Abstract
The extracellular matrix (ECM) is an important contributor to the asthmatic phenotype. Recent studies investigating airway inflammation have demonstrated an association between hyaluronan (HA) accumulation and inflammatory cell infiltration of the airways. The ECM proteoglycan versican interacts with HA and is important in the recruitment and activation of leukocytes during inflammation. We investigated the role of versican in the pathogenesis of asthmatic airway inflammation. Using cockroach antigen (CRA)–sensitized murine models of allergic asthma, we demonstrate increased subepithelial versican in the airways of CRA-treated mice that parallels subepithelial increases in HA and leukocyte infiltration. During the acute phase, CRA-treated mice displayed increased gene expression of the four major versican isoforms, as well as increased expression of HA synthases. Furthermore, in a murine model that examines both acute and chronic CRA exposure, versican staining peaked 8 days following CRA challenge and preceded subepithelial leukocyte infiltration. We also assessed versican and HA expression in differentiated primary human airway epithelial cells from asthmatic and healthy children. Increases in the expression of versican isoforms and HA synthases in these epithelial cells were similar to those of the murine model. These data indicate an important role for versican in the establishment of airway inflammation in asthma.
Introduction
Asthma is a prevalent disease that affects approximately 10% of the population in the developed world and is associated with reversible airflow obstruction, sensitization to aeroallergens, eosinophilic airway inflammation, and acute respiratory exacerbations.1,2 The study of mechanisms underlying the pathogenesis of asthma is challenging as the asthma diagnosis encompasses multiple clinical phenotypes. This has additionally made the identification of reliable biomarkers and discrete susceptibility genes somewhat problematic. 3
In addition to maintaining tissue architecture, there is an emerging appreciation for the role of specific extracellular matrix (ECM) constituents in leukocyte trafficking and modulation of local airway inflammation.4–6 Evidence from both animal and human cell culture models has demonstrated that modulation of hyaluronan (HA), an important ECM component, occurs in the setting of infection and/or a viral mimetic.7,8 Independent studies have shown that the concentration of airway HA is higher in asthmatics and correlates with asthma severity.9–12 Recent studies have reported time-dependent changes in the accumulation of HA in both ovalbumin and cockroach antigen (CRA) sensitization murine models of asthma.13,14 In these two 8-week studies, increases in HA tissue staining correlated directly with the degree of inflammatory infiltrate and peaked at around 1 week following the antigen challenge. Interestingly, the inflammatory changes persisted despite subsequent decreases in the amount of HA in the tissues, suggesting that HA may play an important role in the initiation of the chronic inflammation seen in these models.
Versican is a large chondroitin sulfate proteoglycan that binds to HA, contributes to matrix stability, and modulates tissue inflammation by influencing leukocyte adhesion, migration, and inflammatory cytokine production (see review). 15 Human lung fibroblasts isolated from adults with mild untreated asthma demonstrate greater expression of versican. 16 Furthermore, versican expression in airway biopsy tissues correlates positively with airway hyperreactivity (AHR). Additional in vitro studies of human lung fibroblasts have demonstrated enhanced production of versican following treatment with the viral mimetic polyinosinic:polycytidylic acid (poly I:C) leading to increased adhesion of monocytes to the resultant ECM. 8 Separate studies have also demonstrated the retention of T-cells on a versican-enriched matrix further substantiating a role for versican in the modulation of leukocyte trafficking during the inflammatory process. 17 Taken together, these results suggest that versican may play an important role in the acute and chronic inflammatory changes seen in asthmatic airways; however, studies that link versican expression and inflammatory changes in vivo are currently lacking.
Direct characterization of ECM changes associated with asthma in humans is challenging given the need to study airway biopsy tissues, which are particularly difficult to obtain from children. Fortunately, animal models that recapitulate many of the features of human asthma are available to conduct mechanistic studies. For example, mice sensitized with either ovalbumin or CRA display many of the pathologic airway features of human asthma, including a Th2 cytokine profile, eosinophilic airway infiltration, and AHR.13,14
To investigate further the role of versican in asthmatic airway inflammation, changes in lung versican were assessed in murine models of both acute and chronic exposure to CRA following antigen sensitization. We hypothesized CRA-treated mice would display greater accumulation of versican in their airways, which would correlate with increased inflammatory cell infiltration. In addition, we have compared versican gene expression patterns of the lungs of CRA-treated mice with cultured human bronchial epithelial cells (BECs), differentiated at an air–liquid interface (ALI), obtained from carefully phenotyped children with and without allergic asthma to assess whether the BECs contribute to subepithelial deposition of versican in asthma.
Materials and Method
Acute Exposure CRA-Induced Asthma Model
C57BL/6 mice were purchased (Charles River Laboratories, Wilmington, MA). On days 0 to 2, anesthetized mice were sensitized intranasally with 40 µg of CRA (GREER, Lenoir, NC; B46) in a total volume of 20 µl phosphate-buffered saline (PBS). After 15 days, mice were challenged intranasally with 40 µg of CRA on 2 consecutive days and airway inflammation was assessed 72 hr later. Control mice were treated with PBS under the same protocol. Following sacrifice, lung sections were placed in 1 ml of RNALater and stored at −80C for analysis, and remaining lung tissue was fixed in formalin. All mice were maintained at the Benaroya Research Institute animal care facility under pathogen-free conditions and were used in accordance with applicable regulations with institutional Animal Care and Use Committee approval.
To determine differential cell counts in the lung, single cell suspensions were prepared as follows. Lung was cut into fine pieces using scissors and the tissue fragments digested in 50 µg/ml of Liberase (Roche, Basel, Switzerland) and 10 U/ml of DNase I at 37C for 30 min. Digested samples were filtered through a 100-µm cell strainer and washed with equal volumes of RPMI plus 20% fetal bovine serum (FBS). Red blood cells were lysed with ammonium-chloride-potassium lysing buffer and lung cells centrifuged at 250 × g for 5 min. Cells were resuspended in fluorescence-activated cell sorting (FACS) buffer and counted using a hemocytometer. Differential cell counts were performed by flow cytometry.
Flow Cytometry of Bronchoalveolar Lavage Fluid (BALF) Cells and Tissue Homogenates
Single cell suspensions for determination of differential cell counts in the lung and BALF were prepared as follows. To prevent nonspecific binding of antibodies, cells were incubated with anti-CD16/CD32 (Clone: 2.4G2) in FACS buffer for 15 min at room temperature. The following antibodies were used: anti-Ly6G FITC (1A8), anti-Ly6C PerCpCy5.5 (HK1.4), anti-CD4 BV605 (RM4-5), anti-CD45R BV650 (RA3-6B2), anti-MHCII A700 (M5/114.15.2), anti-CD11b APC-e780 (M1/70), anti-Siglec F PE (E50-2440), anti-CD3 PECy5 (145-2C11), and anti-CD11c PE-Cy7 (N418). Cells were stained with surface antibodies for 12 min at room temperature. Samples were analyzed with BD LSR II flow cytometer (BD Biosciences, San Jose, CA).
Chronic Exposure CRA-Induced Asthma Model
Immunocompetent, nonirradiated, 6- to 8-week-old female BALB/c mice were obtained from the Jackson Laboratory (Bar Harbor, ME). All mice were maintained at the Cleveland Clinic animal care facility under specific pathogen-free conditions using microisolator cages and were used in accordance with applicable regulations after institutional approval. Food and water were available ad libitum. Eight-week-old mice were sensitized by intraperitoneal injection (IP) injection of 5 µg of CRA adsorbed to 20 mg of aluminum hydroxide (Al(OH)3). Two weeks later, mice were challenged with 5 µg of CRA in 50 µl of saline by intranasal delivery once a day for 8 days starting on day 0 and then twice a week until 8 weeks. Mice were sacrificed during the course of antigen exposure at 6 and 12 hr; 1, 2, 4, 6, and 8 days; and 2, 4, 6, and 8 weeks, and lung sections were obtained. Mice without antigen sensitization and challenge were used as controls, and four mice were used for each time point of the study.
Immunohistochemistry
All stainings were performed on an automated immunohistochemistry stainer (Leica Biosystems, Inc., Buffalo Grove, IL). For HA affinity histochemistry, the Bond Intense R Detection kit (Leica Biosystems), a streptavidin–horseradish peroxidase (HRP) system, was used with 4 µg/ml biotinylated HA binding protein in 0.1% bovine serum albumin (BSA)–PBS as the primary.
Versican immunohistochemistry required pretreatment using heat-mediated antigen retrieval with low pH for 10 min and digestion with 0.2 U/ml chondroitinase ABC (Sigma, St. Louis, MO; #C3667) in a buffer containing 18 mM Tris, 1 mM sodium acetate, and 1 mg/ml BSA pH 8.0 for 1 hr at 37C. After digestion, lung sections were incubated for 1 hr with 2 µg/ml rabbit antimouse versican-glycosaminoglycan (GAG) β domain (EMD Millipore, Billerica, MA; #AB1033) in bond antibody diluent, and detection was performed using the Bond Polymer Refine Detection kit (Leica Biosystems) This kit contains a goat antirabbit conjugated to polymeric HRP secondary antibody, 3,3’-diaminobenzidine (DAB) chromogen, and a hematoxylin counterstain. Negative control slides were treated with 2.0 µg/ml ChromPure rabbit IgG (Jackson Immuno Research Labs, West Grove, PA; #011-000-003) in Bond antibody diluent (Leica Biosystems). For F4/80 staining, 1 mg/ml of F4/80 clone A3-1 (Abcam, Cambridge, MA) was used as the primary antibody, and secondary staining was performed as described for versican above.
