Abstract
At the level of the genital system, ovarian neoplasm is the most frequent cause of morbidity and mortality. In the specialized literature, the coexistence of an inflammatory process is admitted from the early stages of the evolution of this pathology. Starting from the importance of this process, both in determinism and in the evolution of carcinogenesis and summarizing the field of knowledge, for this study we considered two objectives: the first was the presentation of the pathogenic mechanism, through which chronic +ovarian inflammation is involved in the process of carcinogenesis, and the second is the justification of the clinical utility of the three parameters, accepted as biomarkers of systemic inflammation: neutrophil-lymphocyte ratio, platelet lymphocyte ratio, and lymphocyte-monocyte ratio in the assessment of prognosis. The study highlights the acceptance of these hematological parameters, with practical utility, as prognostic biomarkers in ovarian cancer, based on the intrinsic link with cancer-associated inflammatory mediators. Based on the data from the specialized literature, the conclusion is that in ovarian cancer, the inflammatory process induced by the presence of the tumor, induces changes in the types of circulating leukocytes, with immediate effects on the markers of systemic inflammation.
Keywords
Introduction
Ovarian cancer was the third most common gynecological cancer globally in 2020. Ovarian carcinoma is the most common type of ovarian cancer, comprising more than 90% of all ovarian cancer cases. The risk of ovarian cancer increases in women with age. Also, family history, with familial cancer syndrome and mutations in the breast cancer susceptibility gene (BRCA), can increase the risk of ovarian cancer. 1
In 2018, 4.4% of cancer deaths among women were due to ovarian neoplasm. 2 The prognosis of ovarian cancer remains poor: the 5-year survival rate is approximately 41% overall and approximately 20% for stage III and IV disease (AJCC 6-th Edition classification). 3 This justifies the interest of research in the field, which aims to identify useful parameters for an early diagnosis and prognosis, to guide personalized treatment.
In recent years, chronic inflammation 4 has been identified as a key factor in the pathogenesis of ovarian cancer. First of all, ovulation 5 itself is a natural inflammatory process, which involves the breaking and cyclic healing of the ovarian cortex, which is considered a factor involved in the occurrence of cancer, and patients with endometriosis or pelvic inflammatory disease have an increased risk of ovarian cancer. 6 On the other hand, oral contraceptives, tubal ligation or hysterectomy, 7 and anti-inflammatory therapy 8 reduce the risk of ovarian cancer. Traditional studies focus on the oncological characteristics of the tumor itself (such as tumor histology, grade, and gene mutations), but recently, studies have begun to investigate host responses and the tumor microenvironment (such as immune cells, fibrocytes, and metabolites). 9 The pathophysiology of ovarian carcinogenesis includes two hypotheses: genetic and inflammatory. Pathogenically, the dominant characteristic of tumor cells is genetic instability, which can increase with each cell generation. This is the expression of genomic mutations whose consequences, if they cannot be removed by intrinsic, cytosolic-nuclear mechanisms, predispose to cancer. The body’s own cells metamorphose, turning from healthy to malignant, by acquiring morpho-functional characteristics much different from those of normal cells, which also explains the ability of the malignant process to “escape” the body’s defense mechanisms. Among the defense processes involved in neoplasias, the development of inflammation is immediate and persistent, initially appearing as a local mechanism, but which over time becomes a systemic one. 10
The numerous published studies investigate, extensively, the importance of the inflammatory reaction, as a prognostic factor of the survival of an oncological patient, and the values of systemic inflammation biomarkers are a means of quantifying its level. Scores based on absolute values of different types of leukocytes are NLR—neutrophil-to-lymphocyte ratio, PLR—platelet-to-lymphocyte ratio, and LMR—lymphocyte-to-monocyte ratio.11-14
Material and methods
The article is based on the analysis of data considered relevant for the chosen topic from the studies identified in PubMed Central (PMC) and MEDLINE Complete (EBSCO).
For a sensitive search strategy, the terms used in search engines were “ovarian cancer and inflammation cell and pathway” and “systemic inflammation markers.” The article focused on updated data about the processes involved in the occurrence of inflammation associated with ovarian cancer, its role in the evolution of cancer, as well as the reflection of the level of local inflammation at the systemic level.
