Abstract
Huntingtin-associated protein-1 (HAP1) was initially identified as a binding partner of huntingtin, the Huntington's disease protein. Based on its preferred distribution among neurons and endocrine cells, HAP1 has been suggested to play roles in vesicular transportation in neurons and hormonal secretion of endocrine cells. Given that HAP1 is selectively expressed in the islets of rat pancreas, in this study, we analyzed the expression pattern of HAP1 in the islets. In rats injected intraperitoneally with streptozotocin, which can selectively destroy β-cells of the pancreatic islets, the number of HAP1 immunoreactive cells was dramatically decreased and was accompanied by a parallel decrease in the number of insulin-immunoreactive cells. Immunofluorescent double staining of pancreas sections showed that, in rat islets, HAP1 is selectively expressed in the insulin-immunoreactive β-cells but not in the glucagon-immunoreactive α-cells and somatostatin immunoreactive δ-cells. In isolated rat pancreatic islets, ×80% of cells expressed both HAP1 and insulin. Expression of HAP1 in the INS-1 rat insulinoma cell line was also demonstrated by immunofluorescent staining. Western blotting further revealed that HAP1 in both the isolated rat pancreatic islets and the INS-1 cells also has two isoforms, HAP1A and HAP1B, which are the same as those in the hypothalamus. These results demonstrated that HAP1 is selectively expressed in β-cells of rat pancreatic islets, suggesting the involvement of HAP1 in the regulation of cellular trafficking and secretion of insulin.
H
Rat HAP1 consists of two isoforms spliced alternately that differ at the C terminus (aa 579–599 in HAP1A vs 579–629 in HAP1B). These two isoforms are expressed at similar levels in rat brain (Li et al. 1995). In human central nervous system, only a single major form of HAP1 has been characterized that has great similarity with rat HAP1A (Li et al. 1998b). In mouse, there are three HAP1 transcripts resulting from alternative splicing: HAP1A, HAP1B, and HAP1C (Nasir et al. 1998,1999).
Earlier studies showed that HAP1 is a widely distributed brain-specific protein with the highest expression level in the hypothalamus (Li et al. 1996; Gutekunst et al. 1998; Page et al. 1998). Subsequent Northern blotting and ISH study revealed a wide range of distribution of the mRNA not only in the brain and the spinal cord but also in the peripheral nervous system and reproductive and pituitary glands (Dragatsis et al. 2000). Our previous immunohistochemical study demonstrated for the first time that HAP1 is expressed in some of the small peptide-, protein-, and amino-acid analog and derivative-secreting endocrine cells, such as cells in the anterior lobe of the pituitary, the parafollicular cells of the thyroid gland, the medullae of adrenal glands, the pancreas islets, and the mucosa of stomach and small intestine (Liao et al. 2005).
Pancreatic islets contain a number of endocrine cells, including α-cells secreting glucagon, β-cells secreting insulin, δ-cells secreting somatostatin, and PP-cells secreting pancreatic polypeptide. It is unknown whether HAP1 is expressed in every kind of islet cell or in a specific kind(s) of islet cell. To reveal the expression pattern of HAP1 in rat pancreatic islets, at first, we observed the effect of intraperitoneal injection of streptozotocin (STZ), a naturally occurring chemical that is selectively toxic to the insulin-producing β-cell of the pancreas in mammals (Like and Rossini 1976), on HAP1 expression in the islets; subsequently, the relationships of HAP1 to α-, β-, and δ-cells in rat pancreatic islets were examined by immunofluorescent double staining; finally, the expression of HAP1 and molecular property of HAP1 in isolated rat pancreatic islet cells and the INS-1 rat insulinoma cell line were analyzed by immunofluorescent staining and Western blotting. The results demonstrated that HAP1, which has the same two isoforms as those in the brain, is selectively expressed in β-cells of rat pancreatic islets.
Materials and Methods
Animals
Male Sprague-Dawley rats (200–220 g) were housed in a temperature- and humidity-controlled environment (12:12 hr light/dark cycle). Animal experiments were conducted in accordance with the Guidelines for Animal Experimentation of the Experimental Animal Science Center of Tongji Medical College, Huazhong University of Science and Technology.
