Abstract
Neural stem cells with self-renewal and multilineage potential persist in the subventricular zone of the adult mammalian forebrain. These cells remain relatively quiescent but, under certain conditions, can be stimulated, giving rise to new neurons. Liver growth factor (LGF) is a mitogen for liver cells that shows biological activity in extrahepatic sites and is useful for neuroregenerative therapies. The aim of this study was to investigate the potential neurogenic activity of LGF in the 6-hydroxydopamine rat model of Parkinson's disease. Proliferation was significantly increased in the subventricular zone and denervated striatum of rats receiving ICV LGF infusions, and 25% of the proliferating cells were doublecortin-positive neurons. Doublecortin-positive cells with the morphology of migrating neuroblasts were also observed in the dorsal and ventral regions of the striatum of LGF-infused animals. Moreover, some newly generated cells were neuronal nuclei-positive mature neurons. LGF also stimulated microglia and induced astrogliosis, both phenomena associated with generation and migration of new neurons in the adult brain. In summary, our study shows that LGF stimulates neurogenesis when applied intraventricularly in 6-hydroxydopamine–lesioned rats. Considering that this factor also promotes neuronal migration into damaged tissue, we propose LGF as a novel factor useful for neuronal replacement in neurodegenerative diseases.
P
Materials and Methods
LGF Purification
LGF was purified from serum of 5-week bile duct–ligated rats following a previously reported procedure (Díaz-Gil et al. 1994). LGF was quantitated by HPLC (Singh and Bowers 1986), and samples with the highest serum LGF concentrations were selected to proceed with the purification process, which involved three chromatography steps using Sephadex G-150, DEAE-cellulose, and hydroxylapatite. Purity, that is, the absence of other growth factors and/or contaminants in the LGF preparation, was also assessed according to standard criteria (Díaz-Gil et al. 1986–1988,1994). All LGF preparations showed a single band in SDS-PAGE. LGF preparations were lyophilized and kept at 4C until use, at which time aliquots were dissolved in saline for pump perfusion.
Animals and 6-OHDA Lesion Surgery
A total of 55 female Sprague-Dawley rats weighing 220–250 g were used according to the European Union Council Directive (86/609/EEC). Under Fluothane anesthesia, rats received two stereotaxic injections of 6-OHDA: one in left substantia nigra (SN) and the other in the medial forebrain bundle (Ungersted 1971). Using a 10-μl Hamilton syringe, 4.0 μl of 6-OHDA (3.6 μg/μl in 0.2 mg/ml l-ascorbate saline) was injected into the left mesostriatal pathway at AP −5.3; ML +2.1; DV −7.8 (in millimeters with respect to bregma and dura; tooth bar at −3.0 mm) and at AP −4.3; ML +1.4; DV −8.7, according to the stereotaxic atlas of Paxinos and Watson (1997). The injection rate was 1 μl/min.
Rotational Behavior
Ten days postlesion, rotational asymmetry was monitored over 15 min after SC injection of apomorphine (0.5 mg/kg diluted in 0.9% saline). Rats rotating at least 100 turns in 15 min were selected for intracerebral infusions. After initiation of infusion, rotation was monitored once a week until the end of the study period.
LGF Infusion
Six weeks later, the unilaterally 6-OHDA–lesioned rats underwent stereotaxic implantation of a 28-gauge infusion cannula (brain infusion kit; Alzet, Cupertino, CA) connected to an osmotic minipump (model 2002; Alzet). The 4.5-mm cannula was stereotaxically implanted into the left ventricle at AP −0.8; ML +1.4; DV −4.5, in millimeters with respect to bregma and dura. Infusion lasted 15 days, at a rate of 0.5 μl/hr. In the ICV-vehicle group (n = 18), the solution consisted of 100 μg/ml rat albumin in 0.9% saline, and in the ICV-LGF group (n = 13), LGF was added to the vehicle solution at a dose of 160 ng/day/rat. Starting 24 hr after the beginning of infusion, the animals were injected daily for 2 weeks with 5-bromodeoxyuridine (BrdU; 50 mg/kg, IP). Animals in the lesion-control group (n = 9) did not undergo minipump implantation and were sacrificed at 6 weeks postlesion.
Tissue Processing
At different time points after the production of the 6-OHDA lesion (6 weeks postlesion and 7 or 15 days after the start of infusion), under deep anesthesia, the animals were perfused through the ascending aorta with 50 ml of isotonic saline, followed by 250 ml of 4% paraformaldehyde. Brains were postfixed in the same solution for 24 hr at 4C, cryoprotected, and frozen before sectioning into 20-μm-thick coronal sections on a cryostat.
Antibodies and Immunochemicals
The following antibodies and final dilutions were used: mouse anti-tyrosine hydroxylase (TH, 1:500; Chemicon International, Temecula, CA), rabbit anti-proliferating cell nuclear antigen (PCNA, 1:75; Santa Cruz Biotechnology, Santa Cruz, CA), mouse anti-5-BrdU (1:25; DakoCytomation, Glostrup, Denmark), rat anti-BrdU (1:25; AbD Serotec, Oxford, UK), rabbit anti-β-tubulin III (1:500; BabCO, Richmond, CA), rabbit anti-glial fibrillary acidic protein (GFAP, 1:200; DakoCytomation, Glostrup), mouse anti-neuronal nuclei (NeuN, 1:1000; Chemicon International), guinea pig anti-doublecortin (Dcx, 1:3000; Chemicon International), rabbit anti-cow S-100 (undiluted; DakoCytomation, Carpinteria, CA), mouse anti-nestin (clone rat 401, 1:20; Developmental Studies Hybridoma Bank, University of Iowa, Ames, IA), peroxidase-labeled isolectin IB4 (Sigma Chemical; St. Louis, MO), biotinylated goat anti-mouse IgG (Zymed Laboratories; South San Francisco, CA), streptavidin–biotin–peroxidase complex and DAB 1 substrate–chromogen system (both from DakoCytomation, Carpinteria), Alexa Fluor-568 goat anti-mouse IgG, Alexa Fluor-488 donkey anti-rat IgG, and Alexa Fluor-488 goat anti rabbit IgG (all from Molecular Probes; Eugene, OR), and fluorescein-conjugated goat antimouse IgG and Cy2- or Cy3- conjugated donkey anti-guinea pig IgG (all three from Jackson ImmunoResearch Laboratories; West Grove, PA).