Quantitative Real-Time Polymerase Chain Reaction (qRT-PCR) for Murine Tissues
All reagents were supplied by Life Technologies (Grand Island, NY) unless otherwise noted. Relative quantitation of V1, V3, Has1, Has2, Has3, Hyal1, and Hyal2 gene expression was performed using Taqman Gene Expression Assays Mm00490173_m1, Rn01493763_m1, Mm00468496_m1, Mm00515089_m1, Mm00515092_m1, Mm00476206_m1, and Mm01230689_g1, respectively. Relative quantitation of V0 and V2 was performed with Taqman custom-designed probes as follows: V0, Forward 5′-CCAAGTTCCACCCTGACATAAATGT-3′, Reverse 5′-GGATGACCACTTACAATCATATCACTCA-3′, and Reporter 5′-TCGACCTGTCTTGTTTTC-3′. V2, Forward 5′-CCAAGTTCCACCCTGACATAAATGT-3′, Reverse 5′-GCATGGGTTTGTTTTGCAGAGATC-3′, and Reporter 5′-CAGAGAAAACAAGACAGGACCT-3′. Briefly, 50 ng cDNA was amplified in 1× Taqman Gene Expression Master Mix with 250 nM Taqman probe in a 20-µl reaction. CD44 amplification was performed using 50 ng cDNA in 1× SYBR Select Master Mix and 1 µM primers. Expression was normalized to eukaryotic 18S rRNA Endogenous Control Part No. 4333760. All reactions were run using the Standard program for 40 cycles on an ABI 7500 Fast Real-Time Thermocycler. All samples were in duplicate. Copy number estimates were generated from a standard curve created by using a selected reference cDNA template and Taqman probe. 18 Primers for CD44, designed with NCBI Primer-BLAST and synthesized by Sigma, are as follows: Forward 5′-GCATCCAACACCTCCCACTA-3′, Reverse 5′-TCCATCGAAGGAATTGGGTA-3′. The assay as described above was repeated with separately prepared samples.
Human Subjects
Atopic asthmatic (n = 25) and healthy nonatopic nonasthmatic (n = 15) children ages 6 to 18 years who were undergoing an elective surgical procedure requiring endotracheal intubation and general anesthesia were recruited. A detailed medical history was obtained at enrollment to ensure that participants met the following inclusion and exclusion criteria. Children with asthma had at least a 1-year history of physician-diagnosed asthma, physician documented wheezing in the 12 months prior to study enrollment, used a short-acting beta-agonist (SABA, that is, albuterol) ≥twice a month or were taking a daily inhaled corticosteroid or leukotriene receptor antagonist, and were born at ≥36 weeks gestation. Healthy subjects were born at ≥36 weeks gestation, had no history of asthma, reactive airway disease, chronic daily cough, or physician-diagnosed obstructive lung disease, and no history of prior treatment with a systemic or inhaled corticosteroid, SABA, or oxygen. Children with asthma had one or more of the following atopic features: history of positive skin prick test, positive radioallergosorbent testing (RAST) for a common aeroallergen (discussed below), elevated serum IgE (>100 IU/ml), history of physician-diagnosed allergic rhinitis, history of physician-diagnosed atopic dermatitis. Healthy subjects lacked a history of any of the above atopic features and were excluded if they had any other atopic comorbidity.
From each subject, a blood sample was drawn and used to measure total serum IgE and RAST allergen-specific IgE to dust mites (Dermatophagoides farinae and D. pteronyssinus), cat epithelium, dog epithelium, Alternaria tenuis, Aspergillus fumigatus, and timothy grass. Subjects had been fasting for at least 6 hr prior to anesthesia. The fraction of exhaled nitric oxide (FENO) was measured according to American Thoracic Society (ATS)/European Respiratory Society (ERS) guidelines 19 using a NIOX MINO nitric oxide analyzer (Aerocrine, Solna, Sweden). Forced vital capacity (FVC), forced expiratory volume in 1 sec (FEV1), and forced expiratory flow between 25% and 75% of FVC (FEF25–75) were measured according to ATS guidelines using a VMAX Series 2130 Spirometer (VIASYS Healthcare, Hong Kong). Spirometry was repeated 15 min following administration of two puffs of albuterol in children with asthma.
Written consent was obtained from parents of subjects, and assent was obtained for children aged ≥7 years. The Seattle Children’s Hospital Institutional Review Board approved this study.
Epithelial Cell Isolation
Immediately after the endotracheal tube was secured, three BEC samples were obtained from subjects while under general anesthesia using 4 mm Harrell unsheathed bronchoscope cytology brushes (CONMED Corporation, Utica, NY). As described by Lane et alf., 20 the unprotected brush was inserted through an endotracheal tube, advanced until resistance was felt, and rubbed against the airway surface for 2 sec. Cells were seeded onto T-25 cell culture flasks precoated with type I collagen. Cultures were maintained at 37C in an atmosphere of 5% CO2 in a humidified incubator. Cells were cultured in bronchial epithelial growth medium (Clonetics BEGM, Lonza, Basel, Switzerland) containing gentamicin and amphotericin B, and further supplemented with penicillin–streptomycin (100 µg/ml; Invitrogen, Carlsbad, CA). Fluconazole (25 µg/ml) was added to primary cultures for the first 96 hr, after which medium was aspirated and replaced with BEGM without fluconazole. BEGM was thereafter changed every 48 hr until the culture reached ~70% to 90% confluence. When P0 flasks became 70% to 90% confluent, cells were passaged into three new P1 T-25 flasks.
ALI Epithelial Cell Cultures
For ALI cultures, we employed the PneumaCult ALI media system (StemCell, Vancouver, British Columbia, Canada). 21 Prior to seeding, cells were incubated with PneumaCult ALI Induction Media for 24 hr in the T-25 flasks according to the manufacturer’s recommendations. Following induction, cells were trypsinized with 2 ml of 0.025% Trypsin–EDTA and then seeded onto Collagen I precoated Corning Costar 12 mm, 0.4 µm transwells (Corning Life Sciences, Tewksbury, MA) at a concentration of 100,000 cells per transwell. Cells were kept in submerged culture using PneumaCult ALI Expansion Media in both the apical and well chambers for 7 days or until confluent following the manufacturer’s guidelines. Once confluent, cells were changed to PneumaCult ALI Maintenance Media in the lower well chamber only, and the remaining apical media was aspirated. ALI Maintenance medium in the basolateral compartment was changed every other day, and cells were differentiated at an ALI for 21 days prior to initiation of cell culture experiments. Each experimental condition per cell line consisted of triplicate transwells.
Epithelial Cell RNA Extraction and qRT-PCR
Total RNA was isolated from airway epithelial cells grown at an ALI. Three transwells from each experimental condition were harvested and pooled to isolate RNA using the RNAqueous kit for total RNA purification from Ambion-Applied Biosystems (Austin, TX). RNA concentration was determined using Nanodrop 1000 (Thermo Scientific, Wilmington, DE). RNA samples (1 µg) were reverse transcribed with M-MLV reverse transcriptase with a combination of random hexamers and oligo-dTs using the SuperScript VILO cDNA Synthesis Kit (Life Technologies). Samples were diluted up to a final volume of 100 µl (10 ng/µl). qRT-PCR was performed using validated TaqMan probes (Life Technologies) for HAS1, HAS2, HAS3, CD44, VCAN, VCAN1, and VCAN3. Assays were performed using the TaqMan Fast Advanced Master Mix reagents and accompanying protocol and the Applied Biosystems StepOnePlus Real-Time PCR System with StepOne Software v2.2.2 (Life Technologies).
Statistical Analysis
The relative expression of hyaluronan synthases (HASs), hyaluronidases (Hyals), CD44, and VCAN isoforms was standardized using 18S rRNA (mouse) or glyceraldehyde 3-phosphate dehydrogenase (GAPDH, human) as a nonregulated reference genes. Analyses of qRT-PCR results were performed using GenEx version 5.0.1 (MultiD Analyses AB, Göteborg, Sweden) based on methods described by Pfaffl. 22 Statistical significance was determined using the Mann–Whitney U test as the data were not normally distributed. The level of significance was set at P < 0.05. Figures were prepared using Prism 6.0 software (GraphPad Software, Inc., San Diego, CA).
Results
Acute CRA Challenge Leads to a Robust Inflammatory Response in Lungs of Sensitized Mice
Flow cytometry analysis of BALF obtained from CRA-challenged mice 72 hr following CRA antigen exposure revealed significantly greater total cell counts compared with BALF obtained from PBS control mice (4.9 × 106 ± 1.6 × 106 vs. 1.2 × 105 ± 1.0 × 104, P < 0.0001; Fig. 1A). Differential analysis of the BALF demonstrated that in CRA mice, these cells were predominantly composed of eosinophils (3.5 × 106 ± 1.2 × 106; 72.7%); however, neutrophils (161,489 ± 58,779; 3.3%), dendritic cells (136,675 ± 49,450; 2.8%), and CD4 T-cells (330,559 ± 105,377; 6.8%) were also all elevated compared with BALF obtained from PBS control mice (P < 0.0001 for each comparison). Similar inflammatory changes occurred in whole lung tissue preparations, which were also quantified using flow cytometry (Fig. 1B). Total cell counts were elevated in lung samples obtained from CRA mice compared with PBS controls (1.4 × 107 ± 2.9 × 106 vs. 3.8 × 106 ± 4.0 × 104; P = 0.0003). Similar to the BALF findings, elevated numbers of eosinophils were also found in the whole lung tissue of CRA-exposed mice compared with PBS controls (3.3 × 106 ± 9.1 × 105 vs. 6.7 × 104 ± 2.0 × 104; P = 0.0007). In addition, multiple other leukocyte populations including neutrophils (616,250 ± 167,050 vs. 134,000 ± 15,620; P < 0.02), dendritic cells (793,500 ± 159,948 vs. 52,167 ± 2539; P < 0.0005), CD4 T-cells (1.2 × 106 ± 221,632 vs. 382,000 ± 28,729; P < 0.03), B cells (1.7 × 106 ± 376,208 vs. 578,667 ± 65,484; P < 0.05), and interstitial macrophages (469,500 ± 76,804 vs. 159,333 ± 11,851; P < 0.04) were found to be elevated in lung tissue obtained from CRA-exposed mice compared with PBS controls. The number of alveolar macrophages was decreased in CRA mice compared with PBS controls (89,875 ± 48,669 vs. 184,333 ± 4333; P < 0.02).
