Abstract
Satellite cells are responsible for postnatal growth, hypertrophy, and regeneration of skeletal muscle. They are normally quiescent, and must be activated to fulfill these functions, yet little is known of how this is regulated. As a first step in determining the role of lipids in this process, we examined the dynamics of sphingomyelin in the plasma membrane. Sphingomyelin contributes to caveolae/lipid rafts, which act to concentrate signaling molecules, and is also a precursor of several bioactive lipids. Proliferating or differentiated C2C12 muscle cells did not bind lysenin, a sphingomyelin-specific binding protein, but noncycling reserve cells did. Quiescent satellite cells also bound lysenin, revealing high levels of sphingomyelin in their plasma membranes. On activation, however, the levels of sphingomyelin drop, so that lysenin did not label proliferating satellite cells. Although most satellite cell progeny differentiate, others stop cycling, maintain Pax7, downregulate MyoD, and escape immediate differentiation. Importantly, many of these Pax7-positive/MyoD-negative cells also regained lysenin binding on their surface, showing that the levels of sphingomyelin had again increased. Our observations show that quiescent satellite cells are characterized by high levels of sphingomyelin in their plasma membranes and that lysenin provides a novel marker of myogenic quiescence.
Keywords
S
Signals released from crushed myofibers, invading macrophages, and connective tissue have been implicated in the initiation of satellite cell activation [reviewed in Charge and Rudnicki (2004)]. However, the molecular mechanisms responsible for the transduction of such extracellular signals in satellite cells remain poorly defined and the potential role of lipid-mediated signaling has not previously been considered in this context.
Lipids have been recognized to play vital roles in various cellular functions. For example, phosphatidylserine is involved in myoblast fusion (van den Eijnde et al. 2001), and phosphatidylethanolamine is essential in cytokinesis (Emoto and Umeda 2000). Although differences in phospholipid composition during myoblast differentiation and fusion have been reported (Kent et al. 1974; Sessions and Horwitz 1983; Anderson 1991; Pediconi et al. 1992), the dynamics of phospholipid composition during satellite cell activation remains unknown.
Sphingomyelin is an integral lipid component of cell membranes in animals. Although sphingomyelin is located in intracellular membranes such as those of the endosomes, lysosomes, and Golgi apparatus and nucleus, significant amounts are also found in the plasma membrane [reviewed in van Meer and Holthuis (2000)]. Sphingomyelin in the plasma membrane forms lipid microdomains with other sphingolipids, cholesterols, and proteins. It has been suggested that these rafts or caveolae lipid microdomains facilitate cytoplasmic signaling by acting to concentrate signaling molecules [reviewed in Simons and Toomre (2000)]. Moreover, sphingomyelin in the plasma membrane also acts as a reservoir of bioactive sphingolipids. Sphingomyelin metabolites, such as ceramide, sphingosine, and sphingosine-1-phosphate, are emerging as important regulators of a variety of cellular events, including cell proliferation, differentiation, and apoptosis [reviewed in Ohanian and Ohanian (2001); Spiegel and Milstien (2003)]. Despite the importance of these sphingomyelin metabolites, few studies have attempted to examine the dynamics of cellular sphingomyelin, and those that have, have relied on indirect biochemical analysis of extracted lipids lacking single-cell resolution [e.g., Kent et al. (1974)].
In this study, we focus on sphingomyelin levels in the plasma membrane of myogenic cells using lysenin, a protein isolated from the earthworm
Materials and Methods
Cell Culture
C2C12 myogenic cells (Yaffe and Saxel 1977; Blau et al. 1983) were maintained in growth medium comprising DMEM (Gibco; Grand Island, NY) containing 20% FBS (Equitec-Bio; Kerrville, TX), 4 mM
To isolate reserve cells from myotubes, C2C12 cells were cultured in differentiation medium for 4-5 days and then all cells were detached using with 0.05% trypsin-0.53 mM EDTA (Gibco; Grand Island, NY) for 5 min at 37C. Cells were then plated in fresh 20% FBS/DMEM in new Petri dishes. After 30 min at 37C, cells were gently rinsed with PBS to remove floating cells (mostly myotubes) and further cultured in 20% FBS/DMEM.
Phospholipid Analysis
To determine the phospholipid content, 8 × 105 C2C12 cells were plated per 300-mm tissue culture dish in growth medium, and 24 hr later, the medium was switched to serum-free differentiation medium. Four days later, reserve cells and myotubes were separated using a modified version of the method of Kitzmann et al. (1998). Briefly, cells were rinsed with PBS containing 0.1 g/liter MgCl2/6H2O and 0.13 g/liter CaCl2/2H2O before incubation with 0.05% trypsin (without EDTA) at room temperature for 5-10 min. Myotubes detach under these conditions and were collected. Residual cells remained attached and were then rinsed with PBS and collected using standard trypsin-EDTA, as detailed previously. Lipids were then extracted from each fraction according to the method of Bligh and Dyer (1959) and were separated on a silica gel 60 TLC plate (Merck; Darmstadt, Germany) using chloroform/methanol/acetic acid/water (100:75:7:4, v/v). Lipids were then visualized with iodine vapor and compared with those of standards before being assayed for phosphorous content with malachite green reagent (Sigma) (Zhou and Arthur 1992).
Isolation and Culture of Mouse Myofibers
Single myofibers with associated satellite cells were prepared as previously described (Rosenblatt et al. 1995). Briefly, adult (~8 weeks old) C57BL/6 mice were killed by cervical dislocation and the extensor digitorum longus muscle was carefully removed. After washing with PBS, the muscles were immersed in 0.6% Collagenase Type I (Worthington Biochemical; Lakewood, NJ) in DMEM and incubated at 38C for 100 min with agitation. Single myofibers were then liberated from the muscle by repeatedly triturating with a Pasteur pipette and then washed in several changes of DMEM. For the analysis of quiescent satellite cells, myofibers were immediately fixed by the addition of 4% paraformaldehyde/PBS for 10 min before washing in PBS. To activate the associated satellite cells, myofibers were cultured in suspension in DMEM containing 10% horse serum/0.5% chick embryo extract at 37C and 5% CO2 for up to 72 hr, and then fixed as described previously.
