Abstract
Galectin-3 (Gal-3) is a multifunctional protein that plays different roles in cancer biology. To better understand the role of Gal-3 and its ligands during colon carcino-genesis, we studied its expression in tumors induced in rats treated with 1,2-dimethylhydrazine (DMH) and in human tissues. Normal colon from untreated rats showed no staining using two specific monoclonal antibodies. In contrast, morphologically normal colon from DMH-treated rats and dysplastic aberrant crypt foci were strongly stained, indicating that increased Gal-3 expression is an early event during the neoplastic transformation in colon cells. Gal-3 was weakly expressed in adenocarcinomas. Overall, the Gal-3 expression pattern observed in the DMH rat model closely resembles that displayed by human colon stained with the same antibodies. We also found that Gal-3 phosphorylation diminishes in serines while increasing in tyrosines during rat colon carcinogenesis. Finally, we showed that Gal-3–ligands expression is strikingly similar in rat and human malignant colon and in non-malignant tissues. In conclusion, the DMH-induced rat colon cancer model displays expression patterns of Gal-3 and its ligands very similar to those observed in human samples. This animal model should contribute to clarifying the role of Gal-3 in colon carcinogenesis and also to finding effective preventive cancer agents based on Gal-3 targeting.
G
Extensive studies have evaluated the diagnostic and prognostic values of Gal-3 in colon carcinoma (Byrd and Bresalier 2004). Several works agree that increased Gal-3 expression in colorectal cancer is associated with a poor prognosis (Schoeppner et al. 1995; Sanjuan et al. 1997; Nakamura et al. 1999; Hittelet et al. 2003; Legendre et al. 2003; Nagy et al. 2003; Endo et al. 2005). Indeed, increased expression of Gal-3 was shown to be associated with metastatic capabilities of colon cancer cells. Moreover, upregulation of Gal-3 in colon cancer cells by stable transfection results in an increase in spontaneous metastasis and liver colonization, whereas downregulation by antisense methodology significantly reduces metastasis (Bresalier et al. 1998). Gal-3 undergoes phosphorylation at Ser-6 and Ser-12 by casein kinase 1 (Huflejt et al. 1993; Mazurek et al. 2000). Phosphorylation of nuclear Gal-3 at Ser-6 is critical for its transport from the nucleus and for its anti-apoptotic functions (Yoshii et al. 2002; Takenaka et al. 2004). Phosphorylation of Gal-3 at Ser-6 also plays a key role in the malignant behavior of BT549 human breast cancer cells and is associated with the activation of specific sets of genes and pathways (Mazurek et al. 2005).
The wide variety of intra- and extra-cellular functions, ligands, and different possible subcellular localizations of Gal-3, as well as not-fully-understood posttranslational regulation, bear witness to a complex role for Gal-3 in cancer biology. Well-characterized clinically relevant animal cancer models are indeed an invaluable resource for unraveling the biology of Gal-3 and its ligands during carcinogenesis. We therefore studied a well-defined experimental model of colon tumourigenesis induced in rats by administration of 1,2-dimethylhydrazine (DMH) (Amberger 1986), one of the most used colorectal cancer animal models. DMH-induced tumors are histologically and pathologically similar to human sporadic colon cancer (Labarriere et al. 1994; Koesters et al. 2001). However, DMH-induced rat lesions and human tumors need to be compared at the molecular level to determine whether those tissues show similar alterations in different pathways. Here we describe the expression pattern of Gal-3 and its ligands, as well as its serine and tyrosine phosphorylation status, during rat colon carcinogenesis. Of importance, the results obtained in DMH-induced colon carcinogenesis strongly resemble those obtained in human cancer, indicating that this could be a novel, clinically relevant model for elucidating the complex role played by Gal-3 in colon cancer.
Materials and Methods
Monoclonal Antibodies
Rat hybridoma M3/38 producing monoclonal antibody (MAb) TIB-166 (IgG2a) against Gal-3 (Ho and Springer 1982) was obtained from the American Type Culture Collection (Rockville, MD). Anti-Gal-3 MAbs A3A12 and A1D6 (IgG1) were generated from mice immunized with recombinant human Gal-3 (Liu et al. 1996). The epitope for MAb A1D6 is contained in the region between aa 36–45 of Gal-3, whereas MAb A3A12 appears to recognize two regions present in aa 1–18 and in aa 36–45 of the molecule (Liu et al. 1996).
Monoclonal anti-phosphoserine and anti-phospho-tyrosine antibodies used in the present study were obtained from Sigma-Aldrich (Saint Quentin, France). These antibodies are widely used in immunoblotting methods and have been reported to specifically bind to phosphoserine (Naz 1999; Brustle et al. 2001; Liu et al. 2001) and phosphotyrosine (Vuillier et al. 1995; Allegrucci et al. 2001). Monoclonal anti-phosphoserine antibody (#P5747, clone PSR-45, mouse IgG1 isotype), raised by immunizing mice against phosphoserine conjugated to keyhole limpet hemocyanin (KLH), specifically reacts against phosphorylated serine. It does not react with non-phosphorylated serine, phosphorylated tyrosine, or threonine, AMP, or ATP. Monoclonal anti-phosphotyrosine antibody (#P5872, clone PT-66, mouse IgG1 isotype) was derived from mice immunized with phosphotyrosine bovine serum albumin (BSA) conjugate. The antibody reacts specifically with phosphorylated tyrosine, both as free amino acid or conjugated to carrier proteins such as KLH or BSA. There is no reactivity with phosphoserine, phosphothreonine, non-phosphorylated amino acids, including serine or threonine, or phosphorylated molecules such as AMP and ATP. Antibody binding is totally inhibited by an excess of free phosphotyrosine or phenylphosphate. No inhibition is observed with the non-reactive phosphorylated compounds listed above.
Animals and Experimental Design
DMH (Sigma), freshly dissolved in PBS, was administered to 6-week-old female Wistar rats at the beginning of the experiment, by subcutaneous injection (15 mg of DMH per kg body weight) as described previously (Berriel et al. 2005). Injections were repeated every week for 8 weeks after the initial DMH administration. The rats were fed on a laboratory chow diet and water ad libitum in a room with temperature maintained at 25C and a 12-hr-light/12-hr-dark cycle. The evaluation of Gal-3 and its ligands was performed on rats divided into the following five groups: (1) rats without treatment (control group); (2) rats killed 16 weeks after the first DMH injection; (3) rats killed 24 weeks after the first DMH injection; (4) rats killed 32 weeks after the first DMH injection; and (5) rats killed 40 weeks after the first DMH injection. Immediately after the animals were killed, colon tissues were removed and fixed in neutral-buffered formalin, coiled up into a “Swiss roll,” and embedded in paraffin for performing histopathological analysis and immunohistochemical evaluation of Gal-3 and its ligands. The “Swiss roll” procedure allows examination of the entire colonic mucosa. To evaluate Gal-3 phosphorylation, grossly evident tumors, non-tumoral mucosa (DMH-treated rats), and normal mucosa (control rats) were rinsed in PBS and stored at −80C until use. This project was approved by the Ethical Committee on Animal Research of the Faculty of Medicine, Universidad de la República, Montevideo, Uruguay.
Clinical Samples
Specimens of colonic adenocarcinoma were obtained from 12 patients of Hospital Pasteur, Montevideo, at the time of surgery. Tissue procurement was in accordance with institutional review board standards and procedures. Pathological examination was performed on all specimens to assess the presence of pre-neoplastic and neoplastic disease. Tumors were included with adjacent and distant non-tumoral mucosa. Samples of tumor tissue were also harvested and stored at −80C until analysis for RT-PCR.