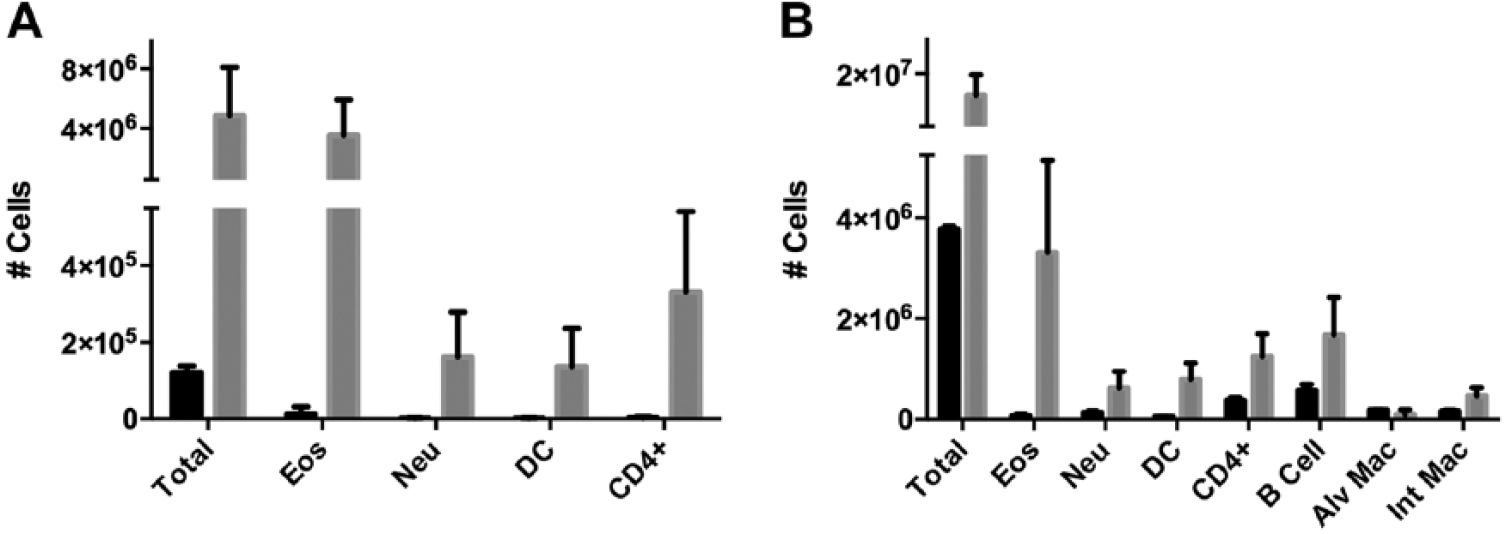
Flow cytometry for leukocytes in both bronchoalveolar lavage fluid (BALF) and lung tissue homogenates. Mice sensitized and challenged with cockroach antigen (CRA) displayed an increased number of all quantified leukocyte subgroups in BALF compared with control mice (P < 0.0001, Panel A). Similarly, leukocyte counts assessed by flow cytometry were elevated in lung tissue homogenates in CRA-treated mice compared with controls for all measured leukocyte classes except alveolar macrophages (P < 0.05, Panel B). Phosphate-buffered saline-treated mice are shown as black bars, CRA mice are shown as gray bars for both panels.
Increased Versican Accumulation in Lungs of Sensitized Mice Challenged Acutely With CRA
Immunohistochemistry
Paraffin-embedded sections from both CRA-treated and PBS control mice were stained for versican using immunohistochemistry. Images of representative sections from the acute CRA exposure model at day 4 are shown in Fig. 2 (n = 4 per group). Comparison of CRA-exposed mice to PBS controls revealed increased staining in the CRA mice (Fig. 2A, H). At higher magnification, intense staining was present in the subepithelial layer surrounding the bronchioles of the CRA-exposed mice, which was not observed in the sections obtained from PBS controls (Fig. 2B, E, I, L). The versican-rich areas around the bronchioles of the CRA mice were infiltrated with leukocytes. Many of these leukocytes in the subepithelial space stained positively for F4/80, a marker for macrophages (Fig. 3I, L), which was not seen in sections obtained from PBS mice. Examination of the perivascular space surrounding the pulmonary veins revealed that versican staining was present in both CRA- and PBS-treated mice; however, the staining was more intense in the CRA mice (Fig. 2C, F, J, M). In addition, CRA-treated mice revealed a substantial perivascular infiltration of leukocytes that was not observed in the PBS mice. In contrast to the subepithelial staining for F4/80 observed in the bronchioles, staining of the cells in the perivascular space did not demonstrate a consistent pattern of increased F4/80 staining, despite increased leukocyte infiltration (Fig. 3C, F, J, M). Finally, examination of the lung parenchyma demonstrated increased versican staining in CRA-treated mice compared with PBS mice (Fig. 2D, G, K, N). Parenchymal staining was most intense in the alveolar septa in the CRA-treated mice. A comparable amount of F4/80 staining was observed in the lung parenchyma of both the CRA- and PBS-treated mice (Fig. 3D, G, K, N).
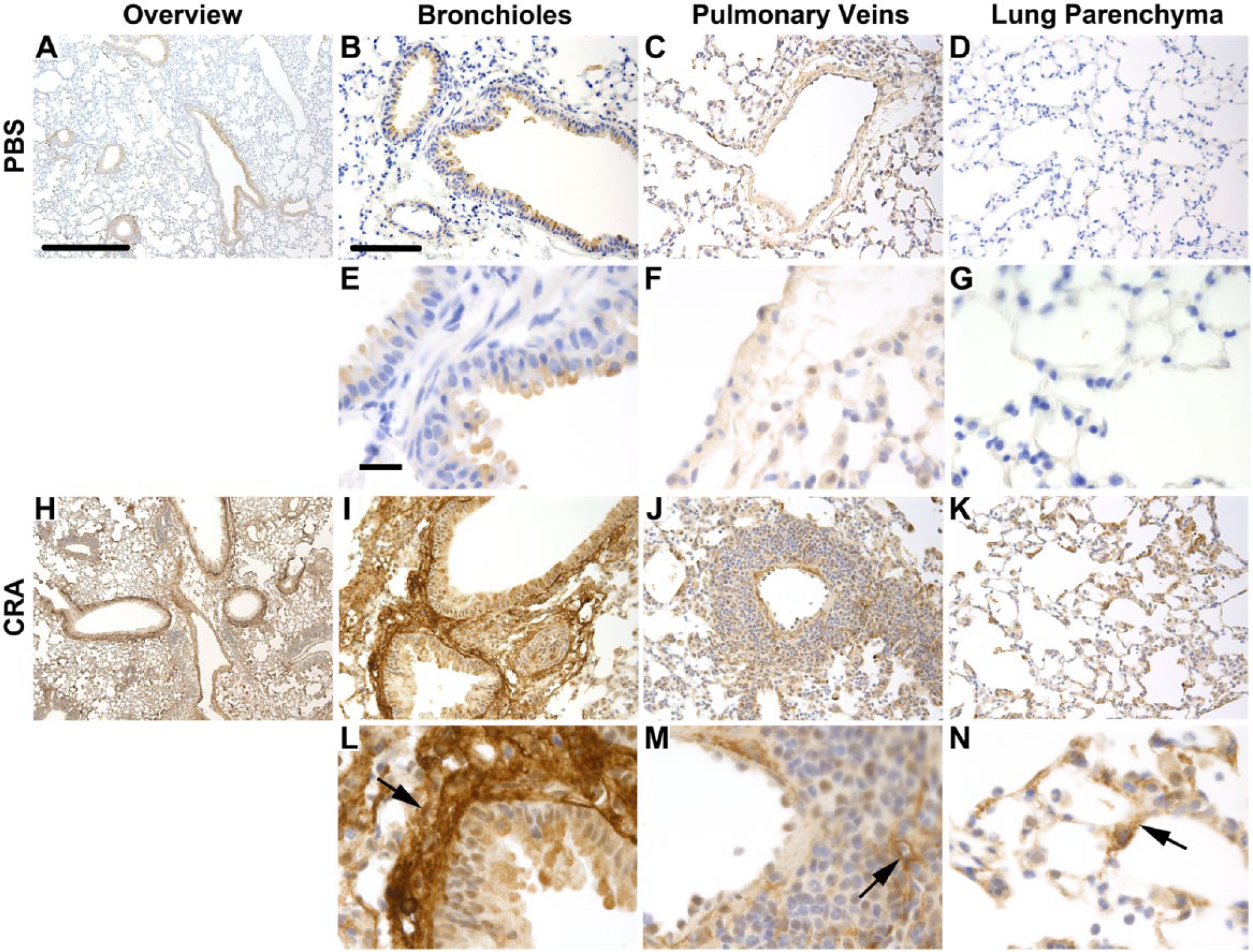
Versican staining in lung tissue sections following treatment with cockroach antigen (CRA). Depicted are representative images of control (Panels A–G) and CRA-treated mice (Panels H–N) stained for versican. Sections obtained from control and CRA-treated mice are shown at low magnification (Panels A and H, respectively; 50×, scale bar = 500 µm). Higher magnification images (200× B–D and I–K, scale bar = 100 µm; 630× E–F and L–N, scale bar = 20 µm) are shown for comparison, including; bronchioles and pulmonary arteries (Panels B, E, I, and L), pulmonary veins (Panels C, F, J, and M), and lung parenchyma (Panels D, G, K, and N). Black arrows denote increased versican staining in the subepithelial space, perivascular space, and parenchyma.
