Abstract
We investigated the localization of alkaline phosphatase (ALP) and three proteins related to intercellular junctions in the McA-RH 7777 rat hepatoma cell line to determine if the formation of junctions between adjacent McA-RH 7777 cells triggers translocation of ALP from cytoplasm to the plasma membrane. Contact between adjacent McA-RH 7777 cells promotes translocation of ALP from the Golgi area of the cytoplasm to the plasma membrane, and also promotes translocation of two proteins, E-cadherin and ZO-1, related to intercellular junctions, from cytoplasm to the plasma membrane. (
γ-G
Dexamethasone and dimethylsulfoxide in cultured fetal rat hepatocytes reportedly promote the differentiation of hepatocytes and translocation of GGT and ALP from cytoplasm to the plasma membrane (Chida 1998). This indicates that translocation of GGT and ALP in hepatocytes is related to development of hepatocyte polarity. Polarity of the plasma membrane in epithelial cells develops as a result of intercellular junctional complexes between adjacent cells (Wodarz 2002). In normal adult rat hepatocytes, ALP is predominantly localized in the bile canalicular domain of the plasma membrane and is considered to hydrolyze phosphate compounds as phospholipids excreted into the bile canalicular lumen. Moreover, the formation of intercellular junctions between adjacent hepatocytes divides the plasma membrane to two domains, apical and basolateral, and produces bile canaliculi as apical spaces. The formation of intercellular junctions between adjacent rat hepatocytes is therefore expected to be closely related to localization of ALP and necessary for ALP function. The present study investigated localization of ALP and three proteins related to intercellular junctions, E-cadherin (adherens junctions), ZO-1 (tight junctions), and connexin-32 (gap junctions) in McA-RH 7777 cells to examine whether the formation of intercellular junctions is related to translocation of ALP from cytoplasm to the plasma membrane after contact between adjacent McA-RH 7777 cells.