Summary of the main advances related to the inflammation—ovarian cancer relationship.
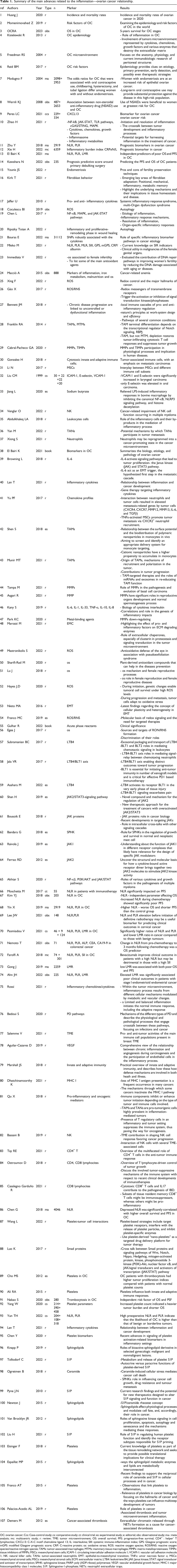
OC: ovarian cancer; Ccs: Case-control study; cs: comparative study; ct: clinical trial; es: experimental study; e: editorial; obs: observational study; ma-: meta analysis; mc: multicentric study; r: review; TME: tumor microenvironment; OS: overall survival; PFS: progression-free survival; CD4+ : helper’ T lymphocites; CXCL13: C-X-C motif chemokine ligand 13; IL: interleukine; SIII: systemic immune-inflammation index; GPS: Glasgow prognostic score; mGPS: modified Glasgow prognostic score; CRP: C-reactive protein; os: oxidative stress; ROS: reactive oxygen species; ROS/RNS: reactive oxygen species/reactive nitrogen species; TAMs: tumor-associated macrophages; MTMs: mammary tissue macrophages; MMPs: matrix metalloproteinases; TIMPs: tissue inhibitors of MMPs; MSCs: mesenchymal stem cells; ICAM-1: circulating intercellular adhesion molecule 1; VCAM-1: vascular cell adhesion molecule 1; NK: natural killer cells; TANs: tumor-associated neutrophils; EMT: epithelial-to-mesenchymal transition; TNFα: umor necrosis factor α; MSCs: mesenchymal stromal cells; MSCs: mesenchymal stromal cells; ECM: extracellular matrix; LTB4: leukotriene B4; JAK: Janus kinase; STAT: signal transducer and activator of transcription; SPHK: sphingosine kinase; PARP: poly (ADP-ribose) polymerase; VEGF: vascular endothelial growth factor; MHC I: major histocompatibility class I molecules; S1P: Sphingosine-1-phosphate; NETs: neutrophil extracellular traps.
For a better understanding of the information presented, we developed a workflow diagram for this study (Figure 1). Workflow diagram for this study.
The relationship between the chronic inflammatory process and the appearance of neoplasia
Pathogenically, the installation and progression of carcinomas, as well as the associated inflammatory response, are favored both by the genetic predisposition and by the environmental characteristics that define, at a given moment, the properties of the area in which the tumor develops. Among these characteristics, generically called tissue homeostasis, regardless of the evolutionary stage of the cancer, an essential role belongs to the existing cell population (endothelial cells, fibroblasts, cells involved in the development of the localized inflammatory process, which later became systemic, etc.). Systemic inflammation must be interpreted as expressing the reparative response of body cells, to the signals carried by the cytokines secreted in excess, with a plurietiological determinism and over a long period of existence of the process.15–20
Physiopathologically, in neoplasias, inflammation has a dual determinism, being involved both in the installation and evolution of cancer, but also occurring as a possible defense reaction against the existing neoplasia. The use of the three parameters/indices/ratio NLR, PLR, and LMR as markers of systemic inflammation requires the exposure of the scientific support regarding the inflammation-neoplasia interference, at least regarding the existence of inflammation as a defense process against neoplasia and only tangentially as a process which intervenes in carcinogenesis.21,22 The appearance and development of the ovarian neoplasm, as a consequence of the existence of a chronic regional inflammation (usually located in the distal part of the Fallopian tube, or at the level of the ovarian epithelium) has as its pathogenic cause the installation of local oxidative stress, just like the evolution of any other inflammatory process, regardless of location.23,24 At any evolutionary moment of carcinogenesis (installation, local invasion or metastasis), the inflammatory process persists, developing together with the immune system, as anti-neoplasia defense systems. Although with a protective function, these