Abstract
Satellite cells are quiescent cells located under the basal lamina of skeletal muscle fibers that contribute to muscle growth, maintenance, repair, and regeneration. Mouse satellite cells have been shown to be muscle stem cells that are able to regenerate muscle fibers and self-renew. As human skeletal muscle is also able to regenerate following injury, we assume that the human satellite cell is, like its murine equivalent, a muscle stem cell. In this review, we compare human and mouse satellite cells and highlight their similarities and differences. We discuss gaps in our knowledge of human satellite cells, compared with that of mouse satellite cells, and suggest ways in which we may advance studies on human satellite cells, particularly by finding new markers and attempting to re-create the human satellite cell niche in vitro.
The satellite cell is defined by its location between the basal lamina and sarcolemma of skeletal muscle fibers. It was identified first in adult frog skeletal muscle (Mauro 1961) and was subsequently found in other vertebrates (Muir et al. 1965), including human (Laguens 1963; Shafiq et al. 1967).
Experiments in rodents demonstrated that satellite cells contribute to the growth and regeneration of skeletal muscle (Moss and Leblond 1971; Bischoff 1975; Cardasis and Cooper 1975a; Konigsberg et al. 1975; Schultz 1976; Snow 1978).
Normally quiescent in adult muscle, satellite cells become activated when muscle is injured and proliferate to generate a pool of muscle precursor cells (mpc) or myoblasts. These can then either repair damaged segments of fibers or fuse together to generate entirely new multinucleated muscle fibers. During the processes of proliferation and differentiation into myofibers, the satellite cell recapitulates the myogenic program that is not an exact recapitulation of muscle development, although the myogenic regulatory factors (MRFs) Myf5, MyoD, MRF4, and myogenin (Figure 1) [reviewed by Weintraub (1993)] are expressed in similar sequence in both processes.
Early interest in satellite cells and their progeny mpc was due to their role in muscle repair and regeneration, which is particularly relevant to the treatment of inherited muscle diseases. Recently, satellite cells have emerged as a model of a postnatal stem cell because the availability of markers and genetically modified mice has allowed investigators to follow in vitro and in vivo the processes of activation, proliferation, differentiation, and self-renewal.
Challenges in Studying Satellite Cells
Despite being of such interest, quiescent satellite cells are very difficult to study. Their scarcity and location under the basal lamina of muscle fibers makes them difficult to isolate. Another major problem relates to the difficulty encountered in attempting to separate them from other cells present within skeletal muscle (e.g., fibroblasts, endothelial cells, interstitial cells, and blood vessel-associated cells), in order to obtain a pure population of satellite cells. Because of these factors, performing experiments that require large numbers of satellite cells can be challenging, even in rodents. These difficulties are significantly compounded when studying human muscle, not only because of the practical difficulties in obtaining muscle biopsies to prepare cells but also because of the lack of markers to distinguish satellite cells from myonuclei and other cells present within skeletal muscle. It is because of these reasons that most of the work done so far is focused on rodents, particularly mice.
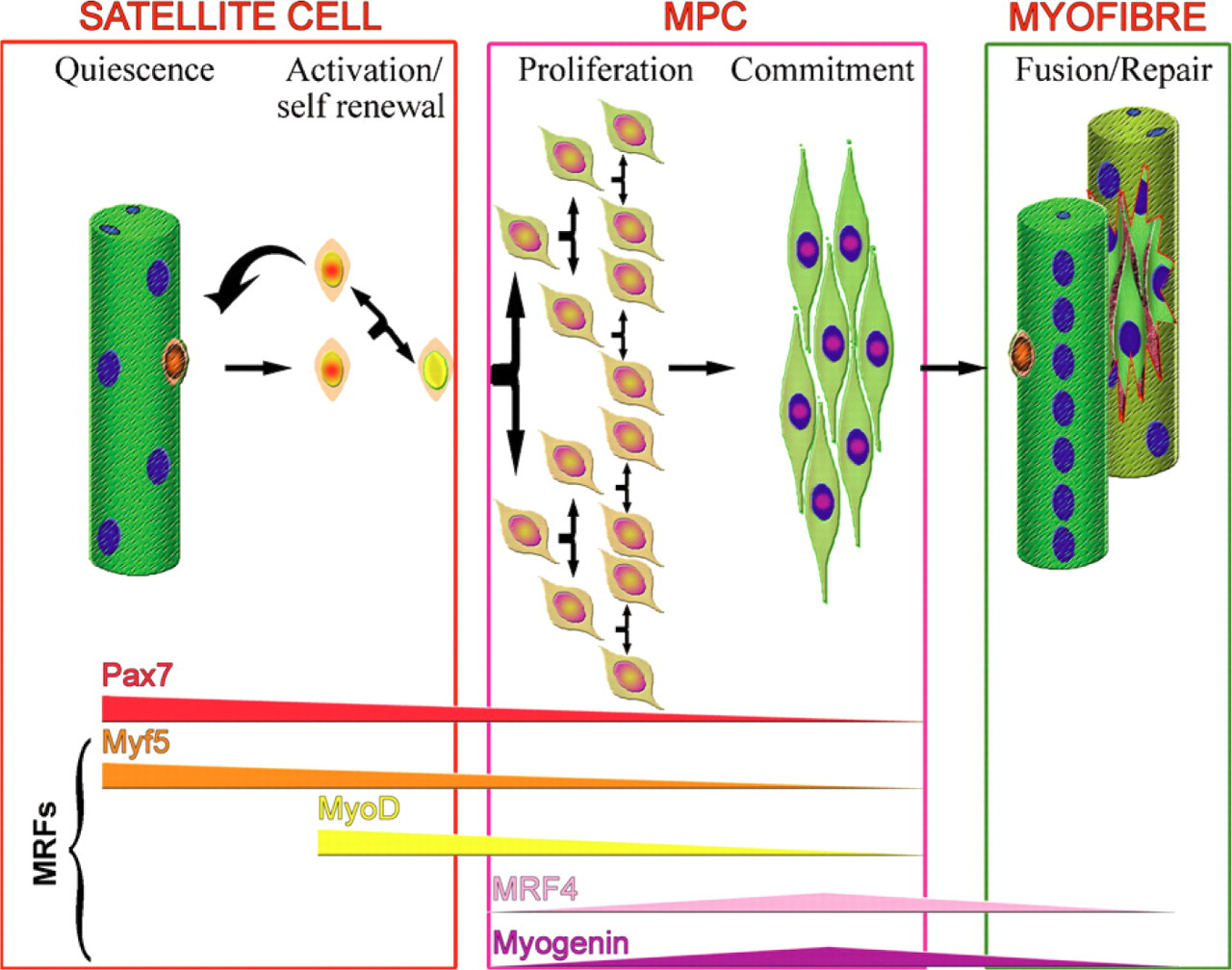
Model of satellite cell activation and progression through the myogenic program. Quiescent satellite cells, underneath the basal lamina of muscle fibers, express Pax7 and Myf5. Upon activation, they upregulate MyoD and divide to produce a pool of muscle precursor cells (mpc). Satellite cell progeny then follow one of two fates. They may down-regulate MyoD and self-renewto give rise to a Pax7+ satellite cell. Alternatively, they may differentiate, down-regulating Pax7, Myf5, and MyoD and expressing MRF4 and myogenin, eventually fusing either to form new or to repair damaged myofibers.
