Abstract
Cell-cell adhesion is crucial not only for mechanical adhesion but also for tissue morphogenesis. Protein 4.1B, a member of the protein 4.1 family named from an erythrocyte membrane protein, is a potential organizer of an adherens system. In adult mouse seminiferous tubules, protein 4.1B localized in the basal compartment, especially in the attaching region of spermatogonia and Sertoli cells. Protein 4.1B localization and appearance were not different in each spermatogenic stage. Developmentally, protein 4.1B was not detected at postnatal day 3 (P3), was diffusely localized at P15, and was found in the basal compartment during the third week. By double staining for protein 4.1B and F-actin, their localizations were shown to be different, indicating that protein 4.1B was localized in a region lower than the basal ectoplasmic specialization that formed the Sertoli–Sertoli junction. By electron microscopy, immunoreactive products were seen mainly on the membranes of Sertoli cells. In the W/Wv mutant mouse, the seminiferous epithelium had few germ cells. Protein 4.1B and β-catenin were not detected, although the basal ectoplasmic specialization was retained. These results indicate that protein 4.1B may be related to the adhesion between Sertoli cells and germ cells, especially the spermatogonium.
S
Protein 4.1B mRNA and Western blotting have also shown expression of protein 4.1B in the testis (Yamakawa and Ohara 2000). The parenchyma of the testis consists of highly convoluted seminiferous tubules. The seminiferous epithelium is a highly specialized, complex epithelium lining the seminiferous tubules, consisting of spermatogenic cells and supporting Sertoli cells (Krstic 1991). Spermatogenic cells go through several cycles of development, including meiotic division and maturation. Spermatogenic stem cells, the spermatogonia, and Sertoli cells lie on the basal lamina. One spermatogonium divides and gives rise to two primary spermatocytes. Immediately after the division, the primary spermatocytes are separated from the spermatogonia by processes of Sertoli cells that run parallel to the basal lamina. These processes divide the seminiferous epithelium into two compartments a basal compartment containing spermatogonia and an adluminal compartment containing the remaining spermatogenic cells. All spermatogenic cells are believed to be surrounded by Sertoli cells, and Sertoli cells are also attached to each other by tight junctions. Therefore, it is of interest to clarify what type of cell or compartment in the testis expresses and localizes protein 4.1B. Here we used a specific protein 4.1B antibody to investigate the localization of protein 4.1B. We found that it was localized in mouse Sertoli cells attaching to the spermatogonia at the basal compartment. It is known that a signal protein relating to adhesion, β-catenin, is expressed in the basal compartment (Lee et al. 2003). In this study, protein 4.1B and β-catenin localizations in W/Wv mutant mice, which have few spermatogenic cells, were also examined, and a difference from that in wild-type mice was found.
Materials and Methods
Antibodies
A rabbit anti-rat protein 4.1B polyclonal antibody was raised against a specific domain of the protein that does not react with other protein 4.1 family members, and affinity-purified as described previously (Yamakawa and Ohara 2000). The anti-protein 4.1B antibody recognizes mouse and rat protein 4.1B. The rabbit polyclonal anti-human β-catenin antibody (Upstate #06–734; Lake Placid, NY) recognized rodent and human β-catenin for immunoblotting and IHC.
Immunoprecipitation and Western Blotting
For normal testis tissues, adult C57BL/6 mice were sacrificed and the testes were rapidly removed. Testes from two mice were lysed with 5 ml lysis buffer containing 1% Triton X-100, 137 mM NaCl, 2 mM EDTA, 20 mM Tris (pH 7.5), and a proteinase inhibitor cocktail (Sigma; St Louis, MO). After 1 hr at 4C, insoluble materials was removed by centrifugation at 2000 × g (20 min, 4C), and the supernatant was cleared by addition of 100 μl protein G-Sepharose (Amersham Pharmacia Biotech; Piscataway, NJ) for 2 hr, followed by centrifugation at 300 × g for 2 min. For immunoprecipitation, 5 μl anti-protein 4.1B antibody was added at 4C overnight; in control experiments no testis sample was added. Protein complexes were precipitated by incubation with 50 μl protein G-Sepharose for 2 hr. Immune complexes were collected by centrifugation at 300 × g, washed five times in lysis buffer, and boiled with Laemmli sample buffer (Bio-Rad; Hercules, CA) and 5% 2-mercaptoethanol (Sigma) for 5 min. Then the prepared samples were loaded on 4–20% gradient SDS-polyacrylamide gel for electrophoresis, and subsequently electrotransferred onto a polyvinylidene fluoride membrane (Millipore; Bedford, MA). After blocking with 2% bovine serum albumin (Sigma) in PBS containing 0.1% Tween-20 (PBS-T) for 2 hr, the electrotransferred membrane was incubated with anti-protein 4.1B antibody at 1:3000 dilution at 4C overnight. After washing with PBS-T, the membrane was incubated with HRP-conjugated goat anti-rabbit IgG antibody (Vector Laboratories; Burlingame, CA) at room temperature for 1 hr. After washing with PBS-T, the labeling reaction was visualized by using an enhanced chemiluminescent substrate (Pierce; Rockford, IL).