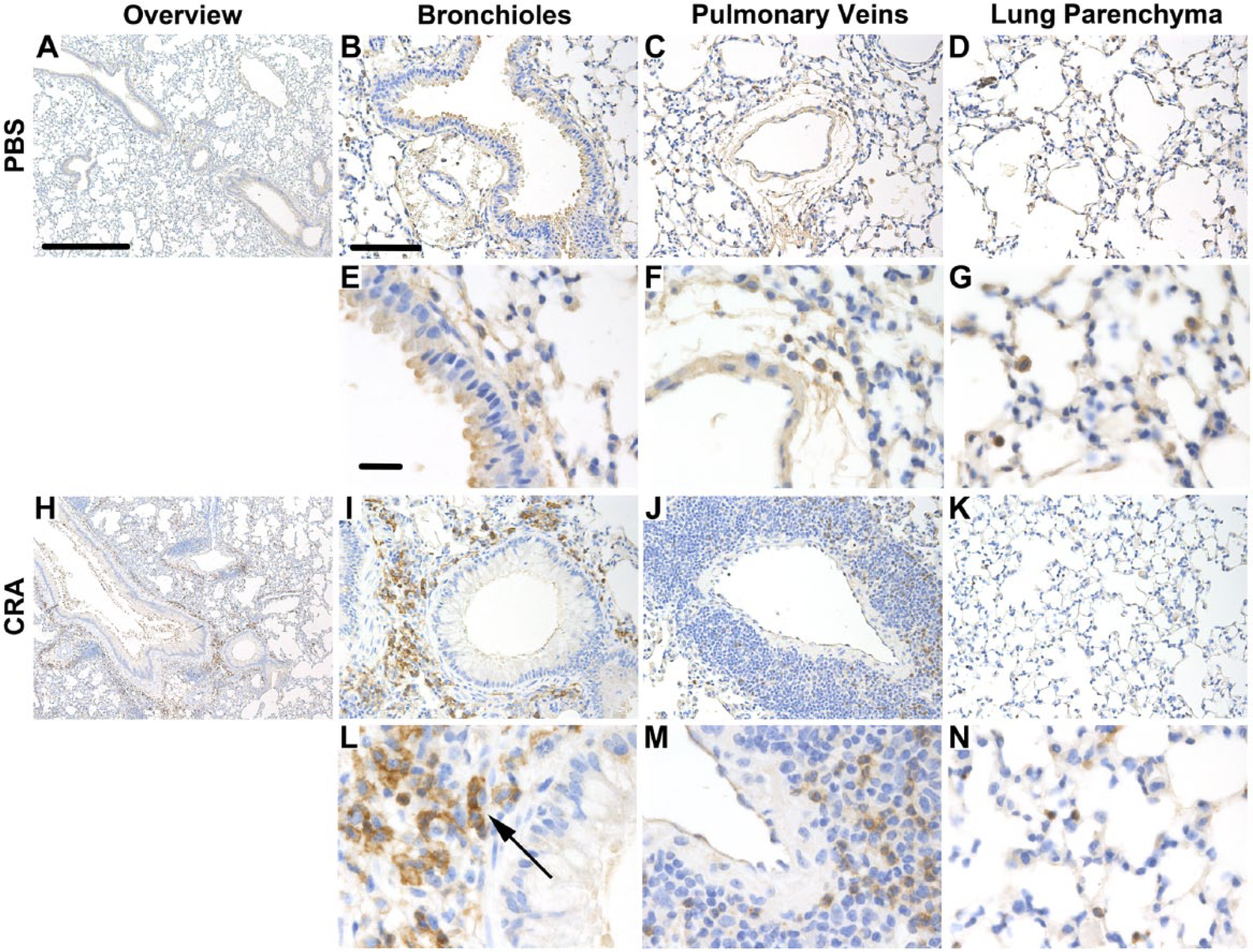
Staining for F4/80 in lung tissue sections following treatment with cockroach antigen (CRA). Shown are representative images of control (Panels A–G) and CRA-treated mice (Panels H–N) stained for F4/80. Sections obtained from control and CRA-treated mice are shown at low magnification (Panels A and H, respectively; 50×, scale bar = 500 µm). Higher magnification images (200× B–D and I–K, scale bar = 100 µm, 630× E–F and L–N, scale bar = 20 µm) are shown for comparison, including bronchioles (Panels B, E, I, and L), pulmonary veins (Panels C, F, J, and M), and lung parenchyma (Panels D, G, K, and N). The black arrow denotes increased F4/80 staining in the subepithelial space.
Gene Expression and Versican
qRT-PCR was performed to evaluate the expression of versican and the involvement of the four major versican isoforms in whole lung homogenates derived from both CRA and PBS mice in the acute 72-hr CRA exposure model. Expression of versican mRNA was elevated in mice exposed to CRA for the V0 (P < 0.003), V1 (P < 0.003), and V2 (P = 0.005) isoforms. A trend toward increased V3 isoform expression was noted in CRA-treated mice; however, this failed to meet statistical significance (P = ns; Fig. 4).
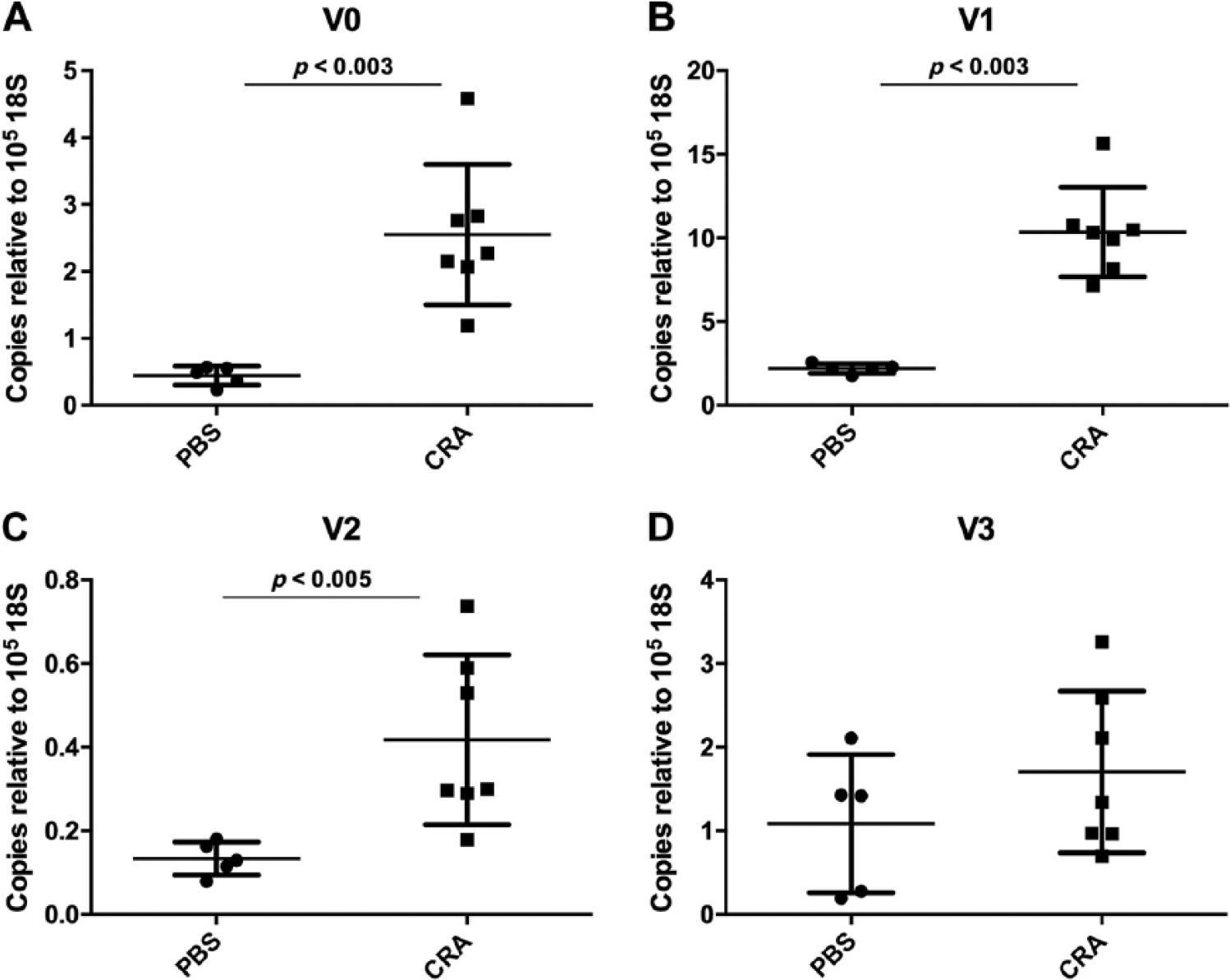
Gene expression of versican isoforms in lung tissue homogenates measured by quantitative real-time polymerase chain reaction (qRT-PCR). Expression of versican mRNA was elevated in mice exposed to cockroach antigen (CRA) for 72 hr compared with control mice for the V0 (Panel A, P < 0.003), V1 (Panel B, P < 0.003), V2 isoforms (Panel C, P < 0.005). Expression of V3 was not significantly different between the groups (Panel D). PBS, phosphate-buffered saline.
Parallel Accumulation of HA in Versican-Enriched Areas in the Lungs of Sensitized Mice Challenged Acutely With CRA
Histological evaluation of tissue sections obtained from both CRA-sensitized mice and PBS control mice 72 hr after CRA challenge was performed using affinity histochemistry for HA. Representative tissue sections are shown in Fig. 5 (n = 4 per group). Increased staining for HA was observed throughout the lung in CRA mice compared with PBS controls (Fig. 5A, H). Closer examination revealed HA staining associated with the bronchioles in both groups of mice. Similar to the pattern of versican staining discussed above, the HA-positive ECM was associated with a large inflammatory cell infiltrate in CRA-exposed mice as described above that was not observed in any of the sections obtained from PBS mice (Fig. 5B, E, I, L) and was greatest in airways closely associated with pulmonary arterial vessels. Pulmonary veins also stained positively for HA in sections from both groups of mice; however, unlike the versican staining, similar patterns of HA staining were noted in both the PBS- and CRA-treated mice (Fig. 5C, F, J, M). Finally, the parenchymal compartment of lung sections obtained from CRA-exposed mice stained intensely for HA compared with PBS mice (Fig. 5D, G, K, N) and was similar to the pattern of versican staining shown above.
