Abstract
The developmental potential of skeletal muscle stem cells (satellite cells) remains controversial. The authors investigated satellite cell developmental potential in single fiber and clonal cultures derived from MyoD iCre/+;R26R EYFP/+ muscle, in which essentially all satellite cells are permanently labeled. Approximately 60% of the clones derived from cells that co-purified with muscle fibers spontaneously underwent adipogenic differentiation. These adipocytes stained with Oil-Red-O and expressed the terminal differentiation markers, adipsin and fatty acid binding protein 4, but did not express EYFP and were therefore not of satellite cell origin. Satellite cells mutant for either MyoD or Myf-5 also maintained myogenic programming in culture and did not adopt an adipogenic fate. Incorporation of additional wash steps prior to muscle fiber plating virtually eliminated the non-myogenic cells but did not reduce the number of adherent Pax7+ satellite cells. More than half of the adipocytes observed in cultures from Tie2-Cre mice were recombined, further demonstrating a non-satellite cell origin. Under adipogenesis-inducing conditions, satellite cells accumulated cytoplasmic lipid but maintained myogenic protein expression and did not fully execute the adipogenic differentiation program, distinguishing them from adipocytes observed in muscle fiber cultures. The authors conclude that skeletal muscle satellite cells are committed to myogenesis and do not spontaneously adopt an adipogenic fate.
Keywords
Satellite cells are skeletal muscle-specific stem cells that reside between the basal lamina and sarcolemma of muscle fibers (Mauro 1961; Sacco et al. 2008). Their critical function in the growth, maintenance, and repair of skeletal muscle is well established (Zammit et al. 2006). Transplantation of a single muscle fiber, with resident satellite cells intact, is sufficient not only to support significant regeneration of the host limb muscle but also to replenish the satellite cell pool (Collins et al. 2005). Key aspects of satellite cell biology, including their robust myogenic potential and self-renewal properties, have been modeled in single fiber culture (Bischoff 1975; Zammit 2008). When single muscle fibers are placed in culture, associated satellite cells are quickly activated, reenter the cell cycle, and express MyoD and other regulatory factors within 24 hr (Yablonka-Reuveni and Rivera 1994; Cornelison and Wold 1997; Zammit et al. 2002). When fibers are plated on appropriate substrates, satellite cells migrate from the fibers and establish proliferative colonies of cells that subsequently differentiate into multinucleated myotubes, recapitulating some of the major events that occur during muscle regeneration (Bischoff 1986; Yablonka-Reuveni and Rivera 1994; Rosenblatt et al. 1995). Compared to mass cultures, single-fiber culturing methods have the advantage of maintaining the anatomical and perhaps physiological relationship between satellite cell and fiber and provide a means of purifying satellite cells from the complex mixture of cell populations found in the muscle interstitium.
Satellite cell developmental plasticity has been the subject of intensive investigation. The historical view that satellite cells are committed myogenic stem cells was influenced by the assumption, based on histological observations, that satellite cells derive from myoblasts in the embryo and by the many functional studies that have demonstrated their robust myogenic activity in culture and in vivo (Zammit et al. 2006). Our recent lineage analysis using MyoDiCre knockin mice is consistent with the notion that satellite cells derive from a myoblast-like progenitor (Kanisicak et al. 2009). Yet, results from a number of investigations using both mass cultures and single fiber cultures have reported non-myogenic differentiation of satellite cells, raising questions concerning the state of satellite cell commitment. For example, satellite cells or myoblasts in mass culture treated with bone morphogenetic protein 2 (BMP2) or BMP4 turn on markers of osteogenic differentiation, such as alkaline phosphatase and osteocalcin, and extinguish markers of the myogenic phenotype (Katagiri et al. 1994; Asakura et al. 2001; Wada et al. 2002). Alternatively, adipogenic differentiation can be induced by inhibition of Wnt signaling (Ross et al. 2000) or by growth in adipogenic media (Wada et al. 2002; De Coppi et al. 2006; Kook et al. 2006; Singh et al. 2007), and this capacity for adipogenic differentiation is increased in satellite cells derived from aged muscle (Guan et al. 2002; Taylor-Jones et al. 2002). Finally, at least some satellite cells cultured in the presence of aged serum exhibit a myogenic to fibrogenic conversion (Brack et al. 2007).
Of particular note, several groups have reported spontaneous adipogenic and osteogenic differentiation in single fiber cultures derived from young adult mice, without the addition of potent factors that promote non-myogenic lineage progression or differentiation (Asakura et al. 2001; Csete et al. 2001; Shefer et al. 2004). Clonal analysis indicated that individual satellite cells and their progeny adopt only a single developmental fate, with adipogenic differentiation representing the phenotype of the majority of clones (Shefer et al. 2004). The satellite cell population is molecularly and functionally heterogeneous (Zammit 2008), and this apparent capacity for non-myogenic differentiation under “standard” growth conditions could represent intrinsic differences in developmental potency. However, cells with adipogenic and osteogenic potential exist in the muscle interstitium (Lounev et al. 2009; Joe et al. 2010; Uezumi et al. 2010), and the evidence that satellite cells represent a source of non-myogenic cells in fiber cultures remains equivocal.
Fibrosis and fatty infiltration are well-known histological features of aging skeletal muscle and represent histopathological hallmarks of dystrophic muscle in humans (Pastoret and Sebille 1995; Pahor and Kritchevsky 1998; Goodpaster and Wolf 2004; Visser et al. 2005; Alexakis et al. 2007; Brack et al. 2007; Delmonico et al. 2009). Defining the developmental potential of satellite cells will clarify their role, if any, in the inter- and intramuscular accumulation of non-myogenic cells types resulting from aging or disease. In addition, understanding whether and under what circumstances satellite cells can adopt divergent fates has important implications for their use in cell-based therapies, where restricting differentiation to desirable developmental pathways is of central importance.
In the present report, we use Cre/loxP lineage analysis to reinvestigate the developmental potential of satellite cells. Satellite cells were specifically and permanently labeled as a consequence of developmental expression of a MyoDiCre knockin allele (Kanisicak et al. 2009; Lounev et al. 2009; Yamamoto et al. 2009), and their fate followed in single muscle fiber and clonal satellite cell cultures. We present evidence that satellite cells do not spontaneously undergo adipogenic differentiation under standard culture conditions and that non-myogenic phenotypes observed in single muscle fiber cultures are likely the result of unintentional co-purification of non-myogenic progenitors from the muscle interstitium.
Materials and Methods
Animals
MyoDiCre knockin mice were developed in our laboratory, and their generation and characterization were described elsewhere (Kanisicak et al. 2009; Lounev et al. 2009). Tie2-Cre transgenic mice (Kisanuki et al. 2001) , Myf-5 null mice (Myf-5 loxP/loxP ; Kassar-Duchossoy et al. 2004; Gayraud-Morel et al. 2007), and MyoD null mice (Rudnicki et al. 1992) were kindly provided by Drs. Tom Sato (University of Texas Southwestern Medical Center), Shahragim Tajbakhsh (Pasteur Institute), and Michael Rudnicki (Ottawa Health Sciences Centre), respectively. FABP4-Cre (He et al. 2003) and R26REYFP (Srinivas et al. 2001) mice were obtained from Jackson Laboratories (Bar Harbor, ME, http://jaxmice.jax.org). FVB mice were obtained from Charles River Laboratories (Wilmington, MA, http://criver.com). All transgenic mouse lines were maintained on an FVB-enriched background. All procedures were approved by the University of Connecticut Institutional Animal Care and Use Committee.
Single Muscle Fiber Isolation and Culture
Single muscle fibers were prepared from the extensor digitorum longus (EDL) muscles of young adult mice (2-4 months) as described previously (Shefer et al. 2004; Shefer and Yablonka-Reuveni 2005) and are referred to as the “standard” muscle fiber isolation procedure throughout the text. Briefly, whole EDL muscles from young adult mice were digested with 0.2% type 1 collagenase (Sigma-Aldrich, St. Louis, MO, http://www.sigmaaldrich.com) diluted with Dulbecco’s modified Eagle’s medium (DMEM; Invitrogen, Carlsbad, CA, http://www.invitrogen.com) for 90 min at 37C. Sterile fire-polished Pasteur pipettes were used to gently dissociate the muscles in horse serum–coated 60-mm dishes containing DMEM. Single EDL muscle fibers were plated individually into 4-well tissue culture plates (Nunc, Rochester, NY, http://nuncbrand.com) coated with Growth Factor Reduced Matrigel (BD Biosciences, San Jose, CA, http://www.bdbiosciences.com) diluted to 1 mg/ml in high glucose DMEM. Muscle fiber culture medium (250 µl/well) consisting of 20% fetal bovine serum (FBS), 10% horse serum, and 1% chick embryo extract (Accurate Chemical, Westbury, NY, http://www.accuratechemical.com) in DMEM supplemented with 1% Pen-Strep (Invitrogen) was then added, and cultures were maintained at 37C, 5% CO2. Muscle fiber culture medium was replaced weekly to allow direct comparison with previous data (Shefer et al. 2004).
