Abstract
Staining for β-galactosidase activity for whole tissues, sections, and cells is a common method to detect expression of β-galactosidase reporter transgene as well as senescence-dependent β-galactosidase activity. Choice of fixatives is a critical step for detection of β-galactosidase activity, subsequent immunostaining, and enzymatic digestion of tissue to dissociate cells. In this report, the authors examined several aldehyde and alcohol fixatives in mouse skeletal muscle tissues for their efficiency at improving detection of β-galactosidase activity as well as detection by immunostaining. In addition, fixatives were also analyzed for their efficiency for collagenase digestion to isolate single muscle fibers on postfixed β-galactosidase-stained whole skeletal muscle tissues. The results show that fixing cells with isopropanol yields the greatest reliability and intensity in both β-galactosidase staining as well as double staining for β-galactosidase activity and antibodies. In addition, isopropanol and ethanol, but not glutaraldehyde or paraformaldehyde, allow for the isolation of single muscle fibers from the diaphragm and tibialis anterior muscles following postfixed β-galactosidase staining. Using this method, it is possible to identify the amount of cells that occupy the satellite cell compartment in single muscle fibers prepared from any muscle tissues, including tibialis anterior muscle and diaphragm.
Keywords
Adult skeletal muscle possesses remarkable regeneration capabilities. After exercise or muscle damage, large numbers of new muscle fibers are typically formed within one week because of the expansion and differentiation of muscle satellite cells (Asakura et al. 2001; Seale et al. 2001; Charge and Rudnicki 2004; Relaix and Marcelle 2009). Satellite cells are a small population that accounts for myogenic stem cells in muscle regeneration. These cells are mitotically quiescent in adult muscle, but they are activated following injury, initiating proliferation to produce myogenic precursor cells, or myoblasts, to mediate the regeneration of muscle tissues. These myogenic precursor cells undergo multiple rounds of cell division prior to terminal differentiation and formation of multinucleated myotubes by cell fusion.
Transplantation of myogenic precursor cells such as satellite cell–derived myoblasts in skeletal muscle is one of the most active areas of research for muscle repair in prominent disease models that include Duchenne muscular dystrophy (DMD) (Tedesco et al. 2010). Transplanted myoblasts are able to fuse with the existing muscle fibers to repair muscle damage. However, for any form of therapy to be effective in the long term, the satellite cell pool needs to be replenished. Current methods of evaluating satellite cell contribution of engrafted cells consist of extracting and developing cross-sectioned host tissue along with double immunostaining using satellite cell–specific markers such as Pax7 (Seale et al. 2000), donor-specific markers such as β-galactosidase (Beauchamp et al. 2000), or green fluorescent protein (GFP; Montarras et al. 2005). It is also important to distinguish between the proliferating satellite cells outside of basal lamina and the quiescent satellite cells under the basal lamina (Grange et al. 2002). For this purpose, double immunostaining with Pax7 and a basal lamina marker laminin is necessary. However, this technique can be very labor-intensive and time-consuming.
Studying intact single muscle fiber cultures is a conventional way to study quiescent and activated satellite cells in their native environment. These fibers still retain the laminin membrane and thus can elucidate the presence of satellite cells (Collins and Zammit 2009). After cell transplantation, isolated single muscle fibers can be stained for β-galactosidase activity to evaluate satellite cell contribution. However, several technical limitations exist with single muscle fiber isolation methods that prohibit examination of satellite cells other than from a limited set of less than ideal muscle tissues. For instance, the muscle tissue needs to be isolated from tendon to tendon, and fiber bundles need to be isolated unharmed to avoid rapid hypercontraction of the fiber. Therefore, isolation of single muscle fibers has normally been done only from muscles such as the extensor digitalis longus (EDL) and forward digitalis brevis due to their small size. However, these muscles have limited uses as models compared to the tibialis anterior (TA) and diaphragm muscles. The TA muscle is preferred for analyzing agents delivered through intramuscular injection such as with myoblast transplants, and the diaphragm muscle is the most severely affected muscle in the mdx mouse model for DMD (Boldrin et al. 2009; Verma et al. 2010). Therefore, it would be of value to be able to study quiescent satellite cells at the single muscle fiber level from these and other useful muscle models.
The aim of this study was (1) to evaluate fixatives that could give suitable results in both β-galactosidase (lacZ) staining as well as immunohistochemistry and (2) to optimize a method for isolating single muscle fibers from various muscle tissues by fixing the muscle prior to isolation of the fibers.
