Abstract
The majority of newly acquired HIV infections are believed to occur following transmission of virus infectivity across mucosal surfaces, although many mechanistic details still remain unresolved. We have used human ex vivo organ cultures and primary cell populations to analyze the cellular and molecular basis for mucosal HIV transmission. By using human palatine tonsil from routine tonsillectomies and semen from HIV-positive donors, we have created an experimental equivalent to oral HIV transmission. HIV infection was readily transferred into tonsillar lymphocytes, but this transmission into lymphocytes was dramatically reduced when the exposed lymphocyte populations were protected by intact mucosal surfaces. In this study, we consider the impact that leukocyte activation and morphological aberrations in surface structure may have on susceptibility to primary HIV infection and introduce novel time-lapse confocal microscopy procedures that begin to reveal the dynamic complexity associated with cell-mediated HIV transmission. This manuscript contains online supplemental material at http://www.jhc.org. Please visit this article online to view these materials.
Keywords
I
Oral transmission of HIV is believed to occur following the introduction of cell-free virions or HIV-infected cells into the oral cavity. Both milk and semen are recognized as potential sources of HIV infectivity for oral transmission by nursing and by receptive oral intercourse, respectively (Crittenden et al. 1992; Dunn et al. 1992; Black 1996; Vernazza et al. 1996; Southern 1998; Semba et al. 1999). Additional evidence supporting a direct connection between exclusive oral exposure and subsequent establishment of disseminated virus infection has been derived from atraumatic oral infections in the SIV/rhesus macaque model system (Baba et al. 1996; Stahl-Hennig et al. 1999). SIV-infected cells have been detected in macaque palatine tonsil, but many details relating to virus exposure and the initiation of primary infection have not been fully resolved.
The palatine tonsil is one of several secondary lymphoid tissues situated in the human pharynx that constitutes part of the mucosa-associated lymphoid tissue (MALT), reviewed in (Perry 1994; Neutra et al. 1996; Perry and Whyte 1998). The bulk of the external surface of the palatine tonsil is protected by a stratified squamous epithelium, but there are periodic invaginations (tonsillar crypts) that increase the total surface area of tonsil ∼6-fold (Perry and Whyte 1998). The crypts are lined by a reticulated epithelium that is populated predominantly by epithelial cells and leukocytes. The specialized composition of cryptal reticulated epithelium is connected with antigen sampling, and functional parallels have been drawn between palatine tonsil and Peyer's patches in the intestine (Neutra et al. 1996). There have been many suggestions that tonsillar crypts may be exploited as a portal of entry by both bacteria and viruses in the oral cavity (Mbopi-Keou et al. 2002), and we have therefore designed a series of experimental systems to analyze the events surrounding exposure and primary infection at the surface of human palatine tonsil. We believe that the human organ culture system can be manipulated to provide unique insights into the process of HIV exposure in the oral cavity, and that this insight will be invaluable in guiding the design of novel strategies to protect mucosal surfaces from both cell-free HIV virions and cell-associated HIV.
Materials and Methods
Sources of HIV Infectivity: Virus Stocks and Virus-infected Cells
A high-titer stock of cell-free HIV was prepared by in vitro passage of a primary patient isolate in heterologous peripheral blood mononuclear cells. This stock (HIV96–480) has a p24 content of 200 pg/ml and has dual tropic properties. The chronically infected T-cell line, H9/HTLVIIIB (Popovic et al. 1984), designated here as H9/HIV, was maintained in complete RPMI medium [RPMI basal medium with 10% heat-inactivated fetal calf serum and antibiotics: 200 units/ml penicillin, 200 U/ml streptomycin, 500 ng/ml amphotericin B (Antibiotic-Antimycotic #15240–062; Invitrogen, Carlsbad, CA)]. For some experiments, exponentially growing H9/HIV cells were labeled with 5-(and 6)-carboxyfluorescein diacetate succinimidyl ester (CFSE; Molecular Probes, Eugene, OR) using standard cell-labeling protocols (Weston and Parish 1990).
Palatine Tonsil Tissue Samples
Tissues were obtained from routine tonsillectomies performed at the Fairview University Medical Center (FUMC), Minneapolis, Minnesota with the assistance of the FUMC Tissue Procurement Facility. The experimental protocols had full Institutional Review Board approval, and individual patient consent for the use of tissue in research applications was obtained prior to initiation of the surgery. Pieces of palatine tonsil were routinely delivered within 1–3 hr of completion of surgery, and any tissue with recognizable macroscopic abnormality was automatically rejected. The tissue was randomly cut into small pieces (∼1–3-mm cubes) with either scissors and forceps or a pair of scalpel blades. The resultant tissue pieces were cultured on nitrocellulose membranes (0.4-μm pore size) placed on top of collagen supports (Gelfoam; Upjohn/Pharmacia, Kalamazoo, MI) or on Falcon transwell membranes (0.4-μm pore size; Becton Dickinson Labware, Franklin Lakes, NJ) at the gaseous/liquid medium (complete RPMI) interface in a standard CO2 incubator (Figure 1). Tissue pieces on transwell membranes supported the outgrowth of primary epithelial cells. On a historical note, our methods for the outgrowth of tonsillar epithelial cells and culture of small tonsil pieces are actually related to procedures that were first published many years ago (Shapiro and Volsky 1983; Ferro et al. 1993). Single-cell suspensions of tonsillar cells, comprised predominantly of B and T lymphocytes, were prepared by forcing small tissue pieces through a wire mesh with a syringe plunger as is routinely performed to disrupt mouse spleen. After extensive washing, the released cells were either cultured in complete RPMI or resuspended in fetal calf serum + 10% DMSO and frozen in liquid nitrogen. In some experiments, suspensions of freshly disrupted tonsil cells were purified away from erythroctyes by banding on Ficoll gradients (Histopaque 1077; Sigma, St Louis, MO).