Animals and Tissue Preparations for IHC
All animals used in this study were handled in compliance with the University of Yamanashi Guidelines for the Use of Animals. W/Wv mice were obtained commercially (Japan SLC; Hamamatsu, Japan). Adult (2–3 month old) postnatal day 3 (P3), P14, P25 C57BL/6 mice, and adult W/Wv mice were anesthetized with pentobarbital and perfused via the heart with 4% paraformaldehyde in 0.1 M phosphate buffer (PB), pH 7.4. The testes were removed and immersed in the same fixative at 4C for 1–2 hr. After rinsing in PBS, they were immersed in 30% sucrose at 4C overnight, then embedded in an OCT compound and sectioned at 4–6-μm thickness in a cryostat for IHC.
Immunofluorescence Microscopy
For IHC analysis by fluorescence microscopy, anti-protein 4.1B (1:500) or anti-β-catenin (1:1000) antibodies were used. Cryosections were first treated with 0.1% Triton X-100 in PBS and 10% goat serum for 30 min each. They were incubated with the primary antibodies at 4C overnight and then with FITC-conjugated anti-rabbit IgG antibodies (Molecular Probes; Eugene, OR) at RT for 1 hr. For the double-labeling study for protein 4.1B and F-actin, sections were immunoreacted with a rabbit anti-protein 4.1B antibody and then with TRITC-conjugated anti-rabbit IgG antibody and FITC-conjugated phalloidin (Molecular Probes). Immunostained sections were analyzed with a fluorescence microscope (BX-61; Olympus, Tokyo, Japan) or with confocal laser scanning microscopy (TCS 4D; Leica, Heidelberg, Germany). For the peptide blocking examination, the anti-protein 4.1B antibody was mixed with the original peptide antigen for making the antibody (20-fold molar ratio of the antigen against the antibody), and this was used as a substitute for the primary antibody.
Immunoelectron Microscopy
A conventional preembedding immunotechnique with peroxidase was used for immunoelectron microscopy, as described previously (Terada et al. 2003). Briefly, 6-μm-thick cryosections, which had been immunostained with the primary anti-protein 4.1B antibody, were incubated with goat anti-rabbit IgG antibody conjugated to biotin and then with horseradish peroxidase (HRP)-conjugated streptavidin (Vector Laboratories). After rinsing in PBS, they were fixed again with 0.25% glutaraldehyde in PB for 10 min. Immunoreactivity was visualized by a metal-enhanced DAB method (Pierce). Sections were treated with 1% osmium tetroxide in PB for 30 min and, after dehydration in a graded series of ethanol and acetone, they were embedded in epoxy resin by the inverted gelatin capsule method. Ultrathin sections at 70-nm thickness were cut on an ultramicrotome (Ultracut E; Reichert, Germany) and collected on copper grids. Sections were stained with uranyl acetate to identify the localization of DAB reaction products and analyzed at an accelerating voltage of 80 kV in an electron microscope (H-7500; Hitachi, Osaka, Japan).