Lysenin Probing and Immunostaining
To label sphingomyelin with lysenin (Sekizawa et al. 1997; Yamaji et al. 1998), cultured cells or myofibers were fixed with 4% paraformaldehyde/PBS for 10 min, blocked with 2% BSA/PBS, and then incubated with 0.5 μg/ml lysenin (Peptide Institute Inc; Osaka, Japan, or Sigma) in 2% BSA/PBS for 60 min. To remove sphingomyelin from the plasma membrane, cells were pretreated with 10 mU/ml bacterial sphingomyelinase from Bacillus cereus (Sigma) at 37C for 1 hr before incubation with lysenin. Where used, BrdU (10 μM) was added to the cultures for 3 hr before fixation.
For immunostaining, fixed cells were permeabilized with 0.5% TritonX-100. For BrdU detection, cells were then treated with 3 N hydrochloric acid for 10 min at room temperature. Cells were then incubated with primary antibodies (mouse monoclonal anti-MyoD clone 5.8A [Dakocytomation; Carpinteria, CA]), anti-myogenin clone F5D (a gift from Dr. W. Wright at University of Texas), anti-sarcomeric myosin heavy chain (sMyHC) clone MF20, anti-BrdUclone G3G4 and anti-PAX7 (Developmental Studies Hybridoma Bank; Iowa City, IA), hamster monoclonal anti-Bcl-2 clone 3F11 (BD Pharmingen; San Diego, CA), and rabbit polyclonal anti-lysenin (Sekizawa et al. 1996) (Peptide Institute Inc; Osaka, Japan). After washes in PBS, primary antibody binding was visualized with Alexa Fluor dye-conjugated secondary antibodies (Molecular Probes; Eugene, OR) for 30 min before washing and mounting in Fluoromount fluorescent mounting medium (DakoCytomation) containing 100 ng/ml 4î,6-diamidino-2-phenylindole (DAPI) or Hoechst 33258. Myofibers were immunostained as above and mounted in Fluoromount fluorescent mounting medium (DakoCytomation) containing 100 ng/ml DAPI.
Western Blotting
