Abstract
Embryonic stem cells (ESCs) provide a convenient model to probe the molecular and cellular dynamics of developmental cell morphogenesis. ESC differentiation in vitro via embryoid bodies (EBs) recapitulates many aspects of early stages of development, including the epithelial–mesenchymal transition (EMT) of pluripotent cells into more differentiated progeny. Hyaluronan and versican are important extracellular mediators of EMT processes, yet the temporal expression and spatial distribution of these extracellular matrix (ECM) molecules during EB differentiation remains undefined. Thus, the objective of this study was to evaluate the synthesis and organization of hyaluronan and versican by using murine ESCs during EB differentiation. Hyaluronan and versican (V0 and V1 isoforms), visualized by immunohistochemistry and evaluated biochemically, accumulated within EBs during the course of differentiation. Interestingly, increasing amounts of a 70-kDa proteolytic fragment of versican were also detected over time, along with ADAMTS-1 and −5 protein expression. ESCs expressed each of the hyaluronan synthases (HAS) −1, −2, and −3 and versican splice variants (V0, V1, V2, and V3) throughout EB differentiation, but HAS-2, V0, and V1 were expressed at significantly increased levels at each time point examined. Hyaluronan and versican exhibited overlapping expression patterns within EBs in regions of low cell density, and versican expression was excluded from clusters of epithelial (cytokeratin-positive) cells but was enriched within the vicinity of mesenchymal (N-cadherin-positive) cells. These results indicate that hyaluronan and versican synthesized by ESCs within EB microenviron-ments are associated with EMT processes and furthermore suggest that endogenously produced ECM molecules play a role in ESC differentiation. This manuscript contains online supplemental material at http://www.jhc.org. Please visit this article online to view these materials.
Keywords
P
During early development, the epithelial–mesenchymal transition (EMT) is a critical process in embryogenesis whereby epithelial progenitor cells transform into mesenchymal cells to migrate and facilitate tissue formation. This transition is typically characterized by a decreased expression of adherens junctions responsible for coupling the cells tightly together, thereby permitting an increase in cell motility. In an effort to further define the potential for using ESCs as an in vitro model of early development, researchers have studied the EMT in ESCs of mouse (Spencer et al. 2007), rhesus monkey (Behr et al. 2005; Denker et al. 2007), and human (D'Amour et al. 2005; Eastham et al. 2007; Ullmann et al. 2007). ESCs that were differentiated using various strategies ranging from feeder-free conditions (Ullmann et al. 2007), definitive endoderm formation (D'Amour et al. 2005), monolayer (Eastham et al. 2007), and spontaneous differentiation (Spencer et al. 2007) have been shown to mimic aspects of EMT events, evidenced in part by the loss of E-cadherin and the tight junction-associated proteins, as well as increased expression of N-cadherin and mesenchymal transcription factors. In addition to the differential expression of these cell junction markers and transcription factors, cell differentiation and tissue formation in the developing embryo are accompanied by spatially distinct alterations in the composition of the ECM (Wan et al. 1984; Ffrench-Constant and Hynes 1989; Fenderson et al. 1993). However, specific ECM molecules associated with EMT processes in ESCs have yet to be defined.
ECM molecules such as hyaluronan and versican are critically important in regulating EMT processes in various tissues (Henderson and Copp 1998; Mjaatvedt et al. 1998; Camenisch et al. 2000; Zoltan-Jones et al. 2003; Toole et al. 2005; Kern et al. 2006). Hyaluronan is a high-molecular mass (106–107 Da) glycosamino-glycan (GAG) composed of repeating disaccharide units of glucuronic acid and N-acetylglucosamine that can be synthesized by three independent hyaluronan synthases (HAS), HAS-1, −2, and −3 (Toole 2004). Hyaluronan has been implicated in several morphogenic cell and tissue events involving EMTs, including embryonic development, wound healing, and angiogenesis (West et al. 1985; Foschi et al. 1990; Trabucchi et al. 2002; Schoenfelder and Einspanier 2003; Allison and Grande-Allen 2006). Versican is a chondroitin sulfate proteoglycan found in a variety of soft tissues and is capable of binding to hyaluronan (Wight 2002). Four different isoforms of versican (V0, V1, V2, and V3) exist as a result of alternative splicing of the two exons that encode the GAG attachment domains (α-GAG and β-GAG) in the core protein. Versican affects cell phenotypes associated with EMT processes, including proliferation and migration (Wight 2002), suggesting that hyaluronan and versican together may play functional roles in EMTs occurring during embryonic development. The objective of this study was to examine the temporal and spatial patterns of hyaluronan and versican expression, accumulation, and organization by mouse ESCs (mESCs) undergoing EB differentiation. The presence and spatial distribution of hyaluronan and versican within differentiating EBs was examined histologically, and hyaluronan and versican extracted from EBs were quantified as a function of differentiation time. The temporal gene expression profile of the individual HAS and
Materials and Methods
Cell Culture
mESCs [D3 cell line (ATCC; Manassas, VA)] were cultured on 0.1% gelatin-coated 100-mm polystyrene cell culture dishes (Corning; Corning, NY) with DMEM (Mediatech; Herndon, VA) supplemented with 15% FBS (HyClone; Logan, UT), 2 mM
Histological Analysis of Hyaluronan and Versican
EBs collected at different stages of differentiation (4, 7, or 10 days) were fixed with 10% formalin for 30 min and embedded in aqueous gel (Histogel; Richard-Allan Scientific, Kalamazoo, MI). Each gel block was processed for histological sectioning through a series of xylene and alcohol rinses prior to being embedded in paraffin. Sections of EB samples (5 μm each) were obtained from different depths (a minimum of 90 μm apart) within the paraffin block and stained with hematoxylin and eosin (H and E) using a Leica AutoStainer XL (Wetzlar, Germany). ESCs fixed with formalin on tissue culture plates were similarly stained with H and E, using the same reagents and incubation times as applied by the autostainer protocol. A minimum of three independent experiments, each containing roughly 3000 EBs, were examined by histological analyses. Paraffin-embedded EBs were also stained using Movat's pentachrome staining procedure after deparaffinization and hydration with distilled water. Sections were incubated in reagents with intermittent rinses performed under running water or ultrapure water, as follows: 1% Alcian blue (20 min), alkaline alcohol (60C, 10 min), Weigert's stain (60 min), Croceine scarlet/acid fuchsin (1 min), 5% phosphotungstic acid (5 min), 1% acetic acid (5 min), 95% ethanol [(EtOH) 1 min], 100% EtOH (2 x; 1 min), alcoholic saffron (7 min), and absolute EtOH (2 x; 1 min). Slides were then cleared in xylene and mounted with coverslips, using Mount Quick mounting medium (Daido Sangyo Co., Ltd.; Tokyo, Japan).