Satellite cells may be studied either in frozen sections of skeletal muscle, or following their isolation from fresh muscle, by growing muscle explants from which muscle cells, including mpc, migrate onto the culture dish substrate (Harvey et al. 1979; Garrett and Anderson 1995; Conboy and Rando 2002; Conboy et al. 2003; Smith and Merrick 2010), or by mincing and enzymatically disaggregating skeletal muscle (Naffakh et al. 1993; Partridge 1997; Yablonka-Reuveni et al. 1999a; Conboy et al. 2003; Montarras et al. 2005). Both methods give rise to a mixture of cell types, but do not necessarily release all cells from the muscle. Flow cytometry has been used to purify both mouse and human satellite cells on the basis of expression of marker proteins (Baroffio et al. 1996; Conboy et al. 2003, 2010; Sherwood et al. 2004; Montarras et al. 2005; Fukada et al. 2007; Pallafacchina et al. 2010), but this method does not give rise to completely pure populations of cells. In addition, enzymatic treatment removes cells from their niche and may strip or alter cell surface markers so that satellite cells prepared in this way may not completely maintain their in vivo phenotype.
An elegant method to prepare rodent satellite cells in their niche on the fiber was developed by Bischoff (1986) and used in many in vitro and in vivo studies (Rosenblatt et al. 1995; Shefer et al. 2004, 2006; Zammit et al. 2004; Collins et al. 2005; Yablonka-Reuveni et al. 2008; Boldrin et al. 2009). Isolated viable muscle fibers bearing their satellite cells under the basal lamina may be either fixed immediately or placed in suspension culture so that the activation, proliferation, differentiation, and self-renewal of satellite cells in their niche can be followed (Beauchamp et al. 2000; Zammit et al. 2004; Collins et al. 2007, 2009; Day et al. 2007; Gnocchi et al. 2009). Fibers may be cultured on a substrate, usually Matrigel (a commercially available mixture of basement membrane components and growth factors), or other substrates, such as collagen (Shefer et al. 2004); under these conditions, satellite cells migrate from the fiber, proliferate, and differentiate into myotubes (Rosenblatt et al. 1995; Blaveri et al. 1999; De Coppi et al. 2006; Yablonka-Reuveni and Anderson 2006; Boldrin et al. 2007; Malerba et al. 2009). Alternatively, single muscle fibers may be carefully washed to eliminate any extraneous cells, and satellite cells removed from them by either physical (Shefer et al. 2004; Collins et al. 2005; Boldrin et al. 2009) or enzymatic (Ono et al. 2009) methods.
Using this single fiber protocol, it is possible to obtain pure populations of satellite cells, albeit in relatively small number. However, a major limitation of this method is that it requires entire freshly dissected muscles to allow the isolated fibers to remain intact and is therefore not easily applicable to human muscle biopsies. Nevertheless, protocols using particularly short muscles (Bonavaud et al. 2002; De Coppi et al. 2006) or fiber fragments (Cardasis and Cooper 1975b) may be suitable for human muscle preparations.
Studies on quiescent satellite cells need to be performed as soon as possible after fiber or cell preparation as they become activated extremely rapidly (Wozniak et al. 2003; Zammit et al. 2004). On activation, satellite cells promptly enter the myogenic program (Cornelison and Wold 1997; Yablonka-Reuveni et al. 2008) and may not therefore retain the capabilities of quiescent satellite cells.
Identification of Satellite Cells
In early studies, satellite cells were identified by electron microscopy on the basis of their position between the basal lamina and sarcolemma of muscle fibers (Cardasis and Cooper 1975a), but this method is technically demanding and not suitable to study large portions of muscle. There is now a panel of reliable markers of satellite cells in mouse, but not in human. However, it must be borne in mind that some are expressed on activated and quiescent satellite cells, whereas others are also expressed on other cell types (Table 1).
The majority of mouse satellite cells can be defined by their expression of Pax7, CD34, caveolin, calcitonin receptor, β1-integrin, M-cadherin, α7-integrin, and nestin (Table 1), but only for the first five of these markers are there commercially available antibodies. Furthermore, it should be noted that nestin expression in quiescent satellite cells has been revealed only by means of green fluorescent protein (GFP) positivity in the nestin-GFP mouse.
In the Myf5nLacZ/+ mouse, which has nuclear-localizing β-galactosidase targeted to the Myf5 locus, quiescent and activated satellite cells are β-galactosidase positive. However, Myf5 protein expression has not been described in quiescent satellite cells, even though Myf5 transcripts can be detected in sorted mouse satellite cells (Day et al. 2007) (Table 1). Possible explanations may be either the protein level is too low to be detected, or instability of the protein, or simply the lack of a clear signal from the antibody used.
Because of the limitations highlighted previously, it is at present unclear whether all of these markers recognize satellite cells in human muscle (Table 1).
