Abstract
Carboxyl ester lipase (CEL) is an enzyme that hydrolyzes a wide variety of lipid substrates, including ceramides, which are known to show inhibitory regulation of pituitary hormone secretion in experimental models. Because no studies on CEL expression in human pituitary and pituitary adenomas have been reported in the literature, we investigated CEL expression in 10 normal pituitary glands and 86 well-characterized pituitary adenomas [12 FSH/LH cell, 17 α-subunit/null cell, 6 TSH cell, 21 ACTH cell, 11 prolactin (PRL) cell, and 19 GH cell adenomas] using IHC, immunoelectron microscopy, Western blotting, and quantitative RT-PCR. In normal adenohypophysis, CEL was localized in GH, ACTH, and TSH cells. In adenomas, it was mainly found in functioning GH, ACTH, and TSH tumors, whereas its expression was poor in the corresponding silent adenomas and was lacking in FSH/LH cell, null cell, and PRL cell adenomas. Ultrastructurally, CEL was localized in secretory granules close to their membranes. This is the first study demonstrating CEL expression in normal human pituitary glands and in functioning GH, ACTH, and TSH adenomas. Considering that CEL hydrolyzes ceramides, inactivating their inhibitory function on pituitary hormone secretion, our findings suggest a possible role of CEL in the regulation of hormone secretion in both normal and adenomatous pituitary cells.
C
One of the most recently reported physiological roles for CEL is its ability to hydrolyze ceramides via a bile salt–independent mechanism (Kirby et al. 2002). The hydrolysis of ceramides lowers their levels in the intracellular compartment and seems to interfere with lipid trafficking in cells (Kirby et al. 2002; Kodvawala et al. 2005). Ceramides are products of sphingolipid turnover and act as lipid second messengers in the regulation of several different biological cell functions, including differentiation, growth suppression, cell senescence, and apoptosis (Mathias et al. 1998). In the pituitary gland, the involvement of ceramides in the fine-tuning of hormone secretion has been suggested by several studies on experimental models (Negishi et al. 1999; Wu et al. 2001; Liu et al. 2002; Chik et al. 2004), and it is conceivable that the regulation of these lipid molecules by lipolytic enzymes, namely CEL, also takes place in the adenohypophysis.
To the best of our knowledge, no CEL protein expression analysis has been previously performed in human pituitary gland and pituitary adenomas. However, consultation of the expressed sequence tag (EST) profiling in the Unigene database (http://www.ncbi.nlm.nih.gov/UniGene/ESTProfileViewer.cgi?uglist=Hs.533258) reveals a high CEL gene expression in the pituitary gland. Based on this, we aimed to characterize CEL expression in the different cell types of the normal human pituitary gland and in a series of 86 well-characterized pituitary adenomas using IHC, immunoelectron microscopy, Western blotting, and quantitative RT-PCR (qRT-PCR).
Materials and Methods
Cases
Ten normal pituitary glands were collected at autopsy from adult patients who had died of non-endocrine disease and who did not display any endocrine disease. Autopsies were performed 24 hr after death. Surgical specimens of 86 well-characterized pituitary adenomas were collected from the files of the Department of Pathology, Ospedale di Circolo, Varese, Italy. The series comprises 12 FSH/LH cell, 17 α-subunit (α-SU)/null cell, 6 TSH cell, 21 ACTH cell, 11 prolactin (PRL) cell, and 19 GH cell adenomas. All tissues were fixed in buffered formalin (formaldehyde 4% and acetate buffer 0.05 M) for 24 hr and routinely embedded in paraffin wax. Five-μm-thick sections were stained with hematoxylin–eosin and Grimelius' Silver Stain for the histopathological examination. All tumors were characterized on the basis of their hormonal expression and associated endocrine symptoms and were classified according to the 2004 WHO classification (DeLellis et al. 2004).
This study was performed in agreement with the clinical standards laid down in the 1975 Declaration of Helsinki and its revision in 1983 and according to the rules of the Ethics Committee of the Ospedale di Circolo.
Immunohistochemistry
IHC was performed using the standard avidin–biotin complex method (Hsu et al. 1981), and the antibodies listed in Table 1 were used. For CEL detection, we used two different antibodies as previously described (La Rosa et al. 2009): the polyclonal anti-CEL antibody raised against a peptide mapping within the internal region of human CEL and the monoclonal anti-BCL10 antibody (clone 331.3) raised against aa 168–233 of the COOH-terminal part of BCL10, which crossreacts with the COOH-terminal portion of CEL. Colocalization studies were performed using double-label immunohistochemical stains as previously described (Mason et al. 1983; Lan et al. 1995). Specificity controls consisted of absorption of antibodies with 10–20 nM of their related antigens, substitution of the primary antibody with non-immune serum of the same species at the same dilution, and use of control tissues with or without the pertinent antigen. Because Santa Cruz Biotechnology (Santa Cruz, CA) does not provide the specific blocking peptide for the antibody directed against the C-terminal portion of BCL10, we performed a Western blotting analysis to check the antibody specificity as previously described (La Rosa et al. 2009).
Immunohistochemical reactions were evaluated semiquantitatively by two independent pathologists (SLR, CC), considering the percentage of positive tumor cells in neoplastic tissue.
