servicio de Neurología, Hospital Ramón y Cajal, Madrid, Spain) for comments and critical reading of the manuscript. We also thank Mr. Santiago López and Miss Macarena Rodríguez for technical help and Mrs. Martha Messman for excellent correction of the manuscript.