Satellite Cell Heterogeneity
There is clear evidence from mouse studies that satellite cells, both within the same muscle and even on the same fiber, are different in terms of their marker expression (Beauchamp et al. 2000; Montarras et al. 2005; Kuang et al. 2007) and/or function (Collins et al. 2005, 2007; Kuang et al. 2007; Sacco et al. 2008; Boldrin et al. 2009). It is also clear that numbers of satellite cells per fiber (Collins et al. 2005; Shefer et al. 2006; Zammit 2008; Ono et al. 2009) and capacity of satellite cells to differentiate in vitro, or contribute to muscle regeneration in vivo (Pavlath et al. 1998; Collins et al. 2005; Montarras et al. 2005; Ono et al. 2009), differ depending on which muscle is used for their isolation. These observations all derive from mouse muscle, and therefore, we do not know whether the human satellite cell pool is, as in the mouse, heterogeneous.
Quantification of Mouse Satellite Cells
Early ultrastructural studies of mouse muscles suggest that 30-35% of fiber nuclei are satellite cells at birth, declining to 5-7% in adults (Allbrook et al. 1971; Cardasis and Cooper 1975a; Schultz 1976). Subsequent studies using either electron microscopy or M-cadherin staining to identify satellite cells in adult mouse soleus muscles give similar numbers of satellite cells (4.6% and 3.4% of nuclei, respectively; Snow 1977; Reimann et al. 2004). Many later studies have relied on counting the number of satellite cells per fiber, based on expression of different marker proteins (Yablonka-Reuveni and Rivera 1994; Beauchamp et al. 2000; Zammit et al. 2004). Nevertheless, even using the same marker, there are differences in the estimated numbers of satellite cells per fiber between laboratories (Collins et al. 2005; Shefer et al. 2006) or even between experiments performed at different times in the same laboratory (Collins et al. 2005, 2007). These discrepancies may be due to age, sex, or strain of mouse.
Quantification of Human Satellite Cells
Comparison of ultrastructural data suggests that there are similar percentages of satellite cells in adult mouse and human muscles—4% ± 2% of all nuclei within the fiber basal lamina of human muscles (Schmalbruch and Hellhammer 1976), which is similar to the 5-7% for mouse satellite cells. More direct comparison is however difficult, as in the mouse, satellite cell number depends on the muscle in which they reside (Collins et al. 2005; Zammit 2008), whereas we lack details in the human.