Materials and Methods
Animals
Heterozygous Myf5+/nLacZ mice (Tajbakhsh et al. 1996) were kindly provided from Shahragim Tajbakhsh. Heterozygous Flk-1-lacZ mice (Ema et al. 2006) were kindly provided from Masatsugu Ema. Homozygous MD6.0-lacZ mice were established in the Stephen Tapscott laboratory (Asakura et al. 1995). Homozygous ROSA26 (B6.129S7-Gt(ROSA)26Sor/J) mice (Zambrowicz et al. 1997) were purchased from the Jackson Laboratory. These mice were bred to maintain the colony in the principal investigator’s lab. For genotyping of Myf5+/nLacZ mice, small pieces of ear tissue were stained with X-gal solution for lacZ expression, or PCR analysis was performed with DNA extracted from tail biopsies. The following PCR primer pair was used for genotyping of Myf5+/nLacZ mice: 5′-TGAAGGATGGACATGACGGAC-3′ (MYFF) and 5′-GGGCGATCGGTGCGGGCCTCTTCGC-3′ (SJT-GAL4). For genotyping of MD6.0-lacZ mice, PCR analysis was performed with DNA extracted from tail biopsies (Asakura et al. 1995). The following PCR primer pair was used for genotyping of MD6.0-lacZ mice: 5′-CCCAAGCTCCGCCCTACTACACTCCTATTGGCTTGAGGCG-3′ (SJT-61) and 5′-GGGCGATCGGTGCGGGCCTCTTCGC-3′ (SJT-GAL4). For genotyping of Flk-1-lacZ and ROSA26 mice, small pieces of ear tissue were stained with X-gal solution for lacZ expression. The animals were housed in an SPF environment and were monitored by the Research Animal Resources (RAR) of the University of Minnesota. All protocols were approved by the Institutional Animal Care and Usage Committee (IACUC) of the University of Minnesota.
Myoblast Culture
Satellite cell–derived myoblasts were isolated from the diaphragm or hind limbs of one- to two-month-old heterozygous Myf5+/nLacZ mice (Asakura et al. 2001). Briefly, after cell dissociation with collagenase (Sigma-Aldrich; St Louis, MO) and dispase II (Roche Diagnostic; Indianapolis, IN) digestion, cells were maintained on dishes coated with collagen (BD Biosciences; Franklin Lakes, NJ) in myoblast growth medium consisting of HAM’s F-10 medium supplemented with 20% FBS and 5 ng/ml basic fibroblast growth factor (FGF; R&D Systems, Minneapolis, MN). The medium was changed daily, and cultures were routinely passaged as they reached 60% to 70% confluence. To maintain the primary characteristics of the cells, all experiments were performed by using cultures that had undergone between three and five passages. The same batch of cells was used for each experiment.
Dissection of Diaphragm and TA Muscles
To dissect the diaphragm, first the intraplural cavity was exposed and the various projections were cut. Next, an incision was made in the intercostal region superior to the diaphragm, encircling the muscle, including the ribs and spine, to obtain any undamaged tissue, taking care not to touch the muscle. To dissect the TA muscle, the fascia was removed and distal tendons were cut. The muscle was teased out and cut at the proximal end, including the knee, to avoid any damage to the muscle fibers.
Fixation
Six different ice-cold fixatives were used in this study: 0.2% glutaraldehyde, 4% paraformaldehyde, 95% ethanol, 100% isopropanol, 100% methanol, and methanol:DMSO (4:1). PBS was used for dilutions of glutaraldehyde and paraformaldehyde solutions. Water was used for dilution of ethanol solution. The culture cells were fixed in the six fixatives individually for 5 min at 4C. The diaphragm and TA muscles were fixed in the six fixatives individually with gentle rocking to assist with the penetration of the fixative for 30 min and 20 min at 4C, respectively.
LacZ Staining and Evaluation
The cultured myoblasts or muscle tissues were washed three times in the lacZ staining buffer, 0.1 M sodium phosphate buffer (pH 7.2), 2 mM MgCl2, 0.01% sodium deoxycholate, and 0.02% Nonidet-P40 immediately after fixation. The samples were transferred to lacZ staining solution, 0.1 M sodium phosphate buffer (pH 7.2), 1 mg/ml of 5-bromo-4-chloro-3-indolyl β-D-galactoside (X-gal), 2 mM MgCl2, 5 mM potassium ferrocyanide, 5 mM potassium ferricyanide, 0.01% sodium deoxycholate, and 0.02% Nonidet-P40 at 37C for 3 hr for cells and overnight for tissues. The intensity quantification from the lacZ staining was performed as follows. Representative images from each lacZ-stained culture cell were captured using the same microscope and camera settings. The captured images were inverted in Image-J software. A circular selection was placed over each cell, and the mean intensity for each nucleus was measured. The size of the area measured was kept the same in all the samples. The values were normalized by the background containing no cells. At least 100 cells were analyzed for each group.
Immunostaining
Cultured cells were fixed and lacZ stained using the fixatives described in the Results section. After lacZ staining, cells were rinsed with washing buffer (PBS with 0.01% Triton X-100). Cells were permeabilized by immersion in 0.2% Triton X-100 in PBS for 10 min. After incubation in the blocking solution (1% bovine serum albumin in PBS) for 30 min, cells were stained with antisarcomeric myosin heavy chain (MHC) antibody (MF20: Developmental Study Hybridoma Bank, Iowa City, IA; 1:20 dilution) followed by biotin-conjugated anti-mouse-IgG antibody (Vectastain, Vector Labs, Burlingame, CA; 1:400 dilution), each diluted in the blocking solution for 1 hr. Then, cells were incubated with the Vectastain Elite ABC Kit (Vectastain), followed by colorization using 3-Amino-9-ethylcarbazole (AEC) chromogen (Sigma-Aldrich) solution (0.05% AEC, 0.015% H2O2 in 0.05 M acetate buffer, pH 5.0). Nuclear counterstaining was performed with 4′,6-diamidino-2-phenylindole (DAPI). Microscopic images were captured by a DP-1 digital camera attached to a BX51 fluorescence microscope with 20× and 40× UPlanFLN objectives (all from Olympus; Center Valley, PA). Photoshop 9.0 (Adobe Systems; San Jose, CA) was used for image processing.