STZ Treatment
Rats were intraperitoneally injected with a single bolus dose of 60 mg/kg STZ (Sigma-Aldrich; St. Louis, MO) solution in citrate buffer; control rats were injected with the citrate buffer alone without STZ. Both the STZ-treated and the control rats were then routinely maintained for 7 days. Blood samples were collected by bleeding rats from the cut tail, and serum glucose levels were measured by a serum glucose reader (Johnson and Johnson Co.; Milpitas, CA).
Tissue Preparation
The rats were deeply anesthetized by intraperitoneal injection of sodium pentobarbital (100 mg/kg body weight) and then perfused via the ascending aorta with 100 ml of 0.01 M sodium phosphate-buffered saline (PBS) 0.85% (pH 7.4), followed by 300 ml of 0.1 M sodium phosphate buffer (pH 7.4) containing 4% para-formaldehyde. After perfusion, pancreas were removed and postfixed in the same fixative at 4C for 4 to 6 hr. The pancreas were soaked in cold 0.1 M phosphate buffer containing 30% sucrose at 4C overnight for cryoprotection, quickly frozen, and cut into sections of 20 μm on a cryostat. Sections were mounted onto slides coated with a mixture of gelatin and chromic alum or poly-
Isolation of Pancreatic Islets
Pancreas were removed from rats sacrificed under anesthesia by intraperitoneal injection of sodium pentobarbital and then minced and washed with Ca2+- and Mg2+-free Krebs-Ringer bicarbonate buffer (KRBB). After the sample was centrifuged for 2 min at 4C at 300 × g, the pellet was resuspended and digested with 0.2% collageneae V (Sigma-Aldrich) in KRBB at 37C for 30 min in an agitating water bath. Digestion was terminated by adding cold KRBB. Digested tissue was filtered through 0.5-mm mesh and then centrifuged for 2 min at 4C at 300 × g. After the supernatant was discarded, the pellet was resuspended in 25% Ficoll (Sigma-Aldrich) with 10 mM HEPES (pH 7.0) and was layered over by various concentrations of Ficoll (23%, 20%, and 11%). Islets were collected from the interface between 11%, 20%, and 23% after centrifugation.
Cell Culture
Rat INS-1 insulinoma cells were cultured at 5% in CO2/95% air at 37C in RPMI-1640 medium (GIBCO-Invitrogen Corp.; Grand Island, NY) containing 11.2 mM glucose and 2 mM
Immunohistochemistry
For immunohistochemical localization of HAP1 or insulin, the sections mounted on gelatin- and chromic alum-coated slides were processed by the avidin-biotin-peroxidase (ABC) method after being rinsed in PBS. To reduce endogenous peroxidase activity and to prevent nonspecific antibody binding, sections were treated in 1% hydrogen peroxide-PBS for 2 hr and then blocked by PBS containing 5% normal goat serum for 30 min after incubation in PBS containing 1% Triton X-100. Thereafter, sections were incubated with a guinea pig polyclonal antibody to a glutathione S-transferase (GST) fusion protein containing aa 278–445 of rat HAP1 (GST-HAP1), EM78 (1:5000; a generous gift from Professor Xiaojiang Li, Department of Human Genetics, Emory University School of Medicine, Atlanta, Georgia), or mouse antibody to insulin (1:1000; Sigma-Aldrich), at 4C for 35 to 40 hr, followed by incubation with biotinylated goat anti-guinea pig immunoglobulin G (IgG) or biotinylated goat anti-mouse IgG (1:200; Vector Labs, Burlingame, CA) at room temperature for 2 hr and avidin–biotin complex (1:100 Vector ABC Elite; Vector Labs) at room temperature for 2 hr. Antibodies and the avidin–biotin complex were diluted with PBS containing 3% Triton X-100 and 5% normal goat serum. Between incubations, the tissues were rinsed in PBS. Finally, the immunoreactive products of HAP1 or insulin were visualized by incubation with 0.02% diaminobenzidine (Sigma-Aldrich) and 0.005% hydrogen peroxide in 0.05 M Tris-HCl buffer for 10 to 15 min. For the control, the primary antibody EM78 was absorbed with excess GST-HAP1 fusion protein or GST. For immunofluorescent double labeling of HAP1/glucagon, HAP1/insulin, or HAP1/somatostatin in rat pancreas, untreated rat pancreas sections were permeabilized and blocked as described above and then incubated with a mixture of two primary antibodies at 4C for 48 hr, followed by incubation with a mixture of two fluorescein-conjugated secondary antibodies at room temperature in the dark for 3 hr. The primary antibodies and secondary antibodies used for immuno-fluorescent double labeling were combined as described in Table 1. After being washed three times with PBS for 10 min each time, sections were mounted with 10% glycerin in PBS and examined and photographed under a laser scanning confocal microscope (Olympus FV500; Tokyo, Japan).