To test whether adipogenic cells in fiber cultures derived from loosely adherent, fiber-associated cells or represented contaminating cells in the bulk medium that were transferred during fiber plating, a modified fiber isolation procedure was employed whereby fibers were subjected to two additional wash steps prior to plating. This minor modification to the standard procedure entailed two serial transfers into horse serum–coated 60-mm dishes containing DMEM before plating the individual muscle fibers or isolating single satellite cells from muscle fibers for clonal analysis. We refer to this method as the “modified” muscle fiber isolation procedure.
Establishment of Clonal Satellite Cell Cultures from Single EDL Muscle Fibers
Satellite cell clonal cultures were prepared essentially as described by Shefer et al. (2004). Single EDL muscle fibers isolated using the two isolation methods described above (standard and modified) were placed in separate 1.5-ml microcentrifuge tubes containing 1 ml of muscle fiber culture medium and triturated four times, using a 20-gauge needle attached to a 1-ml syringe. The resulting cell suspension containing the liberated satellite cells was then evenly distributed into 12 wells of a 24-well tissue culture plate coated with 1 mg/ml Growth Factor Reduced Matrigel. This was expected to result in zero or one satellite cell per well, based on the observation of an average number of seven satellite cells per adult EDL muscle fiber (Zammit et al. 2002; Shefer et al. 2004). After equal dispersion of the satellite cell suspension, an additional 250 µl of muscle fiber culture medium was added to each well. Clones were maintained at 37C, 5% CO2, and culture medium was replaced weekly.
Immunofluorescence
Cultures were fixed at the indicated time points for 10 min in 4% paraformaldehyde (Electron Microscopy Sciences, Hatfield, PA, http://www.emsdiasum.com) in 0.1 M sodium phosphate buffer and then rinsed twice with phosphate-buffered saline (PBS; pH 7.4). Fixed cultures were incubated in PBS containing 5% goat serum (Sigma-Aldrich), 2% bovine serum albumin (Sigma-Aldrich), and 0.2% Triton X-100 (Sigma-Aldrich) for 1 hr at room temperature (RT) to block nonspecific antibody binding. Cultures were then incubated with primary and secondary antibodies diluted with blocking solution at RT for 2 hr and 30 min, respectively. Cultures were rinsed (3 × 5 min) with PBS after incubation with primary and secondary antibodies followed by brief exposure to 100 ng/ml 4′,6-diamidino-2-phenylindole (DAPI; Sigma-Aldrich) to allow visualization of nuclei and a final rinse in PBS before imaging. Single EDL muscle fibers were fixed in 4% paraformaldehyde for 10 min at RT immediately after isolation using the two procedures detailed above. Following fixation, the muscle fibers were immunostained for Pax7 using the staining methods described above for cultured cells.
Primary and Secondary Antibodies
The following antibodies and dilutions were used: mouse monoclonal anti-chicken Pax7 hybridoma supernatant diluted 1:10, mouse monoclonal anti-chicken sarcomeric myosin heavy chain (MyHC) hybridoma supernatant (MF20) diluted 1:10, and mouse monoclonal anti-rat Myogenin hybridoma supernatant (F5D) diluted 1:10 (Developmental Studies Hybridoma Bank, Iowa City, IA, http://dshb.biology.uiowa.edu); mouse monoclonal anti-human MyoD (Clone 5.8A) diluted 1:1000 (BD Biosciences); and rabbit polyclonal anti–green fluorescent protein (GFP), Alexa Fluor 546–conjugated goat anti-mouse IgG and Alexa Fluor 488–conjugated goat anti-rabbit IgG diluted 1:1000 (Molecular Probes, Eugene, OR, http://probes.invitrogen.com).
Detection of Lipid-Filled Adipocytes by Oil-Red-O Staining
Lipid-filled adipocytes were detected with Oil-Red-O (ORO) as previously described, with only minor modifications (Koopman et al. 2001; Shefer et al. 2004). The ORO (Sigma-Aldrich) staining solution was prepared by diluting five volumes of an ORO stock solution (0.5% [w/v] ORO in 60% triethylphosphate [Fisher Scientific, Waltham, MA, http://fishersci.com], filtered through Whatmann paper) with three volumes of water, followed by filtration through a 0.45-µm filter. The working solution was prepared just prior to use. Cultures were fixed as described above and exposed to ORO for 5 min at RT, rinsed twice with PBS, and stained with DAPI. For cells processed for both immunofluorescence and the presence of neutral lipids, ORO staining was conducted after completion of the immunofluorescence staining procedure.
Adipogenic Induction of Single Muscle Fiber Cultures
Single EDL muscle fibers were cultured for 7 days and then switched to adipogenic induction medium (Taylor-Jones et al. 2002; Wada et al. 2002). Adipogenesis-inducing medium consisted of either 10% FBS in DMEM containing 100 µM γ-linolenic acid (GLA; Sigma-Aldrich) or 20% FBS in DMEM supplemented with 10 µg/ml insulin (Sigma-Aldrich), 0.5 mM isobutylmethylxanthine (Calbiochem, Darmstadt, Germany, http://www.emdbiosciences.com), and 1 µM dexamethasone (MP Biomedicals, Solon, OH, http://www.mpbio.com) (collectively referred to as IDX). Cultures were exposed to GLA for a total of 10 days with a medium change after 5 days of induction, whereas those exposed to IDX were induced for 3 days with daily feeding, then switched to 20% FBS in DMEM supplemented with 10 µg/ml insulin for 1 day. Subsequently, cultures were harvested for RNA isolation or processed for immunofluorescence and ORO staining as described above.
Adipogenic Gene Expression Analysis by RT-PCR
Total RNA was isolated using the Stratagene NanoPrep RNA Isolation Kit (Stratagene, La Jolla, CA, http://stratagene.com) according to the manufacturer’s instructions. Total RNA (15 ng) was used to synthesize cDNA using the ProtoScript First Strand cDNA Synthesis Kit (New England Biolabs, Ipswich, MA, http://neb.com) according to the manufacturer’s instructions. PCR reactions were performed using 30 cycles of 94C for 1 min, 60C for 1 min, and 72C for 1 min. Resulting products were separated electrophoretically in 1.5% agarose and visualized by ethidium bromide staining. PCR primers used to amplify cDNA (Guan et al. 2002) are as follows:
Adipsin Fwd 5′ CCCTACATGGCTTCCGTGC 3′
Adipsin Rev 5′ GTAACCACACCTTCGACTGC 3′
FABP4 Fwd 5′ GGGATTTGGTCACCATCCG 3′
FABP4 Rev 5′ CCAGCTTGTCACCATCTCG 3′
GAPDH Fwd 5′ ATTGGGAAGCTTGTCATCAACG 3′
GAPDH Rev 5′ ACCCTGTTGCTGTAGCCGT 3′
Photography and Images
Images were captured using Openlab imaging software (Improvision, Waltham, MA, http://improvision.com) with an Eclipse TE2000-U inverted fluorescence microscope (Nikon Instruments, Melville, NY, http://www.nikoninstruments.com) and a Retiga EXi digital camera (QImaging Corporation, Surrey, BC, http://www.qimaging.com). Brightness and contrast of images were adjusted using Adobe Photoshop Version 7.0 (Adobe Systems, San Jose, CA, http://www.adobe.com).
Statistical Analysis
Experiments were repeated a minimum of three times unless otherwise noted. Data were analyzed using the general linear model procedure of PC SAS Release 8.1 (SAS Institute, Cary, NC, http://www.sas.com). Least squared mean comparisons were made using Fisher’s least significant difference (LSD) test. Means were considered different if p < 0.05.
