Abstract
To better understand the mechanism(s) of leukocyte Ca2+ signaling, we have studied the intracellular locations of two Ca2+-mobilizing receptors, the inositol 1,4,5-trisphosphate receptor and ryanodine receptor, by immunofluorescence microscopy. Our results show that localization differs not only between receptor classes within a cell, but among leukocyte types as well. We also illustrate the importance of preserving labile cellular filaments in maintaining cell integrity by fixation with the Safiejko-Mroczka and Bell protocol, because conventional fixation methods distort receptor patterns. We suggest that the observed differences influence intracellular Ca2+ signaling.
C
Peripheral blood was collected from healthy donors in compliance with the guidelines of the University of Michigan Institutional Review Board for Human Subject Research. Leukocytes were isolated using Ficoll-Histopaque density gradient centrifugation (Sigma Chemical Co.; St Louis, MO), then washed with PBS. Isolated cells were allowed to adhere to cover slips for 15 min at 37C. Cells were fixed for fluorescence microscopy using either 20 min at room temperature with 3.7% paraformaldehyde (Sigma) or the dithiobis (succinimidyl propionate) (DSP) procedure, which provides superior retention of labile cellular filaments (Safiejko-Mroczka and Bell 1996). Briefly, cells were first fixed using DSP (1 μg/ml; Pierce, Rockford, IL) in HBSS for 10 min at room temperature. Cells were extracted using 100% ethanol, followed by 5 min incubation in stabilizing buffer (1 mM EGTA, 4% polyethylene glycol 8000, and 100 mM PIPES at pH 6.9). Samples were washed with PBS, fixed with 3.7% paraformaldehyde for 20 min, then washed again.
Antibodies were conjugated with FITC and tetramethylrhodamine isothiocyanate (TRITC) (Molecular Probes). Anti-IP3R antibody reacting the C-terminal cytoplasmic domain of all three subtypes of the receptor (Chemicon International; Temecula, CA) was conjugated to FITC. Anti-RyR-1,2 antibody (BIOMOL International; Plymouth Meeting, PA) was conjugated to TRITC. Cover slips with fixed leukocytes were washed in PBS, and then blocked for 30 min with 3% BSA (w/v; Sigma) in PBS at room temperature. After blocking, cells were labeled simultaneously with anti-IP3R and anti-RyR at dilutions of 1:20 and 1:50, respectively, in PBS for 30 min at room temperature. Cover slips were washed thoroughly with PBS.
Samples were imaged using a Ziess Axiovert 135 inverted microscope coupled to an intensified digital charge-coupled device camera (Qimaging; Barnaby, BC, Canada). Cover slips were observed using bright field and fluorescence microscopy. To detect FITC, optical filters with an excitation at 482 nm and emission at 530 nm were used. For TRITC detection, optical filters with excitation and emission at 535 nm 590 nm, respectively, were employed. Images were obtained using QCapture image acquisition software (QImaging) and were processed using MicroTome deconvolution software (Vaytek Inc; Fairfield, IA) with an approximate point spread function. Image-Pro Plus (Media Cybernetics; Silver Spring, MD) and Adobe Photoshop 7.0 (Adobe Systems; San Jose, CA) were used to prepare the images.