For immunohistochemical staining, endogenous peroxidase activity was quenched using 0.75% hydrogen peroxide in 100% methanol (20 min), after which the samples were hydrated in a graded ethanol series (100% EtOH–35% EtOH) and rinsed in PBS (pH 7.3). Slides from each block were then incubated in hyaluronidase reaction buffer [50 mM NaOAc, 150 mM NaCl (pH 6.0)] for 1 hour at 37C, with or without hyaluronidase (hyaluronan lyase from
For versican staining, sections were deparaffinized, rehydrated, and quenched as described above; treated for 1 hr at 37C with ABCase [chondroitin ABC lyase (MP Biomedicals); 0.2 U/ml in enriched Tris buffer stock, at final concentrations of 50 mM Tris, 60 mM NaAc, 50 mM NaCl (pH 8), with 0.1 mg/ml BSA] as an antigen retrieval step to degrade chondroitin sulfate GAGs and expose the versican core protein. Slides were rinsed in TBS and then blocked for 1 hr at room temperature in 5% Carnation nonfat dry milk. Primary antibody [rabbit anti-mouse versican β-GAG domain (Millipore; Billerica, MA); 7 μg/ml in 0.1% BSA in PBS] was added and incubated overnight at 4C. After three PBS rinses, a biotinylated goat anti-rabbit secondary antibody (1:400 in 0.1% BSA/PBS; Invitrogen, Carlsbad, CA) was added for 1 hr at room temperature. Samples were developed with a Vector Elite ABC kit with Vector Red substrate, and slides were counterstained with hematoxylin and mounted with coverslips. Negative control samples were prepared identically, with rabbit IgG isotype control (7 μg/ml in 0.1% BSA in PBS; Vector Laboratories) used in place of primary antibody.
To visualize hyaluronan and versican in the same section for localization studies, an immunofluorescence (IF) assay was performed. Deparaffinized histological sections were blocked for 1 hr using 1.5% normal donkey serum (NDS). Samples were incubated with the primary reagents HABR-B (2.5 μg/ml; Sigma) and versican (5 μg/ml β-GAG domain; Millipore) together for 1 hr at room temperature or overnight at 4C. After three PBS rinses, the samples were incubated with Alexa 488-streptavidin (Invitrogen; 1:400 in 1.5% NDS; hyaluronan) and tetramethylrhodamine isothiocyanate (TRITC)-labeled donkey anti-rabbit secondary antibody (Jackson ImmunoResearch, West Grove, PA; 1:400 in NDS; versican) for 1 hr at room temperature prior to Hoechst (10 μg/ml) counterstaining. Samples from all stained batches were then mounted with coverslips, using either low-viscosity mounting medium (immunohistochemical samples; Cytoseal 60; Richard-Allan Scientific) or aqueous mounting medium with antifading agents (IF samples) (Gel/Mount; Biomeda Corp., Foster City, CA). Note that antigen retrieval with ABCase was omitted from these samples; ABCase has hyaluronidase activity, which completely degraded hyaluronan in the samples (data not shown). Hyaluronidase treatment and incubation with rabbit IgG isotype in place of primary antibody served as negative controls for hyaluronan and versican staining, respectively.
Immunohistochemical Analysis of ADAMTS-5, Pan-cytokeratin, and N-Cadherin
For ADAMTS-5 staining, deparaffinized histological sections were blocked for 1 hr at room temperature, using a 2.5% NDS-2.5% BSA solution in PBS. Samples were incubated with rabbit anti-ADAMTS-5 (JSCKNG, 4 μg/ml in 0.1% BSA-PBS; Affinity BioReagents, Rockford, IL) overnight at 4C (negative IgG control: 4 μg/ml in 0.1% BSA-PBS). After three PBS rinses, the samples were incubated with Alexa Fluor 555 (5 μg/ml in 0.1% BSA-PBS) for 1 hr at room temperature prior to Hoechst (10 μg/ml) counterstaining. Samples were then mounted with coverslips using aqueous mounting medium with antifading agents (Gel/Mount).
For pan-cytokeratin and N-cadherin staining, the required antigen retrieval treatments damaged the versican epitope so that colocalization staining could not be performed. Therefore, adjacent sections of each sample were stained for versican as described above. For pan-cytokeratin, antigen retrieval was performed by incubating sections in 40 μg/ml proteinase-K (Fisher Scientific; Pittsburgh, PA) in 10 mM Tris-HCl, pH 8.0 (6 min, room temperature). Samples were then blocked for 1 hr at room temperature in 2% normal goat serum (NGS), followed by incubation with primary antibody recognizing pan-cytokeratin (Dako, Carpinteria, CA; 1:10 dilution in 2% NGS; skin-positive control) overnight at 4C. For N-cadherin staining, heat-induced epitope retrieval was performed by boiling the samples in 10 mM citrate buffer at pH 6.0 for 20 min. Samples were blocked for 1 hr at room temperature in 2% NGS and then incubated with primary antibody recognizing N-cadherin (BD Biosciences, San Jose, CA; 1:30 dilution in 2% NGS; whole-mouse embryo-positive control) overnight at 4C. Both pan-cytokeratin- and N-cadherin-stained samples were visualized by incubation with FITC-labeled goat anti-mouse secondary antibody (Southern Biotech, Birmingham, AL; 1:100 in 2% NGS) for 1 hr at room temperature prior to Hoechst (10 μg/ml) counterstaining. Samples were then mounted with coverslips by using aqueous mounting medium with antifading agents (IF samples; Gel/Mount; Biomeda Corp.).
Microscopy
Histology sections were imaged using a Nikon 80i microscope and a Spot Flex camera (Diagnostic Instruments, Inc.; Sterling Heights, MI). For each antigen sample examined, images for all samples were acquired using the same exposure settings. Slight linear adjustments in brightness were made across entire images using SPOT advanced software to enhance the contrast between signal and background.
Quantitative Hyaluronan Expression in EBs
Hyaluronan was isolated from ESCs, EBs (50,000 cells per sample;
Western Blotting
Protein was isolated from ESCs and day-4, −7, and −10 EBs with extraction buffer [8 M urea, 2 mM EDTA, 50 mM Tris, 0.5% Triton X-100, 0.25 M NaCl, protease inhibitor cocktail (Roche)]. Protein concentrations were measured using a MicroBCA assay (Pierce; Rockford, IL), and samples were stored at −80C prior to analysis. For versican blots, 300 μg of total protein per sample was applied to diethylaminoethyl (Sephacel; Sigma) columns (0.3-ml bed volume), preequilibrated, and washed with 10 bed volumes of wash buffer (8 M urea, 2 mM EDTA, 50 mM Tris, 0.5% Triton X-100, 0.25 M NaCl). The samples were eluted with 3 bed volumes of wash buffer containing 2 M NaCl. Chondroitin sulfate [50 μg/ml; 10 mg/ml stock (Sigma)] carrier was added, and protein was precipitated from the eluate by adding ethanol (95% ethanol-1.3% potassium acetate) for 2 hr at −80C. Precipitated protein was collected by centrifugation for 5 min at 14,000 rpm and rinsed twice with ethanol. Pellets were air dried and then digested with 3 U/ml chondroitinase (10 U/ml stock; Seikagaku, Tokyo, Japan) in 20 mM Tris, 3.6 mM sodium acetate (pH 8.0) for 3 hr at 37C to remove chondroitin chains from the core proteins. Concentrated sample buffer [50 mM Tris-HCl (pH 6.8), 1% SDS, 10% glycerol, 1.7% (v/v) 2-mercaptoethanol] was added, and the samples were boiled for 3 to 4 min prior to being loaded onto a 15-cm × 12-cm 4% to 12% gradient polyacrylamide gel (3.5% stacking gel) and separated overnight at room temperature. Protein was transferred to nitrocellulose membranes and blocked for 2 hr at room temperature in blocking buffer (2% BSA in Tris-buffered saline, 0.1% Tween-20). Membranes were incubated overnight in blocking buffer containing 2% calf serum and 0.25 μg/ml rabbit anti-mouse versican β-GAG domain antibody (Millipore). Membranes were then washed with TBS-T (Tris-buffered saline with 0.1% Tween-20) and incubated for 2 hr at room temperature [1:20,000 goat anti-rabbit IgG antibody conjugated to alkaline phosphatase in blocking buffer (Jackson ImmunoResearch)]. After blots were extensively washed in TBS-T, they were incubated in assay buffer [0.1 M diethanolamine, 1 mM MgCl2, 0.02% sodium azide (pH 10)] for 10 min, followed by incubation with substrate (1:100 CSPD chemi-luminescent substrate, 1:20 Nitro-Block membrane enhancer in assay buffer; Applied Biosystems, Foster City, CA) for 5 min at room temperature. Blots were exposed to autoradiograph film (ISC BioExpress; Kaysville, UT) and developed in a film processor (Fischer Industries Inc.; Geneva, IL). For DPEAAE blots, equal amounts of protein from each sample were ethanol precipitated and electrophoresed on 8% SDS-PAGE with 3.5% stacking gel. Proteins were transferred to nitrocellulose, probed with DPEAAE antibody (1 μg/ml; Affinity Bioreagents, Golden, CO), and detected using an enhanced chemiluminescence kit as described above. As a loading control, the same blot was also probed with an anti-actin N-terminal antibody (Sigma).