McA-RH 7777 cells (American Type Culture Collection; Rockville, MD) derived from Morris rat hepatoma were cultured in α-minimum essential medium containing 10% fetal bovine serum (α-MEM 13.1; Dainippon Phamaceutical, Osaka, Japan). McA-RH 7777 cells were seeded at a concentration of 2 × 104 cells/ml in eight-well collagen-coated BioCoat Culture Slides (Nippon Becton Dickinson; Tokyo, Japan) and cultured for 3 days. Other McA-RH 7777 cells were seeded at a concentration of 105 cells/ml in identical slides and synchronized at S- phase of the cell cycle by culturing in medium containing excess thymidine and hydroxyurea, as described previously (Chida 2000). Synchronized cells were then cultured in α-MEM for 4–8 hr.
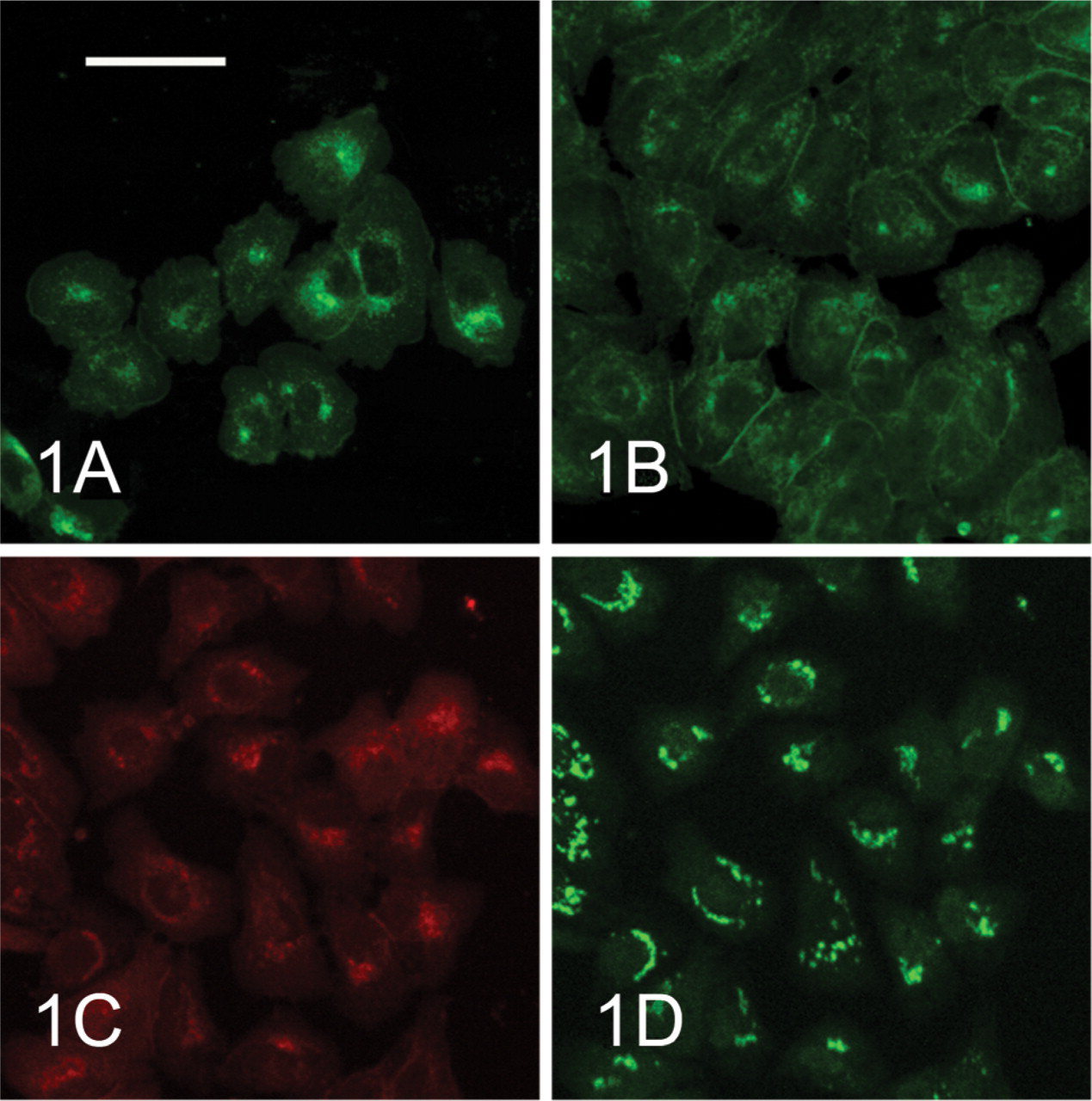
(
McA-RH 7777 cells were fixed in Zamboni solution (2% paraformaldehyde, 15% saturated picric acid in 0.1 M phosphate buffer, pH 7.4) for 10 min at RT. After washing with 0.01 M phosphate-buffered saline solution (PBSS, pH 7.2) containing 0.85% NaCl and 0.05% saponin, cells were immersed in 0.1% Triton X-100 solution dissolved in PBSS for 5 min at RT. Cells were then incubated for 1 hr at RT in anti-rat ALP anti-serum (Chida 1993a, b) diluted 50-fold with PBSS, anti-human E-cadherin monoclonal antibodies (BD Bioscience Pharmingen; San Diego, CA) diluted 500-fold with PBSS, anti-mouse ZO-1 monoclonal antibodies (Chemicon International; Temecula, CA) diluted 1000-fold with PBSS, or anti-rat connexin 32 monoclonal antibodies (Zymed Laboratories; South San Francisco, CA) diluted 250-fold with PBSS. For control, normal rabbit serum or mouse IgG was used in place of antiserum or antibodies in the same solutions. Cells were washed with PBSS and reacted for 30 min at RT with fluorescein isothiocyanate (FITC)-labeled anti-rabbit or anti-mouse IgG antibodies (Medical and Biological Laboratories; Nagoya, Japan). Double staining for ALP and mannosidase II, a Golgi marker, was performed in some cells cultured at low concentration. For double staining, cells were incubated for 1 hr at RT in mixed solution containing anti-rat ALP antiserum diluted 50-fold with PBSS and anti-rat mannosidase II monoclonal antibody (Covance; Research Products, Richmond, CA) diluted 10,000-fold with PBSS. After washing with PBSS, cells were reacted for 30 min at RT with mixed solution containing rhodamine-labeled anti-rabbit and FITC-labeled anti-mouse IgG antibodies (Medical and Biological Laboratories). All cells were mounted in Fluoro-Guard Antifade Reagent (Bio-Rad; Hemel Hempstead, UK) and examined under confocal laser scanning microscopy (Radiance2100; Bio-Rad).