Lack of specific satellite cell markers in the human has led to equivocal and sometimes contradictory reports on their presence and number in human muscle sections. The first antibodies used to identify satellite cells in sections of human skeletal muscle were Leu19 and NKH-1, which recognize CD56, or neural cell adhesion molecule (NCAM; Schubert et al. 1989; Illa et al. 1992; Belles-Isles et al. 1993). NCAM is expressed by quiescent human (Fidzianska and Kaminska 1995) and rat (Irintchev et al. 1994), but not mouse, satellite cells; mouse satellite cells only express NCAM when they become committed to differentiation (Capkovic et al. 2008). Despite NCAM expression not being satellite cell specific (Cashman et al. 1987; Schubert et al. 1989; Mechtersheimer et al. 1992) (Table 1), it has been extensively used for identification of satellite cells on sections of human muscle (Illa et al. 1992; Charifi et al. 2003; Kadi et al. 2006; Doppler et al. 2008; Mackey et al. 2009).
Markers of satellite cells
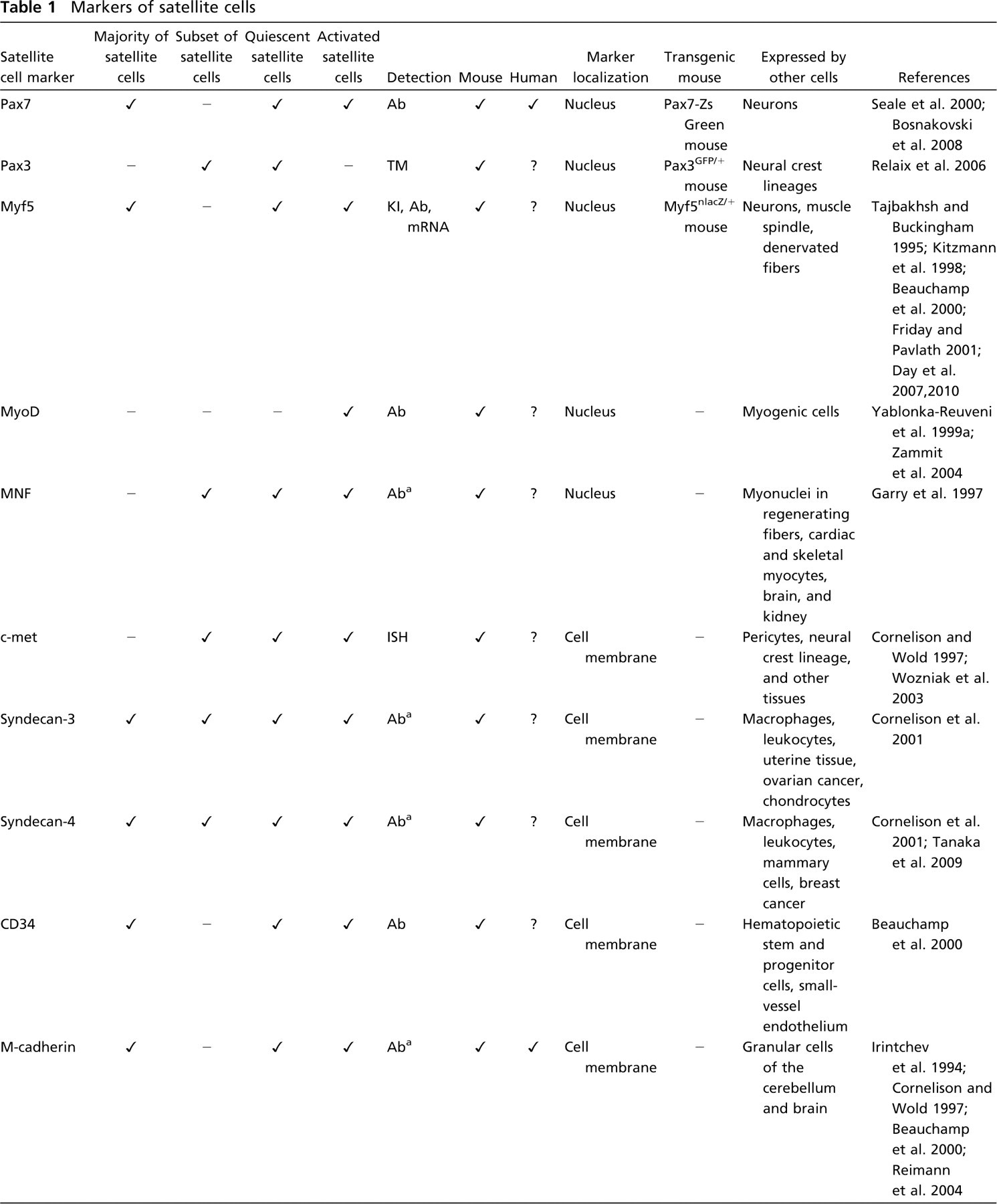
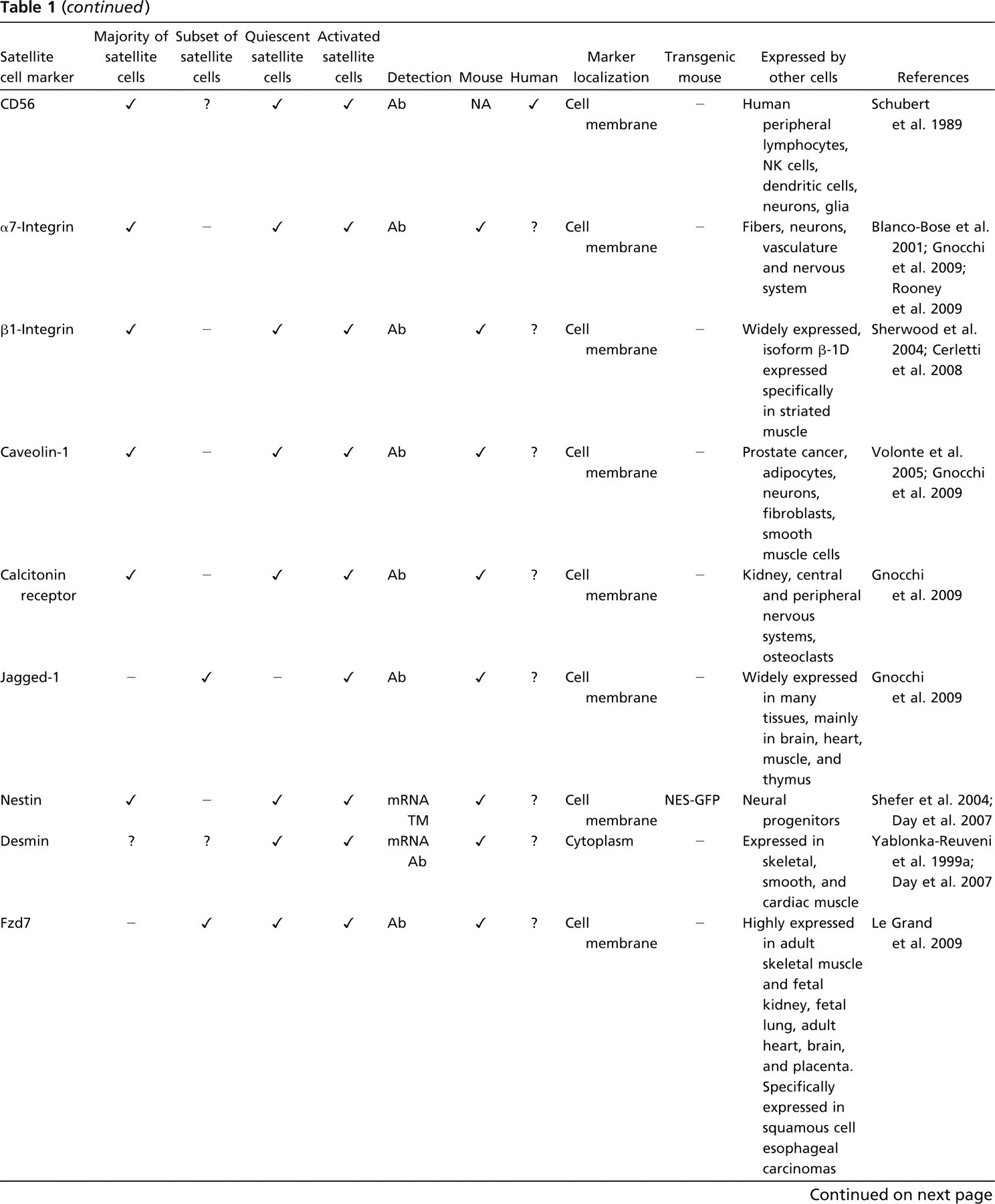
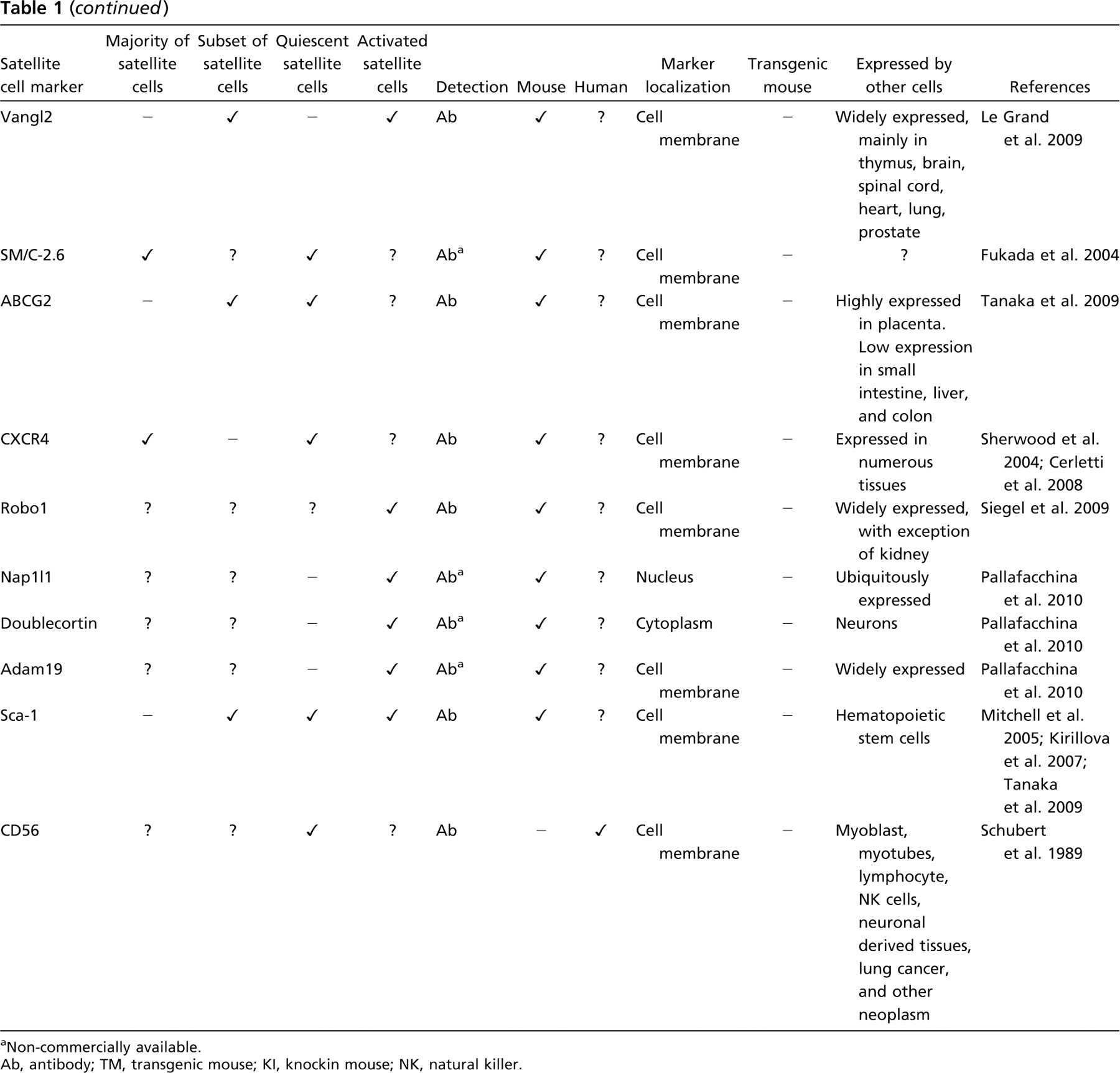
Non-commercially available.
Ab, antibody; TM, transgenic mouse; KI, knockin mouse; NK, natural killer.
M-cadherin, a reliable marker for mouse satellite-cells (Irintchev et al. 1994; Beauchamp et al. 2000), has also been used to identify human satellite cells (Sajko et al. 2004), but this particular antibody is not commercially available and has therefore not been widely used.
Although Pax7 is a reliable mouse satellite cell marker (Seale et al. 2000), in human muscle it appears not to identify all satellite cells; in addition, it may also stain myonuclei (Reimann et al. 2004). In an attempt to distinguish Pax7+ satellite cells from myonuclei, some authors combined Pax7 and NCAM antibodies for satellite cell quantification (Verdijk et al. 2007) or counted a satellite cell as being a NCAM- and/or Pax7-positive cell in a sublaminar position (identified by laminin immunostaining) (Lindstrom and Thornell 2009). The latter study showed that the majority of human satellite cells expressed both Pax7 and NCAM, but there were also small numbers of NCAM+/Pax7- and NCAM-/Pax7+ satellite cells, which may have been either activated or differentiating (Lindstrom and Thornell 2009).