Results
Satellite Cells Emanating from Cultured MyoD iCre/+;R26R EYFP/+ Muscle Fibers Co-express Pax7 and EYFP
Cre/loxP lineage analysis demonstrated that 98% of satellite cells present on freshly isolated EDL muscle fibers from MyoD iCre/+;R26R EYFP/+ mice co-express the satellite cell marker, Pax7 (Seale et al. 2000; Zammit 2008), and EYFP, indicating that the vast majority of satellite cells originate from a MyoD+ precursor (Kanisicak et al. 2009; Fig. 1A-C). Activated satellite cells present on cultured single muscle fibers migrate from their sublaminar position onto the tissue culture surface and begin to proliferate within 1 to 2 days of culture. In these early cultures, activated satellite cells and their daughters can be identified by expression of both MyoD and Pax7 (Shefer et al. 2004; Zammit et al. 2004). To test the sensitivity and specificity of Cre-dependent satellite cell labeling in culture, single EDL muscle fibers were isolated from adult MyoD iCre/+;R26R EYFP/+ mice using the standard isolation procedure (see Materials and Methods), and mononuclear cells were assayed by immunofluorescence for MyoD and Pax7, as well as for EYFP (Fig. 1). All of the MyoD+ cells (Fig. 1D-F; 217 of 217) and almost all Pax7+ cells (Fig. 1G-I; 405 of 409) co-expressed EYFP, demonstrating that myogenic cells from MyoD iCre/+;R26R EYFP/+ mice are efficiently labeled. Whether rare Pax7+;EYFP– cells in culture and on freshly isolated fibers (Kanisicak et al. 2009) represent a distinct subpopulation of satellite cells that have not expressed MyoD in their developmental history or simply represent recombination or detection efficiencies of <100% is unclear. As described previously (Shefer et al. 2004), Pax7– cells were also observed in these muscle fiber cultures and in our study represented 38% (Fig. 1J-L; 251 of 660) of the mononuclear cell population analyzed in 4-day cultures. Importantly, 98.4% of the 251 Pax7– cells assayed failed to express EYFP; the rare Pax7–;EYFP+ cells likely reflect myogenic cells that do not express Pax7 in day 4 cultures, as 100% of EYFP+ cells assayed at later time points were myogenic (see below). Collectively, these data demonstrate the efficiency and specificity of myogenic cell labeling with the MyoD iCre knockin allele.
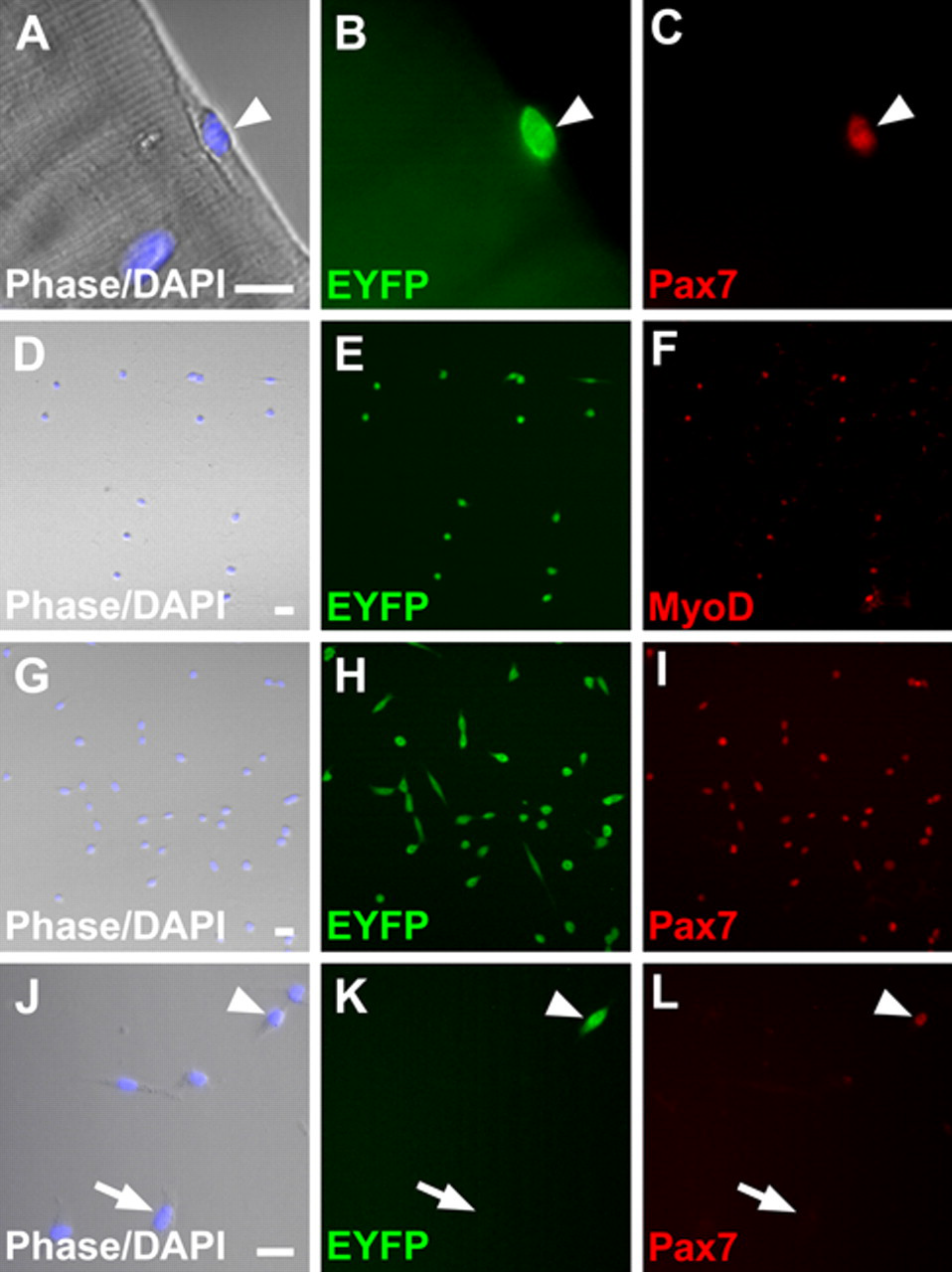
Efficient labeling of satellite cells from MyoD iCre/+;R26R EYFP/+ skeletal muscle. (A-C) Pax7+;EYFP+ satellite cell (arrowhead) on a freshly isolated extensor digitorum longus (EDL) fiber. (D-I) Myogenic regions of 4-day satellite cell cultures showing the concordance of EYFP fluorescence and MyoD (D-F) or Pax7 (G-I) immunofluorescence labeling. (J-L) Four-day cultures comprise Pax7+ (arrowhead) and Pax7– (example at arrow) cells. Pax7– cells are EYFP–. Scale bars: A-C, 5 µm; D-L, 10 µm.
Adipocytes in Muscle Fiber Cultures Are Not Derived from EYFP+ Satellite Cells
We next examined the differentiation potential of EYFP+ satellite cells under standard muscle fiber culture conditions, without the addition of adipogenic media. Single EDL muscle fibers (n = 128) from adult MyoD iCre/+;R26R EYFP/+ mice were isolated using the standard procedure and cultured for 14 days to determine if MyoD iCre -labeled satellite cells could spontaneously differentiate into adipocytes (Asakura et al. 2001; Csete et al. 2001; Shefer et al. 2004). After 14 days in culture, numerous myotubes, as well as large multilocular adipocytes, were observed (Fig. 2A-C). The adipocytes observed in these cultures stained strongly with the neutral lipid stain, ORO (Fig. 2B). Myotubes present in 14-day cultures robustly expressed both EYFP and MyHC (Fig. 2C-E). Of note, we did not observe any EYFP+ adipocytes in cultures derived from MyoD iCre/+;R26R EYFP/+ mice (Fig. 2C-G; n = 128). To confirm that adipocytes were indeed capable of expressing the recombined Cre-dependent reporter allele, we generated FABP4-Cre;R26R EYFP/+ mice in which Cre was driven by regulatory elements of the fatty acid binding protein 4 (FABP4/aP2) gene (He et al. 2003). All of the lipid-filled adipocytes present in 14-day EDL cultures were EYFP+, indicating that these cells can readily undergo Cre-mediated recombination and robustly express the R26REYFP reporter allele (Fig. 2H,I).