Antibodies and antisera used
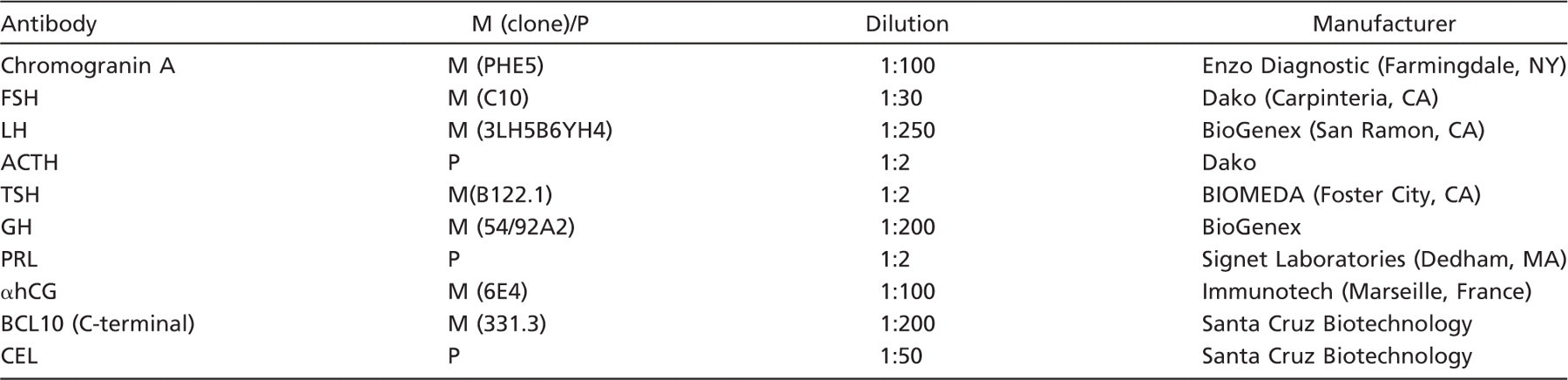
ACTH, adrenocorticotropic hormone; CEL, carboxyl ester lipase; FSH, follicule stimulating hormone; GH, growth hormone; hCG, human chorionic gonadotropin; LH, luteinizing hormone; M, monoclonal; P, polyclonal; PRL, prolactin; TSH, thyroid stimulating hormone.
Immunoelectron Microscopy
For ultrastructural ICC, three normal pituitary glands, two somatotroph adenomas, and one corticotroph adenoma were fixed for 2 hr at 4C in a mixture of 2% paraformaldehyde and 0.5% glutaraldehyde in 0.05 M (pH 7.3) cacodylate buffer and embedded in London White Resin (Polysciences; Warrington, PA). Thin sections, after pretreatment with 1% ovalbumin in 0.05 M TBS (pH 7.4) for 5 min at room temperature, were incubated for 24 hr at 4C with the primary monoclonal anti-BCL10 antibody, then with 18 nm Colloidal Gold-AffiniPure goat anti-mouse IgG (Jackson ImmunoResearch Laboratories, Baltimore, MD) diluted 1:20 for 1 hr at room temperature, and finally counterstained with uranyl acetate and lead citrate. Specificity controls consisted of omission of the first layer, use of tissues with or without pertinent antigens, and substitution of the primary antibody with normal serum and with mouse IgG. For double-label electron microscopy ICC, sections, after incubation with the anti-BCL10 antibody, were incubated with undiluted anti-ACTH antiserum for 24 hr at 4C. Successively, sections were incubated with 18 nm Colloidal Gold-AffiniPure goat anti-mouse IgG diluted 1:20 and with 12 nm Colloidal Gold-AffiniPure donkey anti-rabbit IgG (Jackson ImmunoResearch Laboratories) diluted 1:50 for 1 hr at room temperature. Finally, after counterstaining with uranyl acetate and lead citrate, all thin sections were examined with a Philips (Morgagni 268 D) electron microscope (Eindhoven, The Netherlands).
Electron microscopy immunolabelings were quantified by counting and statistically comparing the number of colloidal gold particles inside and outside secretory granules, as suggested by Mayhew and Lucocq (2008).
In addition to immunoelectron microscopy performed on three cases of pituitary adenomas, we examined the ultrastructural features of 13 of 86 pituitary adenomas.
Western Blotting
For Western blotting analysis, fresh tissues of normal pituitary, salivary gland, lymph node, and pancreas were used. A total protein extract from tissues was prepared by homogenizing ∼0.5 g of tissue in 1 ml of ice-cold PBS with 0.2 mM dithiothreitol and a protease inhibitor cocktail (Roche; Basel, Switzerland). After centrifugation at 12,000 × g for 15 min at 4C, the supernatant was recovered and the protein concentration was quantified by using the Bradford (Bio-Rad; Hercules, CA) method. Protein band immunodetection was performed using the monoclonal anti-BCL10 antibody (dilution 1:1000; Santa Cruz Biotechnology) and the polyclonal anti-CEL antibody (dilution 1:200; Santa Cruz Biotechnology) as primary antibodies and horseradish peroxidase–conjugated secondary antibodies (dilution 1:15,000). Bands were visualized by using enhanced chemiluminescence (ECL Advance Western Blotting Detection Kit; GE Healthcare, Fairfield, CT) as specified by the manufacturer.
Quantitative RT-PCR
qRT-PCR analysis was performed on both fresh (2 normal pituitary glands and 7 adenomas) and formalin-fixed, paraffin-embedded tissues (1 normal pituitary gland and 10 adenomas). Total RNA was extracted and checked for quality from formalin-fixed tissues as previously described (Furlan et al. 2008). Total RNA was prepared from fresh tissues with Tri-reagent (Ambion; Monza, Italy), digested with Turbo DNase (Ambion), loaded in Tris–acetic acid–EDTA agarose gel electrophoresis for quality check, retrotranscribed using the QuantiTect Reverse Transcription Kit (Qiagen; Milan, Italy), and amplified on an ABI Prism 7000 instrument (Applied Biosystems; Monza, Italy) using the TaqMan Universal PCR Master Mix (Applied Biosystems) following the manufacturer's instructions. The following human TaqMan gene expression assays were used: CEL (Hs00426932_m1) and β-actin (Hs99999903_m1). β-Actin expression in each sample was used for normalization. As in preliminary experiments, the efficiencies of each TaqMan gene expression assay were comparable, and the relative quantification of gene expression levels was determined as previously described (Vigetti et al. 2008).
