Abstract
Patients with diabetes are at substantially increased risk for atherosclerosis and clinical cardiovascular events. Because arterial extracellular matrix contains several molecules, including biglycan, versican, hyaluronan, and elastin, that may affect plaque lipid retention and stability, we determined whether diabetes affects plaque content of these molecules in a porcine model of hyperlipidemia and diabetes. Coronary artery sections were studied from non-diabetic normolipidemic (n=11, N-NL), diabetic normolipidemic (n=10, DM-NL), non-diabetic hyperlipidemic (n=16, N-HL), and diabetic hyperlipidemic (n=15, DM-HL) animals. Hyaluronan, biglycan, versican, and apolipoprotein B (apoB) were detected with mono-specific peptides or antisera, and elastin with Movat's pentachrome stain, and contents of each were quantified by computer-assisted morphometry. In the hyperlipidemic groups, diabetes was associated with a 4-fold increase in intimal area, with strong correlations between intimal area and immunostained areas for hyaluronan (R 2 = 0.83, p<0.0001), biglycan (R 2 = 0.72, p<0.0001), and apoB (R 2 = 0.23, p=0.0069). In contrast, median (interquartile range) intimal elastin content was significantly lower with diabetes [N-HL: 5.2% (2.4–8.2%) vs DM-HL: 1.5% (0.5–4.2%), p=0.01], and there was a strong negative correlation between intimal total and elastin areas (Spearman r = −0.62, p=0.001). In this porcine model, diabetes was associated with multiple extracellular matrix changes that have been associated with increased lesion instability, greater atherogenic lipoprotein retention, and accelerated atherogenesis.
A
In contrast, a porcine model recently has been described in which diabetes reproducibly and dramatically increases atherosclerotic lesion formation (Gerrity et al. 2001; Suzuki et al. 2001). This model provides an excellent opportunity to study how diabetes, with or without diet-induced hyperlipidemia, affects extracellular matrix composition in coronary artery lesions. The extracellular matrix plays several key roles in atherogenesis. Hyaluronan, an extracellular matrix glycosaminoglycan, is increased in amount in atherosclerotic and restenotic lesions (Riessen et al. 1996) and has been implicated in tissue macrophage recruitment in other inflammatory diseases (de La Motte et al. 1999). The extracellular matrix proteoglycan, biglycan, may mediate plaque lipoprotein retention (O'Brien et al. 1998, 2004) and also accounts for a large proportion of lesion mass (Gutierrez et al. 1997). Biglycan also may be directly proinflammatory, inasmuch as it recently has been demonstrated to activate macrophages by binding to Toll-like receptors 2 and 4 (Schaefer et al. 2005). Finally, elastin is a major arterial structural protein, and a number of recent studies have suggested that either decreased elastin synthesis (Li et al. 1998a, b) or increased elastin degradation (Liu et al. 2004; Luttun et al. 2004) may promote atherosclerosis.
Thus, the purpose of this study was to characterize, in a porcine model in which diabetes accelerates atherosclerosis, how diabetes and hyperlipidemia affect lesion contents of specific extracellular matrix components, specifically hyaluronan, biglycan, apoB-containing lipoproteins, and elastin.
Materials and Methods
Animals
Coronary arteries were obtained as described previously (Gerrity et al. 2001; Suzuki et al. 2001). Briefly, following weaning, male Yorkshire swine (Clemson University School of Veterinary Medicine, Clemson, SC) were divided into one of four groups: (1) those fed a normal swine chow diet (N-NL), (2) those fed a normal swine chow diet, but made diabetic by daily injections of streptozotocin at 50 mg/kg/dose for 3 days (DM-NL), (3) those fed a high-fat, high-cholesterol diet containing 15% lard and 1.5% cholesterol (N-HL), and (4) those fed the high-fat, high-cholesterol diet and made diabetic by streptozotocin injections (DM-HL). Animals were sacrificed at 20 weeks of age. Proximal coronary arteries were fixed in formalin or methanol Carnoy's solution (60% methanol/30% chloroform/10% glacial acetic acid) and embedded in paraffin wax. In most cases, a segment of the right coronary artery and a segment of the left coronary artery were studied from each animal. Numbers of coronary arteries studied from each group were: N-NL = 11, DM-NL = 10, N-HL516, and DM-HL = 15. The Institutional Animal Care and Use Committee of the Medical College of Georgia approved all animal procedures.
Histological and Immunohistochemical Staining
Serial 6-μm-thick tissue sections were mounted on glass slides, deparaffinized, and then stained with Movat's pentachrome stain for morphology. Immunohistochemistry was performed on adjacent sections using rabbit polyclonal antisera raised against human biglycan (LF-51) [(titer = 1:1000) (O'Brien et al. 1998) (a kind gift of Dr. Larry Fisher, Craniofacial and Skeletal Disease Branch, National Institutes of Dental Research, National Institutes of Health, Bethesda, MD)], or human apoB [(titer = 1:1000) (a kind gift of Dr. Thomas Innerarity, Gladstone Institute, San Francisco, CA)]. Macrophages were detected using monoclonal antibody HB 142.1 [(titer = neat; American Type Culture Collection, Rockville, MD) (Suzuki et al. 2001)]. Hyaluronan was detected using a specific hyaluronan-binding peptide as previously described (Underhill et al. 1993). Versican was detected using a mouse monoclonal antibody raised against human versican (Clone 2-B-1, concentration = 2 μg/ml; catalog # 270428-1, Seikagaku Corporation, Tokyo, Japan) (Sobue et al. 1989).