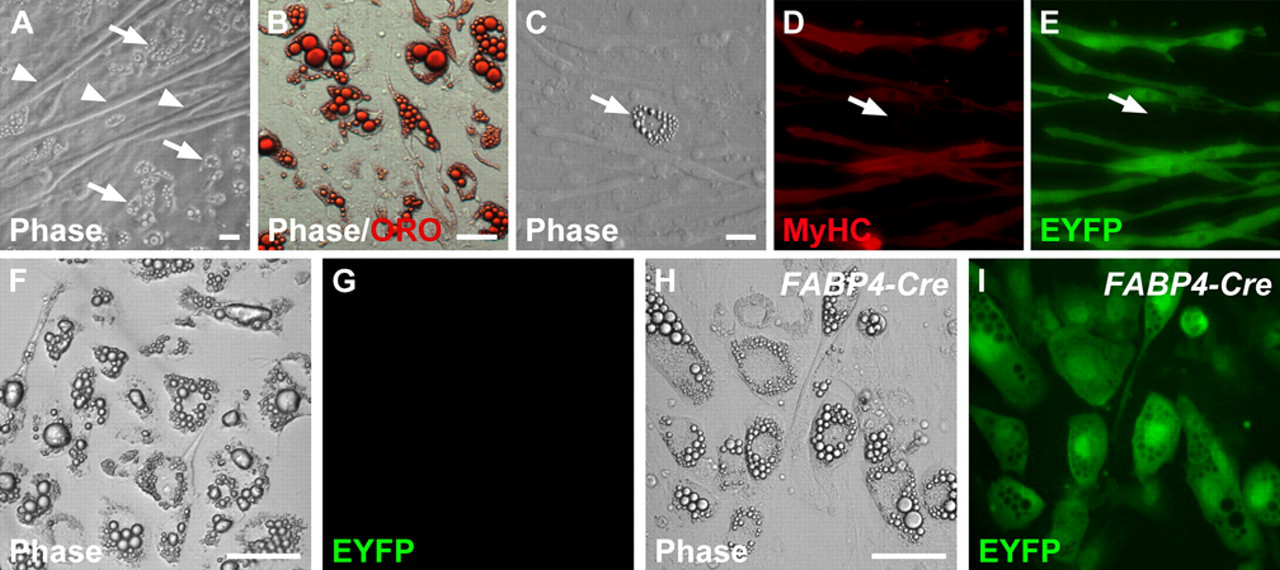
EYFP fluorescence is restricted to myogenic cells in 14-day extensor digitorum longus (EDL) single muscle fiber cultures derived from MyoD iCre/+;R26R EYFP/+ muscle. (A) Region of a culture showing both muscle fibers (arrowheads) and adipogenic cells (arrows). (B) Oil-Red-O (ORO) staining of an exclusively adipogenic region of the culture reveals large, multilocular fat droplets, which typify adipocytes in these cultures. (C-E) Myotubes express both MyHC and EYFP, whereas adipocytes (arrow) express neither. (F, G) An adipocyte-rich region of a culture showing the lack of EYFP fluorescence. (H, I) Control culture derived from FABP4-Cre;R26R EYFP/+ muscle showing robust expression of EYFP in adipocytes. Scale bars: 10 µm.
The results above demonstrate that EYFP+ satellite cells, which represent 98% of Pax7+ satellite cells on freshly isolated EDL fibers (Kanisicak et al. 2009), do not undergo adipogenic differentiation under the growth conditions employed. These data, however, did not address the possibility that adipocytes in single fiber cultures are derived from a small subpopulation of satellite cells that express Pax7 but had not expressed MyoD in their developmental history (Kanisicak et al. 2009) and were therefore EYFP–. To quantitatively assess whether the small population of Pax7+;EYFP– satellite cells on freshly isolated EDL fibers could account for the adipocytes observed in single fiber culture, we undertook a clonal analysis to determine the percentage of fiber-associated cells with adipogenic potential. Individual satellite cell cultures were established by first isolating single EDL muscle fibers from MyoDiCre/+;R26REYFP/+ mice, followed by mechanical disengagement of the satellite cells from their sublaminar position using a 20-gauge needle and a 1-ml syringe (Shefer et al. 2004; Shefer and Yablonka-Reuveni 2005). Single satellite cells were placed in individual wells of 24-well Matrigel-coated culture dishes and maintained in our standard fiber culture medium. Cultures were fixed on day 4 and processed for Pax7 or MyoD immunofluorescence or were grown until day 14 or 28 for analysis of adipogenic and myogenic differentiation. Data from the clonal analysis are summarized in Table 1. A total of 435 clones were produced from 74 individual muscle fibers (5.9 clones per EDL fiber). The majority of clones (267 of 435; 61.4%) were classified as non-myogenic based on the absence of Pax7 staining in 4-day cultures (Fig. 3A,B) or the presence of adipocytes in day 14 and 28 cultures (Fig. 3C,D; data not shown). For comparison, Shefer et al. (2004) observed 58% non-myogenic clones using similar methods for fiber isolation and clonal cultures. At least one non-myogenic clone was produced from 72 of the 74 fibers processed, demonstrating that this phenomenon was not restricted to a subpopulation of fibers and was consistent across all experiments. None of the non-myogenic clones were EYFP+ and had thus never expressed the MyoDiCre allele (Fig. 3A-D; Table 1). Based on ORO staining of day 14 and 28 cultures, all of the non-myogenic clones were classified as adipogenic. In total, 127 of 435 clones (29.2%) were classified as myogenic based on expression of MyoD or Pax7 after 4 days in culture (Fig. 3E-H) or, when cultured for 14 or 28 days, differentiation into multinucleated myotubes (Fig. 3I,J) and expression of MyHC (not shown). One hundred percent of these myogenic clones were EYFP+, and EYFP+ clones were exclusively myogenic. A minority of wells (41 of 435; 9.4%) contained both myogenic and non-myogenic clones. However, we found that all of the myogenic cells were EYFP+ and co-expressed Pax7 on day 4 or differentiated into myotubes on days 14 and 28, whereas the non-myogenic cells were EYFP–. These results are most likely the result of occasional inclusion of two cells per well, with only one being a satellite cell. Similar results were obtained with single muscle fiber and clonal cultures from the predominantly slow-twitch soleus muscle (not shown). We cannot determine whether the small starting population of Pax7+;EYFP– satellite cells (Kanisicak et al. 2009) remained EYFP– throughout the course of the experiments. As such, there remains a formal possibility that these rare Pax7+;EYFP– cells exhibit adipogenic potential. Nevertheless, these studies clearly demonstrate that the great majority of adipogenic cells, which were EYFP– without exception, are not derived from Pax7+ satellite cells.
