Abstract
Our previous work demonstrated an important role of EpCAM in the regulation of pancreatic cell adhesion, growth and differentiation. Here we investigated the consequences of human EpCAM (hEpCAM) overexpression under the control of the MMTV-LTR promoter, known to drive robust gene expression in a number of ductal epithelia, including the pancreas. In this animal model (MMTV-hEpCAM) we uncovered a striking pancreatic phenotype exhibiting a 12-fold increase in the islet cell mass, with normal expression patterns of insulin and the transcription factor PDX-1. Intriguingly, these large islet clusters revealed an altered architectural organization of α- and δ-cells that appeared interspersed with β-cells in the islet cores. This suggests an effect of the hEpCAM transgene on the function of other cell adhesion molecules that we have previously shown to regulate islet cell type segregation. Consistent with this finding, we show that the pancreatic epithelium in MMTV-hEpCAM transgenic mice exhibits a redistribution of β-catenin, a known regulator of E-cadherin-mediated adhesions. Collectively, these results provide an important in vivo validation of hEpCAM signaling properties in normal epithelia and offer unique opportunities to further explore the function of this glycoprotein in select pancreatic cell lineages to elicit islet cell expansion, and/or regeneration in diabetes.
Introduction
EpCAM (Epithelial Cell Adhesion Molecule) (Litvinov et al. 1994a, 1994b)—also known as TACSTD1 (tumor-associated calcium signal transducer 1), CD326 (cluster of differentiation 326), and 17-1A antigen (Balzar et al. 1998, 1999; Schnell et al. 2013a)—was originally discovered in 1979 as a pan-carcinoma antigen in the study of colorectal carcinoma tumor markers (Herlyn et al. 1979; Koprowski et al. 1979). Since its discovery, EpCAM has been found to be upregulated in metastatic cancers of the breast, colon, prostate, ovary, lung, liver and pancreas (Spizzo et al. 2011; Schnell et al. 2013a; Fong et al. 2014), lending to its implication in epithelial cell growth (van der Gun et al. 2010). Interestingly, it has also been observed that EpCAM can be hyperglycosylated in transformed versus normal epithelial cells, pointing to complex regulatory mechanisms of its function in the maintenance and homeostasis of cell–cell interactions in a normal versus oncogenic capacity (Munz et al. 2008). More recently, an alternatively spliced form of EpCAM with deletion of exon 4 was found to be responsible for intestinal wall malformation and the disease termed Congenital Tufting Enteropathy (Sivagnanam et al. 2008; Schnell et al. 2013b), indicating that not only the expression levels but also select domains of EpCAM may impact on distinct cellular phenotypes and functions.
Although the vast majority of studies on EpCAM have focused on its association with tumor cell growth and progression, significant evidence points to fundamental functions of this glycoprotein in normal epithelia. Hence, we previously demonstrated that EpCAM mediates homophilic calcium-independent cell–cell adhesion in primary epithelial cells (Cirulli et al. 1998). In the case of developing epithelial organs, such as the pancreas, the levels of expression of EpCAM are always associated with events of cell proliferation and branching morphogenesis (Cirulli et al. 1998), a finding that suggests a direct implication of this protein in the events of epithelial morphogenesis. Accordingly, we and others have found that EpCAM modulates the adhesive function mediated by classical cadherin/β-catenin complexes through a signaling crosstalk that recruits PI3K, regulates interactions with elements of the cytoskeleton, and affects the stability and function of tight junctions (Winter et al. 2007; Guerra 2012; Wu et al. 2013). These are mechanisms that may be critically relevant for the development of proper tissue architecture and function.
More recent studies have provided additional evidence for multiple pro-growth mechanisms of EpCAM-mediated proliferation. For example, it was shown that a TACE-mediated proteolytic processing generates a short intracellular EpCAM domain (EpICD) that is able to form a complex with FHL2, β-catenin, and Lef-1 to induce gene transcription of oncogenes, such as C-myc and cyclins A/E (Maetzel et al. 2008; Denzel et al. 2009). Furthermore, EpCAM can promote sustained proliferation through a mechanism that counters TGF-β1–induced growth arrest and differentiation, and prolongs epithelial proliferative capability by fostering an epithelial-to-mesenchymal transition (Martowicz et al. 2013; Gao et al. 2015). In other cellular systems, EpCAM regulates epithelial cell motility, a function that is important for both normal tissue development and metastatic cell invasion (Sankpal et al. 2011; Gaiser et al. 2012).
Building on these morphoregulatory properties of EpCAM, we investigated the effects of the transgenic overexpression of EpCAM in the mouse pancreas. In this report, we describe the pancreatic phenotype of a transgenic mouse model expressing the human EpCAM under the control of the MMTV promoter (MMTV-hEpCAM), harboring the LTR hormone-responsive element, which greatly increases gene transcription in response to glucocorticoids, progestins, androgens, and dexamethasone (Cato et al. 1987; Ham et al. 1988; Truss et al. 1992; Richard-Foy and Hager 1987), and is known to drive robust gene expression in multiple ductal and secretory epithelia (Choi et al. 1987; Wagner et al. 2001; Moraes et al. 2007; Yang et al. 2010).
Our results provide the first evidence that the purposeful induction of elevated levels of hEpCAM expression in the pancreatic epithelium results in the development of a strikingly increased pancreatic islet cell mass. These observations have significant implications for future studies focusing on the manipulation of EpCAM expression and/or function, to promote either in vitro islet cell expansion to be used for transplantation or in vivo restoration of residual β-cell beta mass in diabetic patients.
