Abstract
Idiopathic varicocele is closely associated with male infertility or subfertility. Sertoli cell is a very important regulator of spermatogenesis. We investigated the morphofunctional alterations in the Sertoli cell and its possible involvement in the establishment of testicular primary lesion in experimental left-sided varicocele, induced from peripuberty. Twenty-five male peripubertal rats (44 days postpartum [dpp]) were distributed into two groups: control (C) and varicocele (V). Experimental left varicocele was induced in rats through the partial ligature of the left renal vein. Euthanasia was performed at 100 dpp. Testicular histopathology and testosterone plasmatic level were evaluated. Transferrin and vimentin proteins were, respectively, used as immunomarkers of Sertoli cell function and structure. Significant reductions in vimentin and transferrin expressions were noticed in androgen-dependent stages (VII and VIII) of the seminiferous epithelium cycle in V rats; testosterone plasmatic level was also reduced. Bilateral testicular histopathological alterations were found in V rats, mainly massive germ cell desquamation. The histological damage and changes in protein expressions occurred bilaterally. The relevant impairment of the functional and structural characteristics of the Sertoli cell, together with the typical massive germ cell desquamation, indicates that Sertoli cell changes can primarily contribute to the significant testicular dysfunction associated with varicocele.
Introduction
Nowadays, infertility affects around 15% of couples. 1 In this circumstance, varicocele has a relevant role because it constitutes the most common cause of male infertility, affecting 15% to 20% of adults and adolescents around the world.2,3 Among the conditions that compromise the reproductive potential in men, varicocele alone accounts for 35% of cases. 1 It is responsible for 19% to 41% of primary male infertility and for up to 80% of secondary male infertility. 4
It is well known that the abnormal dilatation of pampiniform plexus observed in idiopathic varicocele conduces to an increased vein tortuosity.3,5 In addition, venous stasis and raised testicular temperature have been related to its pathogenesis. Testicular venous hypertension, testicular hypoxia, and subsequent ischemic injuries may also be linked to the establishment of infertility in men with varicocele.6,7 Not surprisingly, the overproduction of free radicals (reactive oxygen species [ROS]) in this condition has been indicated as the main causative factor of changes in the male gamete production, affecting both spermatogenesis and the late maturing of the germinal lineage cells.4,8,9 However, even though it is an important cause of male subfertility, and the mechanisms involved in varicocele spermatogenic damage have been intensely discussed, as formerly mentioned, the primary triggering targets of this damage seem to be still not well defined.
The Sertoli cells, when exposed to either pathological or toxicological conditions, are usually affected and undergo important functional 10 and structural alterations that can compromise the architecture of the seminiferous epithelium as observed in the acute effect of butylparaben11,12 and after nicotine exposure. 13 In these models, a downregulation of both cadherin and vimentin was associated with an impaired spermatogenesis. This impairment could be attributed to a disruption of the junctions between Sertoli and germ cells, conducing to massive desquamation of the seminiferous epithelium. 14 Indeed, through an intimate contact with the germ cells, the Sertoli cells provide an adequate microenvironment for normal spermatogenesis 15 by controlling cellular proliferation and survival.16,17 Thus, to orchestrate the seminiferous epithelium cycles, the Sertoli cell must sort a large variety of substances.18,19
Vimentin (a type III intermediate filament) comprises the predominant type of protein found in the Sertoli cell cytoskeleton during normal spermatogenesis 20 ; the pattern of its distribution is directly related to the morphological integrity of the seminiferous epithelium. 21 The downregulation of vimentin has been linked to decreased spermatogenesis, 12 and thus, vimentin has been considered a structural marker of Sertoli cells.22,23 On the contrary, transferrin is one of many proteins secreted in large quantity by Sertoli cells and represents a good tool for investigating its function.24,25 França et al. 26 highlighted that Sertoli cells are considered the “true nurse cells” because transferrin represents an important molecule in the process of nourishment delivery as it provides mechanisms to transport iron (Fe³) across the blood–testis barrier (BTB), providing support to the differentiation of the germ cells. The molecule is responsible for carrying iron, an important element for the progression of spermatogenesis,27,28 and also has high antioxidant power. 29
Taking into account the possible occurrence of structural and metabolic alterations in Sertoli cells under abnormal conditions that involve oxidative stress, the relevant role of these cells for the establishment and regulation of spermatogenesis, and the scarce number of research works discussing the importance of Sertoli cell damage in the varicocele development,14,30,31 we decided to study the impact of varicocele on the alteration in the function and structure of Sertoli cell and its involvement in the loss of integrity of the seminiferous epithelium. As varicocele has a higher occurrence on the left side5,32,33 and is commonly established early in puberty, 34 but is clinically diagnosed only during sexual maturity, 35 we decided to experimentally induce the left-sided varicocele in peripubertal rats, as this model can provide a scrutiny about the impairment of Sertoli cell in adulthood and its key role in spermatogenic injury triggered from sexual immaturity.
Materials and Methods
Animals
Twenty-five Wistar rats (Rattus norvegicus albinus) were obtained from the Center of Development of Experimental Models for Medicine and Biology (CEDEME/UNIFESP, São Paulo, Brazil). All the rats were maintained in polypropylene cages (40 cm x 30 cm x 15 cm) under wood shavings and appropriate conditions of hygiene, temperature (22–23C), humidity (45–55%), and light (12 hr light/dark). Throughout the experiment, animals had free access to filtered water and appropriate chow (Labina; Purina, Paulínia, São Paulo, Brazil).
Experimental Protocol
The experimental protocol was delineated in accordance with the Ethical Principles of Animal Research described by the Brazilian College of Animal Experimentation (COBEA). The protocol of care and treatment was approved by the Ethical Committee for Animal Research of the Federal University of São Paulo, Brazil (0059/2012).
At peripuberty (44 dpp), the rats were distributed into two experimental groups: varicocele (V, n=15
Right before the surgical procedure, the animals were weighed and anesthetized using ketamine–xylazine 4:1 (Dopalen–Anasedan, 100 mg/kg of body weight) via intraperitoneal injection. Trichotomy and asepsis of the abdominal region were performed in advance, and the following steps were carried out: (1) midline laparotomy incision, (2) careful externalization of the intestinal loops, and (3) exposure of the left kidney, left adrenal vein, inferior cava vein, left spermatic vein, and left renal vein (LRV). The last one was then occluded through a consistent stenosis using a peridural catheter (L = 30 mm, Ø = 0.45 mm × 0.8 mm) and a 3-0 cotton suture as a guide to standardize the reduction produced in the LRV. A 0.8 mm needle or catheter may be more appropriate for constriction of the LRV in adolescent rats; moreover, using a catheter of that diameter, an evident dilatation of the pampiniform plexus is achieved and no left renal atrophy is observed. 37 The catheter was then carefully removed to permit the expansion of the LRV against the suture. The closure of the abdominal wall was performed in two borders using a 4-0 mononylon suture. At the end of the procedure, the animals were monitored for postanesthetic recovery and kept individually in appropriate cages as previously described. Ibuprophen (20 mg/kg of body weight) was administered by gavage for five consecutive postsurgery days for analgesia using a stainless steel needle (18G; Thomas Scientific, Swedesboro, NJ). The general clinical and behavioral characteristics of the rats were monitored daily.