For immunofluorescent double labeling of HAP1/insulin in rat pancreatic islet cells, the isolated islets were further digested into single cells by 0.15% dispase II in Ca2+ - and Mg2+-free KRBB for 3 to 4 min at 37C. The dispersed cells were then plated on glass cover slips, fixed in 4% paraformaldehyde in PBS for 30 min at 4C, and then permeabilized, blocked, and incubated with a mixture of EM78 and mouse anti-insulin antibody at 4C for 48 hr, followed by incubation with a mixture of Rhodamine Red-conjugated donkey anti-guinea pig IgG and FITC-conjugated donkey anti-mouse IgG at room temperature under dark conditions for 3 hr. For immunofluorescent labeling of HAP1 in INS-1 cells, cultured cells were fixed, permeabilized, and blocked as described above, and incubated with EM78 overnight at 4C, followed by incubation with Rhodamine Red-conjugated donkey anti-guinea pig IgG. Photographs were taken with a DXM1200 color digital camera (Nikon; Tokyo, Japan) equipped with an Eclipse TE 2000-S fluorescent microscope (Nikon).
In Situ Hybridization
Total RNA was purified from rat brains with Trizol reagent, and the cDNA fragment located at position 480-789 downstream from the ATG start codon of rat HAP1A (GenBank accession number NM-024133.2) was obtained by RT-PCR using a sense primer (5'-ATAGAATTCCCTGAACACAGCCGCTCG-3') and an antisense primer (5'-ATAAAGCTTGTGCAGCGTCT-CAGCCTTAG-3'). The vector used for the production of the cRNA probe was constructed by insertion of a cDNA fragment of 310 bp containing a HAP1A and HAP1B consensus sequence of rat HAP1A (Li et al. 1995) into an EcoRI-HindIII-digested pSPT19 vector. Using a Dig-RNA labeling kit (Roche Diagnostics; Penzberg, Germany), we synthesized antisense or sense cRNA probes with T7 or SP6 RNA polymerase, which were purified and kept at −80C until use. ISH with the antisense and sense Dig-labeled cRNA probes was performed as previously described (Fujinaga et al. 2004). Briefly, the sections mounted on poly-
Combinations of primary antibodies and secondary antibodies
Western Blotting
Homogenates of the hypothalamus of untreated rat, cultured INS-1 cells, and isolated rat pancreatic islets were resuspended in lysis buffer [50 mM Tris (pH 8.0), 150 mM NaCl, 1% Triton X-100] with protease inhibitor cocktail (Sigma-Aldrich) and phenylmethylsulfonyl fluoride (100 μg/ml) and centrifuged at 12,000 × g for 15 min at 4C. The extracted proteins were then resolved by 10% SDS-PAGE and transferred onto nitrocellulose membrane. The membrane was blocked with 5% skim milk in PBS for 1 hr at room temperature and incubated with EM78 (1:5000) overnight at 4C. The immunoreactive bands were visualized using an enhanced chemiluminescence kit (Pierce Thermo Scientific; Rockford, IL).