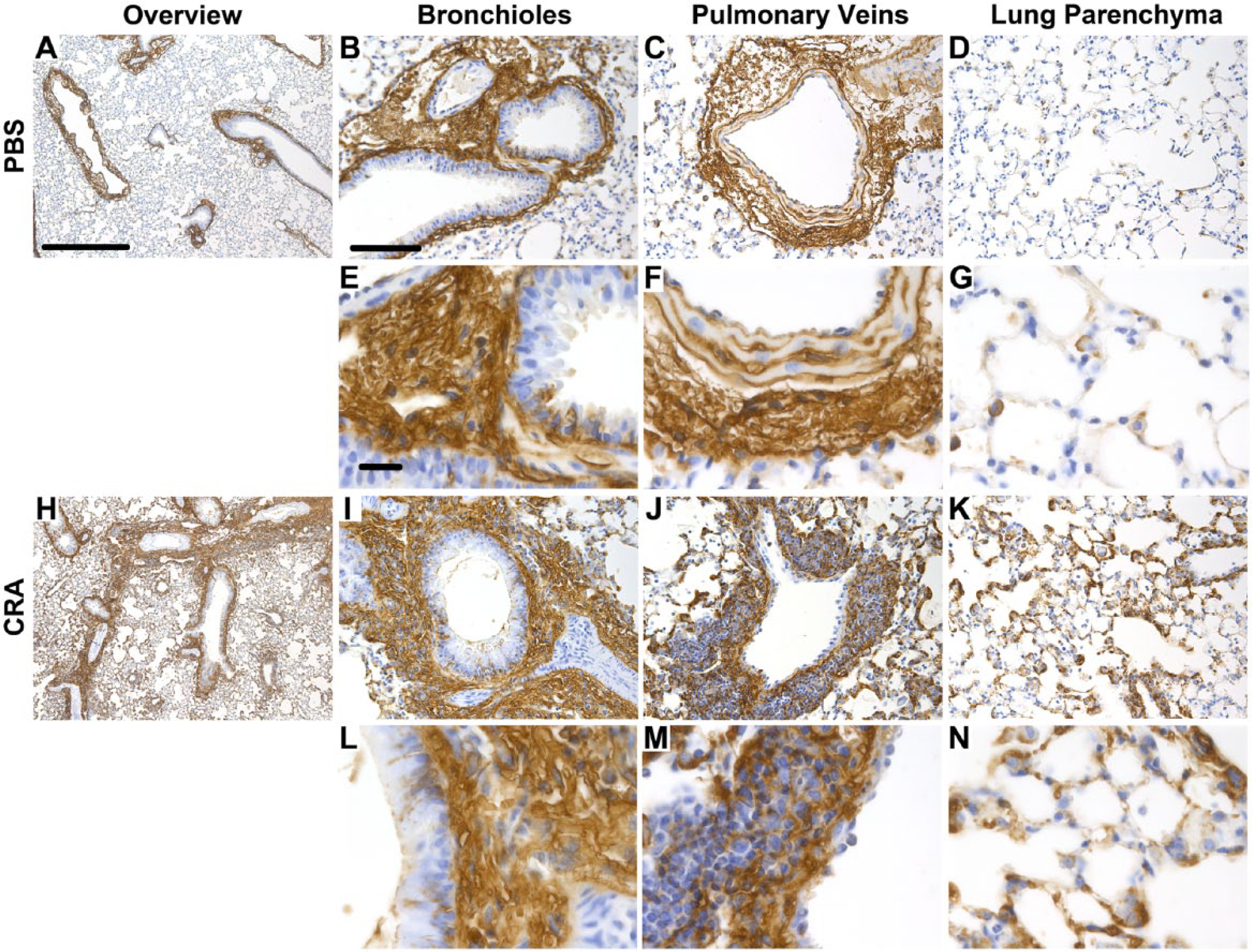
Hyaluronan (HA) staining in lung tissue sections following treatment with cockroach antigen (CRA). Depicted are representative images of control (Panels A–G) and CRA-treated mice (Panels H–N) stained for HA. Sections obtained from control and CRA-treated mice are shown at low magnification (Panels A and H, respectively; 50×, scale bar = 500 µm). Higher magnification images (200× B–D and I–K, scale bar = 100 µm, 630× E–F and L–N, scale bar = 20 µm) are shown for comparison, including bronchioles and pulmonary arteries (Panels B, E, I, and L), pulmonary veins (Panels C, F, J, and M), and lung parenchyma (Panels D, G, K, and N).
Gene Expression and HA
To quantify the expression of the three members of the HAS family (Has1–3), qRT-PCR analysis was performed on whole lung tissue samples obtained from both the CRA- and PBS-exposed mice. No difference in Has1 expression was observed between the CRA-exposed and PBS mice (P = ns; Fig. 6A). Expression of both Has2 and Has3 was significantly increased in CRA-exposed mice compared with PBS controls (P < 0.003 and P < 0.003, respectively; Fig. 6B, C). Given that accumulation of HA is also dependent on turnover by degradation enzymes, expression of the principal Hyals and CD44, a HA receptor involved in Hyal activity, was also examined. Expression of both Hyal1 and Hyal2 was decreased in CRA-treated mice compared with PBS controls (P < 0.02 and P < 0.003; Fig. 6D, E). Conversely, expression of Cd44 mRNA was increased in CRA-treated mice compared with PBS control mice (P = 0.01; Fig. 6F).
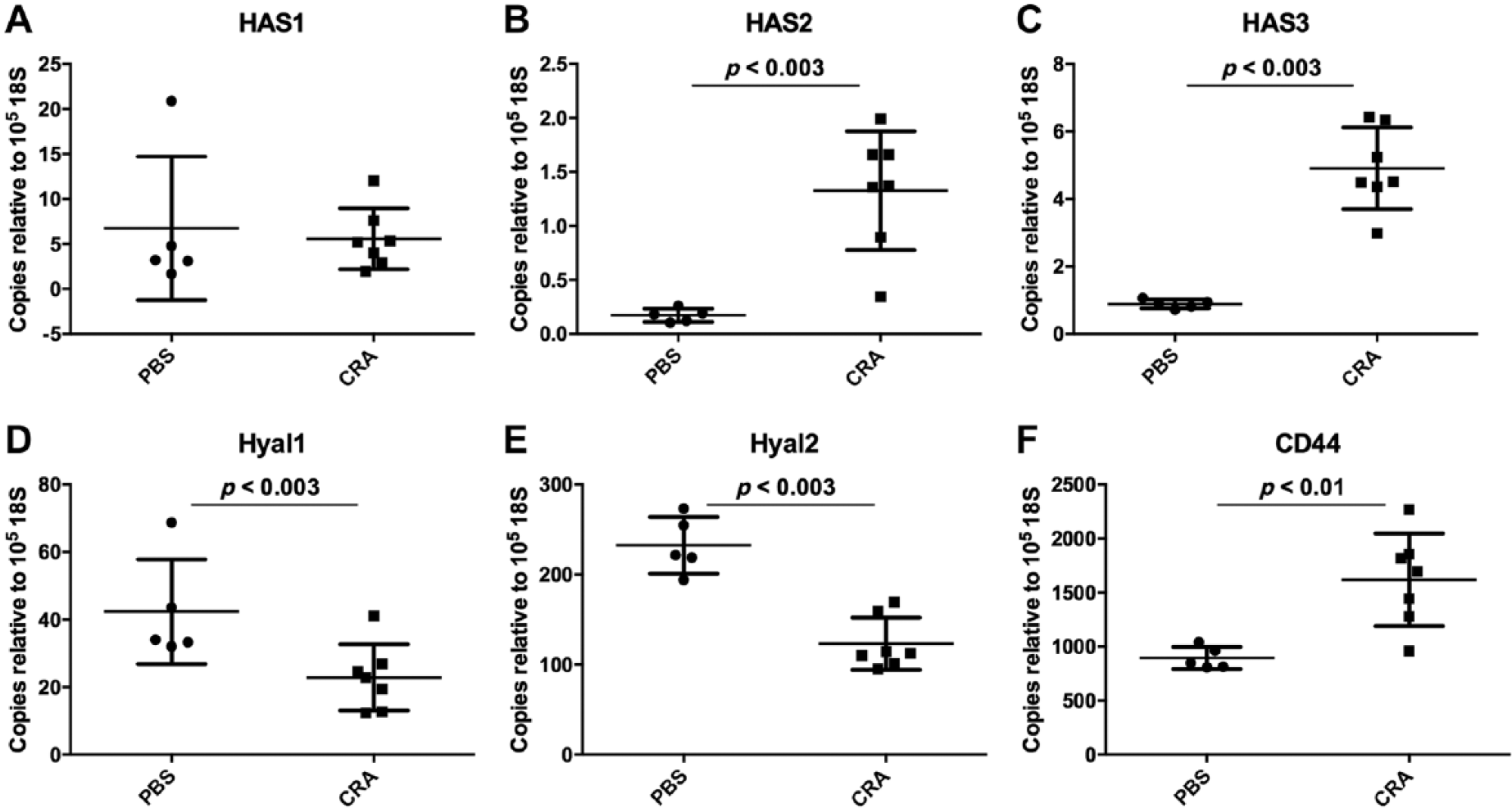
Expression of genes involved in hyaluronan (HA) production and turnover in lung tissue homogenates measured by quantitative real-time polymerase chain reaction (qRT-PCR). Expression of Has1 was not different between control mice and cockroach antigen (CRA)–treated mice (Panel A); however, expression of both Has2 and Has3 was significantly greater in CRA-treated mice (P < 0.003, Panels B and C). Expression of both Hyal1 and Hyal2 was decreased in CRA-treated mice compared with controls (P < 0.02, P < 0.003; Panels D and E) whereas the expression of Cd44 was increased in CRA-treated mice compared with control mice (P < 0.01; Panel F). PBS, phosphate-buffered saline.
