Abstract
Here we report a prenatally detected small supernumerary marker chromosome (sSMC) derived from chromosome 2 as demonstrated by cenM-FISH (centromere-specific multicolor fluorescence in situ hybridization). By application of a recently described subcentromere-specific probe set (subcenM-FISH) for chromosome 2, the presence of a small partial trisomy due to a karyotype 47,XX, + r(2)(::p11.1->q11.2::) was demonstrated. Including this case, a total of 11 patients with sSMC(2) are described throughout the literature. Based on that data, a first genotype/phenotype correlation according to the size and structure of the marker is suggested.
Keywords
A
We followed that scheme in the case of a pregnant woman whose fetus after GTG-banding was found to have the karyotype 45,X[5]/47,XX, + mar[43]/46,XX[2]. The fetus was prenatally studied (amniocytic fluid in week 21 + 6) because it was an ICSI-induced pregnancy and, according to ultrasound examination, had a double bubble sign (i.e., sonographic signs of two cystic structures in the abdomen) and a single umbilical cord artery. The parents did not want a chromosome analysis of their own. Therefore, it could not be determined if the sSMC was inherited or had arisen de novo. The result of cenM-FISH (Figure 1) indicated that the marker was a small ring derived from chromosome 2. This result was confirmed and refined by subcenM-FISH (Figure 2). Therefore, the sSMC could be characterized as 47,XX, + r(2)(::p11.1->q11.2::). The pregnancy was continued and a healthy child was born. No Turner syndrome stigmata were present, and because the parents refused a postnatal chromosome analysis it could not be clarified if a Turner mosaic, as detected prenatally, was also present in the newborn.
This is the eleventh case reported in the literature with an sSMC derived from chromosome 2. In all 11 cases the sSMC is reported to be a small supernumerary ring chromosome (sSRC). This is striking because for sSMCs derived from all other chromosomes apart from chromosome 2 there are always reports of sSMCs and sSRCs (for overview on sSMC see Liehr et al. 2004a and http://mti-n.mti.uni-jena.de/huwww/MOL_ZYTO/sSMC.htm). There is a high probability that this ring chromosome formation is due to low repetitive elements present in the pericentric region of chromosome 2 similar to that of chromosomes 10 and 12 (Jackson et al. 1999; Vermeesch et al. 2003).
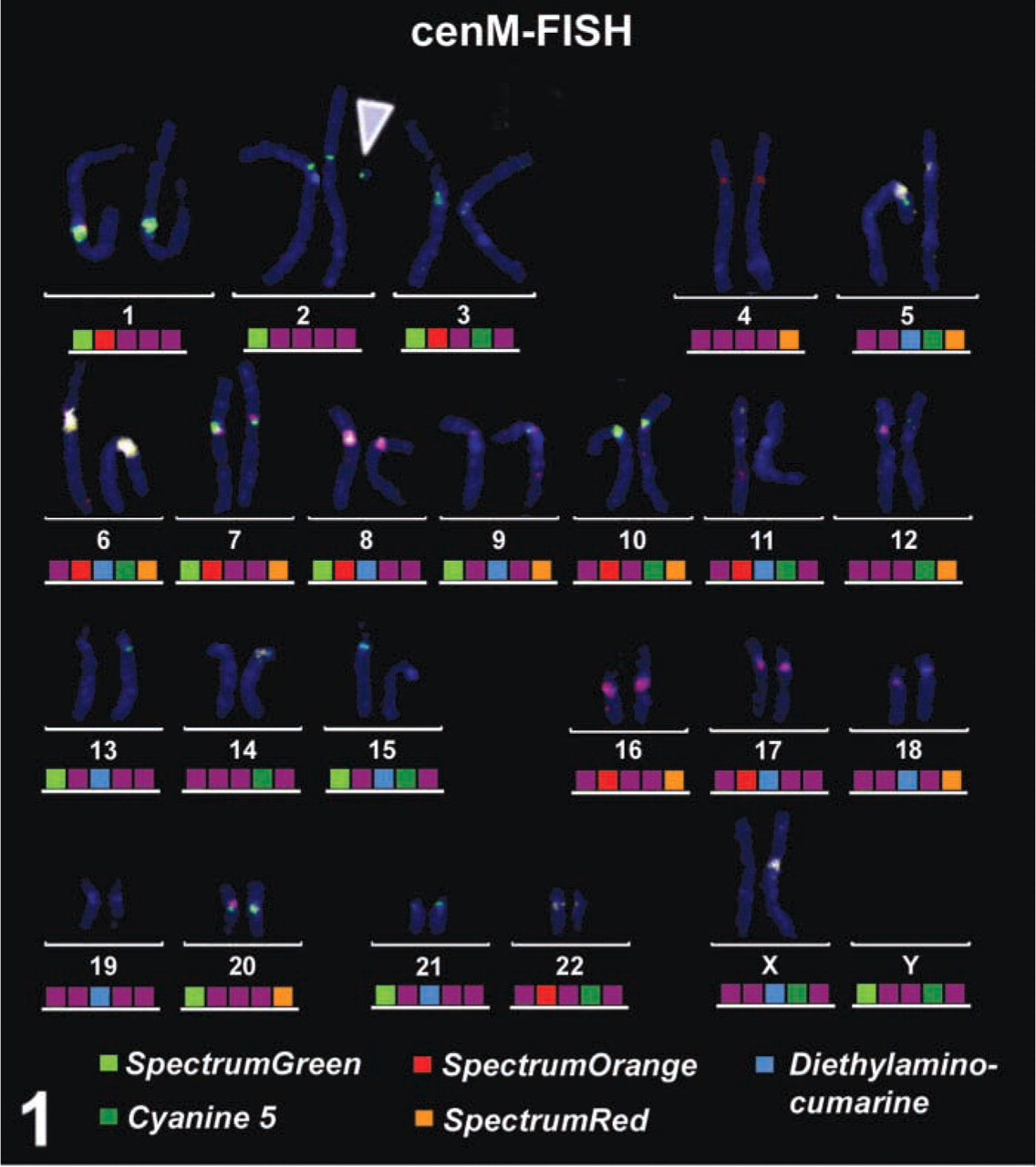
CenM-FISH (centromere-specific multicolor FISH) (Nietzel et al. 2001) result of the case reported here. The sSMC could be characterized as a ring derived from chromosome 2 (arrowhead).