Single Muscle Fiber Isolation
Single muscle fiber isolation from the fixed muscle tissue was performed by a modified protocol from Collins and Zammit (2009) as previously described. The dissected whole-muscle tissue was fixed, rinsed, and lacZ stained as described above. The stained muscle tissue was then rinsed three times with PBS and once with DMEM. The lacZ-stained muscle tissue was digested in a solution of collagenase type I (0.2%, Sigma-Aldrich) at 37C until the muscle tissue had hair-like single fibers projecting from it. For the TA muscle, muscle was periodically triturated and incubated for 90 min. For the diaphragm muscle, the central tendon in the diaphragm disappears after a 10- to 20-min incubation; the tissue was trimmed and further digested for 10 to 20 min to facilitate the release of the single muscle fibers. After sufficient digestion, the collagenase was inactivated by DMEM supplemented with 2% FBS. The fibers were teased out of the fiber bundles by gently titrating with heat-polished, serum-coated Pasteur pipettes with decreasing bore sizes into serial culture plates. Fiber bundles and individual fibers were transferred to PBS to allow for visualization of the lacZ staining.
Statistics
All data are expressed as mean ± SEM. A repeated-measures two-way ANOVA was used to determine differences among experimental groups with time as the repeated factor. If an interaction existed, the Bonferroni post hoc measures were used to determine differences. Values of p<0.05 were considered significant with post hoc comparisons.
Results
Myf5, a myogenic regulatory factor, is a great marker for tracing satellite cells and their progenies, myogenic precursor cells. The Myf5+/nLacZ mice carry nuclear β-galactosidase gene inserted in Myf5 gene locus, where the β-galactosidase gene expression recapitulates the expression of endogenous Myf5 in both satellite cells and myogenic precursor cells (Tajbakhsh et al. 1996; Beauchamp