Results
HAP1 Immunoreactivity and Hybridization Reactivity Are Localized to Cells in the Islet Center of Pancreas
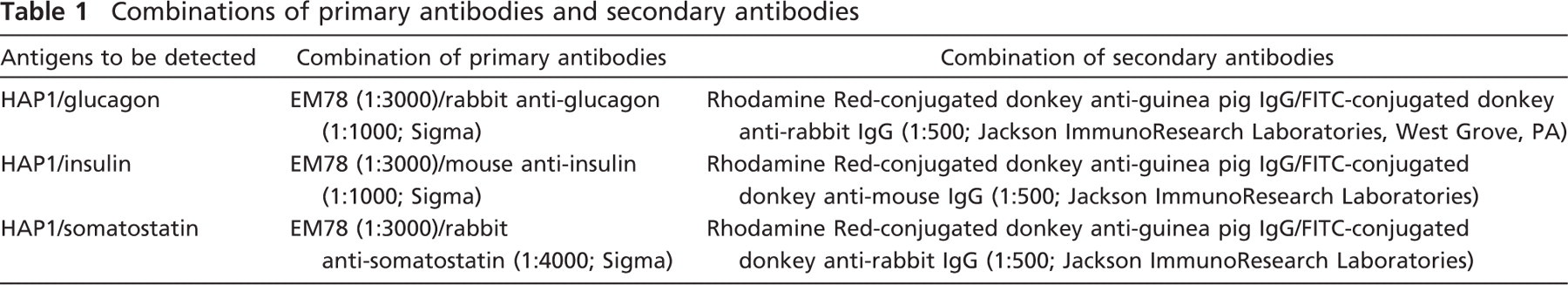
ABC immunohistochemistry of rat pancreas tissue sections, using an antibody generated for HAP1, detected immunoreactive cells that were exclusively present in the endocrine portion of the pancreas, the islets of Langerhans, but not in the exocrine tissue surrounding the islets. The majority of islet cells are HAP1-positive cells, staining being strong in the central portion but negative or weak at the periphery of the islet (Figures 1A and 1B). HAP1-immunoreactive products were localized to individual islet cells as seen by the clustering around the unstained nuclei (Figure 1B). Evidence that the immunostaining in the islets of Langerhans was completely abolished by incubation of EM78 with the excess GST-HAP1 fusion protein (Figure 1C) but not by incubation of EM78 with excess GST (Figure 1D) and the negative staining using preimmune guinea pig serum (data not shown) indicate the specificity of EM78.
In pancreas sections in situ hybridized with the anti-sense probe, HAP1 mRNA could be detected only in the islets. No labeling was observed in the exocrine tissue. The distributive pattern of HAP1 ISH signal in the islets was similar to that of HAP1 immunoreactivity (Figure 1E). No positive signal was detected in the sections in situ hybridized with the sense probe (Figure 1F), demonstrating the specificity of the anti-sense probe used in the present study.
Immunohistochemical and ISH histochemical staining for HAP1 in the pancreas of untreated rat.
STZ Destroys HAP1 Immunoreactive Cells in Pancreas Islets
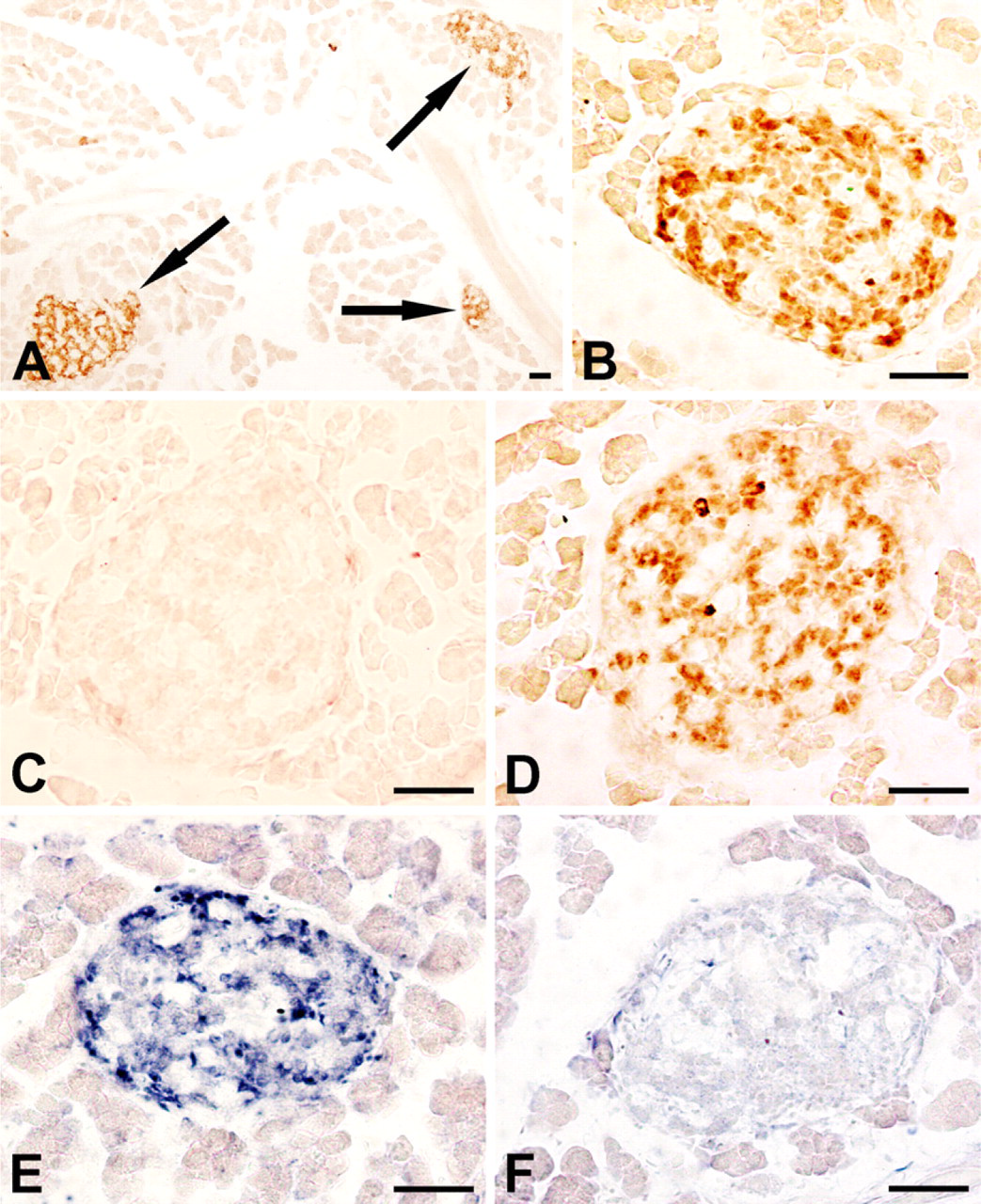