Single-label immunohistochemistry was performed as described previously (O'Brien et al. 1993, 1995, 1996, 1998). Briefly, tissue sections were deparaffinized using xylene and rehydrated with graded alcohols. The slides were blocked with 3% H202 for 5 min, washed with PBS, incubated with the primary antibody for 60 min or overnight, and then washed again with PBS. A biotinylated anti-rabbit secondary antibody was then applied for 30 min, followed by an avidin-biotin-peroxidase conjugate (ABC Elite; Vector Laboratories, Burlingame, CA) for 30 min. The standard peroxidase enzyme substrate 3,3′-diaminobenzidine was added without nickel chloride to yield a brown reaction product or with nickel chloride to yield a black reaction product. The slides were counterstained with either methyl green or hematoxylin to identify cell nuclei. Negative controls included substitution of primary antiserum or antibody with PBS, normal rabbit serum, or irrelevant monoclonal antibodies. Control for hyaluronan staining was predigestion of sections with Streptomyces hyaluronidases (Riessen et al. 1996).
Morphometric Analyses
Digital images were captured using a Nikon CoolPix 990 camera mounted on an Olympus microscope and then transferred to a 2 GHz Pentium 4 computer. Total lesion area, and areas for hyaluronan, biglycan, apoB, and elastin were determined using the ImagePro Plus morphometric analysis program (Version 4.5.1; MediaCybernetics, Silver Spring, MD).
Statistical Analyses
Statistical analyses were performed using GraphPad Prism 3.0 software (GraphPad Prism, Inc.; San Diego, CA). Groups of data with non-Gaussian distributions were compared using the Mann-Whitney U test, and summary data were reported as median and interquartile range (IQR) values. Student's t-test was used to compare groups of data with normal distributions, and summary data were reported as mean values ± standard errors (±SE). A p value <0.05 was considered statistically significant. Pearson correlation coefficients were calculated for groups of data with normal distributions, and Spearmen correlation coefficients were calculated for groups of data with non-Gaussian distributions.
Results
Total Intimal (“Lesion”) Areas
Effect of Diabetes in Normolipidemic Groups. The intimal areas of each coronary arterial cross-section were determined using Movat's pentachrome-stained sections, on which the black elastin staining made it possible to readily identify the internal elastic lamina. Within each of the four groups (N-NL, DM-NL, N-HL, and DM-HL), intimal areas did not follow a normal distribution, so groups were compared using the non-parametric Mann-Whitney U test. In the non-diabetic, normolipidemic group (N-NL), the median intimal area was very small [median (IQR): 0 (0–20,690) μm2]. With diabetes alone (DM-NL), median intimal area increased to 21,390 (0–57,980) μm2, but this difference did not reach statistical significance (p=0.21 vs N-NL), probably due to the relatively small numbers of arteries in each group. Also, this increase in intimal area was not due to formation of classical atherosclerotic lesions, inasmuch as neither lipid accumulation nor macrophage infiltration was detected.
Effect of Diabetes in Hyperlipidemic Groups. Atherosclerotic lesions were seen in the two hyperlipidemic groups. Total cholesterol levels (mean ± SD) seen with the high-fat, high-cholesterol diet did not differ between the non-diabetic (N-HL: 491 ± 96 mg/dl) and diabetic (DM-HL: 555 ± 111 mg/dl) groups (p=0.19). However, blood glucose levels (mean ± SD) were much higher in hyperlipidemic animals with diabetes (DM-HL: 351 ± 76 mg/dl) than in animals with hyperlipidemia alone (N-HL: 84 ± 17 mg/dl) (p<0.0001). In the non-diabetic group (N-HL) median (IQR) intimal area was 20,460 (11,250–69,730) μm2. However, superimposition of diabetes on hyperlipidemia (DM-HL) was associated with a significant 6-fold increase in median (IQR) intimal area to 122,400 (44,780–421,200) μm2 (p=0.014 vs N-HL). The results for lesion areas for all four groups are summarized in Table 1.
Effect of Diabetes and Hyperlipidemia on Hyaluronan Accumulation
Effect of Diabetes in Normolipidemic Groups. The increased intimal areas in the DM-NL group were due primarily to a prominent accumulation of extracellular matrix that stained blue with Movat's pentachrome stain, indicating accumulation of glycosaminoglycans. The increased content of glycosaminoglycans also was present in the upper media (Figures 1A and 1B). These areas stained intensely for hyaluronan (Figures 1C and 1D). There was a trend toward higher intimal hyaluronan content, expressed as percent intimal area [median (IQR)], in the diabetic, as compared with the non-diabetic normolipidemic groups [DM-NL: 33.1% (0–51.8%) vs N-NL: 0% (0–14.8%)], although the difference did not reach statistical significance (p=0.095).