Materials & Methods
Development of Transgenic Mice Expressing Human EpCAM
The Human EpCAM SmaI–BglII fragment of GA733-2 cDNA (kindly provided by Dr. A. Linnenbach; Szala et al. 1990) was subcloned into the steroid inducible MMTV LTR promoter region of the vector pJ5Ω (ATCC® 37721™), as shown in Figure 1A. MMTV LTR-hEpCAM mice were generated using the GA733-2 cDNA fragment of the pJ5Ω vector, digested using PvuII-Pst I to yield a 2.6-kb linear fragment. The fragment was recovered by electro-elution, extracted twice with phenol–chloroform and subsequently precipitated with ethanol. After reconstitution in T10E01 and dialyzed against TE at a final concentration of 5 µg/ml, this fragment was used for pronucleus injection of mouse eggs isolated from super-ovulated (C57Bl/LiARij x CBA/BrARij) F1 female mice mated to (C57Bl/LiARij x CBA/BrARij) F1 males, as described (Hogan et al. 1986). Injected oocytes were cultured in vitro overnight, and 2-cell stage embryos were implanted into one of the oviducts of 1-day pseudo-pregnant (C57Bl/LiARij x CBA.BRARij) F1 females and carried to term. Three weeks after birth, tail biopsies of the offspring were taken for DNA analysis. Total genomic DNA was prepared from tail biopsies. All experiments were performed on mice no older than 3 months. All procedures, including generation, housing and use of animal subjects were reviewed and approved by the Office of Animal Welfare at the Leiden University Medical Center (Leiden, The Netherlands), and by the Institutional Animal Care and Use Committee at University of Washington (Seattle, WA).
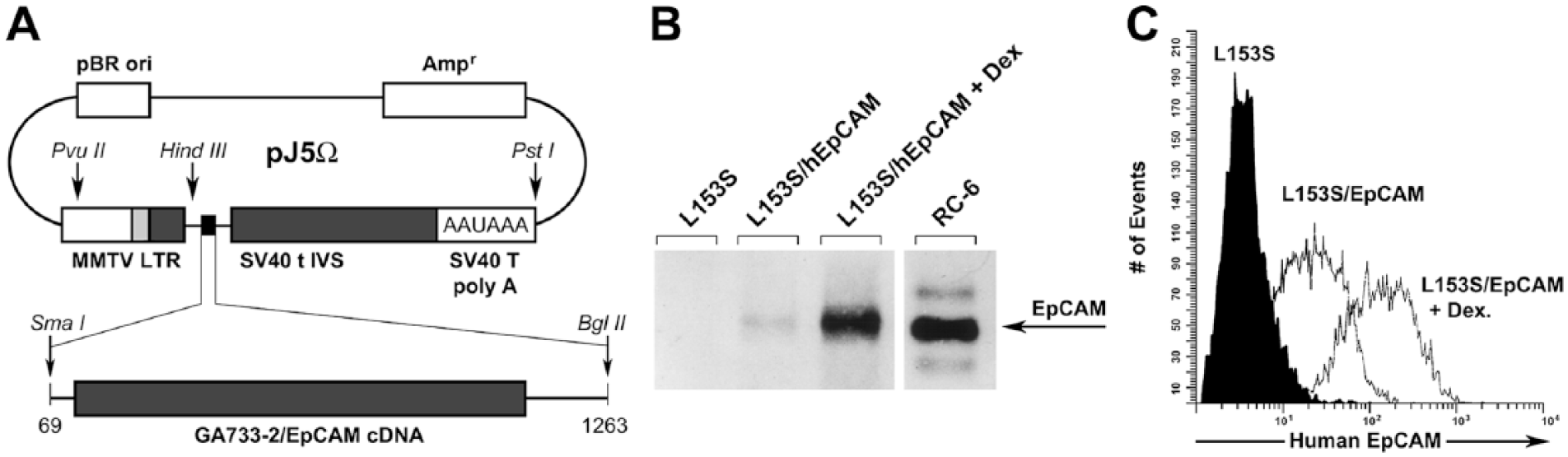
MMTV-hEpCAM construct and in vitro expression. (A) Map of vector pJ5Ω with MMTV LTR-human EpCAM cDNA insert used for transgenic mice production and in vitro experiments. (B) Western blotting of hEpCAM transfected in mouse mammary carcinoma cell line (L153S). Lane 1: Control L153S cells; lane 2: L153S cells transfected with the MMTV LTR-hEpCAM construct; lane 3: MMTV LTR-hEpCAM-transfected L153S cells after 24 hr induction with 100 µM dexamethasone in the media; lane 4: immortalized human mammary cell line RC-6 expressing endogenous hEpCAM. (C) FACS analysis of control L153S cells and MMTV LTR-hEpCAM transfectants showing cell-surface translocation of hEpCAM, before (L153S/hEpCAM) and after (L153S/hEpCAM + Dex) further induction by 100 μM dexamethasone.
Cell Lines
For select experiments, murine mammary carcinoma cells (L153S) (Schuuring et al. 1990) or human EpCAM-expressing carcinoma cell line RC-6 (American Type Culture Collection; Rockville, MD) were used. All cell lines were cultured in Dulbecco’s modified Eagle’s medium supplemented with 10% FCS, 100 U/ml penicillin and 100 U/ml streptomycin (all from Gibco/BRL; Breda, The Netherlands). For transfection, 2–5×105 cells were cultured in 35-mm Petri-dishes (Greiner B.V.; Alphen a/d Rijn, The Netherlands). Cells were then transfected using the DOTAP reagent (Boeringer Mannheim, Mannheim, Germany), according to the manufacturer’s protocol. The hEpCAM cDNA-containing plasmid (pJ5Ω) was mixed with the plasmid pSV2neo (Spivak et al. 1984) in a 10:1 ratio and used for each transfection. After 24 hr, the DOTAP/DNA-containing medium was replaced by fresh culture medium. The cells were cultured for an additional 48 hr, transferred to 2×100-mm Petri-dishes (Greiner B.V.), and further grown in the presence of 300 µg/ml of G418 (Gibco-BRL). After 14–18 days, G418-resistant clones were isolated, and flow cytometry was used to detect the expression of human EpCAM on the cell surface using the anti-EpCAM mAb 323/A3. EpCAM positive clones were pooled and used in subsequent experiments.