Randomly cut pieces of tonsil were infected by careful application of 5-μl drops of the cell-free virus stock, such that only the drops came into contact with the tissue surface. The HIV infections were performed as soon as possible after preparation of the tissue pieces, generally within 1–2 hr of receiving the tonsil into the laboratory. In this experimental scheme, with randomly cut small tonsil pieces, no efforts were made to keep track of orientation relative to the external surface and, therefore, infection occurred at cut surfaces. In contrast, to study infection exclusively at intact tonsillar surfaces (experiments shown in Figures 3E–3G and Figure 6) we used much larger tissue pieces with stratified squamous epithelium (10–20 mm2) and covered cut tissue surfaces with a protective agarose seal (Maher et al. 2004). The tissue surfaces remained moist throughout the infection period but, in most cases, no deliberate effort was made to wash away the virus inoculum. Infections were terminated by transferring tissue grown on collagen supports into tissue cassettes followed by immersion in either 10% formalin or Streck Tissue Fixative (STF; Streck Laboratories, La Vista, NE). These fixed tissue pieces were embedded in paraffin and sectioned (5 μm) for general assessment of tissue architecture (hematoxylin and eosin staining). Single-cell in situ hybridization analysis was performed exactly as reported previously (Zhang et al. 1999); hybridization specificity was confirmed by the use of matched SIV-specific probes as negative controls. Cells were counterstained lightly with hematoxylin and mounted in Permount (Fisher Scientific; Atlanta, GA) for microscopic evaluation.
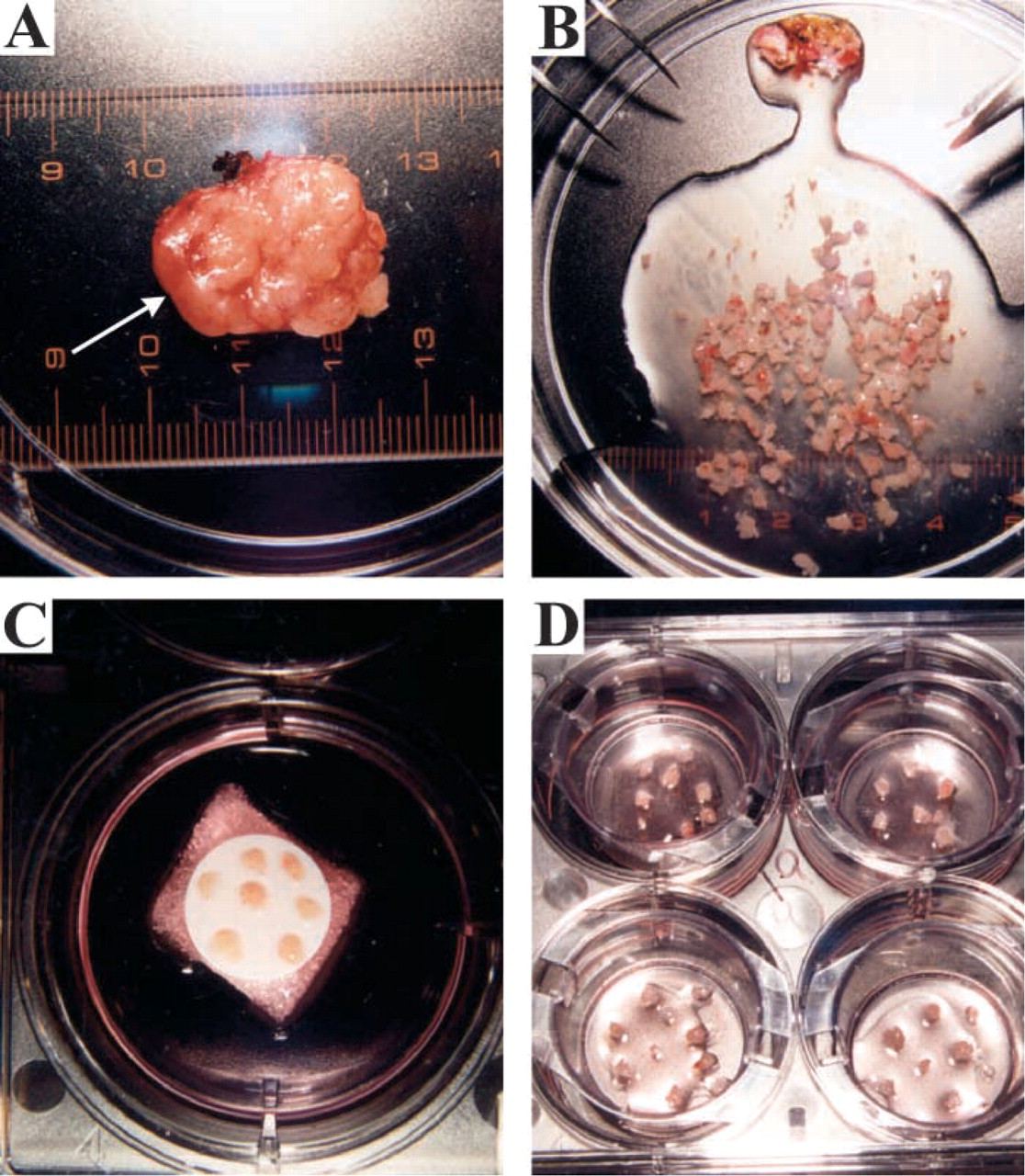
Preparation and culture of palatine tonsil tissue pieces. (
Processing of Seminal Samples
Freshly expressed semen samples were transported to the laboratory at room temperature and processing was initiated within 1–4 hr of sample collection. The semen was mixed by pipetting to ensure homogeneity, and the seminal cells were collected by centrifugation (10 min, 1500 rpm, 15C). The cell-free supernatant (seminal plasma fraction) was carefully removed and dispensed into Eppendorf tubes for long-term storage at −80C. The seminal cell pellets were washed twice with complete Hanks’ Balanced Salt Solution (HBSS) (30 ml/wash) and then resuspended either in complete RPMI for experimental infection transfer or in HBSS for spotting onto microscope slides. The cellular content of semen can differ widely from donor to donor and, in addition to mature spermatozoa, there are variable numbers of immature round germ cells and leukocytes. The leukocyte population consists primarily of neutrophils, although T cells and macrophages may also be present.
PCR Amplification of HIV Target Sequences
Randomly cut pieces of tonsil were positioned on nitrocellulose membranes on top of collagen supports, and HIV infections were initiated with cell-free virus stocks of the primary patient isolate, HIV96–480. The infected tissue pieces were incubated at the gaseous/liquid interface and, at various times after infection, tissue pieces were removed from the membranes and immediately disrupted in Trizol (Invitrogen; Carlsbad, CA). Total RNA was recovered in accordance with the manufacturer's recommended procedure. cDNA was synthesized using a random primer (Invitrogen), and PCR amplifications were performed under standard hot-start conditions with conserved primers from the HIV-1 gag coding region: forward primer: 5'-GTCAGCCAAAATTACCCTATAGTGCAGAAC; reverse primer: 5'-ACATAGTCTCTAAAGGGTTCCTTTGGTCCT with 2.5 mM MgCl2, and 40 two-step cycles (94C, 45 sec; 72C, 120 sec) followed by a final extension for 10 min at 72C. Amplification products were analyzed by agarose gel electrophoresis and visualized by ethidium bromide staining and UV illumination.