IHC and Detection of Isolectin IB4
Coronal sections obtained from the infusion site (AP −0.8), preoptic hypothalamic region (AP −1.4), and sustantia nigra (AP −5.3) of each brain were mounted on silane-coated slides, treated with 10 mM sodium citrate, pH 6.0, at 95C for 5 min, and blocked with 5% normal goat serum. Primary antibodies were applied for 24 hr at room temperature, and most of them were visualized using immunofluorescence procedures. In some cases, anti-nestin antibody was visualized by means of an immunoperoxidase technique. For double immunolabeling with BrdU, neural cell markers were detected before BrdU immunostaining. For detection of incorporated BrdU, sections were incubated in 2 N HCl at 37C for 30 min and rinsed in Tris-buffered saline.
One series of sections was processed sequentially for immunofluorescent detection of BrdU, followed by a peroxidase method for histochemical detection of isolectin IB4, a marker of microglia and macrophages.
Morphometric Analysis
For quantitative estimation of BrdU-, neural antigen-, and isolectin IB4-immunoreactive cells, a systematic random sampling of 25 fields of view (objectives X20 or X40) was carried out under fluorescence microscopy with the aid of the Computer Assisted Stereology Toolbox (CAST) grid system (Olympus; Ballerup, Denmark). In each animal, measurements were performed (a) in the SVZ and striatum of two or three representative sections, 1 mm apart, at the infusion site, and (b) around the third ventricle (3rd V) in two sections, 1 mm apart, in the hypothalamic preoptic region.
Quantitative assessment of TH-immunoreactive fibers in striata and dopaminergic neurons in SN was performed as previously described (Reimers et al. 2006).
Statistical Analysis
The results represent the mean ± SEM from four to nine individual animals. Statistical analyses were performed using ANOVA, followed by the Bonferroni multiple comparison test, and the difference was considered significant when p ≤ 0.05.
Results
ICV Infusion of LGF Induces Proliferation and Nestin Re-expression in the SVZ of 6-OHDA–lesioned Rats Intracerebral infusion of trophic factors stimulated the proliferation of neural precursors located in the SVZ of the lateral ventricles of rodents. PCNA is a nuclear protein expressed in the early G0/G1 and S phases of the cell cycle. Unilaterally, 6-OHDA–lesioned rats showed PCNA immunoreactivity in the dorsal and central regions of the SVZ (Figures 1A and 1D), whereas nearly no PCNA-positive cells were found at the most ventral level (Figure 1G). ICV infusion of LGF over 7 days increased the number of PCNA-positive cells throughout the infused SVZ (Figures 1C, 1F, and 1I). PCNA-positive cells were also observed within the striatal and septal parenchyma of LGF-treated rats (Figures 1F and 1I). Animals receiving ICV infusion of vehicle showed increased proliferation in the dorsal and central regions of the SVZ (Figures 1B and 1E), but the effect was significantly less marked than in the ICV-LGF group.
Nestin is an intermediate filament protein used as a biological marker to identify neural progenitor cells (Lendahl et al. 1990; Ernst and Christie 2005). Nestin immunoreactivity was localized in the SVZ of 6-OHDA–lesioned rats (Figures 1A, 1D, and 1G). ICV infusion of vehicle for 7 days raised nestin expression in the dorsal and central regions of the SVZ (Figure 1B), whereas no significant changes were observed in the most ventral region of the SVZ (Figures 1E and 1H). In LGF-treated rats, nestin immunolabeling was significantly increased throughout the SVZ (Figures 1C, 1F, and 1I). In addition, LGF expanded the population of nestin-positive cells within the striatal parenchyma, where a certain number of cells coexpressed nestin and PCNA (Figure 1F, inset).
Nestin and PCNA expressions were also analyzed in the SVZ of naïve rats receiving ICV infusion of vehicle or LGF. PCNA immunoreactivity was similar in the infused and contralateral sides of ICV-vehicle–infused animals. However, ICV-LGF infusion in naïve rats slightly increased proliferation in the infused side in three of seven animals analyzed independently (data not shown). Because the SVZ, striatum, and septum of these three animals were damaged, we could argue that the proliferative effect observed was probably caused by the insertion of the cannula. On the other hand, neither the ICV-vehicle–treated animals nor the ICV-LGF–infused animals showed increased nestin immunoreactivity in the SVZ or nestin-positive cells invading the striatal parenchyma (data not shown). Taken together, these results indicate that, in the absence of lesion, LGF is unable to stimulate the proliferation of nestin-expressing cells and their migration from the SVZ.