Immunohistochemical Staining
Tissue sections of 5 μm were stained with hematoxylin and eosin. Slides were examined by two pathologists (DM and MR) and classified as normal mucosa, dysplastic change (adenomas of low and high grade), or adenocarcinoma, in agreement with World Health Organization classification of tumors (Hamilton and Aaltonen 2000). After deparaffinization, each sample was incubated with the appropriate dilution of each MAb overnight at 4C. After three washes in PBS, the quenching of endogenous peroxidase activity was performed with 3% H2 O2 in PBS for 20 min. Finally, sections were incubated with a peroxidase-conjugated anti-mouse immunoglobulin antibody (Sigma) (for A3A12 and A1D6) or a peroxidase-conjugated, anti-rat immunoglobulin antibody (Sigma) (for TIB-166) for 30 min at room temperature. Reactions were revealed with diaminobenzidine (Sigma), and slides were counterstained with Mayer's hematoxylin and mounted with a synthetic resinous medium. Between each step, sections were washed in PBS. For every assay, negative controls using PBS without primary antibody were included. To evaluate the expression of Gal-3 during colon carcinogenesis, we performed a semi-quantitative analysis of cell immunostaining. Each sample was scored as follows: 0, for negative samples or with less than 10% of tissue stained; 1, for samples with between 10% and 39% of tissue stained; 2, for samples with between 40% and 79% of tissue stained; and 3, for samples with more than 80% of tissue stained. The signal intensity was scored as strong (3), moderate (2), weak (1), or null (0). The total immunostaining score is the result of the addition of both parameters.
Analysis of Gal-3 Phosphorylation by Western Blot
Tissue samples (100 mg) of colonic mucosa from untreated rats, morphologically normal colon mucosa from DMH-treated rats, and colon carcinomas were homogenized in 50 mM Hepes, pH 7.4, containing 1% Triton X-100 and a protease inhibitor cocktail, at 4C. Soluble proteins were extracted by centrifugation at 100,000 g for 60 min at 4C and measured using the Bradford method (Bradford 1976). Equal samples of proteins (7 mg) were incubated overnight at 4C with the appropriate amount of anti–Gal-3 MAb (TIB-166). Protein G coupled to Sepharose beads (Sigma) was added to samples and then incubated for a subsequent 2 hr at 4C. Immunocomplexes bound to protein G were separated by centrifugation and extensively washed with 50 mM Tris-HCl, pH 7, and 1% Triton X-100. Samples were dissolved in sample buffer containing dithiotreitol and boiled for 7 min. Proteins were resolved by SDS-PAGE on 10% polyacrylamide gels, electroblotted onto polyvinylidene difluoride membranes, and probed with the appropriate antibody [mouse anti-phosphoserine (1:2000), mouse anti-phosphotyrosine (1:2000), or rat MAb anti–Gal-3 (1:50)] for 90 min at 4C, followed by 90 min incubation with horseradish peroxidase (HRP)-conjugated secondary antibody [anti-mouse IgG or anti-rat IgG (1:20,000) (Sigma Chemical)]. As a negative control, the same procedure was performed omitting primary antibodies. Immunoreactive bands were detected by a chemiluminescence procedure (ECL Plus Western Blotting Detection System; Amersham Biosciences, Little Chalfont, UK). Quantification of bands was achieved by densitometry using Storm 840 (Amersham Pharmacia Biotech; Little Chalfont, UK).
Detection of Gal-3 mRNA Using RT-PCR
Total RNAwas extracted from malignant human colon tissues with Tri-Reagent (Sigma-Aldrich) according to the manufacturer's instructions. First-strand cDNA was synthesized by using Moloney murine leukemia virus reverse transcriptase (Amersham; Piscataway, NJ). Briefly, 4 μg of RNAwas added to 200 U enzyme in the presence of 2 μl 20 mM of each deoxynucleotide triphosphate (dNTP) and 1 μl 250 μg random hexamers in a 20-μl total reaction volume. After incubation at 37C for 1 hr, the mixture was heated to 96C, snap-cooled, and stored at −20C. One μl of cDNA was added to a final volume of 25 μl of a PCR mixture containing 20 mM Tris-HCl, pH 8.4, 50 mM KCl, 2.5 mM MgCl2, 200 μM dNTP, 300 nM each primer, and 1 U Taq DNA polymerase (Life Technologies; Carlsbad, CA). Primer sequences are shown in Table 1. Amplification was performed under the following conditions for 35 cycles: 1 min at 95C, 1 min 40 sec at 58C, and 2 min at 72C. After a final 10 min extension period at 72C, 15 μl of PCR products was analyzed by electro-phoresis on 2% agarose gels and visualized by ethidium bromide staining under ultraviolet light. The human colon cancer HT29 cell line was used as positive control.
Evaluation of Gal-3 Ligands in Colon Tissues Using Recombinant Gal-3
The recombinant human Gal-3 was expressed in Escherichia coli from a construct based on the pET 30 Ek/Lic vector (Novagen; Madison, WI), and purified on a lactosyl-Sepharose column described previously (Ahmed et al. 1996). The recombinant Gal-3 was conjugated to HRP as reported earlier with some modifications (Ahmed et al. 2002). Briefly, HRP (4 mg) was activated by incubation with 1 mg sulfosuccinimidyl 4-[N-maleimidomethyl]cyclohexane-1-carboxylate (Pierce; Rockford, IL) in 0.5 ml PBS (pH 7.2) for 30 min at 37C, and separated by a desalting column. For conjugation, the purified Gal-3 (0.5 mg in 0.5 ml of azide-free PBS/0.1 M lactose) was mixed with the activated HRP. After overnight incubation at 4C, the conjugation mixture was dialyzed with azide-free PBS and purified by affinity chromatography on a lactosyl-Sepharose column as indicated above. The purified Gal-3–HRP conjugate was dialyzed with azide-free PBS and stored in 1% BSA–50% glycerol at −20C. The expression of Gal-3–accessible binding sites in colon tissues was evaluated by histochemistry. After the quenching of endogenous peroxidase activity, endogenous Gal-3–ligands interactions were competitively disrupted by adding saturating doses of lactose, and then sections were incubated with the Gal-3–HRP conjugate (10 μg/ml) for 60 min at room temperature. After three washes in PBS, reactions were revealed with diaminobenzidine as described above. Haptenic inhibition with 10 mM lactose was a test for sugar-dependent binding.