Immunocytochemistry and Immunofluorescence
The following antibodies were used at a 1:100 dilution: HIV p24 (clone Kal-1; DAKO, Carpinteria, CA), S100 (rabbit polyclonal antibody, DAKO), Ki67 (Novocastra Laboratories; Newcastle, UK), and CD45RO (clone UCHL-1; Biogenex, San Ramon, CA). Negative controls for immunocytochemistry and immunofluorescence were appropriate for the primary antibody and confirmed specific detection of the desired antigen: HIV p24, matched, non-infected tissue; S100, omission of the primary antibody; and CD45RO, an isotype control antibody (mouse IgG; Vector Laboratories, Burlingame, CA). Secondary antibodies were conjugated to biotin for ABC amplification (Vector Laboratories) or cyanine 3 and cyanine 5 (Jackson ImmunoResearch Laboratories; Westgrove, PA) for fluorescent signals. Standard detection procedures were used; briefly, samples were prepared, processed through antigen retrieval and peroxidase blocking steps as needed, blocked with 1.5% serum, and incubated with the appropriate primary antibody overnight at 4C. Samples were then washed, re-blocked, and incubated with the appropriate secondary antibodies and detection systems for 1 hr at room temperature and washed and mounted in Permount and stored at room temperature or, for immunofluorescence, samples were mounted in Vectashield Hardset (Vector Laboratories) and stored at 4C until viewed.
Confocal Microscopy
Fluorescent images were collected with a Bio-Rad 1024 Laser Scanning Confocal microscope (Bio-Rad; Hercules, CA) equipped with a krypton/argon laser and processed with Confocal Assistant (written by Todd Clerke Brelje, University of Minnesota, Minneapolis, MN) and Adobe Photoshop (Adobe Systems Inc.; San Jose, CA). The time-lapse images were obtained by serial collection of a z-series repeated at 4-min increments.
Scanning Electron Microscopy (SEM)
Tissue was fixed in 2.5% glutaraldehyde in 0.1 M sodium cacodylate buffer, pH 7.4, for 10 hr, rinsed twice in the same buffer, and postfixed in 1% osmium tetroxide, again in the same buffer, for 45 min. The samples were then dehydrated through a graded series of alcohols, critical-point dried from liquid CO2, and coated with 2–3 nm platinum by indirect ion-beam sputtering before examination in a Hitachi S-4700 field-emission SEM (Hitachi High Technologies America; Pleasanton, CA).
Results
As a first approach to explore the susceptibility of tonsil lymphocytes to HIV infection, randomly cut pieces of palatine tonsil (1–3 mm3 pieces with no discernible external epithelium) were maintained in organ culture for 4–14 days (Figure 1). There was some cell loss from the tissue pieces—both cell death and leukocyte emigration occurred during the first 72 hr, but the cellular content stabilized after that time. Cell viability in the organ culture systems was confirmed by finding that many of the tissue pieces acquired a surface coating of epithelial cells after 5–7 days in culture. As expected from previously published studies (Glushakova et al. 1995), when cut pieces of tonsil were infected with cell-free HIV, there was a time- and virus-dose-dependent increase in the number of HIV-infected cells (primarily CD4+ T cells), as judged by accumulation of intracellular HIV p24 antigen. At day 1 postinfection with a primary patient isolate of HIV, HIV96–480, p24 signal was not detectable (Figure 2A), but by days 4 and 6, there were numerous clusters of cells that were strongly positive for intracellular p24 (Figures 2B and 2C). A 10-fold dilution of the HIV96–480 inoculum resulted in at least 10-fold lower numbers of infected cells at day 6, but the apparent intensity of p24 staining per cell was not affected by dilution of the virus inoculum (Figures 2D–2F). HIV infection was also established in tonsil tissue pieces using cell-free virions from the tissue-culture-adapted HIV-1 strain, LAI, at generally comparable frequencies (data not shown), thereby validating the use of the primary patient isolate for these experimental HIV infections.
Active replication of the HIV96–480 primary patient isolate in experimentally infected tonsil pieces was confirmed by RT-PCR amplification of HIV target sequences. When RNA was extracted from tissue pieces immediately after exposure to the cell-free virus, a weak HIV-specific signal was detected that represented amplification of HIV genomic sequences in the HIV96–480 inoculum (Figure 3A). In comparison, RT-PCR amplification with an RNA template preparation extracted 6 days after infection showed a 10- to 50-fold increase in HIV RT-PCR product (Figure 3A). HIV RNA was also detected in the supernatant from HIV-infected tonsil pieces collected at 6 days postinfection (Figure 3A).
Additional independent confirmation of HIV96-480 replication within cut tissue pieces was obtained from a combined immunocytochemistry/in situ hybridization procedure (Zhang et al. 1999). Cytoplasmic HIV RNA was observed in both activated T cells (Ki67+ cells) and resting T cells (Ki67- cells) in the tonsil organ culture system (Figure 3B). This finding exactly matched the results obtained from a rhesus macaque model for SIV sexual transmission, wherein SIV replication was detected in both activated and resting T cells in proximity to exposed mucosal surfaces and in the draining lymph nodes of vaginally infected macaques (Zhang et al. 1999). The susceptibility of tonsillar lymphocytes to direct HIV infection was also demonstrated using tonsil-cell suspensions. Following exposure to cell-free HIV96–480, infected T cells were readily detected at day 7, in the absence of any exogenous cytokine stimulation of the tonsillar lymphocyte population (Figure 3C). The role of an intact epithelium in impeding the initiation of primary HIV infection was demonstrated by comparing adjacent pieces of tissue from the same tonsil. These tissue pieces were embedded in agarose to cover all cut tissue surfaces (Maher et al. 2004), and then the external epithelial surfaces were damaged to varying degrees prior to exposure to HIV96–480 virions. In these experiments, ∼5- to 10-fold more infected cells were detected in tissue pieces either lacking epithelium or with damaged epithelium at the time of infection when compared with tonsil pieces that retained an undamaged epithelial surface during exposure to infectious HIV (Figures 3E–3G).