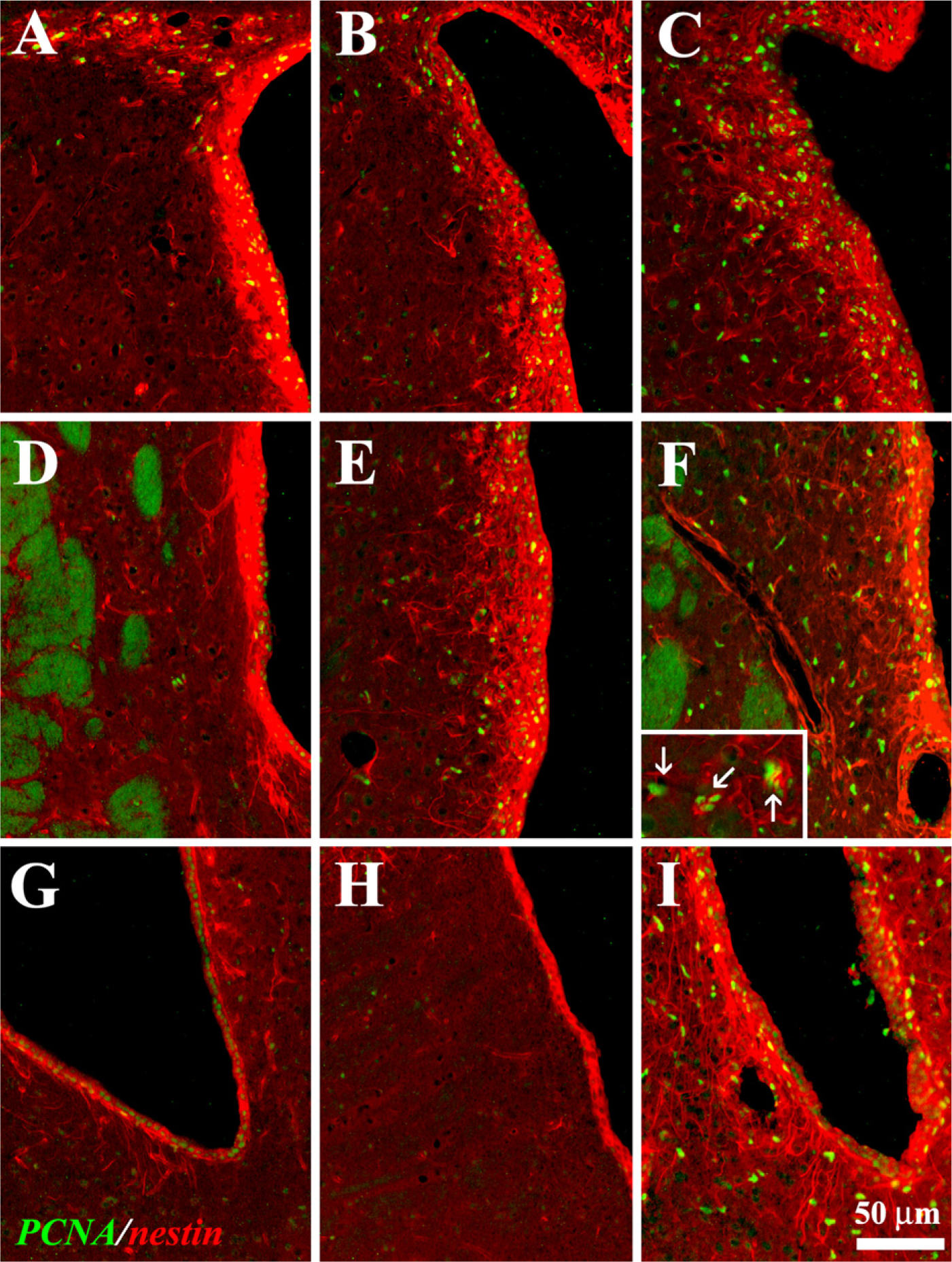
Liver growth factor (LGF) stimulates proliferation and nestin expression in 6-hydroxydopamine (6-OHDA)–lesioned rats. The subventricular zone (SVZ) and dopamine-denervated striatum of hemi-parkinsonian rats receiving LGF infusions (
LGF Promotes the Proliferation of Neuroblasts and Their Migration Into the Striatum of 6-OHDA–lesioned Rats
Neural precursors from the SVZ give rise to a population of new neurons, the final destination of which is the olfactory bulb. Doublecortin (Dcx) is a microtubule-associated protein expressed by migrating neuroblasts. The anti-Dcx antibody labeled clusters of cells in the lateral dorsal horn and SVZ of unilaterally 6-OHDA–lesioned rats (Figure 2A). Most of these Dcx-positive cells were double labeled with anti-nestin (Figure 3A), indicating that they corresponded to the neurogenic cell population of neural progenitors located in the SVZ of the lateral ventricles (Yang et al. 2004). The administration of vehicle into the lateral ventricle of unilaterally 6-OHDA–lesioned rats did not modify the number [27 ± 3.7 and 30 ± 6.2 Dcx-positive cells/mm2 (n = 5) in the non-infused and vehicle-infused side, respectively] or distribution of Dcx-positive cells, analyzed at the level of the cannula. However, ICV infusion of LGF for 15 days increased the number of Dcx-positive cells 1.63 ± 0.29-fold (n = 5) in the infused side and affected their distribution. In general, Dcx-positive cells were observed in the dorsal region of the striatum of LGF-infused animals (Figure 2D). However, some animals showed a significant number of Dcx-positive cells invading the dorsal (Figure 2C1) and ventral (Figure 2C2) regions of the striatal parenchyma. In animals receiving LGF infusion, Dcx-positive cells coexpressed neither nestin (Figure 3B) nor GFAP (Figure 3G), and they showed the morphology of migrating cells with long leading processes. Besides the striatum, Dcx immuno-reactivity was also observed protruding into the lateral ventricles of ICV-LGF–infused rats (Figure 2B).