According to Turner, 36 the percentage of failure to induce varicocele using the technique described above is around 10%. Therefore, at the euthanasia day, the rats submitted to ELV were checked for the expansion of the left spermatic vein; only rats with such expansion were included in the V group.
Testosterone Plasmatic Dosage
On the euthanasia day (100 dpp), an intraperitoneal injection of heparin (Clexane, 100 IU/kg of body weight) was given to the rats for their blood collection. After 15 min, 10 mg/kg of lidocaine and also 120 mg/kg of body weight of thiopental sodium (Amental; Strides Arcolab Ltd.; Bangalore, Karnataka, India) were intraperitoneally injected into the animals.38,39 Then, after laparotomy, a blood sample was collected from the inferior cava vein, thus confirming euthanasia. Blood samples were centrifuged at 4500 rpm (Eppendorf, model 5430R) for 12 min. The plasma collected was stored in duplicates at −20C. Testosterone plasmatic levels were measured using chemiluminescence (Kit COBAS 05200067190, reference number 178782) with an analytical sensitivity of 6.08 ng/ml.
Testicular Weight and Histological Preparation
Immediately after euthanasia, the right and left testes were collected and weighed (Marte semi-analytical balance, Model AS 1000). Relative testicular weight was calculated (milligrams of testicular weight/100 g of body weight). Then, testicular fragments were fixed by immersion in Bouin’s solution for 24 hr, following all the recommendations reported by Russel 40 and Nolte et al., 41 and further prepared for paraffin embedding following a standard protocol. Testicular cross-sections with 4 and 6 µm thicknesses were used for histopathological and immunohistochemical analyses, respectively. Non-consecutive testicular sections of each thickness were obtained with an interval of seven 10-µm-thick sections between each chosen section.
Histopathological Analysis
For histopathological analysis, two sections (4 µm) of testis from each rat were submitted to hematoxylin and eosin (H&E) stain. Two hundred H&E-stained tubular sections per rat (100/testicular section) were randomly and systematically analyzed using Leica Q-Win V3 image analysis system coupled to a microscope (Olympus BX-50) under a 20× objective lens. The following parameters were assessed: (1) cellular debris and cell sloughing into the tubular lumen, (2) basal portions of the seminiferous epithelium detached from the tubular lumen, (3) intraepithelial vacuolization, and (4) retention of late spermatids (in stages IX and X of the seminiferous epithelium). The tubular sections were categorized, rated, and classified according to the characteristics of the histological alterations observed; then, a crescent score was established (from 1 to 4) according to the frequency of the alterations found. Scores were expressed in percentage and categorized as follows: 1 = 0 to 25%; 2 = 26% to 50%; 3 = 51% to 75%; 4 = 76% to 100%, according to Consentino et al. 42 and Oliva and Miraglia. 43
Immunolabeling of Transferrin and Vimentin
Two non-consecutive testicular sections (6 µm) per animal were placed in silanized slides, deparaffinized, hydrated, and submitted for the inactivation of the endogenous peroxidase by immersion in hydrogen peroxide solution (3%). After that, the slides were treated for labeling of each protein. For detecting transferrin in the seminiferous epithelium, the slides were previously treated with 15% bovine serum albumin (BSA) for 20 min, aiming to prevent background; then, they were incubated with the anti-transferrin primary antibody (1:5000; rabbit anti-rat, A110-124-1; Bethyl Laboratories, Montgomery, TX) for an hour. For detecting vimentin expression, the slides were immersed in citrate buffer and kept in a microwave for 15 min for heat-induced antigen retrieval; then, they were treated with 10% BSA for 20 min and incubated with the anti-vimentin primary antibody (1:150; mice anti-rat, sc-6260; Santa Cruz Biotechnology, Inc, Dallas, TX) for 2 hr. After that, all the slides were incubated with the biotinylated secondary antibody for 30 min and then with peroxidase conjugated with avidin for further 30 min (LSAB, K0690; Dako, Carpinteria, CA). All the incubations were performed in humid chamber at room temperature, and the slides were washed with phosphate buffer (0.05 M pH 7.4) between intervals. The slides were finally treated with the chromogen 3′3-diaminobenzidine (DAB K3468; Dako) and Harris’ Hematoxylin as a counterstaining. Regarding the analysis of both proteins (vimentin and transferrin), only seminiferous epithelium cells displaying characteristic dark brown staining were considered positive. Negative controls were obtained by omitting the primary antibody.44,45
Stereological Analysis: Volume Density of the Immunolabeled Proteins
The volume density (Vv) is a stereological parameter that characterizes the relative occupation of a component in a tissue or organ. 46 According to the basic principle of stereology, “the area density of the profiles (AA), which measures the relative occupation of the test-area for the area of the images of the structure under evaluation, is comparable to its volume density (Vv),” that is, to the density of profiles in relation to the space; as a result, Vv and AA have a similar interpretation.46–50 Therefore, using the image analysis system, the immunolabeled areas of each protein studied (in tubular sections from the left and right testes) were delineated in the captured images to generate the calculation of Vv. As a result, the Vv was calculated by the ratio between the total positive area in the seminiferous epithelium (Ae+) observed for each protein and the total analyzed area in the tubular sections (Att), that is, (Ae+/Att). Although these proteins are also expressed in the interstitial tissue, only immunolabeling detected in the seminiferous epithelium was considered.
The Vv of transferrin and vimentin immunolabeling found in the seminiferous epithelium was calculated separately. For each one of the proteins, we sampled four different sections per animal (right and left testes, two sections each), and 50 fields per testicular section were systematically and randomly scrutinized, totaling 100 fields per testis. As a result, an average of 700 tubular sections for each side was carefully examined (1400 per animal). This analysis was performed using Leica Q-Win V3 image analysis system coupled to a microscope (Olympus BX-50) under a 20× magnification objective lens.
Besides the analysis of the Vv of vimentin expression in the indistinctive stages of epithelium seminiferous cycle (global analysis), we also investigated its specific Vv in the androgen-dependent stages (VII and VIII stages) as the pattern of distribution of vimentin in the Sertoli cells varies according to the seminiferous epithelium stage 20 ; in addition, this is an interesting parameter to relate to the testosterone levels. The same analysis was performed to detect transferrin expression in androgen-dependent stages because it is maintained in vivo by testosterone 51 and for knowing whether the mRNA levels of transferrin receptors are maximum in those stages. 52
Statistical Analysis
The results were submitted to the statistical tests provided by the software SigmaPlot 11.2 (Systat Software Inc.; San Jose, CA). All the data obtained in our experimental evaluation presented normal distribution and were analyzed using the Student’s t-test to compare the V and C groups. Data obtained from the ipsilateral testis (Vl = left testis) and from the contralateral testis (Vr = right testis) to the varicocele were, respectively, compared with each other and with the mean value of the C group (obtained from the left and right testes) using the test one-way analysis of variance. The differences were considered significant when p≤ 0.05. When significant differences were observed in a particular parameter, the Student–Newman–Keuls multiple comparison test was used.
Results
Animals were monitored daily throughout the study. No clinical alterations, as chromodacryorrhea, and changes in behavioral or eating patterns were observed. No statistical significance was detected between the body weight means of V and C groups (Table 1).