Effect of diabetes on lesion areas in normolipidemic and hyperlipidemic animals
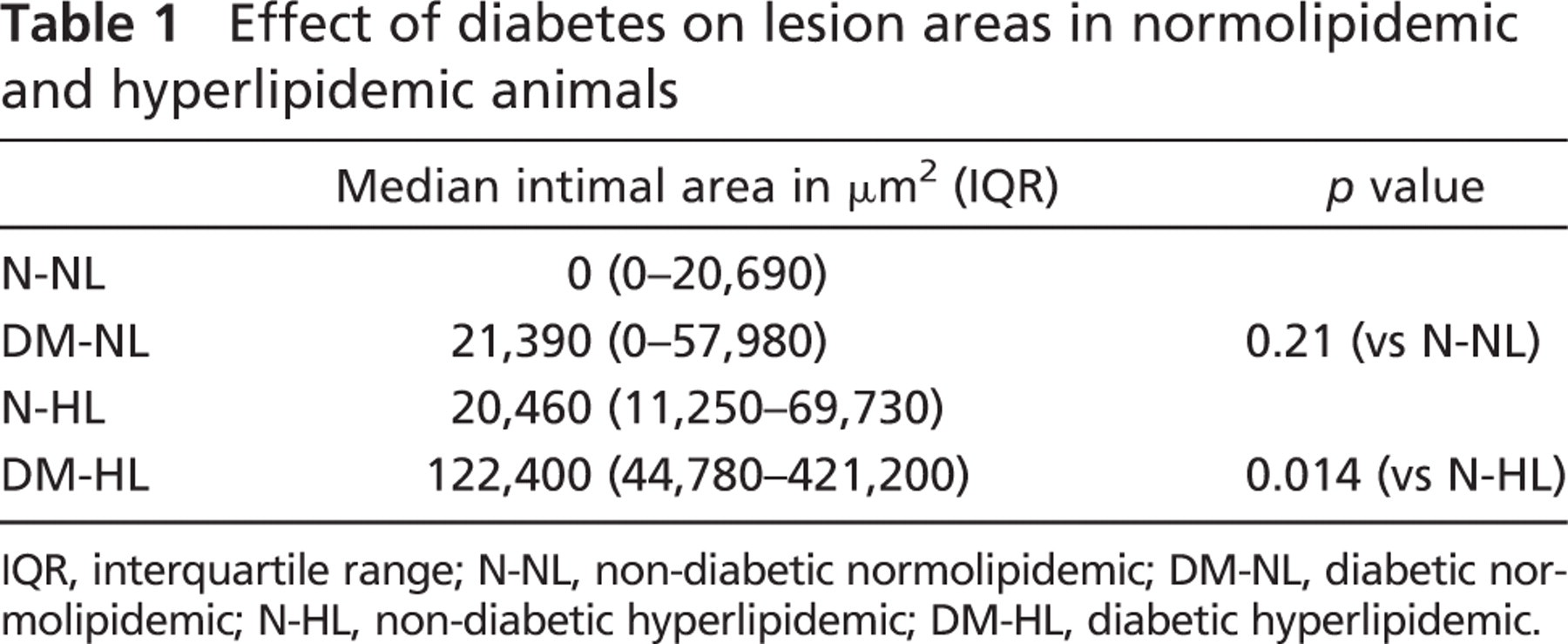
IQR, interquartile range; N-NL, non-diabetic normolipidemic; DM-NL, diabetic normolipidemic; N-HL, non-diabetic hyperlipidemic; DM-HL, diabetic hyperlipidemic.
Effect of Diabetes in Hyperlipidemic Groups. Hyperlipidemia alone substantially increased intimal hyaluronan accumulation in both nondiabetic (N-HL) and diabetic (DM-HL) animals (Figure 2, right panels). However, when expressed as a percent of total lesion area, the median (IQR) hyaluronan contents of non-diabetic and diabetic hyperlipidemic lesions were identical [NHL: 38.5% (23.0–53.2%) vs 40.3% (26.2–62.1%), p=0.86] (Figure 3A). However, there was a strong correlation between intimal area and hyaluronan content for both the non-diabetic and diabetic hyperlipidemic lesions (R 2 = 0.83, p<0.01) (Figure 3B), suggesting that the increased intimal area seen in diabetic lesions is associated with a proportionate increase in hyaluronan.
Effect of Diabetes on Biglycan
Effect of Diabetes in Normolipidemic Groups. Biglycan was not detected in the intima of N-NL or DM-NL coronary arteries, but was a prominent component of both N-HL and DM-HL intima (Figure 2, center panels).
Effect of Diabetes in Hyperlipidemic Groups. However, when expressed as a percent lesion area, the median (IQR) biglycan contents of non-diabetic and diabetic hyperlipidemic lesions were similar [N-HL: 26.1% (12.9–45.2%) vs DM-HL: 24.1% (10.3–47.9%), p=0.92] (Figure 3C)]. In addition, there was a strong correlation between intimal area and biglycan content for both the non-diabetic and diabetic hyperlipidemic lesions (R 2 = 0.72, p<0.01) (Figure 3D), suggesting that similar to hyaluronan, the increase in lesion size seen with diabetes was accompanied by a proportionate increase in biglycan.
Relationship Between Intimal Area and LDL Deposition
Effect of Diabetes in Normolipidemic Groups. Immunohistochemistry was performed to identify apoB, the primary apolipoprotein molecule on LDL particles. ApoB also is present on VLDL particles and thus should be an excellent surrogate for “atherogenic” lipoprotein deposition. No apoB was detected in the intima of either normolipidemic group (N-NL or DM-NL).
Effect of Diabetes in Hyperlipidemic Groups. However, apoB was a prominent component of the intima of both hyperlipidemic groups (N-HL and DM-HL). When expressed as a median (IQR) percent of total lesion area, the apoB contents of non-diabetic and diabetic hyperlipidemic lesions were similar [N-HL: 14.5% (4.9–17.4%) vs DM-HL: 16.0% (6.2–50.5%), p=0.40] (Figure 3E). Similar to the findings for hyaluronan and biglycan, there was a relationship between apoB content and intimal area for the non-diabetic or diabetic groups, although the correlation was not as strong (R 2 = 0.23, p=0.0069) (Figure 3F).