et al. 2000). Myf5+/nLacZ mice were then used to test the efficiency of lacZ staining for satellite cell-derived myoblasts and muscle tissues. Myoblasts were isolated and cultured from the Myf5+/nLacZ mouse skeletal muscle. Cells were then fixed with aldehyde (glutaraldehyde and paraformaldehyde) and alcohol (isopropanol, ethanol, and methanol) fixatives followed by X-gal staining for β-galactosidase activity. The results are summarized in Table 1 and Figure 1A. Figure 1B shows the quantification of the staining intensity normalized to 0.2% glutaraldehyde because this method has been routinely used as the standard (Ma et al. 2002). When cells were fixed with 4% paraformaldehyde (PFA) or 95% ethanol, lacZ staining detected in myoblast nucleus was considerably weaker than the control (69.1 ± 7.8% or 44.3 ± 3.7%, respectively). Importantly, 100% isopropanol showed the strongest lacZ staining intensity in myoblasts (130.3 ± 17.7%). By contrast, 100% methanol and methanol:DMSO (4:1; data not shown) fixation resulted in no lacZ+ staining.
Summary for Different Fixatives Used for Double Staining
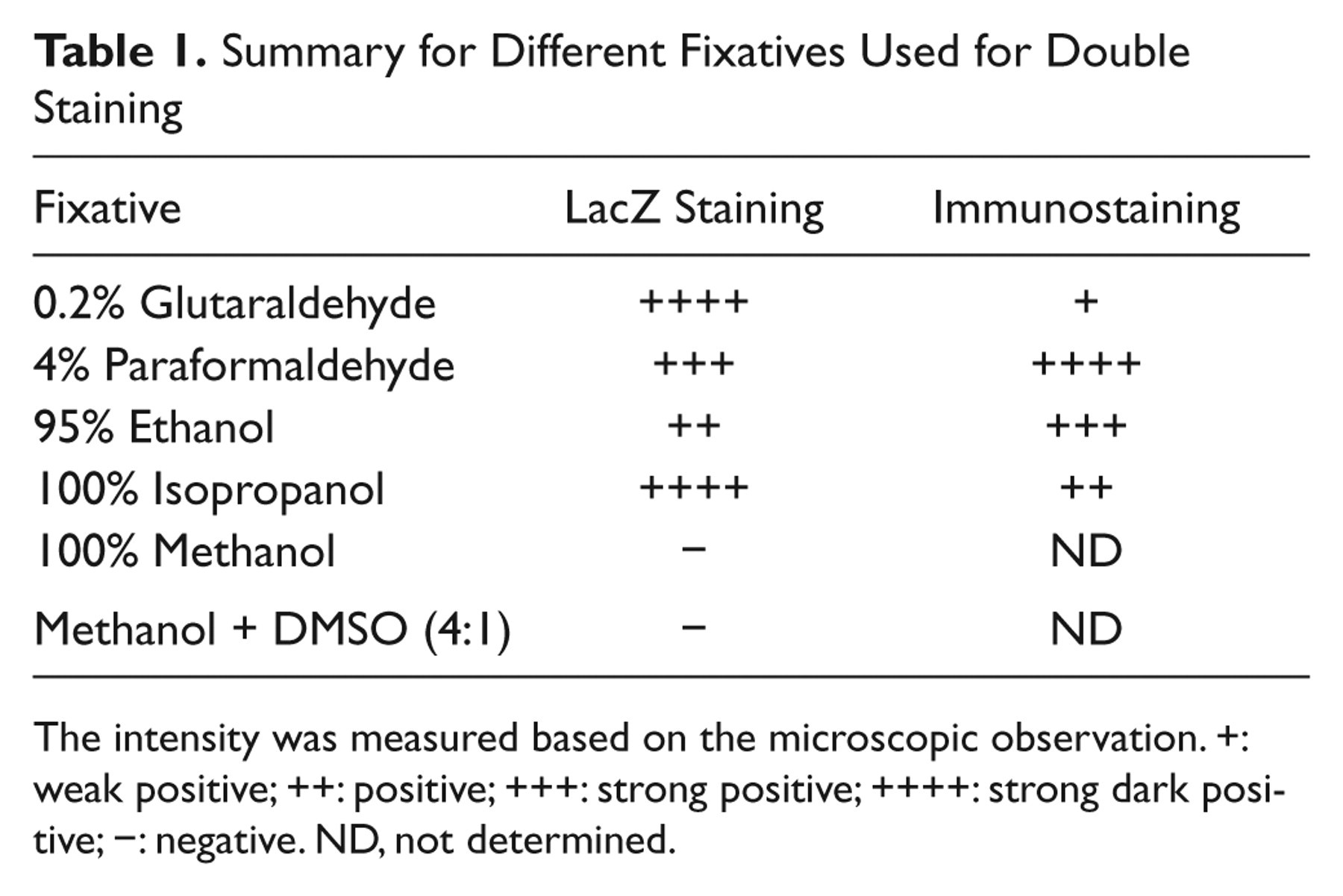
The intensity was measured based on the microscopic observation. +: weak positive; ++: positive; +++: strong positive; ++++: strong dark positive; −: negative. ND, not determined.
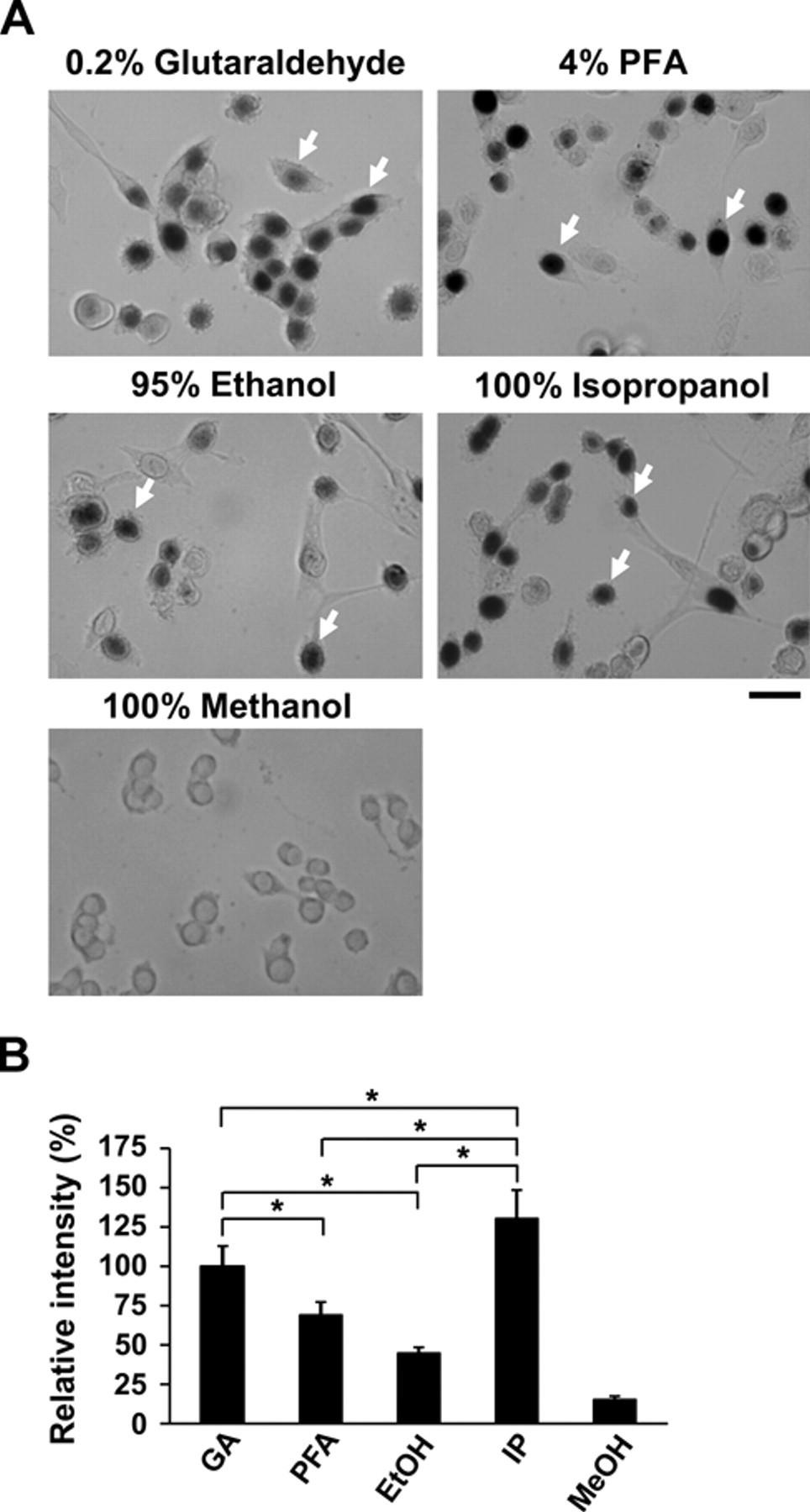
Different fixatives used for lacZ staining in myoblasts. (A) Myoblasts from the Myf5+/nLacZ mice were isolated and cultured. The cells were fixed with different fixatives followed by lacZ staining. Arrows show lacZ-positive myoblasts. (B) LacZ staining was quantified by Image-J, and relative intensities were compared. GA, 0.2% glutaraldehyde; PFA, 4% paraformaldehyde; EtOH, 95% ethanol; IP, 100% isopropanol; MeOH, 100% methanol. Asterisks indicate experimental pairs where differences between the compared values were statistically significant (p<0.05) by ANOVA test. Bars indicate 25 µm.