All rats given bolus injections of STZ displayed symptoms of diabetes. The average glucose level was 26.2 mmol/liter in STZ-treated rats, which was significantly higher than that of the control rats injected with citrate buffer alone (6.3 mmol/liter). ABC immunohistochemistry showed a decreased number of HAP1-positive cells and also the decreased HAP1 immunoreactivity in these positive cells in the pancreas sections of STZ-treated rats (Figure 2A). The change of insulin staining pattern was correlated with that of HAP1 in these rats (Figure 2B), but the staining pattern for HAP1 and insulin in the control rats remained similar to those in normal rats (data not shown).
HAP1 Immunoreactivity Colocalizes With Insulin Immunoreactivity in Pancreas Islets
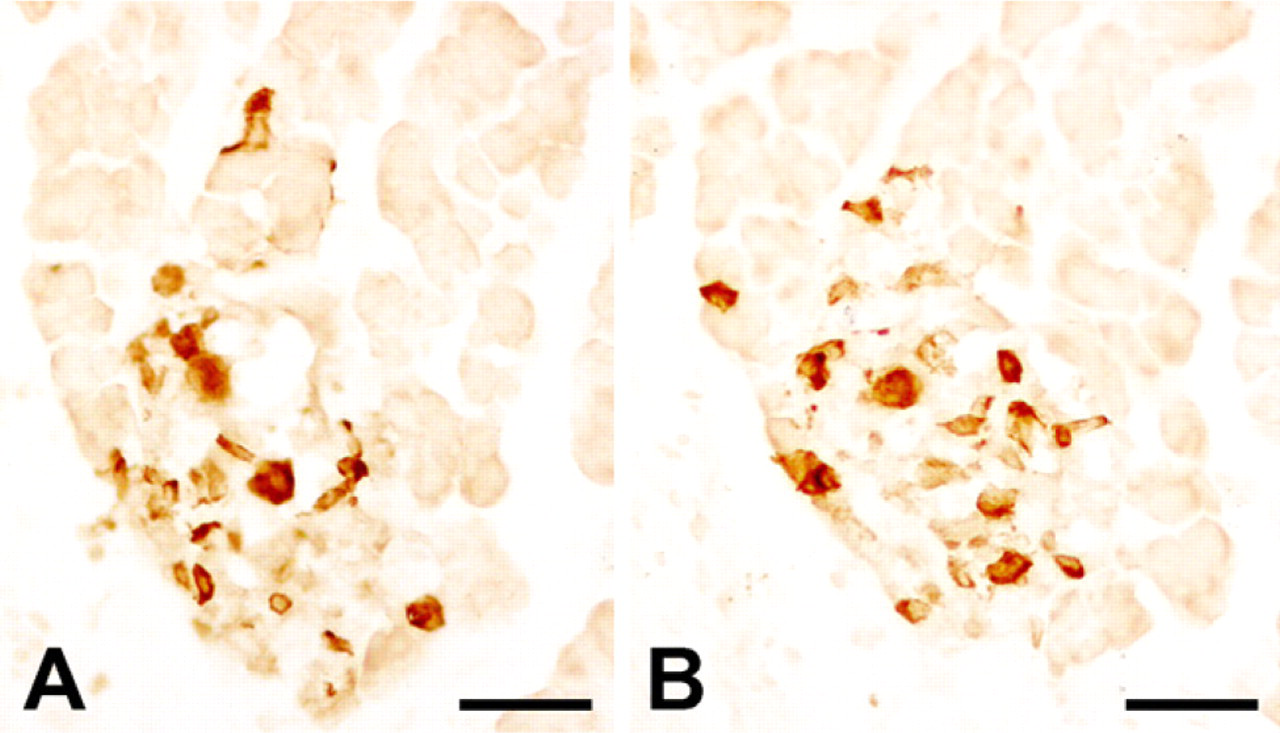
To examine the distribution of HAP1 in the islets, double immunofluorescent staining was performed with tissue sections from rat pancreas by using antibodies to HAP1/glucagon, HAP1/insulin, or HAP1/somatostatin. For antigen identification in the costained tissue, Rhodamine Red was used to detect HAP1, and FITC was used to detect glucagon, insulin, or somatostatin. HAP1-immunoreactive cells (stained red) were also positive for insulin (Figures 3B1–B3) but distinct from glucagon-immunoreactive cells (Figures 3A1–A3) and somatostatin-immunoreactive cells (Figures 3C1–C3) (stained green).
Immunohistochemical staining for HAP1 and insulin in the pancreatic islets of an STZ-treated rat.
Islet β-Cells Express Both HAP1A and HAP1B
In the isolated rat pancreatic islets, immunofluorescent staining using EM78 detected that ×80% of dispersed single islet cells were immunoreactive for HAP1 (Figures 4A and 4D). Double labeling using EM78 and insulin antibody showed that these HAP1-immunoreactive islet cells also expressed insulin (Figures 4A–4C). Immunofluorescent staining using antibody to HAP performed on cultured INS-1 cells revealed that HAP1 immunoreactivity