Each lane on the Western blots shown represents protein lysates from an EB culture collected at the designated time point. Lysates from three separate cultures per time point were examined for each independent experiment.
Quantitative RT-PCR (qRT-PCR)
RNA was extracted from undifferentiated ESCs and EBs at days 4, 7, and 10 of differentiation using an RNeasy mini-kit (Qiagen; Valencia, CA). Complimentary DNA was reverse transcribed from 1 μg of total RNA, using an iScript cDNA synthesis kit (Bio-Rad; Hercules, CA), and real-time RT-PCR was performed using SYBR Green technology with a MyiQ cycler (Bio-Rad). Beacon designer software was used to design forward and reverse primers (Invitrogen) (see Supplemental Table ST1) for the pluripotency markers
For SuperArray RT2 Profiler PCR array analysis, cDNA synthesis was performed using a SuperArray RT2 First Strand kit (SABiosciences; Frederick, MD). First, the genomic DNA elimination mixture was prepared by mixing 0.5 μg of RNA with the 5x genomic DNA elimination buffer and RNase-free water and incubated at 42C for 5 min. The RT cocktail (5x RT buffer 3, primer and external control mixture, RT enzyme mixture 3, and RNase-free water) was prepared (10 μl) and added to the elimination buffer mixture. Each sample was then placed in an iCycler thermal cycler (Bio-Rad) for synthesis (15 min at 42C, 5 min at 95C) and diluted with RNase-free water. RT-PCR was performed by first preparing the experimental cocktail (1275 μl of 2x SuperArray RT2 qPCR Master Mix, 102 μl of diluted first-strand cDNA synthesis reaction, 1173 μl of RNase-free water) and then distributing it across all wells of the PCR 96-well array. The array was tightly sealed with optical thin-wall 8-cap strips and run in a MyiQ cycler (Bio-Rad) with a two-step cycling program (1 cycle of 10 min at 95C; 40 cycles of 15 sec at 95C; and 40 cycles of 1 min at 60C) and melt curve. Fold changes in gene expression were analyzed using the threshold cycle (ΔΔCt) method of quantitation, whereby samples of EBs from different time points (days 4, 7, and 10) were compared to undifferentiated ESC values after individual sample values were normalized to GAPDH levels.
Statistics
For paired comparisons, Student
Results
Embryoid Body Differentiation
Adherent colonies of undifferentiated mESCs were dissociated into single cells and reaggregated in suspension culture to form cell spheroids constituting putative EBs (see Supplemental Figure SF1). As expected, the resulting EBs increased in size and adopted a more differentiated morphology over the course of differentiation, indicated by the formation of a primitive endoderm layer on the periphery of EBs (shown by solid arrows in Supplemental Figure SF1), as well as cystic cavities (hollow arrows, Supplemental Figure SF1) that developed in the interior of the EBs. Phenotypic differentiation of the EBs was evident as a reduction in the expression of the