To further characterize the fixatives, immunohistochemical staining was performed after lacZ staining for the fixatives that were able to stain for β-galactosidase activity in myoblast culture. After lacZ staining, the cells were stained for sarcomeric MHC, a marker for differentiating myocytes. These MHC+ cells were elongated and/or multinucleated and did not express lacZ in their nucleus. By contrast, MHC− myoblasts displayed round shape and expressed lacZ in their nucleus (Fig. 2). Although 4% PFA yielded the best results for immunostaining, cells fixed with 100% isopropanol and 95% ethanol also performed efficiently. However, cells fixed with 0.2% glutaraldehyde contained many lacZ+ myoblasts that were nonspecifically stained with the anti-MHC antibody.
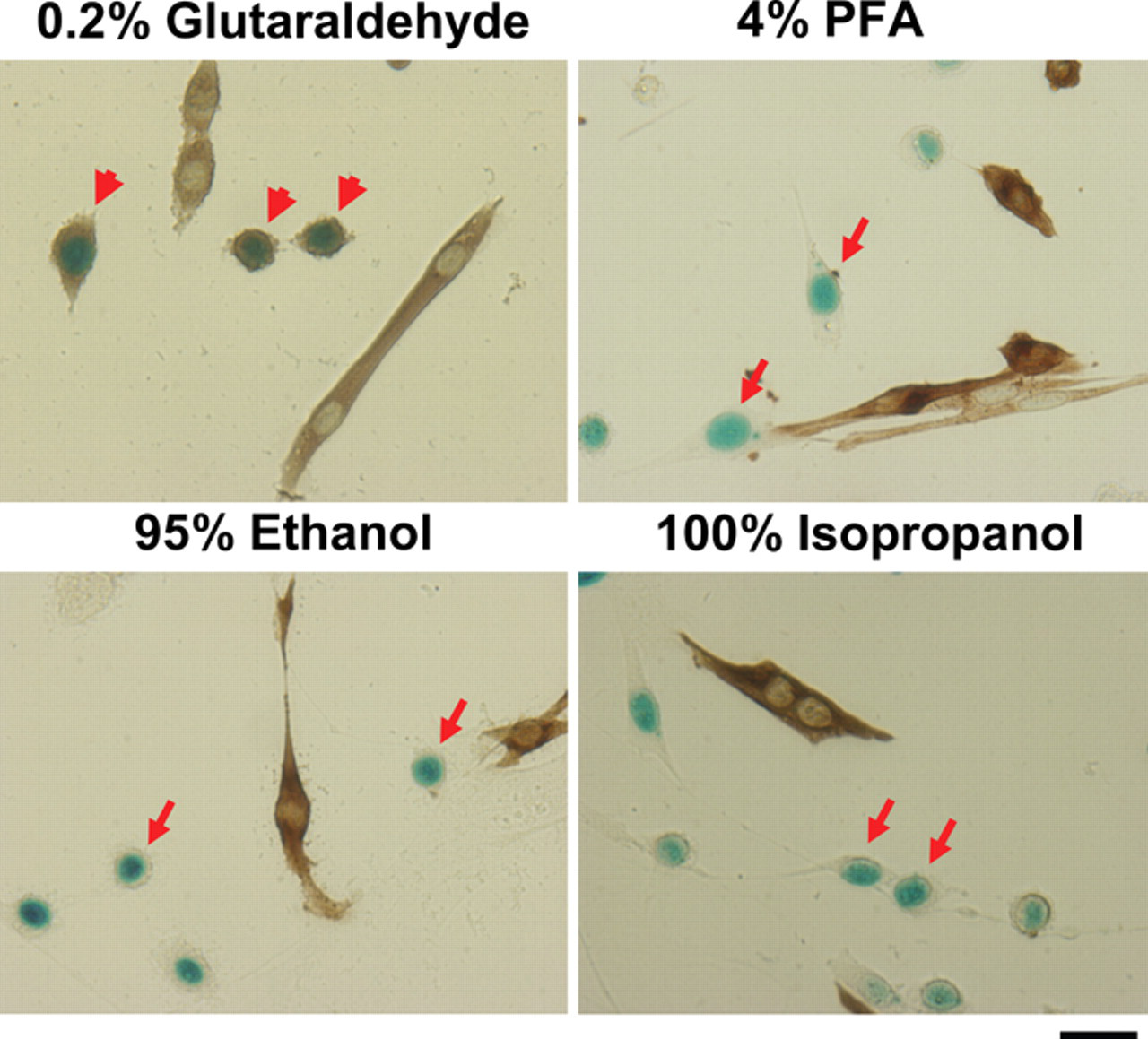
Different fixatives used for double staining of lacZ and antibody. Myoblasts isolated from Myf5+/nLacZ mice were cultured and fixed with different fixatives followed by lacZ staining and immunostaining with antisarcomeric myosin antibodies (brown color). Arrows show lacZ+ myoblasts. Arrowheads show lacZ+ myoblasts nonspecifically stained with anti–myosin heavy chain (MHC) antibody. Bars indicate 25 µm. PFA, paraformaldehyde.
