Abstract
Neural transdifferentiation is increasingly recognized in neural crest and neural stem cell tumors. Neuronal differentiation has been anecdotally described primarily in somatotroph cell adenomas associated with acromegaly, but its prevalence in adenomas and relationship to adenoma type has not been completely established. In this study we performed a retrospective morphological and immunohistochemical analysis of neurofilament, phosphoneurofilament, Neu-N, class III tubulin, and Hu in WHO grade I pituitary adenomas. Limited numbers of cells with neuronal features and neuron-associated epitopes may be more common in pituitary adenomas than previously recognized. These may occur in many forms of adenomas including somatotroph, lactotroph, mixed somatotroph and lactotroph, null cell/gonadotroph cell and, rarely, corticotroph cell adenomas.
Keywords
T
The presence of ganglion cells in pituitary adenomas has long been recognized in association with growth hormone (GH) or prolactin-secreting adenomas. These nerve cells have been designated as neuronal choristoma (Rhodes et al. 1982; Scheithauer et al. 1983; Horvath et al. 1994; Iwase et al. 1995; Sharma et al. 1999), hamartoma (Horvath et al. 1994; Kroh and Marchel 2000), gangliocytoma (McCowen et al. 1999; Geddes et al. 2000; Pace and Perentes 2001; Kurosaki et al. 2002), or neuronal metaplasia (Scheithauer et al. 1999; Vidal et al. 2002; Moro et al. 2004; Thodou et al. 2004; Kontogeorgos et al. 2006). Rarely, acromegaly may develop in a setting of a GH-secreting pituitary adenoma associated with the presence of neuronal cells with or without associated neuropil. In the one series published to date, 11/15 cases of pituitary adenoma associated with neuronal metaplasia expressed GH (Kontogeorgos et al. 2006). Occasional cases also concomitantly express prolactin (Thodou et al. 2004) and GH or prolactin alone. In these tumors, cells with a neuronal phenotype, sometimes accompanied by neuropil, are found without hypothalamic involvement (Geddes et al. 2000). Recent studies have shown that these neuronal cells express GH and/or prolactin and in one case dopamine receptor mRNA, suggesting that neuronal cells are derived from adenoma cells. Ultrastructural features in some cases are similar to adenomas. Even less frequently, the presence of cells with intermediate or morphological neuronal differentiation has been associated with Cushing's syndrome (Geddes et al. 2000; Vidal et al. 2002). Thus, limited evidence suggests that neuron-like cells are neuroepithelial cells expressing neuronal genes and exhibiting neuronal differentiation. The phenotype of cells with neuronal differentiation is not extensively characterized. One previous study found no neurofilaments in the normal adenohypophysis or adenomas (Halliday et al. 1990), but neurofilament has been seen in others (Kontogeorgos et al. 2006). In the present study we evaluated a series of GH, lactotroph and mixed, null or gonadotroph, and corticotroph cell adenomas for apparent neuronal differentiation and used antibodies to differentiated neural epitopes and epitopes expressed early in neuroembryogenesis to characterize the cells found and to screen for additional expression of neural-associated epitopes.