Body Weight and Testicular Biometric Data (Mean Values of Both Testes) Obtained From Rats of the Control (C) and Varicocele (V) Groups and of the Ipsilateral (Vl) and Contralateral (Vr) Testes to the Varicocele at 100 dpp.
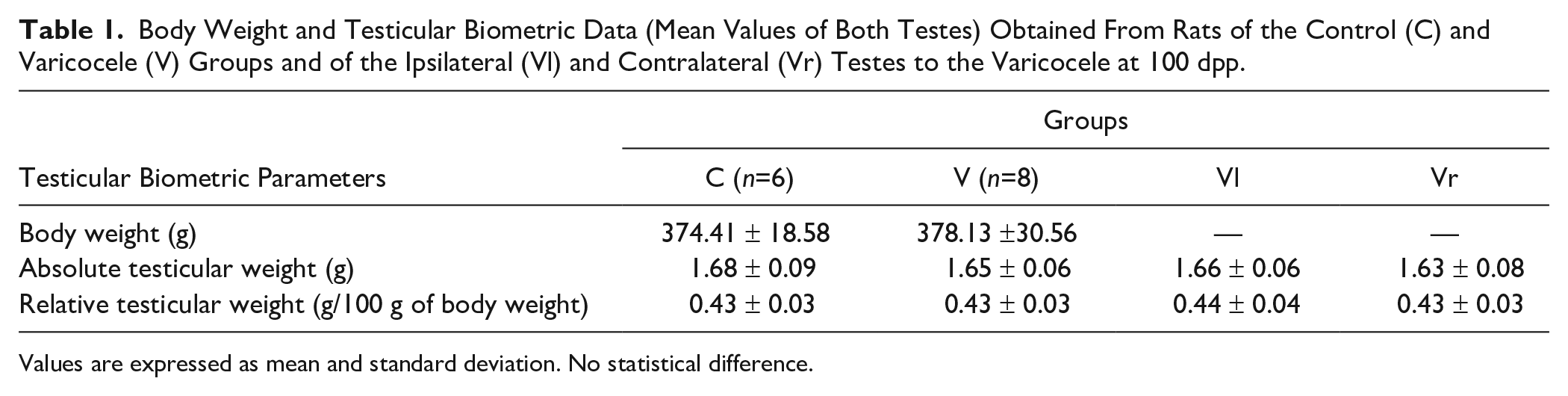
Values are expressed as mean and standard deviation. No statistical difference.
Biometric Data
Testes from animals pertaining to groups C and V showed no altered macroscopic characteristics, such as shape, color, or morphological aspects. Absolute and relative testicular weights were not significantly different either, even when the mean of both testes of each animal was considered (Table 1); a similar result was noticed when the mean values obtained from the right (Vr) and left (Vl) testes from varicocelized rats were compared with the mean values obtained from both testes of animals of the C group (Table 1).
Histopathological Analysis
Animals of the C group presented a normal aspect in the seminiferous epithelium as expected for the age, showing germinal cell lineage organized in typical cell associations, containing spermatogonia, spermatocytes, and spermatids in various steps of spermiogenesis. In addition, mature Sertoli cells displayed a characteristic euchromatic and polymorphous nuclei with evident nucleoli, lying on the basal region of the tubular section (Figs. 1A and B). In these rats, only a few tubular sections displayed desquamated epithelial cells in the tubular lumen. On the contrary, in the varicocelized animals, we frequently noticed sections containing cellular debris and an intense desquamation of epithelial cells into the lumen, including some Sertoli cells, which was evidenced by their nuclei (Fig. 1C; Table 2). Moreover, these animals showed disorganized seminiferous epithelium; some of them presented associations that characterize stages IX, X, and XI within the same tubular section, which is not found in normal rats (Fig. 1D). Elongated sperm heads were also arrested among the germ cells in IX and X postspermiation stages (Fig. 1D). Displacement of parts of the more basal portion of the seminiferous epithelium into the tubular lumen, cell depletion (Fig. 1E), and intraepithelial vacuolization (Fig. 1F) were also commonly seen in the varicocelized rats. Such alterations were significantly more frequent in the varicocelized compared with the control rats, as shown in Table 2. Some of these alterations were more frequent in the Vl (left) testis, such as the tubular sections containing massive desquamated germ cells in the lumen.
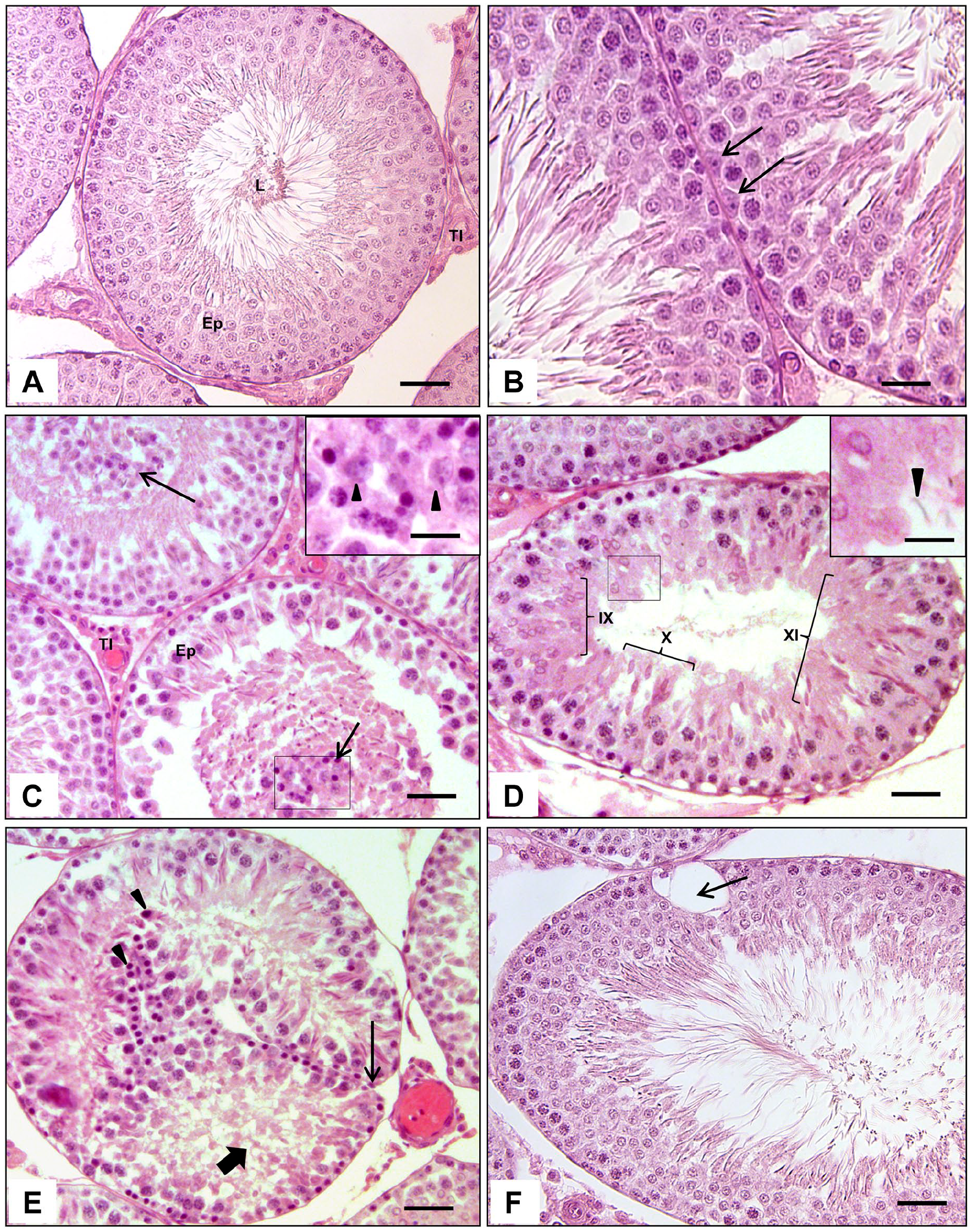
Photomicrographs of testicular cross-sections of control (A and B) and varicocelized (C, D, E and F) rats at 100 dpp, stained by the hematoxylin and eosin method (H&E). Observe in the rat testis of the C group (A) an organized seminiferous epithelium (Ep), an interstitial tissue with a normal aspect (TI), and a tubular lumen with a moderate amplitude, with no cells detached into it (L). Scale bar = 40 µm. In a higher magnification, note the normal characteristics of the germ cells and the Sertoli cell nuclei (arrows—B). Scale bar = 15 µm. In the varicocelized animals, observe the high number of epithelial cells desquamating into the tubular lumen (arrows—C), including some Sertoli cells, of which the nuclei can be easily visualized (arrow heads—inset). In (D), observe the tubular section containing a quite disorganized seminiferous epithelium, containing germ cell associations of different stages (IX, X, and XI) (arrows—D), and some spermatid heads retained in the basal region (arrow head—inset). (E) shows whole portions of seminiferous epithelium clearly detached from the basal layer of some tubules and folding into the tubular lumen (thin arrow—E); cellular depletion (thick arrow—E) and many degenerating germ cells desquamating into the tubular lumen (arrow heads — E). In (F), an intraepithelial vacuolization (arrow—F) can be evidenced. Scale bar = 35 µm.