To further complicate comparison between studies, the following parameters have been used by different authors for normalization of the satellite cell count: number of myonuclei (Crameri et al. 2004; Eriksson et al. 2005; Sinha4-Iikim et al. 2006), length of muscle fiber (Sinha-Hikim et al. 2003; Sajko et al. 2004), and fiber cross-sectional area (Charifi et al. 2003; Dreyer et al. 2006; Kadi et al. 2006; Verdijk et al. 2007; Lindstrom and Thornell 2009). The correlation with the cross-sectional fiber area allows processes like muscle growth, pathological events, and ageing to be taken into account (Sajko et al. 2004), but it has been suggested that ideally both satellite cells per fiber and satellite cells per number of myonuclei should be used together for a more accurate measurement (Lindstrom and Thornell 2009).
In conclusion, it is still not clear if different antibodies detect all satellite cells and if satellite cell number determined by antibody staining and electron microscopy concur. In addition, it remains to be determined if differences between immunostaining of human and mouse satellite cells are an indication of species-specific differences, or merely reflect that the antibody itself is species specific. There is therefore a pressing need to standardize the identification and quantification of satellite cells in transverse sections of skeletal muscle so that comparisons of satellite cell numbers in different muscles, or the same muscles in individuals of different age and sex or individuals suffering from different pathological conditions, can be made.
Control of Satellite Cell Activation and Proliferation
Nearly all the work done on the processes of quiescence, activation, and self-renewal has been done on mouse, not human, satellite cells [reviewed by Dhawan and Rando (2005), Collins (2006), and Zammit (2008)]. We therefore summarize briefly findings on rodent satellite cells, but whether the same mechanisms apply to the human remains to be demonstrated.