Cells were lysed in SDS-sample buffer (50 mM Tris-HCl, pH 6.8, 2% SDS, 10% glycerol, 50 mM dithiothreitol, 0.1 mM phenylmethansulfonylfluoride, bromophenol blue), followed by boiling for 3 min. Fifty μg of protein were analyzed by 15% SDS-PAGE and transferred to polyvinylidene fluoride membranes. Membranes were blocked with Odyssey Blocking Buffer (LI-COR; Lincoln, NE) and incubated for 1 hr with primary antibodies. Membranes were washed and incubated with Alexa-Fluor 680-conjugated secondary antibodies and analyzed with an Odyssey Infrared Imaging System (LI-COR).
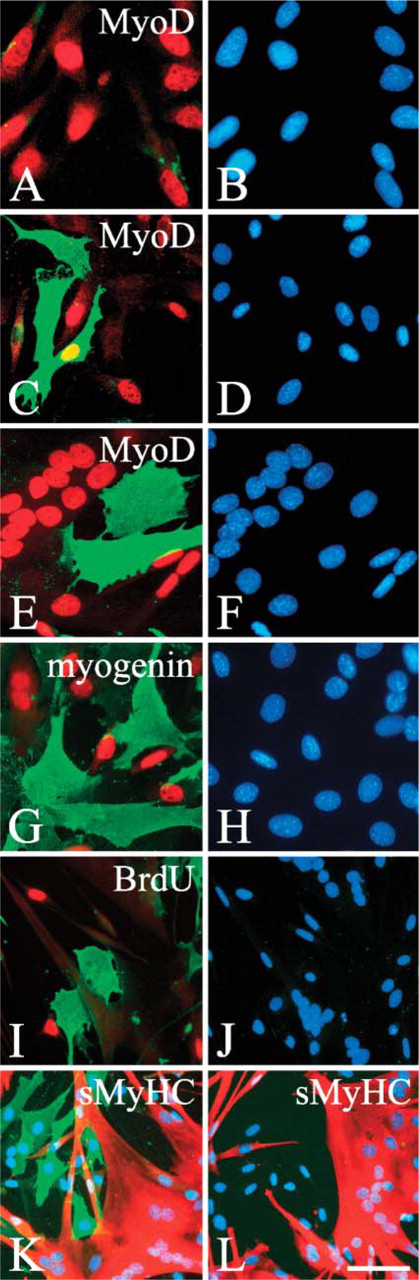
Sphingomyelin levels significantly increase in C2C12 reserve cells. When proliferating C2C12 cells were fixed, probed with lysenin, and immunostained (
Results
To detect cell surface sphingomyelin, we used lysenin, a peptide isolated from the earthworm
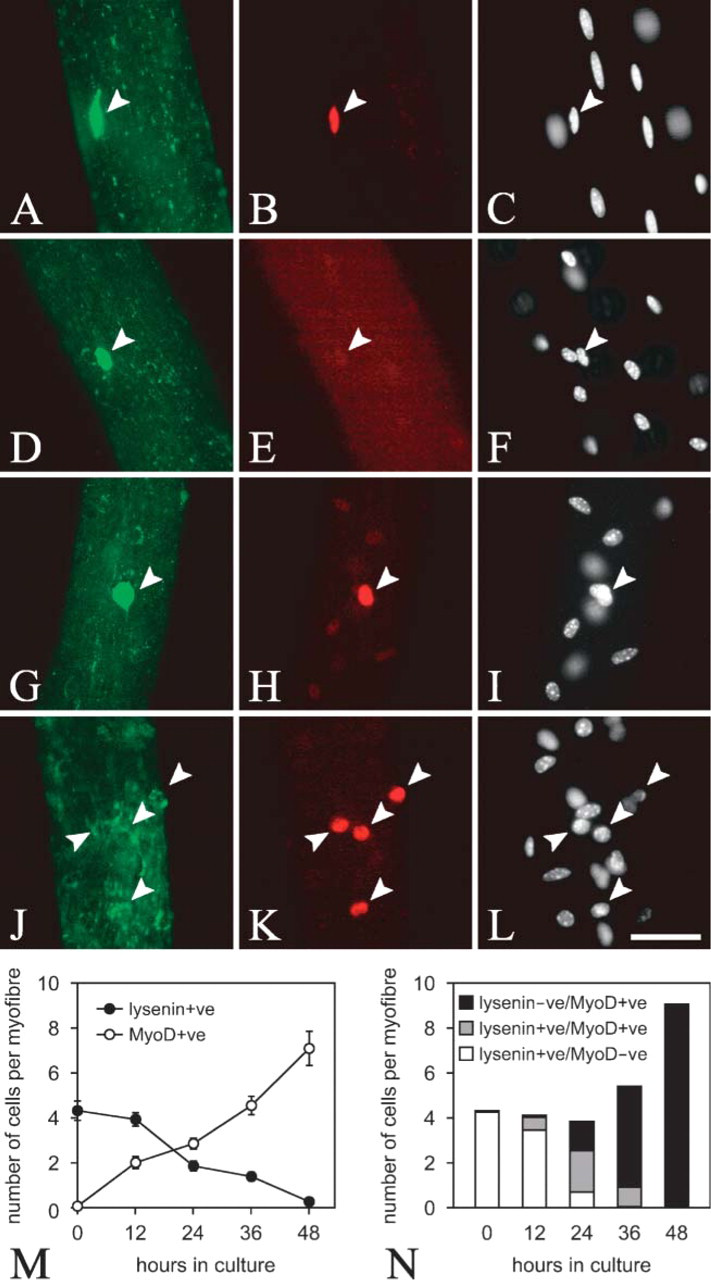
When C2C12 cells were switched to differentiation medium, lysenin-positive (lysenin+ve) cells appeared within 24 hr (Figures 1C and 1D and quantified in Figure 2A). At this time, the lysenin+ve cell population contained both MyoD+ve and MyoD-negative (MyoD-ve) cells (Figures 1C and 1D and quantified in Figure 2A). After 4 days however, lysenin+ve mononucleated cells invariably became MyoD-ve (Figures 1E and 1F and quantified in Figure 2A). On serum deprivation, most C2C12 cells undergo terminal differentiation (Figure 2B), but differentiated mononucleated cells identified with myogenin (Figures 1G and 1H) and multinucleated myotubes (Figures 1E, 1F, and 1K) did not label with lysenin. Differentiation, however, is not the only response to serum withdrawal. Other myogenic cells stop dividing, downregulate MyoD, and escape immediate differentiation to form mononucleated reserve