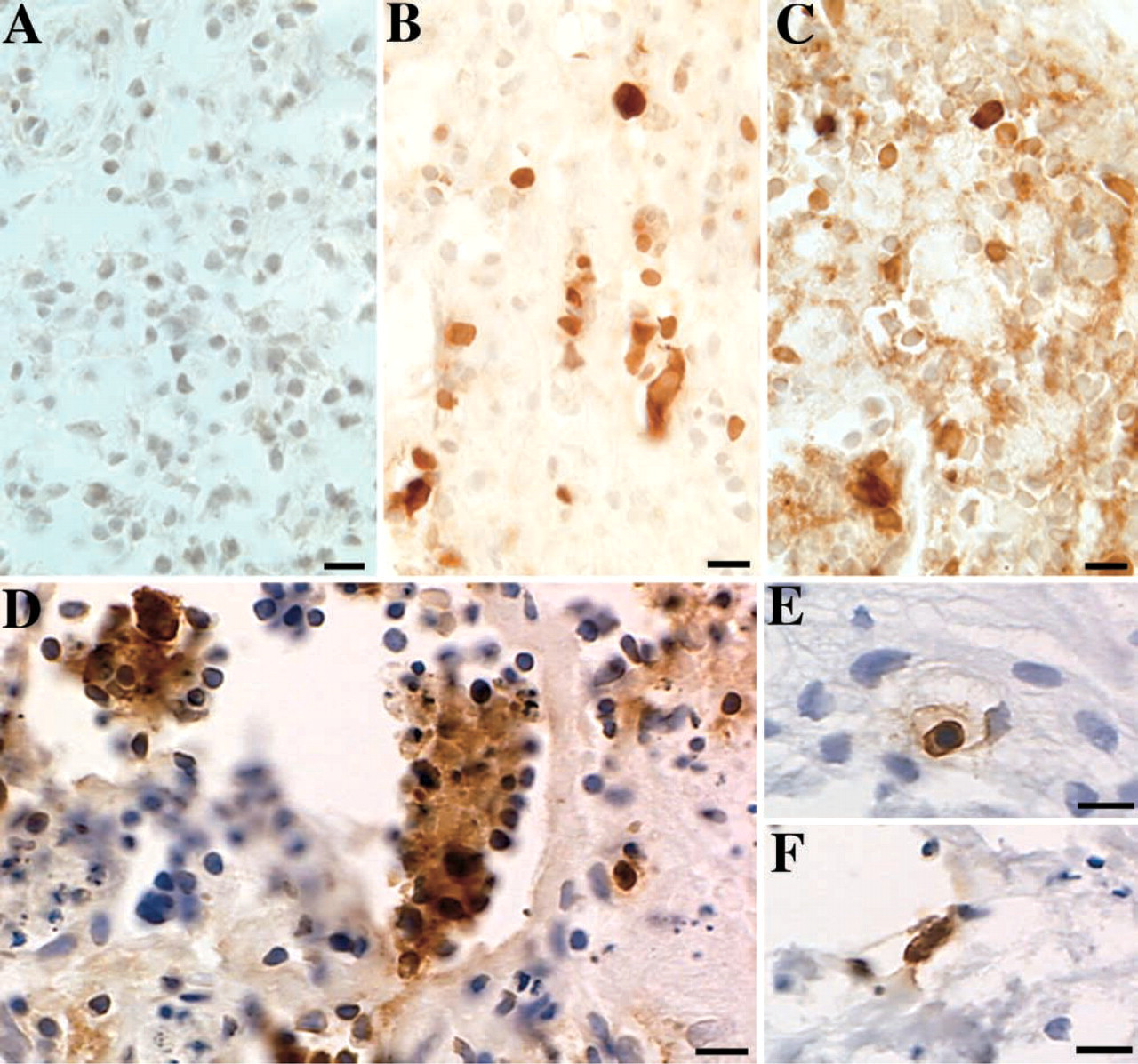
Time- and dose-dependent accumulation of HIV p24 gag in T cells contained within infected tonsil pieces. Small, randomly cut pieces of tonsil were infected with cell-free HIV (HIV96–480 primary patient isolate) and cultured for the indicated times. Tissue pieces were fixed in Streck Tissue Fixative (STF) and embedded in paraffin. Five-μm sections were processed for immunocytochemical detection of HIV p24 (brown indicates positive signal). These results are representative of more than eight independent tonsil infections with HIV96–480, the primary patient isolate. Bars = 10 μm. (
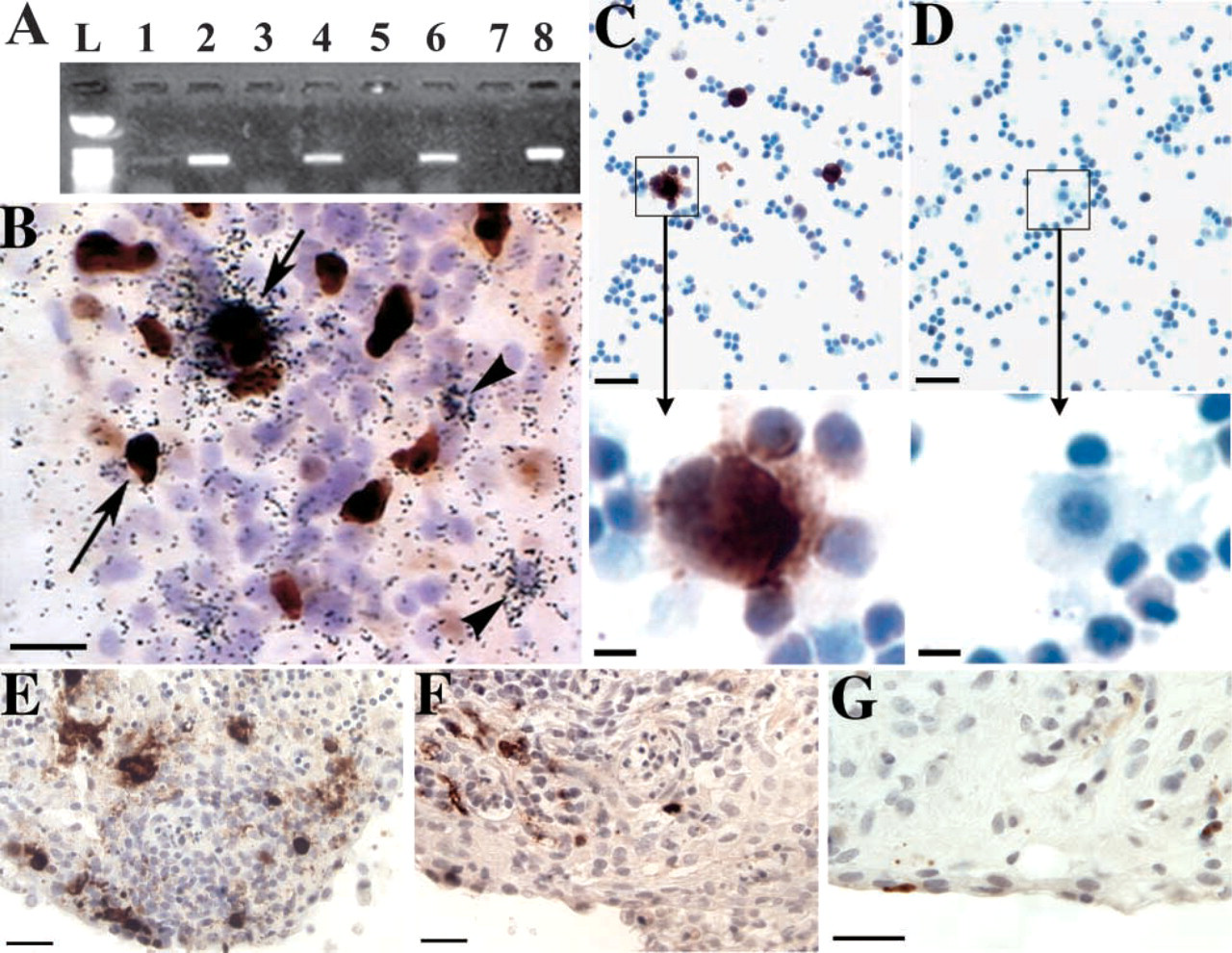
RT-PCR amplification of HIV RNA extracted from infected tonsil pieces. Small, randomly cut pieces of tonsil tissue were infected with HIV96–480 (virus inoculum equivalent to 1.0 pg of p24), cultured for the indicated times, and then processed for total RNA extraction and RT-PCR. L: ladder; 1: HIV-infected, day 0, tissue lysis immediately after addition of the virus; 2: HIV-infected, day 5; 3: no added HIV, day 5; 4: HIV-infected, day 13; 5: no added HIV, day 13; 6: HIV-infected, day 6, culture supernatant; 7: no added HIV, day 6, culture supernatant; 8: HIV PCR positive control. (
To begin to reconstruct elements of biological complexity that are associated with natural routes of HIV-1 transmission, we have attempted to transfer HIV infection into tonsillar T cells using semen from HIV-seropositive individuals. Because of concerns regarding potential cytotoxic properties of seminal plasma (Fiore et al. 1997a), we have routinely separated seminal fluid into two fractions: seminal cells and cell-free seminal plasma. Coculture of viable seminal cells (a mixture of mature spermatozoa, immature round germ cells, and leukocytes) with a single-cell suspension of primary tonsillar cells resulted in transfer of HIV infection into the tonsillar T-cell population and accumulation of p24 antigen in the culture supernatant. Soluble p24 levels increased 100-fold between day 5 (17.5 pg/ml) and day 17 (1.75 ng/ml) of culture for the exposed tonsillar lymphocyte populations. There was no detectable accumulation of p24 when matched populations of tonsillar cells were exposed to cell-free seminal plasma from the same semen donor. In subsequent experiments, we determined that 80% to 90% of tonsillar lymphocytes were killed by overnight incubation in seminal plasma (10% v/v seminal plasma in complete RPMI; 22 hr at 37C in a standard CO2 incubator). Thus, oral exposure to HIV infectivity in semen may involve a balance between direct cytotoxic effects of seminal plasma and cell-to-cell interactions that can successfully transfer HIV infectivity into tonsillar T cells.
As another indicator of tonsillar lymphocyte susceptibility to HIV infection and the ability of infected tonsillar lymphocytes to release infectious progeny HIV virions, we used the day 17 (1.75 ng/ml p24) filtered tissue culture supernatant described above as a virus inoculum to transfer HIV into new tonsillar lymphocyte suspensions, derived from two completely independent adult donors. In both cases it was possible to observe the time-dependent appearance of HIV infection in the exposed tonsil-cell populations (p24 immunocytochemical staining of infected T cells, data not shown). This demonstration of the production of infectious HIV within tonsillar lymphocyte populations provides a useful experimental correlate for the observation of HIV shedding in the oral cavity of some HIV-seropositive individuals (Zuckerman et al. 2003) because infected tonsillar T cells could be regarded as a potential source of HIV to contribute to oral shedding.