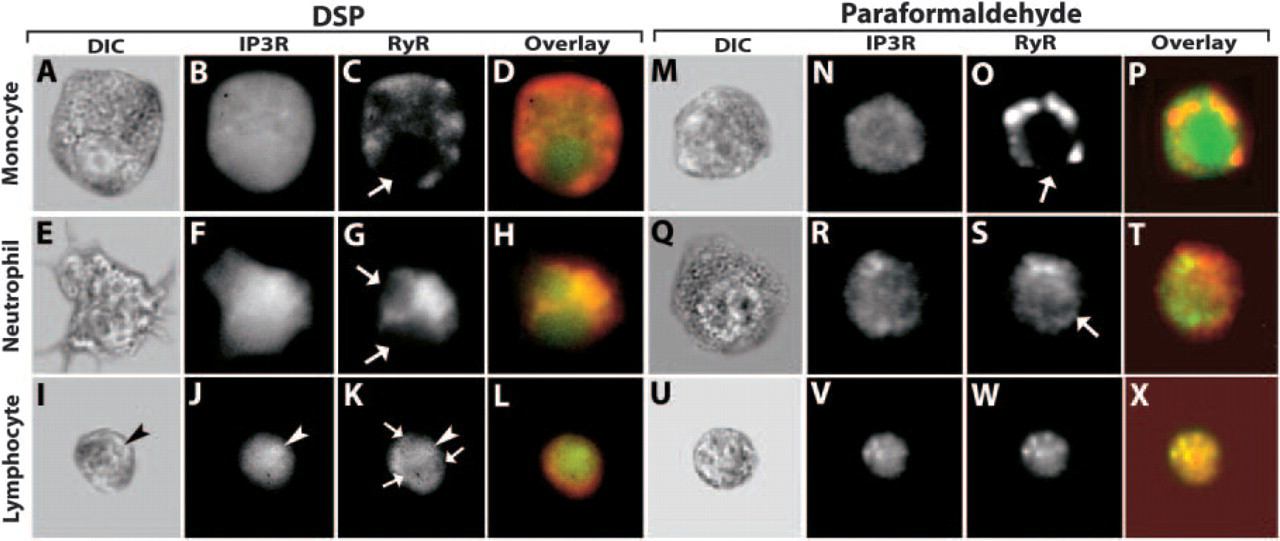
Intracellular distribution of IP3R and RyR varies among human leukocytes. Cover slips with adherent leukocytes were fixed with the DSP (
The IP3R's intercellular locations were determined by immunofluorescence staining of human neutrophils, monocytes, and lymphocytes. In monocytes, the IP3R was present as a diffuse, uniform pattern throughout the cytoplasm and nucleoplasm (Figure 1B). In neutrophils, the IP3R was found throughout the cell, including the interior of the nucleus (Figure 1E). The pattern in lymphocytes was distinct from that of other leukocytes. As with neutrophils and monocytes, the IP3R was distributed throughout the entire cell (Figure 1J). However, these cells were not labeled with nonimmune serum. In contrast to neutrophils and monocytes, lymphocytes exhibited an area of bright intensity inside the nucleus that morphologically corresponded to the nucleolus (arrowhead, Figure 1J), with brightness decreasing toward the cell surface. This unexpected morphological feature is further illustrated in Figure 2. This figure shows quantitative line profile analyses of IP3R labeling of lymphocytes. Enhanced labeling is associated with the nucleolus. Hence, the IP3R is heterogeneously distributed within and among leukocytes.
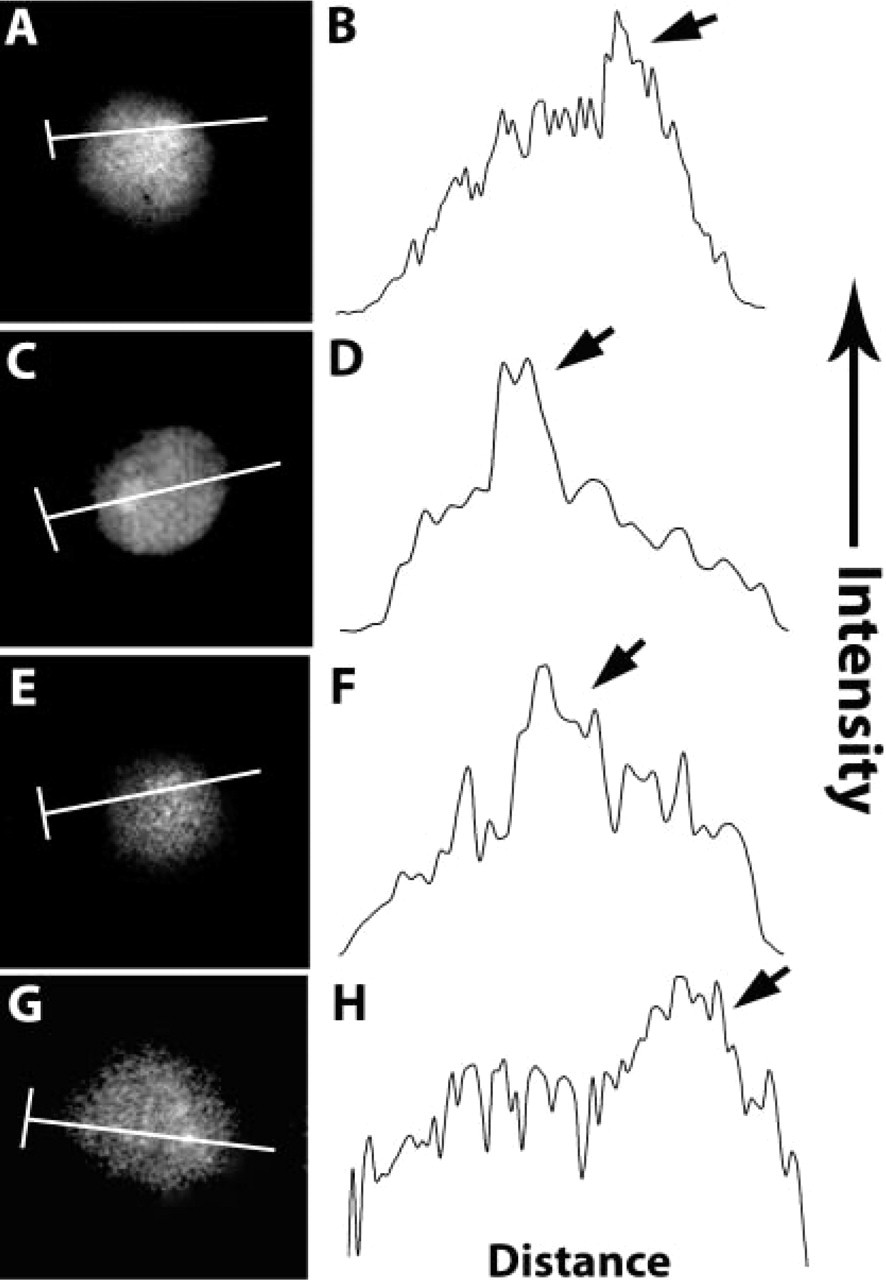