Antibodies and Immunohistochemistry
The mAbs 323/A3 (Edwards et al. 1986), 17-1A (Herlyn et al. 1979; Koprowski et al. 1979), GA733 (Herlyn et al. 1991), and K931 (De Leij et al. 1994), which are reactive with human EpCAM, were provided by Centocor Inc. (Malvern, PA). The EpCAM-directed mAb MOC31 was kindly supplied by Dr. L. de Leij (University of Groningen, Groningen, The Netherlands). The mAb KS1/4 (Bumol et al. 1988; Perez and Walker 1989; Schön et al. 1993), reacting with the extracellular domains of EpCAM, was a gift from Dr. Ralph Reisfeld (The Scripps Research Institute, La Jolla, CA). The mAb directed against murine EpCAM, G8.8 (Nelson et al. 1996), was kindly provided by Dr. Andy Farr (University of Washington, Seattle, WA). The other antibodies used were rabbit anti-glucagon (Cell Signaling Technology; Beverly, MA), guinea pig anti-insulin (Dako; Glostrup Denmark), goat anti-PDX1 (Beta Cell Biology Consortium; Nashville, TN), mouse anti-E-cadherin (36/Ecadherin; BD Biosciences, Franklin Lakes, NJ), rabbit anti-somatostatin (Dako), mouse anti-β-catenin (14/Beta-Catenin; BD Biosciences). Fluorophore-conjugated secondary antibodies were from Jackson Immunoresearch Laboratories (West Grove, PA).
Flow Cytometry
Determination of human EpCAM translocation to the cell surface of transfected L153S cells was performed using the mAb KS1/4, followed by detection with FITC-conjugated goat-anti-mouse IgG (Southern Biotechnology; Birmingham, AL), as described elsewhere (Cirulli et al. 1998).
Dexamethasone Induction of MMTV hEpCAM
For dexamethasone induction of cell lines, 1 µM dexamethasone was added to the culture medium for 24 hr. Cells were then analyzed by flow cytometry or western blotting. Although MMTV-hEpCAM transgenic mice expressed significant levels of the human EpCAM under basal conditions, possibly due to the action of mouse endogenous glucocorticoids, progestins, and androgens (Cato et al. 1987; Ham et al. 1988; Truss et al. 1992; Richard-Foy and Hager 1987), we treated mice with dexamethasone (1 µg per gram body weight) that acts on the LTR hormone-responsive element of the MMTV promoter and further increases in vivo transcription of any transgene downstream of this promoter, as previously described (Sadikot et al. 2001). The expressed human EpCAM transgenic protein was immunoprecipitated using human-specific mAb 323/A3.
Immunoprecipitation and Immunoblotting
Transfected cell lines were seeded at equal density on 100-mm Petri-dishes 48 hr before lysis, and cultured for the last 24 hr in the presence or absence of dexamethasone (1 µg/ml) in the medium. Cells grown to 70% to 90% density, or tissue isolates from either wild type or transgenic mice, were lysed in a buffer containing 0.5% Triton X-100, 50 mM Tris pH 7.4, 100 mM NaCl, 1 mM CaCl
Immunohistochemistry, Immunofluorescence and Morphometric Analysis
Sections from paraffin blocks of 4% paraformaldehyde-fixed adult pancreas of three-month-old wild type or MMTV-hEpCAM transgenic mice were used for immunohistochemistry and immunofluorescence staining. Slides were deparaffinized in 2×5 min incubation in Citrisolv and then rehydrated with 2×5 min in 100% ethanol, and then 1 min each in 95%, 80%, 70% ethanol followed by 2 min in ddH2O. The slides were then transferred to 1×PBS for 1 min, and then incubated for 30 min in citrate buffer (0.1M sodium citrate, 0.05% Tween-20, pH 6.0) that had been preheated to 90°C. Tissue sections on slides were then outlined with a PAP pen, and incubated with PBS–50 mM glycine for 15 min to quench residual formaldehyde-generated reactive groups, and then blocked in PBS containing 1% BSA and 5% donkey serum for 1 hr at room temperature. Sections were then incubated overnight at 4°C with primary antibodies at the following dilutions: rabbit anti-glucagon, 1:200; guinea pig anti-insulin, 1:400; rabbit anti-somatostatin, 1:100; goat anti-PDX-1, 1:400; mouse anti-E-Cadherin, 1:100; mouse anti-β-catenin, 1:50 (see antibodies section). Sections were washed with PBS containing 0.1% BSA, 0.05% Tween-20, and then incubated for 45 min at room temperature with fluorophore-conjugated secondary antibodies, all diluted 1:200. Slides were then washed and mounted with VectashieldTM (Vector Laboratories; Burlingame, CA) for imaging and analysis on a Nikon Eclipse 90i fluorescence microscope (Nikon; Tokyo, Japan) connected to a Photometrics CoolSNAP HQ2 CCD camera using the NIS-elements AR software for image acquisition and morphometric analysis.
For quantitative assessment of pancreatic islet size and islet cell type distribution, sections from four wild type and four MMTV LTR-hEpCAM pancreata were analyzed, with a minimum of 35 sections/animal collected at 200-µm intervals. To assess the architectural arrangement of islet cell types within pancreatic islets, we counted the number of α- and δ-cells located in the “outer third” and “inner two thirds” of the hormone-positive area in each islet cluster, a morphometric approach that we previously reported to accurately define normal regionalization of islet cell types within islet clusters (Cirulli et al. 1994).