cells (Yoshida et al. 1998). Because the mononucleated cells that bound lysenin after 4 days in differentiation medium were also MyoD-ve (Figures 1E and 1F and quantified in Figure 2A) and did not incorporate BrdU and so were no longer cycling (Figures 1I and 1J), these cells were identified as reserve cells. Because only 8.1 ± 1.2% (mean ± SEM,
To ensure that lysenin was faithfully reporting sphingomyelin levels, reserve cells were treated with 10 mU/ml bacterial sphingomyelinase for 60 min at 37C to digest sphingomyelin, and then incubated with 0.5 μg/ml lysenin. As expected, lysenin+ve immunosignals disappeared when reserve cells were pretreated with bacterial sphingomyelinase, confirming that the lysenin specifically labels sphingomyelin (Figures 1K and 1L). Thus sphingomyelin is highly expressed in reserve cells, but significantly less so in proliferating and differentiating myogenic cells.
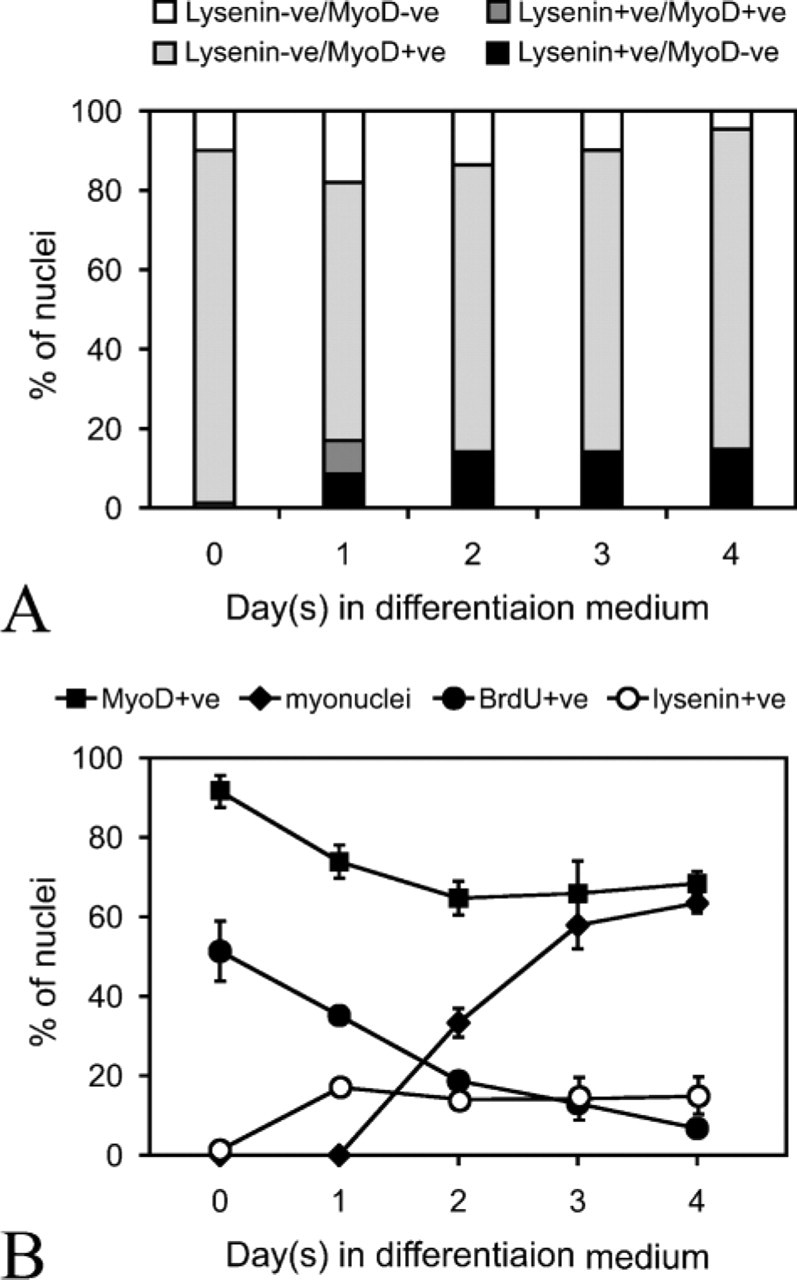
Kinetics of cell surface sphingomyelin during differentiation of C2C12 cells. (
When stimulated with serum, reserve cells reinduce MyoD expression, proliferate, and subsequently differentiate (Yoshida et al. 1998). Reserve cells were separated from differentiated cells and reseeded in fresh growth medium. Lysenin binding was still present on the surface of stimulated reserve cells after 60 min, when most of the cells remained MyoD-ve (Figures 3A and 3B and quantified in Figure 3I), and also after 3 hr, when MyoD expression was beginning to be induced (Figures 3C and 3D and quantified in Figure 3I). By 24 hr, however, significant number of cells did not bind lysenin (Figures 3E and 3F and quantified in Figure 3I), and by 48 hr practically no cells did (Figures 3G and 3H and quantified in Figure 3I). Therefore, sphingomyelin levels in the plasma membrane of reserve cells decrease as the cells are stimulated to proliferate.