was distributed in the cytoplasm, including processes of INS-1 cells; no nuclear staining was observed (Figure 4E). To examine whether the HAP1 protein expressed in islet cells and INS-1 cells have the same molecular property as that expressed in the brain, protein extracts from isolated rat islets, INS-1 cells, and rat hypothalamus were analyzed by Western blotting. As expected, the EM78 recognized two isoforms in the protein preparation of both islet cells and INS-1 cells, which have the same molecular sizes as HAP1A and HAP1B detected in rat hypothalamus protein preparation, respectively (Figure 4F).
Discussion
Initial studies showed that HAP1 is a protein distributed mainly in the neuronal cells, and is highly expressed in the limbic-related forebrain regions and midline/periventricular brainstem regions, with dramatic enrichment in the pedunculopontine nuclei, the accessory olfactory bulb, and the hypothalamus. In contrast, little expression of HAP1 is detected in the striatum and thalamus (Li et al. 1995,1996; Page et al. 1998; Gutekunst et al. 1998; Martin et al. 1999). Then, Dragatsis et al. (2000) studied the HAP1 gene expression in developing and adult mice by Northern blotting and ISH, and detected HAP1 mRNA not only in the nervous system but also in the reproductive system and pituitary glands (Dragatsis et al. 2000). By using immunohistochemistry, we have also shown that HAP1 is highly expressed in the rat endocrine system, including pancreatic islets (Liao et al. 2005). In the present study, HAP1 expression in both protein and mRNA levels in the pancreatic islets was further demonstrated by using immunohistochemistry and ISH histochemistry, respectively.
Double immunofluorescent staining for HAP1 and one of the islet hormones: glucagon, insulin, and somatostatin, in pancreas islets of untreated rat.