Collectively, the preceding results highlight the importance of a fully intact tonsillar epithelium to provide a physical barrier between HIV virions and the otherwise readily susceptible tonsillar lymphocytes. However, as part of an ongoing survey of the morphological characteristics of tonsillar epithelium, we have examined a representative series of tonsils that were immediately fixed on receipt into the laboratory. Somewhat surprisingly, localized irregularities, including lymphocyte invasion of the epithelium, were commonly observed in the stratified squamous epithelium covering the external surface of the palatine tonsil in 10 of 11 randomly selected tonsils (Figure 4A). Immunocytochemical staining with an antibody directed against CD45RO (clone UCHL-1) (Janossy et al. 1989) confirmed that activated T cells could readily be detected within and immediately below the stratified squamous epithelium (Figure 4B). Furthermore, in examining tonsil pieces by SEM, it was possible to identify occasional distinctive cells located at the tonsil surface and to recognize an abrupt transition in the topographical structure of the surface (Figures 4C and 4D). Considered together, these structural aberrations may have profound implications for HIV transmission by allowing potentially susceptible lymphocyes to be situated in proximity to infectious inocula that had been deposited onto the external surface of the tonsil.
As another experimental approach to evaluate the protective importance of an intact stratified squamous epithelium, we have developed culture conditions to expand populations of primary tonsillar epithelial cells. Epithelial cell populations proliferate outwards from the edges of tonsil tissue pieces placed on 0.4-μm transwell membranes and, although the cell populations are not homogeneous, a majority of cells express cytokeratins and form tight junctions (Maher et al. 2004). By virtue of this proliferative ability, these tonsil epithelial cells are likely to be derived from the basal epithelial layer and/or tonsillar reticulated epithelium (Perry and Whyte 1998). On the basis of the histopathological complexity that we observed at the tonsil surface (Figure 4), we were interested in assessing whether cell-free or cell-associated HIV infectivity could bind to primary tonsillar epithelial cells, as a prelude to establishment of primary HIV infection. We were unable to find any consistent evidence for infection of primary tonsil epithelial cells by cell-free HIV (data not shown), although it was possible to visualize stable binding of HIV virions to these primary cell populations (Maher et al. 2004). When primary tonsil epithelial cells were cocultured with HIV-infected lymphocytes (H9/HIV), we observed numerous lymphocyte–epithelial cell complexes, formed by fusion of the HIV-infected lymphocytes to the epithelial cell layers and the appearance of multinucleated giant cells containing high levels of HIV p24 gag protein (Figure 5). This process of absorption of HIV infectivity by the primary epithelial cells may be significant for HIV transmission across mucosal surfaces, especially when the surface is not fully protected by stratified squamous epithelium.