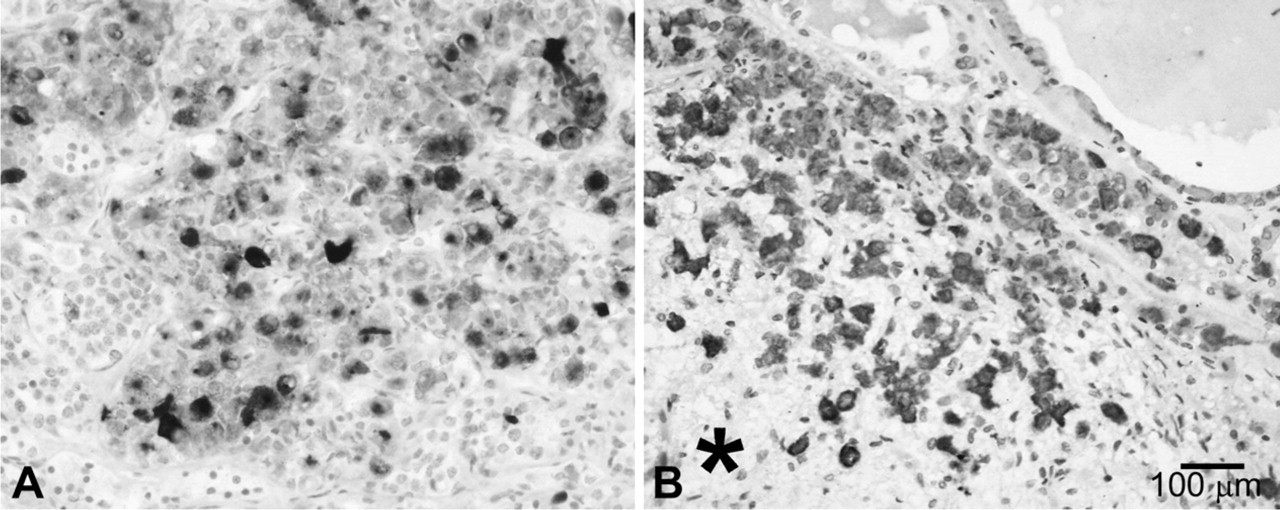
Carboxyl ester lipase (CEL) expression in normal pituitary gland detected using the anti-COOH-terminal BCL10 antibody (
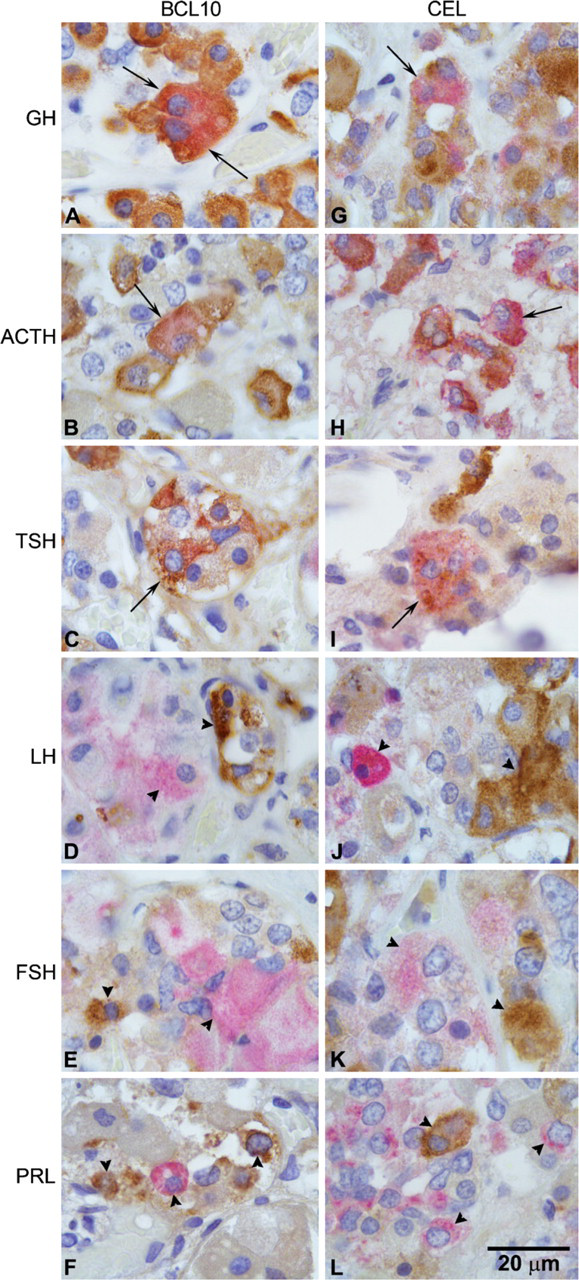
Double-label immunostainings demonstrate that the immunoreactivity for the COOH-terminal part of BCL10 (left column) and for CEL (right column) colocalizes with GH, ACTH, and TSH but not with LH, FSH, and prolactin. (
Statistical Analysis
The different expression of BCL10 and CEL among functioning and silent adenomas and the correlation between CEL expression and clinicopathological features were evaluated using the Fisher's exact test.
According to Mayhew and Lucocq (2008), the distribution of colloidal gold particles was statistically evaluated using the χ 2 analysis.
Results
Several cells of normal adenohypophysis showed an intense cytoplasmic immunoreactivity (IR) for CEL, detected by using both anti-CEL and anti-COOH-terminal BCL10 (clone 331.3) antibodies (Figure 1). The neurohypophysis did not display any CEL IR. Double-label IHC demonstrated that CEL expression was confined to a fraction of GH, ACTH, and TSH cells, whereas the other pituitary endocrine cell types including FSH, LH, and PRL cells were CEL-negative (Figure 2). Western blotting analysis confirmed the specificity of both antibodies used in recognizing CEL protein; indeed, when pituitary protein extracts were loaded, the two antibodies revealed the expected band at 105 kDa corresponding to the molecular mass of CEL (Figure 3).