Satellite cells are awakened from quiescence in response to normal physiological stimuli, e.g., exercise (Darr and Schultz 1987), and mechanical stretch (Tatsumi et al. 2002; Wozniak et al. 2003) and by pathological degeneration of muscle fibers, as seen for example in muscular dystrophy. These events lead to the activation of specific signaling pathways. Stretch-induced satellite cell activation is mediated by nitric oxide (NO) (Wozniak et al. 2003) that activates hepatocyte growth factor (HGF) (Tatsumi et al. 1998, 2002; Anderson and Pilipowicz 2002). Notch receptors on the satellite cell membrane play a crucial role in regulating self-renewal of satellite cells (Conboy et al. 2003; Kuang et al. 2007), and the wnt pathway is involved in both activation and self-renewal (Fuchs et al. 2004; Brack and Rando 2007; Le Grand et al. 2009) of mouse satellite cells. Sphingosine 1 phosphate induces mouse satellite cells to enter the cell cycle (Nagata et al. 2006). Growth factors such as fibroblast growth factor (FGF) (DiMario and Strohman 1988; DiMario et al. 1989) and insulin-like growth factor (IGF)-1 (Hill and Goldspink 2003) play a part in satellite cell proliferation and muscle regeneration (Charge and Rudnicki 2004). Other signals that are involved in controlling satellite cell function include stromal cell-derived factor (SDF)-1 that binds to CXCR4 and CXCR7 receptors on myogenic cells (Melchionna et al. 2010) and M-cadherin (Irintchev et al. 1994) that is involved in both satellite cell quiesence (Irintchev et al. 1994) and fusion into muscle fibers (Charrasse et al. 2007). Other cells within skeletal muscle may influence satellite cells, e.g., smooth muscle cells and fibroblasts secrete angiopoietin 1 that is involved in satellite cell quiescence and self-renewal (Abou-Khalil et al. 2009). However, the control of satellite cell quiescence, activation, proliferation, differentiation, and self-renewal within adult skeletal muscle in vivo has yet to be fully elucidated.
Satellite Cell Contribution to Skeletal Muscle Regeneration
Evidence that there are some satellite cells that fulfill the definition of a stem cell—a cell that is able to give rise to differentiated progeny and to self-renew (Ramalho-Santos and Willenbring 2007)—came from work in which quiescent satellite cells prepared from genetically modified mice were grafted into dystrophic mouse muscles and contributed both to regenerated muscle fibers and to functional satellite cells (Collins et al. 2005).
In mouse, the regenerative capacity of satellite cells does not appear to depend on the niche, as quiescent satellite cells removed from their fiber retain their ability to regenerate skeletal muscle and functionally reconstitute the satellite cell pool when grafted into dystrophin-deficient host mice (Collins et al. 2005; Boldrin et al. 2009; Ono et al. 2009). Similar work on human satellite cells has not been performed because of the difficulties in obtaining sufficient human satellite cells for grafting and particularly in determining if the donor cells had given rise to satellite cells, which requires the availability of a reliable human-specific satellite cell antibody.
However, as it is impractical to obtain satellite cells directly from donor muscle for treatment of patients, expansion in vitro would be necessary. A major limitation of the use of mpc to treat muscular dystrophies is that they lose their regenerative capacity following tissue culture. In vitro expansion of donor mouse (Montarras et al. 2005) and chicken satellite cells (O'Neill and Stockdale 1972) for only a short time significantly reduces the number of muscle fibers they form in vivo, probably because they commence myogenic differentiation. Similar to mouse, the regenerative capacity of human mpc is reduced after they have been expanded in vitro (Cooper et al. 2003; Brimah et al. 2004), which may be as a result of senescence during the culture period (Decary et al. 1996). This suggests that expansion of both mouse and human mpc in vitro may cause stem-like properties to be outweighed and therefore lost. Stem cell potential may also be affected by tissue culture conditions, e.g., signals from the substrate, medium components, growth factors, the cells themselves, or prior events in the life history of that particular population of cells. Whether there are ways to maintain the stem cell subpopulation in vitro, e.g., by using low-oxygen conditions as in the case of pluripotent stem cells (Millman et al. 2009), or using growth factors and substrates to re-create the niche (Cosgrove et al. 2009), remains to be investigated.
Nevertheless, there are some mouse mpc that retain their ability to regenerate skeletal muscle following a limited period of tissue culture. It is known that only a minority of cultured mouse mpc survive in vivo following intramuscular grafting, and these were the cells that were non-proliferating in vitro (Beauchamp et al. 1999). Within irradiated (but not in non-irradiated) dystrophic mdx hosts, the surviving donor cells proliferated, but nevertheless their contribution to regenerated muscle fibers was inefficient (Beauchamp et al. 1999) compared with freshly isolated satellite cells (Collins et al. 2005). For technical reasons, it has not been possible to follow the kinetics of human mpc grafted into mouse muscles, but similar to mouse mpc (Morgan et al. 1989, 1990, 1993; Watt et al. 1991; Gross and Morgan 1999), human mpc contribute to regenerated muscle fibers in immunodeficient mouse hosts (Huard et al. 1994; Brimah et al. 2004; Silva-Barbosa et al. 2005; Ehrhardt et al. 2007). However, human mpc repopulate host mouse muscle even less effectively than mouse mpc; fewer fibers of donor origin are found when the same number of human (Brimah et al. 2004) and mouse mpc (Morgan et al. 2002; Cousins et al. 2004) are grafted. This suggests that human myoblasts do not regenerate as effectively as mouse myoblasts, unless the difference in efficiency is related to the xenografts. Furthermore, neither mouse (El Fahime et al. 2000) nor human (Skuk et al. 2006) mpc are able to migrate far from the injection site, which is another major limitation of their use for therapeutic applications.
Interestingly, the pretreatment of host muscle (Brimah et al. 2004) and the host mouse strain (Cooper et al. 2001; Morgan et al. 2002; Silva-Barbosa et al. 2005) affects the number of donor-derived muscle fibers formed, and this effect seems to differ for human and mouse mpc. For example, human mpc contribute to more fibers of donor origin in cryoinjured rather than irradiated host muscles (Brimah et al. 2004), whereas the opposite is true for mouse satellite cells (Boldrin et al., unpublished data). Mouse mpc contribute to significantly more muscle fibers of donor origin in mdx nu/nu than in C5-/Rag2-/γ chain-host mice (Morgan et al. 2002). This implies that mouse and human mpc respond differently to an in vivo environment; human mpc may not undergo an expansion phase within the host mouse muscle and therefore neither regenerate skeletal muscle nor reconstitute the satellite cell niche efficiently (Ehrhardt et al. 2007).