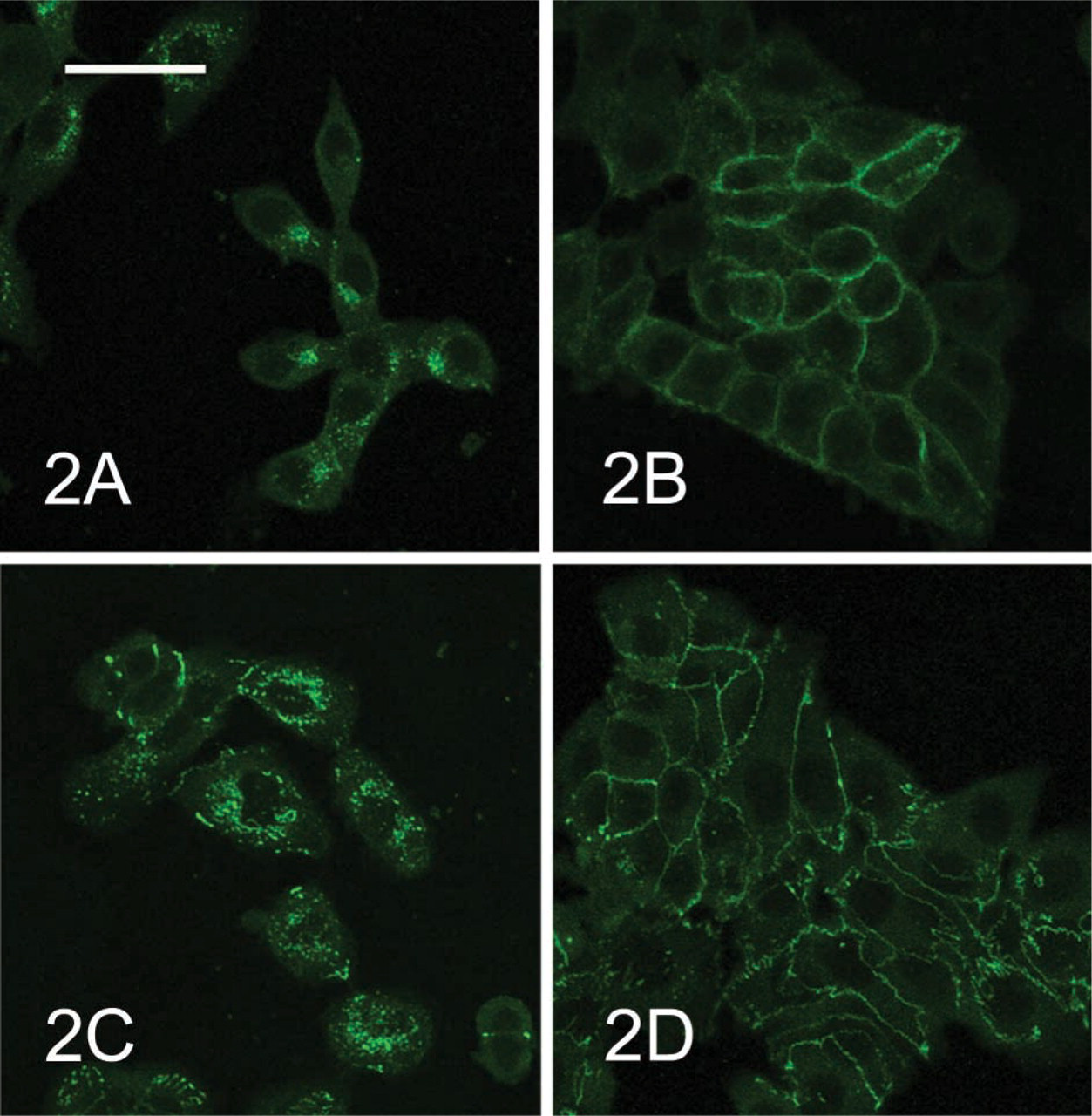
(
In McA-RH 7777 cells cultured at low concentration, ALP was restricted to cytoplasm around the nucleus (Figure 1A). Moreover, double staining for ALP and mannosidase II indicated locations of ALP in cytoplasm basically consistent with those of mannosidase II (Figures 1C and 1D). During the S–G2 stages of the cell cycle in synchronous McA-RH 7777 cells, ALP was localized at the borders between adjacent cells and in cytoplasm around the nucleus (Figure 1B).