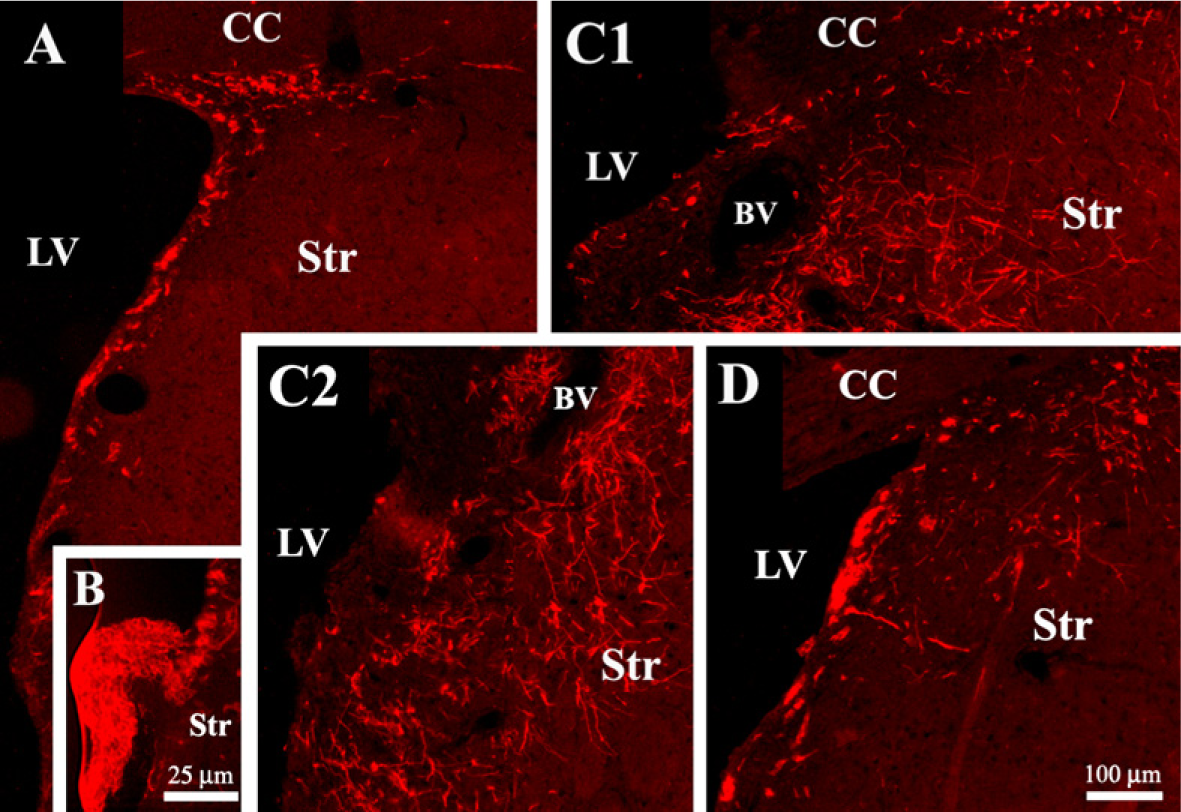
Neurogenic activity of LGF in 6-OHDA–lesioned rats.
To determine whether Dcx-positive cells were newly generated during LGF infusion, some animals received BrdU (50 mg/kg/day, IP) for 15 days. At the end of the treatment, the animals were sacrificed, and double immunolabeling experiments for BrdU and neuronal antigens were performed. IHC analysis showed a significant increase in the total number of BrdU-positive cells in the SVZ and striatum of LGF-treated rats compared with the ICV-vehicle group (Figure 4A). Double immunostaining experiments showed that 25 ± 5.6% (n = 7) of BrdU-positive cells in the striatal parenchyma were immunopositive for Dcx. Most of these newborn neurons showed a round morphology and were located near the SVZ and blood vessels (Figure 3C). In addition, Dcx-/BrdU-positive cells showing the morphology of migrating neuroblasts were also observed in the denervated striatum of ICV-LGF–infused animals (Figure 3C, inset). NeuN is a nuclear antigen expressed by mature neurons. Interestingly, 31 ± 6% (n = 6) of BrdU-positive nuclei costained with anti-NeuN (Figures 3D and 3E), indicating that newly generated neurons may differentiate into a mature phenotype in the striatum of 6-OHDA–lesioned rats.
Another population of cells that proliferated during LGF infusion was that composed of GFAP-positive astrocytes, which represented 24 ± 2% (n = 10) of the total population of BrdU-positive cells in the striatum (Figure 3H). GFAP immunostaining showed the presence of astrocytes with slender processes in the striatum of ICV-vehicle–infused animals (Figure 3F). In contrast, GFAP-positive cells in the striatal parenchyma of 6-OHDA–lesioned rats receiving ICV-LGF infusion showed cell body hypertrophy (Figure 3G). These results strongly suggest that ICV infusion of LGF also promotes glial reactivity in 6-OHDA–lesioned animals. In addition to neurons and astrocytes, 22 ± 2% (n = 7) of the cells in the striatum of ICV-LGF–infused animals were BrdU-positive cells that were immunopositive for nestin (Figure 5B). Approximately 5% of these cells were located in the blood vessels and showed the morphology of endothelial cells (Figure 5B, inset).
LGF Stimulates the Proliferation of Microglia in the Lateral and Third Ventricles of 6-OHDA–lesioned Rats
As shown in Figure 5, cushion-like thickenings of BrdU-positive cells were observed protruding into the lateral ventricles of LGF-infused rats. These cells were separated from the striatal parenchyma by a layer of ependymal cells that were immunopositive for S-100 and GFAP (Figures 5D and 5E). Although Dcx and GFAP immunoreactivity was detected within these growing structures, double immunostaining experiments showed that only a few BrdU-positive cells costained with anti-Dcx (Figure 5C) or anti-GFAP (Figure 5E). Moreover, none of these BrdU-positive cells were double labeled with anti-nestin (Figure 5B) or anti-S-100 (Figure 5D). We reported previously that intrastriatal delivery of LGF activates microglia in the striatum of 6-OHDA–lesioned rats (Reimers et al. 2006). Because the SVZ contains microglia, these cells could be stimulated during ICV-LGF infusion. As shown in Figure 5F, rounded cells that were immunoreactive for the specific marker of microglia and macrophages, isolectin-IB4 (Boya et al. 1991), were present in the proliferating structures of the ICV-LGF–infused animals. This cell population was double labeled with anti-BrdU and represented as much as 30% of the total BrdU-positive cells within these structures (Figure 4C).