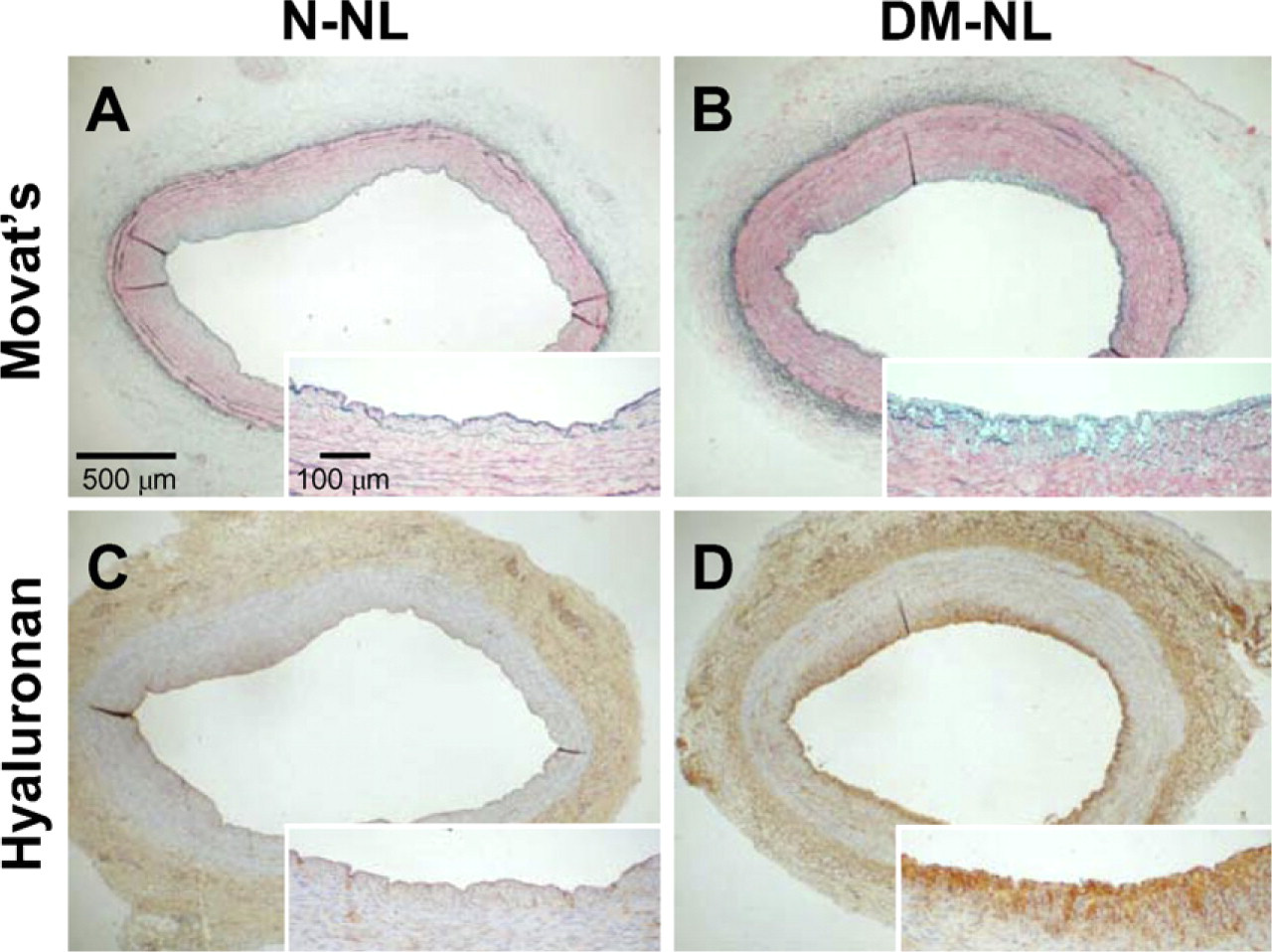
Effect of diabetes on hyaluronan accumulation in normolipidemic animals. Shown are coronary artery segments from a non-diabetic normolipidemic animal (N-NL) (
Lesion Cellularity
Effect of Diabetes in Normolipidemic Groups. Total intimal cell contents were counted by hand to determine whether diabetes was associated with a change in lesion cellularity. No macrophages were detected in the intima of either normolipidemic group (N-NL or DM-NL).