Proportion of Sphingomyelin is Higher in Reserve Cells than in Myotubes
Changes in the other components of the plasma membrane, such as glycolipids and cholesterols, can interfere with the efficient binding of lysenin to sphingomyelin (Yamaji et al. 1998, Ishitsuka et al. 2004). To ensure therefore that the differences in lysenin binding observed during reserve cell activation accurately reflects a drop in sphingomyelin levels, we directly quantified their phospholipid composition. After 4 days in serum-free differentiation medium, many myotubes were present, as identified by sMyHC content (Figure 4A) and morphology (Figure 4C), whereas reserve cells expressed Bcl-2 (Figure 4B). Cultures were partially trypsinized to separate myotubes from reserve cells (Kitzmann et al. 1998). Western blot analysis confirmed that this separation was effective with sMyHC, myogenin, and MyoD detectable in both isolated myotubes and total C2C12 cultures, whereas Bcl-2 was enriched in the reserve cell fraction as expected (Dominov et al. 1998) (Figure 4D). Analysis of separated myotube and reserve cell fractions for total phospholipid composition using thin layer chromatography showed that sphingomyelin made up 7.4 mol% of the total phospholipid content in reserve cells, but only 3.8 mol% in myotubes (Table 1). This increase in sphingomyelin content of noncycling reserve cells is consistent with the increase of sphingomyelin in the plasma membrane, as revealed by the binding of lysenin to their surface (Figures 1E and 1F).
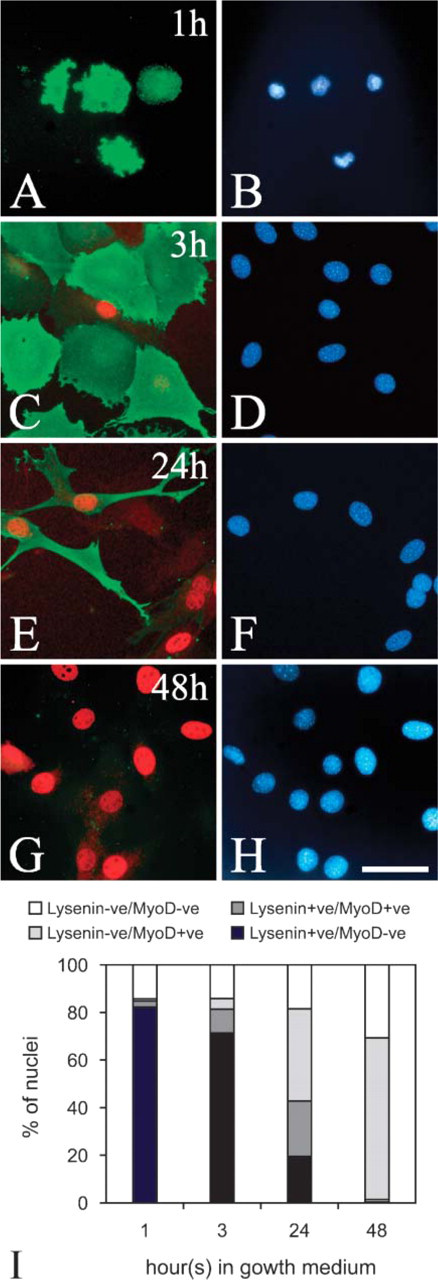