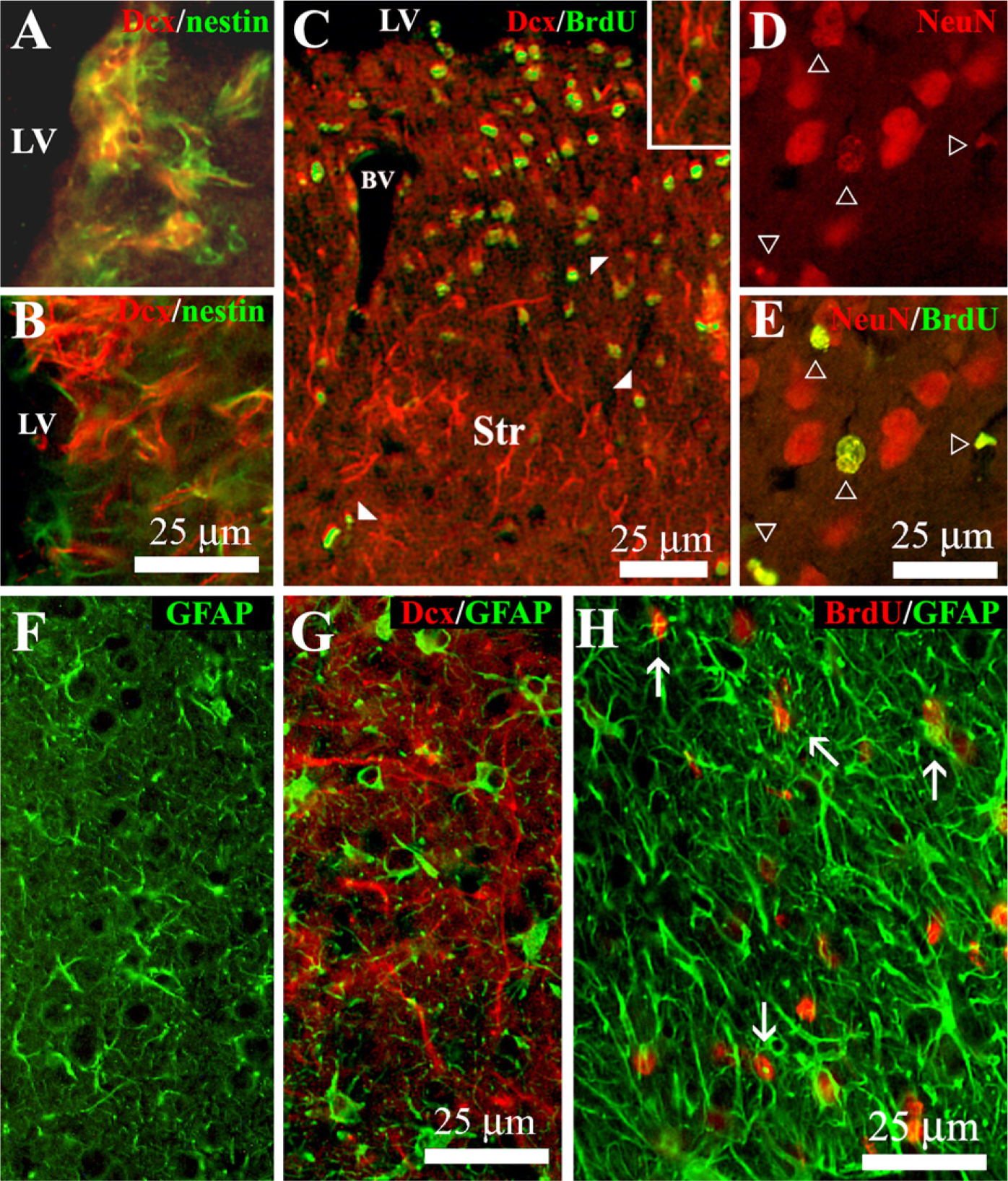
LGF promotes neuroblast proliferation and glial reactivity in 6-OHDA–lesioned rats.
LGF also stimulates the proliferation of microglia in the third ventricle. As shown in Figure 4B, a significant increase in BrdU incorporation was observed on both sides of the lateral wall of the third ventricle of ICV-LGF–infused animals compared with the ICV-vehicle group (Figure 3B). Moreover, double immunostaining experiments showed that >40% of these BrdU-positive cells colabeled with rounded isolectin IB4-positive cells (Figures 4C and 5G).
Effects of LGF on Tyrosine Hydroxylase–positive Innervation
In a previous study, we showed that intrastriatal infusion of LGF increases TH immunolabeling in the striatum of 6-OHDA–lesioned rats (Reimers et al. 2006). To evaluate the degree of degeneration of the nigrostriatal DA projection before LGF infusion, a lesion-control group (n = 9) was sacrificed 6 weeks postlesion. IHC analysis of coronal sections at two different levels of the striatum showed that striata from unilaterally 6-OHDA–lesioned rats contained no TH-positive neurons, and only 5.5 ± 1.9% of the structure corresponding to the ventral-most region of the striatum showed TH-positive innervation. ICV infusion of LGF for 7 or 15 days did not affect this parameter because only 8 ± 2% of the striatum showed TH-positive innervation. In addition, no TH-positive or TH-/BrdU-positive neurons were observed in the striatal parenchyma of LGF-infused animals at these time points. On the other hand, no changes in the total number of TH-positive neurons were detected in the SN of ICV-LGF–infused animals compared with ICV-vehicle rats.