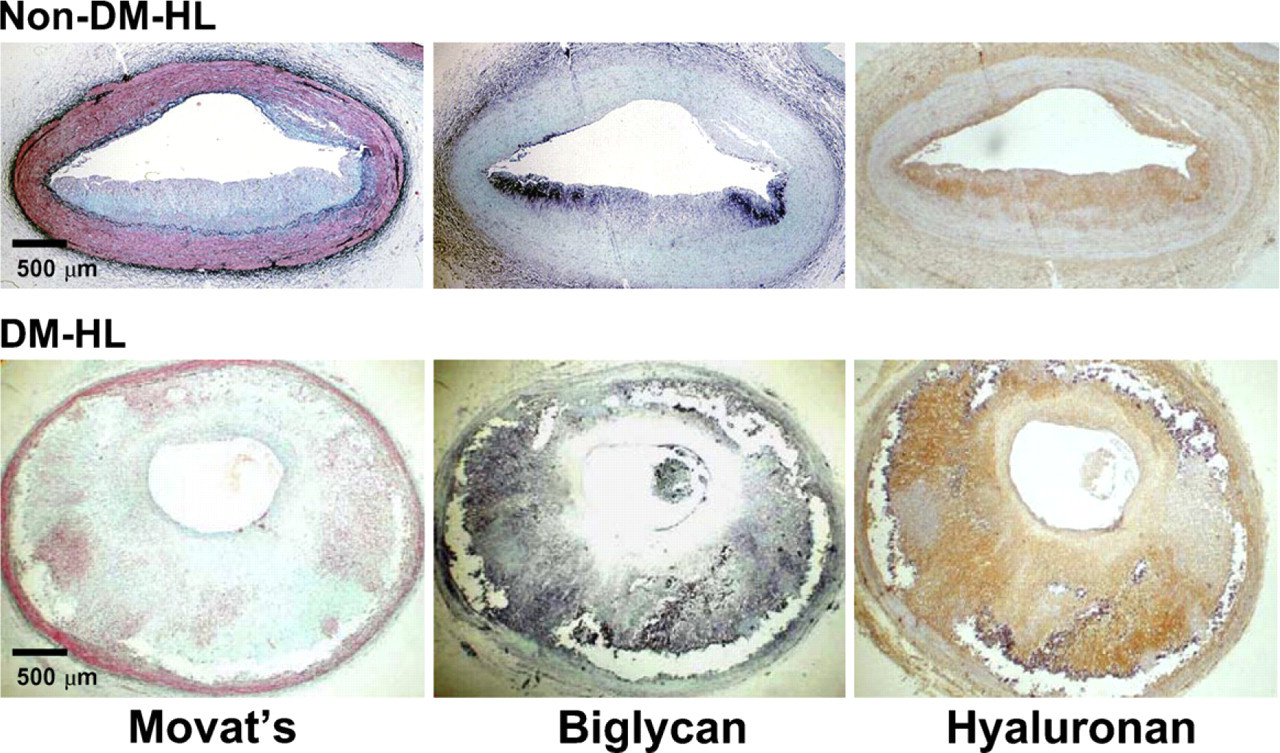
Increased biglycan and hyaluronan accumulation in hyperlipidemic animals. Shown are photomicrographs of adjacent sections stained with Movat's stain (left panels), immunostained for biglycan (black reaction product, center panels), or stained with hyaluronan-binding protein to identify hyaluronan (brown reaction product, right panels) from coronary artery segments from a non-diabetic hyperlipidemic animal (Non-DM-HL, upper panels) and from a diabetic hyperlipidemic animal (DM-HL, lower panels). Both biglycan and hyaluronan are detected in atherosclerotic intima of both hyperlipidemic groups. Counterstains: methyl green (center panels) or hematoxylin (right panels).
Effect of Diabetes in Hyperlipidemic Groups. For the N-HL and DM-HL groups, there was no difference in total intimal cellularity, expressed as cells/intimal area in μm2. For the N-HL group, median (IQR) cellularity was 0.020 (0.014–0.029) cells/μm2, whereas for the DM-HL group, median cellularity was 0.023 (0.018–0.028) cells/μm2 (p=0.57). Total macrophage area was determined for the subset of coronary segments that had been fixed in methanol Carnoy's solution, rather than formalin (N-HL = 8, DM-HL = 7). Median (IQR) intimal macrophage immunostained area actually appeared slightly lower for diabetic coronary segments [N-HL = 0.0060 (0.0047–0.0075) % vs DM-HL = 0.0023 (0.0015–0.0068) %], although this difference was not statistically significant (p=0.12).
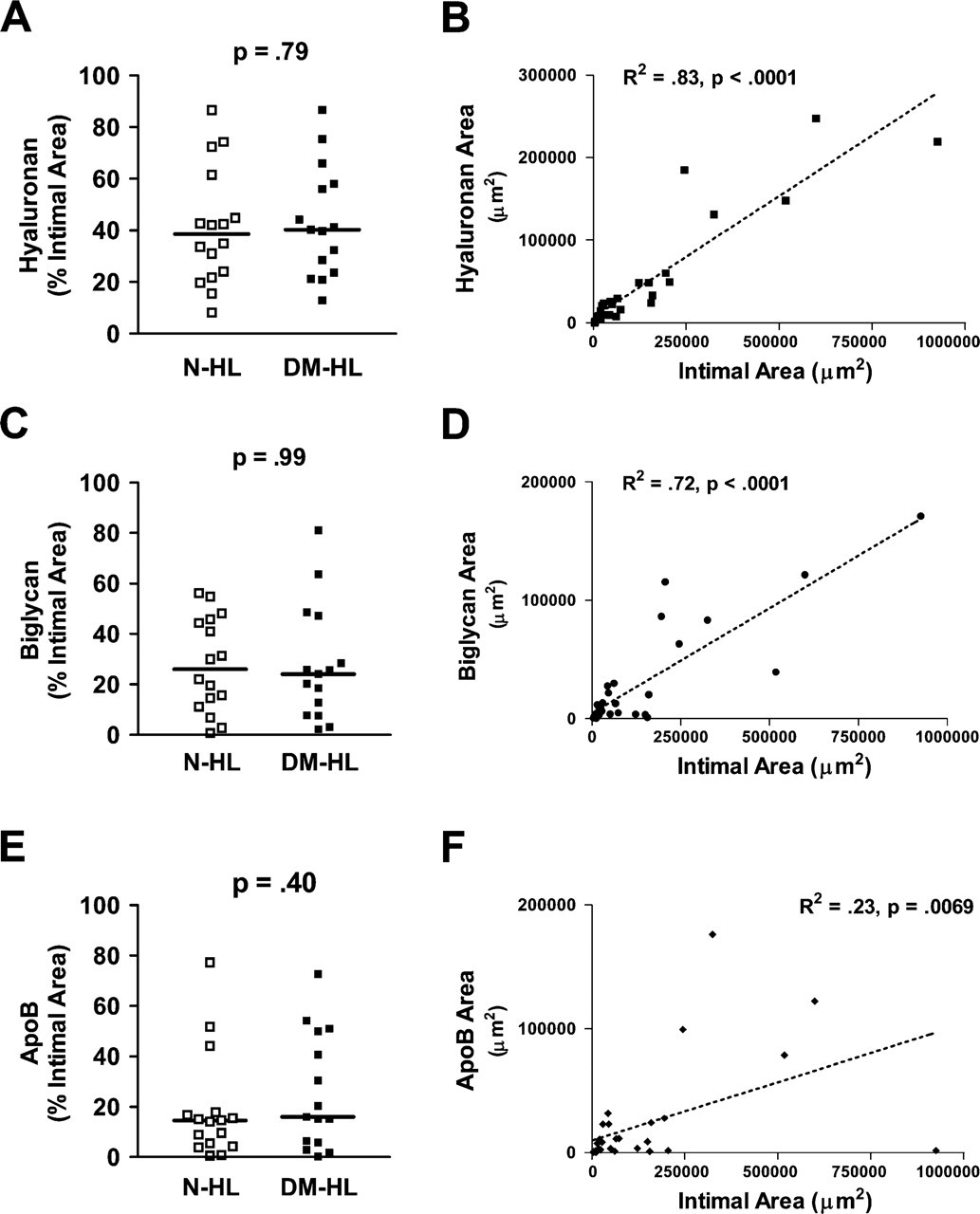
Comparison of hyaluronan, biglycan, and apoB areas in non-diabetic and diabetic porcine coronary artery intima. Shown are comparisons, for non-diabetic hyperlipidemic animals (N-HL) and diabetic hyperlipidemic animals (DM-HL), of percent (mean ± SEM) intimal areas with positive immunostaining for hyaluronan (
Elastin Content