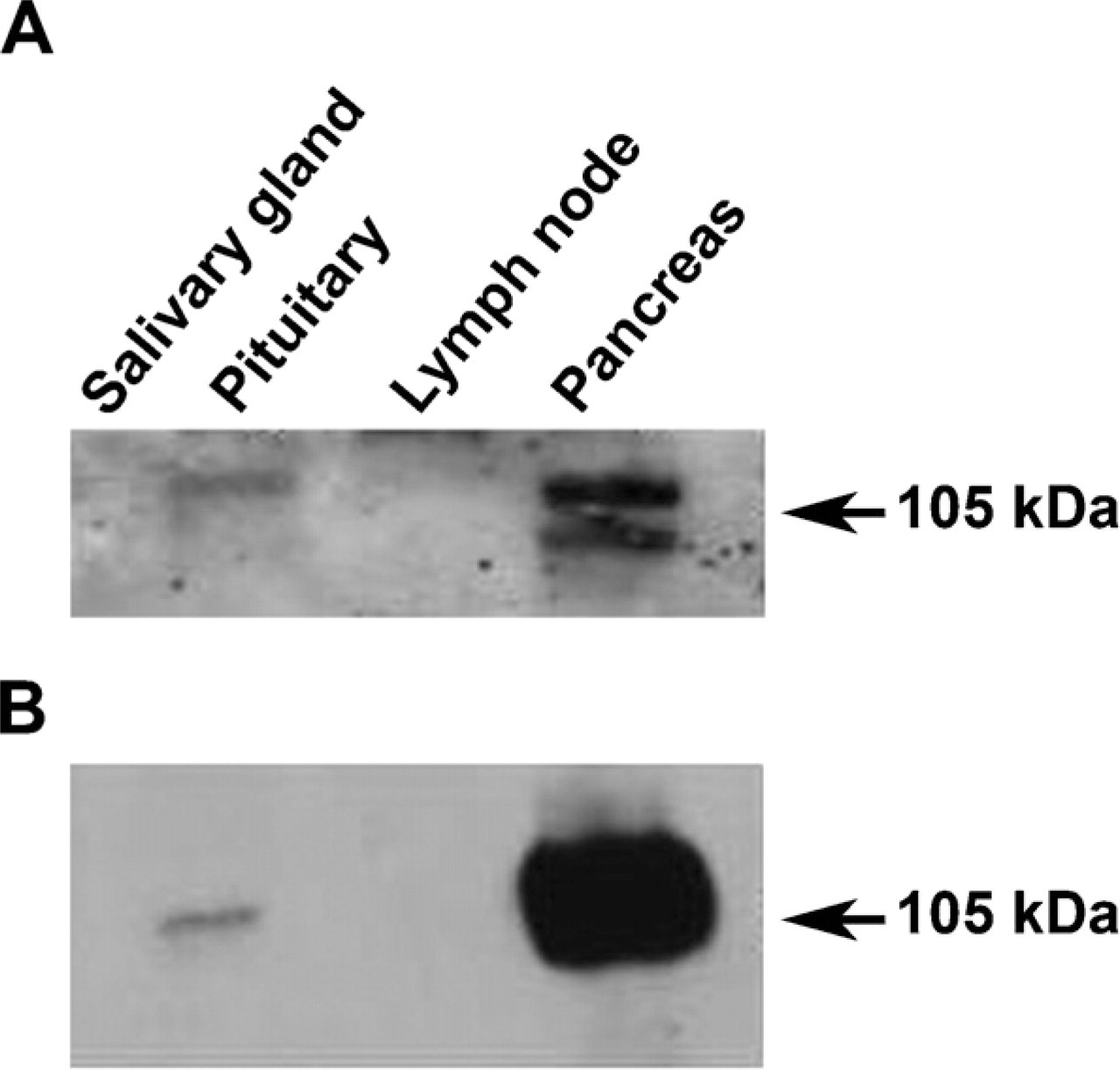
Western blotting analysis of protein extracts from human salivary gland, pituitary, lymph node, and pancreas using the anti-CEL antibody (
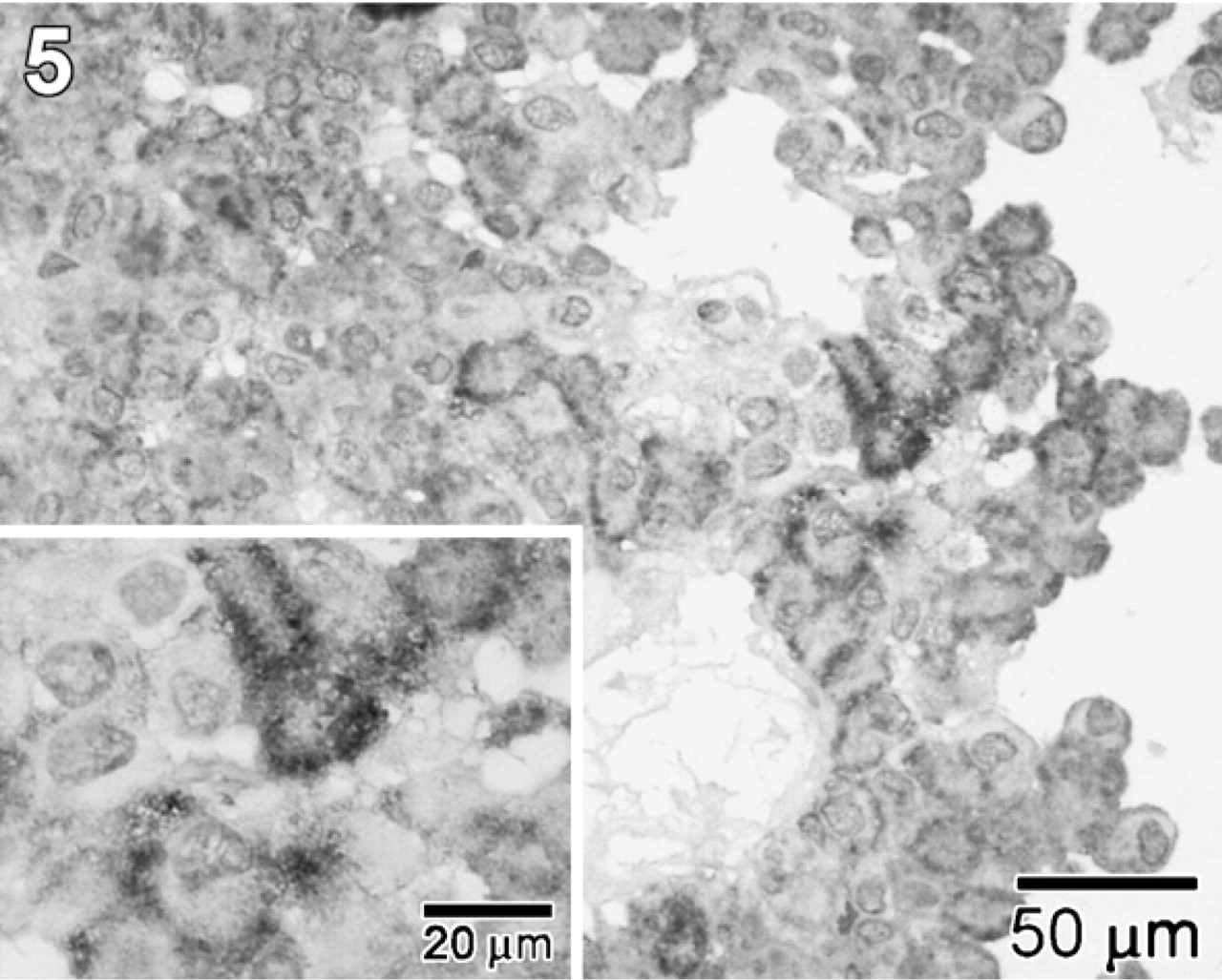
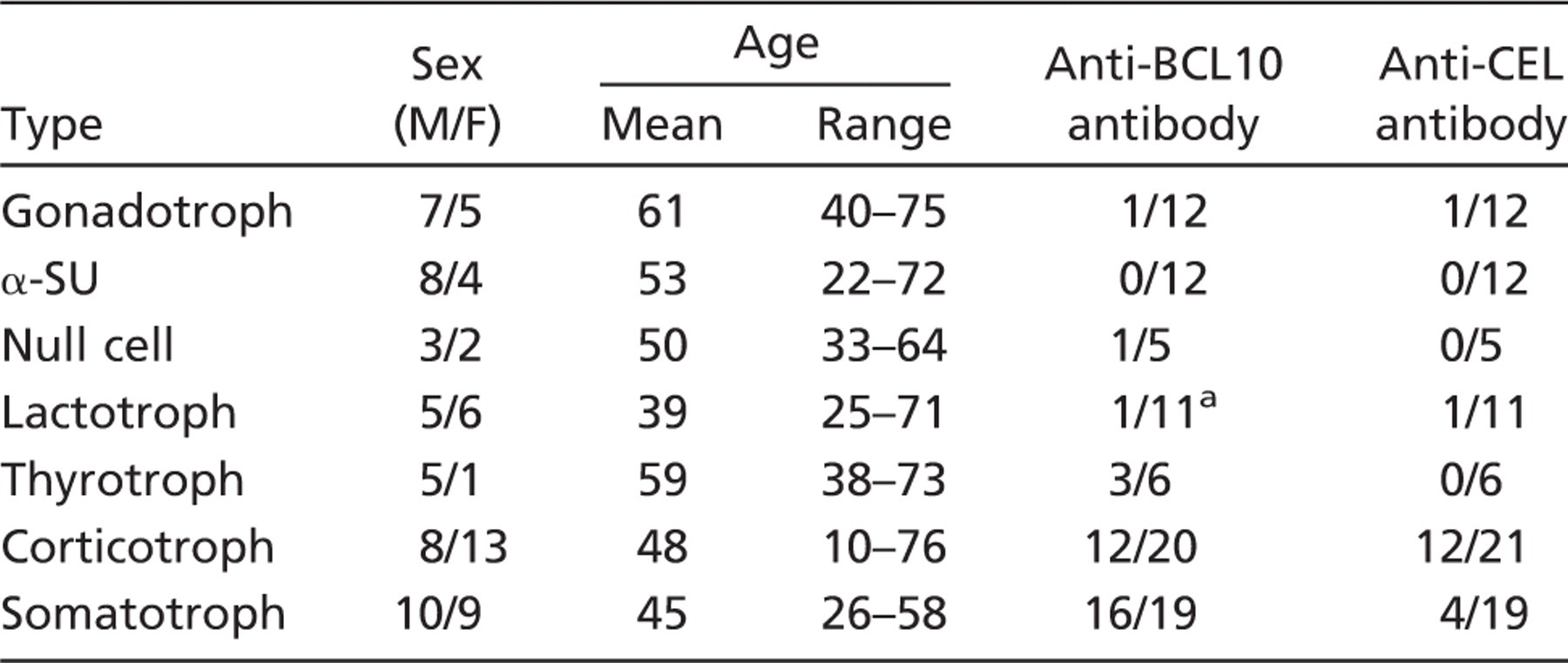
CEL expression was found in the majority of GH (Figure 4), ACTH (Figure 5), and TSH cell adenomas, whereas in gonadotroph, α-SU, PRL, and null cell adenomas, the expression of this enzyme was consistently absent. Actually, with the exception of one gonadotroph and one null cell adenoma showing scattered CEL-positive cells, gonadotroph, α-SU, and null cell adenomas were CEL-negative when tested with the two antibodies. Among 11 PRL cell adenomas, only one case showed a significant number of CEL-immunoreactive cells, whereas in six cases only rare and scattered CEL-immunoreactive cells were found.

When the immunostainings obtained with the two different antibodies used in the study were compared, the anti-BCL10 antibody turned out to be more sensitive than the anti-CEL antibody in identifying the enzyme expression, both in terms of the number of positive cells and in terms of intensity of the reaction. This was particularly evident when we considered GH cell and TSH cell adenomas (Table 2).