Percentage of Tubular Sections Showing the Seminiferous Epithelium Alterations and the Score of the Frequency (1–4) of These Changes in Animals of the Control (C) and Varicocele (V) Groups, Considering the Ipsilateral Testis (Vl = left) and the Contralateral Testis (Vr = right) to the Varicocele.

Score: 1 = 0–25%; 2 = 26–50%; 3 = 51–75%; 4 = 76–100%. Statistical tests: analysis of variance and Student–Newman–Keuls. Values are expressed as mean and standard deviation.
Vl and Vr > C (p≤ 0.001); #Vl > Vr (p≤ 0.05).
Immunohistochemical and Stereological Analysis
Vv of Transferrin in the Seminiferous Epithelium
A significant reduction was observed in the transferrin immunolabeling of the V group seminiferous epithelium compared with that of the C group. The difference was significant either for global evaluation (when the mean of both testes was compared in both groups) or when the Vl and Vr testes to the varicocele were, respectively, compared with the mean value obtained from the C group (Figs. 2A and B, 3A and B). There was also a significant difference when the right and left testes of the V group were compared with each other (more conspicuous damage in the Vl testis).
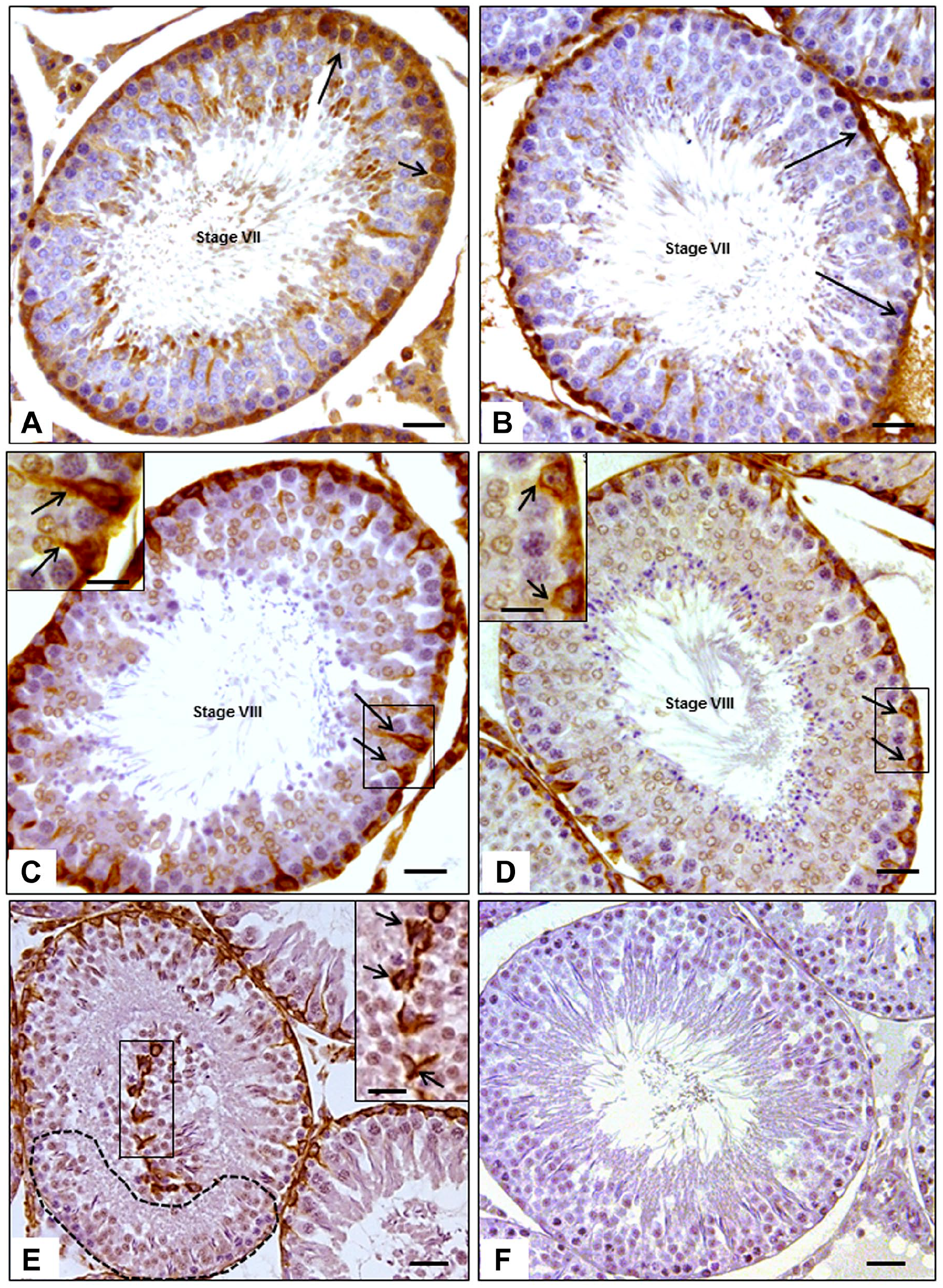
Photomicrographs of testicular cross-sections of control and varicocelized rats at 100 dpp showing the immunolabeling of transferrin (A and B) and vimentin (C, D and E), counterstained with Harris’ Hematoxylin. Note that the transferrin immunolabeling in the seminiferous epithelium (arrows) of a tubular section, in an androgen-dependent stage (stage VII), is stronger and wider in the testis of the control rat (A) than in the varicocelized rat (B). The vimentin immunolabeling in the cytoplasm of Sertoli cells (arrows), in sections of androgen-dependent seminiferous tubules (stage VIII), is also stronger and wider in the animal of the control group (C) than in varicocelized rat (D), in which the labeling is more restricted to the perinuclear region. Scale bar = 25 µm; scale bar of the inset = 10 µm. Also observe the presence of vimentin-positive material in the tubular lumen of a section of varicocelized animal (E), besides a region without labeling of vimentin (dotted area). Scale bar = 40 µm; scale bar of the inset = 10 µm. The negative control (incubated without primary antibody) shows no markings (F). Scale bar = 40µm.