Results
Expression of MMTV-driven hEpCAM in Murine Cell Lines
To investigate whether the construct containing the hEpCAM cDNA was expressed in murine cells, and whether the expressed protein would be immunologically similar to native human EpCAM, L153S murine carcinoma cells were transfected with the pJ5Ω/EpCAM construct, in which hEpCAM cDNA is under the control of the MMTV-LTR. Figure 1A depicts the construct used to generate MMTV promoter-driven human EpCAM transgene, harboring the LTR hormone-responsive element that greatly increases gene transcription in response to glucocorticoids, progestins, androgens, and dexamethasone (Cato et al. 1987; Ham et al. 1988; Truss et al. 1992; Richard-Foy and Hager 1987). Western blotting analysis of stable transfected lines revealed hEpCAM-specific immunoreactivity of a molecular mass similar to that detected in human carcinoma RC-6 cells expressing native hEpCAM (Fig. 1B). The slight difference in mobility of the transgenic hEpCAM expressed in murine cells (as compared to EpCAM expressed by human carcinoma RC-6 cells) is likely due to slightly different glycosylation of the molecule, since inhibition of glycosylation resulted in an identical molecular masses (data not shown). Treatment of L153S/hEpCAM cells with dexamethasone (Dex) caused a significant increase in hEpCAM expression (Fig.1B; L153S/EpCAM + Dex), thus demonstrating the inducibility of the transgene in these transfectants. The human RC-6 cell line was used as a positive control for hEpCAM immunoreactivity (Fig.1B; RC-6). Analysis of the same transfectants by FACS demonstrated cell-surface translocation of the transfected hEpCAM (Fig. 1C). Induction with dexamethasone significantly increased translocation of hEpCAM to the cell surface, further validating the integrity and functionality of the hEpCAM transfection construct (Fig. 1C). Collectively, these results demonstrate the integrity and functionality of our construct MMTV-driven hEpCAM.
In Vivo Demonstration of Transgenic hEpCAM Expression
Following our in vitro validation studies, we generated transgenic mice to study the functional impact of hEpCAM overexpression in various epithelial tissues. Following pronucleus injection of the Pvu II and Pst I fragment from the pJ5Ω/hEpCAM construct (Fig. 1A) into fertilized mouse oocytes, and their subsequent transplantation into pseudo-pregnant female mice, we were able to identify seven transgenic founder mice (9/1, 9/4, 9/5, 9/6, 9/7, 9/8, and 9/9). DNA samples from these founder mice analyzed by Southern blotting revealed that three transgenic strains (9/1, 9/6, and 9/3) contained intact copies of the MMTV-hEpCAM construct that were successfully transmitted to germ line, and expressed the transgene in their tissues. However, the progeny of transgenic line 9/1 harbored the highest copy number of the transgene concatamer (Fig. 2A). Northern blot analysis of RNA isolated from different epithelial tissues from the various MMTV-hEpCAM transgenics revealed that line 9/1 exhibited the highest levels of the transgene expression in the pancreas (Fig. 2B), with detectable, albeit lower, levels in other epithelial tissues, such as the mammary gland, liver, and kidney. This expression profile was validated at the protein level by western blotting performed on total protein isolates prepared from various epithelia, and immunoprecipitated with the human-specific anti-EpCAM 323/A3 mAb (Fig. 2C). These results demonstrate high levels of expression of the hEpCAM transgene in the pancreas, and lower levels in other epithelia (Fig. 2C). Based on these results, we focused our analysis on the 9/1 transgenic line, henceforth referred to as MMTV-hEpCAM transgenics, to study the effects of hEpCAM overexpression in the pancreas. For these morphometric studies, pancreatic tissue was isolated before and after dexamethasone induction. As shown in Figure 2D, indirect immune-peroxidase staining using the human-specific anti-EpCAM KS1/4 mAb revealed a mosaic distribution of the hEpCAM transgene, with the most prominent immunoreactivity detected in ductal structures and acinar tissue (Fig. 2D). Induction of the transgene with dexamethasone resulted in a significant increase in hEpCAM-specific immunoreactivity throughout the pancreatic epithelium (Fig. 2E), thus providing direct evidence for substantial expression of hEpCAM in situ, both under steady-state and after dexamethasone treatment. To simultaneously detect endogenous murine EpCAM and the human hEpCAM transgene, pancreatic sections from 12-week-old MMTV-hEpCAM mice and control wild type (WT) littermates were stained by double immunofluorescence using the mouse-specific mAb G8.8 and the human-specific mAb 323/A3. These studies revealed that in, MMTV-hEpCAM mice (Fig. 2I–2K), the expression of the hEpCAM transgene appears homogeneously expressed throughout the exocrine compartment, both acinar (Fig. 2J–2K) and ductal (data not shown), and is virtually undetectable in islet cells (Fig. 2J, circled area), suggesting that the MMTV promoter is no longer active in endocrine cells once they have differentiated from ductal progenitors during embryonic life (data not shown). No specific immunoreactivity for the human hEpCAM transgene was detected in WT tissue (Fig. 2G), further validating the specificity of mAb 323/A3 for human EpCAM. Conversely, immunoreactivity for the endogenous murine EpCAM was readily detectable in islet cells and, at lower levels, in acinar cells, in both MMTV-hEpCAM (Fig. 2I, circled area) and age-matched WT littermate pancreata (Fig. 2F, circled area).