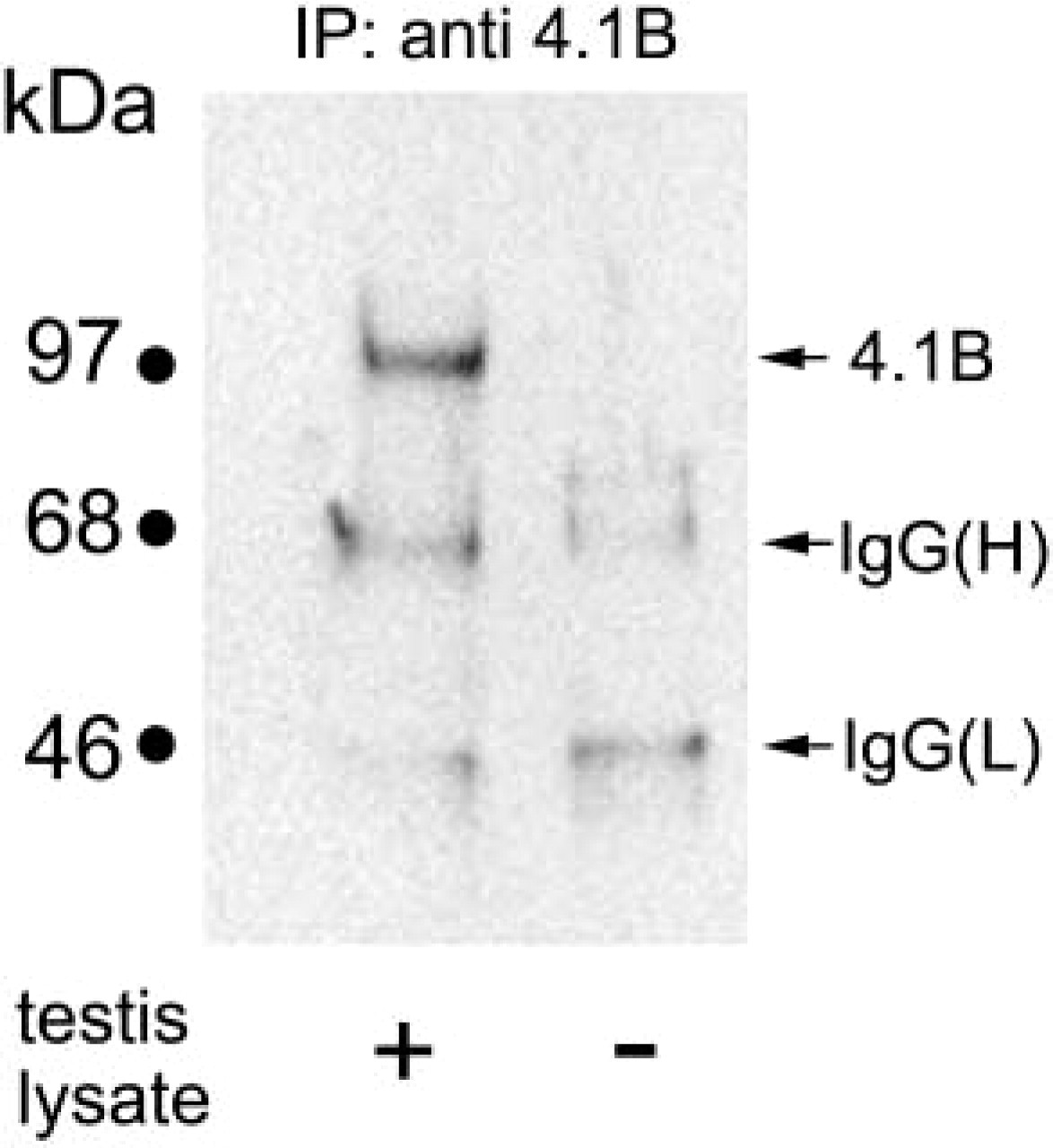
Specificity of protein 4.1B (4.1B) antibody to testis. Adult mouse testis tissues were lysed with Triton X-100, immunoprecipitated with 4.1B antibody, and Western-blotted using the same antibody. A 110-kD band is recognized with the 4.1B antibody (left lane). This band does not appear for the same amount of 4.1B antibody without testis lysate (right lane). The 65-kD and 30-kD bands are believed to be IgG heavy chains [IgG(H)] and light chains [IgG(L)], respectively.

Immunolocalization of protein 4.1B in the basal compartment of the mouse seminiferous epithelium. Paired DIC (
Results
Distribution of Protein 4.1B in Testis
The specificity of the anti-protein 4.1B antibody was examined in the mouse testis. Because Western blots of the mouse testis probed with anti-protein 4.1B showed little signal, we performed immunoprecipitation to concentrate the reactive proteins with the antibody. A single band with apparent molecular weight 110 kD was detected in the sample of immunoprecipitated testis tissues with the anti-protein 4.1B antibody (Figure 1, left lane) but not in the sample of the antibody itself (Figure 1, right lane) or in the sample of testis attached to protein G–Sepharose beads without the antibody (data not shown).
Protein 4.1B detected with our antibody was present in adult mouse seminiferous tubules (Figure 2). There was no immunoreactivity in the ductus epididymidis or in the ductuli efferentes (data not shown). In the seminiferous tubules, the protein 4.1B-specific staining was restricted to the basal compartment of the seminiferous epithelium containing spermatogonia attached to Sertoli cells (arrowheads in Figures 2a–2c). The staining pattern was arch-like on the basement membranes, and each spermatogonium nucleus was below the arc structure. In the adluminal compartment, in which spermatocytes, spermatids, and spermatozoa were differentiating along Sertoli cells, protein 4.1B immunoreactivity was faint compared with that in the basal compartment. The emergence and disappearance of the protein 4.1B staining were not different in the seminiferous epithelial cycle [e.g., stage X (after spermiation) shown in Figures 2d–2e and stage V (apical spermatid accumulation) shown in Figures 1f and 1g (Oakberg 1956)]. This immunoreactivity was eliminated by the original peptide that was used as an antigen for making the antibody (Figures 2h and 2i). This suggests that protein 4.1B is consistently localized in the basal compartment of the mouse seminiferous epithelium.