Materials and Methods
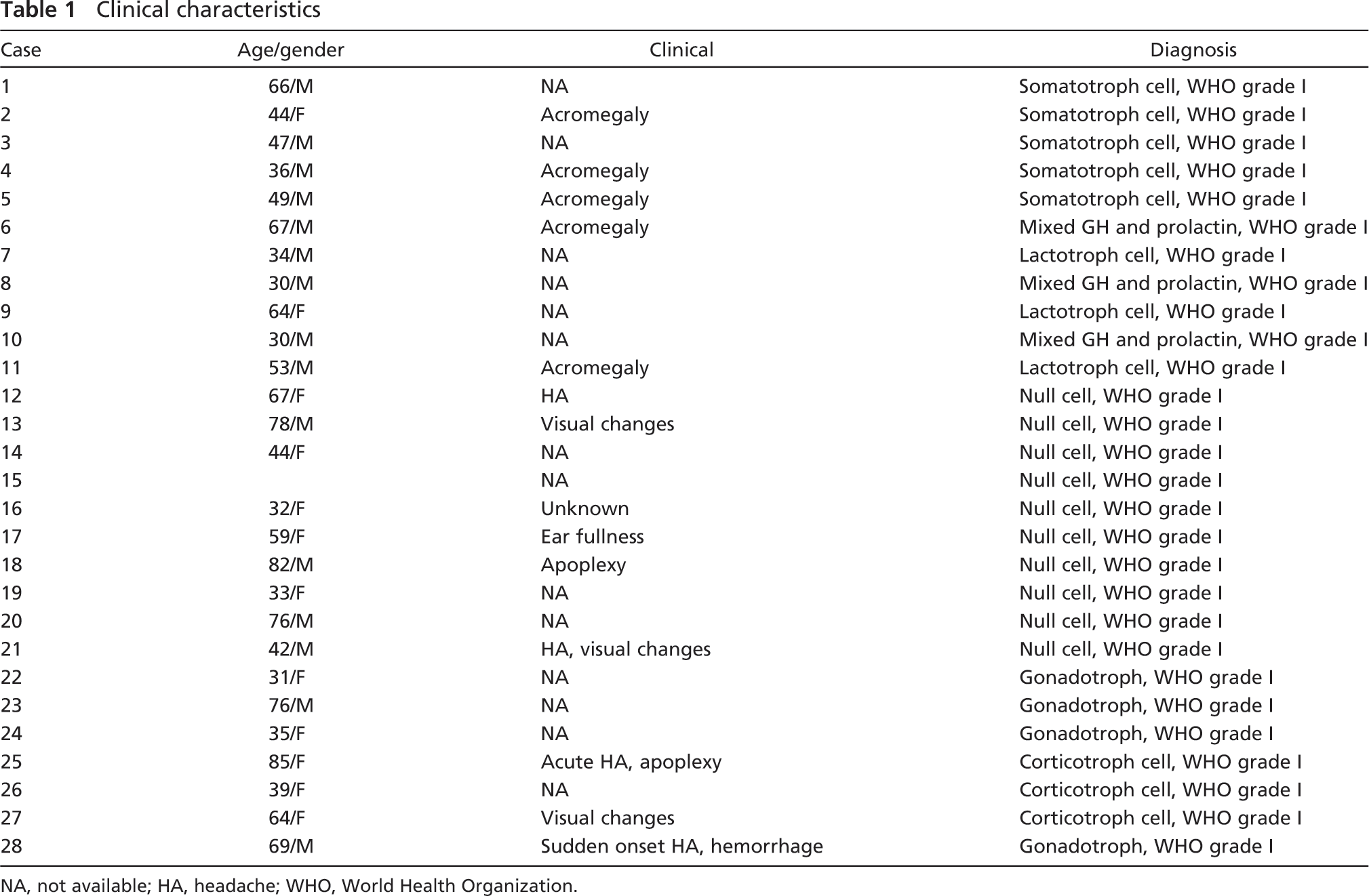
Twenty seven cases classified by World Health Organization (WHO) guidelines (Al-Shraim and Asa 2006) as somatotroph, lactotroph, mixed GH and prolactin cell, null cell, gonadotroph, or corticotroph cell adenoma (WHO grade I) were retrieved from the archives of the University of Rochester Medical Center, or in two cases consultation material was blindly and independently evaluated for morphological and immunohistochemical evidence for neuronal differentiation by two of us (MDJ and XF). In some centers, the latter would be classified as adenomas with neuronal transdifferentiation/metaplasia or neuronal choristoma. Patient characteristics and diagnoses are summarized in Table 1. Immunohistochemistry was performed with monoclonal antibodies to (1) neurofilament (NF, Covance; Berkley, CA), an antibody that reacts with a non-phosphorylated epitope in neurofilament H; (2) phosphoneurofilament (PNF; Covance) that reacts with a phosphorylated neurofilament H and, to a lesser extent, neurofilament M found in neuronal cell bodies (Giasson and Mushynski 1996); (3) Neu-N (clone A60, 1:100; Chemicon, Temecula, CA) that reacts with a neuron-specific nuclear protein in most postmitotic neuronal cell types (Kruger et al. 2002); (4) class III β-tubulin (Covance), an antibody made against brain microtubules that does not react with β-tubulin in glial cells (Draberova et al. 1998); and (5) anti-Hu, HU-1C/D (clone 16A11, 1:200; Molecular Probes Invitrogen, Eugene, OR) (Gultekin et al. 1998). These antibodies did