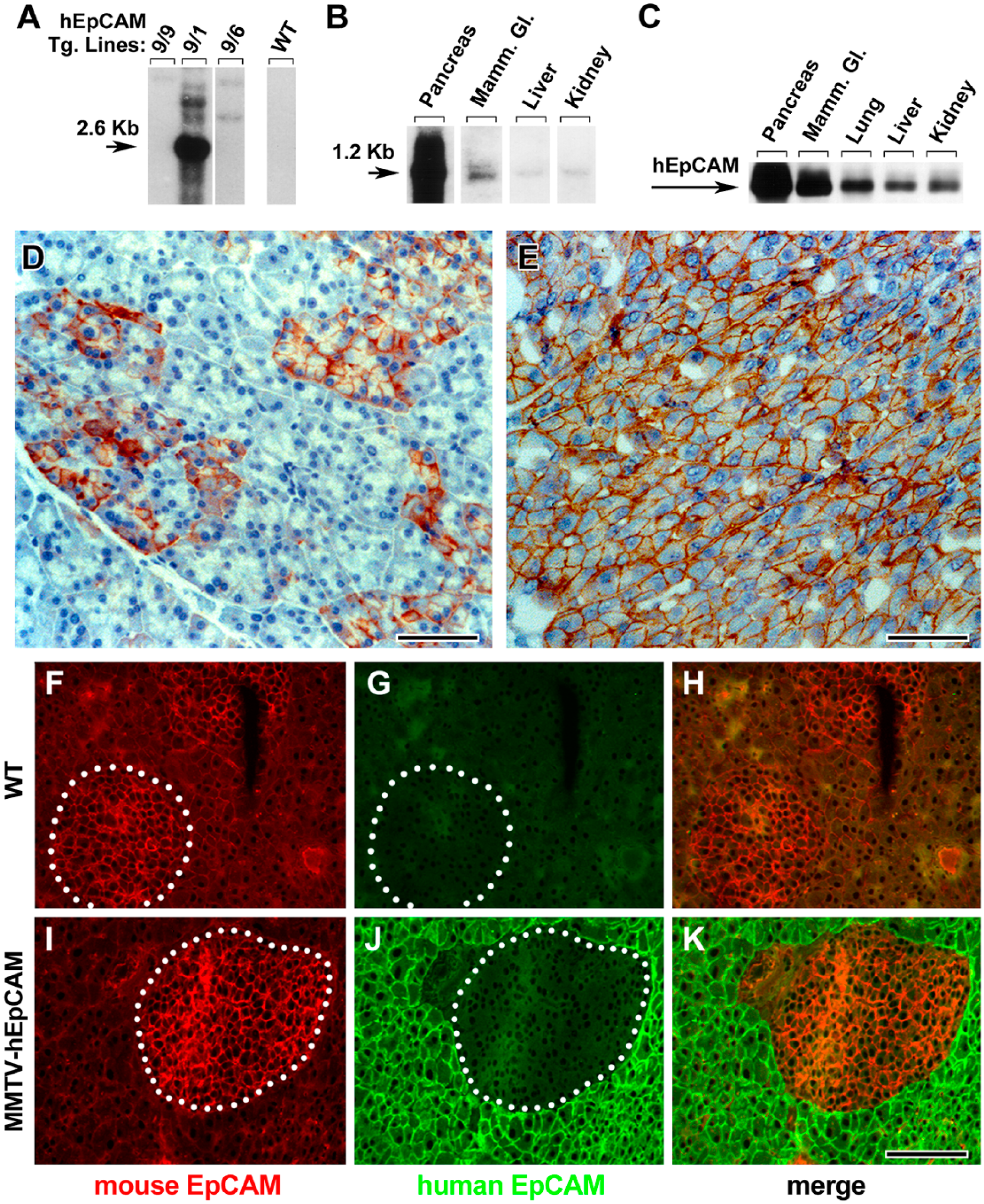
Expression profile of the hEpCAM transgene in vivo. (A) Southern blot analysis of transgenic MMTV LTR-hEpCAM strains of transgenic mice, with line 9/1 exhibiting the highest copy number of the transgene concatamer. (B) Northern blot performed on RNA isolated from selected epithelial tissues from the transgenic line 9/1 showing the highest levels of hEpCAM transcripts in the pancreas. (C) Western blotting for hEpCAM performed on immunoprecipitates from whole tissue lysates of various epithelial tissues after induction by dexamethasone. (D, E) Identification of hEpCAM-specific immunoreactivity in cryosections from the pancreas of MMTV-hEpCAM transgenic mice before (D) and after (E) dexamethasone induction. Wild type (F–H) and MMTV-hEpCAM transgenic pancreas (I–K) stained with double immunofluorescence for rat mAb G8.8, which is specific for the murine EpCAM (F, I; red), and mouse mAb 323/A3, which is specific for human EpCAM (G, J; green). Circled areas in F, G, I and J identify islet tissue. Scale (D) 50 µm; € 40 µm; (K) 100 µm.
Overexpression of hEpCAM in the Pancreatic Epithelium Results in Significantly Enlarged Islets of Langerhans
To maximize the downstream effects of hEpCAM over-expression in the pancreas, we reasoned that, although significant expression of the transgene was detected in MMTV-hEpCAM mice, induction by dexamethasone would have further enhanced its expression, and possibly have an impact on the morphology of the pancreas. Hence, weaning-age, 3-week-old MMTV-hEpCAM transgenic mice were treated with a single dose of dexamethasone (1µg per gram body weight) and reared to adulthood for morphometric analysis. At 12 weeks of age, these MMTV-hEpCAM mice exhibited a remarkable pancreatic phenotype, with a significant 5-fold increase in overall organ size as compared with WT littermates (data not shown). Interestingly, immunostaining for insulin identified a striking increase in insulin-positive areas (Fig. 3A, 3B), with some islet clusters in the MMTV-hEpCAM pancreata occupying entire pancreatic lobuli (Fig. 3D), as compared with WT controls (Fig. 3C). Extensive morphometric analysis performed at 200-µm intervals throughout the pancreas revealed close to a 12-fold increase of the overall insulin positivity in MMTV-hEpCAM mice, relative to control mice (Fig. 3E). Monitoring of these mice over 1.5 years yielded no evidence of increased incidence of tumors in any of the epithelial tissues in which the MMTV promoter has been previously shown to be active (Choi et al. 1987; Wagner et al. 2001; Moraes et al. 2007; Yang et al. 2010). Additionally, measurements of blood glucose levels in the MMTV-hEpCAM transgenic mice revealed that they remained euglycemic, with no signs or symptoms of hypo- or hyperglycemia (data not shown). Consistent with our previous work demonstrating that upregulation of EpCAM is always associated with pancreatic progenitors and islet cell growth (Cirulli et al. 1998), the results obtained with the MMTV-hEpCAM transgenic mice indicate that the purposeful overexpression of this cell adhesion molecule may have a positive regulatory function on the establishment of an enlarged islet cell mass.