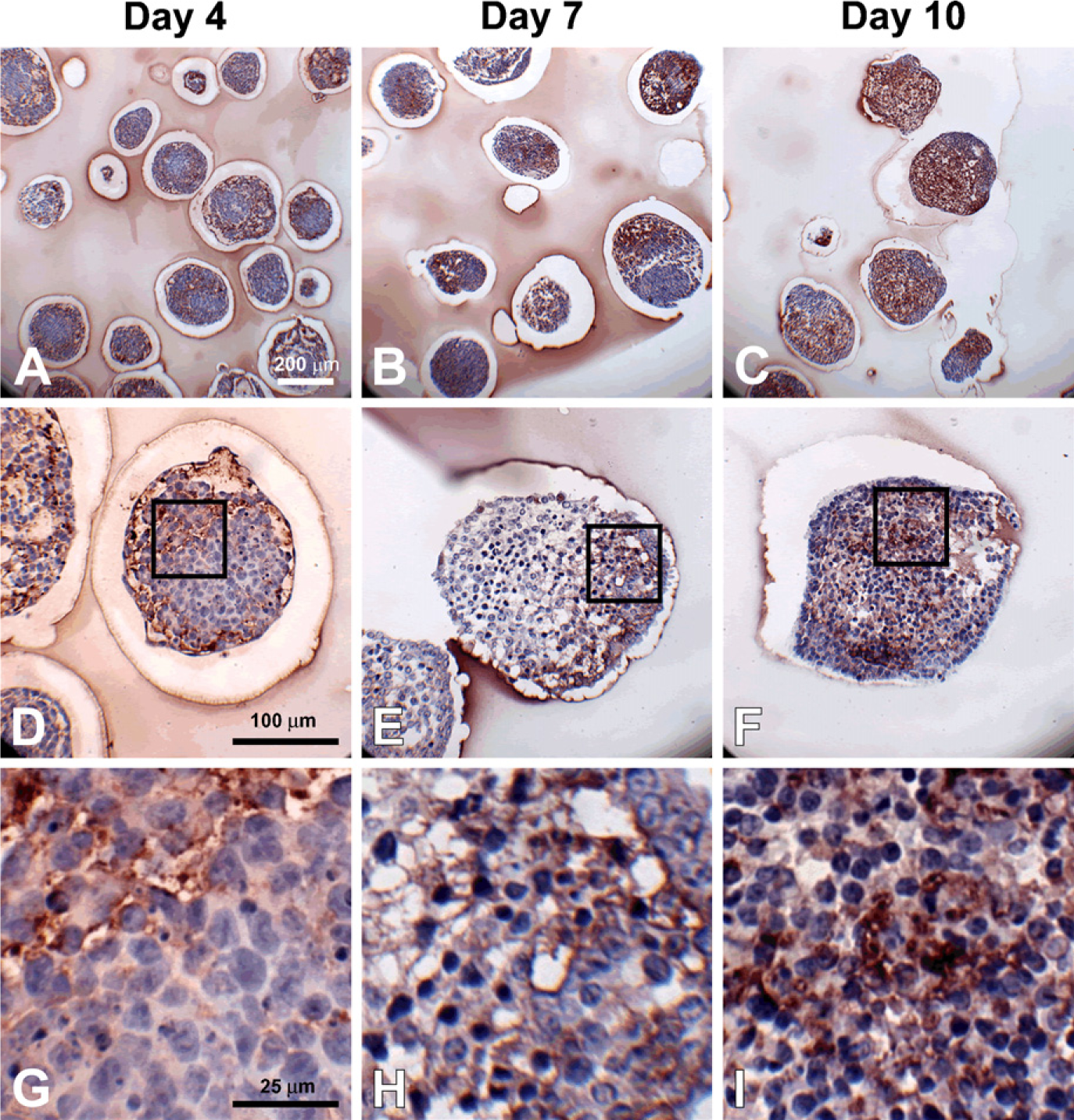
Hyaluronan accumulation during embryoid body (EB) formation. Sections (5 μm) from day 4, 7, and 10 EBs were stained with hyaluronan binding protein (dark red color); hematoxylin-stained nuclei are shown in blue. Hyaluronan was increasingly prominent with differentiation time (
Hyaluronan and Versican Accumulation and Distribution Within Differentiating EBs
To specifically examine the spatial distribution of hyaluronan and versican within EBs, immunohisto-chemistry was performed to examine EBs at different stages of differentiation (days 4, 7, and 10). After only 4 days of differentiation, very little hyaluronan was detected in EBs (Figures 1A, 1D, and 1G), but that which was present was predominantly localized around the periphery of most of the cell aggregates. By differentiation day 7, hyaluronan was clearly detectable more extensively throughout EBs and most often appeared to be enriched primarily on one side of an EB (Figures 1B, 1E, and 1H). After day 10, hyaluronan was found to be distributed throughout almost the entire interior of EBs (Figures 1C, 1F, and 1I). Interestingly, the accumulation of hyaluronan appeared to be greatest in areas of lower cell density within EBs. Similarly, versican (detected with an antibody recognizing the V0/V1 isoforms of versican) was minimally present within EBs at day 4 of differentiation (Figures 2A, 2D, and 2G) but was distributed throughout a large portion of EBs by day 7 of differentiation (Figures 2B, 2E, and 2H). After 10 days of differentiation, versican was detected throughout the majority of EBs and appeared most concentrated within less dense areas of cells (Figures 2C, 2F, and 2I), in patterns similar to those observed for hyaluronan localization. Thus, the expression levels of hyaluronan and versican within EBs exhibited similar temporal and spatial patterns over the course of differentiation.
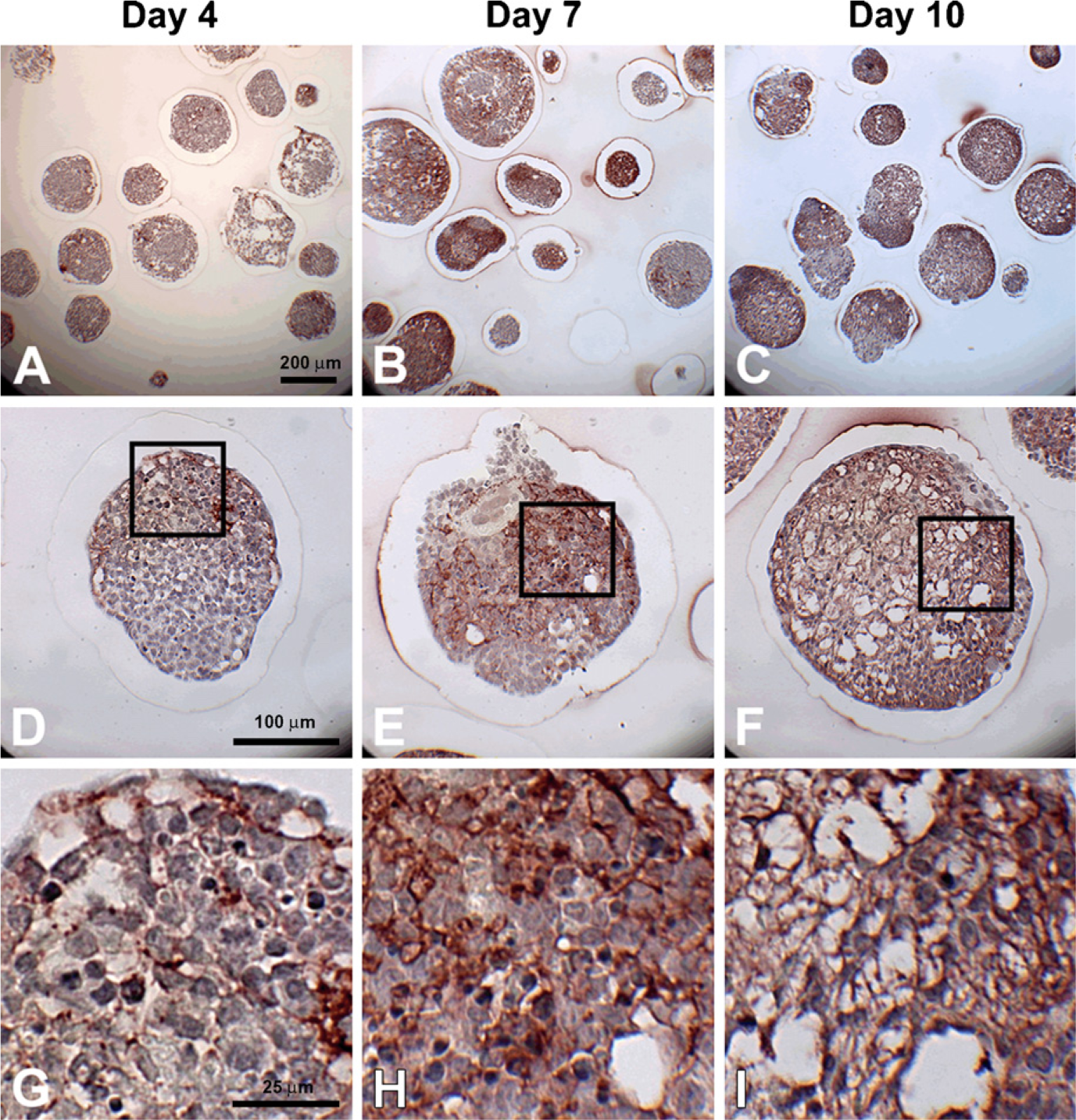
Versican accumulation during EB formation. Sections (5 μm) from day 4, 7, and 10 EBs were stained with anti-versican antibody (dark red color); hematoxylin-stained nuclei are shown in blue. Some versican expression can be seen by day 4 (
Accumulation of Hyaluronan During EB Formation
To quantify the amount of hyaluronan synthesized and retained within EBs and released into the medium over the course of differentiation, hyaluronan produced by EBs was assessed using a hyaluronan binding protein quantification kit (Figure 3). Hyaluronan accumulated within EBs as a function of time, such that the relative amount of hyaluronan by day 10 of differentiation increased significantly compared with that of earlier time points (ESCs,