In a final series of experiments, we have used confocal microscopy to begin to describe the sequence of events that unfolds after HIV exposure at mucosal surfaces. HIV-infected cells could bind stably to the epithelial surface of live tonsil pieces (Figure 6A), and in this image the infected lymphocytes are positioned at the entrance to a crypt, in proximity to S100-positive dendritic cells situated sporadically throughout the palatine tonsil (Noble et al. 1996; Papadas et al. 2001; Maeda et al. 2002). Although dendritic cells have been recognized as participating in one uptake mechanism for cell-free HIV virions through virion binding to DC-SIGN (Geijtenbeek et al. 2000; Pohlmann et al. 2001), the role of dendritic cells in the uptake of HIV-infected foreign cells is far less defined. The dynamic complexity of cell–cell interactions at epithelial surfaces was best revealed by time-lapse confocal microscopy. We observed some lymphocytes migrating across the epithelial surface and other lymphocytes stably bound to the epithelial surface within 60 min of exposure (Figure 6B). We are currently attempting to identify the ligand–receptor interactions that may facilitate lymphocyte binding to epithelial surfaces, but we have observed that fluorescently labeled latex spheres (4-μm diameter) can also bind to epithelial surfaces or become trapped on epithelial surfaces (data not shown). However, it is extremely unlikely that the cell distortions visible as cells migrated across the tonsil surface could reflect nonspecific interactions (Figure 6B). In the context of HIV transmission it must be recognized that both specific and nonspecific mechanisms may contribute to lymphocyte binding and retention at epithelial surfaces and that such binding may be a prelude to transfer of HIV infectivity across an otherwise intact mucosal barrier.
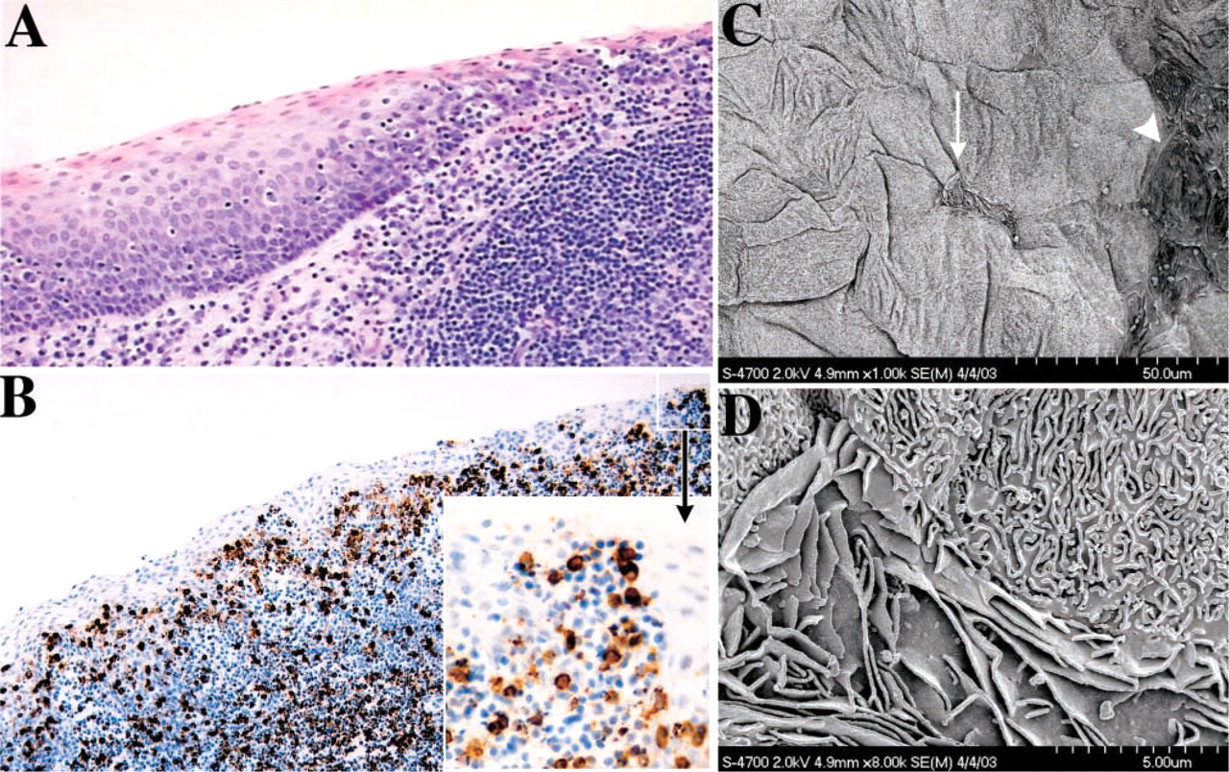
Microscopic surface irregularities in tonsillar epithelium. (
Discussion
Organ cultures provide a versatile experimental system for studying the primary events surrounding HIV transmission and the initiation of HIV infection in human cells. Partially overlapping information could be obtained by experimental analysis of tissue biopsies taken from newly exposed individuals but, in the context of community-acquired infections, characterization of the inocula may not be possible and anti-retroviral therapy may be instigated at the time of patient presentation. Thus, from the standpoint of both controlling and then adjusting experimental variables, there are numerous opportunities and benefits to be derived from studies relying on human organ cultures now that the systems have been validated (Glushakova et al. 1995,1999; Collins et al. 2000; Greenhead et al. 2000; Gupta et al. 2002; Zhao et al. 2004). We have shown that cell-free HIV (HIV96–480, a low passage primary patient isolate) can establish infections in tonsillar leukocytes when infectivity is applied directly to the surface of cut tonsillar tissue (Figure 2 and Figures 3A, 3B, and 3E) and that primary suspensions of tonsillar lymphocytes are readily susceptible to cell-free HIV infection (Figure 3C). We have also confirmed that HIV infectivity present in semen can initiate infection in tonsil cells in a process that must resemble receptive oral intercourse.