The environmental factors that modulate donor mouse or human myoblast or satellite cell-derived muscle regeneration and self-renewal have yet to be fully determined. Certainly, other cells present within skeletal muscle, e.g., macrophages (Gordon 1995; Tidball 1995; Chazaud et al. 2003; Malerba et al. 2009), microvascular components (Rhoads et al. 2009), nerves (Tatsumi et al. 2009), smooth muscle cells, fibroblasts (Abou-Khalil et al. 2009), and the fiber itself, together with growth factors, gases (e.g., NO) (Anderson and Pilipowicz 2002; Tatsumi et al. 2002), or connective tissue components produced by them (Silva-Barbosa et al. 2008), as well as systemic factors (Conboy et al. 2005; Brack et al. 2007; Brack and Rando 2007), influence the capacity of satellite cells to survive, proliferate, migrate, regenerate muscle fibers, and self-renew.
There is evidence that growth factors such as IGF-1 (Mourkioti and Rosenthal 2005), leukemia inhibitory factor (Kurek et al. 1998), HGF (Miller et al. 2000), and FGF promote endogenous regeneration (Kurek et al. 1998; Yablonka-Reuveni et al. 1999b; Miller et al. 2000) or donor human myoblast-derived regeneration in the mouse (Brimah et al. 2004). However, not all growth factor isoforms have the same effect—for example, in some reports, IGF-6 has no effect on (Fiore et al. 2000) or even impairs (Floss et al. 1997) muscle regeneration; similarly, different FGF isoforms may achieve different effects (Neuhaus et al. 2003). Even the concentration of a particular growth factor and its interaction with other factors, e.g. HGF and myostatin, may be crucial for its effect on satellite cells (Yamada et al. 2010).
Function of Aged Satellite Cells
Satellite cells are lost with age in both mouse and man (Renault et al. 2002; Kadi et al. 2004; Sajko et al. 2004; Brack et al. 2005; Shefer et al. 2006; Collins et al. 2007; Verdijk et al. 2007). Although satellite cells from aged mouse muscles have a reduced capacity to self-renew (Day et al. 2010), there is a satellite cell fraction that retains muscle stem cell characteristics in aged mouse muscle and, if grafted into a young muscle environment, is still capable of muscle regeneration and self-renewal to the same extent as young satellite cells (Collins et al. 2007). However, the equivalent experiments on human satellite cells have not been performed.
Skeletal muscle mass is lost, and there is a decline in the ability of muscle to regenerate with increasing age in both mouse and humans (Alnaqeeb and Goldspink 1987; Cartee 1995; Grounds 1998; Bross et al. 1999). Whether this is due to defects in the environment (either local or systemic), or in muscle satellite cells, or both has been the subject of much recent debate.
In the mouse, satellite cells from aged muscle show impaired activation (Betters et al. 2008; Leiter and Anderson 2010) and increased apoptosis (Collins et al. 2007); similarly, aged human satellite cells are less capable of activation in vitro compared with young satellite cells (Renault et al. 2002). It has been suggested that accumulation of lipofuscin with age on human myofibers and satellite cells could result in a delay in satellite cell activation (Renault et al. 2002; Nakae et al. 2004; Leiter and Anderson 2010). The different expression of muscle actin isoforms in cultures of human mpc from individuals of different ages supports the view that old human satellite cells indeed differ from young human satellite cells (Lancioni et al. 2007).
The aged muscle environment impedes muscle regeneration (Conboy et al. 2005; Solomon and Bouloux 2006; Brack and Rando 2007; Carlson et al. 2009), possibly because of systemic or local levels of wnt or TGF-β1 (Carlson et al. 2009), but it may be modified by preirradiation to allow efficient donor-derived satellite cell regeneration and self-renewal (Boldrin et al. 2009).
Satellite Cell Response to Exercise and Contribution to Skeletal Muscle Hypertrophy
In response to exercise, satellite cells become activated and increase in number (Armand et al. 2003; Parise et al. 2008). Human satellite cells of both young and old individuals respond similarly to exercise, increasing in number and activation status (Crameri et al. 2004; Kadi et al. 2005; Mackey et al. 2009; Verdijk et al. 2009) and contributing to muscle hypertrophy (Kadi et al. 1999; Kadi and Thornell 2000). Interestingly, resistance exercise seems to have a different hypertrophic effect on men and women (Kosek et al. 2006), with satellite cell number only increasing in young men (Petrella et al. 2006).
In the mouse and rat, it was demonstrated that satellite cells are required for hypertrophy of overloaded skeletal muscles (Rosenblatt and Parry 1992; Rosenblatt et al. 1994; Snijders et al. 2009). Growth factors that cause muscle hypertrophy include particular isoforms of IGF-1 (Barton et al. 2002, 2010; Goldspink 2003), but only when muscle is growing or regenerating (Shavlakadze et al. 2010).
Muscle atrophy is mediated by interacting signaling pathways (Glass 2003, 2005), including FoxO (Southgate et al. 2007) and nuclear factor kB (Li et al. 2009). Myostatin induces muscle atrophy (Lee and McPherron 2001), and its inhibition results in muscle hypertrophy (McPherron and Lee 1997; Lee and McPherron 2001; Bogdanovich et al. 2002). However, satellite cells do not seem to be involved in muscle hypertrophy mediated by this pathway (Amthor et al. 2009). In rodent models of unloading-induced muscle atrophy, satellite cells initially become activated (Ferreira et al. 2006), but eventually decrease in number (Schultz et al. 1994; Mozdziak et al. 2000; Hawke and Garry 2001; Jejurikar et al. 2002; Jejurikar and Kuzon 2003), and those that remain are dysfunctional (Mitchell and Pavlath 2004). These studies have implications not only for repair and maintenance of skeletal muscle during periods of immobilization but also for using satellite cells to repair muscle that is already affected by disuse atrophy as, for example, in muscular dystrophies.