Analysis of Satellite Cell Clones Prepared with Different Muscle Fiber Isolation Methods
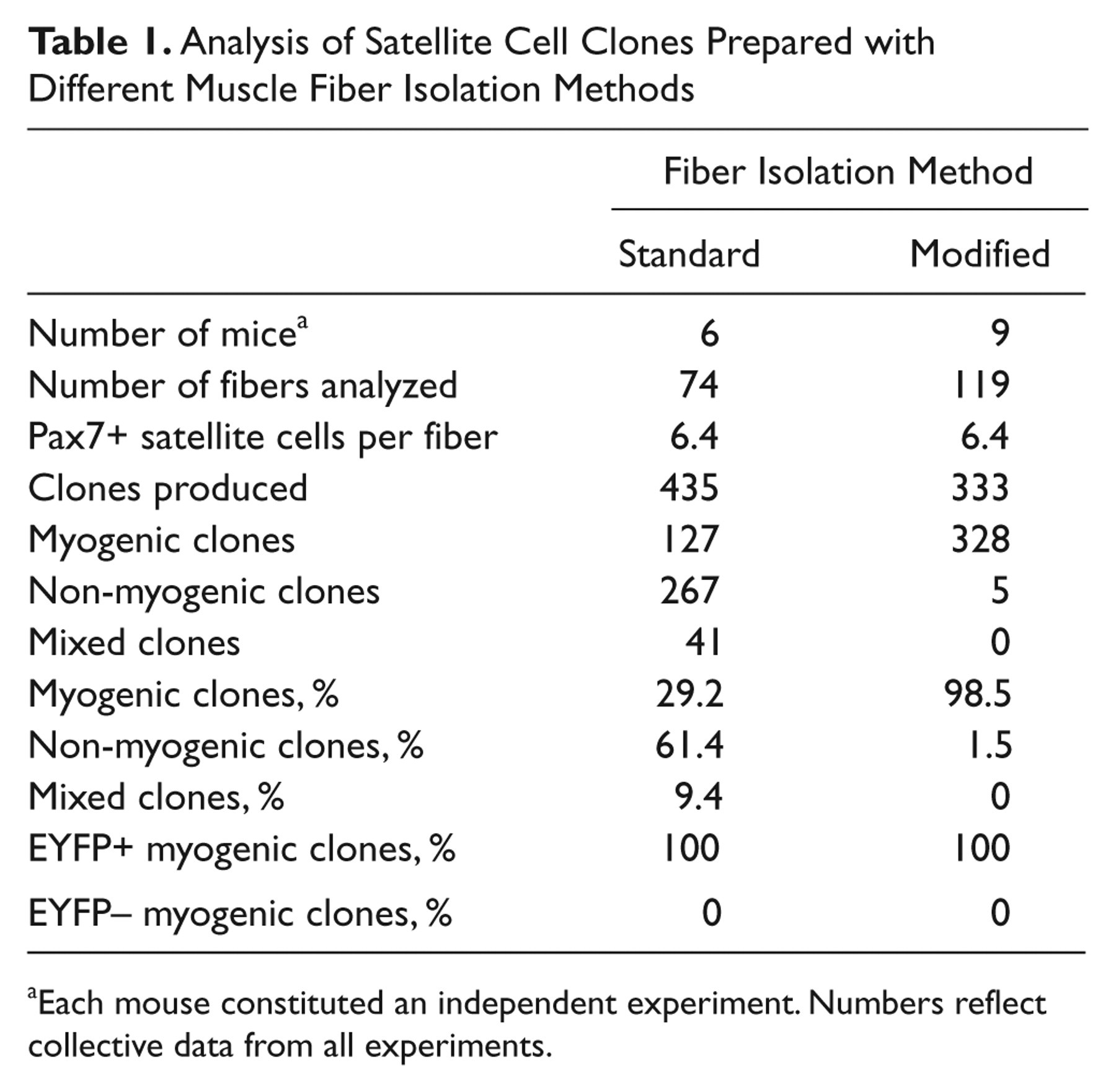
Each mouse constituted an independent experiment. Numbers reflect collective data from all experiments.
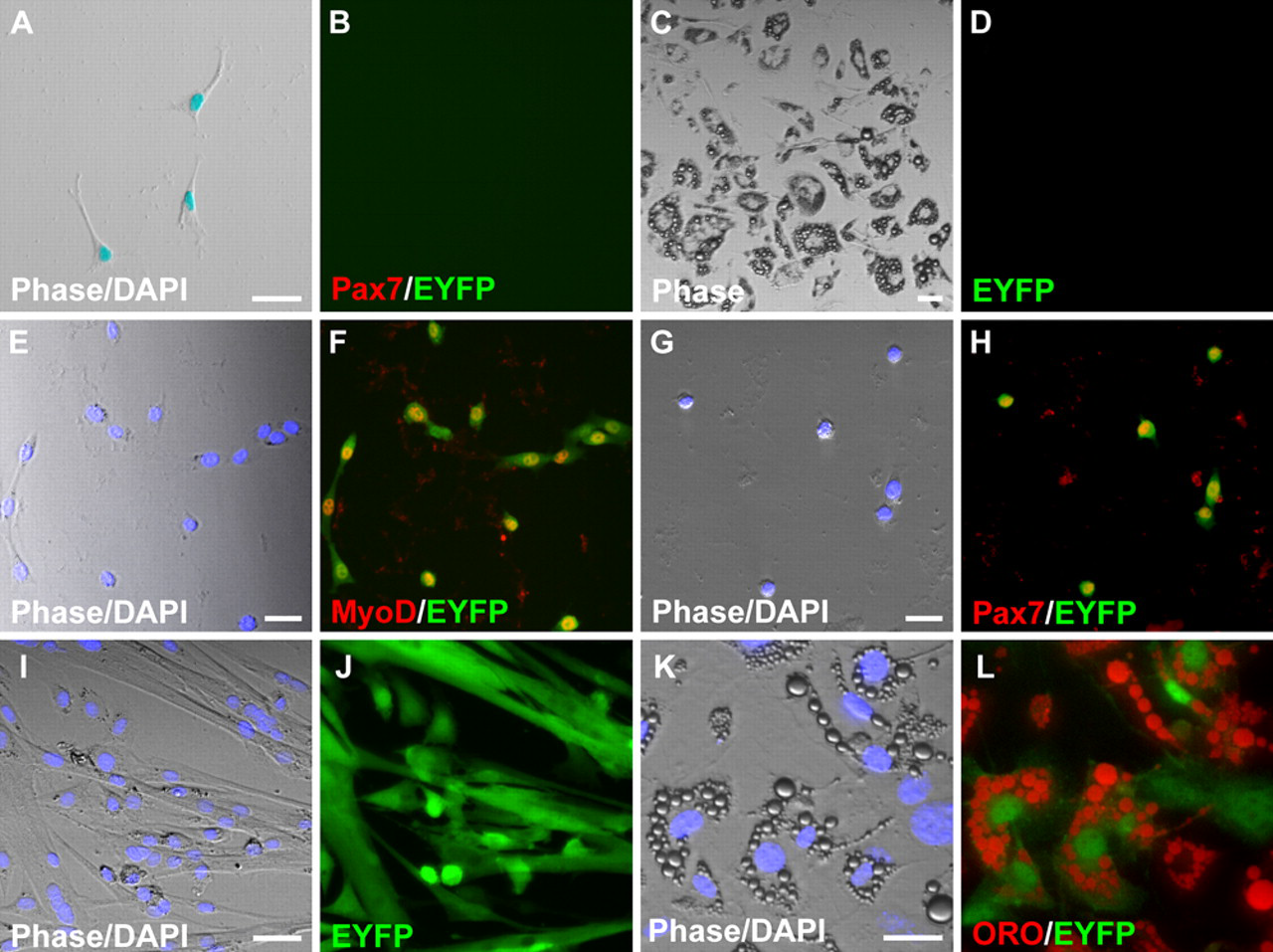
Myogenic programming of satellite cells in clonal cultures. (A-J) Cultures derived from MyoD iCre/+;R26R EYFP/+ muscle. (A, B) Non-myogenic Pax7−;EYFP– clone in a day 4 culture. (C, D) Adipogenic clone in a 14-day culture. All adipocytes are EYFP–. (E-H) EYFP+ myogenic clones in 4-day cultures stained for MyoD (E, F) or Pax7 (G, H). Yellow nuclei in merged images demonstrate co-expression of EYFP and the myogenic markers. (I, J) Differentiated myogenic clone in a 14-day culture. All myotubes are EYFP+. (K, L) The majority of ORO+ adipocytes in cultures derived from Tie2-Cre;R26R EYFP/+ muscle are EYFP+. Scale bars: 10 µm.
Adipocytes Are Derived from Contaminating Cells That Co-purify with Muscle Fibers
Because of the large proportion of unrecombined non-myogenic clones observed in our muscle fiber and clonal cultures, we hypothesized that there were unlabeled, contaminating cells being transferred to the cultures with the muscle fibers. To evaluate this possibility, a minor modification was made to the muscle fiber isolation procedure (Shefer et al. 2004; Shefer and Yablonka-Reuveni 2005), whereby fibers isolated as above were subjected to two additional transfers into horse serum–coated 60-mm dishes containing DMEM before plating the individual muscle fibers or isolating single satellite cells from muscle fibers for clonal analysis. Inclusion of these additional wash steps (modified fiber isolation procedure) did not alter the average number of Pax7+ satellite cells per EDL fiber (Table 1; n = 31 fibers; p = 0.97). To determine the proportion of mononuclear cells co-expressing EYFP and Pax7 after 4 days in culture, individual EDL muscle fibers (n = 16) were isolated from MyoD iCre/+;R26R EYFP/+ mice, and mononuclear cells were assayed by immunofluorescence. All (n = 1323) of the mononuclear cells analyzed co-expressed EYFP and Pax7, indicating that inclusion of additional fiber wash steps removed the large number of Pax7−;EYFP– cells observed in single muscle fiber cultures described above. For clonal analysis, 333 clones from 119 EDL muscle fibers were evaluated, and the data are summarized in Table 1. We found that 98.5% of the clones were myogenic, based on expression of Pax7 on day 4 or MyHC expression and myotube formation on days 14 and 28. As expected, all myogenic clones were EYFP+, whereas the 5 non-myogenic clones observed were EYFP– and comprised cells that were Pax7– at day 4 or contained ORO+ adipocytes on day 14. Day et al. (2010) also observed a reduced frequency of non-myogenic cells in single fiber culture when additional fiber wash steps were incorporated. Careful inspection of muscle fibers prepared using methods on which our standard isolation procedure was based did not reveal adherent contaminating cells (Shefer et al. 2004). However, without the additional washing steps, non-adherent cells with adipogenic potential are present in the fiber suspension medium prior to plating (data not shown), and the unavoidable transfer of small quantities of this media to the culture dishes during fiber plating is likely responsible for adipogenic cells in these fiber cultures.