The level of sphingomyelin in the plasma membrane falls as reserve cells activate. After 4-5 days in differentiation medium, mononucleated cells were passaged and reseeded into fresh growth medium. An hour after stimulation, reserve cells still bound lysenin (green), but remained MyoD-ve (
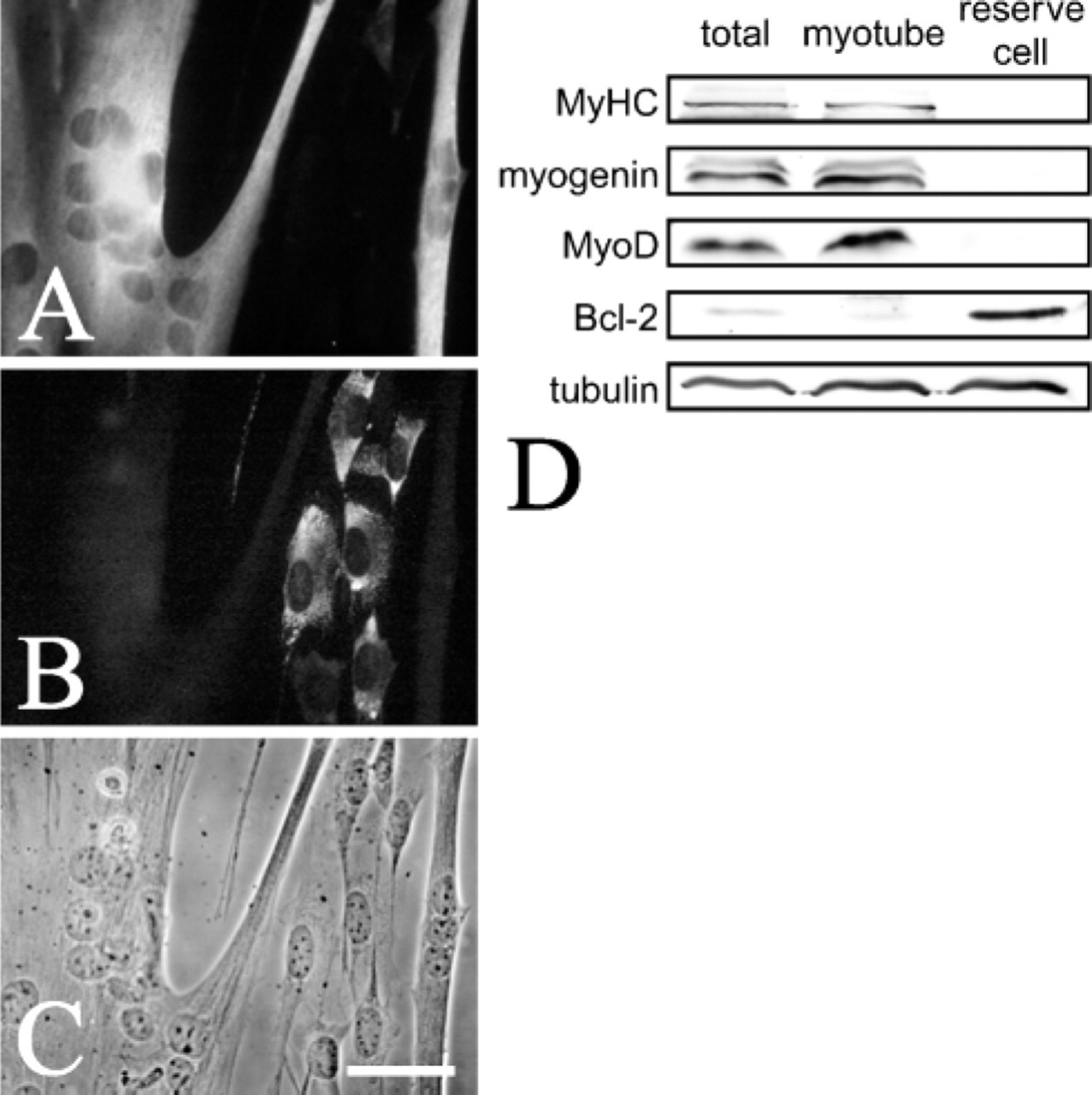
Reserve cells and myotubes can be effectively separated. To analyze phospholipid content (Table 1), it was first necessary to establish that reserve cells and myotubes could be separated. Immunostaining of cultures after 4 days in serum-free differentiation medium showed that myotubes containing sMyHC (
High Levels of Sphingomyelin in the Plasma Membrane Are Characteristic of Quiescent Satellite Cells
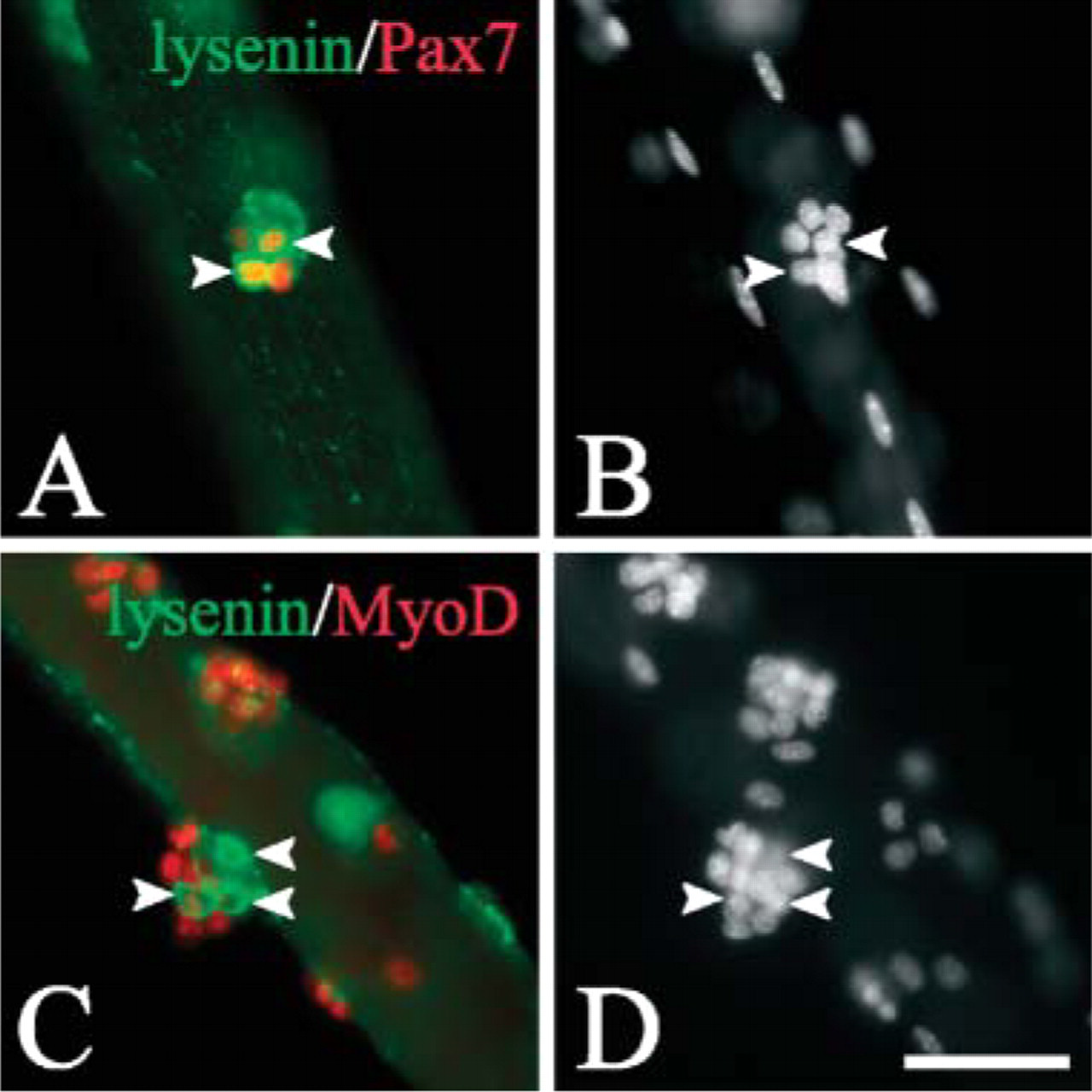
Our observations on reserve cells show that sphingomyelin levels in the plasma membrane of noncycling myogenic reserve cells are high, but then fall as they activate. To see if quiescent myogenic cells in vivo also have high levels of sphingomyelin in their plasma membranes, adult extensor digitorum longus myofibers and their associated quiescent satellite cells were isolated, immediately fixed, and incubated in 0.5 μg/ml lysenin. Immunostaining showed that quiescent satellite cells on the edge of the myofiber, identified by Pax7 expression (Seale et al. 2000), had strong cell surface lysenin labeling (Figures 5A-5C), showing that they too have high levels of sphingomyelin in their plasma membranes. Notably, almost all (98.2 ± 0.8%, mean ± SEM,
Phospholipid composition of myotubes and reserve cells
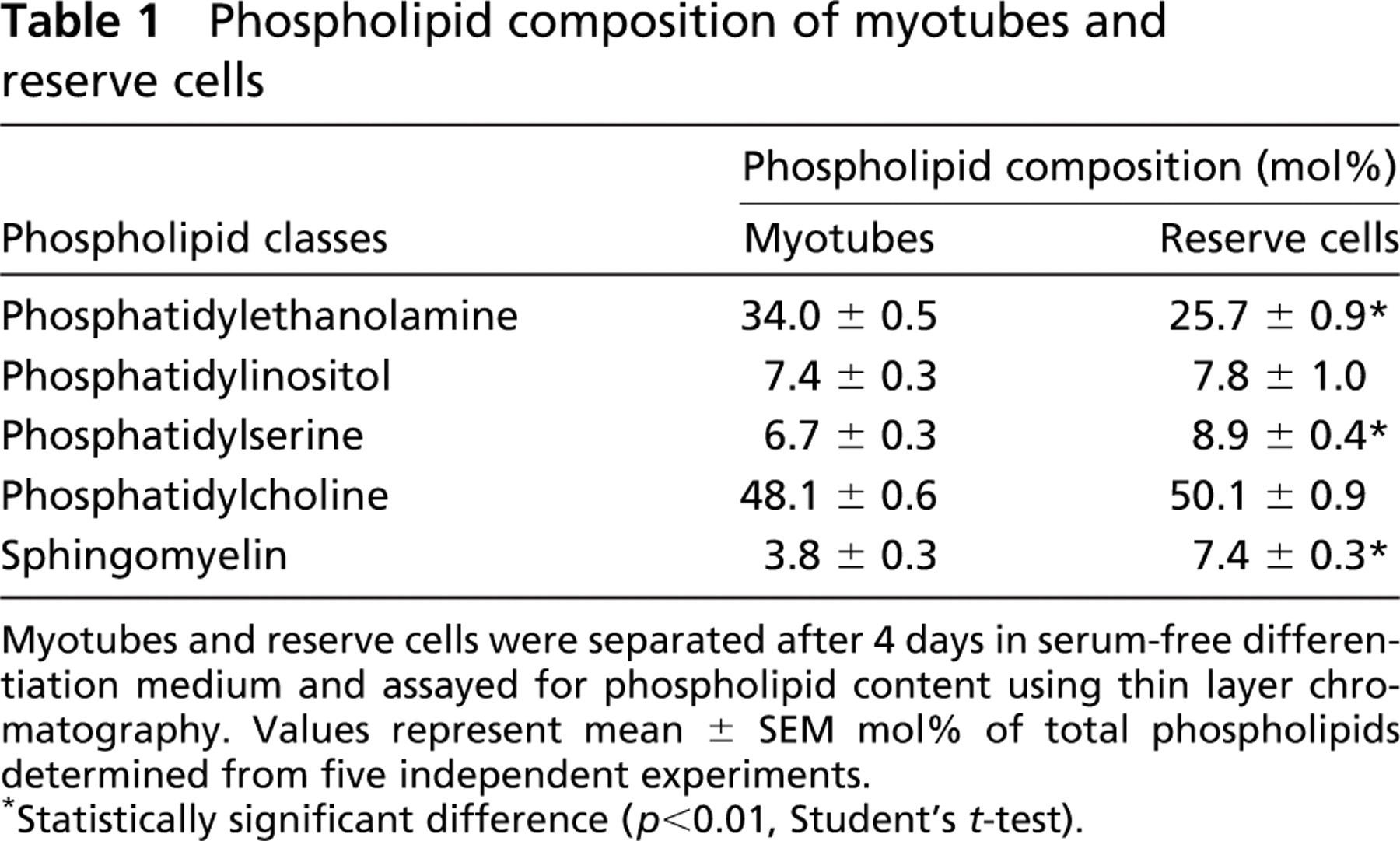
Myotubes and reserve cells were separated after 4 days in serum-free differentiation medium and assayed for phospholipid content using thin layer chromatography. Values represent mean ± SEM mol% of total phospholipids determined from five independent experiments.