processes also lead to the disruption of redox homeostasis, exacerbating oxidative stress.25,26 In cancers, the increase in oxidative stress is the result of the synergistic action of at least two types of histochemical processes. The first one excessively generates oxidizing species, as a result of the specificity of cancer cells to synthesize ATP macromolecules anaerobically, glycolytically, both through their own multiplication/development, and through the destructive invasion, enzymatically, of normal tissue, peri tumoral or that at the site of metastasis, a process dependent on the stage of development of the neoplasia (localized or metastatic). To these secondary or residual products of the various stages of tumor development, cumulatively, a second synthesizing source of oxidizing species is added, from the development of the associated inflammatory process. Thus, inflammation, although it occurs as a defense process, aiming to limit “tumor aggression,” constitutes a new synthesizing source of ROS/RNS, which we can generically name “redox quota generated by defense.” The process is a sequential one, unfolding in cascade, with evolution from the stage of limited, local-reactionary defense to that of systemically expressed inflammation. In the pathophysiological determination of any inflammation, two types of processes are included, derivative and independent: one alterative-destructive and another reactive-limiting.27,28
The destructive-alterative process, which develops, is formed by the existing defense processes against the cancer cell, a non-self structure for the body. The installation of neoplasia and regional invasion induces morphological degradation both at the cellular and interstitial level, leading to areas of necrosis, with compromised tissue metabolism. Locally, the action is the consequence of the release in the matrix of the interstitium of enzymes with a destructive role, such as metalloproteinases (MMP), but also of the increase in the concentration of active biological compounds such as cytokines or chemokines.29,30
The local development of the inflammatory process is the expression of the conjugated action of at least two categories of compounds: - ICAM-1 (intercellular adhesion molecule-1) and VCAM-1 (vascular cell adhesion molecule-1), but also selectins (P, E, L), generators of chemoattraction for the figured elements that circulate lamellarly in the vessels of the normal peritumoral tissue or in those developed by angiogenesis, activated by cancer development.31,32 - others, the majority, act as mediators of inflammation
33
(Table 2). PDGF: platelet-derived growth factor; FGF: fibroblast growth factor; VEGF: vascular endothelial growth factor; IGF: insulin-like growth factor; EGF: epithelial growth factor; IL: interleukine; IFN: interferon; TNFα: tumor necrosis factor; TGF: transforming growth factor; MCP: membrane cofactor protein; MIP: macrophage inflammatory protein.

The human body responds to the action developed by the biologically active compounds, through a set of changes, which constitute the reaction syndrome, consisting of vascular-circulatory reactions, which include permeability disorders, that allow the formation of inflammatory exudate. The recognition and targeting of cells of the mononuclear system, capable of phagocytosis (in particular, monocytes, neutrophils), by chemoattractants, is the result of their affinity/attraction to the phospholipid layer of the cell membrane.
The inflammatory process begins with the expression of adhesion molecules, responsible for the marginalization of mononuclear cells, with phagocytic capacity, and those of immune defense, and through the synergistic intervention of the cytokine VEGF (vascular endothelial growth factor), diapedesis is triggered (Figure 2). The interface between neoplasia and chronic inflammatory process.