Tie2 expression identifies a tumor-associated population of pericyte-like cells with adipogenic potential in culture (De Palma et al. 2005), and Tie2-Cre-labeled cells in skeletal muscle respond to BMP2 stimulation and contribute to heterotopic ossification in vivo (Lounev et al. 2009). We used Tie2-Cre transgenic mice (Kisanuki et al. 2001) to test whether a Tie2+ progenitor with adipogenic potential resides in skeletal muscle tissue. EDL muscle fibers were isolated from Tie2-Cre;R26R EYFP/+ mice using the standard isolation procedure, and cells from 14-day cultures were assayed for EYFP fluorescence and ORO staining. A large proportion (>68%; 230 of 337) of the multilocular, ORO+ adipocytes observed in 14-day muscle fiber cultures were EYFP+ (Fig. 3K,L), indicating that a substantial proportion of the contaminating adipogenic cells in these fiber cultures derive from Tie2+ precursors. Whether EYFP– adipocytes originate from a distinct progenitor population or reflect inefficient Cre-mediated recombination of the reporter allele remains to be determined.
Satellite Cells Mutant for MyoD or Myf-5 Do Not Spontaneously Adopt an Adipogenic Fate
We also investigated the adipogenic potential of satellite cells derived from MyoD-null (Rudnicki et al. 1992) and Myf-5-null (Kassar-Duchossoy et al. 2004) mice. A number of studies have documented the myogenic capacity of satellite cells in these mutant genetic backgrounds, although the extent and kinetics of myogenic differentiation varied (Yablonka-Reuveni et al. 1999; Cornelison et al. 2000; Schuierer et al. 2005; Gayraud-Morel et al. 2007; Ustanina et al. 2007). Previous studies, however, did not exclude the possibility of fate changes within a subpopulation of the satellite cell pool, which is known to be heterogeneous (Zammit 2008). To evaluate this possibility, single EDL myofibers were isolated from MyoD iCre/-;R26R EYFP/+ and Myf-5loxP/loxP ;MyoD iCre/+;R26R EYFP/+ mice, and satellite cells were assayed for expression of Pax7 and EYFP by immunofluorescence on freshly isolated, fixed fibers and after 4 days in culture. Consistent with previous reports (Yablonka-Reuveni et al. 1999; Cornelison et al. 2000; Gayraud-Morel et al. 2007; Kanisicak et al. 2009), a greater number of satellite cells were associated with freshly isolated MyoD-mutant fibers than with fibers derived from phenotypically wild-type (MyoD iCre/+;R26R EYFP/+) littermates (11.9 vs 6.0; p < 0.001), whereas the number of satellite cells per fiber was not increased in Myf-5-mutant fibers that harbored a single wild-type MyoD allele (5.6 vs 5.2; p > 0.05). Both on freshly isolated fibers (not shown) and in 4-day cultures, virtually all of the Pax7+ mononuclear cells assayed from mice lacking MyoD (Fig. 4A-D; n = 368) or Myf-5 (Fig. 4E-H; n = 362) co-expressed EYFP, demonstrating the efficiency of MyoD iCre -dependent satellite cell labeling in these mutant backgrounds. Differentiated cultures of MyoD iCre/-;R26R EYFP/+ fibers prepared by the modified method to eliminate contaminating adipogenic progenitors contained extensive networks of EYFP+;MyHC+ muscle fibers by day 21 in culture, as well as EYFP+ mononuclear cells that were weak or negative for MyHC (Fig. 4I-L). These mononuclear cells most likely represent reserve cells (Yoshida et al. 1998; Day et al. 2007) or reflect the delayed differentiation kinetics of MyoD-mutant satellite cells (Yablonka-Reuveni et al. 1999; Cornelison et al. 2000; Schuierer et al. 2005). Of note, no ORO+ adipogenic cells (EYFP+ or EYFP–) were observed in satellite cell cultures derived from a total of 48 EDL fibers in three independent experiments. Similar results were obtained with Myf-5-mutant fibers that were heterozygous for MyoD (data not shown).
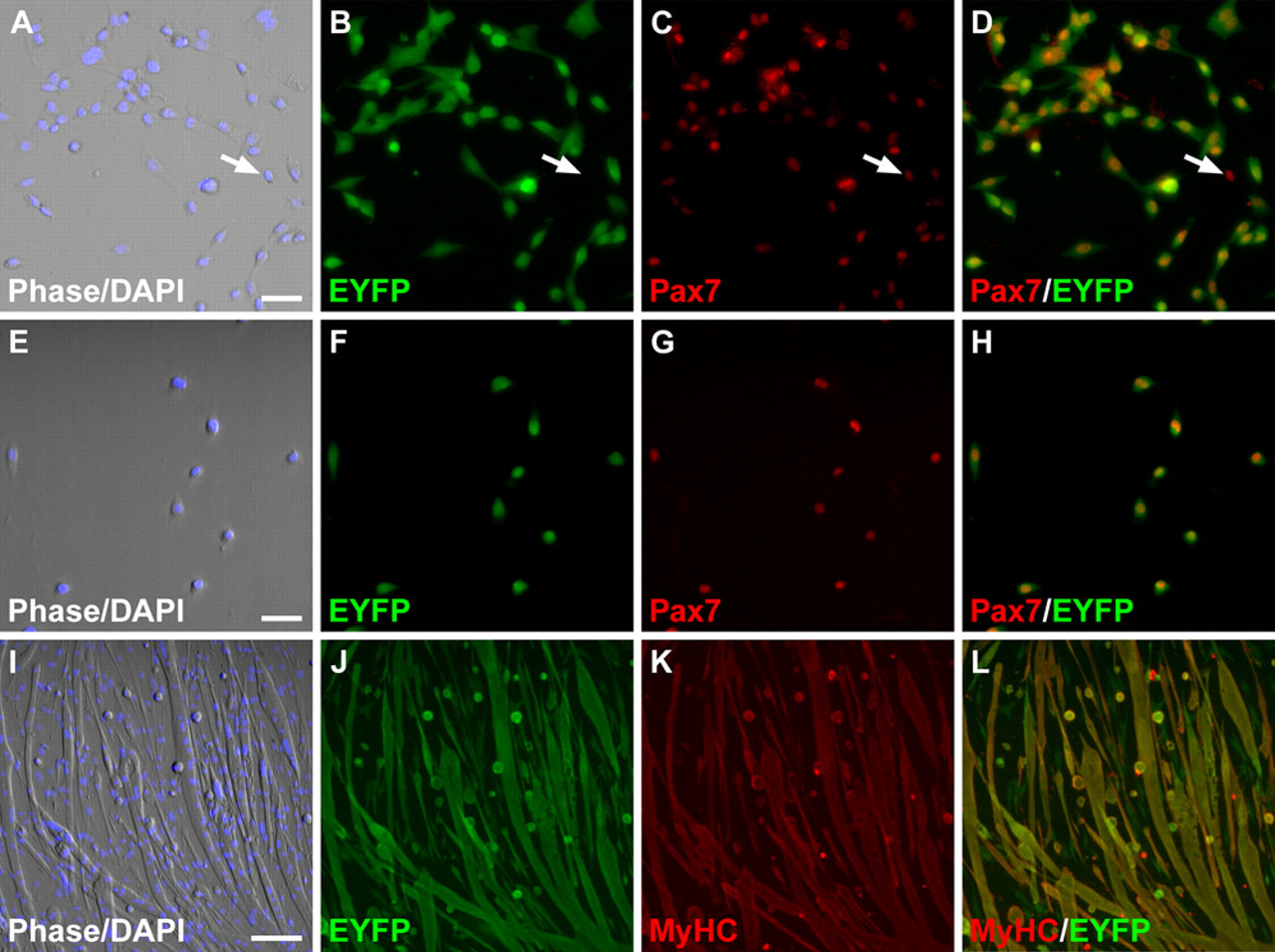
Myogenic programming is maintained in satellite cells mutant for MyoD or Myf-5. (A-H) Examples of 4-day single fiber extensor digitorum longus (EDL) cultures derived from MyoD iCre/-;R26R EYFP/+ (A-D) or Myf-5 loxP/loxP ;MyoD iCre/+;R26R EYFP/+ (E-H) muscle. Immunofluorescence images show co-localization of Pax7- and EYFP-expressing cells. The arrows in panels A to D identify a rare Pax7+;EYFP– cell. (I-J) A 21-day single fiber EDL culture derived from MyoD iCre/–;R26R EYFP/+ muscle. Cultures at this stage comprise predominantly MyHC+;EYFP+ fibers, although a few mononuclear MyHC-EYFP+ cells are interspersed throughout the culture (see text for details). No ORO+ adipogenic cells were observed in these cultures. Scale bars: A-H, 10 µm; I-L, 50 µm.