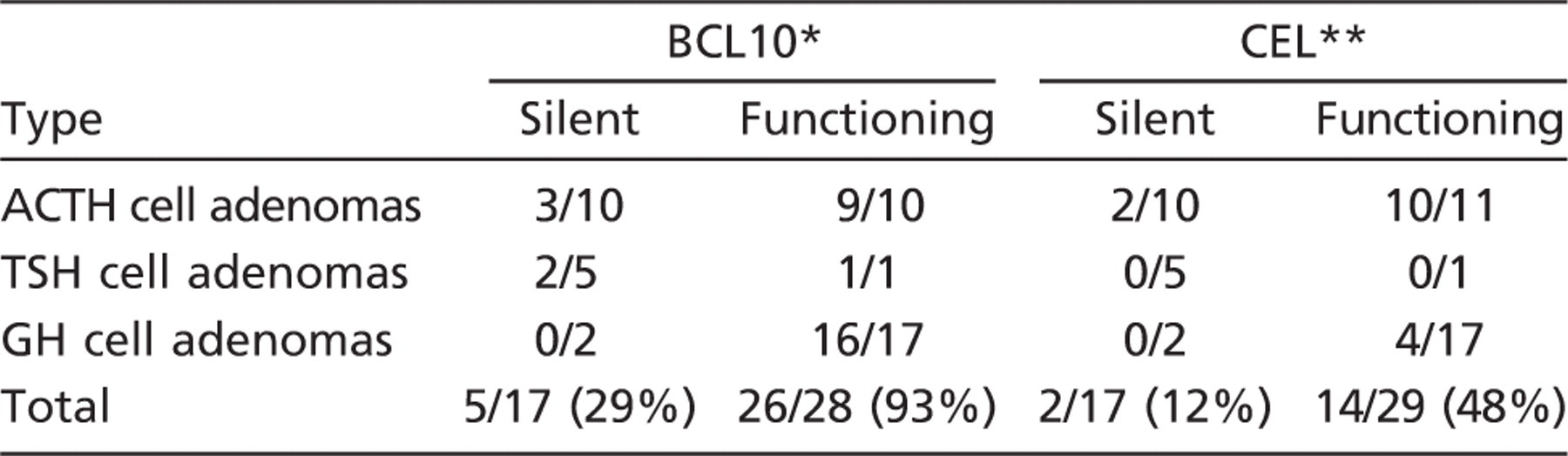
Interestingly, the comparison between the immunohistochemical results and the clinicopathological profile of the adenomas showed that CEL was preferentially expressed in functioning adenomas, with the exception of PRL cell adenomas, whereas the silent ones were almost totally negative (Table 3). Conversely, we did not find any correlation between CEL expression and other clinicopathological features, including sex, age, tumor size, extrasellar extension, mitotic count, and Ki-67 proliferative index (data not shown).
At the ultrastructural level, CEL IR was confined to secretory granules of GH, ACTH, and TSH cells with a mainly peripheral juxtamembrane intragranular distribution, as demonstrated in Figure 6, which shows an example of CEL expression in a normal ACTH cell and in a GH cell adenoma. The nucleus, mitochondria, Golgi apparatus, and other cytoplasmic organelles did not show CEL IR. Quantitative analysis of immunoelectron microscopy labelings demonstrated a statistically significant difference (
In addition, we found a positive relationship between the number of secretory granules, CEL expression, and clinical function in the 13 adenomas ultrastructurally investigated (11 sparsely granulated and 2 densely granulated). In particular, among the 11 sparsely granulated adenomas, only 2 (18%) showed CEL expression and were associated with the endocrine syndrome (hyperthyroidism in one case and acromegaly in the other). Conversely, both densely granulated adenomas (100%) displayed strong CEL expression and clinically evident endocrine syndrome.
CEL expression identified using both anti-COOH-terminal BCL10 and anti-CEL antibodies in the 86 different types of pituitary adenomas
In 6 of 10 cases considered negative, rare scattered positive cells were observed. M, male; F, female; α-SU, α-subunit.
The qRT-PCR results showed a high CEL mRNA expression in both fresh and formalin-fixed samples from normal pituitaries and from GH and ACTH cell adenomas, whereas CEL expression was low or insignificant in the other adenoma types, including FSH/LH, α-SU, and null cell tumors, perfectly in accordance with the immunohistochemical findings (Table 4; Figure 7). It is to be noted that, in terms of threshold cycle (
Discussion
As far as we are able to ascertain, this is the first study demonstrating CEL expression in normal human pituitary gland and in pituitary adenomas using multiple experimental approaches including IHC, immunoelectron microscopy, Western blotting, and qRT-PCR. All techniques used confirmed that CEL is expressed in the normal pituitary gland and in specific types of pituitary adenomas. For immunohistochemical localization, we used two different antibodies recognizing CEL protein: the polyclonal anti-CEL antibody and the monoclonal anti-COOH-terminal BCL10 antibody; the latter recognizes aa 156–205 of the COOH-terminal part of BCL10. This sequence maintains a high degree of homology with aa 564–608 of CEL (La Rosa et al. 2009). The anti-BCL10 antibody seemed more sensitive in detecting CEL in both normal and adenomatous pituitaries; this finding is in line with recent studies demonstrating a high sensitivity of the anti-COOH-terminal BCL10 in identifying CEL in pancreatic acinar cell carcinoma, pancreatic metaplasia of the esophageal and gastric mucosa, and colonic adenocarcinoma showing pancreatic acinar cell differentiation (La Rosa et al. 2009; Chiaravalli et al. 2009). Because these two antibodies have been produced in different species using different immunogens, their different sensitivity in recognizing CEL is not surprising, and it may be due to the fact that they do not exactly recognize the same epitope. The immunohistochemical results were also confirmed by the Western blotting experiments, which clearly showed the presence of a protein, identified with both the anti-CEL and anti-COOH-terminal BCL10 antibodies, having the molecular mass of a previously characterized CEL enzyme. Although we did not perform an ISH study to demonstrate the CEL mRNA in both normal and adenomatous pituitary cells, qRT-PCR clearly demonstrated the presence of the mRNA coding for CEL protein in both fresh and formalin-fixed pituitary tissues showing CEL at the immunohistochemical investigation.