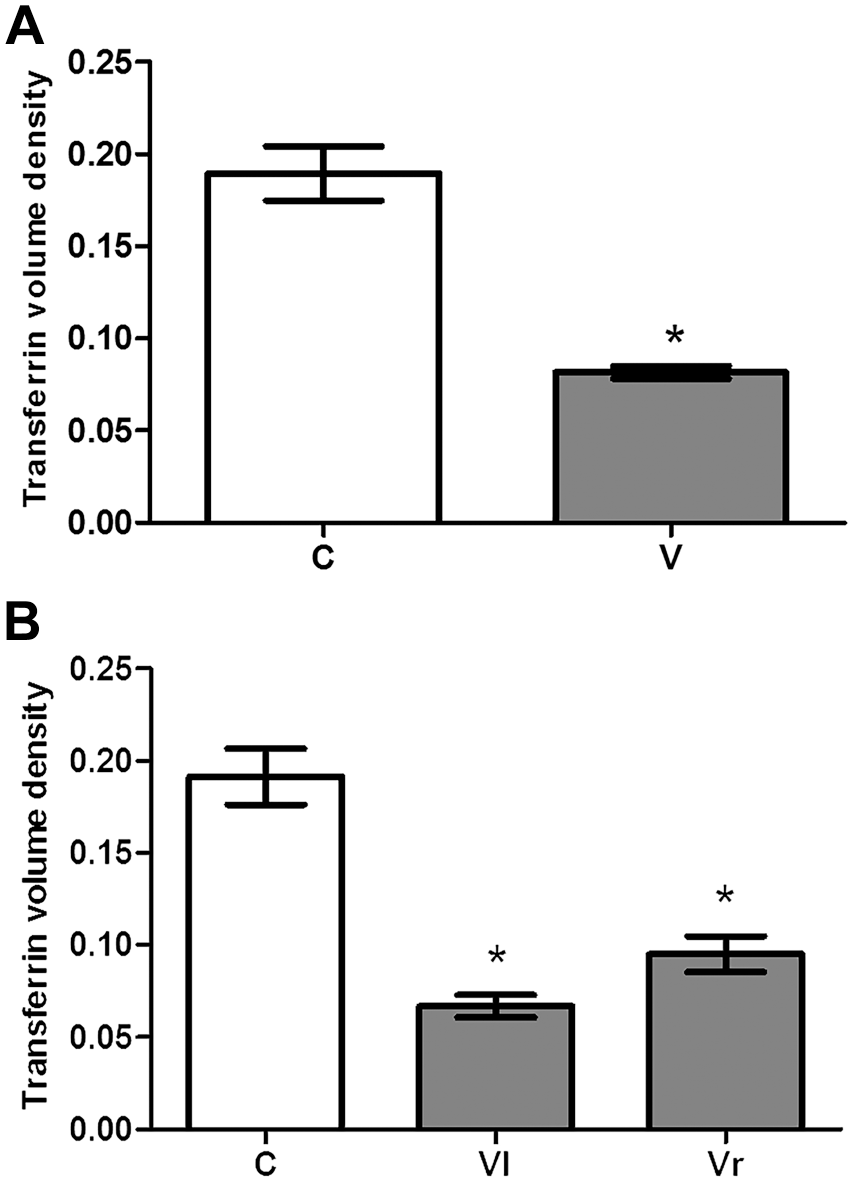
Volume density of the transferrin immunolabeling of animals of the control (C) and varicocele (V) groups, considering the global evaluation (A) and the separated data of the ipsilateral testis (Vl = left testis) and the contralateral testis (Vr = right testis) to the varicocele (B), in tubular sections of different stages of the seminiferous epithelium cycle. Values are expressed as mean and standard deviation (n=6). Statistical tests—(A) Student’s t-test:
A significant reduction was also noticed in transferrin labeling in androgen-dependent stages of the seminiferous epithelium (VII and VIII) in the V group compared with the C group (Fig. 4A and B). The reduction occurred when the mean values of both testes were compared between the two groups (Fig. 4A). A decrease in this parameter was also observed in the Vl and Vr testes to the varicocele when each one was compared with the mean value obtained from the C group. Similarly, a significant difference was noticed when the left and right testes of animals of the V group were compared among them (Fig. 4B) so that it was more evident in the left testis.
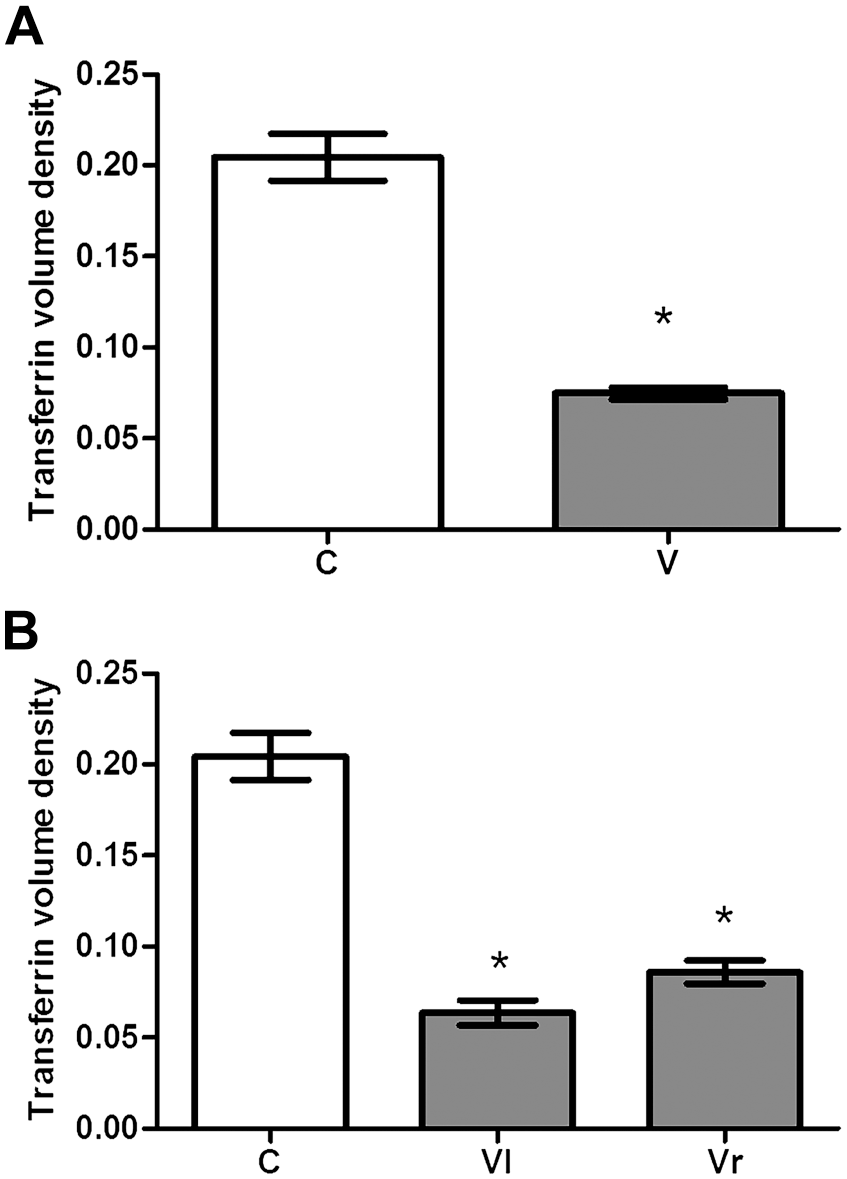
Volume density of transferrin immunolabeling in the androgen-dependent stages of the seminiferous epithelium (VII and VIII) in the animals of the C (control) and V (varicocele) groups, considering the global evaluation (A) and the data obtained from testes that were ipsilateral (Vl = left testis) and contralateral (Vr = right testis) to the varicocele (B). Values are expressed as mean and standard deviation (n=6). Statistical tests—(A) Student’s t-test: *V < C (p≤ 0.05); (B) analysis of variance and Student–Newman–Keuls: *Vl < Vr < C (p≤ 0.05).