Satellite Cells Can Be Stimulated to Accumulate Lipid but Do Not Undergo Terminal Adipogenic Differentiation
Data presented thus far show that mouse satellite cells do not spontaneously enter the adipogenic pathway under standard growth conditions. Yet, under adipogenic conditions, cultured satellite cells/myoblasts are known to accumulate lipid and express adipogenic-specific genes (Teboul et al. 1995; Asakura et al. 2001; Guan et al. 2002; Taylor-Jones et al. 2002; Wada et al. 2002; De Coppi et al. 2006). As mice used in the present study were on an enriched FVB genetic background, which is distinct from that used in previous studies, we sought to determine whether satellite cells were resistant to adipogenic conversion, which might explain their inability to spontaneously adopt an adipogenic phenotype. Muscle fibers were isolated using the modified isolation procedure to eliminate contaminating cell types and cultured for 7 days in standard medium, followed by culturing in adipogenesis-inducing medium (100 µM GLA in 10% FBS;DMEM) for an additional 10 days (Wada et al. 2002). Induced cultures were immunostained for EYFP followed by ORO staining. Satellite cells expressing EYFP readily accumulated ORO+ lipid droplets, giving them an adipocyte-like morphology (Fig. 5A,B). Interestingly, however, expression of MyoD and myogenin was not repressed (Fig. 5C-F), indicating that cells treated with adipogenic media retained myogenic programming, consistent with previous findings (Taylor-Jones et al. 2002; Uezumi et al. 2010). To further investigate the adipogenic potential of MyoDiCre -labeled satellite cells, we exposed the cells to a second adipogenesis-inducing cocktail (IDX) shown to promote terminal adipogenic differentiation in 3T3-L1 preadipocytes (MacDougald and Lane 1995; Cowherd et al. 1999; Guan et al. 2002). The IDX induction procedure also resulted in significant accumulation of lipid in EYFP+ satellite cell progeny. As in the case of GLA-induced cultures, the IDX-induced, ORO+ cells also expressed MyoD (Fig. 5G,H) and myogenin (Fig. 5I,J). We also found that although mixed cultures containing contaminating adipocytes robustly expressed the terminal adipocyte differentiation markers, adipsin and FABP4, apparently pure satellite cell cultures grown under adipogenesis-inducing conditions did not (Fig. 5K). These results indicate that the EYFP+ satellite cell progeny, while accumulating lipid, did not undergo complete adipogenic differentiation. Our experiments also show that ORO positivity alone is not an adequate marker of adipogenic differentiation and that it should be used in conjunction with markers of terminal adipogenic differentiation.
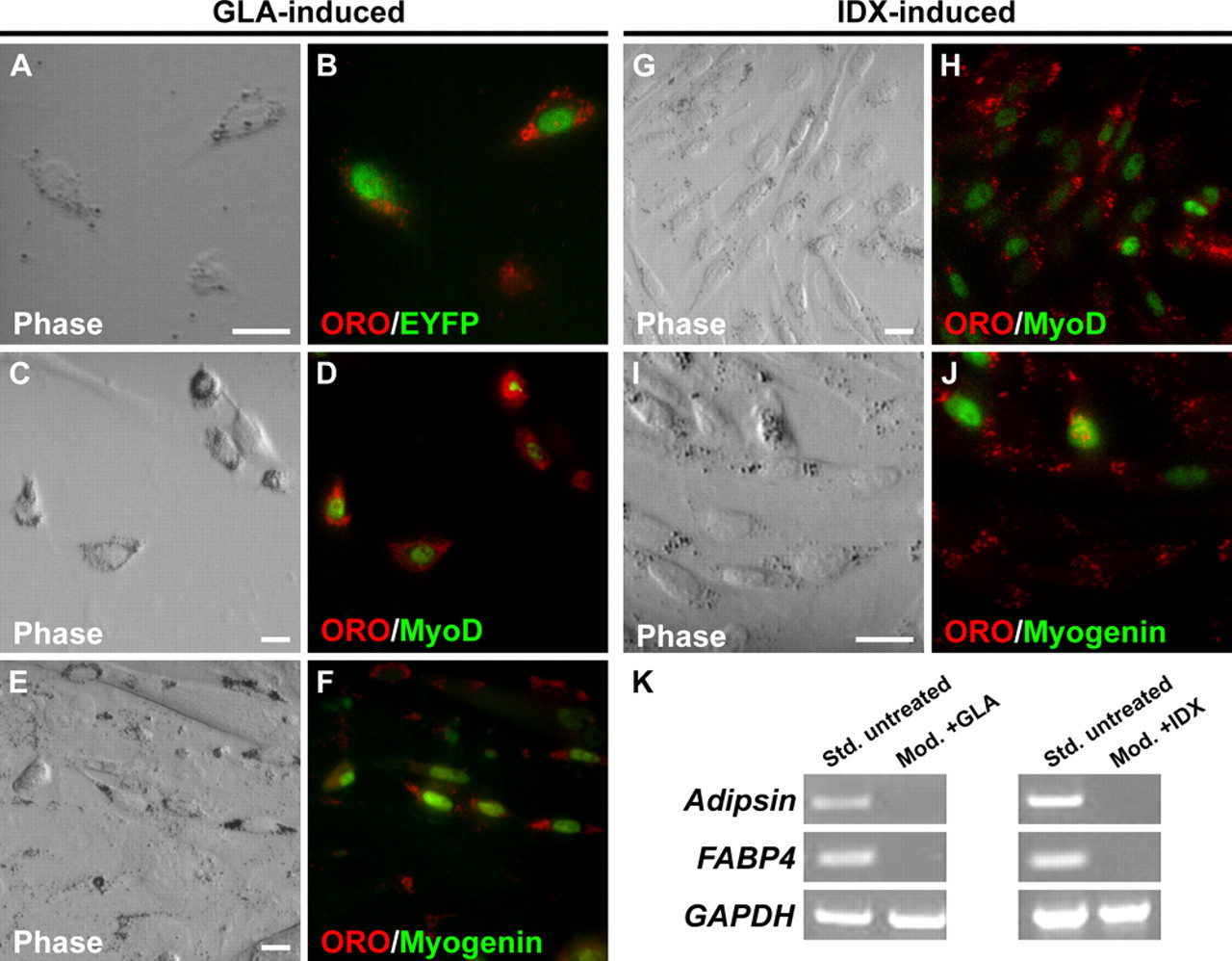
Satellite cells exposed to adipogenesis-inducing reagents accumulate lipid but do not terminally differentiate. Cultures were prepared using the modified fiber isolation procedure to eliminate contaminating adipocytes. (A, B) Single fiber extensor digitorum longus (EDL) culture derived from MyoD iCre/+;R26R EYFP/+ muscle showing ORO+ lipid accumulation in EYFP+ cells exposed to γ-linolenic acid (GLA). (C-J) Single fiber cultures derived from wild-type muscle. Both GLA- and insulin, dexamethasone, isobutylmethylxanthine (IDX)-treated satellite cells express MyoD (C, D, G, H) and myogenin (E, F, I, J). Scale bars: 10 µm. (K) RT-PCR analysis of adipocyte terminal differentiation marker expression in standard (Std., untreated) and modified (Mod. + GLA or IDX) cultures.