Satellite Cells in Pathological Conditions
Satellite cell dysfunction has been implicated in the muscular dystrophies, and in cases where there is a mouse model of the human dystrophy, comparisons may be made between satellite cells in mouse and human muscles with the same genetic defect. In Duchenne muscular dystrophy (DMD), lack of functional dystrophin leads to sarcolemma fragility and to continuous cycles of muscle degeneration and regeneration, resulting in regeneration failure, loss of muscle mass and function, and progressive substitution of muscle tissue with fibrotic and adipose tissue (Rando 2001; Emery 2002). In contrast, skeletal muscle regenerates at first effectively in the dystrophin-deficient mouse model of DMD, the mdx mouse (Coulton et al. 1988ab;, Stedman et al. 1991), although older mdx mice show a degree of defective regeneration (Zacharias and Anderson 1991) and their muscle eventually deteriorates with age (Pastoret and Sebille 1995). It is not therefore clear whether there is really a difference in dystrophic mouse/human satellite cell regenerative capacity, and if so, whether it is caused by differences in the satellite cells themselves, or the local muscle (e.g., extent of fibrosis), or systemic environment, or even by the genetic background of the mouse model (Fukada et al. 2010). Whether satellite cell numbers are altered in dystrophic muscle is difficult to determine, as accurate satellite cell quantification in dystrophic muscle is complicated by substitution of muscle fibers by fibrotic and adipose tissue (Desguerre et al. 2009). In addition, “branching” of regenerated myofibers (Bradley 1979; Blaveri et al. 1999) makes quantification of satellite cell numbers per fiber difficult. Nevertheless, based on observations on skeletal muscle sections, it has been reported that satellite cell number is greater in muscles of patients with DMD (Wakayama et al. 1979; Ishimoto et al. 1983; Watkins and Cullen 1988; Maier and Bornemann 1999) and neurogenic atrophy, but not in other muscular dystrophies (Becker muscular dystrophy, limb-girdle dystrophy) or inflammatory myopathies (Maier and Bornemann 1999). Numbers of M-cadherin+ satellite cells (calculated as the ratio of M-cadherin+ cells/total cell nuclei) were greater in mdx than in C57B1/10 mouse muscles (Yamane et al. 2005), but their activation state was not determined. There is evidence that satellite cells in muscles of DMD patients may be in a more activated state (Wakayama et al. 1979; Watkins and Cullen 1988; Maier and Bornemann 1999), as they seem to be in mdx muscles (Bhagavati et al. 1996; Ikemoto et al. 2007) and be detrimentally influenced by the pathological environment (Watkins and Cullen 1986). This state of constant activation and contribution to the repair of necrotic muscle fibers may deplete the stem cells from the satellite cell pool (Heslop et al. 2000).
Although human DMD mpc have reduced proliferative capacity (Blau et al. 1983; Melone et al. 1999), they can differentiate in vitro into myotubes and normal mpc (Blau et al. 1983), but only when contaminating fibroblasts have been removed from the preparation (Delaporte et al. 1984). The mpc from mdx EDL muscles have been reported to differentiate to the same extent as mpc from age-matched control animals, but mpc from some mdx mice displayed poor differentiation (Schuierer et al. 2005). Although these mice are of the same genetic background, there may be a difference in the muscle pathology between both muscles in the same mouse and the same muscle of different mice. However, the kinetics of differentiation of mdx mpc seems to be accelerated—normal mouse mpc proliferated faster and differentiated earlier than mdx mpc (Cheng et al. 1996), in accordance with work showing that mpc derived from mdx flexor digitorum brevis and diaphragm muscles had accelerated differentiation (Yablonka-Reuveni and Anderson 2006). However, there is no evidence that satellite cell-derived mpc fail to proliferate at later stages of the mdx pathological process (Bockhold et al. 1998). Different dystrophies may affect satellite cells in different ways, as suggested in mpc derived from biopsies of patients with myotonic dystrophy type II, which proliferate, but fail to differentiate (Pelletier et al. 2009).
Finally, in vitro experiments have suggested that satellite cells may actually be contributing to muscle pathology by transdifferentiating into adipocytes or fibroblasts (Asakura et al. 2001; Brack et al. 2007). However, the satellite cell population may be contaminated with other cell types, and recent in vivo experiments have provided evidence that adipocytes and fibroblasts in skeletal muscle derive from interstitial cell progenitors rather than from satellite cells (Joe et al. 2010; Uezumi et al. 2010). There is therefore a need to study human satellite cell number, activation, and differentiative and self-renewal ability in muscles of patients with muscular dystrophies and in particular to address the contribution of satellite cells to fat and fibrotic tissue in different pathological conditions. These studies would be invaluable for our understanding of why skeletal muscle fibers are lost in muscular dystrophies.
Conclusions
Although human and mouse satellite cells express similar markers, it has been difficult to determine whether their phenotype and functions are equivalent. This is mostly due to the difficulty in isolating quiescent human satellite cells and the lack of specific antibodies for their unequivocal identification. In particular, it is not clear whether human satellite cells are indeed effective muscle stem cells. Both mouse and human skeletal muscles are capable of regeneration, but whether human satellite cells can self-renew following injury to give rise to functional satellite cells has not been demonstrated. In order to progress work on human satellite cells, one first needs to ascertain whether newly discovered markers of mouse satellite cells (Fukada et al. 2007; Gnocchi et al. 2009; Pallafacchina et al. 2010) are expressed by human satellite cells. Then, if there was a method to induce cultured mpc to give rise to quiescent satellite cells (e.g., by re-creating the niche in vivo), these markers could be verified and used to either subfractionate human satellite cells or study their activation and differentiative potential in vitro.
Isolation of human satellite cells and investigation of their in vitro and in vivo properties, similar to the extensive work that has been performed on mouse satellite cells, would pave the way to either using donor satellite cells as a therapy for muscular dystrophies or enhancing the function of endogenous satellite cells in dystrophic or aged muscles.
Footnotes
Acknowledgements
L.B. is funded by the Muscular Dystrophy Campaign and J.E.M. holds a Wellcome Trust University award. J.E.M. and F.M. are principal investigators of the Medical Research Council Centre for Neuromuscular Diseases.
We thank Dr. Francesco Conti for his critical reading of the manuscript.