Increases in Versican Accumulation in Lungs of Sensitized Mice with Chronic CRA Challenge
To investigate versican accumulation in the lungs of mice chronically exposed to CRA, we examined lung tissue from mice repeatedly challenged with CRA over 8 weeks. Sections were stained for versican as described above (Fig. 7). Similar to our findings for the acute CRA exposure model, increased versican staining was found only in the CRA-exposed mice. Versican staining was visible as early as 6 hr of CRA exposure and continued to increase with time peaking at 8 days of exposure; however, versican staining was noted throughout the duration of the 8-week exposure (Fig. 7). Closer examination of versican accumulation demonstrated that enhanced staining in the epithelial cells surrounding the airways was identified as early as 6 hr. The area of the basement membrane was positive for versican by 12 hr. Versican accumulation gradually increased over the first week of the exposure, reaching its maximum by day 8. Versican remained elevated in the subepithelial compartment through 8 weeks; however, versican was notably less than the peak observed at day 8. Staining for versican in the perivascular space lagged slightly behind that observed in the subepithelial space, but was noted as early as the 12-hr time point. Peak versican staining of the pulmonary veins occurred slightly earlier than peak versican accumulation in airway occurring at the 6-day time point. Similar to the pattern observed in the subepithelial space, versican associated with the pulmonary blood vessels was seen throughout the 8-week exposure. Versican accumulation in the lung parenchyma followed a similar pattern, first becoming visible in 6 to 12 hr and remaining elevated throughout the 8-week exposure. Interestingly, inflammatory cell infiltration in the subepithelial and perivascular spaces was most prominent after the 8-day time point, that is, coinciding with peak versican accumulation. These findings closely mirrored the time course of HA deposition as previously reported. 14
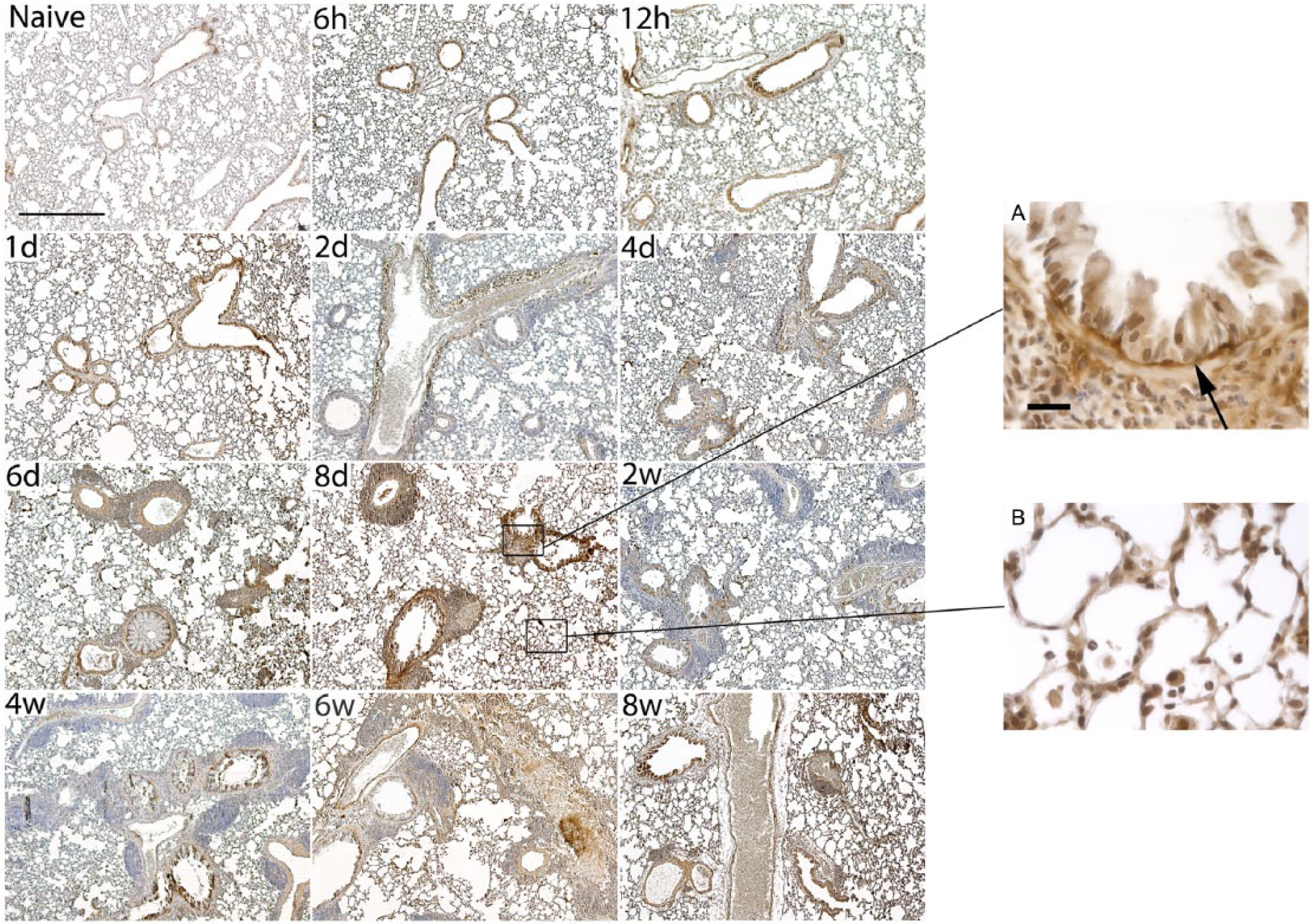
Time course of versican staining in mice sensitized and challenged with cockroach antigen (CRA) over an 8-week period. Shown in this figure are histological sections obtained from mice sensitized by intraperitoneal injection injection of 5 µg of CRA starting at 8 weeks of age (50×, scale bar = 500 µm). Mice were subsequently challenged with 5 µg of CRA in 50 µl saline by intranasal delivery once a day for 8 days starting on day 0 and then twice a week until 8 weeks. Mice were sacrificed during the course of antigen exposure at 6 and 12 hr; 1, 2, 4, 6, and 8 days; and 2, 4, 6, and 8 weeks. Staining for versican was apparent as early as 6 hr post challenge; however, peak staining was observed between 6 and 8 days of CRA challenge. Inset Images A and B show higher magnification (630×, scale bar = 20 µm) views during peak versican staining. The black arrow denotes increased subepithelial versican staining.
In addition, sections were also stained for F4/80 to highlight the presence of macrophages (Fig. 8). F4/80 staining closely mirrored the pattern observed for versican over the duration of the 8-week exposure paradigm. F4/80-positive cells began to appear around the airways and blood vessels within the first 24 hr of the exposure. Similar to versican, F4/80 staining peaked at day 6 for the vasculature and day 8 for the subepithelial space. Cells positive for F4/80 were also abundant in the interstitial space at day 8, but decreased through the rest of the 8-week exposure.
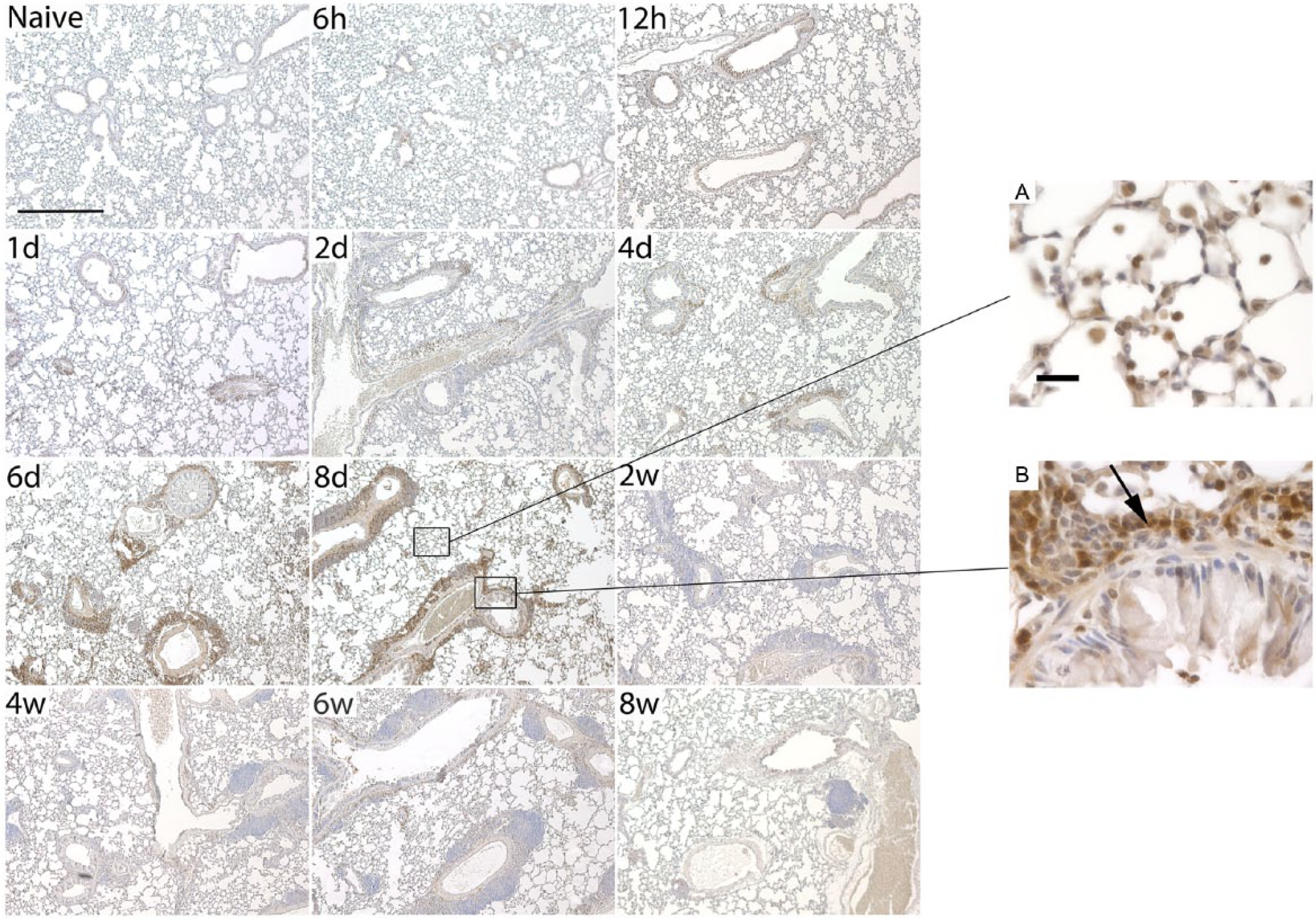
Staining for F4/80 in mice sensitized and challenged with cockroach antigen (CRA) over an 8-week period. Shown in this figure are histological sections from mice following a chronic CRA exposure occurring over an 8-week period (50×, scale bar = 500 µm). Mice were sacrificed during the course of antigen exposure at 6 and 12 hr; 1, 2, 4, 6, and 8 days; and 2, 4, 6, and 8 weeks. Staining for F4/80 was apparent within the first 24 hr following challenge; however, peak staining was observed between 6 and 8 days of CRA challenge. Inset Images A and B show higher magnification (630×, scale bar = 20 µm) views during peak F4/80 staining. The black arrow denotes increased F4/80 staining in infiltrating cells in the subepithelial space.
Greater Capacity for HA and Versican Synthesis by BECs From Asthmatic Children
Given the increased synthesis of versican and HA in lung tissue obtained from CRA-sensitized mice, localized to the subepithelial layer of the bronchioles, we investigated versican and HA expression patterns of BECs obtained from asthmatic and healthy children. These BECs were differentiated at an ALI for 4 weeks to establish a pseudostratified columnar epithelium similar to in vivo airway epithelium. Clinical characteristics of the subjects whose BECs were analyzed are presented in Table 1. qRT-PCR analysis of gene expression for the versican isoforms for the human BECs was performed. Notably, pan-versican (VCAN) transcription was found to be significantly elevated in the asthmatic BECs compared with those from healthy controls (P < 0.0001; Fig. 9A). Furthermore, V1 and V3 transcripts were also elevated in BECs derived from asthmatic children (P < 0.004 and P < 0.02, respectively; Fig. 9B, C).
Subject Characteristics.