E-cadherin and ZO-1 in McA-RH 7777 cells cultured at low concentration were also restricted to cytoplasm around the nucleus (Figures 2A and 2C). E-cadherin and ZO-1 during the S–G2 stages of the cell cycle in synchronous McA-RH 7777 cells were localized at the borders between adjacent McA-RH 7777 cells (Figures 2B and 2D). However, connexin-32 was not detected in McA-RH 7777 cells cultured under either of the above conditions.
We previously reported that ALP and GGT in McA-RH 7777 cells are localized at the same sites as Golgi markers, Golgi 58K protein and a substance that reacts to wheat germ agglutinin (WGA) (Chida 2000). The present study demonstrated, by double staining of ALP and mannosidase II, a Golgi marker, that ALP in McA-RH 7777 cells cultured at low concentrations is localized in the Golgi areas of the cytoplasm. This means that ALP in McA-RH 7777 cells is synthesized in the rough endoplasmic reticulum around the nucleus and then accumulates in Golgi complexes, and is transferred to the plasma membrane by the trigger of contact between adjacent cells. Furthermore, this study showed that E-cadherin and ZO-1 are also translocated from cytoplasm to the plasma membrane after contact between adjacent McA-RH 7777 cells. This indicates that contact between McA-RH 7777 cells promotes the formation of intercellular junctions similar to adherens junctions at the cell borders. However, the present study cannot draw any conclusions regarding the formation of tight junctions at cell borders between adjacent McA-RH 7777 cells, because ZO-1 is a cytoplasmic protein that links with transmembrane proteins in tight junctions and also interacts with E-cadherin (Watabe et al. 1994) and several signaling proteins. During the S–G2 stages of the cell cycle in synchronous McA-RH 7777 cells in the present study, E-cadherin and ZO-1 were localized only in the plasma membrane between adjacent cells, whereas ALP displayed dual localization in the cytoplasmic and plasma membrane. Accordingly, E-cadherin and ZO-1 may be translocated to cell surfaces through these passages in different ways from that of ALP. The present study cannot therefore conclude whether ALP translocation is associated with formation of intercellular junctions similar to adherens junctions. A previous study detected localization of connexin-32 at cell borders between adjacent cells in fetal rat hepatocytes cultured in dexamethasone-supplemented medium (Chida 1998). However, connexin-32 was not detected in the present study despite the use of the same methods as in that previous study. This suggests that McA-RH 7777 cells do not express connexin-32. Gap-junctional intercellular communications reportedly decrease during hepatocarcinogenesis in rats (Tsuda et al. 1995). The present observations may therefore suggest that the formation of connexin-32-mediated gap junctions may not be related to translocation of ALP from the Golgi area of cytoplasm to the plasma membrane.
In mouse or rat livers, colchicine administration or bile duct ligation leads to morphological changes of the junctional complex of hepatocytes and to partial breakdown of the permeability barrier between the canalicular lumen and the intercellular space between adjacent hepatocytes (Metz et al. 1977; Rassat et al. 1982). ALP in rat liver becomes localized not only in the bile canalicular membrane but also in the basolateral membrane by the above treatment (Araki et al. 1995; Chida et al. 1995). These facts indicate that translocalization of ALP in the plasma membrane of rat hepatocytes is closely related to the formation of intercellular junctions between adjacent hepatocytes. However, at present we cannot assert whether the formation of adherens junctions between adjacent McA-RH 7777 cells triggers translocation of ALP from Golgi complexes to the plasma membrane. Moreover, tyrosine phosphorylation of junctional proteins reportedly disrupts the junctional complex (Rao et al. 2002). However, whether ALP functions as a phosphatase enzyme to protect intercellular junctions from such tyrosine phosphorylation of junctional proteins is unknown.