The consequence of the increased migration of leukocytes is the infiltration of the intra- and peritumoral tissue or, in the case of extirpation of the tumor, of the area on which it was intervened. The triggered inflammatory response is characterized by an increase in the neutrophil population that mediates a pro-carcinogenic effect, by promoting angiogenesis, cancer cell migration, with local invasion and metastasis.36,37
In addition, neutrophils indirectly regulate the epithelial-mesenchymal transition, through the production of oncogenic cytokines and chemokines, such as interleukins (IL)-1β, IL-6, IL-12, and TNF-α, which promote the recruitment and further activation of other inflammatory cells.38-41
However, in the leukocyte infiltrate, monocytes predominate, which, through metamorphosis, become macrophages. These macrophages, termed tumor-associated macrophages (TAMs), are phenotypically and functionally distinct from pre-existing tumor tissue macrophages (MTMs), as TAMs express the adhesion molecule VCAM1 and do not exhibit an “alternatively activated” phenotype.28,42,43
Tissuely, they secrete numerous biologically active products, such as those in Table 2, but also myeloperoxidases (MMP),44,45 which have a synergistic action with the released cytokines. 46
Thus, these hemoproteins will contribute, at the tumor level, to the destruction of the extracellular matrix, facilitating the mobilization of tumor cells and metastasis.47,48
In parallel, the released MMPs cause the exacerbation of the synthesis of pro-oxidant agents (ROS) which, reacting with NO, develop another locally destructive species, nitrogen derivatives (NOS). Excessive local synthesis of the two types of agents with pro-oxidative properties leads, through consumption, to the quantitative insufficiency of antioxidant defense systems, such as “scavenger” enzymes (superoxide dismutase (SOD), catalase, GSH-peroxidase (GSHPx), GSH-reductase and GSSG), but also by other non-enzymatic antioxidants.49–51
Modification at the level of the tumor cell, of the way of controlling the synthesis of mediators involved in the development of inflammation, such as the activation of leukopoiesis and thrombocytopoiesis
Oxidative stress is caused by an imbalance between the production and accumulation of ROS in cells and the ability of the biological system to detoxify them. Cancer cells exhibit aberrant redox homeostasis, but while ROS are pro-tumorigenic, high ROS levels are cytotoxic. The functions of cells in the tumor microenvironment, cancer-associated fibroblasts (CAFs), tumor-associated macrophages (TAMs), and T cells, are affected by oxidative stress. 52
At the level of the latter, there is a change in the way of controlling the synthesis and release of cytokines, some of which have a role as mediators of inflammation and immune defense. Thus, tissue oxidative stress allows the continuation of the effect of a. Cancer proliferation, the pathogenic mechanism promoting invasion and metastasis.
53
b. The development of regional inflammation, as a defense process, but which, in progress, becomes systemic inflammation, manifested by changing the blood count and by increasing the serum values of inflammation markers, such as C-reactive protein, alpha-2 globulinemia, and orosomucoid.
In conditions of oxidative stress, the target of ROS/RNS is the derivative of sphingomyelin, arachidonic acid, which, through multi-enzymatic catalysis, generates most of the mediator compounds of inflammation, but also of carcinogenesis. Figure 2 predominantly highlights the effects developed by the main four groups of messenger compounds, with a role in autocrine or paracrine signaling, resulting from the enzymatic biodegradation of arachidonic acid: prostaglandins, prostacyclins, thromboxanes, and leukotrienes, molecules produced especially by leukocytes.
In a next step, the resulting compounds activate “redox signaling,” which allows a sequential and modulated process to unfold and “apply” the signals carried by them.54–56
In order to carry out such a process, cells have both specific structures, with the role of redox sensors, as well as the mechanisms of signal transduction, respectively transcription.