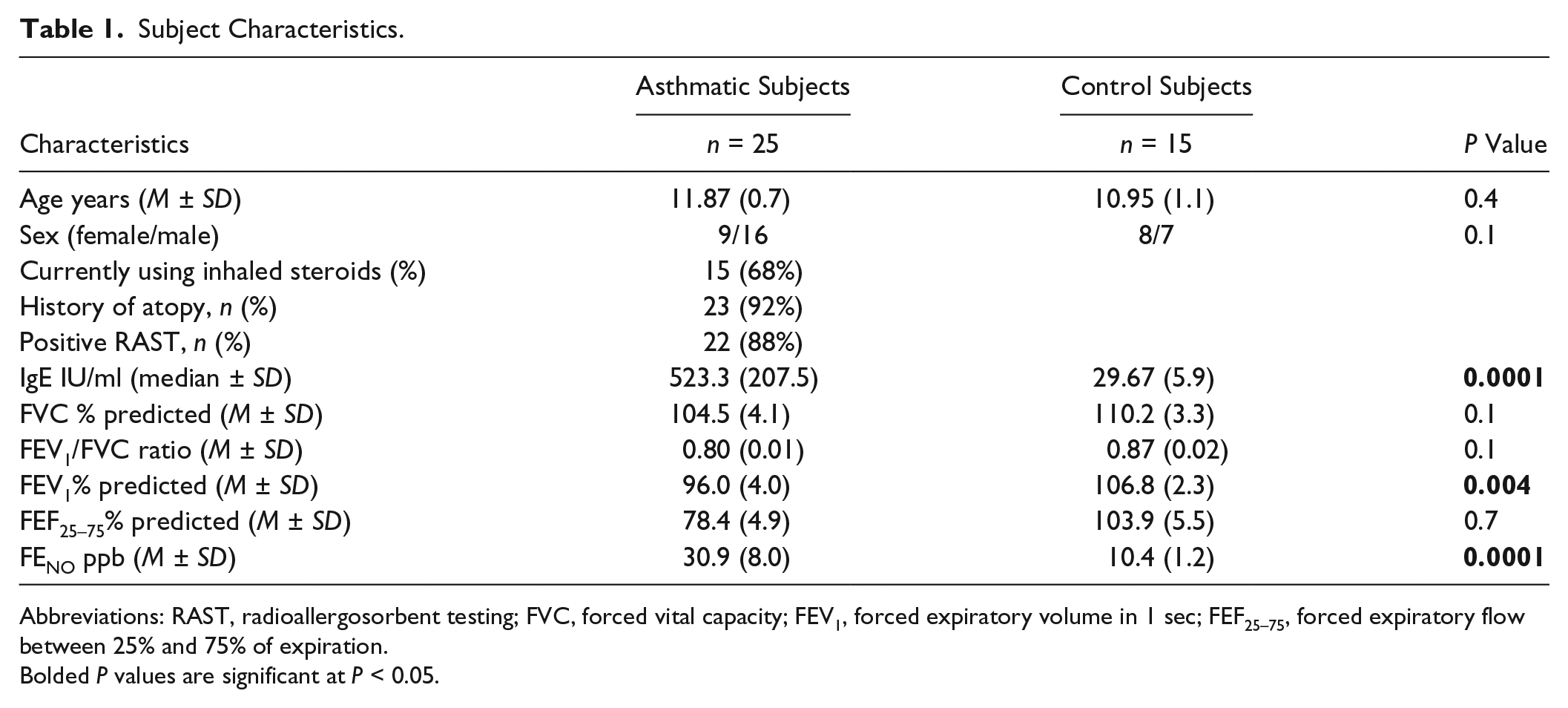
Abbreviations: RAST, radioallergosorbent testing; FVC, forced vital capacity; FEV1, forced expiratory volume in 1 sec; FEF25–75, forced expiratory flow between 25% and 75% of expiration.
Bolded P values are significant at P < 0.05.
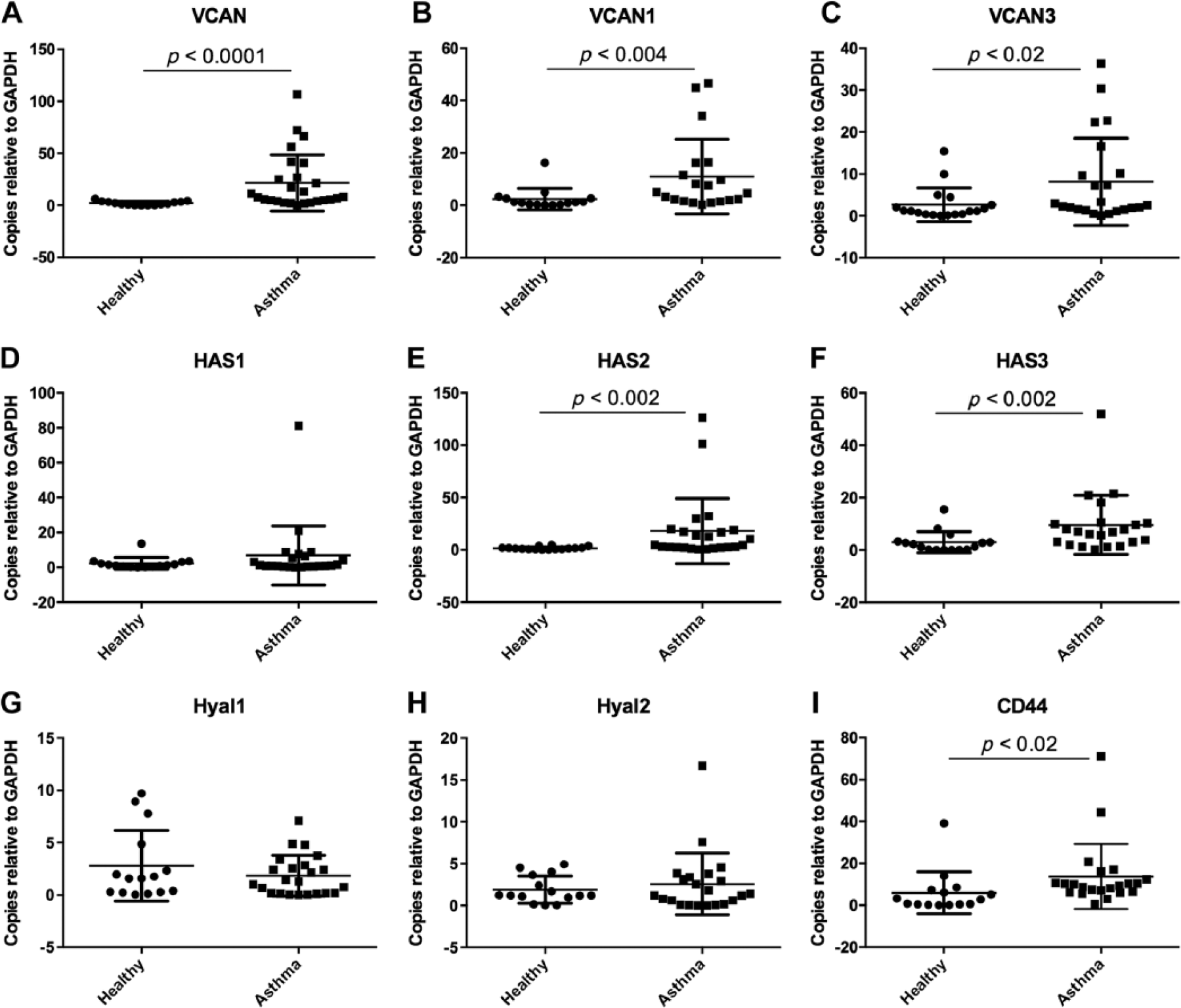
Expression of versican isoforms and enzymes involved in hyaluronan (HA) production and turnover in primary differentiated human bronchial epithelial cells (BECs) obtained from healthy and asthmatic children. Expression of the pan-versican transcript (VCAN) was elevated in asthmatic BECs compared with healthy controls (P < 0.0001; Panel A). Expression of versican isoforms V1 and V3 were also elevated in the asthmatic BECs (P < 0.004 and P < 0.02; Panels B and C). Expression of HAS1 was not different between the groups (Panel D); however, expression of both HAS2 and HAS3 was elevated in asthmatic BECs (P < 0.002; Panels E and F). No significant differences were noted in the expression of HYAL1 and HYAL2 between the groups (Panels G and H). Expression of CD44 was increased in asthmatic BECs compared with healthy controls (P < 0.02; Panel I). Abbreviation: GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
Examination of HAS showed increased expression of HAS2 (P < 0.0002; Fig. 9E) and HAS3 (P < 0.002; Fig. 9F) transcripts in the BECs from asthmatic children compared with BECs from healthy children. No differences were observed in HAS1 expression between the two groups (Fig. 9D). In contrast to the whole lung samples obtained from CRA-sensitized mice, BECs obtained from the asthmatic children displayed no differences in the expression of HYAL1 or HYAL2 compared with healthy controls (P = ns; Fig. 9G, H). Similar to the CRA-treated mice, the expression of CD44 was increased in BECs from asthmatic children (P < 0.02; Fig. 9I).
Discussion
In the present study, in a murine model of asthma, we have demonstrated that versican accumulates in the subepithelial space of bronchioles and perivascular spaces during the acute phase of CRA exposure and that this increase is accompanied by increases in the expression of the V0, V1, and V2, but not the V3 isoforms. We further show that with longer chronic exposure to CRA, versican accumulation is persistent throughout the exposure and precedes the infiltration of leukocytes and establishment of inflammation. Increases in versican staining were accompanied by increased F4/80 staining, a marker for macrophages, in both the acute and chronic exposure models, suggesting a possible contribution of macrophages to versican accumulation. 23 We further show that these changes parallel HA accumulation, as demonstrated by others, and peak following 8 days of repeated CRA exposure. 14 In addition, we have compared the expression of versican and HA in our in vivo murine model with an in vitro cell culture model that utilizes differentiated primary human BECs obtained from both healthy and asthmatic children. We found elevated expression of the VCAN, VCAN1, and VCAN3 isoforms, as well as HAS2 and HAS3, suggesting that the respiratory epithelia may be an additional source of these ECM molecules.