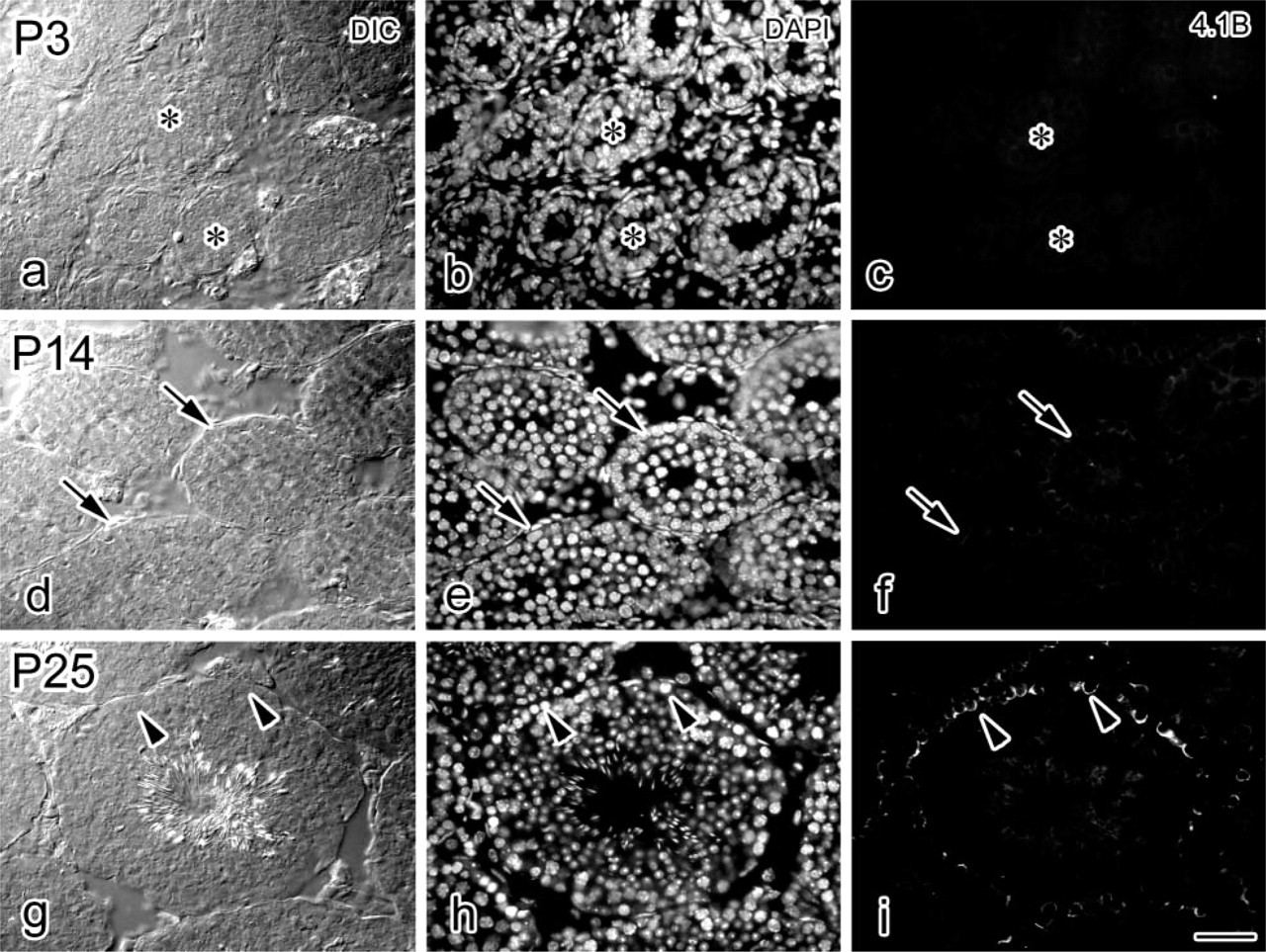
Developmental expression and localization of protein 4.1B in the mouse seminiferous epithelium. Paired DIC, DAPI, and protein 4.1B images at P3 (
Protein 4.1B Appearance and Localization During Development
We next extended the IHC analysis of protein 4.1B expression to examine testes at early developmental stages (Figure 3). Protein 4.1B was not detected in P3 testis (Figures 3a–3c); weak protein 4.1B expression was detected as short-distance lines or dots in the cells in P14 testis (Figures 3d–3f). The expression became much more pronounced in P25 testis, and the localization was in the basal compartment as described above (Figures 3g and 3i). The blood–testis barrier was reported to start to form around P15 (Cyr et al. 1999), suggesting that protein 4.1B expression and localization may be related to the maturation of the seminiferous epithelium.
Protein 4.1B Localization and the Basal Ectoplasmic Specialization
To determine the localization of protein 4.1B in the seminiferous epithelium, confocal microscopy of double-stained tissue with protein 4.1B (Figure 4a) and phalloidin (Figure 4b) was performed. The distribution of actin filaments, as determined by fluorescent phalloidin staining, was used as a marker for basal and apical junction sites. Actin was concentrated both at the apical Sertoli–germ cell ectoplasmic specializations (ESs) (S/Gj in Figure 4b) and the basal Sertoli–Sertoli cell ESs (S/Sj in Figure 4b). The protein 4.1B staining was partially overlapped in the basal S/Sj at the region of the top of the arced shape. At the region of the bottom of the arced shape by protein 4.1B staining, phalloidin staining did not overlap. This indicates that protein 4.1B is probably not a component of ESs.
Ultrastructurally, Protein 4.1B Localized Under the Plasma Membranes of Sertoli Cells
To determine the cellular localization of protein 4.1B in the basal compartment (BC in Figure 5 inset) more precisely, IHC by the pre-embedding method with a DAB reaction was performed (Figure 5). All spermatogenic cells were surrounded by Sertoli cells extending from the basal lamina to the lumen of a seminiferous tubule. The basal side of Sertoli cells (SC in Figure 5) was in contact with spermatogonia and spermatogenic stem cells (Sg in Figure 5). DAB reaction products were observed mainly in the Sertoli cells adjacent to the spermatogonia (arrows in Figure 5). In some regions, Sertoli cells were detached from the spermatogonia, probably because of cell shrinkage due to chemical fixation and alcohol dehydration. In such regions, it was clear that DAB reaction products were under the plasma membranes of Sertoli cells. This suggests that protein 4.1B localizes on the Sertoli cell membranes, adhering to the spermatogonia.