Vv of Vimentin in the Seminiferous Epithelium
The expression of the protein vimentin was evident in the cytoplasm of Sertoli cells and more intensely in the perinuclear region, but also observed in the apical region of these cells (Figs. 2C and D). The detached Sertoli cells, seen in the tubular lumen, also presented vimentin immunolabeling (Fig. 2E). Statistically significant alterations were not observed in the global evaluation of vimentin (Vv) between the V and C groups. Moreover, no differences were detected comparing the Vv in the testes that were VI and Vr to the varicocele with each other and with the C group (Figs. 5A and B).
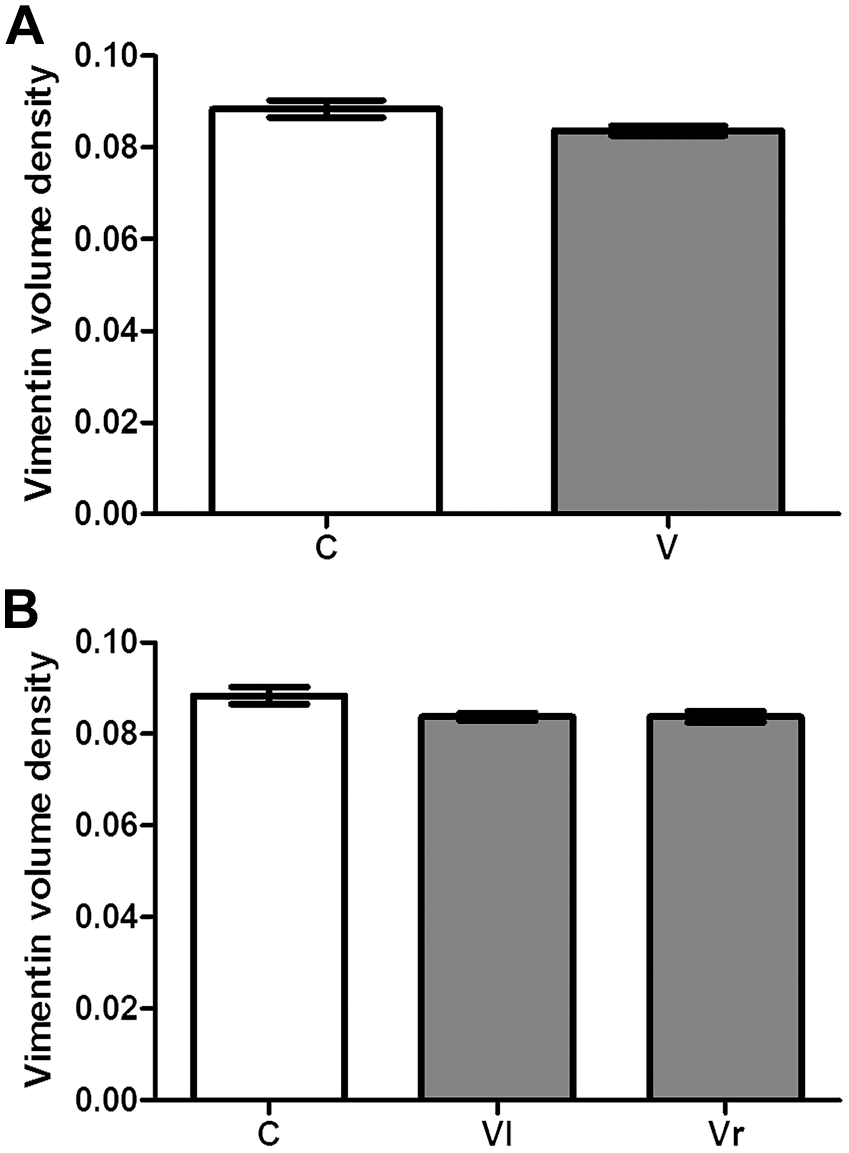
Volume density of the vimentin immunolabeling in the seminiferous epithelium in different stages of the seminiferous epithelium cycle in animals of the control (C) and varicocele (V) group, considering the global evaluation (A) and the data obtained from testes that were ipsilateral (Vl = left testis) and contralateral (Vr = right testis) to the varicocele (B). Values are expressed as mean and standard deviation (n=6). Statistical tests: (A) Student’s t-test; non-significant differences. (B) Analysis of variance; non-significant differences.
However, in the androgen-dependent stages (VII and VIII), vimentin immunolabeling was significantly reduced in the animals of the V group compared with those of the C group (Fig. 6A). The volume densities of this protein in Vl and Vr testes to the varicocele were also reduced compared with those obtained in the C group (Figs. 4C and D, 6A and B). A significant reduction of this parameter was also noticed in the left testis in comparison with the right testis of varicocelized rats (Fig. 6B).