not react with glial and multiple epithelial or mesenchymal tissues. Antigen unmasking was performed by the following method: preheating at 95C-99C using Dako antigen-retrieval solution (Dako; Carpinteria, CA), citrate buffer, pH 6.1, or in high pH antigen-retrieval solution in a Black and Decker steamer (Model HS800; Black and Decker, Shelton, CT) for 30 min followed by a 15-min cool-down period. Immunoreactivity was detected and visualized with horse anti-mouse IgG Bio-tin (Vector Laboratories; Burlington, VT) and streptavidin-HRP (Jackson ImmunoResearch Laboratories; West Grove, PA). Slides were developed with AEC+ (Dako) (Sabattini et al. 1998).
Clinical characteristics
NA, not available; HA, headache; WHO, World Health Organization.
Sections were scored as exhibiting morphological features of neurons (+) if their size was more than twice the size of the diameter of adenoma cells, and if they exhibited abundant cytoplasm with or without obvious Nissl substance, and prominent nucleoli. Cells with expanded somas relative to other adenoma cells and prominent nucleoli were designated “possible” neurons. Adenomas without these features were designated (−). Immunoreactivity to each antibody was also scored as “+” (1 or less cell per ×10 field), “++” (>1 to scattered cells per ×10 field), or absent (−). In cases of discordance between reviewers, the negative designation was accepted over “rare” immunoreactive cells.
Results
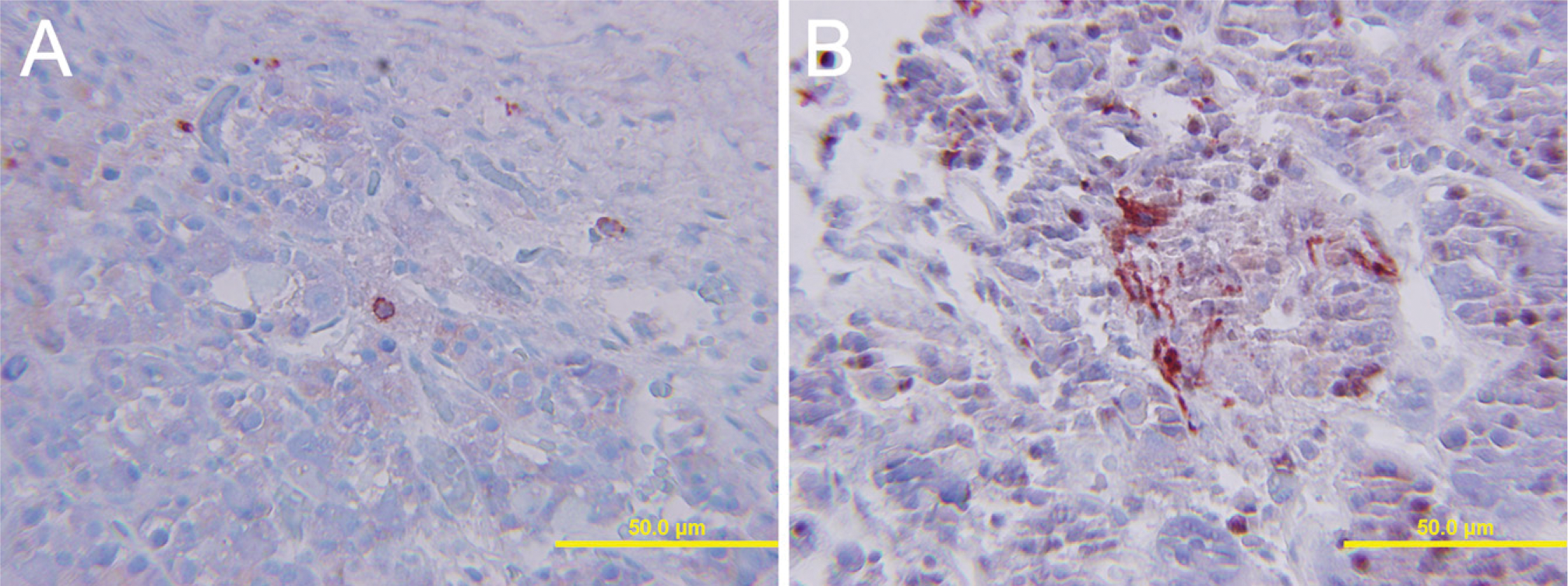