Versican Accumulation and Proteolytic Processing During EB Formation
To further analyze versican synthesis and accumulation by differentiating EBs, proteoglycans were isolated and examined by Western blot analysis using a primary anti-mouse versican β-GAG domain antibody, which detects only the V0 and V1 versican isoforms. Isoforms V0 and V1 usually appear as two high-molecular-mass products that migrate in the 350- to 450-kDa range on SDS-PAGE (Sandy et al. 2001; Kenagy et al. 2006; Seidelmann et al. 2008). For ESCs or EBs after 4 days of differentiation, little, if any, immunoreactivity appeared in the gels above the 250 molecular weight marker where V0 and V1 usually run (Figure 4A). However, by day 7, there was a distinct increase in the amount of protein recognized by the β-GAG antibody, which was further increased at day 10 (Figure 4A). Several bands also appeared near or below the 250-molecular-weight marker in the 10-day EB cultures, most likely representing degradation products of versican (Sandy et al. 2001; Kenagy et al. 2006; Seidelmann et al. 2008). Therefore, to assess whether versican proteolysis occurred during EB differentiation, immunoblotting was performed using an antibody that specifically recognizes DPEAAE, a cleavage neoepitope of versican generated by specific members of the ADAMTS family of enzymes (Sandy et al. 2001). The neoepitope was not detected in undifferentiated ESCs but appeared at day 4 of differentiation and increased in signal intensity through day 10 (Figure 4B). Similarly, gene expression of
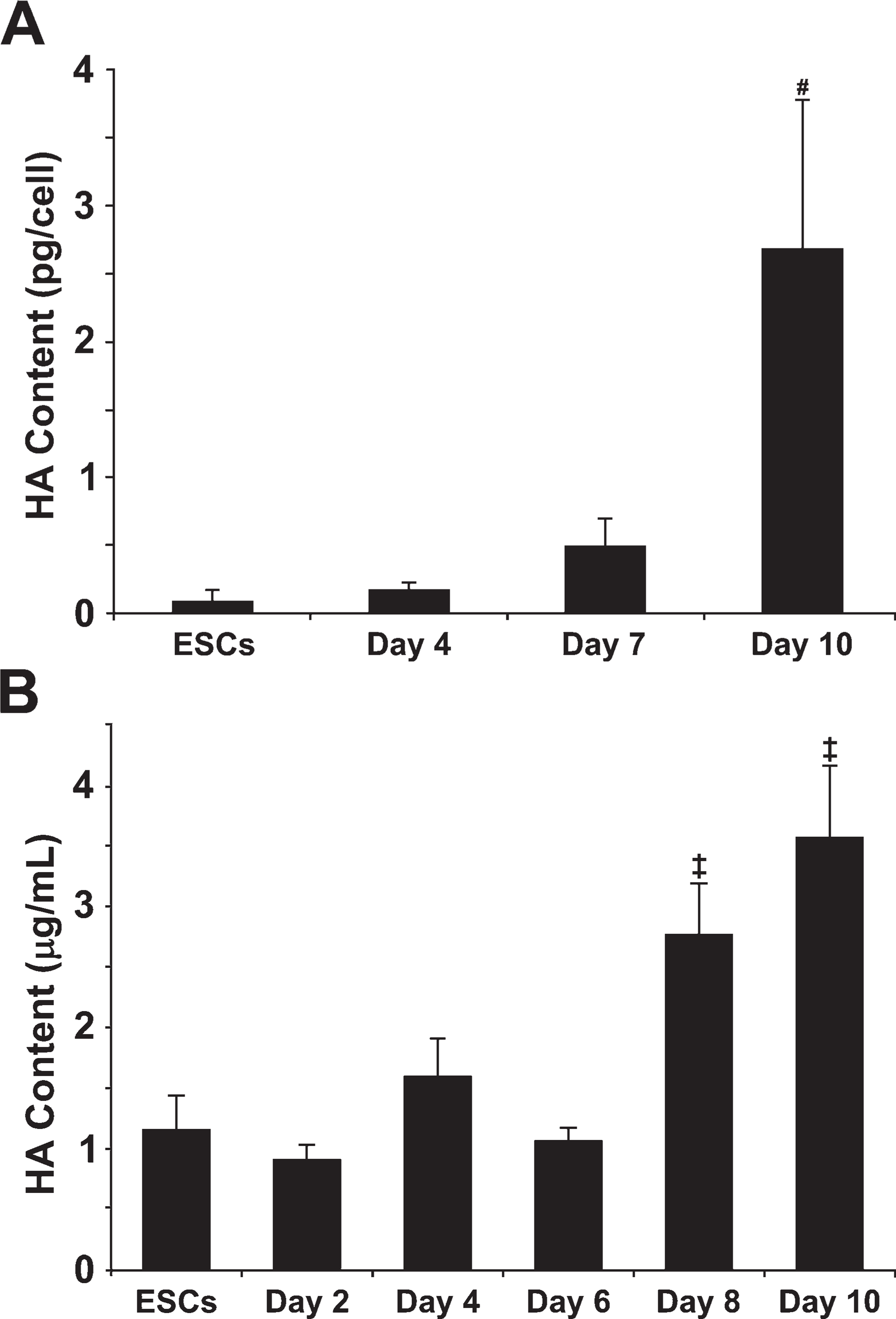
Quantification of hyaluronan synthesis by EBs. A quantitative hyaluronan (HA)-binding protein-based kit was used to measure hyaluronan retained within embryonic stem cells (ESCs) and day 4, 7, and 10 EBs (
Hyaluronan Synthase and Versican Gene Expression
The relative expression levels of the different HAS genes remained fairly consistent over the course of early EB differentiation (Figure 5A), with expression of
Similar to the patterns of HAS expression, the relative expression levels of the different versican isoforms were consistent over time (Figure 5B). Undifferentiated ESCs and differentiating EBs expressed relatively high levels of
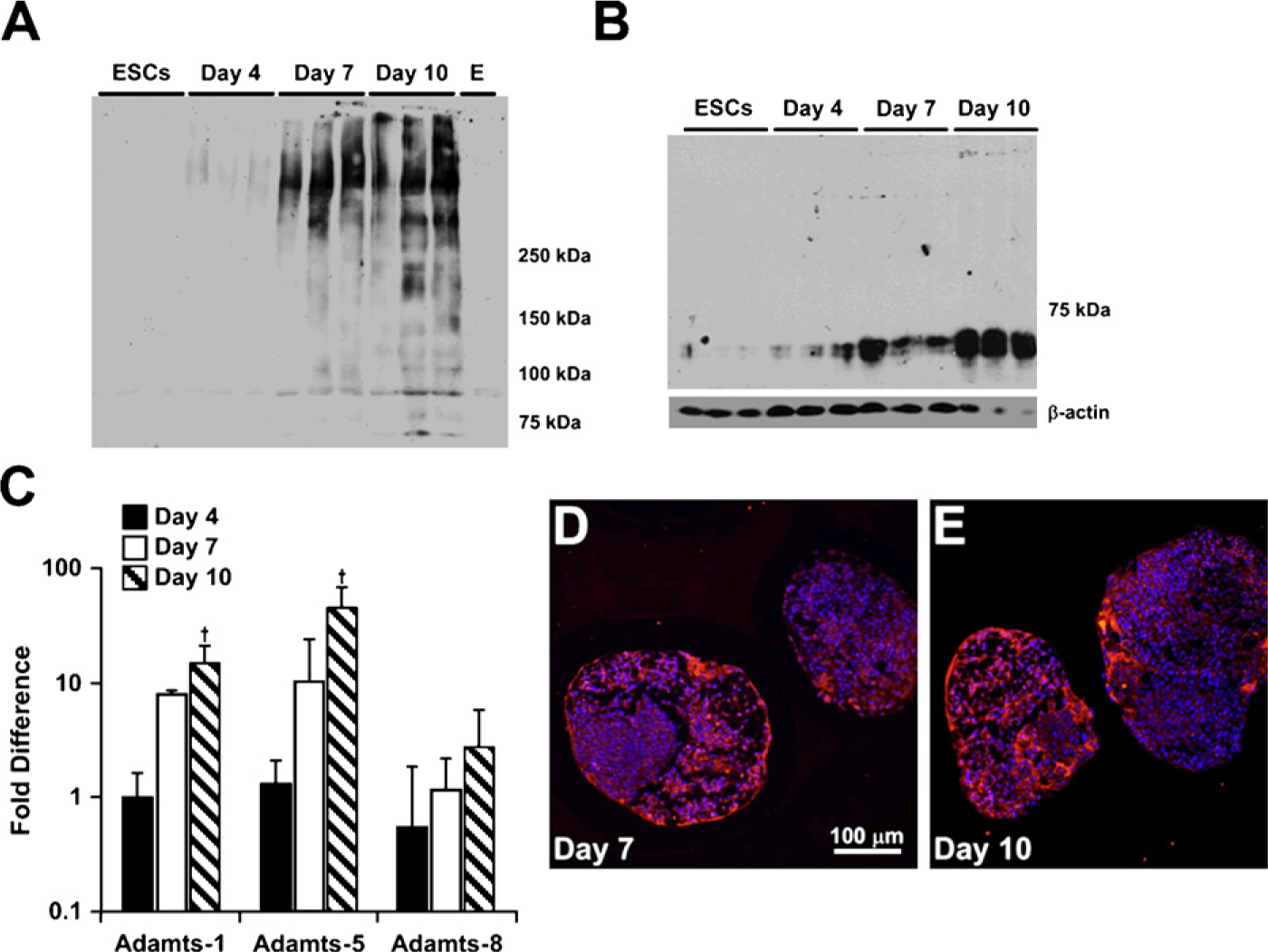
Analysis of versican synthesis and proteolysis in differentiating EBs. Purified, chondroitinase-treated protein lysates from triplicate samples of ESCs and day 4, 7, and 10 EBs were probed with rabbit anti-mouse versican β-GAG domain antibody (
Hyaluronan and Versican Localization