Statistically significant difference (
Sphingomyelin Levels Drop during Satellite Cell Activation
Next, we investigated the levels of sphingomyelin during satellite cell activation. Incubation of isolated myofibers in serum stimulates the associated satellite cells to activate, as shown by the induction of MyoD (Yablonka-Reuveni and Rivera 1994; Beauchamp et al. 2000). Coimmunostaining of quiescent satellite cells showed that the majority were lysenin+ve and MyoD-ve (Figures 5D-5F and quantified in Figures 5M and 5N), but, on stimulation, MyoD+ve satellite cells rapidly appeared, with a concomitant drop in the number of cells binding lysenin (Figures 5G-5I and quantified in Figures 5M and 5N). After 48 hr, lysenin staining was absent from virtually all activated MyoD+ve satellite cells (Figures 5J-5L and quantified in Figures 5M and 5N). Satellite cells immunostaining for lysenin and myogenin were mutually exclusive, showing that sphingomyelin levels drop significantly before the cells enter terminal differentiation (data not shown).
Some Satellite Cells Again Increase the Level of Sphingomyelin in Their Plasma Membranes
We have recently shown that when satellite cells are activated on an isolated myofiber, most are destined for differentiation. Some, however, maintain Pax7, downregulate MyoD, and stop cycling (Zammit et al. 2004), thus reacquiring characteristics of quiescence. Although lysenin immunostaining was universally lost in activated/proliferating satellite cells (Figures 5J-5L and quantified in Figures 5M and 5N), some satellite cell progeny later reacquired lysenin binding (Figure 6). Lysenin+ve cells tended to express Pax7 (Figures 6A and 6B), but not MyoD (Figures 6C and 6D). At this time in culture, most MyoD+ve satellite cells also express myogenin (data not shown; Zammit et al. 2004) and are thus committed to differentiation. Therefore the increased levels of sphingomyelin in the plasma membrane of Pax7+ve/MyoD-ve satellite cell progeny to levels characteristic of quiescent satellite cells is further evidence that some satellite cells have returned to a quiescent-like state.
Sphingomyelin levels are high in the plasma membranes of quiescent, but not activated, satellite cells. Coimmunostaining of freshly isolated extensor digitorum longus myofibers demonstrates that the majority of associated Pax7+ve (red) quiescent satellite cells (arrowheads) bind lysenin (green) on their surface (
Discussion
In this study, we have examined the levels of sphingomyelin at the cell surface of myogenic cells using the sphingomyelin-specific binding protein lysenin (Yamaji et al. 1998). Cycling C2C12 cells are either MyoD+ve or MyoD-ve (Yablonka-Reuveni and Rivera 1997), but do not bind lysenin, showing that proliferating myogenic cells have little sphingomyelin in their plasma membranes. This remains the case in differentiated myocytes and myotubes. However, other myogenic cells respond to serum withdrawal by downregulating MyoD and escaping immediate differentiation to become reserve cells (Yoshida et al. 1998). In the course of this process, the levels of sphingomyelin in their plasma membranes of reserve cells significantly increased. Indeed, direct measurement showed that reserve cells contained almost twice as much sphingomyelin as myotubes. The differences in the level of sphingomyelin may appear smaller than expected from lysenin staining, but only ~50% of cellular sphingomyelin is located in the plasma membrane [reviewed in van Meer and Holthuis (2000)], with the rest in intracellular membranes. In addition, the low levels of sphingomyelin on the surface of myotubes may be below the level of detection with lysenin at the concentration used in this study. Taken together, our data suggest that quiescence is accompanied by a specific increase in sphingomyelin levels in the plasma membrane. We are examining the changes in sphingomyelin metabolism during the activation of satellite cells to determine how much breakdown or synthesis of sphingomyelin occurs during this event.
Some satellite cell progeny reacquire lysenin binding. Coimmunostaining of extensor digitorum longus myofibers that had been in culture for 72 hr showed Pax7+ve (red) satellite cells, some of which bound lysenin (green) on their surface (arrowheads in
Muscle satellite cells contribute myogenic cells to growing muscle to provide new myonuclei, but then become quiescent in normal adult muscle (Schultz et al. 1978). The rapid isolation of intact myofibers allows their associated satellite cells to be examined while still quiescent (Yablonka-Reuveni and Rivera 1994; Beauchamp et al. 2000). Using this preparation, we found that virtually all quiescent satellite cells bound lysenin, demonstrating that they too have high levels of sphingomyelin in their plasma membranes. The culture of myofibers in mitogen-rich medium stimulates their associated satellite cells to activate, proliferate, and subsequently differentiate, and this process is accompanied by a significant decrease of sphingomyelin on their cell surface. However, although lysenin is useful for identifying quiescent satellite cells on isolated myofibers, it should be noted that sphingomyelin is present to varying degrees in the membranes of all cells. Significantly, sphingomyelin levels fall rapidly on activation, whereas most other commonly used molecular markers for satellite cells, including M-cadherin, Pax7, and the Myf-5/β-galactosidase fusion protein, persist in proliferating cells (Irintchev et al. 1994; Beauchamp et al. 2000; Seale et al. 2000).