Effect of Diabetes in Normolipidemic Groups. Movat's stain, with which elastin is stained black, was used to determine the elastin content of hyperlipidemic non-diabetic and diabetic lesions. There was no difference in intimal elastin content in normolipidemic groups (N-NL and DM-NL).
In contrast to the findings for hyaluronan, biglycan, and apoB in hyperlipidemic animals, diabetes was associated with a decrease in the intimal elastin content of lesions (Figure 4), expressed as percent of total intimal area. Specifically, the median (IQR) intimal elastin was higher in non-diabetic intima than in diabetic intima [N-HL: 5.2% (2.4–8.2%) vs DM-HL: 1.5% (0.5–4.2%), p=0.01] (Figure 5A). However, in the media of these coronary segments, median (IQR) elastin contents were similar [N-HL: 8.8% (7.3–12.7%) vs DM-HL: 7.5% (6.6–14.3%), p = 0.46] (Figure 5B). Thus, the relative loss of elastin appeared to be confined to the intima, but not to the media. However, qualitatively, there appeared to be increased elastin fragmentation, defined as obvious discontinuity in elastin fibers on Movat's stain, in both the intima and media of DM-HL as compared with N-HL lesions (comparison of Figures 5A and 5B).
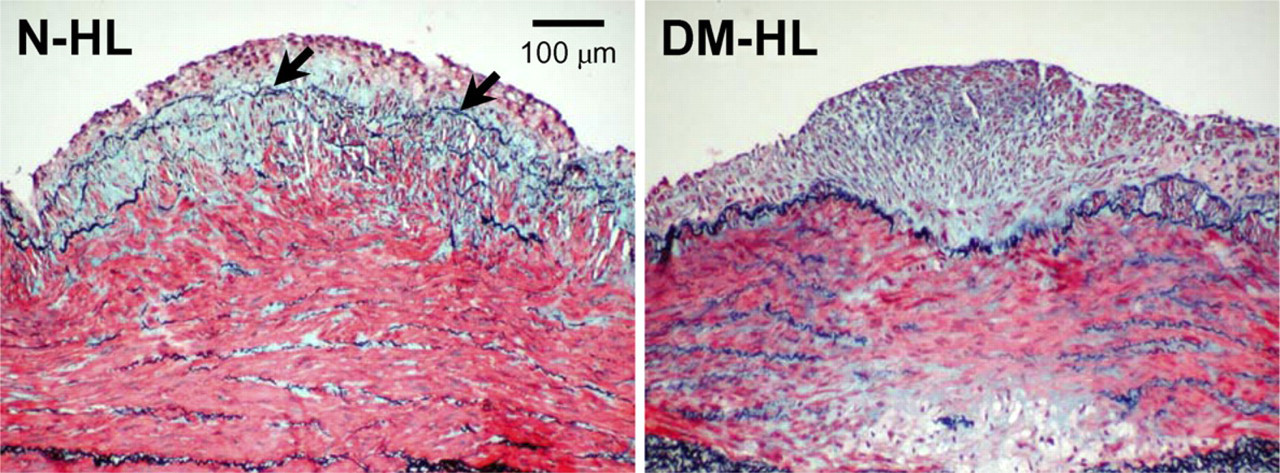
Decrease in intimal elastin staining. Shown are photomicrographs of Movat's staining of similar-sized lesions from a non-diabetic hyperlipidemic animal (N-HL, left panel) and a diabetic hyperlipidemic animal (DM-HL, right panel). Black elastin staining is readily apparent in the intima of the non-diabetic animal (black arrows, left panel), but is much less prominent in the intima of the diabetic animal (right panel).
Importantly, for the two hyperlipidemic groups combined (N-HL and DM-HL), there was a strong negative correlation between total intimal areas and intimal elastin areas (Spearman r = −0.62, p=0.001), but no correlation between total medial areas and medial elastin areas (Spearman r = −0.24, p=0.18), again suggesting that loss of elastin with lesion progression is confined to the intima.