Next, diaphragm and TA muscles were dissected, fixed, and stained for β-galactosidase activity (Fig. 3A). Although 0.2% glutaraldehyde- and 4% PFA-fixed tissues had the stronger intensity, 95% ethanol- and 100% isopropanol-fixed muscles displayed more lacZ+ cells in whole-mount tissues due to bleaching effects. Both 0.2% glutaraldehyde- and 4% PFA-fixed diaphragm muscle failed to disassociate even with collagenase treatment after 4 hr. By contrast, 100% isopropanol- and 95% ethanol-fixed tissues were readily disassociated. In the diaphragm muscle, the central tendon became transparent and eventually disappeared compared to the 0.2% glutaraldehyde- and 4% PFA-fixed tissue, which remained white and hardened. After incubation, the fibers started projecting outwards from both diaphragm and TA muscle tissues, suggesting the connective tissue was successfully digested (Fig. 3B). Then, both muscle tissues were triturated to yield rigid single muscle fibers. The fibers were brittle and, therefore, triturated along the fiber length to avoid breaking the fibers into smaller pieces. These isolated single muscle fibers did not become hypercontracted, which is a common problem associated with isolating fibers from unfixed tissue (Collins and Zammit 2009). Using this technique, it was possible to isolate single muscle fibers that retained lacZ+ cells in the satellite cell compartment. Therefore, isopropanol and ethanol fixatives may allow isolation of single muscle fibers from any postfixed muscle following lacZ staining.
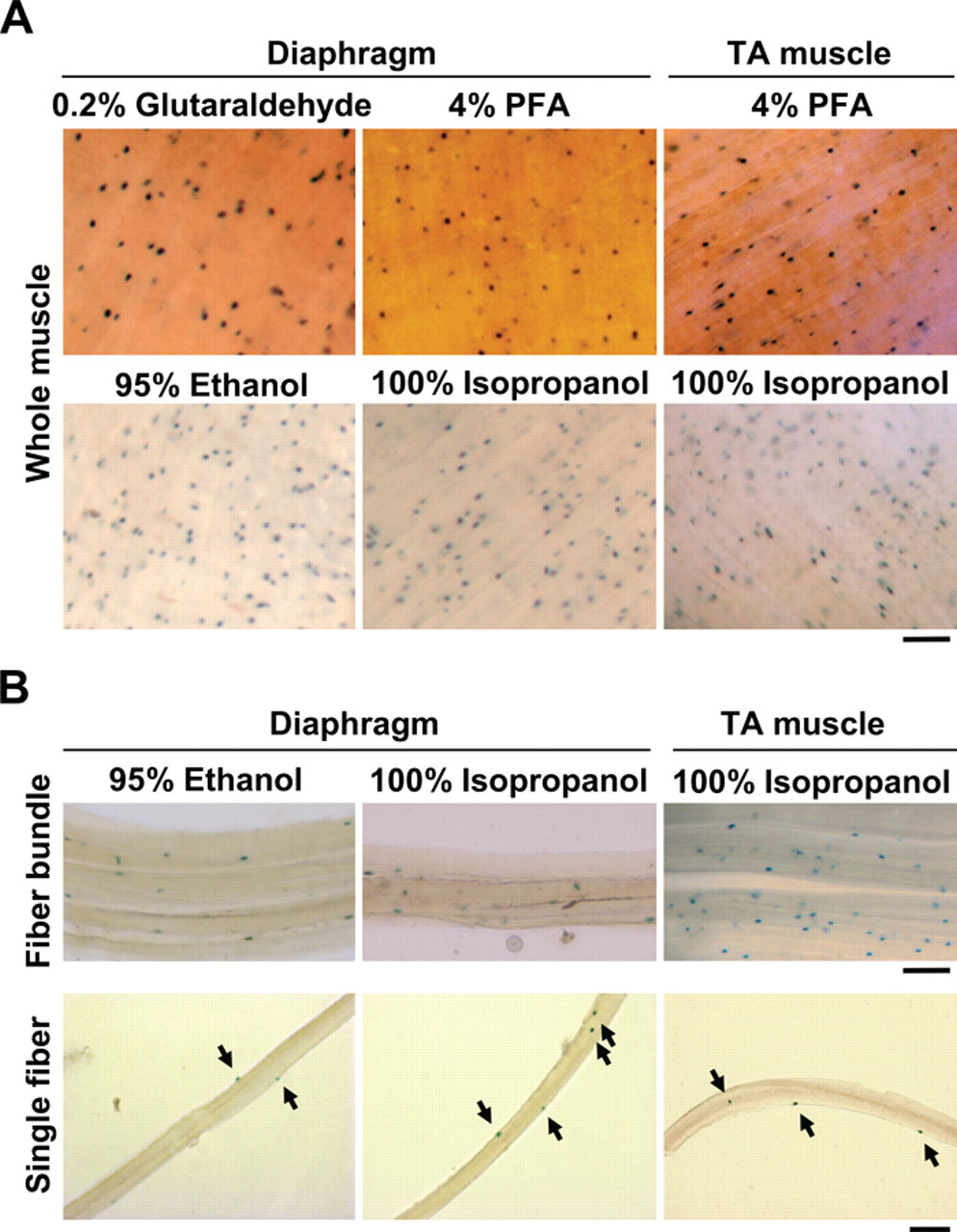
Single muscle fiber isolation after using different fixatives for lacZ staining. (A) Diaphragm muscle isolated from Myf5+/nLacZ mice was fixed with different fixatives followed by lacZ staining (whole muscle). (B) After lacZ staining, diaphragm muscle was digested with collagenase to dissociate to muscle bundles and subsequently single muscle fibers. Arrows show lacZ-positive satellite cells on isolated single muscle fibers. Bars indicate 100 µm. PFA, paraformaldehyde; TA, tibialis anterior.