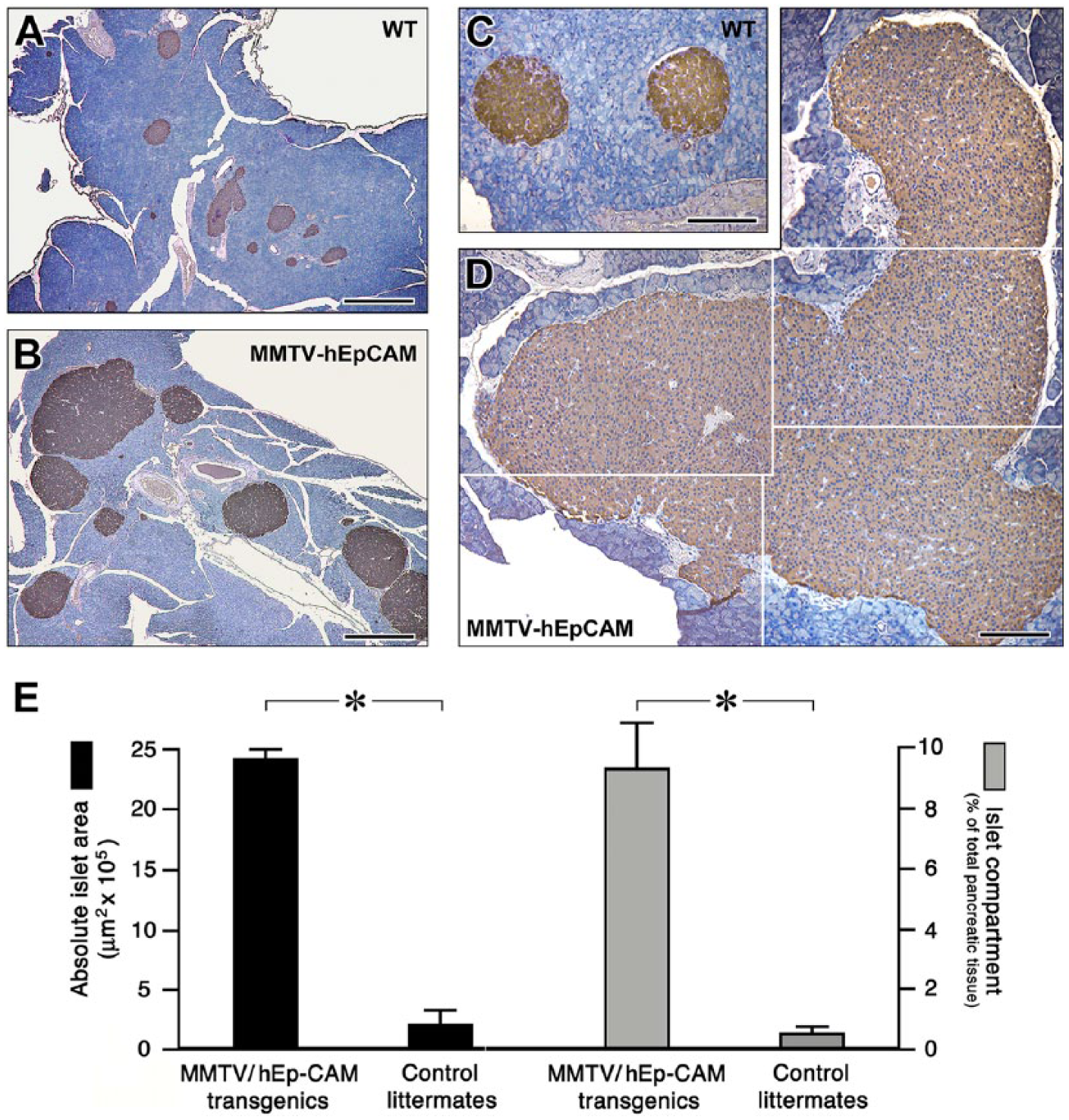
Pancreatic phenotype of MMTV-hEpCAM transgenic mice. (A, B) Representative low magnification images of wild type (A) and MMTV LTR-hEpCAM transgenic pancreas (B) stained for insulin by immunoperoxidase (brown). Note the larger area of insulin-positive cells in hEpCAM transgenics. (C, D) Higher magnification images acquired from wild type (C) and MMTV LTR-hEpCAM transgenic pancreata (D) displaying an example of extremely large islet tissue. (E) Morphometric analysis of wild type and MMTV LTR-hEpCAM transgenic pancreata, expressed as absolute islet area (left, y-axis) and % of total pancreatic tissue (right, y-axis). Data were compiled from sections collected from four wild type and four MMTV LTR-hEpCAM pancreata. A minimum of 35 sections/animal were collected at 200-µm intervals for staining and analysis. *p<0.001. Scale (A, B) 600 µm; (C, D) 200 µm.
Islet Cell Type Distribution Is altered in MMTV-hEpCAM transgenic mice
In rodents, pancreatic islet cell types have been shown to exhibit a highly conserved architectural organization, with β-cells located in the core and the non-β-cells (α-, δ- and pp-) predominantly arranged at the periphery (Orci and Unger 1982; Halban et al. 1987). To determine if hEpCAM overexpression in our model affected islet architecture, pancreatic sections from MMTV-hEpCAM transgenic mice and wild type littermates were analyzed by multiple immunofluorescence for insulin, glucagon and somatostatin. These experiments revealed that the typical islet architecture of wild type mice (Fig. 4A) is altered in the large islet clusters identified in the MMTV-hEpCAM transgenic pancreata, with α- and δ-cells present in significant numbers throughout the islet area (Fig. 4B). To validate this qualitative assessment in both MMTV-hEpCAM transgenics and control littermates, we counted the number of α- and δ-cells located in the “outer third” and “inner two thirds” of the hormone-positive area in each islet cluster, a morphometric approach that we previously reported for the characterization of islet cell type architectural organization (Cirulli et al. 1994), and compiled the results for each islet cell type as percentage of their respective regional localization. The results of this analysis revealed that, in wild type pancreata, 92.1% of the α-cells were restricted to the outer third of the overall islet area, with only 7.9% of cells found in the inner islet area. However, in MMTV-hEpCAM transgenics, only 64.8% of the α-cells were found to occupy this outer region, with up to 35.2% identified in the inner regions of the islet area (Fig. 4C). A similar distribution was also observed for δ-cells, with up to 41% found in the inner region of MMTV-hEpCAM islets as compared with only 13% recorded in wild type mice (Fig. 4C). Interestingly, we also observed that the absolute number of α-cells and δ-cells in MMTV-hEpCAM islets was increased by 7.1- and 4.2-fold, respectively (n=31 islets scored from four MMTV-hEpCAM transgenics; n=22 islets scored from three WT mice). At present, we do not have a mechanistic explanation for the effects of the hEpCAM transgene on the distribution and compartmentalization of islet cell type within islet clusters. Based on our previous studies on the role of select cell adhesion molecules on the architectural organization of islet cell types (Cirulli et al. 1993; 1994), it is possible that the overexpression of hEpCAM alters the balance of cell–cell interactions, thus resulting in a defective cell type segregation.