Versican
Intimal versican areas were compared only for the two hyperlipidemic groups. Among the two hyperlipidemic groups, there was a trend toward increased versican content with diabetes [N-HL: 3.7% (0.1–17.3%) vs DM-HL: 0% (0–0.9%)], but the difference between groups did not reach statistical significance (p=0.08).
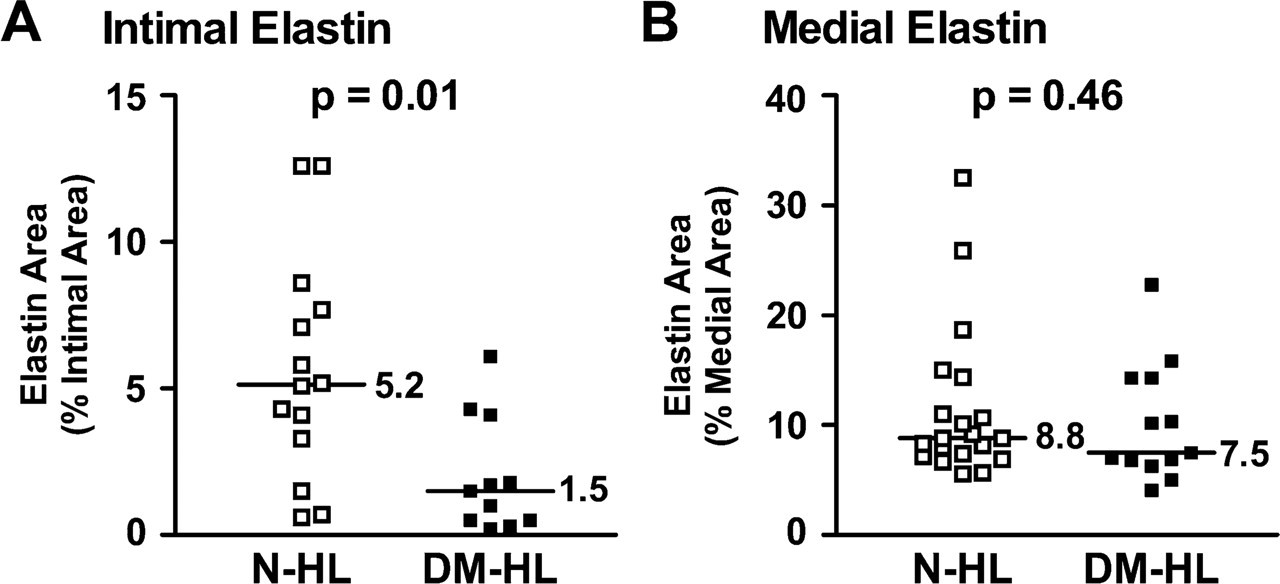
Decrease in relative intimal elastin content with diabetes in hyperlipidemic animals. Shown are comparisons, for non-diabetic hyperlipidemic animals (N-HL) and diabetic hyperlipidemic animals (DM-HL) of percent (mean ± SEM) areas with positive histochemical staining for elastin in the intima (
Discussion
This study confirms that diabetes dramatically increases lesion size in hyperlipidemic animals, but also demonstrates that diabetes has differential effects on arterial extracellular matrix components. Specifically, diabetes is associated with proportionate increases in hyaluronan and biglycan, but also in a substantial decrease in elastin content. Although the relative amounts of biglycan and hyaluronan are similar in non-diabetic and diabetic hyperlipidemic lesions, the fact that the absolute amounts of biglycan and hyaluronan are 4-fold greater in diabetic hyperlipidemic lesions indicates that specific factors associated with the diabetic state dramatically increase lesion biglycan and hyaluronan accumulation. The observation that relative elastin content is decreased in intima of animals with diabetes may be consistent with accelerated intimal elastin degradation, rather than decreased elastin production, because diabetes was not associated with a relative change in medial elastin content. In addition, there was qualitatively more elastin fragmentation in lesions from animals with diabetes as compared with those from animals without diabetes.
Hyaluronan Accumulation With Diabetes
Increased hyaluronan has been observed in the vasculature of human subjects with diabetes (Heickendorff et al. 1994), and there is a significant correlation between hyaluronan aortic content and diabetes but not between hyaluronan aortic content and age. Interestingly, hyaluronan synthesis by cultured human aortic smooth muscle cells is stimulated when these cells are exposed to sera from subjects with type 1 (Jarvelainen et al. 1987) and type 2 (Jarvelainen et al. 1986) diabetes. However, the specific serum factors responsible for this increase are not known. Factors associated with diabetes, such as high plasma glucose, lead to increased hyaluronan production in rat glomeruli, and stimulate mesangial cell hyaluronan production (Mahadevan et al. 1995). Hyaluronan fragments have been shown to bind to macrophages and stimulate chemokine expression (McKee et al. 1996). Diabetes induced by streptozotocin in the rat model is accompanied by elevated levels of glomerular hyaluronan and monocyte adhesion (Wang and Hascall 2004). Increased arterial lesion hyaluronan accumulation has been reported in humans, both in atherosclerosis (Levesque et al. 1994) and following mechanical arterial injury (Riessen et al. 1996). The functions of hyaluronan in arterial tissue are not known, but hyaluronan content is increased at sites of plaque erosion (Kolodgie et al. 2002). In addition, hyaluronan fragments have been shown to bind and activate macrophages through binding to the CD44 receptor (de La Motte et al. 1999; Pure and Cuff 2001). Thus, hyaluronan could mediate increased plaque inflammation. Consistent with this hypothesis, lack of CD44 has been associated with decreased atherosclerosis in the apoE-deficient mouse (Cuff et al. 2001). In addition, Baynes and colleagues demonstrated that reactive carbonyls, which are a feature of the diabetic state (Baynes and Thorpe 2000), can fragment hyaluronan (Jahn et al. 1999). However, the specific mechanisms by which diabetes stimulates hyaluronan accumulation are not known.