Primer sequences
Results
Gal-3 Is Predominantly Expressed in Early Stages of DMH-induced Rat Colon Carcinogenesis
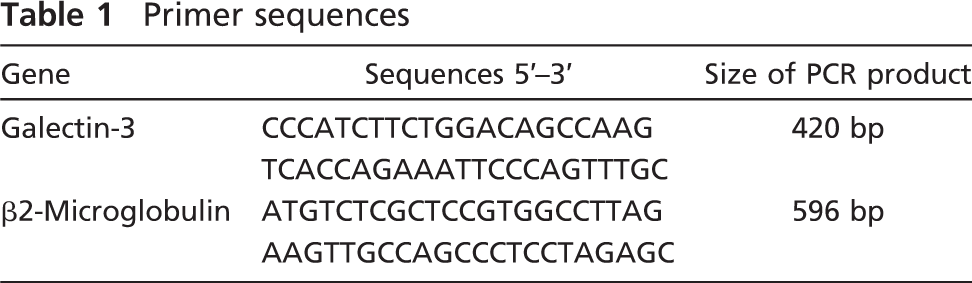
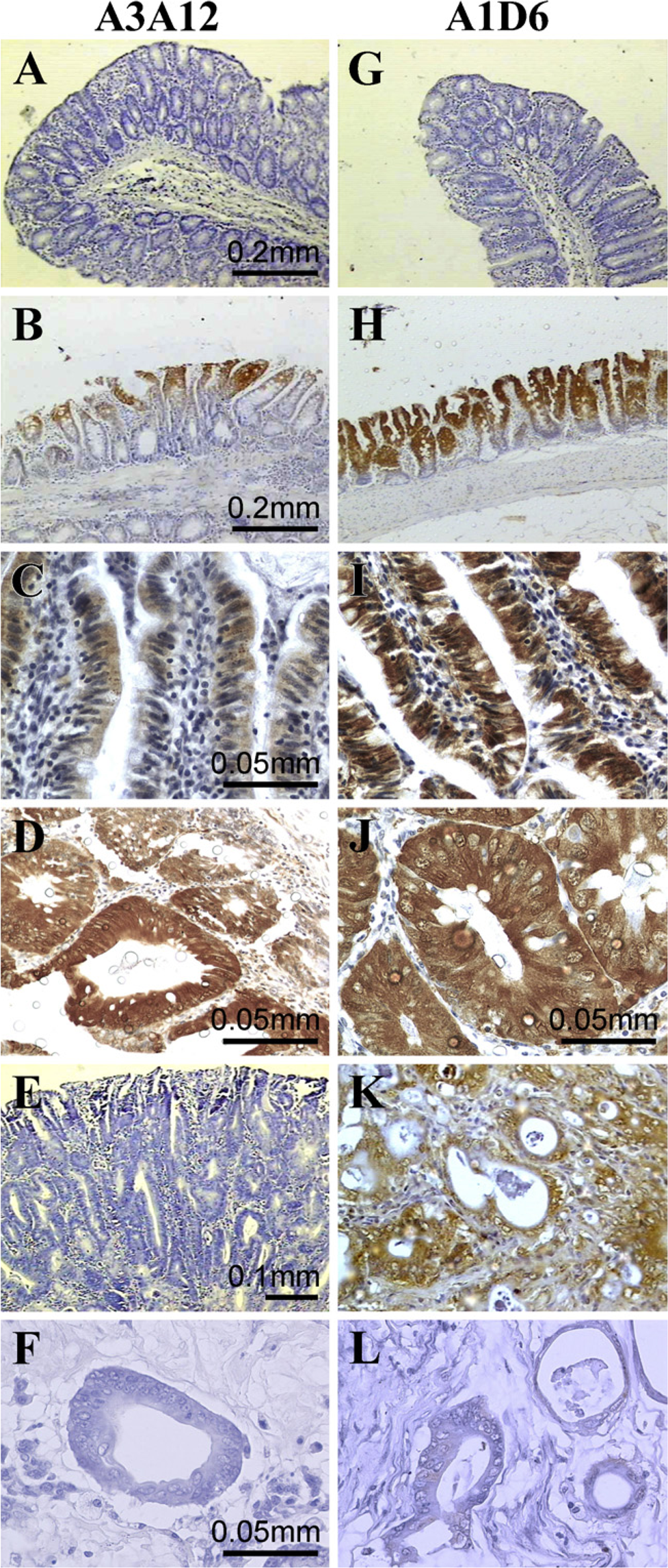
To determine whether Gal-3 is expressed in the DMH rat colon carcinogenesis model, immunohistochemistry experiments were performed at different time points before and after tumor induction. Development of colon adenocarcinoma was observed in 0/7 rats at week 16, in 3/7 rats at week 24, in 5/6 rats at week 32, and in 7/7 rats at week 40 after DMH administration. Metastases were found in lymph nodes, viscera, and peritoneum (animals sacrificed at week 40). To avoid epitope-restricted profiles, which might not reflect the whole Gal-3 antigen behavior, we used two different well-characterized anti–Gal-3 MAbs (MAbs A3A12 and A1D6) able to recognize human and rat Gal-3 (Liu et al. 1996). In immunohistochemistry experiments, we carefully analyzed colonic tissues from naive (untreated) rats, morphologic normal colon from DMH-treated rats, as well as DMH-induced dysplastic aberrant crypt foci [ACF (pre-malignant lesions)], DMH-induced adenocarcinoma, and hepatic metastasis from DMH-induced colonic adenocarcinoma. Normal colon from untreated rats showed an extremely modest, almost negative staining with both MAbs (Figures 1A and 1G). In fact, barely 5% of cells (Figure 2) showed some weak staining. In clear contrast, morphologically normal colon from DMH-treated rats (16 weeks of carcinogenesis) showed an intense staining with A3A12 and A1D6 (Figures 1B and 1H). About 75–90% of the crypts and superficial cells were positive for both MAbs (Figure 2). Although both MAbs gave very similar results regarding the staining intensity, the pattern was quite different. Indeed, whereas A3A12 staining was concentrated in the superficial crypt third, A1D6 staining was evident in almost all of the crypt, except its deepest part (Figures 1B and 1H and 1C and 1I), which contains the most immature enterocytes. Goblet cells were negative for both MAbs (Figures 1B and 1H and 1C and 1I). Similar results were obtained in morphologically normal colon from DMH-treated rats at 24 and 40 weeks of carcinogenesis (Figure 2 and data not shown). These results suggest that Gal-3 expression could be an early event during rat colon carcinogenesis. In agreement with this speculation, the premalignant lesions dysplastic ACF (Byrd and Bresalier 2004; Orlando et al. 2008) showed an intense staining with both A3A12 and A1D6 MAbs that covered ×90% of cells (Figures 1D and 1J and Figure 2). Qualitatively, A3A12 and A1D6 stained almost the entire crypt, including the deepest part (Figures 1D and 1J). Interestingly, we observed a dramatic diminution in the staining of A3A12 and A1D6 in malignant tumors, as compared with ACF and morphologically normal colon from DMH-treated rats (Figures 1E and 1K and Figure 2). Although all malignant tumors were positive for A1D6, the percentage of stained cells was approximately 25%. The contrast was more evident for A3A12 staining, where only 5% of the tumor cells were stained positively. A similar finding was obtained in metastatic tissues, which were stained negatively (A3A12) or barely positively in 10% of cells (A1D6) (Figures 1F and 1L and Figure 2).
Galectin-3 (Gal-3) expression during rat colon carcinogenesis. Anti–Gal-3 monoclonal antibodies (MAbs) A3A12 and A1D6 were probed against colonic rat tissues. Positive cells are shown in brown (DAB staining) and counter-colorated with hematoxylin (blue staining). Images are representative of different animals for each condition.
Semi-quantification of A3A12 and A1D6 staining during rat colon carcinogenesis. Semi-quantification was performed by two independent investigators (DM and MR). After immunoperoxidase staining with anti–Gal-3 MAbs, tissue sections were evaluated. Six different animals were studied in each group.