Results are listed in Table 2. Three somatotroph cell adenomas had morphological neuronal transdifferentiation (Figure 1 and Figure 2), and two had cells suspicious for transdifferentiation. Of these, 80% showed neurofilament immunostaining, 100% displayed PNF (Figure 1 and Figure 2), and one showed Neu-N immunoreactivity. All exhibited tubulin and one exhibited Hu immunostaining. Possible morphological evidence of neuronal transdifferentiation was found in two mixed GH and prolactin cell adenomas. These tumors displayed multiple tumor markers. In mixed GH and prolactin cell adenomas, three of four adenomas displayed PNF. All displayed tubulin, but none showed Hu immunoreactivity. One adenoma with possible morphological evidence suggestive of neuronal differentiation also displayed Neu-N. Neither lactotroph adenomas exhibited possible neuronal transdifferentiation, but both displayed some cells with PNF and tubulin immunostaining. In the null cell adenomas, morphological evidence of neuronal differentiation was limited, but 5/10 (50%) displayed NF and 7/10 (70%), including the cases with suspicious morphology, displayed PNF, eight of nine displayed tubulin (88%), and one displayed Neu-N immunostaining. None of the corticotroph cell adenomas exhibited easily recognized morphology consistent with neuronal transdifferentiation, but one displayed PNF, tubulin, and Hu immunostaining (Figure 3).
Immunophenotype of neuron-like cells in pituitary adenomas
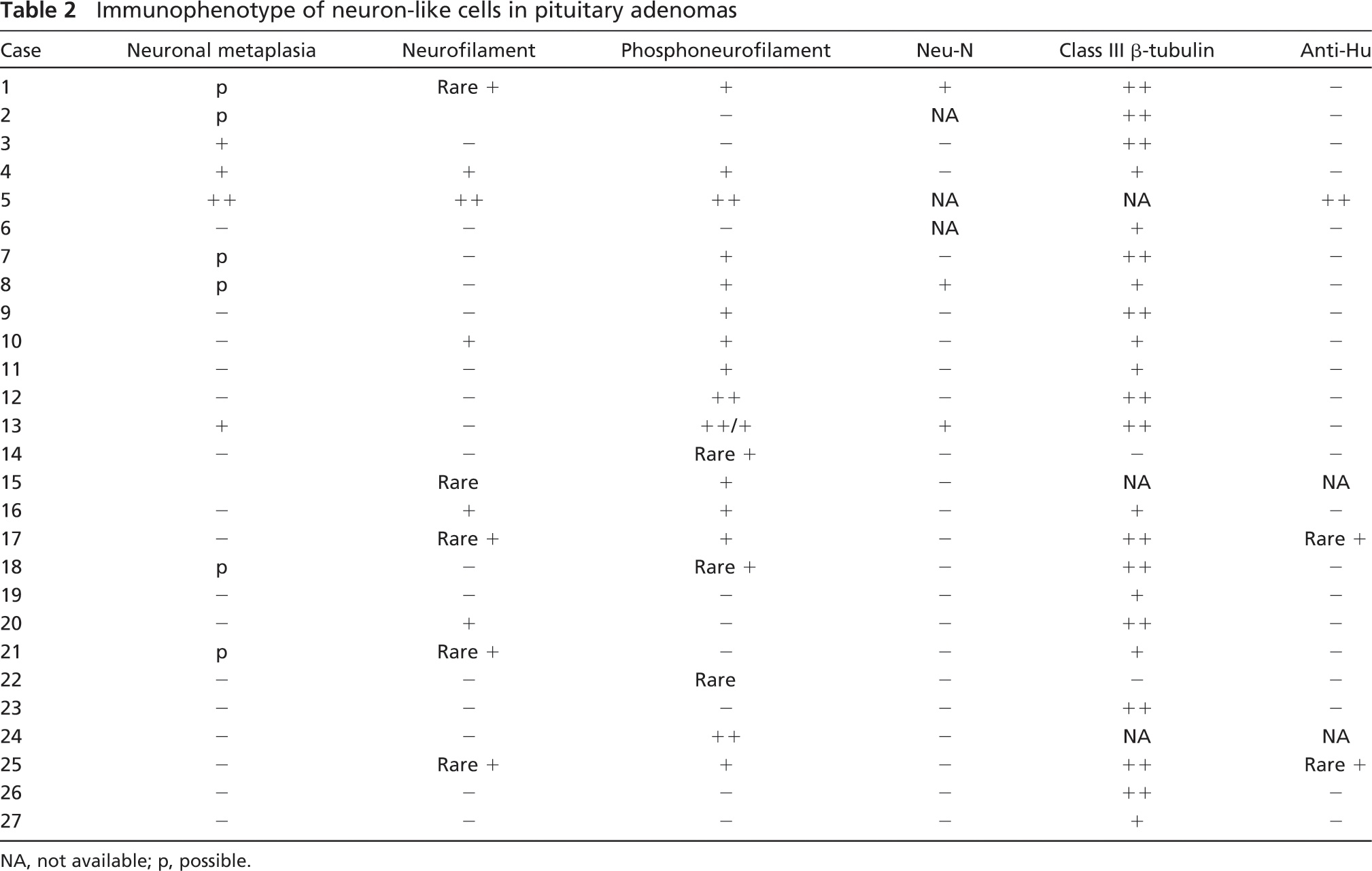
NA, not available; p, possible.
Discussion