IP3R labeling is enhanced in the lymphocyte nucleolus. Cells were fixed with DSP, washed in PBS, then blocked with BSA. Cells were labeled simultaneously with FITC-anti-IP3R and TRITC-anti-RyR. Line profile intensities of IP3R staining were measured along a line drawn through the nucleolus, and was plotted as intensity over distance. (
The RyR was labeled simultaneously by staining the same cells with a second fluorescent antibody, which allowed unambiguous comparisons of receptor distributions. We found pronounced differences in the labeling patterns of the two receptors. In monocytes, the RyR was present in clusters, which localize primarily near the border of the cell membrane (Figure 1C). Smaller, less intense clusters of RyR extend into the cell's interior—though it appears that the RyR is excluded from the nucleus (arrow, Figure 1C). Similarly, the RyR was not evident in the neutrophil's nucleus (Figure 1F). The extranuclear localization of RyR in neutrophils was distinct from that of monocytes. The RyR appeared near the nuclear envelope and extended into the cytoplasm. Interestingly, in neutrophils, the RyR is most intense close to the nucleus and does not appear to extend to the plasma membrane. In lymphocytes, as with neutrophils, there appears to be a brighter intensity of RyR labeling near the nucleus, which decreases toward the cell membrane (arrows, Figure 1K). Contrary to what is seen in the other two cell types, the RyR is also present in the lymphocyte nucleus. The RyR labeling is brightest near the nucleolus and decreases in the vicinity of the nuclear envelope.