Gal-3 Expression in Human Colon Cancer Closely Resembles the Pattern Observed in the DMH Rat Model
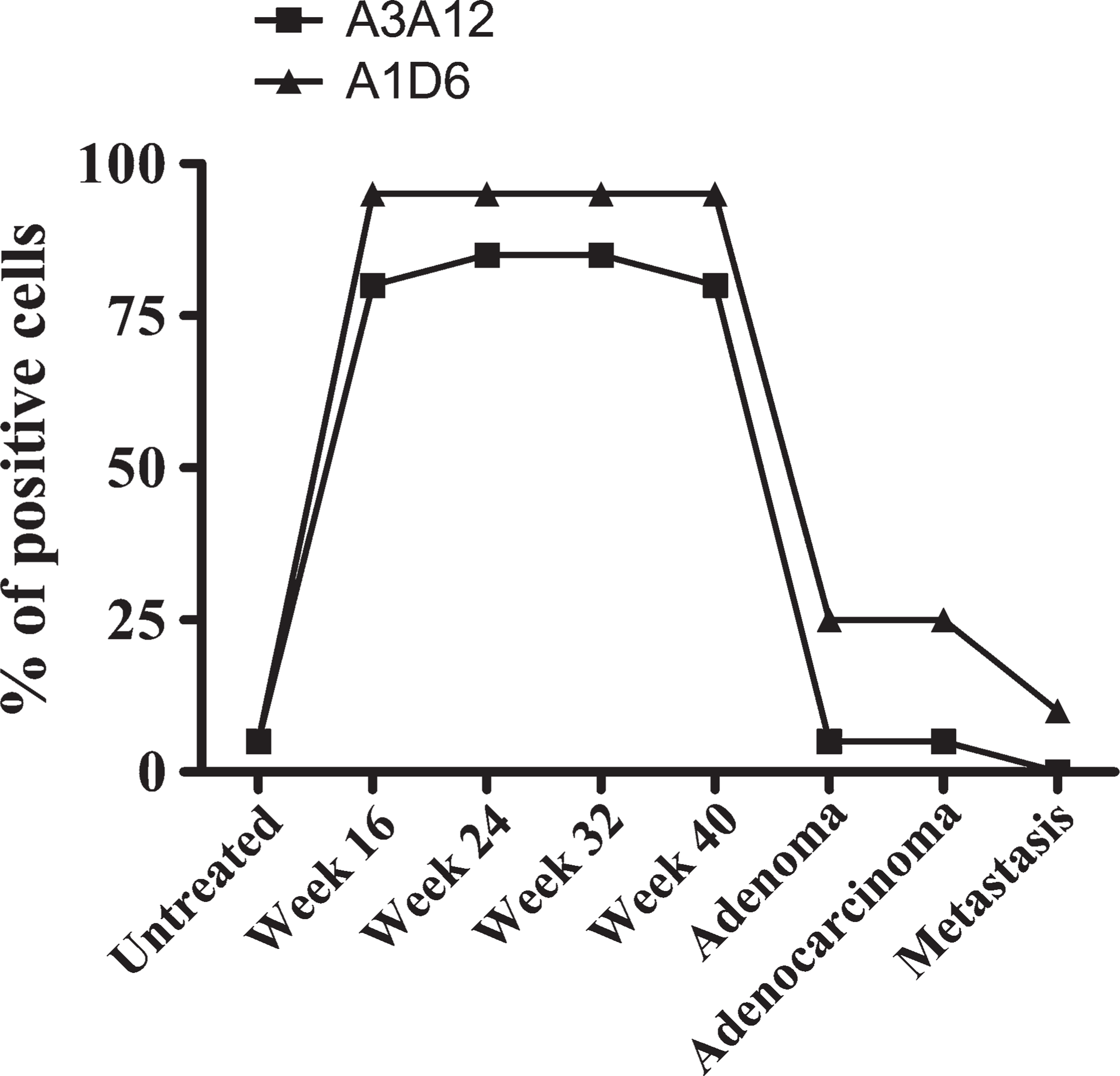
To compare the staining pattern of anti–Gal-3 antibodies between the DMH-induced rat colon model and human colon tissues, we studied 12 human colon adenocarcinoma samples, as well as morphologically normal colon mucosa surrounding the tumor and distant from it. We observed that morphologically normal colon mucosa distant from the tumor was almost negative for A3A12 (<5% of cells) and A1D6 (10% of cells) immunostaining (Figures 3A and 3F). This result was similar to that observed in colon tissues from untreated rats (Figures 1A and 1G). In contrast, morphologically normal colon mucosa surrounding the tumor showed significant staining with both antibodies (50–65% of cells) (Figures 3B and 3G) in a manner similar to that seen in morphologically normal colon from DMH-treated rats (Figures 1B, 1C, 1H, and 1I). Interestingly, well-differentiated human colon tumors showed more-significant A3A12 and A1D6 staining than poorly differentiated cancers (data not shown). Furthermore, malignant tumors showed very different immunostaining when comparing A3A12 and A1D6 MAbs (Figures 3D, 3I, 3E, and 3J and Figure 4). Although some tumors were negative or barely positive for both antibodies (Figure 4, samples identified as MA and 25), A3A12 staining was globally weaker than that for A1D6, whereas similar intensity of staining by both was observed in the positive control. Indeed, some tumors were negative/weakly positive for A3A12 but clearly positive for A1D6 (Figure 4, samples 17, 21, and 18). Again, these observations in human cancer strongly resemble the results obtained in the rat model. From the point of view of the subcellular immunostaining pattern, A3A12 showed a diffuse cytoplasmic staining in all samples (normal and neoplastic) (Figures 3B and 3E), whereas A1D6 showed nuclear and cytoplasmic immunoreactivity in crypts’ enterocytes adjacent to the infiltrant tumor (Figure 3G). With A1D6, it was also observed that as the displastic and neoplastic changes increased, the immunostaining pattern became mainly cytoplasmic in most atypical cells (Figure 3J). As observed in the rat model, not only the staining intensity but also the staining pattern was clearly different when comparing A3A12 and A1D6 MAbs.
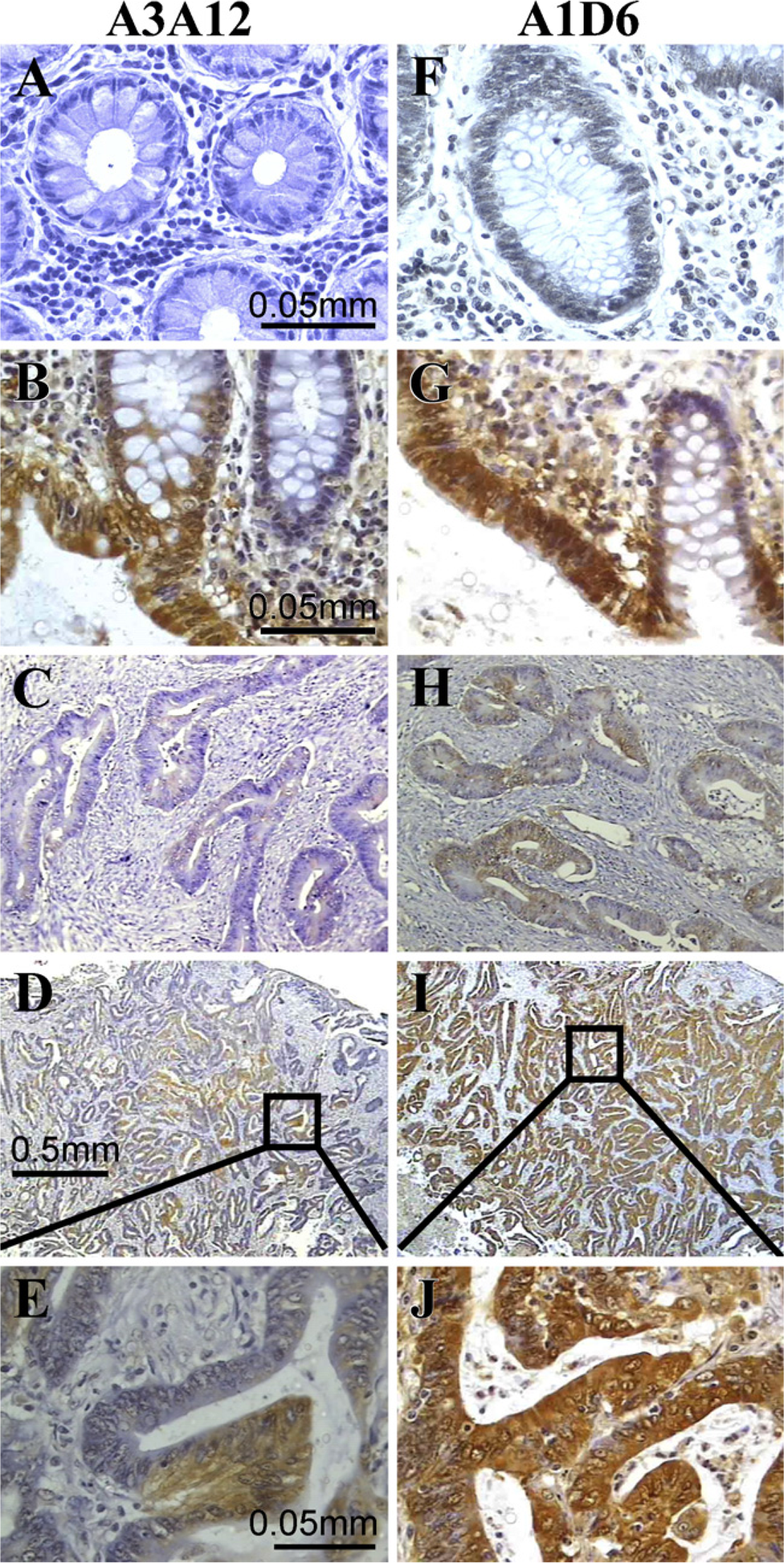
Gal-3 expression in human colonic tissues. Anti–Gal-3 MAbs A3A12 and A1D6 were probed against colonic human tissues. Positive cells are shown in brown (DAB staining) and counter-colorated with hematoxylin (blue staining). Images are representative of samples from twelve different patients.