Pancreatic islets contain several kinds of endocrine cells, including α-cells, β-cells, δ-cells, and PP-cells. The present immunohistochemical and ISH histochemical observations found that the majority of islet cells are HAP1-expressing and that both immunoreactivity and ISH reactivity of HAP1 were strong at the central portion but negative or weak at the outer edge of the islet in untreated rat, suggesting that HAP1 might be localized mainly in pancreatic β-cells. In agreement with the finding in the untreated rats, the results from examination on the effect of STZ on expression of HAP1 in the islet indicates that HAP1 expresses in β-cells sensitive to STZ. In the islets of STZ-treated rats, a dramatic decrease in the number of HAP1-immunoreactive cells was parallel with that of insulin-immunoreactive cells. Furthermore, double immunofluorescent staining showed that HAP1 was colocalized with insulin but not with glucagon and somatostatin in the islet sections of untreated rats and that all the isolated rat single islet cells that expressed HAP1 were also reactive to the anti-insulin antibody. It is therefore demonstrated that HAP1 is selectively expressed in β-cells but not in α-cells and δ-cells of rat pancreatic islets. INS-1 cells, a rat insulinoma cell line, were immunofluorescently stained positive for HAP1, which further supports the fact that HAP1 is expressed in islet β-cells.
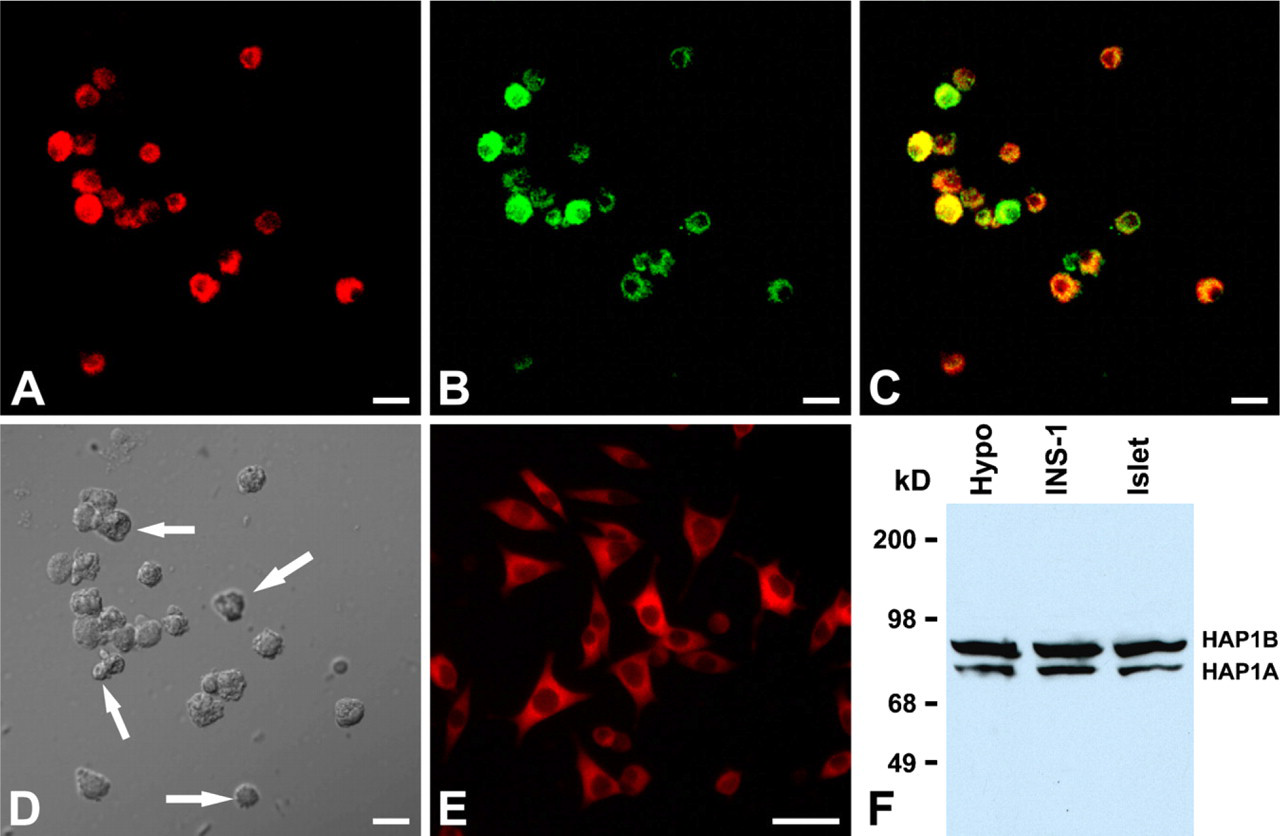
HAP1 immunoreactivity in the isolated rat islet cells and INS-1 cells.