Last, we examined whether the lacZ detection protocol using 100% isopropanol is as sensitive with other transgenic mice using promoters other than Myf5 as drivers for β-galactosidase expression. The whole diaphragm and TA muscle dissected from MD6.0-lacZ, Flk-1-lacZ, and ROSA26 mice were fixed with 4% PFA or 100% isopropanol followed by lacZ staining. The MD6.0-lacZ mice carry MyoD 6-kb upstream fragment-driving nuclear lacZ transgene and express β-galactosidase in myonuclei in myofibers (Asakura et al. 1995). The Flk-1-lacZ mice carry the β-galactosidase gene inserted in the Flk-1 gene locus and express β-galactosidase in the cytoplasm of endothelial cells and developing hemangioblasts (Ema et al. 2006; Verma, et al. 2010). The ROSA26 mice carry the β-galactosidase gene inserted in the ROSA26 locus and ubiquitously express β-galactosidase in the cytoplasm of all cell types (Zambrowicz et al. 1997). Similar to the results obtained from Myf5+/nLacZ mice, all muscle tissues were stained at a higher intensity for lacZ when 100% isopropanol was used compared to 4% PFA (Fig. 4), indicating that 100% isopropanol fixative gives sufficient staining intensity for β-galactosidase activity regardless of the cell types or the intracellular β-galactosidase localization.
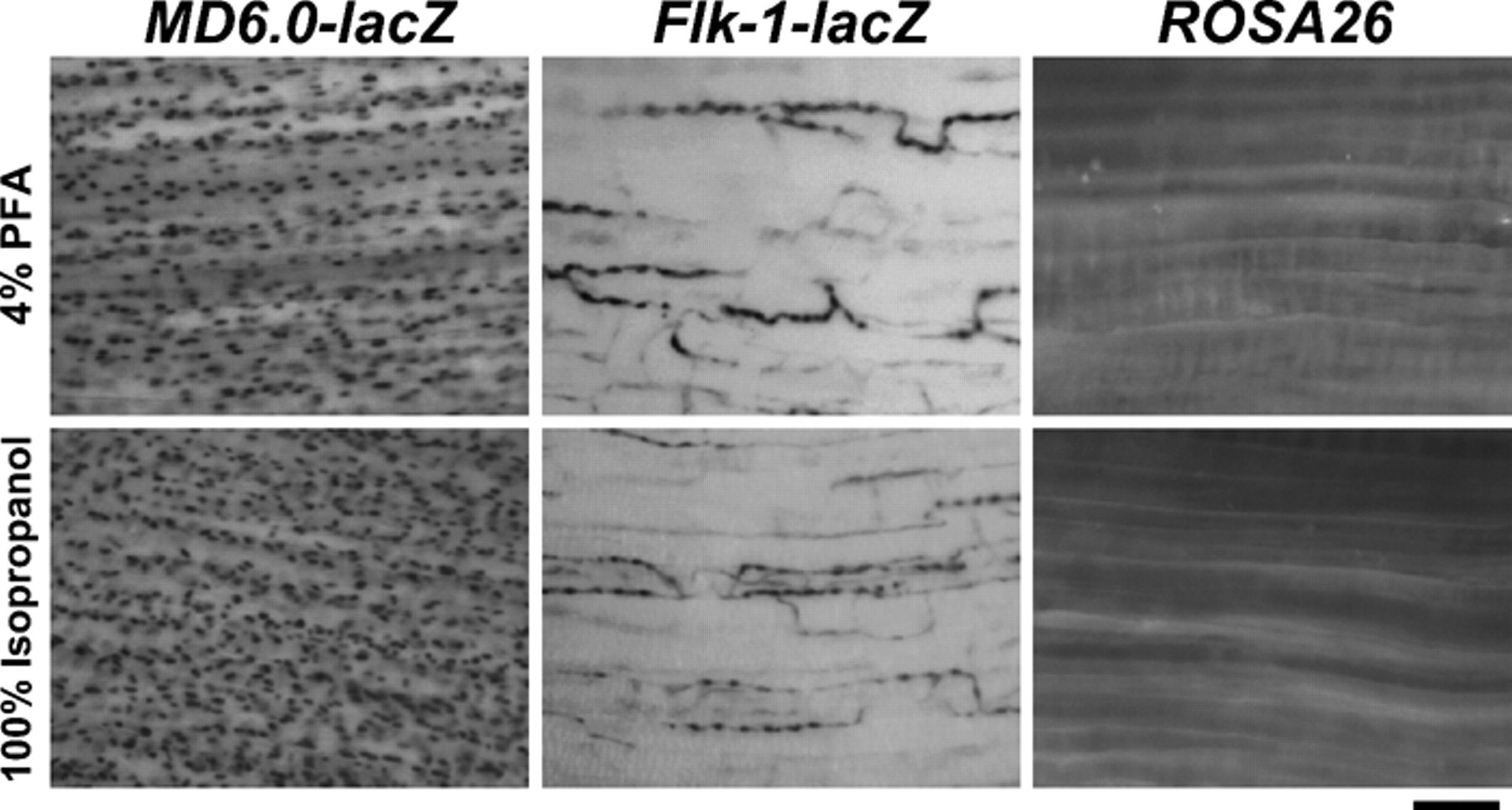
Different transgenic lines for lacZ staining following 4% paraformaldehyde (PFA) or 100% isopropanol fixation. Tibialis anterior muscles isolated from MD6.0-lacZ, Flk-1-lacZ, and ROSA26 mice were fixed with 4% PFA or 100% isopropanol followed by lacZ staining (whole muscle). Bar indicates 100 µm.