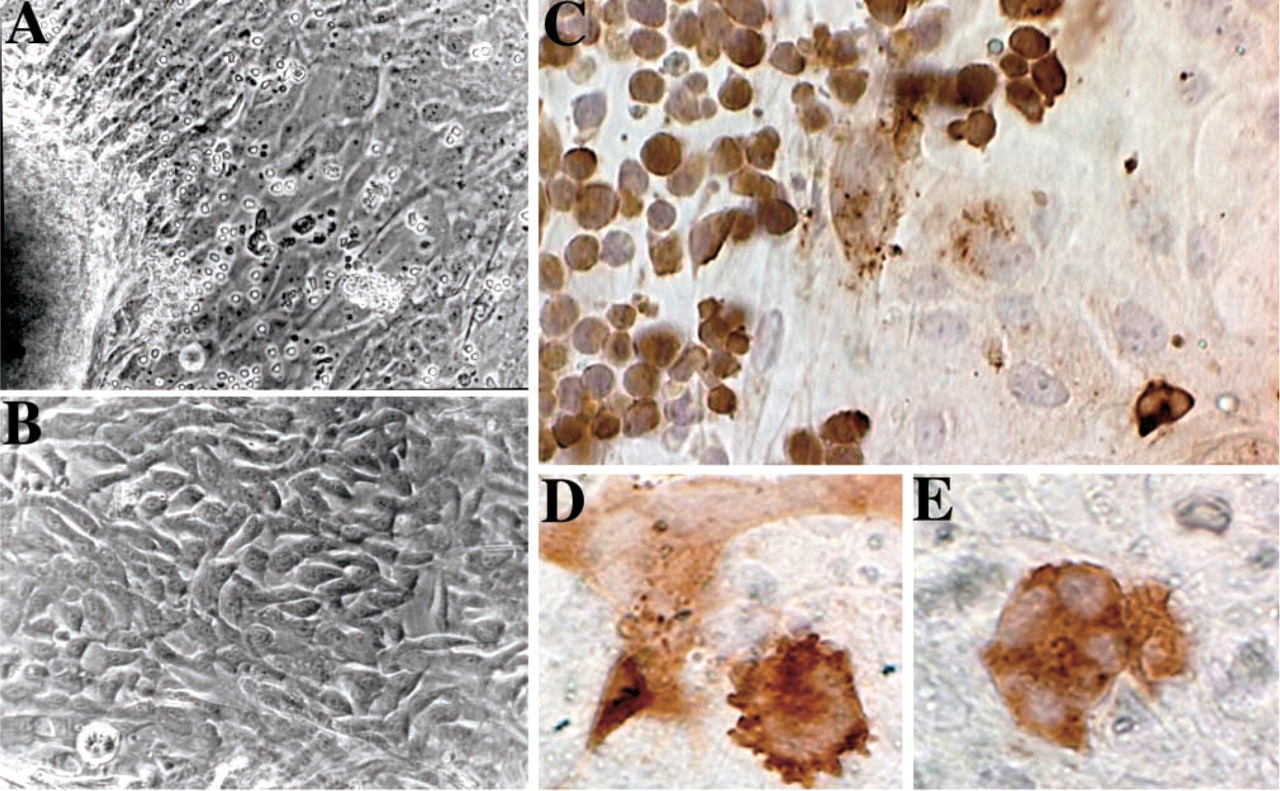
HIV infection of primary tonsillar epithelial cells. (
In total, the findings reported here provide strong support for the identification of tonsillar tissue as a portal of entry for HIV infectivity and site for primary HIV infection, following oral exposure. Several other viruses, including polio virus, JC virus, cytomegalovirus, Epstein–Barr virus (EBV), Kaposi's sarcoma herpes virus, and human herpes virus 6, are believed to use tonsil as the site of primary infection (Goudsmit et al. 1982; Monaco et al. 1996; Mbopi-Keou et al. 2002). These viruses probably employ different mechanisms to traverse the tonsillar epithelium, but for EBV there is some evidence that tonsillar epithelial cells may be susceptible to productive virus infection (Borza and Hutt-Fletcher 2002). In the same general context, it is interesting to note that tonsillar inflammation has been implicated as one possible mechanism whereby consumption of meat products from cattle affected by bovine spongiform encephalopathy could lead to the onset of variant Creutzfeld–Jakob Disease in young adults (Nailon and Ironside 2000; Hilton et al. 2002).
We believe that multiple mechanisms may account for HIV transmission across the tonsillar epithelium, and that there will be a recognizable connection between the specific properties of the HIV inoculum (cell-free virions or cell-associated infectivity) (Crittenden et al. 1992; Quayle et al. 1997; Fiscus et al. 1998), the macro- and microarchitecture of the target tissue at the exposed sites, and the mechanism of infection transfer. If the epithelium has been transiently damaged either by recent trauma or by previous or ongoing microbial infection, then HIV infectivity may gain direct access to tonsillar leukocytes in a process that is largely analogous to our experimental infection of cut tonsillar tissue pieces in organ culture (Figure 2 and Figures 3A, 3B, and 3E). With cell-associated infectivity, HIV-infected cells in the inoculum could fuse to surface epithelial cells (Figure 5) and then, at least for reticulated epithelium, release of virions from the basolateral epithelial cell surface would position HIV infectivity in immediate proximity to CD4+ leukocytes. In addition to fusion at the epithelial surface, infected donor cells may even invade the epithelium. Although any such invading donor cells should rapidly be removed by phagocytosis, the process of local recruitment of activated recipient cells in response to recognition of foreign antigens may serve to facilitate the initiation of primary HIV infection. Any temporary disruption or relaxation of tight junctions formed between epithelial cells could allow paracellular transport of virions or infected cells across the epithelial surface. The process of transcytosis has been carefully described for transfer of HIV virions across monolayers of cultured epithelial cell lines in vitro (Bomsel 1997; Belec et al. 2001), but it is far from clear if transcytosis is involved in HIV uptake in natural infections. Any one of these distinct uptake mechanisms may be sufficient to establish primary HIV infection at an exposed mucosal surface. However, the combination of different uptake mechanisms with the morphological irregularities that we have observed in epithelial surfaces can be projected to lead to increased susceptibility for primary HIV infection.