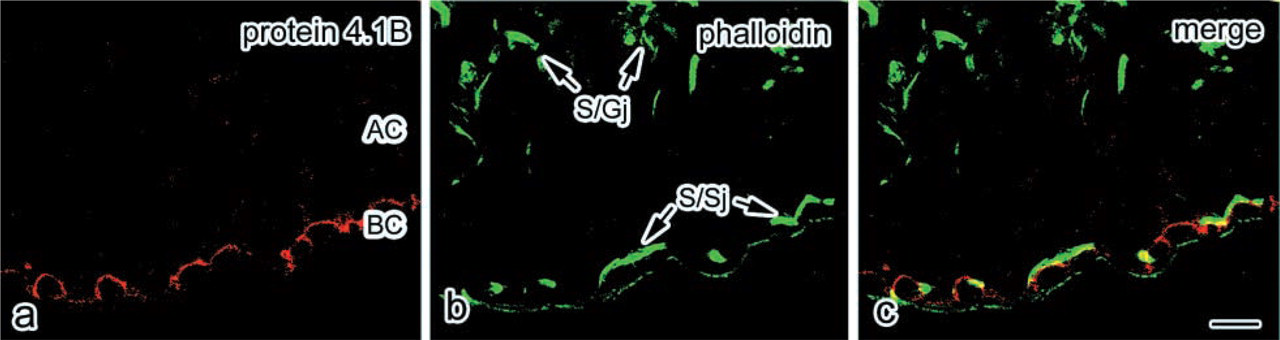
Localization of protein 4.1B and F-actin in the mouse seminiferous epithelium. Confocal laser micrographs of a cross-section from a seminiferous epithelium in a normal adult mouse. The section was stained for protein 4.1B with TRITC (
Protein 4.1B Expression and Localization in a W/Wv Mouse Model
Germ cell-depleted W/Wv mutant mice, which contained only Sertoli cells in their seminiferous tubules (Ogawa 2001), provide a good model to exclude the role of germ cells. Hence, to understand whether germ cells were required to mediate protein 4.1B expression and location, IHC for protein 4.1B in W/Wv mice was performed. In W/Wv mice, the basal ectoplasmic specialization (i.e., Sertoli–Sertoli cell junction) was reported to retain its structure (Kurohmaru et al. 1992). The phalloidin staining (Figure 6f) confirmed this concept, although stained longitudinal lines in W/Wv mice were longer than those in the wild-type mice (Figure 6c). However, protein 4.1B immunostaining in the W/Wv mutant mice (Figure 6l) was absent relative to controls (Figure 6i). This indicated that the immunostaining of basal ectoplasmic specialization was different from the immunostaining pattern of protein 4.1B. β-Catenin is a protein reported to be a factor in the adherens junction of the seminiferous epithelium, especially localizing in the basal compartment (Lee et al. 2003). Like protein 4.1B, the β-catenin immunostaining in the W/Wv mutant mice (Figure 6r) was also absent relative to controls (Figure 6o). These results suggest that protein 4.1B may function in Sertoli-germ cell adhesion in a complex that also includes β-catenin.
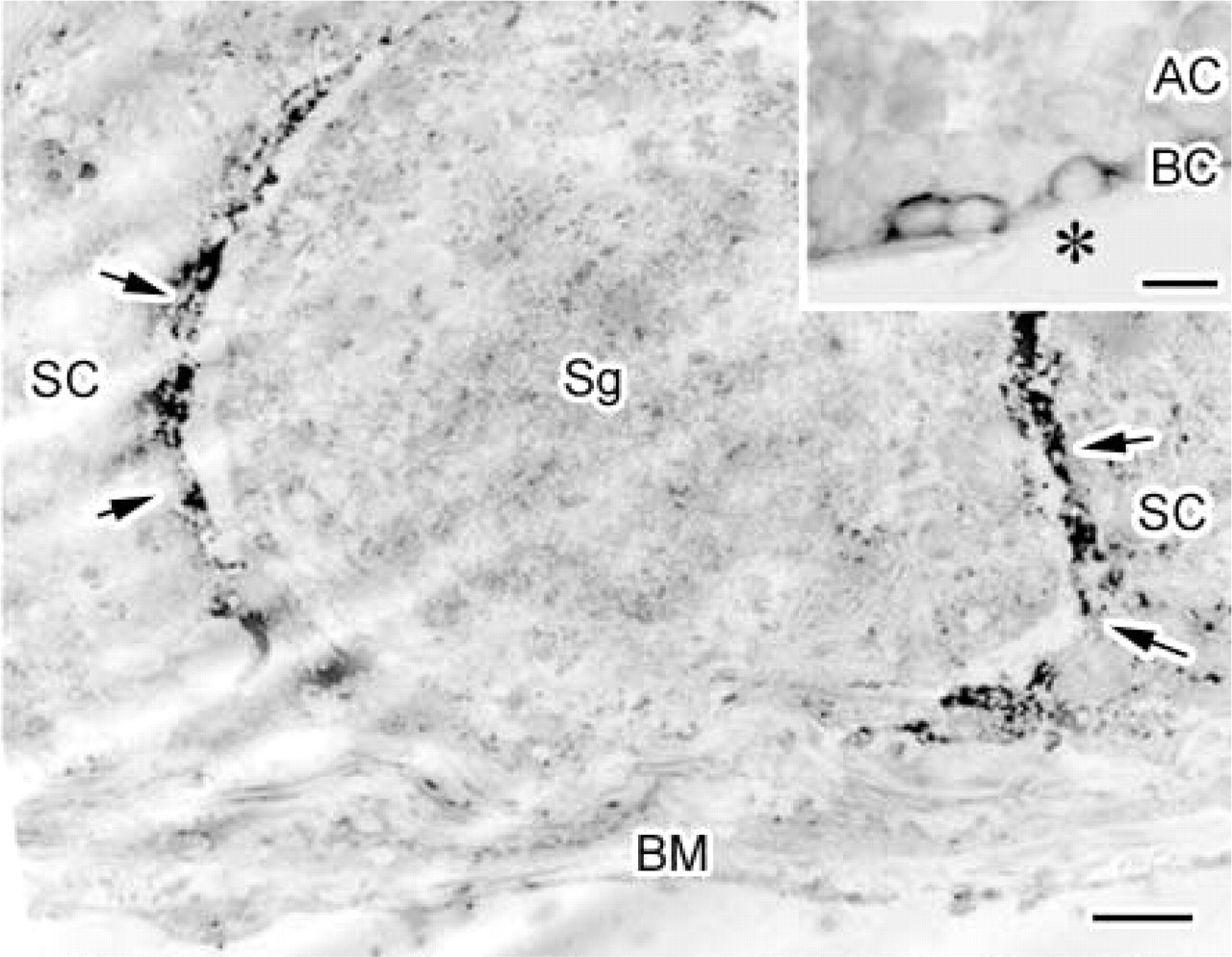
Protein 4.1B on plasma membranes of Sertoli cells facing the spermatogonium. Immunoelectron microscopic localization of protein 4.1B in the mouse seminiferous epithelium with an anti-protein 4.1B antibody, an HRP-conjugated secondary antibody, and subsequent DAB staining. (
Discussion
In this study we demonstrated the distribution of protein 4.1B in the regions related to Sertoli cell–spermatogonium adhesion in the mouse seminiferous epithelium, as shown in Figure 7 (the left side of the figure shows the wild-type). Protein 4.1B was shown to be related to cell–cell adhesion in paranodes (axon–oligodendrocyte contact), islet endocrine cells, small and large intestines, and renal proximal tubules. Consistent with these observations, our results support the idea that protein 4.1B is related to cell–cell adhesion.