Different CEL expressions, detected using both anti-COOH-terminal BCL10 and anti-CEL antibodies, between silent and functioning pituitary adenomas
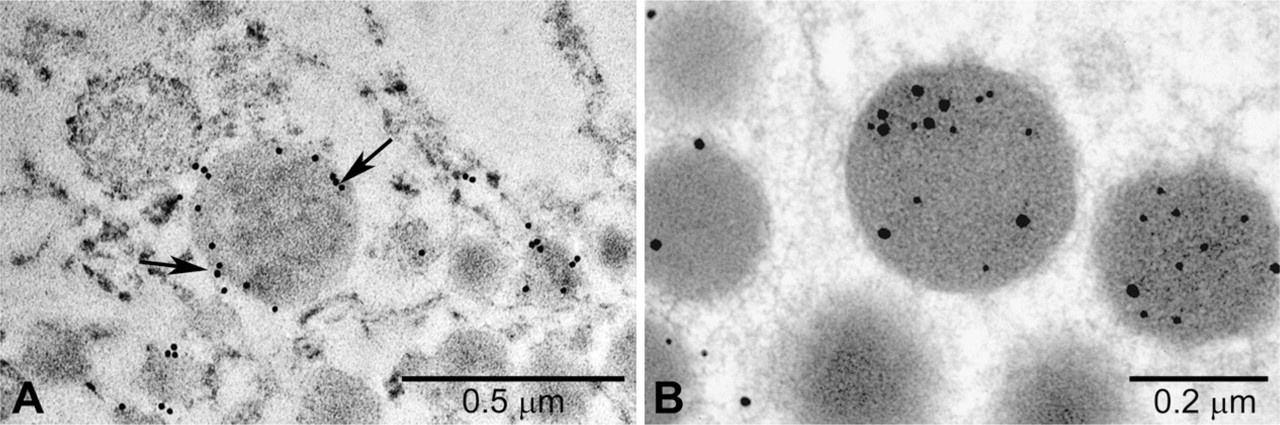
Electron microscopy ICC performed on a GH adenoma (
It is of interest that CEL has recently been found to be able to hydrolyze ceramides, thus inactivating their physiological actions (Kirby et al. 2002). Ceramides are precursors of all glycolipids containing sphingosine (i.e., sphingomyelin, cerebrosides, globosides, sulfatides, and gangliosides) and can be formed by using the de novo pathway or by hydrolysis of sphingomyelin or cerebrosides. CEL is able to degrade ceramides through hydrolysis, forming sphingosine and fatty acid (Hui et al. 1993). This enzymatic activity seems to be the most relevant in vivo, because CEL-knockout mice show an altered chylomicron secretion due to the alteration of ceramide metabolism (Kirby et al. 2002). Moreover, ceramides are potential promoters of Golgi disassembly and are capable of disrupting protein trafficking through intracellular secretory pathways (Rosenwald and Pagano 1993; Fukunaga et al. 2000). It is interesting to recall that ceramides, in addition to several biological functions including differentiation, growth suppression, cell senescence, and apoptosis (Mathias et al. 1998), have recently been found to be able to modulate pituitary hormone secretion in experimental models (Negishi et al. 1999; Wu et al. 2001; Liu et al. 2002; Chik et al. 2004). Ceramides show inhibitory effects by reducing growth hormone-releasing hormone–stimulated GH release through intracellular elevation of Ca2+ in rat anterior pituitary cells (Negishi et al. 1999). Moreover, Chik et al. (2004) demonstrated that ceramide is able to inhibit GH release in GH3 cells through inhibition of the L-channel. They also found that treatment of GH3 cells with sphingomyelinase, which induces the hydrolysis of sphingomyelin and generates ceramide endogenously (Hannun 1994), also causes inhibition of L-channels as observed with ceramide treatment. In addition, Liu et al. (2002) showed that, in GH4 pituitary cells, GH is able to induce the formation of ceramides via a Gαi3/Gβγ-dependent pathway, in the context of an autocrine feedback regulation.
Clinicopathological features and immunohistochemical profile of the 17 adenomas investigated using quantitative RT-PCR (Figure 7)
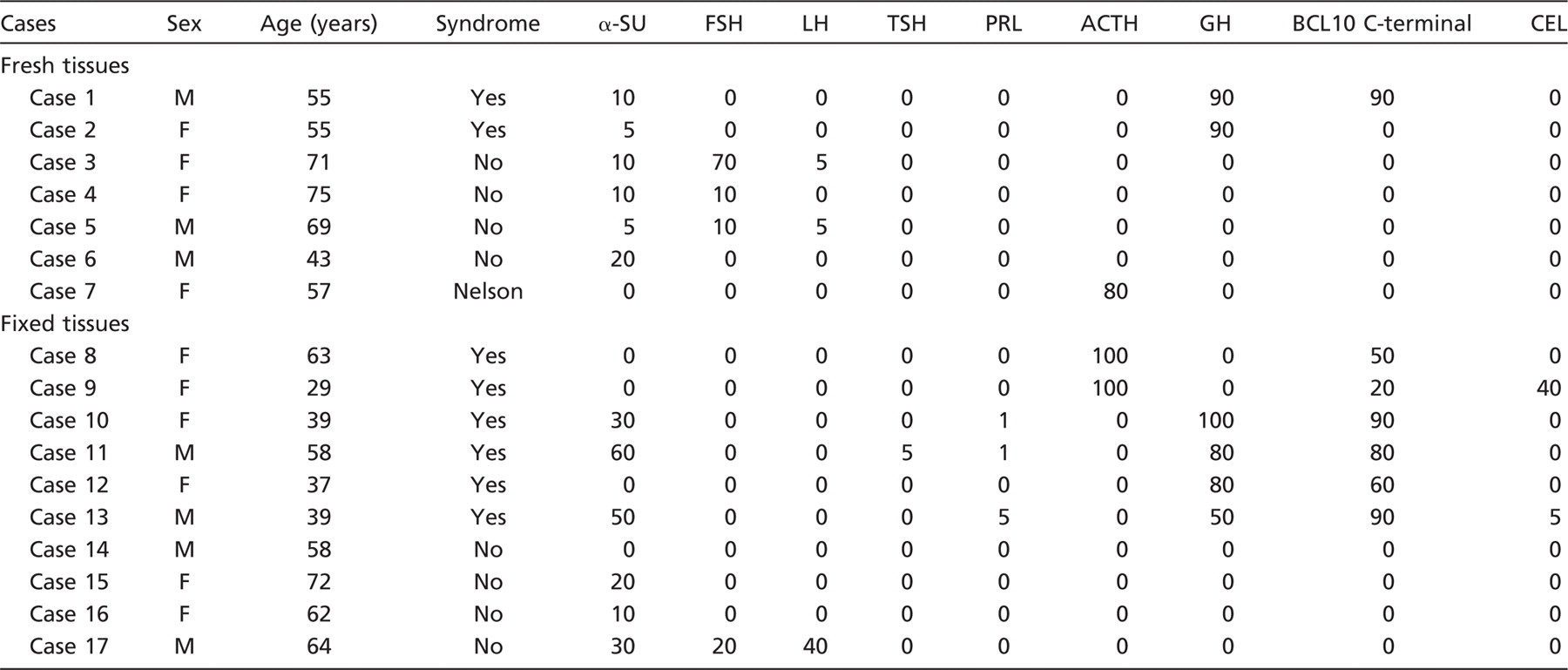
The numbers indicate the percentage of immunoreactive cells.