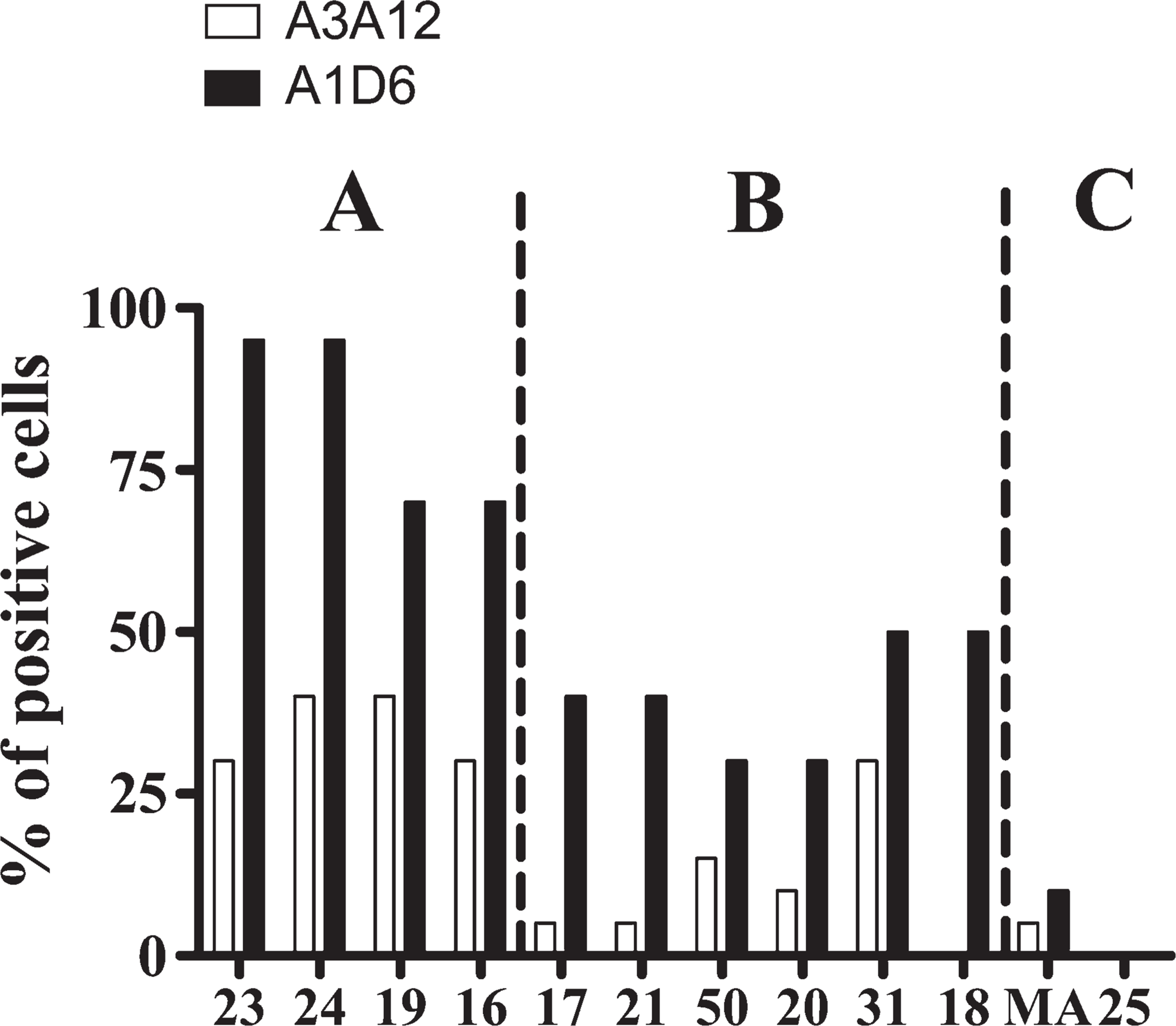
Semi-quantification of A3A12 and A1D6 staining in human colon adenocarcinomas. Each number on the x-axis represents one patient.
Some A3A12-negative Human Tumors Express Gal-3
The fact that some rat and human colon tumors were negative/weakly positive for A3A12 but positive for A1D6 raises the hypothesis that Gal-3 posttranslational modifications might alter immune reactivity of the MAb A3A12. However, another explanation could be that at least in these samples and by this methodology, A1D6 recognizes a non–Gal-3 epitope. To further analyze this issue, we decided to study Gal-3 expression in the A3A12–negative/A1D6–positive tumors at the mRNA level as well as in a third well-characterized anti–Gal-3 MAb (TIB-166) in immunohistochemistry experiments. RT-PCR experiments clearly showed that the A3A12–negative/A1D6–positive tumors (samples 17, 21, and 18) do express Gal-3 at the mRNA level (Figure 5A). In addition, those tumors showed strong immunoreactivity with MAb TIB-166 (Figure 5B), displaying a pattern similar to that of A1D6 (not shown). We can therefore conclude that A3A12–negative/A1D6–positive tumors do express Gal-3. Gal-3 posttranslational modifications could be one possible explanation for the differential immunostaining patterns observed with A3A12 and A1D6 MAbs as well as for the decreased immunostaining of these MAbs in malignant tissues.
Gal-3 Phosphorylation Diminishes in Serine Residues While Increasing in Tyrosine Residues During Rat Colon Carcinogenesis
It is well known that Gal-3 serine phosphorylation plays a major role in Gal-3 functions. In addition, Gal-3 tyrosine phosphorylation was also found in the period of repair of rat hepatocytes injured by CCl4 and in the rat hepatoma Ac2F cell line (Yamazaki et al. 2001). To identify the phosphorylation status of Gal-3 during carcinogenesis, we analyzed serine and tyrosine phosphorylation in Gal-3 at different stages of rat colonic malignant progression. Cell lysates from untreated rats, morphologically normal colon mucosa from DMH-treated rats, and colon carcinomas were immunoprecipitated using the anti–Gal-3 MAb TIB-166 and further blotted and assayed with TIB-166 and anti-phosphoserine– or anti-phosphotyrosine–specific antibodies. The phosphoserine/Gal-3 and phosphotyrosine/Gal-3 ratios were established by densitometric analysis of Western blot–stained bands for each sample analyzed (Figure 6). We observed a significant decrease of Gal-3 serine phos-phorylation together with an increase in Gal-3 tyrosine phosphorylation in colon carcinoma as compared with colonic mucosa from untreated rats and morphologically normal colon mucosa from DMH-treated rats (Figure 6). Gal-3 serine phosphorylation and Gal-3 tyro-sine phosphorylation are therefore dynamically and differentially regulated during rat colon carcinogenesis.