Findings presented here suggest that cells with neuronal features and expression of neuronal-associated epitopes may be more common in pituitary adenomas than previously recognized and occur not only in GH cell adenomas but also other types such as lactotroph, null cell, gonadotroph cell, and corticotroph cell adenomas. These findings are consistent with growing recognition of neuronal transdifferentiation in other neural crest tumors. The infrequent recognition of neuronal forms may, in part, be due to (1) infrequent immunohistochemical evaluation for its presence; (2) prominent nucleoli, particularly GH secreting adenomas, obscuring its presence; (3) widespread synaptophysin immunoreactivity that may obscure the presence of coexisting small neurons; and (4) the presence of intermediately differentiated neuronal forms (as seen in ganglion cell tumors) that are difficult to recognize.
A previous study found neurofilament protein in stroma of a tumor with “incomplete neuronal differentiation” (Thodou et al. 2004), neuropil (Vidal et al. 2002), and neuropil fibrils of pituitary “gangliocytomas” and dot-like NF immunoreactivity in some adenoma cells associated with these cases (Kontogeorgos et al. 2006). In most cases, in the present study neurofilament in adenoma and neuron-like cells appeared to be phosphorylated. PNF is seen in intermediately and differentiated neurons (Pant and Veeranna 1995). The presence of PNF immunoreactivity has not been described in adenomas or in cells with neuronal metaplasia. Its significance, if any, remains to be established. At least in neurons, phosphorylation of neurofilaments is thought to increase stability to axons (Shea et al. 2003).