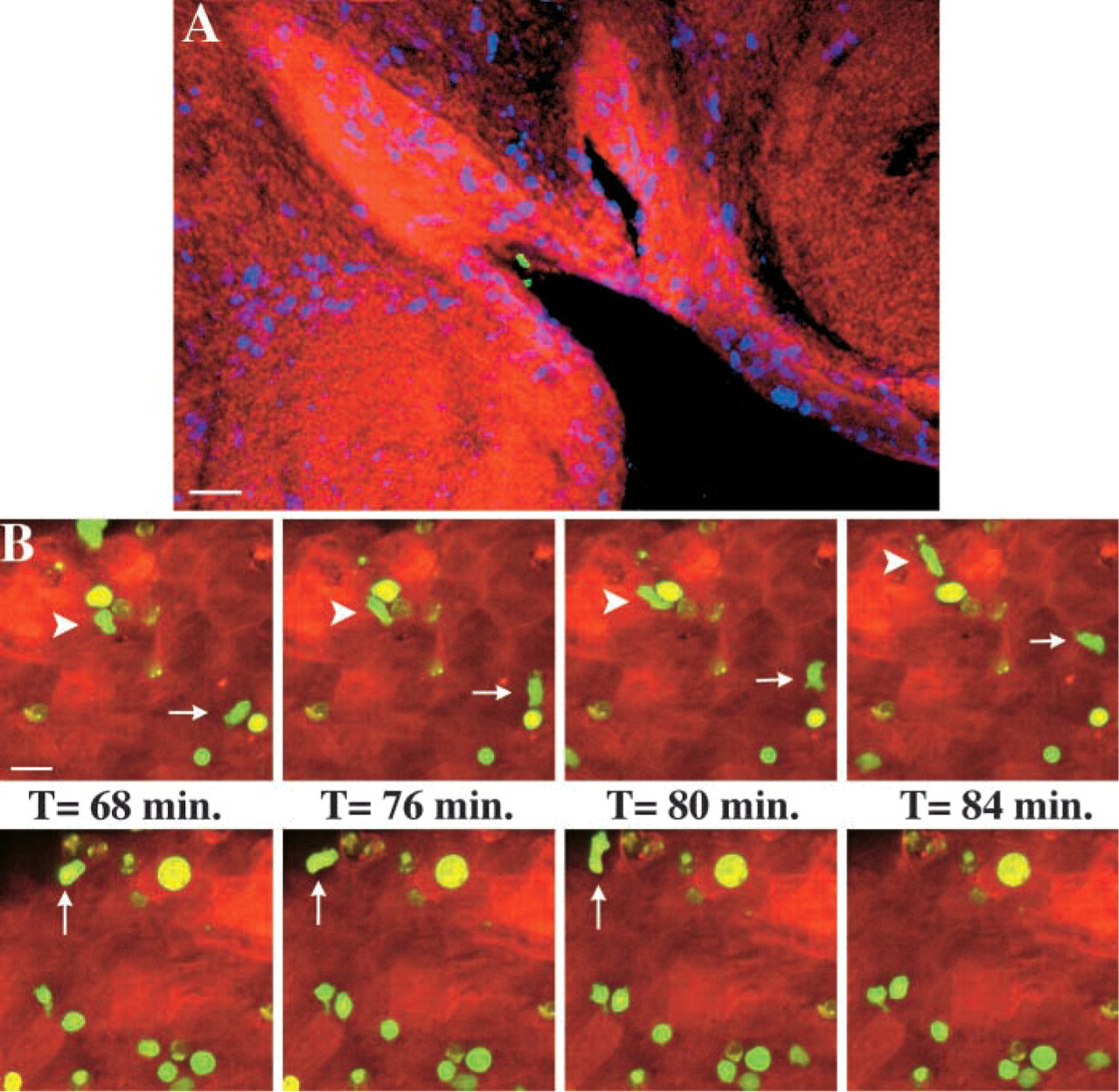
Interactions between HIV-infected lymphocytes and epithelial cells at the tonsil surface. (
On one level of thinking, much of the HIV transmission in the ongoing global HIV pandemic is readily understandable because virus is spread from person to person either by intimate sexual contact or by exposure to infected blood. In the case of mucosal exposure to HIV infectivity, there appears to be considerable protective benefit from an intact epithelial surface. Conversely, physical trauma or preexisting microbial infections can cause extensive change in the characteristics of mucosal surfaces and lead to elevated susceptibility to primary HIV infection. There is a clear association between STDs in the female reproductive tract and acquisition of primary HIV infection and similar connections between preexisting infections and HIV infection can be projected to occur in the oral cavity (Fiore et al. 1997b; Hillier 1998; Sturm-Ramirez et al. 2000). In this respect, it must be noted that the tonsil tissue used in these experiments cannot necessarily be considered as “normal.” Most of the tissues showed mild to moderate hyperplasia and had been removed from individuals with chronic tonsillitis or sleep apnea. In both clinical conditions, inflammation may have contributed significantly to tonsillar enlargement. The combination of alterations in the mucosal surface and activation and redistribution of tonsillar lymphocytes may dramatically enhance susceptibility to primary HIV infection, following oral exposure. Looking into the future, we anticipate that human mucosal organ culture systems will support a unique role in the evaluation of new protective strategies designed to curtail the spread of HIV and that the binding events and surface aberrations we have described will need to be addressed with the goal of achieving comprehensive protection against HIV.
Footnotes
Acknowledgements
These studies were supported in part by funds provided by the National Institute of Dental and Craniofacial Research (National Institutes of Health), Grants DE-12934 and DE-15090 and the Great Lakes Regional Center for AIDS Research.
We would like to thank our Microbiology colleagues, Dr. Ashley Haase, Dr. Hae-Sun Park, Dr. Kathryn Staskus, Dr. Pamela Skinner, and Stephen Wietgrefe, and Dr. Stefan Pambuccian in the Department of Surgical Pathology, University of Minnesota Medical School, for invaluable advice and insightful critiques. This work would not have been possible without the skillful assistance of Ms. Diane Rauch and Ms. Sarah Bowell in the Tissue Procurement Facility, Fairview University Medical Center (FUMC), Minneapolis, Minnesota, and Matthew Larson in the Department of Medicine (FUMC) who were collectively responsible for the primary interactions with the patients. We greatly appreciate the willingness of many anonymous patient donors who have provided samples for research purposes. We also acknowledge general technical assistance provided by Ms. Julie Horbul, SEM instruction and assistance provided by Chris Frethem, and essential contributions provided by the staff and resources within the Bioimaging and Processing Laboratory and the Supercomputing Institute at the University of Minnesota.