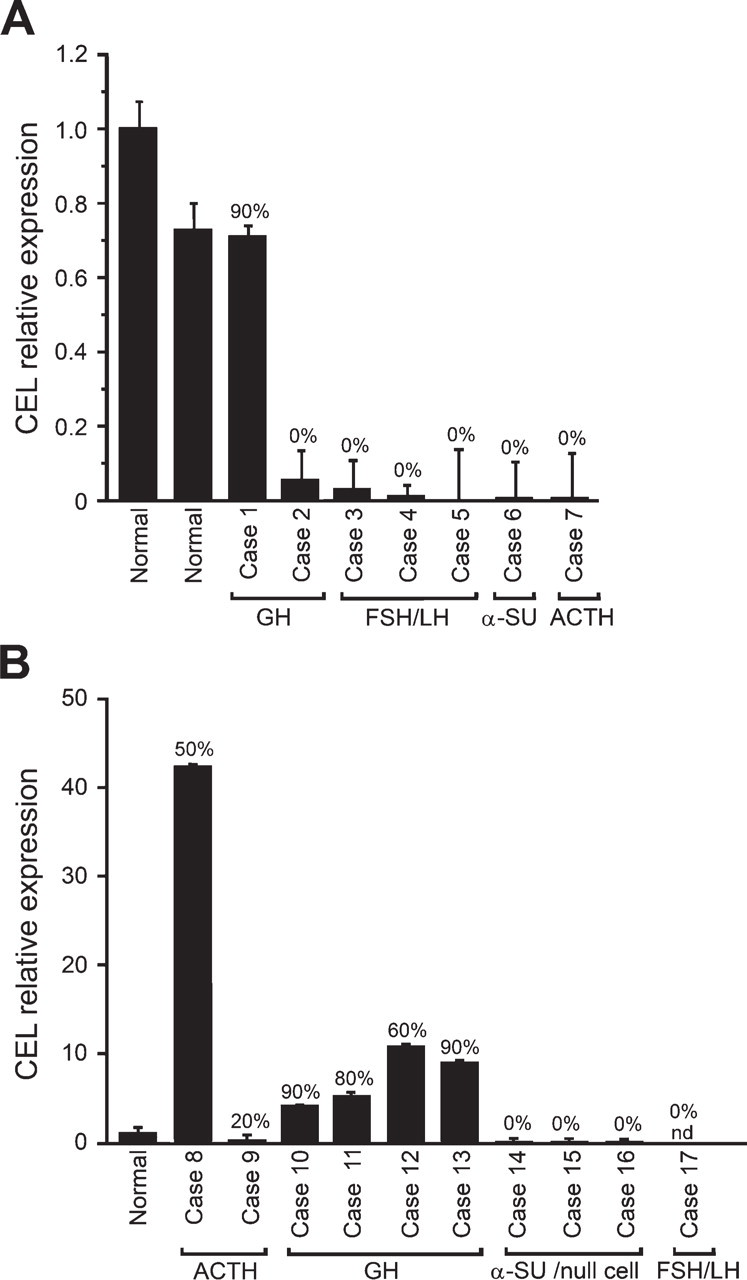
Relative quantification of CEL mRNA by quantitative RT-PCR in frozen (
Considering the link between CEL enzymatic activity and ceramide function and between ceramide and pituitary hormone secretion, our aim was to establish whether normal and neoplastic pituitary cells express CEL. As far as we know, there are no published studies analyzing the expression of CEL in the human pituitary. However, CEL expression is suggested by means of EST profiling in the Unigene database (CEL accession number HS.533258). In the normal pituitary, we have found a specific distribution of CEL in GH, ACTH, and TSH cells, and in adenomas, CEL was mainly expressed by functioning GH, ACTH, and TSH cell adenomas. However, the reason for this specific distribution with restriction to these specific cell types is not known to date. We have found a good correlation of CEL expression between normal and neoplastic GH, ACTH, and TSH cells. This finding suggests that CEL may participate in mechanisms regulating hormone secretion by inactivating ceramide. At the ultrastructural examination, CEL was mainly observed at the periphery of secretory granules close to their lipid-rich membrane, and this is in agreement with the action of this enzyme on lipid structures. This ultrastructural localization of CEL within secretory granules also suggests an enzymatic action on ceramides, which are a component of the lipid membrane and are known to modulate GH release (Negishi et al. 1999). So, it may be speculated that CEL, through the modulation of ceramide content in secretory granule membranes, could regulate hormone release by mediating the fusion of secretory granules with plasma membrane or by modulating Ca2+ concentration in the cytoplasm.
Among tumors, we have found that CEL was preferentially expressed in functioning densely granulated adenomas rather than in silent ones, even if there seemed to be no specific relationship between the clinical characteristics of the endocrine syndrome and the level of CEL expression, neither in terms of number of immunoreactive cells nor in terms of mRNA amount. Taken together, our experiments suggest a possible involvement of CEL in the regulation of pituitary hormone secretion via ceramide modulation. This is not surprising considering that the inhibitory action of ceramides on hormone secretion has been demonstrated in rat somatotroph and GH3 cells (Negishi et al. 1999; Chik et al. 2004) and that CEL inactivates ceramide through hydrolysis (Kirby et al. 2002). In the normal gland, CEL expression could be restricted to a specific functional status as indirectly suggested by the fact that not all GH, ACTH, and TSH cells express CEL. However, further investigations, including functional studies, are needed to verify these hypotheses.
In conclusion, this is the first study demonstrating that human pituitary cells and pituitary adenomas express CEL, which may modulate mechanisms of hormone secretion by inactivating ceramides, which are lipid second messengers able to inhibit pituitary hormone secretion. However, further functional studies are needed to prove this hypothesis, which is based on morphological and molecular biology evidence.
Footnotes
Acknowledgements
This study was supported in part by a grant from the University of Insubria, Varese, Italy.