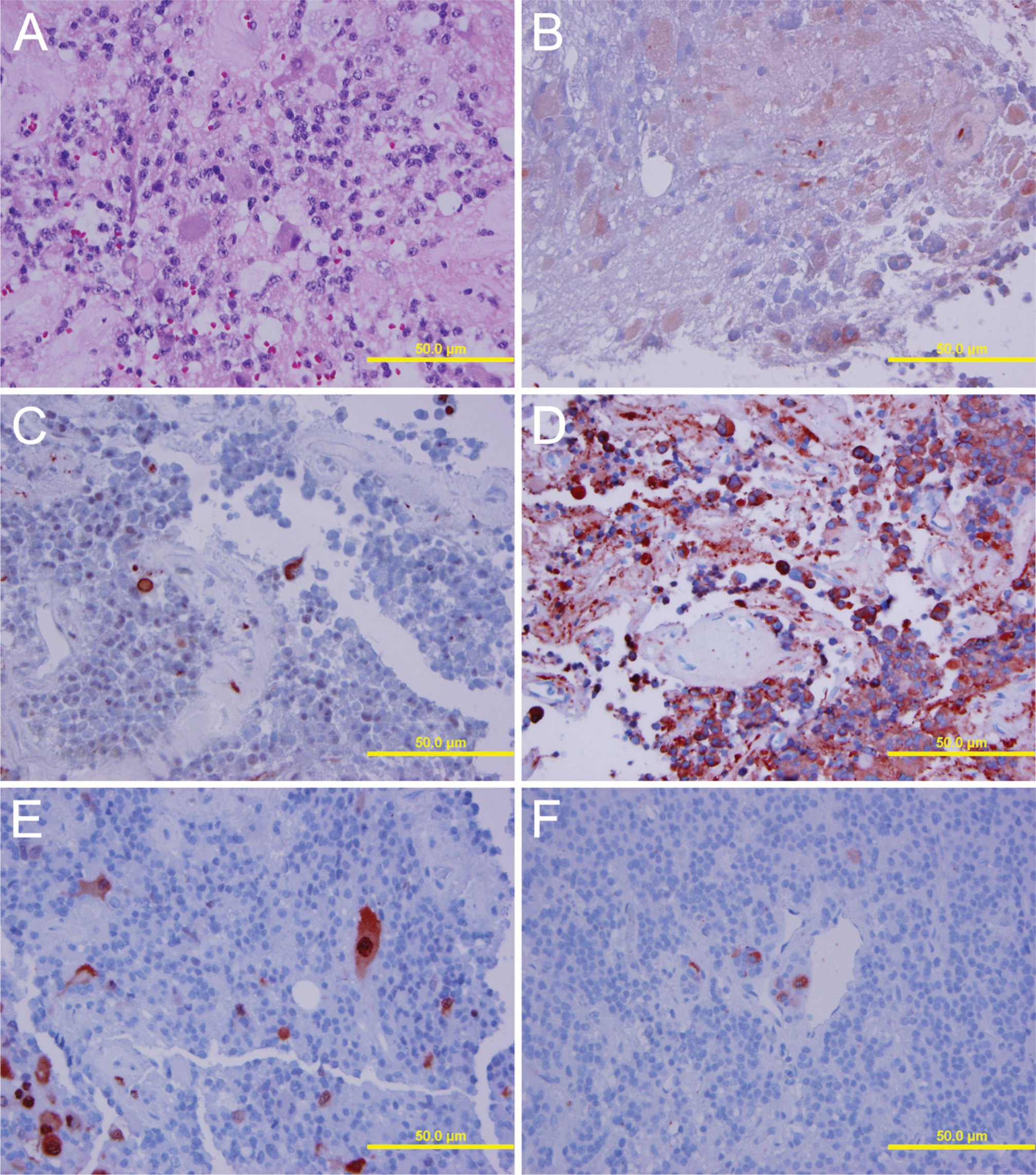
Case 5. Neuronal differentiation and neuropil, hematoxylin and eosin stain (
Case 4. Neuronal differentiation in a GH cell pituitary adenoma exhibiting immunoreactivity for neurofilament (
Neu-N immunostaining was seen in only a few adenomas, all of which had other evidence of neuronal differentiation. Three of five also displayed phosphoneurofilament immunoreactivity in cells with neuronal features. This suggests that, in most cases, the neuronlike cells do not express Neu-N and/or are not terminally differentiated.