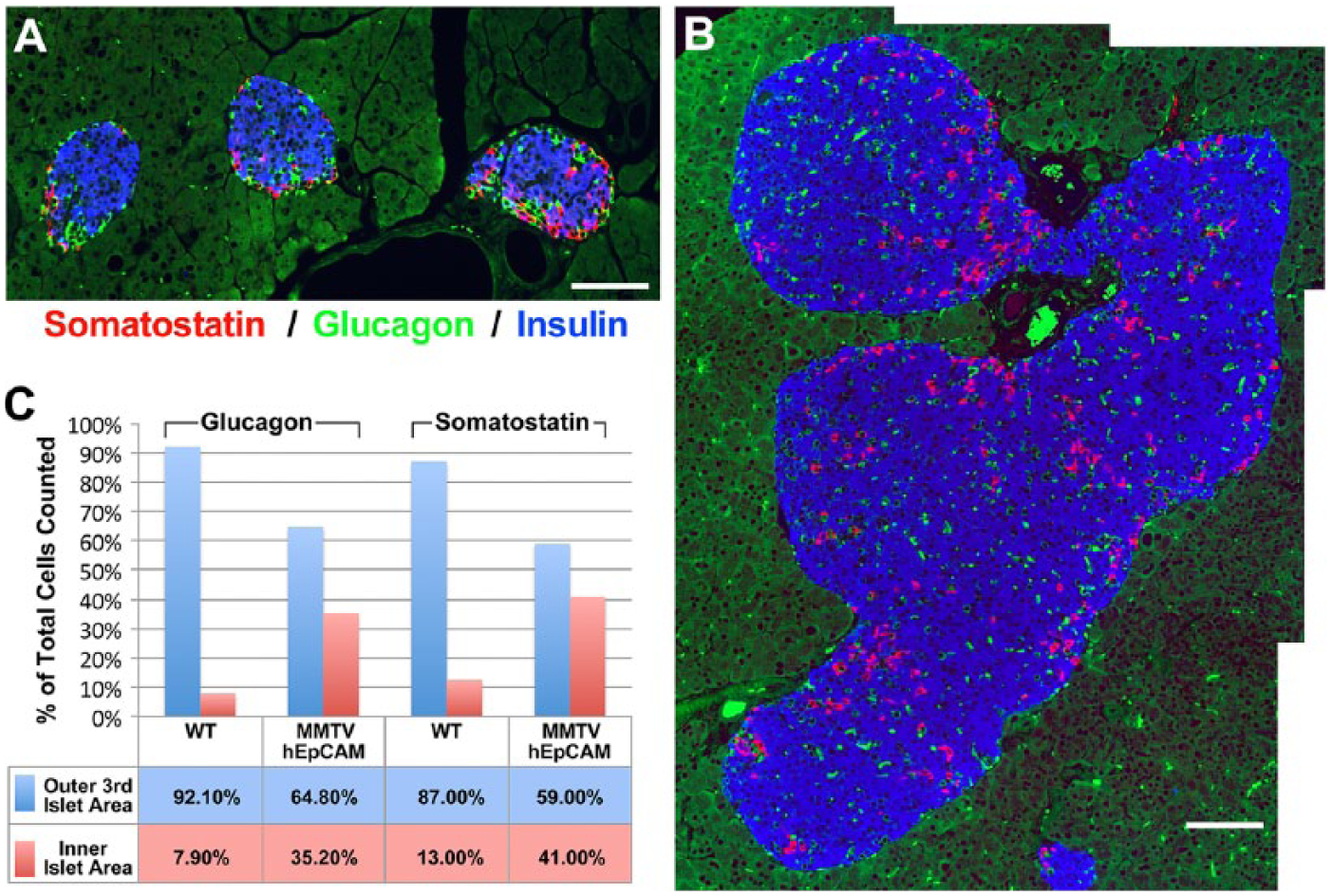
Islet cell type architectural organization is altered in MMTV-hEpCAM transgenic mice. (A) Wild type pancreatic islets identified by immunofluorescence for somatostatin (red), glucagon (green), and insulin (blue), displaying the typical cell type distribution with β-cells (blue) in the core and most of the α-cells (green) and δ-cells (red) located at the periphery. (B) Pancreatic islets in the pancreas of MMTV-hEpCAM transgenics show an altered cell type distribution, with significant numbers of α-cells and δ-cells found throughout the islet clusters. (C) Quantification of α- and δ-cell distribution in the peripheral outer third (blue bars) and inner islet area (red bars) in wild type and MMTV-hEpCAM transgenics. Note the significant loss of islet cell type segregation in the MMTV-hEpCAM transgenic islets. Scale, 150 µm.