Versican is a chondroitin sulfate-rich proteoglycan that exists in at least four different isoforms generated by alternative splicing of its different domains (see review). 15 In healthy tissue, including the lung, versican content is typically low; however, it increases dramatically in the context of disease and inflammation, and is known to interact closely with HA, TSG-6, and CD44.24,25 In addition, studies have confirmed that increases in versican expression influence cell adhesion, proliferation, migration, and survival as well as regulation of key inflammatory response.26–28
Versican accumulation in the subepithelial layer in airways of atopic asthmatics has been described previously and correlates with the degree of AHR to methacholine challenge. 16 In addition, increased versican accumulation is associated with both small and large airway remodeling seen in autopsy specimens following fatal asthma exacerbations. 29 In a separate, more recent study, subjects who had uncontrolled asthma demonstrated increased versican accumulation in biopsy specimens from their central airways compared with healthy subjects or those who had well-controlled asthma. 30 Interestingly, these same subjects also demonstrated an increased number of myofibroblasts per unit area in tissue samples. Of note, lung fibroblasts obtained from asthmatic adults have been shown to produce greater amounts of proteoglycans including versican in vitro compared with lung fibroblasts obtained from healthy adults. 31 Additional studies have confirmed the presence of versican in the sputum of adults with severe asthma, which correlates negatively with their FEV1, indicating a positive correlation with airway obstruction. 9
Few studies have investigated the role of versican in animal models of lung disease. As mentioned, increased versican expression by lung macrophages leading to increased lung versican accumulation has been described in an lipopolysaccharide (LPS)-induced mouse model of acute lung injury; however, these effects were not specific to airway disease. 26 In another study, Brown Norway rats were utilized in an ovalbumin sensitization model to describe increased proteoglycan deposition in the airways and BALF. 32 Increased versican staining was observed in the airways and blood vessels of the ovalbumin-exposed rats, which co-localized with alpha-smooth muscle actin staining, suggesting an association with myofibroblasts in these tissues. Furthermore, these changes were not reversible following treatment with budesonide, a commonly used inhaled corticosteroid. 32
While the presence of versican in asthmatic airways has been reported previously, as discussed above,29,30 no studies exist to date demonstrating that versican is produced directly by the airway epithelium. Our finding that human BECs can directly express versican is intriguing given the subepithelial pattern of versican accumulation in both mouse exposure models studied. In addition, increased staining of epithelial cells was observed at early time points in the chronic CRA exposure model, which localized to the epithelial cells. Indeed, exposure to CRA via the airway epithelium has been shown to activate the expression of Th2 cytokines, 33 which may result from activation of the toll-like receptor (TLR) 2, 34 TLR4, 35 or proteinase-activated receptor-2 (PAR2) pathway. 36 However, it is important to point out that the underlying mesenchymal cells and possibly other infiltrating cell types such as macrophages likely also contribute to accumulation of versican in vivo. The possibility that BECs could have an impact on the subepithelial accumulation of versican either directly or indirectly via intracellular signaling mechanisms is currently unknown and merits further investigation.
The results of the present study are consistent with previous reports13,14 of a robust, predominantly eosinophilic, inflammatory response in the airways and BALF of mice, albeit using a slightly different CRA sensitization and exposure paradigm. We observed significant leukocyte infiltration in the airways and blood vessels of CRA-treated mice that was closely associated with areas of increased versican and HA staining. Our finding of increased versican accumulation in the CRA-treated mice is the first evidence of such an occurrence in a murine CRA sensitization model. We demonstrate a robust staining pattern for versican in several compartments of the lung in the CRA-treated mice; however, the accumulation at the subepithelial layer is perhaps the most striking as this would correlate best with the pathological findings of ECM deposition in that compartment in human asthma. In addition, the enhanced expression of multiple versican isoforms in both the murine and human epithelium is also a novel finding. Although the relative contributions of the various isoforms of versican are not presently well understood, future studies identifying the unique contributions of versican to airway disease will provide further insight into this question.
In addition to the enhanced versican accumulation following CRA sensitization, we have also confirmed increases in HA accumulation in CRA-exposed mice. 14 Increases in HA observed in CRA-treated mice were accompanied by increased expression of Has2 and Has3 that occurred in parallel with significant decreases in Hyal1 and Hyal2 expression in whole lung lysates obtained from CRA mice. When compared with expression patterns of differentiated human BECs obtained from asthmatic children, similar expression patterns emerged for HAS2, HAS3, and CD44; however, the expression of HYAL1 and HYAL2 was not different in BECs from healthy controls. The latter finding is likely explained by the use of whole lung lysates in the mouse studies compared with isolated BECs in the human studies as leukocytes are known to be a major source of enzymes that degrade the ECM including the Hyals. 37 Separating the relative contributions from the various cell types in the mouse studies was not possible.
Previous studies have examined the role of HA in the inflammatory changes observed in animal models of lung injury and allergic asthma. Bleomycin administration causes diffuse lung fibrosis in mice that is associated with increased levels of HA in the lung tissue and BALF; however, the injury seen in this model is more diffuse and is not specific to the airways. 38 Many other lung injury models have been described in animal studies; however, models of antigen sensitization and subsequent challenge have been the most effective at replicating the airway pathology seen in asthma. Using a murine model of ovalbumin sensitization, Cheng and colleagues have reported airway eosinophilia and increased HA accumulation. 13 More recent work by this group, using a CRA sensitization and subsequent chronic exposure paradigm, has demonstrated a similar time-dependent accumulation of HA in the lungs of CRA-treated mice that peaked at 6 days in the tissue sections and 8 days in the BALF. 14 HA accumulation in this study correlated with downregulation of Hyal1 and Hyal2 with sustained elevated expression of Has2 despite an early peak and decline in Has1 expression. The time periods correlated very closely with observation of inflammatory cell infiltration of the lung tissue, which consisted predominantly of eosinophils, as demonstrated by tissue staining in the first week of the experiment. However, by 8 weeks, a more lymphocytic infiltrate predominated, and HA staining gave way to increased collagen staining, suggesting a progression of airway remodeling over time. 14 In the present study, we compared versican expression in this model of chronic 8-week CRA exposure with our more acute 72-hr CRA exposure model. Our findings are complimentary to the previously reported results 14 and add to the growing understanding of proinflammatory matrices in the development of asthmatic airway inflammation.
Another emerging concept regarding the role of versican and HA in airway inflammation is that HA- and/or versican-enriched matrices have been shown to be an important “landing strip” for leukocytes during the establishment and maintenance of inflammatory processes in the airway. Lauer and colleagues demonstrated that treatment of the airway epithelial cells with either poly I:C or tunicamycin, a potent inducer of endoplasmic reticulum oxidative stress, led to increased production of HA; 39 however, the HA accumulation localized to different regions of the polarized epithelium. In this model, treatment with tunicamycin led to HA accumulation on the apical surface of the ALI culture, while treatment with poly I:C led to HA accumulation in the basement membrane of these cultures. In a monocyte adhesion assay, only the tunicamycin-treated cultures, that is, displaying apical HA accumulation, demonstrated increased adhesion of monocytes at 4C. The increased monocyte adhesion was abrogated by either treatment with exogenous Streptomyces Hyal or restoration of the endogenous monocyte Hyal activity by returning them to 37C. Given the physical limitations of the model system, the adherence of monocytes to the HA accumulated in the basement membrane was not feasible. While several other studies have demonstrated an important role for HA-dependent binding of monocytes in other models of inflammation,7,40,41 far fewer studies have investigated the role of versican in this process. A previous study from members of our group has demonstrated that cultured human lung fibroblasts treated with poly I:C produce an ECM that is enriched with versican and HA, which adheres monocytes. 8 Monocyte binding in this model was inhibited by both antiversican antibodies and digestion of the ECM with Hyal, suggesting that both versican and HA are important for monocyte adhesion. In a separate study, versican was also found to modulate the HA-dependent binding of T lymphocytes to ECM produced by fibroblasts treated with poly I:C. 17 Taken together, these studies suggest an important role for versican in the modulation of inflammatory processes. Whether these effects occur in conjunction with or independently from the effects of HA remains an active area of investigation.
Another aspect of the present study is the finding that F4/80 staining closely mirrors the temporal and anatomical staining pattern observed for versican. In a recent study, isolated lung macrophages were shown to upregulate versican synthesis in response to stimulation with LPS despite low levels of expression at baseline. 26 Furthermore, histological sections from mice exposed to LPS demonstrated increased versican staining, suggesting that macrophages can contribute to the production of versican in an acute lung injury model. In a separate series of experiments, a similar pattern of staining using anti-ER-HR3, a different marker of monocytes/macrophages, was reported; however, the presence of versican was not evaluated. 14 Thus, we may only speculate on this association based on the present data. However, given the evolving concepts of the role that inflammatory cells such as macrophages play on ECM generation and remodeling, future studies aimed at directly investigating the link between the presence of macrophages and accumulation of ECM will be an important area of investigation.
Our study does have several important limitations. First, the intranasal administration of CRA to the mice can lead to variable amounts of the compound reaching the lungs, despite careful standardization of the exposure protocol. We had the benefit of pathological examination of the lung tissue for each of the animals that was studied and observed pathological changes indicative of airway inflammation in all the CRA mice studied while no pathology was confirmed in any of the PBS control mice. Second, the examination of whole lung lysates does not allow for isolation of the various contributions by different cell types; however, we were able to compare the data obtained from the mouse studies with isolated differentiated BECs obtained from children with and without atopic asthma. While there are likely contributions from multiple cell types to the accumulation of both HA and versican, we have shown that epithelial cells are capable of contributing to the production of versican. The latter is of particular interest given the accumulation of versican in the subepithelial space. Furthermore, this leaves open the possibility of developing inhaled therapeutic compounds to target the epithelium in this process. Unfortunately, our study was not designed to disrupt the production of versican in the context of CRA sensitization; however, future studies in transgenic versican knockout mice should provide greater information on the contributions of versican in asthmatic airway disease.
In summary, we demonstrated that, in addition to enhanced production of HA, mice exposed to a CRA sensitization/exposure protocol demonstrated increased versican accumulation in their airways. Furthermore, we observed increased versican expression in differentiated human BECs obtained from asthmatic as compared with healthy children. Given the association of versican and airway inflammation, we hypothesize that versican contributes to the inflammatory response observed in asthmatic airways either in concert or independently of the effects of HA. We have also demonstrated similar patterns of staining for versican and the macrophage marker F4/80, raising the question of macrophage involvement in versican accumulation in the lung. Additional studies to determine whether aspects of asthmatic airway inflammation drive versican accumulation or conversely are versican-dependent will be the focus of future investigation.
Footnotes
Acknowledgements
The authors thank Virginia M. Green, PhD, for careful editing and preparation of the manuscript.
In Memorium
The authors dedicate this study to the memory of Dr. Mark Lauer, a valued friend and colleague at the Lerner Institute, Cleveland Clinic, whose untimely death cut short his influential and inspirational work on extracellular matrix and asthma.
Author Contributions
MAA, JSD, CWF, SFZ, and TNW were responsible for the conception and design; SRR, GK, AS, GC, MAA, MJM, JSD, CWF, SFZ, and TNW contributed to the analysis and interpretation of data; SRR, GK, AS, and GC performed experiments; and SRR, GK, MJM, JSD, CWF, SFZ, and TNW were responsible for drafting, editing, and reviewing manuscript.
Competing Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Parker B. Francis Fellowship (S.R.R), National Institutes of Health Grants P01 HL098067 (J.S.D., S.F.Z., C.W.F., T.N.W.), R56 HL128361 (J.S.D.), R01 AI068731 (S.F.Z.), P01 HL081064 and P01 HL103453 (M.A.A.), and the University of Auckland, Faculty Development Fund (M.J.M.).