Immunofluorescent affinity labeling of both hyaluronan and versican within the same sections confirmed that hyaluronan and versican were not detectable in most EBs at 4 days of differentiation, although when present, hyaluronan localized in the same areas as versican (Figures 6A, 6D, 6G, and 6J). By day 7, hyaluronan and versican were distributed in regions of the EBs that primarily contained a lower density of cells (Figures 6B, 6E, 6H, and 6K). The appearance of a largely fibrillar network of overlapping hyaluronan and versican was clearly distinguishable by 10 days of differentiation throughout much of the interior of EBs (Figures 6C, 6F, 6I, and 6L). Thus, at each of the time points examined, hyaluronan and versican were consistently distributed in a similar spatial pattern within differentiating EBs.
Epithelial and Mesenchymal Phenotypes Within EBs
Based on the observed accumulation and overlapping patterns of hyaluronan and versican expression in EBs during differentiation, as well as differences in cell morphology in regions containing hyaluronan and versican, the spatial distribution of epithelial [cytokeratin-positive (cytokeratin+)] and mesenchymal [N-cadherin-positive (N-cadherin +)] cells within EBs were examined relative to versican-rich regions. After 10 days of differentiation, EBs clearly contained distinct populations of cytokeratin+ and N-cadherin1 cells. Cytokeratin+ cells were primarily localized around the periphery of EBs, and versican appeared to be completely excluded from such areas within the same EB (Figures 7A and 7B). In contrast, N-cadherin+ cells were predominantly found within the interior of EBs in regions that overlapped with the presence of versican (Figures 7C and 7D).
Discussion
These studies provide evidence that spatial and temporal patterns of hyaluronan and versican accumulation and organization by ESCs within EBs are associated with epithelial–mesenchymal transitions occurring within the aggregates of differentiating cells. ESCs express several HAS and versican isoforms throughout the course of EB differentiation, concomitant with an increasing accumulation of hyaluronan and versican over the first 10 days of differentiation. The overlapping spatial patterns of expression suggest that hyaluronan and versican are physically associated within EBs and moreover that the overlapping patterns of accumulation of these molecules are found predominantly in regions of EBs containing cells actively undergoing EMT differentiation. Overall, these results not only suggest a specific role for hyaluronan and versican in the morphogenesis of primitive pluripotent stem cells but also demonstrate that dynamic changes in the endogenous expression, accumulation, and processing of ECM molecules by differentiating ESCs occur coincidentally with cell phenotype changes. These results indicate that an improved understanding of the molecular composition of embryonic extracellular matrices could yield new insights into developmental biology and provide novel strategies for the directed differentiation of stem cells.