In the seminiferous epithelium, tight junctions between Sertoli cells form a functional blood–testis barrier between 10 and 19 days of age. Several molecules relating to the tight junction in the seminiferous epithelium have been identified. Occludin was reported to appear in band-like localization at the base of the tubules at P14 (Cyr et al. 1999). The zonula occludens (ZO)-1 concentrates in the tight junction regions of barriers (Byers et al. 1991). In addition, claudin-11/Osp (oligodendrocyte-specific protein) was found to be necessary for spermatogenesis by generating a paracellular physical barrier of tight junctions (Gow et al. 1999). The tight junction between Sertoli cells, together with a subsurface cistern of the endoplasmic reticulum and a layer of closely packed actin filaments that is sandwiched between the plasma membrane and the subsurface cistern, have collectively been defined as the basal ectoplasmic specialization (Toyama et al. 2003). As reported previously (Kurohmaru et al. 1992), F-actin was recognized in the ectoplasmic specialization of the W/Wv Sertoli cell and appeared similar to that in normal mice in this study. In addition to F-actin, one of the intermediate filaments, vimentin, in the W/Wv mouse Sertoli cells was already reported to distribute around the nucleus and extended toward the tubular lumen, similar to normal mice (Kurohmaru et al. 1992), indicating the subcellular distribution of F-actin and intermediate filaments in W/Wv mouse. Sertoli cells were not thought to be strikingly affected by lack of spermatogenic cells. We initially assumed that cytoskeletal proteins, including protein 4.1B, might maintain Sertoli cell stability, even without germ cells. However, after we examined the W/Wv mutant mouse seminiferous epithelium, the protein 4.1B immunoreactivity was found to be absent relative to controls, as shown in Figure 6. Therefore, protein 4.1B may have a role in adhesion between Sertoli cells and germ cells, but not for the Sertoli–Sertoli junction (i.e., tight junction), as shown in Figure 7a. The result of the confocal microscopic study (Figure 4) also supported this idea.