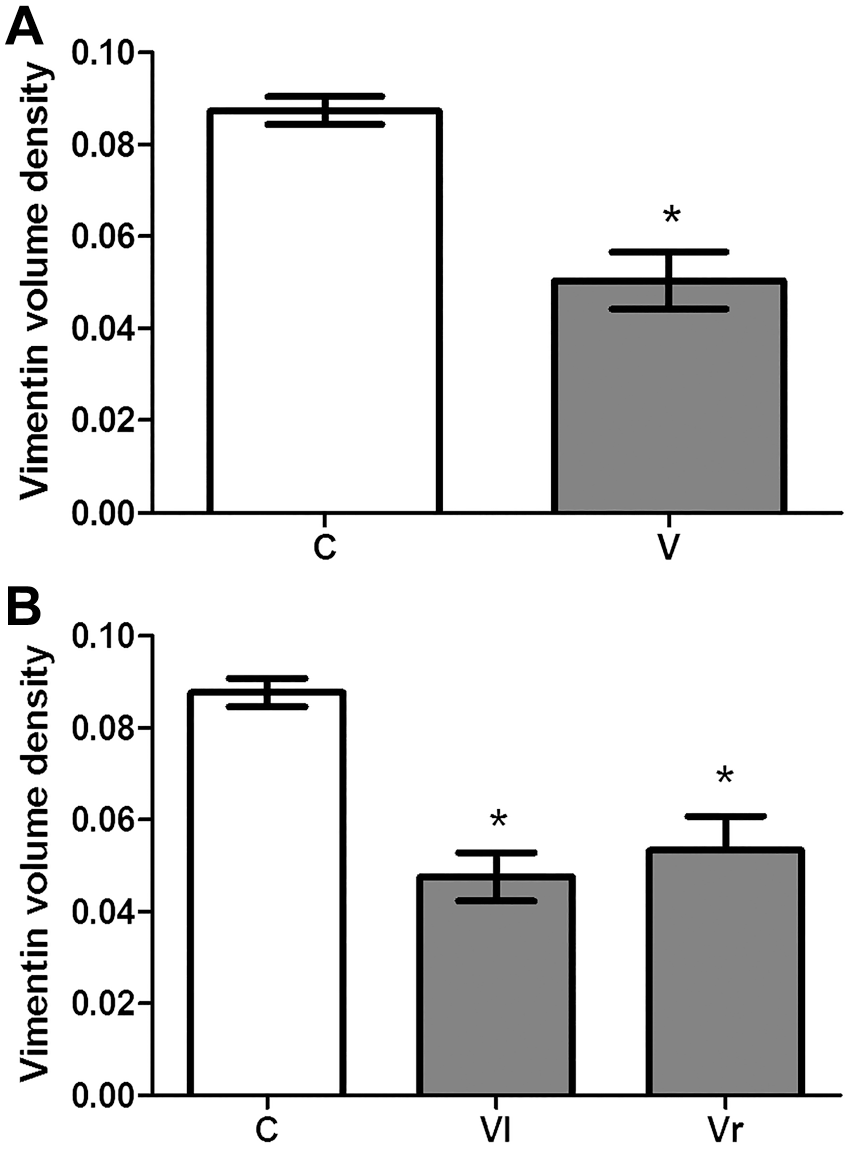
Volume density of vimentin immunolabeling in the seminiferous epithelium in the androgen-dependent tubules (VII and VIII) in the animals of the control (C) and varicocele (V) groups, considering the global evaluation (A) and data obtained from the testes that were ipsilateral (Vl = left testis) and contralateral (Vr = right testis) to the varicocele (B). Values are expressed as mean and standard deviation (n=6). Statistical tests—(A) Student’s t-test: *V < C (p≤ 0.05); (B) analysis of variance and Student–Newman–Keuls: *Vl < Vr < C (p≤ 0.05).
The negative control image, by primary antibody omission, is also shown in Fig. 2F.
Plasmatic Levels of Testosterone
A significant reduction of the testosterone plasma levels was observed (p≤0.05) in the animals of the V group compared with the C group (Fig. 7).
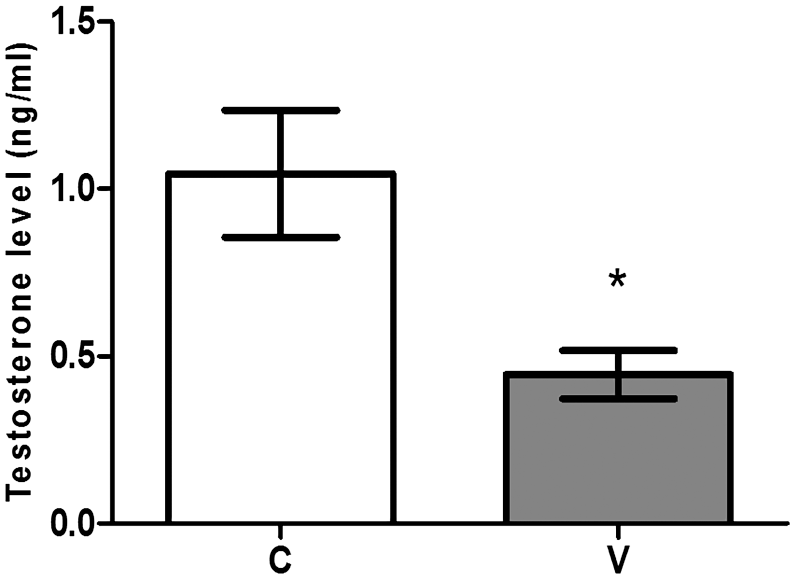
Plasmatic levels of testosterone in the animals of the control (C, n=10) and varicocele (V, n=15) groups. Values are expressed as mean and standard deviation. Statistical tests—Student’s t-test: *V < C (p≤ 0.05).
Discussion
The experimental model of induced varicocele using rats is a habitual procedure due to the anatomical similarity and distribution of blood flow compared with humans. In this study, we could control the time of establishment and development of the varicocele, from peripuberty, using an optimized experimental model with the aim to contribute to the understanding of the reproductive pathophysiology in adolescence.37,53 In addition, the effects of varicocele on the testicular structure in rats can vary according to the time of varicocele onset: at prepuberty (4 weeks), adolescence (7 weeks), or adulthood (12 weeks); not surprisingly, the testicular damage seems to be more conspicuous at adolescence. 54 Over the years, spermatogenic damage caused by varicocele seems to be progressive.31,55
There are some references indicating that the occurrence of bilateral palpable varicocele can be higher than 50%. 4 In the current research, left-sided varicocele led to a bilateral alteration of testicular structure in adulthood.55,56 Changes in the testicular expression of vimentin and transferrin (in androgen-dependent tubules) occurred in both the left (Vl) and right testes (Vr) of varicocelized rats and happened along with evident damage in the seminiferous epithelium. However, as expected, a worse lesion was observed in the Vl testis, in comparison with the Vr testis.