Summary of all available cases with sSMC(2) a
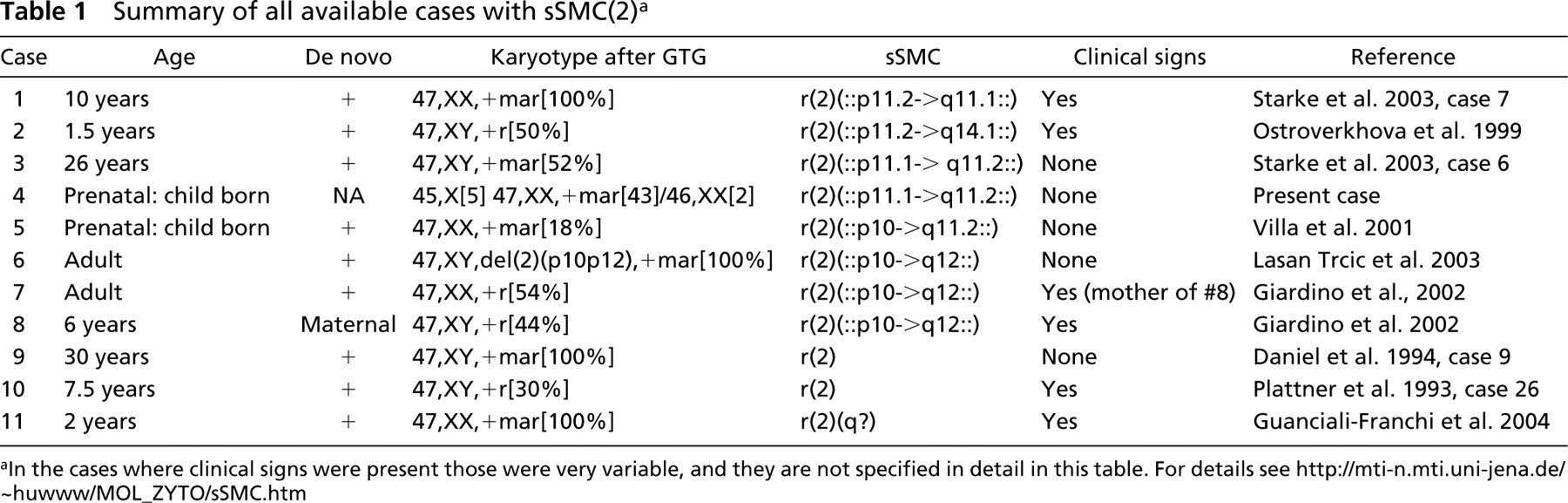
aIn the cases where clinical signs were present those were very variable, and they are not specified in detail in this table. For details see http://mti-n.mti.uni-jena.de/~huwww/MOL_ZYTO/sSMC.htm
Five of the 11 cases reported in the literature and summarized in Table 1 are without clinical symptoms. However, only eight of those 11 cases were characterized in detail for their chromosomal content. Among those characterized comprehensively, there is a prominent correlation concerning presence of centromere-near sequences of 2p11.2 plus clinical abnormalities (cases 1 and 2) and absence of clinical signs plus presence of proximal sequences of 2q11.2 (cases 3, 4, and 5). Small partial trisomies of material derived distally from 2q11.2 already lead to clinical signs (cases 7 and 8). Case 6 has no clinical abnormalities and an sSMC similar to that of cases 7 and 8, but in case 6 there is no partial trisomy because a balanced karyotype 47,XY,del(2)(p10p12), + r(2)(::p10->q12::) was diagnosed. Therefore, we can speculate that case 9 consists of 2p centromeric and/or 2p11.2-derived material. Case 10 may include 2p11.2 or material distal from 2q11.2 and for case 11, for which the presence of 2q material is proven, the latter suggestion made for case 10 may also be true.