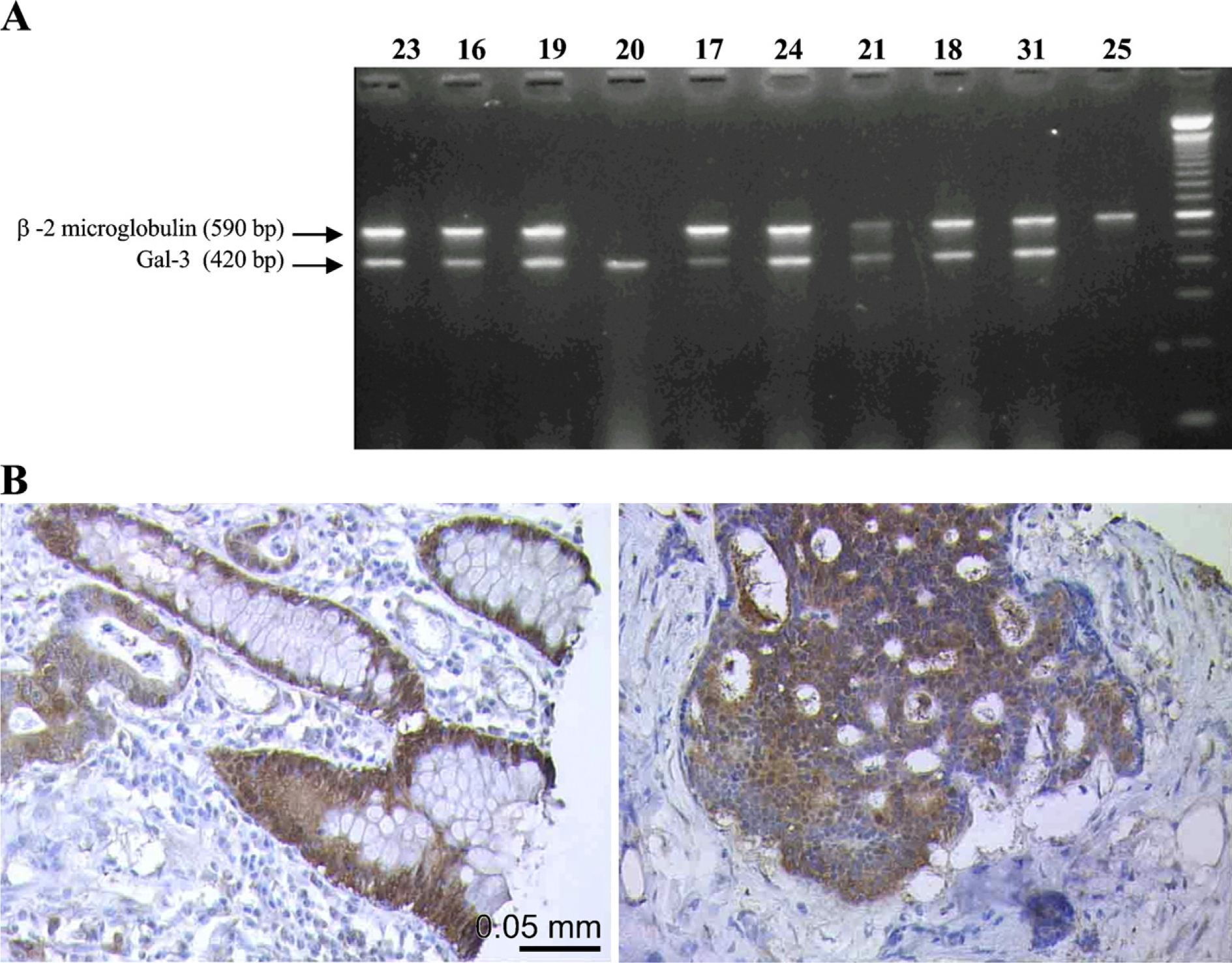
A3A12-negative human colon adenocarcinomas express Gal-3.
Gal-3 Ligand Expression in Malignant and Non-malignant Colon
Several biological functions of Gal-3, mainly at the extracellular level, depend on its capacity to bind β- galactosides. Consequently, we studied Gal-3 ligand expression in the rat model, comparing human malignant and non-malignant colonic tissues. To study this issue, we analyzed the staining pattern of recombinant Gal-3 coupled to HRP (Figure 7). The profile of Gal-3 ligand expression in rat (Figures 7A–7C) and human (Figures 7D–7F) colon tissues was therefore studied. In colon tissue from untreated rats (Figure 7A) and in morphologically normal human colon mucosa distant from the tumor (Figure 7D), we observed Gal-3–HRP staining in apical membrane and secretory vesicles of goblet cells as well as in the apical membrane of absorptive epithelial cells, whereas basal cytoplasm was negative. According to the results shown above regarding Gal-3 expression (Figures 1C, 1I, 3B, and 3G), Gal-3 was restricted to basal cytoplasm and was negative in secretory vesicles of goblet cells. Thus, Gal-3 and its ligands seem to be topologically separated in goblet cells. In morphologically normal colon from DMH-treated rats (Figure 7B) and in human colon tissue adjacent to the tumor (Figure 7E), the same staining pattern was observed, although the intensity was more significant. Furthermore, Gal-3–HRP staining was also evident in the lamina propria of peritumoral human tissues. In colon carcinoma, the desmoplastic stromal reaction was also stained with Gal-3–HRP (Figures 7C and 7F). Our results therefore showed that Gal-3–ligands expression is strikingly similar in rat and human malignant and non-malignant tissues. Interestingly, colon carcinoma expressed Gal-3 ligands at the extracellular matrix only in the late phases of tumorigenesis. This result suggests an important role for Gal-3–ligand interactions in cancer local invasion and metastasis.
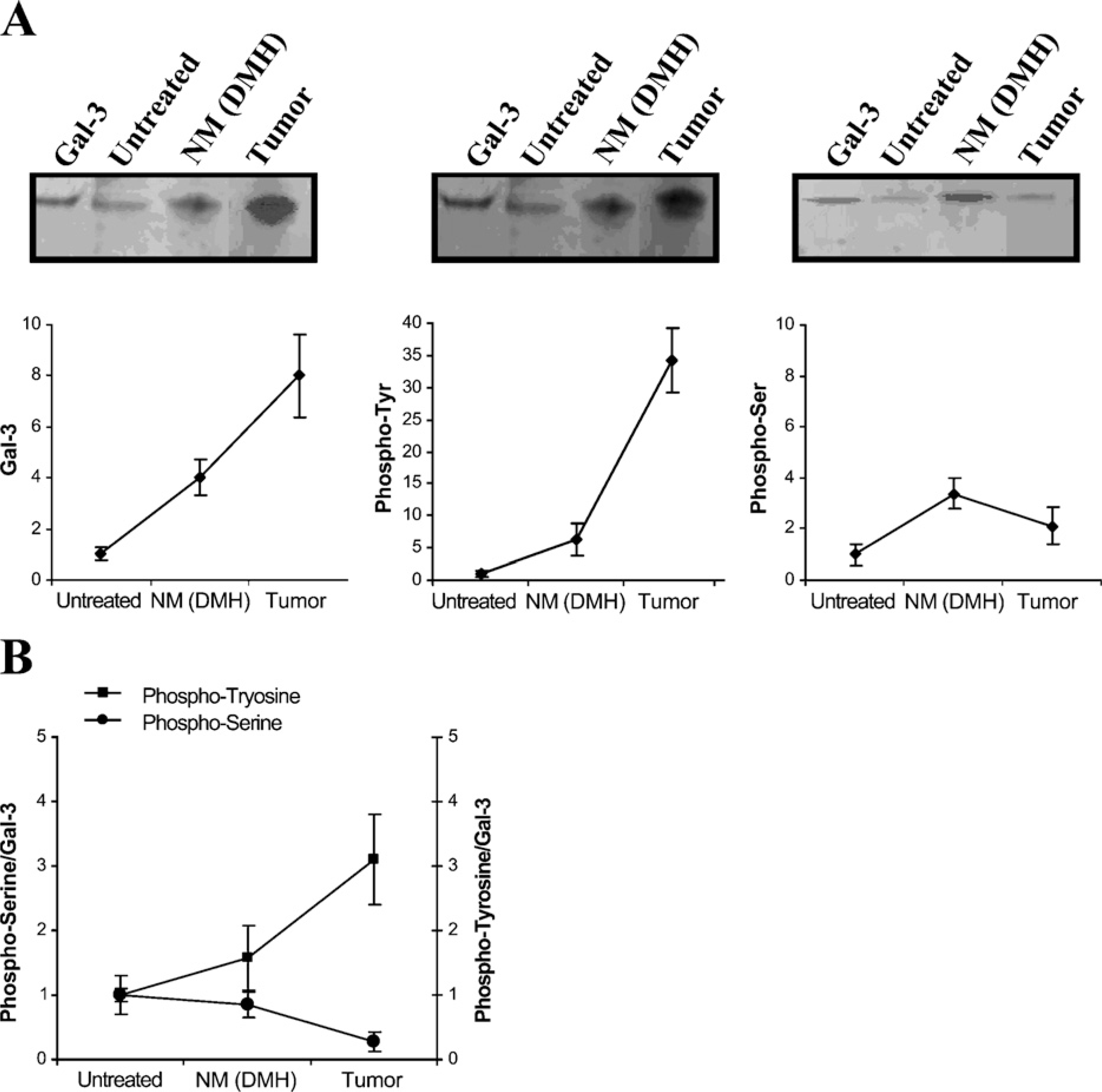
Gal-3 phosphorylation and expression pattern during rat colon carcinogenesis. Cell lysates from untreated rats, normal mucosa from DMH-treated rats, and colon carcinomas were immunoprecipitated using the anti–Gal-3 MAb TIB-166. After that, Western blot analyses of phosphoserine, phosphotyrosine, and Gal-3 were performed with appropriate antibodies.