Discussion
In this report, we compared the efficiency of staining for β-galactosidase expressing in myoblasts and muscle tissues, after fixation with several different fixatives. In addition, we examined whether myoblasts could be immunostained following lacZ staining postfixation with a variety of fixatives. Using the methods described in this report, we have developed a system for isolating and analyzing single muscle fibers from the diaphragm and TA muscles. This protocol can be adapted for other muscles with modifications to ensure the appropriate fixation and digestion times.
It is known that aldehydes form covalent bonds among amino acid side chains, thereby cross-linking and stabilizing the proteins with the cytoskeleton (Leong and Leong 2007). Although this cross-linking preserves tissue and cell morphology with minimal shrinking, it also inhibits the degradation of the collagen connective tissue, making it impossible to release the single muscle fibers. In addition, aldehyde fixatives also have a masking effect on many antigens, which hinders immunohistochemical identification. By contrast, alcohols such as ethanol and isopropanol are protein precipitants that cause coagulation and aggregation of the proteins. This leads to improved immunostaining of cytoskeletal proteins (Battifora and Kopinski 1986; Troiano et al. 2009). Although ethanol is frequently used as a fixative, we show that isopropanol may also be a better fixative in the case of lacZ staining due to improved signal intensity with both β-galactosidase as well as immunostaining. In addition, muscle tissue fixed in isopropanol can be used to isolate single fibers to analyze satellite cells.
β-galactosidase reporter transgene has been extensively used for characterizing developmental processes for specific cell differentiation and developmental biology (Asakura et al. 1995; Tajbakhsh et al. 1996; Zambrowicz et al. 1997; Beauchamp et al. 2000; Ema et al. 2006) as well as labeling donor cells for transplantation experiments (Heslop et al. 2001; Asakura et al. 2002; Collins and Zammit 2009; Boldrin et al. 2009). The detection of the reporter gene with the appropriate transgene can be used to identify the amount of donor cells that occupy the satellite cells compartment after transplantation of myogenic precursor cells into any muscle tissues, including TA muscle and diaphragm.
Previously, we demonstrated that satellite cell–derived myoblasts lacking MyoD are able to engraft better as well as contribute to the satellite cell compartment after cell injections compared to wild-type cells (Asakura et al. 2007; Hirai et al. 2010). Therefore, it is important to analyze the ability of these cells to contribute to the satellite cell pool after transplantation. Here, we offer an experimental scheme for using the technique established in this article. Myf5+/nLacZ mouse-derived myogenic cells will be delivered to the TA muscle by intramuscular injection. The muscle tissue will be dissected one month after cell delivery and subjected to fiber isolation described in this article following lacZ staining. The total number of lacZ+ engrafted cells located underneath basal lamina will be counted from each single muscle fiber preparation. The number is a direct enumeration of the amount of cells that engraft and contribute to the self-renewing satellite cell pool. Our lacZ staining for whole-muscle tissue more readily allows us to isolate all lacZ+ single muscle fibers without losing the rare population of engrafted lacZ+ single muscle fibers. Taken together, our modified lacZ staining techniques can be broadly used for evaluation of lacZ-expressing cells in tissues for developmental biology, including embryos, as well as for cell transplantation experiments in skeletal muscle and other systems, including mdx muscular dystrophy model mice.
Footnotes
Acknowledgements
We thank Dr. Shahragim Tajbakhsh for providing Myf5+/nLacZ mice, Dr. Masatsugu Ema for providing Flk-1-lacZ mice, Yoko Asakura for genotyping, and Christopher Tastad for critical reading of the manuscript.
The authors declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The authors disclosed receipt of the following financial support for the research and/or authorship of this article: This study was supported by the Muscular Dystrophy Association (MDA) Research Grant and the Gregory Marzolf Muscular Dystrophy Training Grant.