For comparison, staining was performed in an identical manner on leukocytes fixed using a conventional paraformaldehyde procedure. Some alterations in cell morphology were evident using bright-field microscopy (Figures 1M, 1P, and 1U). Changes in the intracellular distributions of Ca2+-mobilizing receptors were examined. Although IP3R staining in monocytes was diffuse as in Figure 1B, it did not clearly extend to the cell surface (Figure 1N). RyR labeling became markedly clumped (Figure 1O). Neutrophil labeling was more dramatically disrupted. In comparing Figure 1Q with Figure 1E, it is clear that labile cytoskeletal structures, such as filopodia, are retained by the DSP protocol, but not paraformaldehyde fixation. IP3R labeling was not uniform. Conventionally fixed cells exhibited IP3R clustering and the location of the nucleus was more clearly defined (Figure 1R). Furthermore, the nucleus was less clearly defined after RyR labeling (arrow, Figure 1S) of paraformaldehyde-fixed cells. In comparing Figures 1R with 1S, there is little that varies. The most dramatic change occurs in the lymphocyte. IP3R and RyR expression (Figures 1J and 1K, respectively) appeared to be the same in paraformaldehyde-fixed cells, but the nature of the staining was quite different in comparison to DSP-fixed cells. Rather than a gradient in the IP3 label, the pattern was uniform, with the exception of several bright clusters around the outer edge, none of which obviously corresponded to the nucleolus (Figure 1V).
This study demonstrates differential trafficking of the IP3R and RyR in hematopoietic cells, which may contribute to an intracellular “wiring” diagram that underlies immune function. Previous studies from this laboratory using high-speed fluorescence microscopy have demonstrated a variety of intracellular Ca2+ signaling patterns (Kindzelskii and Petty 2003; Petty 2004). The differential intracellular trafficking of the IP3R and RyR may be one factor contributing to these signal patterns. For example, the increased IP3R density associated with lymphocyte's nucleolus may contribute to the intense Ca2+ wave initiated near this site during antigen stimulation (Li Q, et al., unpublished data). We suggest that the differences in Ca2+ mobilizing receptor distribution are linked with cell types.
Previous studies are consistent with our findings. For example, the IP3R resides in both the cytoplasm and nucleus in other cell types (Echevarria et al. 2003; Huh and Yoo, 2003). Sugiyama et al. (1994) showed that the expression profiles of IP3R subtypes shows cell-specific variations among hematopoietic cell lines. Northern and dot blot analyses revealed that the expression profiles for each cell type changes during differentiation (Sugiyama et al. 1994). RyR expression levels vary among cell types (Furuichi et al. 1994). We propose that epigenetic differences in receptor localization among cell types influence spatiotemporal characteristics of traveling Ca2+ waves and signal targeting within cells. The interplay among these receptors may tailor a cell's signaling properties to facilitate intracellular communication.
We also illustrate the importance of preserving labile cellular filaments and, thereby, the integrity of other cell structures by fixation with DSP. Organelles such as the endoplasmic reticulum are attached to the cytoskeleton. When cells are fixed with conventional chemical means, cytoskeletal structures depolymerize, which may allow organelles to assume artifactual shapes and locations. Because the IP3R and RyR are found in the endoplasmic reticulum (Pozzan et al. 1994), the ER's preservation is vital in assessing IP3R and RyR distributions. By employing this technique, we showed that the intracellular distributions of IP3R and RyR varies among monocytes, neutrophils, and lymphocytes. By double-labeling cells with anti-IP3R and anti-RyR, we also found that their expression is markedly different in the same cell, which is true for all three cell types used. DSP fixation allows observations of intracellular receptors that are lost with conventional techniques. In contrast, we found that fixation with paraformaldehyde creates artifactual clumps of IP3R and RyR in lymphocytes and neutrophils—presumably because of disruption of microtubules. This finding suggests that maintaining the integrity of these structures by rapid DSP fixation may be especially important when labeling lymphocytes, because they are more dramatically affected than other cell types studied.
Footnotes
Acknowledgements
This work was supported by the NIAID and National Multiple Sclerosis Society.