Discussion
The role played by Gal-3 in colon cancer biology is starting to be understood. Data from several independent groups support the idea that Gal-3 promotes tumor development (Liu and Rabinovich 2005; Nakahara and Raz 2008). As mentioned in Eude-Le Parco et al. (2009), Gal-3−/- mice could have acquired some adaptive mechanisms during development in response to the absence of Gal-3, and one possibility is the compensation by other galectin members. In addition, Gal-3 can function in various cell types, including epithelial cells, stromal cells, and immune cells, and can have multiple functions. Although existing evidence suggests that Gal-3 in general promotes tumorigenesis, it is possible that in certain cancer models, such as those described by Eude-Le Parco and coworkers, the expression of this lectin in cancer cells promotes cancer initiation, progression, and metastasis, but its function in the cancer microenvironment and/or immune cells contributes to the suppression of these processes. As a result, no phenotype has been observed in Gal-3−/- mice. It is to be noted that other investigators, using Gal-3−/- mice (e.g., Abdel-Aziz et al. 2008), have shown that Gal-3 plays a role in tumorigenesis in other cancer models. Furthermore, its regulation, including phosphorylation and subcellular localization, is only partially understood. Recent evidence suggests that expression of Gal-3 in prostate cancer is regulated by promoter methylation (Ahmed et al. 2007). The pattern of Gal-3 expression in colorectal carcinoma has been a matter of debate, because some investigators have found decreasing Gal-3 levels in colon carcinoma progression, whereas others have observed increased Gal-3 content in human colorectal carcinomas (Califice et al. 2004). Technical issues can explain, at least partially, those discrepancies (van den Brule et al. 2004). Nevertheless, those contradictory results have led us to believe that a well-characterized animal cancer model could be an important tool for characterizing Gal-3 in colon cancer. Indeed, investigation of Gal-3 expression at different steps of carcinogenesis might make it possible to associate a certain expression pattern with a given molecular function. Additionally, this animal model should be pertinent to human malignant colon pathology, to better translate this knowledge into therapeutic and/or diagnostic approaches. DMH-treated animals develop sequential formation of histopathological lesions similar to those observed in spontaneous carcinogenesis in humans (Chang 1984), thus allowing the study of various stages of colon cancer formation (preneoplastic lesions, adenomas, carcinomas) from the earliest stages of tumor development. We found that A3A12 and A1D6 staining during DMH-induced rat colon carcinogenesis showed a particular kinetic pattern. Morphologically normal colon exposed to the carcinogen, as well as premalignant lesions (dysplastic ACF), dramatically increased Gal-3 expression, compared with colonic tissue from untreated rats, indicating that an increment in Gal-3 expression is an early event during carcinogenesis. These results agree with those observed by some authors for human colon carcinogenesis (Irimura et al. 1991; Schoeppner et al. 1995; Hittelet et al. 2003). Gal-3 also appeared very early during the first steps of the neoplastic transformation in diethylstilbestrol-induced renal tumors in male Syrian hamsters (Saussez et al. 2005). Remarkably, we found that immunostaining with A3A12 and A1D6 MAbs in human tissues indicates a kinetic evolution similar to that shown in rat carcinogenesis. Indeed, as observed in rat tissues, our results support the speculation that human colonic mucosa might increase A3A12 and A1D6 immunostaining during tumorigenesis while diminishing it in the poorly differentiated forms of cancer. However, tumors and metastasis showed diminished immunoreactivity for both MAbs used in this study, as compared with premalignant tissues. This is in agreement with the results reported by Lotz et al. (1993) and Sanjuan et al. (1997), who reported a decreased expression of Gal-3 related to the neoplastic progression of colon cancer. However, opposite results in human tissues were obtained by Schoeppner et al. (1995) and Nakamura et al. (1999), who found that metastases expressed a higher level of Gal-3 than the primary cancers from which they evolved.
Unexpectedly, we found significant differences in the staining pattern between A3A12 and A1D6 MAbs. In fact, the distribution of reactivity was distinct along the crypt-to-surface axis, in that A3A12 staining was concentrated in the upper third of the crypt and surface cells, whereas A1D6 staining was evident in almost all of the crypt, except in the cells at the base, which were negative. Furthermore, the subcellular immunostaining pattern was also different between the two MAbs. This is an important issue; using the polyclonal antibody anti–Gal-3, van den Brule et al. (2000) reported that the cytoplasmic/nuclear Gal-3 expression pattern is associated with tumor progression in prostate cancer. Indeed, the tool used to detect Gal-3 can give different subcellular localizations, implying different prognostic values. In malignant tissues, we showed that A13A12 staining was, in general, weaker than A1D6 staining. We found that some A3A12-negative tumors do express Gal-3, demonstrated at the mRNA and protein levels (Gal-3 identified using MAbs TIB-166 and A1D6). One possible explanation for these differential staining patterns could be that posttranslational modifications of Gal-3, differentially regulated during colon carcinogenesis, could modify the epitope recognized by the A3A12 MAb. Among other posttranslational modifications, Gal-3 cleavage might explain differential reactivity with the A3A12 and A1D6 antibodies. Further work is needed to establish whether this is the case. Whereas the epitope for MAb A1D6 is contained in the region between aa 36–45 of Gal-3, MAb A3A12 appears to recognize independently two regions present in aa 1–18 and in aa 36–45 of the molecule (Liu et al. 1996). Gal-3 displays phosphorylable serines (Ser-6 and Ser-12) and probably phosphorylable tyrosines (Tyr-41 and Tyr-45) within the antigenic sequence recognized by A3A12 (Figure 8). It remains to be determined whether Tyr-41 and Tyr-45 are indeed phosphorylated in Gal-3. In this context, we observed that Gal-3 phosphorylation diminishes in serines while increasing in tyrosines during rat colon carcinogenesis. Therefore, our results raise the possibility that Gal-3 phosphorylation (probably at tyrosine residues) is involved in the modification of the A3A12 binding site in colon carcinomas. It is important to note that phosphorylated Gal-3 retains immunological recognition by MAb TIB-166 (Mazurek et al. 2000). Another explanation could be that the epitope recognized by A3A12 may include phosphorylated serines within the aa 1–18 sequence (Figure 8). Further studies will determine whether a cancer-associated phosphorylation pattern could affect the binding of these antibodies to Gal-3.
Perhaps one of the most interesting observations reported here is that Gal-3 can be phosphorylated at tyrosine residues and that this process is associated with malignant transformation. Although Gal-3 tyro-sine phosphorylation has been reported in rat hepatocytes injured by CCl4 and in the rat hepatoma Ac2F cell line (Yamazaki et al. 2001), to our knowledge, Gal-3 tyrosine phosphorylation has never been described in malignant cells. Intriguingly, since the original observation in 2001 that Gal-3 can be tyrosine phosphorylated, no further reports have confirmed this observation. A species-restricted (rat) Gal-3 tyrosine phosphorylation might explain why this phenomenon has not been more widely studied. Previously, it was found that DMH treatment induced an increase of tyrosine-phosphorylated proteins in apparently normal rat colon, as well as in invasive rat colon carcinoma tissues, compared with untreated normal colon (Schwartz et al. 1995). After DMH administration, intense phosphotyrosine positivity (Schwartz et al. 1995) and a marked protein tyrosine kinase activity (Schwartz et al. 1991) were noted in colonic cells from the upper crypt zones. Protein tyrosine phosphorylation plays a key role in the generation of signals necessary for cellular events such as growth, migration, and invasion of malignant cells (Vlahovic and Crawford 2003). The results reported here should be taken as an incentive to proceed further in the biochemical and functional characterization of Gal-3 tyrosine phosphorylation during colon carcinogenesis. More information is available regarding phosphorylation of Gal-3 at Ser-6, which has important functional relevance. It was found that this phosphorylation significantly reduces the interaction of Gal-3 with its ligands (Mazurek et al. 2000), is critical for Gal-3 transport from the nucleus and for its anti-apoptotic functions (Yoshii et al. 2002; Takenaka et al. 2004), and also plays a key role in the behavior of malignant breast cancer cells (Mazurek et al. 2005). Our results suggest that Gal-3 serine phosphorylation could be an early event in the neoplastic progression of colon cells. Taking into account that almost all reported works on Gal-3 serine phosphorylation are based on the use of cell lines, the experimental model reported here could be very useful in evaluating in vivo the role of this phosphorylation during colon carcinogenesis.