Sertoli cells play many specialized functions, which are important to provide the required microenvironment for the development of the germ cells. 26 The first described functions were nursing and supportive roles that guarantee the maintenance of testicular homeostasis, nutrition, secretion of growth factors, and synchronization of clones of germ cells in specific steps of differentiation and spermiation.57,58
Vimentin, the main intermediary filament in mammal Sertoli cells, is a fundamental component of its cytoskeleton and has a role in maintaining the integrity of the seminiferous epithelium. 20 Thus, it is possible that, in varicocele, early loss of germ cells from the epithelium and their early detachment into the lumen were a secondary effect, as a consequence of the rupture of junctions between Sertoli and germ cells. In addition, the observation of detachment of some folded portions of seminiferous epithelium from the peripheral tubular region into the tubular lumen also indicates that the distribution of vimentin was affected primarily. This protein distribution normally changes according to the cycle of seminiferous epithelium in a stage-dependent manner and can be associated with hemidesmosome-like structures attached to the basal lamina. 59 In the current report, there was a conspicuous reduction of vimentin expression in tubules at androgen-dependent (VII–VIII) stages in varicocelized rats. Alterations in vimentin biochemical properties may be a probable mechanism involved in the loss of adherence among germ cells or to transfer a death signal to germ cells and subsequently to a decrease in intratesticular testosterone levels (ITLs), 60 which may have occurred due to a primary disturbance in the paracrine regulation of Sertoli cells. 61
The histopathological results presented here also showed vimentin labeling in the perinuclear region of damaged Sertoli cells that were sloughed into the tubular lumen of varicocelized rats along with a massive quantity of germ cells. The desquamation of germ cells into the luminal compartment was previously seen by Cameron et al., 14 in testicular biopsies from 21 otherwise healthy men (22–37 years old) diagnosed with varicocele. They also described structurally abnormal Sertoli–germ cell junctional complexes using electron microscopy and suggested that Sertoli cell could be a primary target in the varicocele. However, the age of the samples of biopsied testes was probably too heterogeneous, varying by 15 years; moreover, time of varicocele onset was not considered, and consequently, the degree of spermatogenic damage probably varied as well. Furthermore, the morphology of the Sertoli cell changes depending on the stage of the seminiferous epithelium cycle and staging the fragments of biopsied testicular tissue, especially under a transmission electron microscope, are limiting factors. In addition, no specific damage to the cytoskeleton components of Sertoli cells was investigated by these authors, as we show here. Also, we recently observed that vimentin loss is one of the consequences of testicular tissue damage after nicotine exposure in rats. 13 In fact, disruption of vimentin filaments can cause immature germ cell sloughing23,62 after exposure to different xenobiotics. In this study, the cell association of different stages of the seminiferous epithelium cycle was sometimes abnormally observed in a same tubular section, which is another sign of alteration in the testicular cycle that is controlled by the Sertoli cell. 63
The effects of oxidative stress outbalance caused by varicocele as well as its role in reproductive damage have already been well documented and recognized as a pathogenic mediator in testicular damage.4,64 Although the oxidative stress can be quite harmful to any germ cell,4,8,9 Sertoli cells may also be a target of the excessive oxidative stress, and thus, considering their nursing function, a primary injury in these cells can be an important trigger for the spermatogenic damage. For instance, increased ROS formation observed in varicocelized rats alters the expression of α-catenin and E-cadherin, leading to seminiferous epithelium degenerative changes, suggesting the damage of the BTB. 65 Indeed, damage of Sertoli–Sertoli cell junctional complexes may lead to a dysfunction of BTB and consequently to an alteration of microenvironment of seminiferous tubule. 66 Thus, four proteins—α-catenin, E-cadherin, 65 vimentin and transferrin—signalize a primary damage to the Sertoli cell in varicocele.
Another condition that may lead to the impairment of the Sertoli cell function in varicocelized rats is the decrease in testicular blood flow. 67 The venous stasis and raised testicular temperature have been also related to varicocele pathogenesis. Zhang et al. 68 suggested that heat stress produced by cryptorchidism in monkeys alters the cytoskeletal components of the Sertoli cell. They observed a disorganized distribution of vimentin and loss of its extension associated with the perinuclear area, which may increase the vulnerability of Sertoli cells in a stage-dependent manner. 59 Regarding this issue, Vallés et al. 69 showed in vivo (using rats) and in vitro (using mouse-derived cells) that daily repeated exposures to moderate hyperthermia, in regular intervals, cause irreversible injuries by provoking harmful consequences on the Sertoli cell lipid homeostasis. According to these authors, high temperatures affect Sertoli cell mitochondrial physiology as there is an increased degree of membrane depolarization and ROS overproduction. 4 This could be one of the mechanisms to harm the Sertoli cell in varicocele, but the range of damage and the consequences to intracellular integrity may vary and still need to be deeply investigated.
Using transferrin immunolabeling30,70 we showed that left-sided varicocele caused a decrease in both global and VII/VIII stage-specific transferrin in rats, indicating a functional alteration in the Sertoli cell. Under normal conditions, transferrin mRNA levels vary twofold across the complete cycle, being high during the stages XII to XIV and achieving its maximum at stages VII and VIII. 52 In addition, there is a positive correlation between the reduction of seminal transferrin and disruption in spermatogenesis. 71 Indeed, the positive correlation between the reduction of seminal transferrin and the disruption in spermatogenesis 71 points out that varicocele impairs the Sertoli cell function.
Similarly, another study has already shown that patients with varicocele usually present increased levels of serum follicle-stimulating hormone in response to a drop of inhibin production, 72 which may also reflect in Sertoli cell malfunction and altered production and secretion of androgen-binding protein. 73 In fact, concerning the plasmatic testosterone level, a reduction was observed in the present study, as already observed by Zheng et al. 31 and Razi et al. 74 Adequate intratesticular testosterone levels (ITLs) maintain the integrity of the seminiferous epithelium in androgen-dependent stages (VII and VIII). Thus, the evident decrease in transferrin and vimentin Vv in VII/VIII androgen-dependent stages of the seminiferous epithelium cycle suggests that an alteration of the ITLs may also have occurred. The key role played by the Sertoli cell in paracrine regulation within the testicular tissue, through the secretion of many control signals, can also affect the Leydig cell function. Thus, besides the disturbance to the spermatogenesis, injured Sertoli cells can cause deficient testicular activin and inhibin signaling, both products of Sertoli cells, altering important signals to testosterone production by Leydig cells. 61
As Sertoli cells play a fundamental role in the spermatogenic process, the precise understanding of the conditions, necessary for the proper functioning of these cells, as well as their respective basic molecular mechanisms involved in testicular functions, are the basis to identify the main events that occurred in this process.26,75 On the contrary, this places the Sertoli cell as a potential and central target in many adverse situations. The current research is the first experimental study focusing on the Sertoli cell as a primary target in adult testicular damage in rats, which were varicocelized from peripuberty (41 days). Although Pan et al. 76 have investigated the integrity of BTB in varicocelized rats, they induced varicocele when the animals were 12 weeks old. In their study, they observed a downregulation of claudin-11 protein and the disruption of this barrier in the varicocelized rats, which reinforces the idea that the Sertoli cell plays a key role in this type of damage.
The final involvement of increased ROS production and of the deficiency of the antioxidant enzyme system in the Sertoli cell damage may also play an important role; thus, future studies should investigate whether the oxidative stress caused by varicocele is involved in the alterations in vimentin and transferrin production by the Sertoli cells and other proteins. In addition, using the same experimental model, the study of Sertoli cell products in the fluid of rat vas deferens, such as transferrin and inhibin B, could be also performed to confirm the role of Sertoli cells as a primary target in cases of subfertility.
The investigation on what are the possible primary targets in adverse situations can be the key to understanding their pathophysiological mechanisms, contributing to achieve new strategies to help the huge number of people who suffer from subfertility.
Footnotes
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
ACV carried out animal surgery, tissue collection and fixation, paraffin embedding, and stereological analysis of the immunolabeled testicular cross-sections. CCP performed immunohistochemistry reactions, contributed to manuscript drafting, and selected the photomicrographs and arranged it in the manuscript. TBM helped in animal surgery and in all histological processing. RELC performed the statistical analysis. JNS checked the references and contributed to the revision of the second corrected version of the manuscript. VV contributed to final revision of the manuscript and English translation. SMM designed and coordinated the study, drafted the manuscript, and reviewed the final version of the manuscript. All authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - Brasil (CAPES)- finance code 001.