HAP1 in the rat brain possesses two alternatively spliced isoforms, HAP1A and HAP1B, with molecular masses of 85 and 75 kDa, respectively (Li et al. 1995). Analysis of the isolated rat islets and INS-1 cells by Western blotting in the present study detected two HAP1 isoforms that have the same molecular sizes as those detected in rat hypothalamus. Thus, like rat brain tissue, islet β-cells express both HAP1A and HAP1B.
The secretory machinery of endocrine cells, including islet β-cells, shares many properties with that of the neurons. For example, both endocrine cells and neurons contain secretory vesicles, including synaptic microvesicles and large-dense core vesicles (LDCV) (Thomas-Reetz and De Camilli 1994; Regazzi et al. 1995). Synaptic vesicles are the predominant secretory vesicles in neurons, and the converse is true for the endocrine cells (Thomas-Reetz and De Camilli 1994). It is known that insulin is transported in pancreatic β-cells in LDCV, the secretory granules, and released from the β-cells by exocytosis. This excytosis is mediated by the action of the exocytotic machinery and finally results in the fusion of the secretory granule membrane with the plasma membrane, followed by release of the granule content, insulin, into the extracellular space to reach the blood circulation (Lang 1999). Similar to exocytosis in neurons, insulin release from granules is triggered by calcium influx through voltage-dependent channels (Zhang et al. 1998). In addition, β-cells also contain membrane receptors upon which other hormones and neurotransmitters act (Fridlyand et al. 2007; Smith 2009).
Although the precise function of HAP1 remains unclear, mounting evidence suggests that HAP1 might participate in intracellular trafficking in neurons, including transport of organelles, vesicles, or molecules, endocytosis of membrane receptors (Gutekunst et al. 1998; Li et al. 1998a,2002; Tang et al. 2003; Gauthier et al. 2004; Kittler et al. 2004; Li and Li 2005; McGuire et al. 2006; Rong et al. 2006,2007; Sheng et al. 2006). In light of the similarity of β-cells with neurons in intracellular trafficking, the expression of HAP1 in β-cells strongly suggests that HAP1 may play an important role in the trafficking of secretory granules, channel proteins or membrane receptors in β-cells.
Huntingtin is also expressed in rodent islets as well as in clonal β-cells (Björkqvist et al. 2005). Nuclear inclusions of mutant huntingtin, which may be associated with the pathology in HD patients and transgenic mice, exist in the islets with the predominance in β-cells of R6/2 mice, the first HD transgenic model (Björkqvist et al. 2005). Furthermore, exocytosis is abolished in β-cells but not in α-cells of R6/2 mice (Björkqvist et al. 2005). An enhanced and aberrant interaction between mutant huntingtin and β-tubulin has been found to disrupt insulin vesicular transport and further impair insulin secretion in β-cells (Smith et al. 2009), which may contribute to frequent development of diabetes in HD patients (Podolsky et al. 1972) and HD transgenic mice (Hurlbert et al. 1999; Jenkins et al. 2000). Kinesin and dynein, the molecular motors crucial for an effective microtubule-based transport, regulate sustained insulin release (Donelan et al. 2002; Varadi et al. 2003). Huntingtin interacts with these motor proteins and mediate axonal transport of vesicles or other molecules (Gunawardena et al. 2003; Gauthier et al. 2004; Trushina et al. 2004; Caviston et al. 2007). The association of huntingtin with molecular motors is dependent on the presence of HAP1, and the huntingtin-mediated vesicular transport requires HAP1 (Gauthier et al. 2004). It is therefore indicated that impairment of vesicular transport caused by mutant huntingtin may be contributed by the abnormal interaction of mutant huntingtin with HAP1 (Gunawardena et al. 2003; Gauthier et al. 2004; Trushina et al. 2004). In view of this, the abnormal interaction of mutant huntingtin with HAP1 in islet β-cells may be also responsible for the disruption of insulin vesicular transport and insulin secretion in HD-related diabetes.
Footnotes
Acknowledgements
This study was supported by grants 30225024 (to HL) and 30671106 (to ML) from the National Natural Science Foundation of China.