In neoplasms, the impairment of hematopoiesis and the function of the immune system is the expression of the activation of membrane receptors BTL1 and BTL2, respectively, high and low affinity receptors, to which one of the most powerful pro-inflammatory cytokines (LTB4) binds. 57
BLT1 has long been known to be a chemoattractant receptor for neutrophils, and recent studies have identified BLT1 expression on multiple cell types (macrophages, activated T cells, mast cells), greatly expanding the potential roles of LTB4. This diversity of expression has led to the current concepts of yin and yang regulation of cancer, as the activity of immune cells influencing both pro (neutrophils, myeloid-derived suppressor cells) and anti-tumor outcomes (CD8+ effector T cells) can be controlled by the axis LTB4/BLT1. BLT2 expression on epithelial cells has been implicated in pro-tumorigenic signaling by promoting chemotherapy resistance, angiogenesis, inflammation and metastasis. Becoming active by fixation, the information carried by LTB4 is cytosolic transduced into the nucleus, where the signals act as modulators of transcription.58,59
The pathway that disrupts the intracellular transmission, is the phosphorylation of tyrosine, threonine, serine residues or functional-structural proteins, catalyzed by cytosolic kinases of the Janus-associated kinase family type (JAK1, JAK2, JAK3, and TYK2).60,61 JAK1 and, in particular, JAK2 are involved in signaling through cytokines and growth factors, affecting hematopoiesis and immune system function. The control of activation at the membrane level, by JAK2, is exercised by two domains: functional JH1 and JH2, which negatively regulates the first domain. The presence of a mutation in the latter domain allows the activation of JH1 and implicitly JAK2 kinase through phosphorylation62–64. Phosphorylated JAK2 activates the signaling pathway: STAT, MAPK, ERK (Ras-Raf-MEK-ERK pathway), and PI3/akt which, on the one hand, modulates the synthesis and release of some of the cytokines involved in inflammation, and, on the other hand, induces growth proliferation, differentiation and cell survival, including at the level of red bone marrow. 65
The processes of multiplication and differentiation, at the hematogenous medulla level, are expressed peripherally, by changing some of the parameters of the blood count, such as platelets, neutrophils, and monocytes, figurative elements that are integrated in the calculation of systemic markers of inflammation. This constitutes the scientific foundation, which allows the practitioner to evaluate, indirectly, the dimensions of inflammation in cancer, and these parameters NLR, PLR, and LMR are potential clinical prognostic markers. NLR has been repeatedly reported as a prognostic factor in ovarian cancer patients in advanced stages of the disease,11,66–69 in patients with other tumor locations70,71 or may be useful in selecting patients for different types of immunotherapy.66,72
Mechanisms of antineoplastic defense by activating innate immune cells
A scientific argument, about the fact that the values of markers of systemic inflammation can provide useful information to medical practice, in evaluating the prognosis of the patients with genital neoplasms, highlighted by several authors,73,74 is also the fact that in neoplasia, in parallel with the inflammatory process, there is another, of immune defense.
This is because lymphocytes, by migrating into the tumor microenvironment, become part of the peritumoral inflammatory infiltrate, and intervene in the antitumor immune reaction, by inducing cytotoxic cell death and by inhibiting the proliferation and migration of tumor cells.16,44,75
From another point of view, the fact that neoplasia evolves regardlessof type and location, invading and metastasizing, demonstrates that the immune defense is not strong enough to cause apoptosis of the cancer cell, probably due to the latter’s ability to secrete biologically active compounds, that prevent such an action.
The interaction between defense mechanisms and the malignant tumor, is also reflected at the peripheral level, by involving the figurative elements of the blood, with direct consequences on the markers of systemic inflammation. 76
The conditionality of the two processes is based on the expression, by the cancer cells, of their own antigens, which leads to the activation of the cells involved in the elaboration of the immune response.77,78
In an initial step, the cells of the histocompatibility system, exercising their role, identify the antigens expressed by antigen-presenting cells (APCs), present them to those involved in the innate immune system, and then to those responsible for adaptive immunity.79,80
The innate immune system, through monocytes that have become macrophages, through neutrophils and Natural Killer (NK) lymphocytes, releases extracellular cytokines, chemokines, oxygen free radicals, MMPs, their natural inhibitors (TIMPs), which will ensure antineoplastic defense by acting on various levels: destruction and inhibition of DNA repair capacity, angiogenesis promotion, inhibition of COX2 activity, etc.81,82
Mechanisms of antineoplastic defense by activating acquired immunity
A much more complex process of acquired immunity begins through the interaction of antigen-presenting cells (APC) with one of the tumor-specific antigens, an activating process for the generation of antibodyproducing cell clones. This category includes B lymphocytes and T lymphocytes, of which helper (CD4+) and cytotoxic T (CD8+) are important.83–85 The differentiation to these two lymphocyte subtypes is controlled by IL12, secreted by APC. The type CD8+ lymphocyte isendowed with phagocytic capacities for dysfunctional/destroyed cells and cancer cells, identifiable as non-self, while CD4+ lymphocytes produce a Th1-type immune response, which consists of the secretion of cytokine IFN-γ, an activator of monocyte metamorphosis in macrocytes, which leads to anaugmentation of the entire immune response.86,87
The involvement of lymphocytes in the defense against cancer cells, scientifically justifies their use in medical practice, the numerical value being involved in the calculation of the three ratios: NLR, MRL, and PLR.13,71,73,88
Platelets, as a factor involved in the defense system through the development of antineoplastic inflammation
Physiopathologically, in neoplasms, the research in daily medical practice of the number of circulating platelets, their volume and the calculation of the PLR index, is supported by the involvement of these elements figured in: - induction of mesenchymal-epithelial transition; - favoring tumor metastasis, by activating the synergistic interaction between the TGF-β1/Smadand NF-κB pathways or by activating YAP1 signaling;89,90 - in the growth and progression of ovarian carcinomas, via proliferative effect, an aspect observed both in vivo and in vitro;
91
- in the immune system.