Discussion
In the present study, we used Cre/loxP-based methods to permanently and specifically label satellite cells from adult skeletal muscle so that their developmental potential could be assessed in culture without the potential confounding influence of co-purifying, non-myogenic contaminating cells. Although the robust myogenic potential of satellite cells has been well established for several decades, more recent studies documented the presence of adipogenic (Asakura et al. 2001; Csete et al. 2001; Shefer et al. 2004) and osteogenic (Asakura et al. 2001) cells in single fiber cultures that were concluded to be of satellite cell origin. We used MyoD iCre knockin mice, which can be used to permanently label essentially all satellite cells in adult skeletal muscle (Kanisicak et al. 2009), to reinvestigate satellite cell developmental potential in single fiber cultures.
We demonstrated that satellite cells from young adult mice are committed to myogenesis and do not spontaneously adopt adipogenic or other non-myogenic fates in culture. Because rare Pax7+;EYFP– satellite cells associated with EDL fibers (2% of Pax7+ satellite cells; Kanisicak et al. 2009) could not be traced, we cannot rule out the formal but unlikely possibility that EYFP– satellite cells represent a distinct population that has adipogenic potential. Clearly, however, the vast majority and perhaps all adipocytes in single fiber cultures are the result of co-purification of adipocyte progenitors, which can be removed by inclusion of additional serial wash steps prior to plating. Our experiments did not distinguish whether contaminating cells were loosely adherent to the fibers or were transferred to cultures in the bulk fluid that accompanies fiber plating, although careful inspection of fibers isolated by similar methods did not reveal the presence of adherent contaminating cells (Shefer et al. 2004). These data highlight the potential difficulty of obtaining uncontaminated muscle fiber preparations, particularly for downstream applications that require large-scale fiber isolation, such as fluorescence-activated cell sorting (FACS) analysis of fiber-associated cells (Sherwood et al. 2004).
The absence of MyoDiCre -dependent labeling of adipocytes in single fiber culture and in clonal cultures of FACS-isolated cells (Kanisicak et al. 2009) indicates that adipogenic cells arise from a lineage distinct from the myogenic lineage and is consistent with recent FACS analyses, which identified non-myogenic progenitor cells in the muscle interstitium capable of adipogenic differentiation (Joe et al. 2010; Uezumi et al. 2010). Adipogenic progenitors isolated from skeletal musculature by these groups share the marker profile, CD31-CD45-Sca1+CD34+PDGFRα+, although whether they represent identical cell populations is not yet clear. RT-PCR analysis also revealed expression of Tie2 (Uezumi et al. 2010), a receptor tyrosine kinase for angiopoietins 1 and 2 that is expressed by endothelial cells and subsets of hematopoietic cells and mesenchymal progenitors (De Palma et al. 2005). Interestingly, more than half of the adipocytes in our fiber cultures derived from Tie2-Cre;R26R EYFP/+ muscle were EYFP+, raising the possibility that these cells are related to adipogenic cells identified by independent means. Labeled adipocytes in culture likely were derived from Tie2+ progenitors in the musculature rather than resulting from Tie2 induction after plating because Tie2-Cre-labeled adipocytes were observed in vivo following intramuscular injection of FACS-fractionated cells derived from Tie2-Cre;R26NG/+ (Yamamoto et al. 2009) musculature (unpublished observations). Although low-level Tie2 expression has been detected in satellite cells of Tie2-GFP transgenic mice (Motoike et al. 2000) by FACS (Abou-Khalil et al. 2009), we have not observed Tie2-Cre-dependent labeling of cells of the myogenic lineage, including embryonic myoblasts, quiescent and proliferative satellite cells, mature muscle fibers, and nascent muscle fibers following cardiotoxin-induced injury of the tibialis anterior muscle (data not shown), further confirming that adipocytes in our cultures derive from a distinct, non-myogenic lineage.
MyoD, Myf-5, and MRF4 collaborate to establish the myogenic lineage (Rudnicki et al. 1993; Kassar-Duchossoy et al. 2004), and expression of either MyoD or Myf-5 alone is sufficient to establish myoblast identity and support near-normal embryonic myogenesis (Braun et al. 1992; Rudnicki et al. 1992; Rudnicki et al. 1993; Kablar et al. 2003). Satellite cell progenitors in the embryo transit through a MyoD+ stage (Kanisicak et al. 2009), consistent with the view that satellite cells become committed to the myogenic program prenatally. However, MyoD and Myf-5 are induced in satellite cells as an early response to muscle injury (Zammit 2008), and it is formally possible that this induction is required to establish or maintain satellite cell myogenic programming. Interestingly, Myf-5-null mice exhibit adipocyte accumulation and fibrosis following freeze-injury (Gayraud-Morel et al. 2007), although the cellular origin of these non-myogenic cell types is uncertain. We showed here that satellite cells deficient for either MyoD or Myf-5 maintain myogenic programming in culture, as all EYFP+ cells from MyoDiCre/– ;R26REYFP/+ and Myf-5loxP/loxP ;MyoDiCre/+ ;R26REYFP/+ muscle were myogenic. The MyoDiCre allele is active in MyoD and Myf-5-mutant mice, and satellite cells are labeled at the time of fiber isolation (Kanisicak et al. 2009), eliminating the possibility that EYFP– adipocytes were derived from satellite cells that failed to activate the MyoD locus in these mutant backgrounds. Whether functional compensation between MyoD and Myf-5 explains persistent myogenic programming in these mutant backgrounds remains to be explored.
At present, there is no clear evidence that satellite cells can undergo permanent fate changes to non-myogenic lineages. We showed here that satellite cells exposed to adipogenesis-inducing medium fail to express terminal markers of adipogenic differentiation and maintain expression of myogenic regulatory factors, consistent with previous findings (Taylor-Jones et al. 2002; Uezumi et al. 2010). Although satellite cell-derived myoblasts from aged mice show a greater propensity for adipogenic differentiation than younger myoblasts, they also do not fully execute the adipogenic program, and myogenic gene expression is not effectively repressed (Guan et al. 2002). Chronic states of hyperglycemia are known to stimulate de novo lipogenesis in muscle fibers (Aas et al. 2004; Guillet-Deniau et al. 2004). In this regard, lipid accumulation in satellite cells cultured under high glucose conditions in adipogenic medium probably reflects a metabolic response to hyperglycemic conditions rather than a lineage conversion from myogenic to adipogenic fates. In addition, whereas myoblasts treated with BMP2 or BMP4 induce expression of osteogenic markers such as alkaline phosphatase and osteocalcin and downregulate myogenic markers (Katagiri et al. 1994; Katagiri et al. 1997; Asakura et al. 2001; Wada et al. 2002), maintenance of the osteogenic phenotype and repression of the myogenic phenotype is dependent on the continual presence of BMPs (Katagiri et al. 1994). These observations imply that although adipogenic media and BMPs are potent modulators of muscle cell phenotype in culture, satellite cells/myoblasts maintain a memory of their committed myogenic state. Although the effect of pro-adipogenic conditions on satellite cell fate and phenotype in vivo remains unclear, neither quiescent nor activated satellite cells contribute appreciably to heterotopic ossification following intramuscular injection of BMP2 (Lounev et al. 2009).
Our study did not investigate satellite cell potential in vivo or the pathophysiological role of satellite cells, if any, in diseases and conditions characterized by increased inter- and intramuscular fatty infiltrates. Duchenne muscular dystrophy patients with advanced disease exhibit pronounced fatty infiltrates and fibrosis of unknown etiology in skeletal muscle. Intramuscular accumulations of adipocytes of unknown origin also are observed in age-related sarcopenia, obesity, and type 2 diabetes and are associated with a decline in measures of muscle function (Pahor and Kritchevsky 1998; Goodpaster and Wolf 2004; Visser et al. 2005; Delmonico et al. 2009). In addition, satellite cells exhibit a Wnt-dependent increased capacity for fibroblastic differentiation in culture when exposed to aged serum, providing a possible explanation for increased fibrosis of aged muscle (Brack et al. 2007). In vivo lineage analyses in mouse models will address the role of satellite cells in the accumulation of non-myogenic cell types associated with skeletal muscle in aging and disease.
Footnotes
Acknowledgements
We thank Cathy Cogswell for expert assistance with maintenance of the mouse colony, members of the Goldhamer lab for helpful discussions during the course of this work, Dr. Shahragim Tajbakhsh (Pasteur Institute) for Myf-5loxP mice, Dr. Michael Rudnicki (Ottawa Health Sciences Centre) for MyoD knockout mice, and Dr. Tom Sato (UT Southwestern) for Tie2-Cre transgenic mice.
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: This work was funded by grants to DJG from the National Institutes of Health (AR052777) and the Muscular Dystrophy Association.