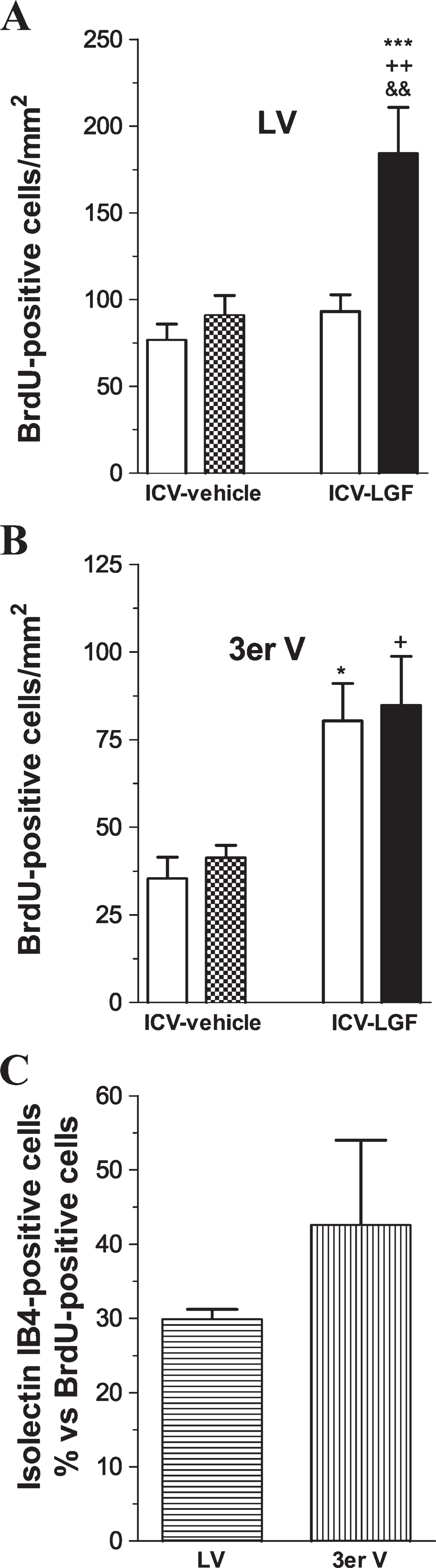
LGF increases BrdU incorporation in 6-OHDA–lesioned rats. ICV infusion of LGF (ICV-LGF) significantly increased the total number of BrdU-positive cells in the SVZ and denervated striatum ipsilateral to LGF infusion (
To determine whether ICV administration of LGF could restore the motor function of rats with unilateral 6-OHDA lesions, apomorphine-induced rotations were evaluated once a week during the infusion period. As we reported previously, at 6 weeks postlesion, apomorphine-induced rotational behavior reached a plateau that was maintained for several weeks (Reimers et al. 2006). Animals receiving ICV-LGF infusion did not show any significant change in apomorphine-induced rotations during the period of infusion (15 days of LGF) compared with the lesion-control group or with ICV-vehicle–infused rats (data not shown). These results indicate that LGF neither restored motor behavior nor impaired PD symptoms during the period of treatment analyzed in this study.
Discussion
In this study, we showed that ICV infusion of LGF stimulates the proliferation of SVZ neuronal precursors and their invasion of the denervated striatum of 6-OHDA–lesioned rats. In addition, LGF activates microglia and induces astrogliosis, two phenomena that have been associated with the generation of new neurons and their migration into the adult brain.
NSCs with self-renewal and multilineage potential persist in the SVZ of the mammalian adult forebrain. In vivo administration of growth factors (i.e., epidermal growth factor, tumor growth factor-α, or VEGF) and neurotrophins increased their expansion (Craig et al. 1996; Kuhn et al. 1997; Schänzer et al. 2004) and was effective in promoting neurogenesis in the healthy (Palmer et al. 1999; Pencea et al. 2001; Jin et al. 2002) and damaged brain (Fallon et al. 2000; Teramoto et al. 2003; Cooper and Isacson 2004; Mohapel et al. 2005; Wang et al. 2007). Our preliminary studies suggested that LGF could stimulate the proliferation of multipotent progenitor cells localized in the SVZ of the lateral ventricles of 6-OHDA–lesioned rats (Bazán et al. 2005). These data confirmed our previous observations because LGF increased PCNA and nestin immunoreactivity in the SVZ surrounding the infused lateral ventricle. Other studies have shown that the insertion of a canula into the lateral ventricle increases the proliferation of SVZ cells (Weinstein et al. 1996), suggesting that proliferation might be induced by mechanical disruption of the SVZ. As we showed here, ICV infusion of vehicle also increased PCNA immunostaining in the SVZ of hemiparkinsonian rats. However, the level of proliferation was lower than that detected after LGF infusion. Under our experimental conditions, ICV infusion of LGF also stimulated nestin expression in the SVZ of 6-OHDA–lesioned rats. Because nestin and PCNA colabeling was observed, our results clearly indicate that LGF stimulates the proliferation of neural precursors located in the SVZ of 6-OHDA–lesioned rats. On the other hand, doublets and triplets of nestin-/PCNA-positive cells were also visualized in the striatal parenchyma, suggesting that LGF also stimulates the migration of proliferating neural precursors into the denervated striatum of 6-OHDA–lesioned rats.