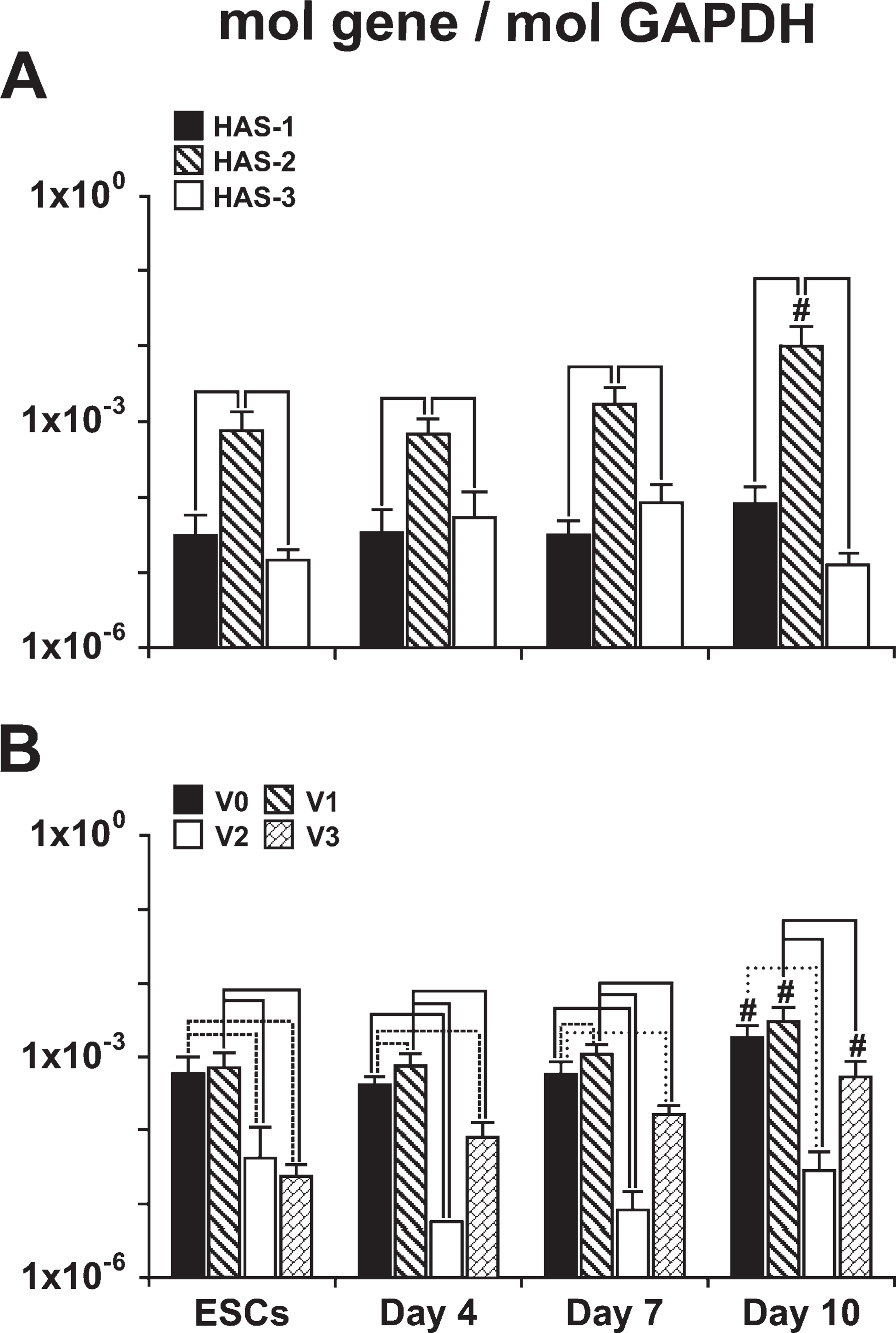
Quantification of hyaluronan synthase (HAS) and versican gene expression. qRT-PCR was used to compare expression levels of HAS isoforms 1, 2, and 3 and versican splice variants V0, V1, V2, and V3 during the course of EB differentiation.
The most significant and novel finding of the present study is that the timing and spatial distribution of hyaluronan and versican expression within EBs correlate with the appearance and localization of mesenchymal (N-cadherin+) cell phenotypes and is excluded from regions of epithelial (cytokeratin+) cells (Figure 7). Previous studies have demonstrated that ESC differentiation within EBs recapitulates many aspects of gastrulation, including the transformation of epithelial pluripotent cells (resembling primitive ectoderm) into migratory mesenchymal cells (resembling primitive streak) (Behr et al. 2005; D'Amour et al. 2005; Denker et al. 2007; Eastham et al. 2007; Spencer et al. 2007; Ullmann et al. 2007). Thus far, however, dynamic changes in prevalent ECM molecules produced within EBs, such as hyaluronan and versican, have not been directly examined or correlated with distinct populations of cells. It has been previously demonstrated that coincident expression of hyaluronan and versican can stimulate cell migration by creating a viscoelastic, hyaluronan-versican-rich, malleable extracellular environment (Wight 2002; Wight and Merrilees 2004; Allison and Grande-Allen 2006; Ricciardelli et al. 2007). Furthermore, a hyaluronan–versican matrix may influence cell fate by enriching for signaling molecules and growth factors that regulate cell proliferation and differentiation. The importance of hyaluronan and versican in directing EMT processes is clear from a number of examples in cell and developmental biology. Whereas HAS-2−/- mouse embryos fail to undergo EMT during cardiac morphogenesis (Camenisch et al. 2000), overexpression of hyaluronan by HAS-2 recombinant adenoviral infection of normal epithelial cells induces an EMT (Zoltan-Jones et al. 2003). In addition, versican plays an essential role in the EMT of the endocardial mesenchymal cushion (Mjaatvedt et al. 1998; Kern et al. 2006), as well as mesenchymal condensation and hair induction (Kishimoto et al. 1999). Thus, hyaluronan and versican appear to regulate the transformation of epithelial cells to mesenchymal phe-notypes at various stages of development.
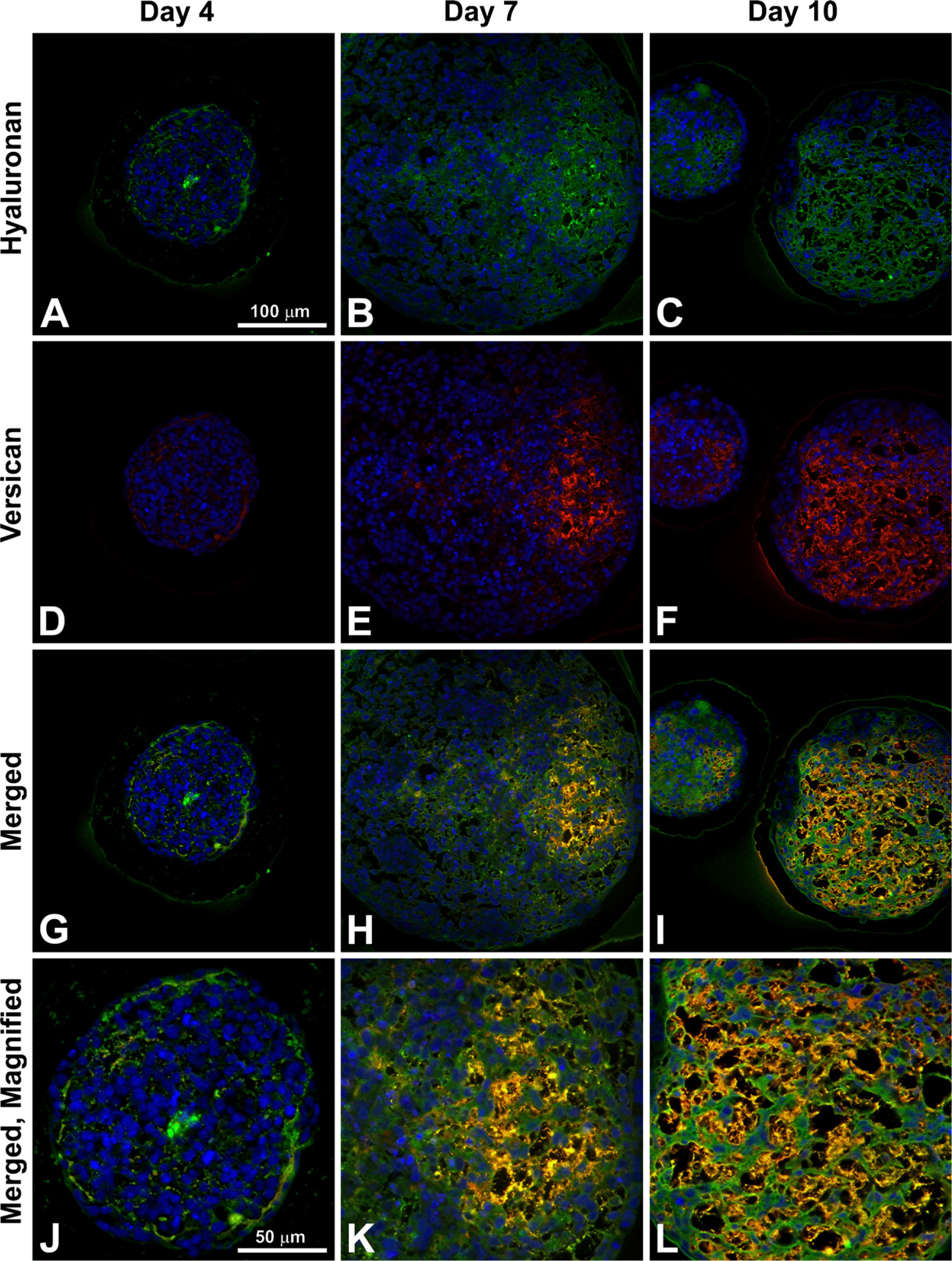
Hyaluronan and versican spatial distributions. EBs were stained for both hyaluronan (green;
As noted above, the expression of HAS isoforms, in particular HAS-2, and the synthesis of hyaluronan play important roles in early embryological development of numerous tissues. Although targeted inactivation of both the
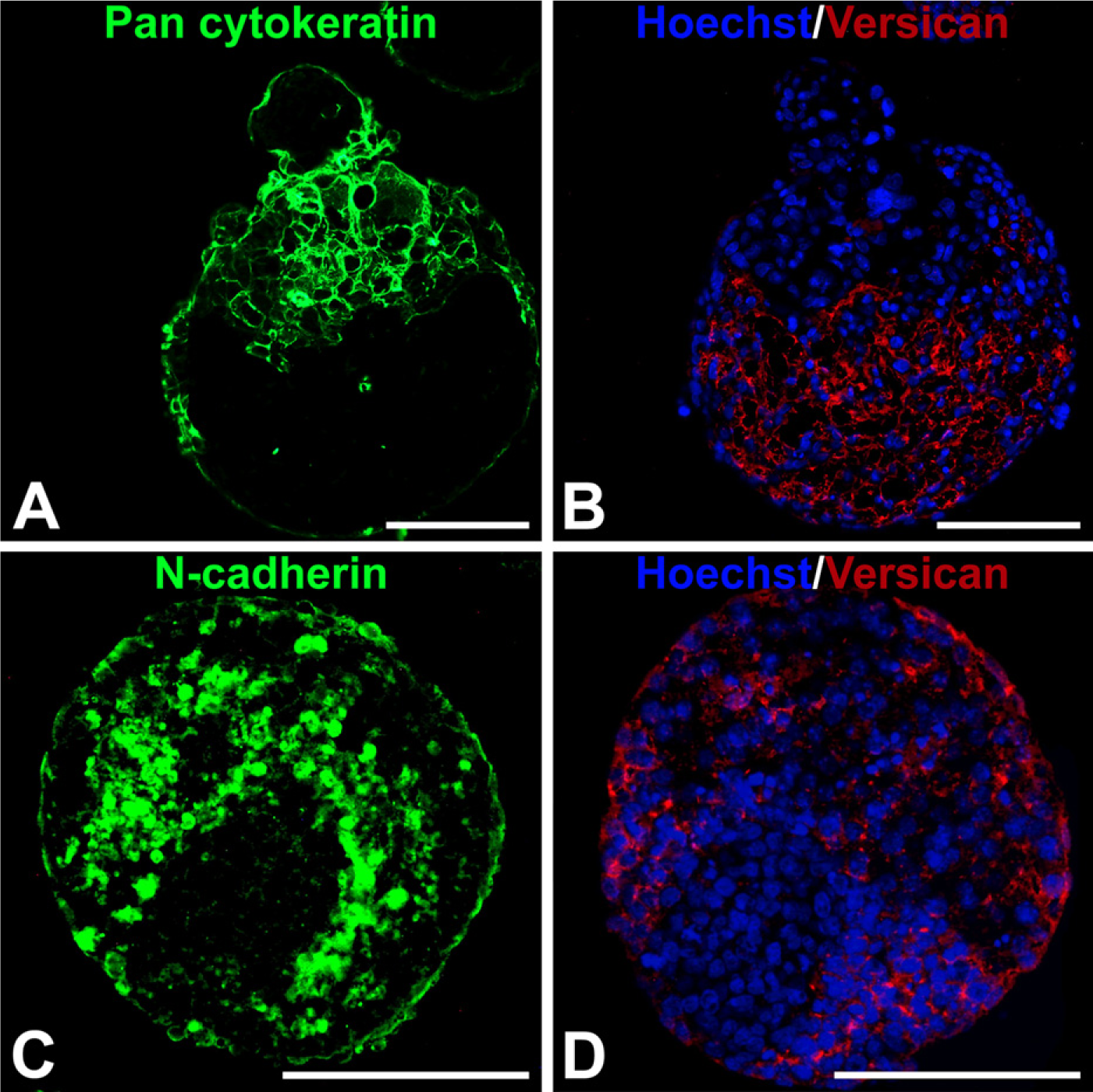