Potential ligands for Gal-3 include mucins derived from colon cancer cells, Thomsen Friedenreich antigen, laminin, lysosomal-associated membrane proteins 1 and 2, IgE, and Mac-2-binding protein. Our results show that rat and human colon carcinomas express Gal-3 ligands at the extracellular matrix only in late phases of tumorigenesis. This result is in agreement with the proposed role for Gal-3–ligand interactions in cancer local invasion and metastasis. Furthermore, mononuclear cell tumoral infiltration was evident mainly at late stages. Indeed, intratumoral monocyte-macrophages as well as lymphocytes might be modulated by Gal-3–ligand interactions at the extracellular matrix. Finally, our observations might constitute a model for studying Gal-3–ligand interactions. For instance, the biological impact of Gal-3 modifications such as sialylation and sulfation could be assessed by studying the binding of modified Gal-3 to ligands expressed by DMH-induced tumors. In addition, it is important to note that Gal-3 displays high affinity for A and H histo–blood group antigens (Bachhawat-Sikder et al. 2001), which modify their expression in colon mucosa from rats treated with DMH. Normal rat colon showed strong expression of A antigen, but weak expression of H antigen. In contrast, it was found that dysplastic glands were strongly stained with anti-H reagents, whereas their A antigen was lost (Hallouin et al. 1997).
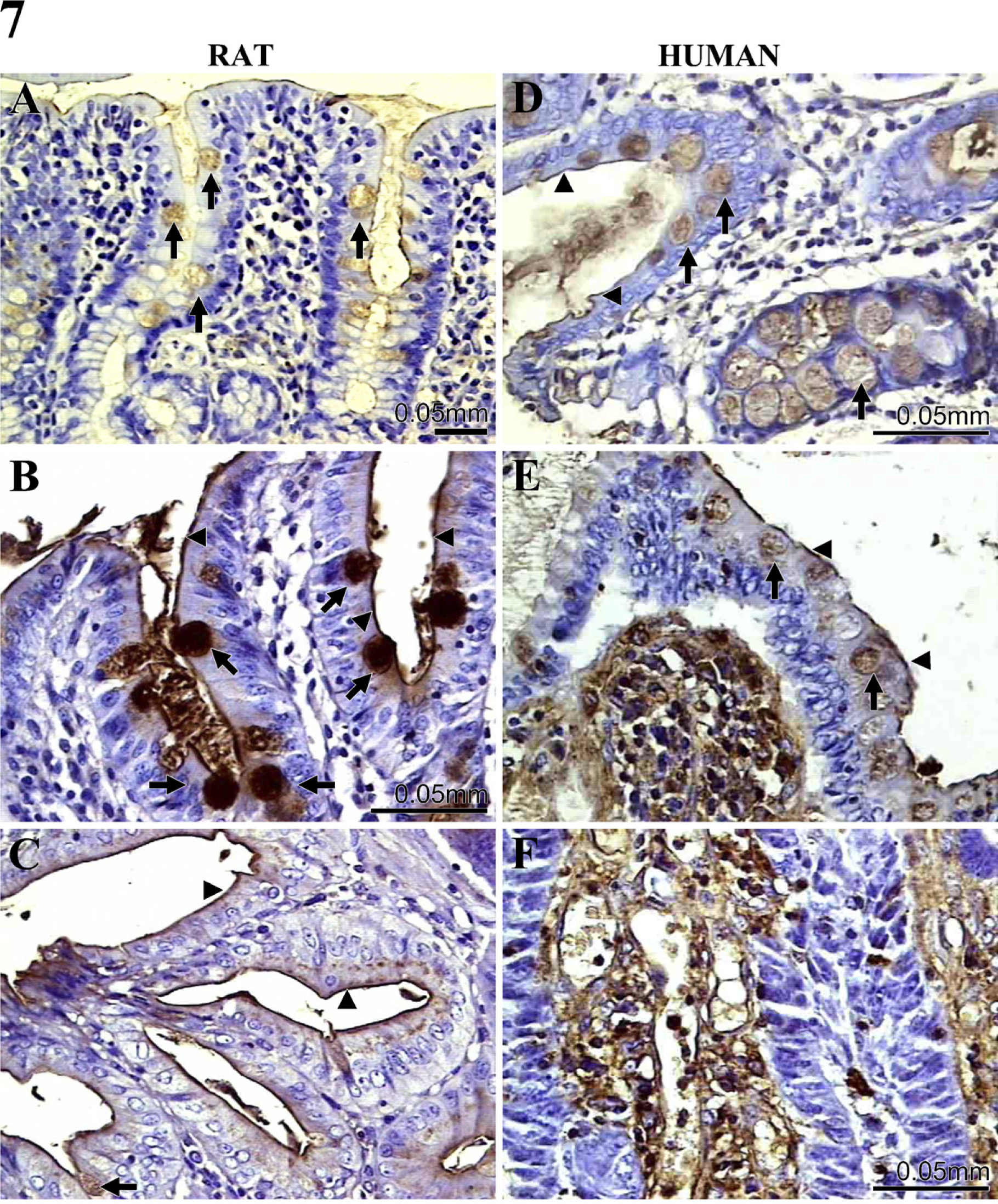
Gal-3 ligand expression during DMH-induced rat colon carcinogenesis and in human malignant and non-malignant colon. Gal-3 ligands were identified by incubating Gal-3–horseradish peroxidase (brown staining) in rat
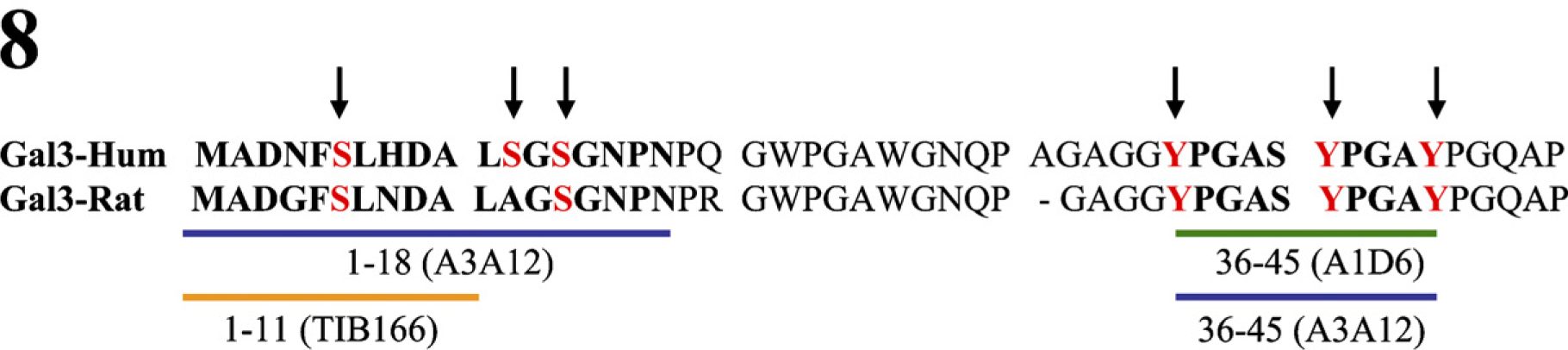
Alignment of human and rat Gal-3 amino acids sequence (aa 1–50) depicting the epitopic sequences recognized by A3A12, A1D6, and TIB166 MAbs. Already described (serine) and putative phosphorylable (tyrosine) residues are indicated by arrows.
In conclusion, we describe here a rat colon cancer model that displays Gal-3 and ligand expression patterns very similar to those observed in human samples. Differences observed between distinct MAbs might be explained by posttranslational Gal-3 modifications that could be attributed to phosphorylation. Given the complexity of Gal-3 biology in cancer, the new clinically relevant model described here is an interesting tool for shedding light on Gal-3 function in colon carcinogenesis.
Footnotes
Acknowledgements
This work was supported by the Comisión Honoraria de Lucha Contra el Cáncer, Montevideo, Uruguay.