Biglycan and Versican Accumulation in Diabetes
As was seen with hyaluronan, the relative content of biglycan did not differ between lesions of animals with or without diabetes. However, on an absolute basis, super imposition of diabetes on hyperlipidemia was associated with a 4-fold increase in lesion biglycan content. This also suggests that factors associated with the diabetic state may stimulate increased biglycan accumulation in lesions. One possibility is oxidative stress (Baynes and Thorpe 2000; Pennathur et al. 2001), which has been shown to selectively increase biglycan expression by smooth muscle cells in vitro (Chang et al. 2000). The biglycan promoter contains binding sites for several oxidant-sensitive nuclear transcription factors, including AP-1, NF-κB, and AP-2 (Wegrowski et al. 1995; Ungefroren and Krull 1996). However, the exact mechanism responsible for the increased biglycan accumulation in diabetes also is not known.
In contrast, the suggestion that the chondroitin sulfate proteoglycan (CSPG), versican, may be increased in diabetic hyperlipidemic lesions is consistent with previous studies in rat tissues reporting increased CSPG content in glomerular basement membranes with diabetes (Karasawa et al. 1997) and in livers with insulin resistance (Olsson et al. 2001), as well as increased CSPG content in aortas of dogs with alloxan-induced diabetes (Sirek et al. 1980). However, the mechanism by which diabetes might lead to increased CSPG accumulation in these tissues is not known.
“Atherogenic” Lipoprotein Deposition and Intimal Area
For all hyperlipidemic lesions, there was a statistically significant correlation between intimal area and the amount of “atherogenic” lipoproteins (as assessed by apoB immunostaining), although the correlation between intimal area and apoB content was not as strong as that for hyaluronan and biglycan. However, the relative amounts of intimal apoB in lesions from hyperlipidemic animals with or without diabetes were not significantly different. This finding is in contrast to those of one study performed on human atherectomy specimens, which reported that lipid areas comprised 2% vs 7% of plaque area in lesions of subjects without or with diabetes, respectively (Moreno et al. 1999). In that study, “lipid” areas were determined from sections stained with the Masson's trichrome histochemical stain (Moreno et al. 2000), which does not specifically stain lipid, and therefore were probably necrotic cores, rather than lipid per se. In the current study, immunostaining for apoB was used to specifically identify retained atherogenic lipoproteins. Thus, the differences in conclusions from the present study and the previous study (Moreno et al. 2000) may be due to differences in the methods used to identify plaque “lipids.”
Diabetes and Decreased Intimal Elastin
Strikingly, diabetes was associated with a disproportionately lower relative content of elastin in the intima. Whether the relatively lower intimal elastin content seen with diabetes is the result of reduced elastin synthesis or accelerated elastin degradation is not known. Interestingly, elastin-deficient mice die of obstructive arterial disease (Li et al. 1998a), and arterial thickness is increased in both mice and humans hemizygous for elastin deficiency (Li et al. 1998b), possibly as a consequence of either increased wall stress or loss of an inhibitory effect of elastin on intimal growth (Li et al. 1998a, b). It is of interest that hyaluronan fragments induce metalloelastase expression by macrophages (Horton et al. 1999), indicating a possible relationship between increased hyaluronan and decreased elastin. Similarly, a number of elastin-degrading enzymes are increased in atherosclerosis (Liu et al. 2004), whereas in contrast, a deficiency in matrix metalloproteinase-9 is associated with protection against atherosclerosis in apoE-deficient mice (Luttun et al. 2004). Thus, lower elastin content, whether through decreased elastin production or increased elastin breakdown, appears to promote atherosclerosis. In addition, there is precedent for a diabetes effect on arterial elastin content, inasmuch as both decreased aortic elastin content and increased aortic elastin degradation have been reported in a rat model of alloxan-induced diabetes (Kwan et al. 1988).
To summarize, diabetes dramatically accelerates coronary atherosclerosis in a hyperlipidemic porcine model of atherosclerosis. This increase in lesion size is due, in part, to proportionate increases in intimal content of the extracellular matrix molecules hyaluronan and biglycan. In contrast, intimal elastin content is decreased with diabetes. The net effect of increasing the amounts of “non-structural” matrix components, such as hyaluronan and biglycan, while “structural” proteins, such as elastin, are decreased, may be increased wall stress and further acceleration of atherogenesis. Moreover, increased hyaluronan content has been associated with plaque erosion in humans (Kolodgie et al. 2002). Thus, the extracellular matrix composition changes seen in porcine diabetic lesions would be consistent with the recognition that humans with diabetes are more likely to suffer acute coronary syndromes, i.e., myocardial infarction or unstable angina, that result from rupture or ulceration of “unstable” atherosclerotic plaques. However, it is likely that additional factors, such as increased calcification, might play important roles in mediating the increased propensity to ulceration or rupture of lesions in subjects with diabetes.
Footnotes
Acknowledgements
This work was supported in part by Grants DK-02456 (to TNW, AC, and KDO) and HL-55798 (to RGG) from the National Institutes of Health, Bethesda, MD, and Mary Gates Undergraduate Research Awards (to CJ and H-JC) from the University of Washington.