Even though uniparental disomy (UPD) must always be considered in case of an sSMC (Liehr et al. 2004a) and UPD 2 was excluded only for cases 5 and 8 in Table 1, we provide evidence for a first genotype/phenotype correlation of one small subgroup of sSMCs, those derived from chromosome 2 (Figure 3). It must be checked whether such a relatively simple correlation can be confirmed by further cases, e.g., for sSMC derived from chromosome 3 the situation seems to be more complex and is not yet resolved (Liehr et al. 2004c).
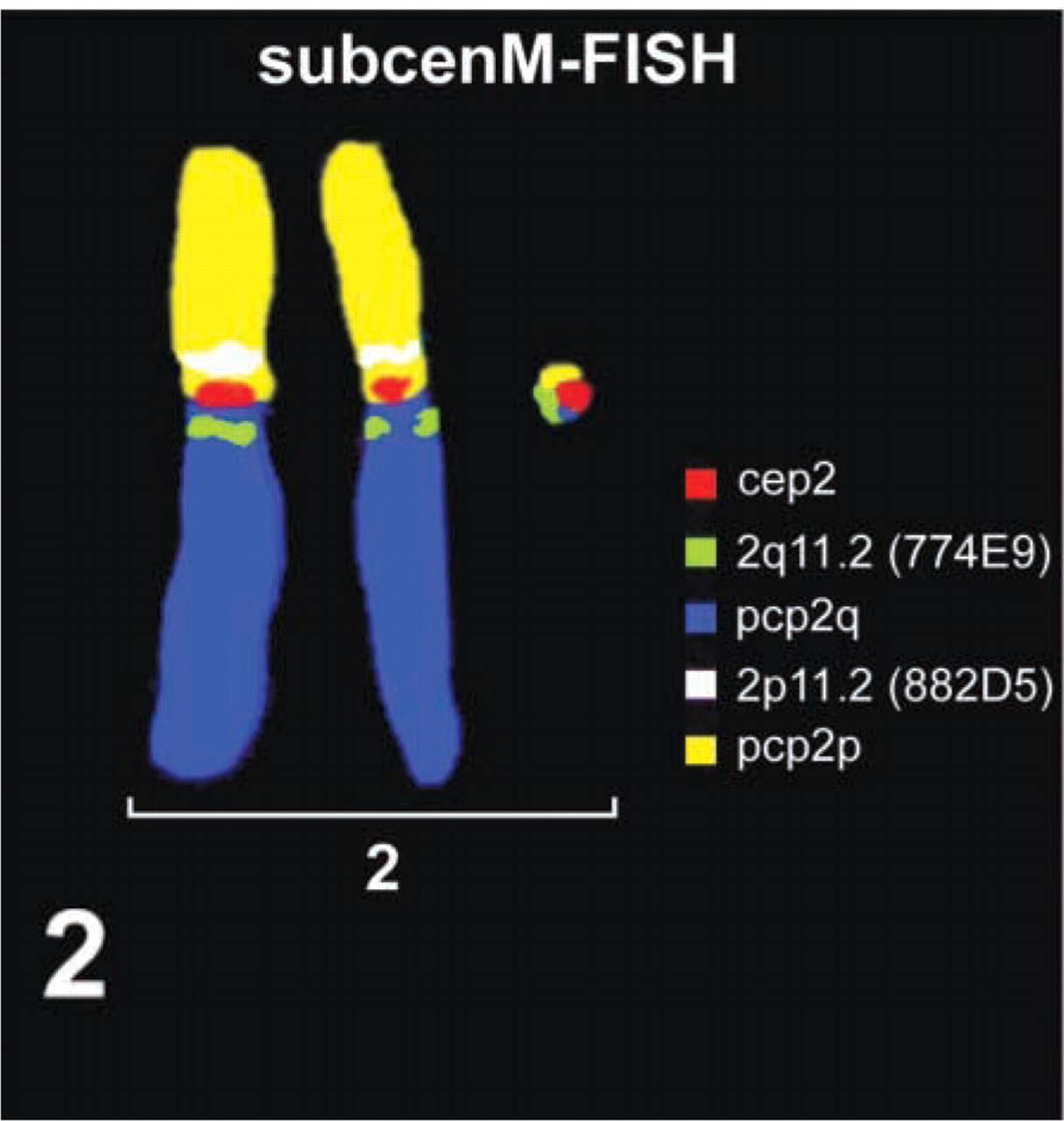
SubcenM-FISH characterized the sSMC as r(2)(::p11.1->q11.2::) (Starke et al. 2003).

Summary from the literature of the partial trisomies in well-characterized cases with sSMC 2. The case numbers are according to Table 1; the present case is marked by an arrowhead. Clinical signs are to be expected when the sSMC contains sequences derived from 2p11.2 or distal from 2q11.2. Green color, no clinical signs; red color, clinical signs.
Footnotes
Acknowledgements
Supported in part by Dr Robert Pfleger Stiftung and the EU (1CA2-CT-2000–10012).
The case was kindly provided by Drs M. Kosskiewicz and H. Tittelbach (Nürnberg, Germany).