Anti-Hu antibodies react against RNA-binding proteins that are expressed early in the development of the nervous system and are associated with neuronal differentiation and function (Gultekin et al. 1998). In normal tissue they are restricted to neurons. They have been used as a marker of neurogenic precursors in neural crest cells (Marusic 1993). Anti-Hu antibodies have been found to react with nuclei and weakly with cytoplasm of neurocytomas and gangliogliomas (Gultekin et al. 1998). Serum anti-neuronal nuclear autoantibodies associated with paraneoplastic syndromes have been shown to react with frozen but not formalin-fixed sections of pituitary adenomas (Laeng et al. 1998). In the present study, anti-Hu immunoreactivity was found in cells with morphological or immunohistochemical features of neuronal differentiation in four cases. Nonetheless, this was not widespread. These RNA-binding proteins are expressed in high levels earlier than synaptophysin, and thus it is not unexpected that their expression might be variable in cells with varying stages of neuronal differentiation.
Rare or scattered class III β-tubulin expression was seen in 88% of cases. Using a high dilution, the antibody reacted with cells exhibiting neuronal morphology but also in larger cells and some cells without overt neuronal differentiation. Expression was seen in all of the adenomas displaying phosphoneurofilament immunoreactivity and in an additional 33% of cases where phosphoneurofilament was not seen (Furuhata et al. 1993). This expression, seen in conjunction with phosphoneurofilament expression, appears to support the presence of variable neuronal transdifferentiation in some adenomas. Thus, its detection in some smaller adenoma cells is of uncertain significance but may reflect an intermediate level of differentiation. Class III β-tubulin expression has been observed in >5% of cells in 43/61 pituitary adenomas in a previous study but without characterization of the scattered adenoma cells showing immunoreactivity (Furuhata et al. 1993).
The origin of cells exhibiting neuronal features is not certain. Previous studies have suggested that they represent metaplasia of adenoma cells. Transdifferentiation of pituitary cells has also been described. Hormonal changes during pregnancy stimulate lactotroph cell hyperplasia and convert somatotrophs to prolactinsecreting cells (Vidal et al. 2001; Kakeya et al. 2002). Diethylstilbestrol increases lactotrophs by transdifferentiation of gonadotrophic cells (Shukuwa et al. 2006). Somatotroph conversion into thyrotrophs after experimental production of a hypothyroid state has also been described (Radian et al. 2003). Adenoma cell transdifferentiation into neurons has not been established, but adenoma cells are known to express some neuronal epitopes, particularly synaptophysin (Pierret et al. 2006), and may have that potential. The presence of PNF in some cells maintaining features of adenoma cells also supports that possibility. Nonetheless, it is also possible that these cells arise from pleuripotential stem cells in the pituitary with a capacity for neural differentiation. Adult stem cell differentiation into neural cells has been described (Pierret et al. 2006). It has recently been shown that ∼1.5% of adenohypophyseal cells are “stem/progenitor cells” (Carbajo-Perez and Watanabe 1990; Taniguchi et al. 2002; Chen et al. 2005), which could give rise to pituitary adenomas and also to neuronal components in them (Chen et al. 2005). Circulating CD14 (+) monocyte-derived multipotential cells have also been shown to differentiate into neurons (Kodama et al. 2006).