Cellular expression of epithelial–mesenchymal phenotype markers relative to versican localization. Adjacent 5-μm sections from day 10 EBs were fluorescently stained with pan-cytokeratin or N-cadherin primary antibody (FITC-labeled secondary, green) and versican (isoforms V0 and V1; TRITC-conjugated secondary antibody) (red); nuclei were counterstained with Hoechst dye (blue). Cytokeratin localized primarily in the periphery of the EBs, with some evidence of invasion into the EB interiors, while versican was localized exclusively in cytokeratin-negative areas (
In addition to changes in HAS expression and hyaluronan synthesis, differentiating ESCs also exhibit temporal changes in versican isoform expression, associated with increasing versican content within EBs over the course of differentiation. In the developing embryo, versican plays an important role in early cell migration events during neural crest formation and cardiac morphogenesis (Henderson and Copp 1998; Dutt et al. 2006); thus, it is not surprising that versican is expressed by EBs undergoing similar differentiation processes in vitro. Compared with several other hyaluronan-binding proteoglycans, including aggrecan, neurocan, and brevican, previous work demonstrated that the transcript of versican was the only transcript to show a considerable enhancement in EBs compared with ESCs (Nairn et al. 2007). In the present study, however, gene expression of each of the four different versican isoforms was independently examined. Isoforms
Differentiating ESCs offer a unique model for studying various aspects of developmental biology. Analyzing the dynamics of ECM synthesis, accumulation, and organization during development may yield new insights into the molecular regulatory mechanisms responsible for stimulating morphogenic processes, such as EMT, which is important for embryonic cell differentiation and organ development. In a broader context, these results also indicate that phenotypic differences between differentiating ESCs may be associated with the local composition of the ECM and, therefore, regulation of endogenous ECM production within EBs could provide a novel approach to directing ESC fate.
Footnotes
Acknowledgements
This research was funded by the Georgia Tech/Emory Center for the Engineering of Living Tissues (GTEC, National Science Foundation EEC-9731463), the National Institutes of Health (NIH) (R21 EB007316), and the American Heart Association (0665265B). R.N. is supported by a National Science Foundation fellowship. T.N.W. (R24 HL64387-06A1) and M.W.R. (F32 HL083593) were also supported by the NIH.
The authors thank Elizabeth Krauth at the Georgia Institute of Technology, Atlanta, GA, for technical assistance.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