The cadherin/catenin complex and its intracellular attachment site in the testis, considered to be the functional unit for actin-based cell–cell adhesion junctions, has been the subject of controversy with regard to cell-cell adhesion in the basal compartment of the seminiferous epithelium (Cheng and Mruk 2002; Lee et al. 2003; Lui et al. 2003). It was shown that N-cadherin co-localized with β-catenin in the seminiferous epithelium near the basal and lower third of the adluminal compartment (Lee et al. 2003). Another previous study found that pan-cadherin and β-catenin antibodies did not react with the ectoplasmic specialization but rather with desmosome-like junctions that are present at basal junctional complexes between Sertoli cells and at sites of attachment to spermatogenic cells (Mulholland et al. 2001). In this study, in addition to protein 4.1B, we also examined the immunostaining of β-catenin in W/Wv mice for the first time and found it absent relative to controls, as shown in Figure 6r. From these results, in the mouse seminiferous tubule, one of the protein 4.1B functions may be to offer the Sertoli cell-spermatogonia adhesion in relation to β-catenin. The biochemical interaction between protein 4.1B and β-catenin in testis is under investigation. No intramembranous molecule that related to protein 4.1B has been identified in the testis. In paranodes, Caspr-1 and Caspr-2 were reported to interact with protein 4.1B, as band 3 and protein 4.1R in erythrocytes (Gollan et al. 2002; Denisenko-Nehrbass et al. 2003). The structural and functional importance of adherens junctions in the seminiferous epithelium in relation to the cadherin/catenin