Why is the transition from quiescence to activation in myogenic cells associated with a change in the levels of sphingomyelin in the plasma membrane? Because activation itself is rapid, the change in sphingomyelin observed in this study might be secondary to the activation. It would be important to examine the relationship between sphingomyelin and the certain stimuli known to involved in satellite cell activation such as hepatocyte growth factor (Allen et al. 1995), mechanical stretch (Tatsumi et al. 2001), and nitric oxide (Anderson and Pilipowicz 2002). One possible role for sphingomyelin in the plasma membrane is to act to concentrate signaling molecules. It is well established that sphingolipids, together with cholesterol and signaling molecules, are organized into lateral assemblies within cell membranes. These assemblies, including lipid rafts and caveolae, are emerging as important centers for cell signaling [reviewed in Simons and Toomre (2000)] and could act as platforms to coordinate molecules needed to initiate myogenic cell activation. Alternatively, the loss of sphingomyelin may reflect its metabolism to produce bioactive lipids. It has been proposed that sphingomyelin in the plasma membrane acts as a store that can then be cleaved to generate ceramide, sphingosine, or sphingosine-1-phosphate, which have been shown to act as second messengers in a variety of cell types (Ohanian and Ohanian 2001). Ceramide, generated by agonist-induced sphingomyelin hydrolysis, has been implicated in cell differentiation, growth arrest, and apoptosis (Hannun 1996). Ceramide is further metabolized into sphingosine-1-phosphate, which is mitogenic in diverse cell types and has been shown to oppose ceramide-mediated apoptosis (Spiegel and Milstien 2003). Recent studies have revealed that sphingolipids are also active in skeletal muscle. Sphingosine controls muscle contraction by regulating calcium concentration in myofibers (Sabbadini et al. 1999), whereas ceramide has an inhibitory effect on insulin-like growth factor-I-induced protein synthesis in mouse myogenic C2C12 cells (Strle et al. 2004). Neutral sphingomyelinase catalyses the cleavage of sphingomyelin to produce bioactive lipid metabolites and is present in skeletal muscle (Ghosh et al. 1998); therefore, the sphingomyelin in the plasma membrane could be accessed for signaling purposes. It is then intriguing to speculate that bioactive lipids may also play a role in satellite cell activation.
When both reserve cells and satellite cells were stimulated with serum, sphingomyelin levels in their plasma membranes dropped significantly. However, at later times in culture, a limited number of satellite cell progeny reacquired lysenin binding, showing that the level of sphingomyelin in their plasma membranes had again increased. The effective response to repeated injury shows that the satellite cell pool is maintained (Sadeh et al. 1985; Luz et al. 2002), but there is debate at present about how this is achieved. It has been proposed that satellite cells may be part of a hierarchical system and merely represent a committed myogenic precursor that is restricted to providing myonuclei. In this system, the replacement of satellite cells occurs from a stem cell located within the muscle interstitium (Gussoni et al. 1999; Asakura et al. 2002) or outside muscle tissue (Fukada et al. 2002; LaBarge and Blau 2002), but there is no evidence that this accounts for more than a very minor contribution. More likely is that satellite cells self-renew, as originally proposed by Moss and Leblond (1971). Indeed we have recently shown that satellite cell progeny maintained on isolated myofibers adopt divergent fates in culture. When they activate, satellite cells coexpress Pax7 with MyoD and most then proliferate, downregulate Pax7, and differentiate. In contrast, other proliferating cells maintain Pax7 but lose MyoD and withdraw from both the cell cycle and immediate myogenic differentiation, characteristics consistent with a quiescent state (Zammit et al. 2004). Similar observations have been made in chicken during muscle growth, showing that this may be a widespread mechanism in vertebrates (Halevy et al. 2004). The demonstration that some of these Pax7+ve/MyoD-ve cells also reacquire lysenin binding on their surface, a characteristic of quiescence in myogenic cells, is further evidence that these cells have reentered a quiescent state.
In conclusion, sphingomyelin levels are high in the plasma membrane of quiescent satellite cells and then fall as they activate and proliferate. These results implicate lipid rafts/caveolae as platforms to coordinate activation signals or the involvement of bioactive lipid metabolism in the process of activation and the subsequent return to quiescence. This increase in sphingomyelin levels on some satellite cells that appear to be reentering quiescence supports the conclusion that they are able to self-renew. Assaying the sphingomyelin level in the plasma membrane using lysenin therefore provides a positive marker of quiescence that actually drops during activation, in contrast to most other molecular markers of satellite cells.
Footnotes
Acknowledgments
This work was supported by a research grant (11B-1) for Nervous and Mental Disorders and by a grant for research in Brain Science from the Ministry of Health, Labor and Welfare, Japan, and by a grant from the Fugaku Trust for Medical Research. The Japan Scholarship Foundation funded Y.N., whereas P.S.Z. was supported by The Muscular Dystrophy Association and the Medical Research Council.
We thank Jon Beauchamp and Terry Partridge for invaluable discussions and their comments on the manuscript and Drs. Y. Miyagoe-Suzuki and S. Takeda (National Institute of Neuroscience, National Center of Neurology and Psychiatry) for advice on single myofiber isolation. P.S.Z. acknowledges the invaluable assistance of the late, great John Peel. The monoclonal antibodies, MF20, G3G4, and PAX7 were obtained from the Developmental Studies Hybridoma Bank developed under the auspices of the National Institute of Child Health and Human Development and maintained by The University of Iowa, Department of Biological Sciences, Iowa City, Iowa.