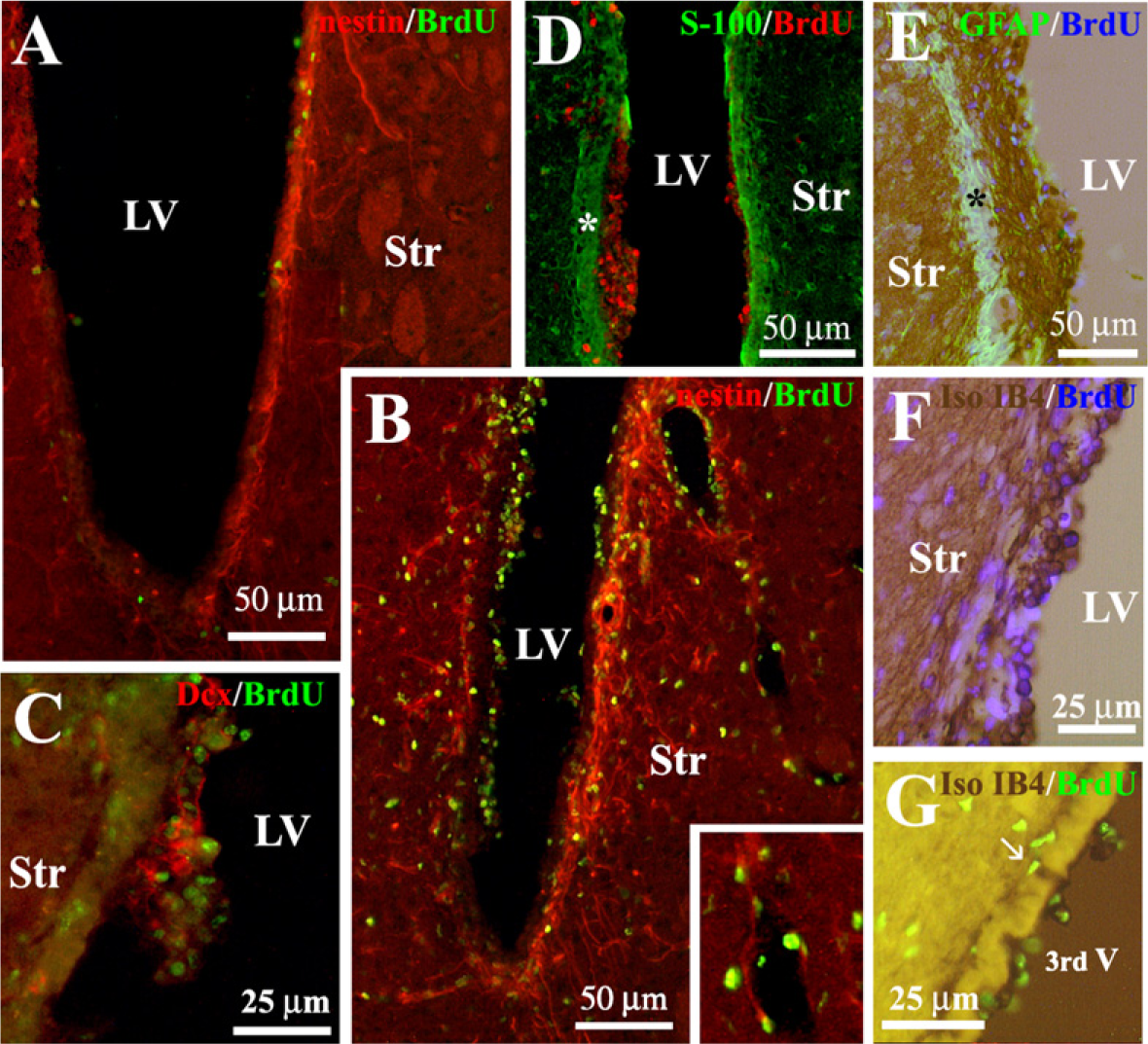
LGF stimulates the proliferation of microglia inside the lumen of the lateral and third ventricles of 6-OHDA–lesioned rats. As shown in
Neural precursors from the SVZ give rise to a population of Dcx-positive neurons, the final destination of which is the olfactory bulb (Yang et al. 2004; Ernst and Christie 2005). As these results showed, BrdU incorporation was significantly increased in the striatum of LGF-infused animals, and double immunostaining experiments showed that >20% of the BrdU-positive cells colabeled with Dcx expressing cells. The majority of the newborn neurons in the striatal parenchyma showed a round morphology and were located near the SVZ, suggesting that the neurogenic pool of neural precursors divides and their progeny migrate into the neighboring parenchyma after ICV infusion of LGF. Dcx-positive cells showing long leading processes were also found invading the denervated striatum of ICV-LGF–infused animals. Although only a few of these Dcx-positive cells coexpressed BrdU, we can argue that they were newly generated during infusion of the factor, because Dcx expression has been considered a marker for neurogenesis (Couillard-Despres et al. 2005), and BrdU incorporation only provides a relative estimation of the number of proliferating cells for different reasons such as the length of the S-phase, which is a fraction of the entire cell cycle, the number of cells dividing when BrdU is not present for their labeling, and the survival of newly generated cells, because cell death is a normally occurring process in neurogenic regions (reviewed in Taupin 2007). The absence of colocalization with nestin or GFAP confirms that Dcx-positive cells invading the striatum are not multipotent neural precursors or GFAP-positive astrocytes. However, the morphology of most of these Dcx-expressing cells, in combination with the lack of nestin and GFAP expression, clearly indicates that they represent a population of migrating neuroblasts. We should mention that Dcx immuno-reactivity was not detected in the striatum of ICV-vehicle–infused animals or when vehicle or LGF were infused in the left lateral ventricle of naïve animals (our unpublished data). Taken together, these results suggest that LGF confers on the denervated striatum some properties that allow the migration of the newborn neurons.
The striatum of ICV-LGF–infused rats also showed BrdU-positive cells that colabeled with anti-NeuN, a nuclear antigen expressed by mature neurons. As proposed in other studies, new neurons in the parenchyma could be generated from multipotent progenitor cells that reside in parenchymal structures of the adult fore-brain (Laywell et al. 2000; McGinn et al. 2004). Although we cannot exclude that possibility, our previous studies do not support this hypothesis because the intrastriatal delivery of LGF did not promote the generation of new neurons in the striatal parenchyma (Reimers et al. 2006). On the other hand, the fact that these newly generated neurons showed lower NeuN immunoreactivity than adult striatal neurons (Figure 3D) may suggest that they probably represent a population of maturing neurons derived from the migrating neuroblasts mentioned above.
The generation of new neurons in the denervated striatum of 6-OHDA–lesioned rats is of therapeutic interest. However, no BrdU-/TH-positive neurons or behavioral improvement was observed 7 or 17 days after the start of LGF infusion. Because rats receiving intrastriatal-LGF infusion showed higher TH immunoreactivity and improved rotational behavior 1 month after the end of the infusion (Reimers et al. 2006), a longer period postinfusion might be needed to determine whether newborn neurons could acquire a dopaminergic phenotype capable of improving function in 6-OHDA–lesioned rats. Besides, newly generated neurons may require additional midbrain or forebrain cues to differentiate into mature dopamine neurons, as recently suggested for the intracerebral infusion of TGF-α (de Chevigny et al. 2008).