system has been described above. p120 catenin localization in the basal compartment was also recently reported (Golenhofen and Drenckhahn 2000; Johnson and Boekelheide 2002). Another adhesion system, the nectin/afadin system (Ozaki-Kuroda et al. 2002), and the integrin system (Mulholland et al. 2001) were found in relation to Sertoli–germ cell and Sertoli–Sertoli cell adhesion, although the lack of protein 4.1B immunostaining at the ectoplasmic specialization indicates that relationships to them are unlikely. The relationship of protein 4.1B with these four systems (Caspr, cadherin/catenin, nectin/afadin, and integrin) is an open question. Although maintenance of the spermatogonia in the basal compartment of the seminiferous epithelium is one of the important and interesting biological phenomena during spermatogenesis, very few studies have been performed to examine the participating molecules and the mechanism by which this event is regulated. On the basis of our IHC study, we propose a model of the Sertoli–spermatogonia relationship in mouse seminiferous tubules by the protein 4.1B-including membrane skeletons, as shown in Figure 7c. Further examinations of protein 4.1B localization and function may expand our knowledge of spermatogenesis pertinent to junction structuring in the seminiferous epithelium.
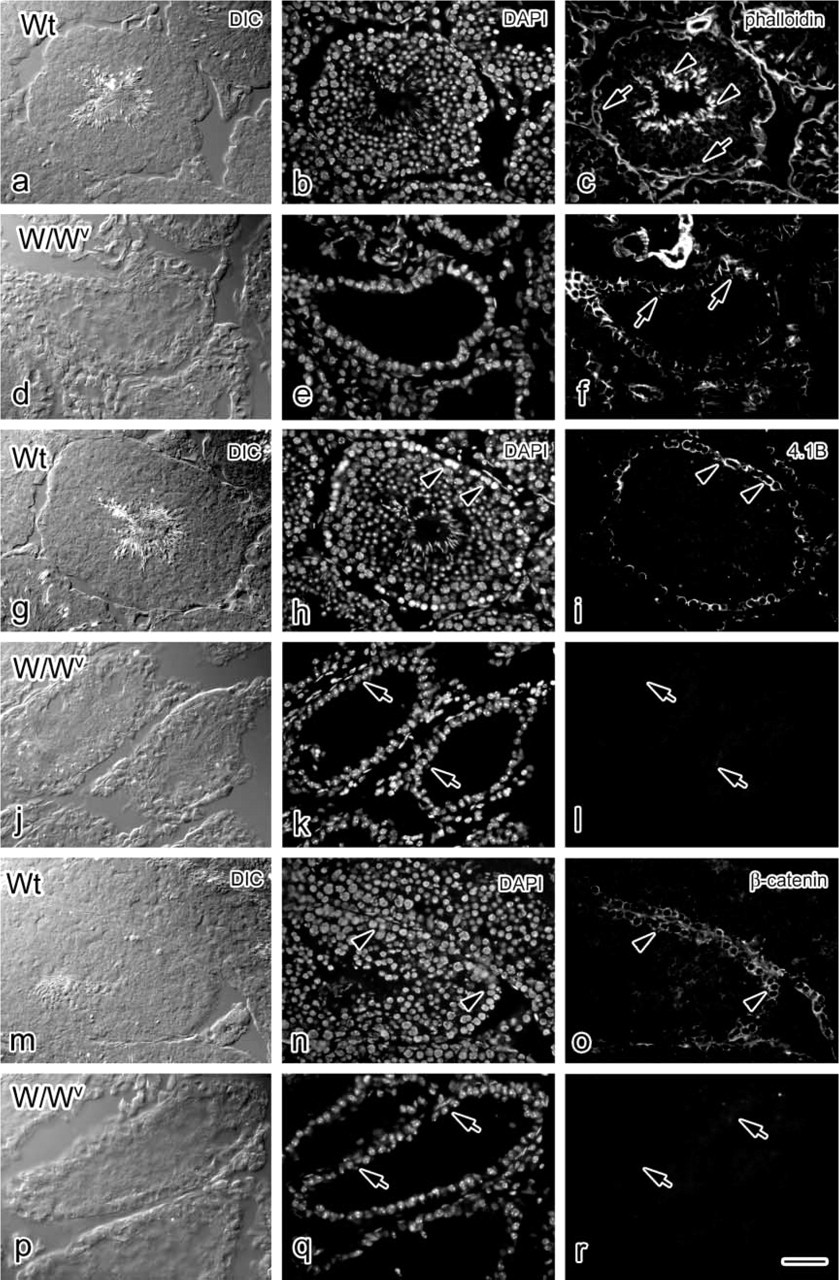
Changes in protein 4.1B and β-catenin IHC reactivity in the W/W mutant mouse seminiferous epithelium. Paired DIC, DAPI, and fluorescence micrographs showing localization of F-actin (
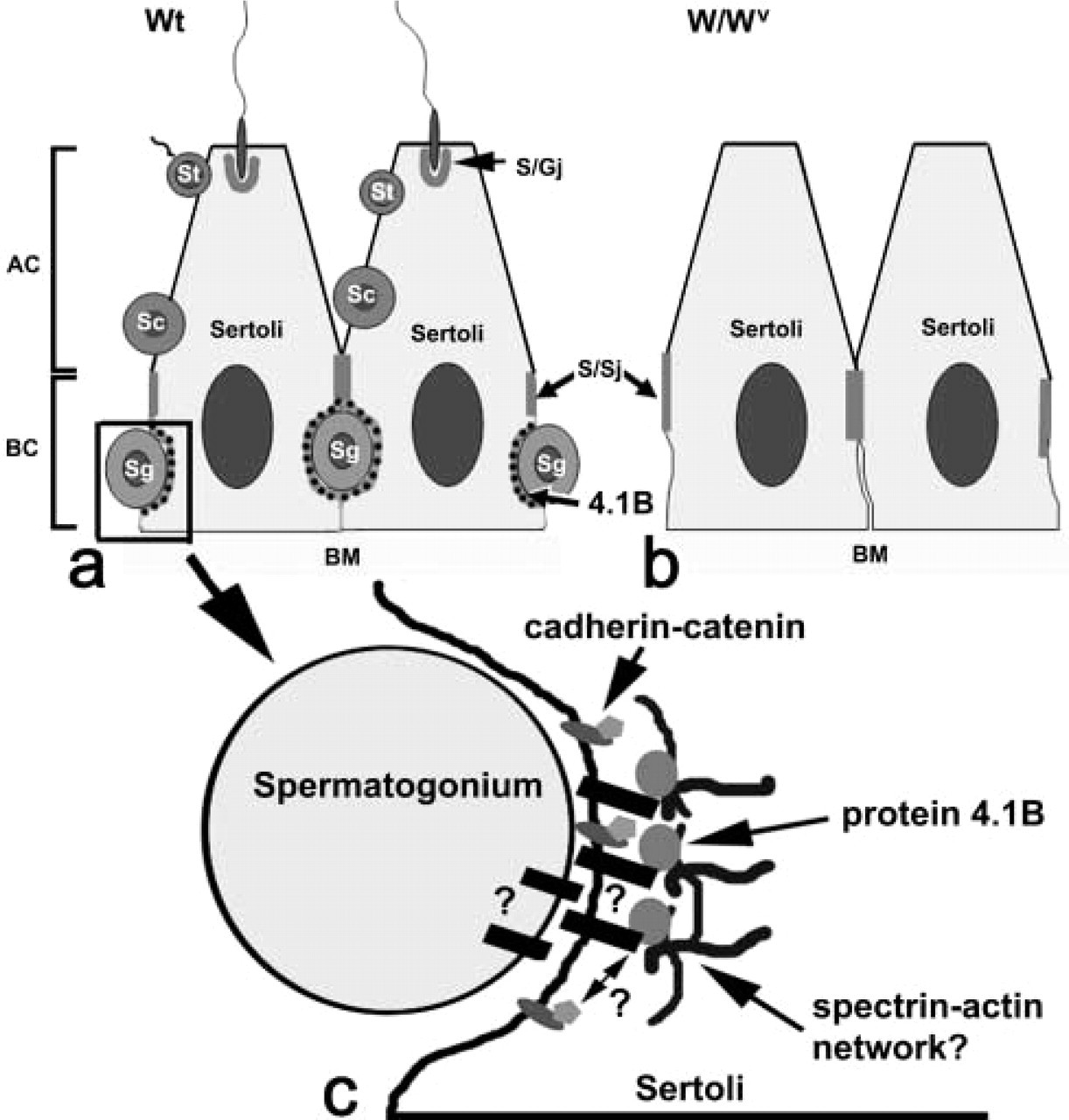
Spermatogenesis in mouse testis and the localization of protein 4.1B. In normal (