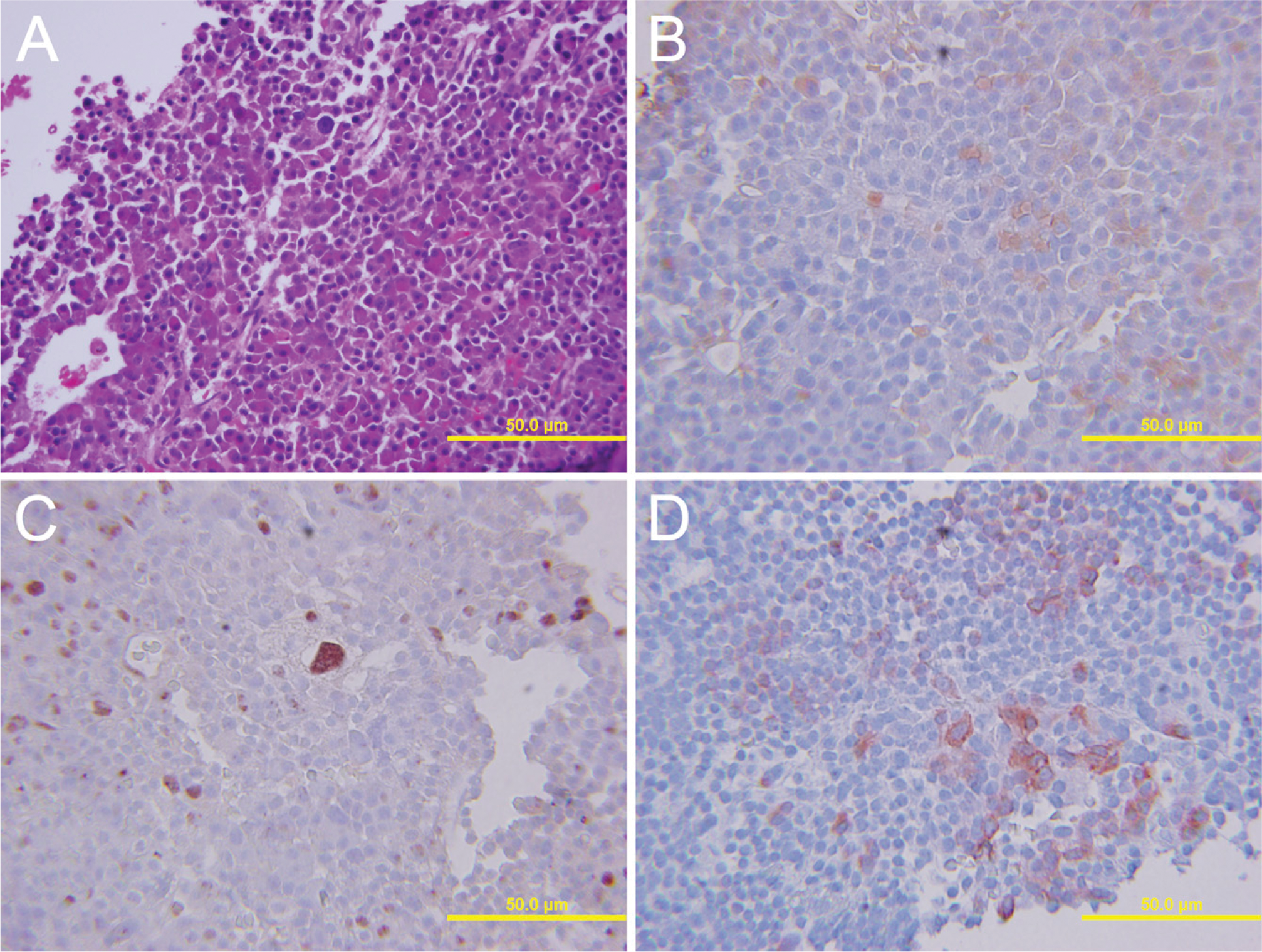
Case 25. Corticotroph cell pituitary adenoma with scattered neuronal differentiation (
The clinical significance of the finding of neuronal differentiation in pituitary adenomas remains to be established. Nonetheless, in some malignant neuroepithelial tumors such as neuroblastomas and medulloblastomas, neuronal differentiation is associated with a better prognosis (Hedborg et al. 1995; Cai et al. 2000; Yamaguchi et al. 2007). Recent gene profiling studies of high-grade gliomas have shown that a molecular subtype (proneuronal type) in a tumor displaying neural lineage mainly showed longer survival (Phillips et al. 2006). In the present study, long-term follow-up was not available for many patients biopsied a decade earlier and followed at off-site clinics. Prospective studies may clarify if this feature is associated with a reduced rate of recurrence and less postoperative recurrence.