An important issue was to determine the target cell that mediates LGF-induced neurogenesis and neuronal migration in 6-OHDA–lesioned rats. Our previous studies indicated that LGF is devoid of any proliferating or differentiating effect in cultures of NSCs (Bazán et al. 2005). Recently, we suggested that LGF stimulates axonal growth in 6-OHDA–lesioned rats through the activation of microglial cells (Reimers et al. 2006). Activated microglia have been associated with the pathogenesis of several neurodegenerative diseases including PD (Teismann and Schulz 2004). Additionally, these cells may play a key role in neurogenesis (Walton et al. 2006; Ziv et al. 2006; Ninkovic and Götz 2007) and can direct the migration and glial differentiation of neural precursors (Aarum et al. 2003; Butovsky et al. 2006; Nakanishi et al. 2007). As has been reported for other trophic factors (Kuhn et al. 1997; Pencea et al. 2001; Cooper and Isacson 2004), cushion-like thickenings of BrdU-positive cells were observed in the lateral ventricles of LGF-infused rats. Although some of these newly generated cells colabeled with Dcx or GFAP, many of them immunostained for isolectin IB4. Proliferation is part of the activation response of microglia (Graeber et al. 1988). Activation of microglia also results in a morphological transformation from a ramified to a macrophage-like morphology similar to that found in our study (Vilhardt 2005). Under our experimental conditions, the insertion of a canula into the lateral ventricle could be partially responsible for the activation of microglia, but we found no activated isolectin IB4-positive cells in the lateral ventricles of ICV-vehicle infused animals. Moreover, because our experimental model of PD does not disrupt the brain at the level of the third ventricle, the presence of rounded proliferating microglia surrounding this structure after ICV-LGF infusion strongly suggests that this cell type is a target for LGF in 6-OHDA–lesioned rats.
We should point out that LGF activity is mediated by the upregulation of TNF-α, mRNA, and protein expression in rat liver (Díaz-Gil et al. 2003). Interestingly, activated microglia are able to synthesize and release TNF-α (Wang et al. 2003; Suzuki et al. 2004), a cytokine involved in the proliferation, migration, and differentiation of neural precursors (Wu et al. 2000; Ben-Hur et al. 2003; Katakowski et al. 2007). Because microglia originate from the invasion of monocytes in early development (Ling and Wong 1993) and LGF induces TNF-α release in cultured human monocytes (Díaz-Gil JJ, unpublished data), we can argue that this cytokine could be the molecular effector that mediates LGF-induced neurogenesis in our experimental model of PD. Further experiments are warranted to determine whether LGF-activated microglia are able to synthesize and release TNF-α in 6-OHDA–lesioned rats.
Emerging evidence suggests that astrocytes play a key role in controlling multiple steps of adult neurogenesis (Ma et al. 2005; Mori et al. 2005; Barkho et al. 2006). On the other hand, neuroblast migration occurs in association with reactive astrocytes and blood vessels in rodents (Teramoto et al. 2003; Ohab et al. 2006; Thored et al. 2006; Yamashita et al. 2006; Bovetti et al. 2007). In the striatum of ICV-LGF–infused animals, most of the Dcx-positive cells with the morphology of migrating neuroblasts were not associated with blood vessels (Figure 3C). However, these cells lay on a bed of astrocytes that proliferated during the factor infusion and showed cell body hypertrophy, two features of glial reactivity. The association of astrocytes with migrating neuroblasts was only observed when neuroblasts migrated into the damaged striatal tissue (Thored et al. 2006; Yamashita et al. 2006). Because the denervated striatum is a damaged tissue, our results suggest that reactive astrocytes might be responsible for neuronal migration in our experimental model of PD. Interestingly, a recent study suggested that the release of TNF-α by activated microglia stimulates the synthesis of monocyte chemoattractant protein-1 in astrocytoma cells (Widera et al. 2004), a chemokine that plays a critical role in neuroblast migration after focal cerebral ischemia (Yan et al. 2007).
Besides microglia and astrocytes, other cell types could mediate LGF-induced neurogenesis and neuronal migration in 6-OHDA–lesioned rats. The first targets of LGF in liver are portal vein endothelial cells (Díaz-Gil et al. 2003). Endothelial cells stimulate the self-renewal of NSCs, as well as neuronal differentiation of neural progenitors through the production of soluble factors and direct contact (Palmer et al. 2000; Jin et al. 2002; Doetsch 2003; Shen et al. 2004; Li et al. 2006; Gama Sosa et al. 2007; Teng et al. 2008; Guo et al. 2008). In the striatum of ICV-LGF–infused animals, ∼20% of the BrdU-positive cells were located near the blood vessels, but only 5% of these cells showed the morphology of endothelial cells, suggesting that this cell type is not the main target for LGF in our experimental model of PD. Nevertheless, on the basis of our study, we cannot exclude the possibility that LGF could stimulate endothelial cells to synthetize and release some factors involved in neurogenesis. For instance, LGF stimulates endothelial cells in vivo and in vitro to produce TNF-α that mediates LGF-induced proliferation in rat liver (Díaz-Gil et al. 2003).
In summary, our study showed that LGF stimulates neurogenesis when infused into the lateral ventricle of 6-OHDA–lesioned rats. Because this factor also promotes the migration of newly generated neurons into the damaged striatum, we propose LGF as a novel factor that may be useful for neuronal replacement in neurodegenerative diseases such as PD.
Footnotes
Acknowledgements
This study was funded by the Fondo de Investigaciones Sanitarias (FISS PI060315) and Fundaciòn Mapfre Medicina. R.G.G. was the recipient of a Pfizer fellowship, and S.B. and M.R.S. were recipients of Finnova fellowships from the Comunidad Autònoma de Madrid, Spain.
We thank Dr. Carlos L. Paíno (Servicio de Neurobiología, Hospital Ramòn y Cajal, Madrid, Spain) for his comments and critical reading of the manuscript. We also thank Cristina Miranda for her technical help and Martha Messman for her excellent correction of the manuscript.