89
The argument that supports the roles attributed to platelets in ovarian carcinomas, especially in high-grade serous ones, is the result of published studies, which consistently identify, preoperatively, the presence of thrombocytosis.38,90,92,93
The circulating platelet excess can be explained by the role of these acellular figured elements, as reactants of inflammation, the hyperproduction being induced by the pro-inflammatory cytokines: IL-1, IL-6, and IL-11.40,94
The involvement of platelets in the process of carcinogenesis and associated inflammation results from the action they induce at the level of sphingolipids of cell membranes, represented mainly by ceramides (CER), glucosylceramide (GlcCer), sphingosine (SPH), and sphingosine-1-phosphate (S1P) 95
Selectively, cellular oxidative stress is a stimulating factor of sphingosine and/or ceramide synthesis, and among these, the distinctive role in modulating the process of development, progression, metastasis, and even in the chemoresistance of neoplasia, belongs to CER.96,97 In cells, there is a rheostat, namely, CER-S1P, whose relative position is determined by the concentration of oxidative agents that regulate the interconversion of ceramide-sphingosine-S1P. 98 CERs are generated by enzymatic hydrolysis of sphingomyelin by sphingomyelinase, and S1P by both metabolic conversions of ceramide and phosphorylation of SPH which is incorporated from the interstitial space, sphingosine kinase-catalyzed reaction (SPHK).97,99,100 Inactivated platelets possess extremely active SPHK, which allows the quantitative influence of rheostat parameters, because S1P is not lysed, which leads to its accumulation. Through activation, human platelets have been shown to release S1P.98,99,101,102 Ceramide and sphingosine are pro-apoptotic, while S1P promotes cell survival. 98 Through such a mechanism, platelets can direct the cell to a program of growth, neovascularization, migration, inhibition of apoptosis, leading to neoplastic proliferation or a survival program, achievable by depressing the processes mentioned, but also by activating the defense system by inflammation.96,98,103 The participation of platelets in anti-tumor defense system, is reflected by the change in the number of circulating platelets, in the sense of thrombocytosis and implicitly the modification of the marker of systemic inflammation PLR, indirectly illustrating the intensity of the peritumoral inflammation process.94,104–107
Being a review of the data published worldwide, regarding the relationship between inflammation and ovarian cancer and the practical implications of markers of systemic inflammation on prognosis, this paper has a limitation derived from the type of study used—a narrative review type.
This paper tries to support with scientific arguments the usefulness, in daily clinical activity, of markers of systemic inflammation, which are easy to calculate, being reports obtained from available blood tests, and do not require expensive and time-consuming procedures for determination. Related to the clinical significance of the mentioned biomarkers, the published studies allow us to state that the determination of their values guides the clinician in evaluating the prognosis and sometimes helps to select patients for a specific therapy.
Conclusions
This study is a synthesis of knowledge regarding the conditionality in neoplasms, between the degree of inflammation at the tumor level and the number of circulating figurative elements, which appear in the calculation formulas of the three ratios: NLR, MRL, and PLR and whose values reflect the degree of involvement of the anti-tumor defense of the immune system. The use of markers of systemic inflammation as prognostic biomarkers in different types and locations of neoplasms could effectively reflect the inflammation and immune status in vivo, which have been demonstrated to be associated with the progression and prognosis of tumors. Furthermore, prognostic markers and scores based on systemic inflammation not only identify patients at risk but also provide well-defined therapeutic targets for future clinical trials.
Footnotes
Author contributions
All authors have equal contributions to the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