Overexpression of hEpCAM Does Not Alter PDX-1 Expression in Pancreatic Islets
Based on the results presented above, although insulin expression appears to be unaffected in transgenic MMTV-hEpCAM, we nonetheless investigated the expression profile of PDX-1, a transcription factor well known to regulate the development, differentiation and function of pancreatic β-cells. For these immunolocalization experiments, pancreatic sections from wild type and MMTV-hEpCAM mice, immunostained for insulin and PDX-1, revealed that the expression of PDX-1 transcription factor is readily detected in β-cells of MMTV-hEpCAM mice (Fig. 5B, 5C), in a pattern that appears qualitatively indistinguishable from that observed in wild type islets (Fig. 5A). This can be easily appreciated in single channel images showing the typical nuclear-restricted localization of PDX-1-specific immunoreactivity in wild type (Fig. 5A) and MMTV-hEpCAM islets (Fig. 5C). These results suggest that hEpCAM overexpression has no negative effect on the differentiation of β-cells.
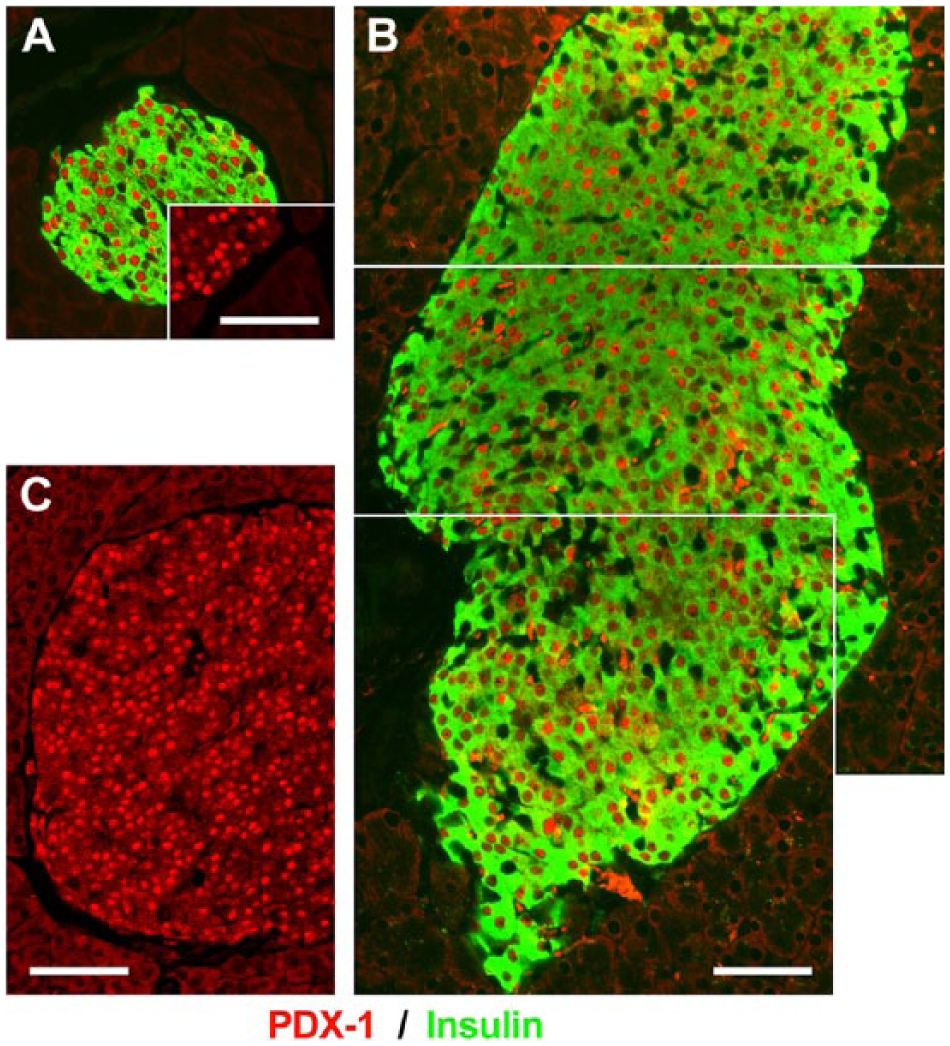
Normal expression profile of PDX-1 in MMTV-hEpCAM pancreata. Immunofluorescence for PDX-1 (red) and insulin (green) in wild type (A) and MMTV-hEpCAM pancreata (B–C). A typical nuclear restricted localization of PDX-1 can be readily appreciated in both wild type (A, red channel inset) and MMTV-hEpCAM islets (B and C, red). Scale, 150 µm.
Overexpression of hEpCAM in MMTV-hEpCAM Mice Causes Redistribution of β-Catenin
Based on our previous in vitro studies demonstrating that EpCAM can negatively regulate cadherin-mediated cell–cell interactions through redistribution of the pool of cadherin-associated catenins and recruitment of the PI3K (Winter et al. 2007), we investigated the possible effects of hEpCAM overexpression in MMTV-hEpCAM mice on the cellular localization of β-catenin, a crucial regulator of cadherin-mediated adhesions (Huber et al. 1996). As expected, wild type pancreata revealed that β-catenin expression is readily detected in all epithelial cells, both endocrine and exocrine, with a prominent fluorescence signal restricted to cell-cell boundaries (Fig. 6A, 6B). In contrast, in MMTV-hEpCAM pancreata, β-catenin-specific immunoreactivity—although retained in most exocrine cells—appeared significantly reduced in islet cells (Fig. 6C, 6D). Interestingly, both β-catenin and E-cadherin-specific immunoreactivities appeared to be redistributed from the cell surface to the cytosol in MMTV-hEpCAM pancreata, with a spotted pattern best visible in the single red channel (Fig. 6, compare panels B and D, and panels F and H). This in vivo expression pattern is in agreement with our previous in vitro studies showing a redistribution of the β-catenin pool in hEpCAM-transfected cell lines (Balzar et al. 1998; Winter et al. 2007), and supports a direct role for hEpCAM in the remodeling of cell–cell adhesion domains that promote downstream Wnt/β-catenin nuclear translocation and signaling, as recently shown in vitro by Maetzel and colleagues with FHL2 and Lef-1 (Gostner et al. 2001; Maetzel et al. 2009).
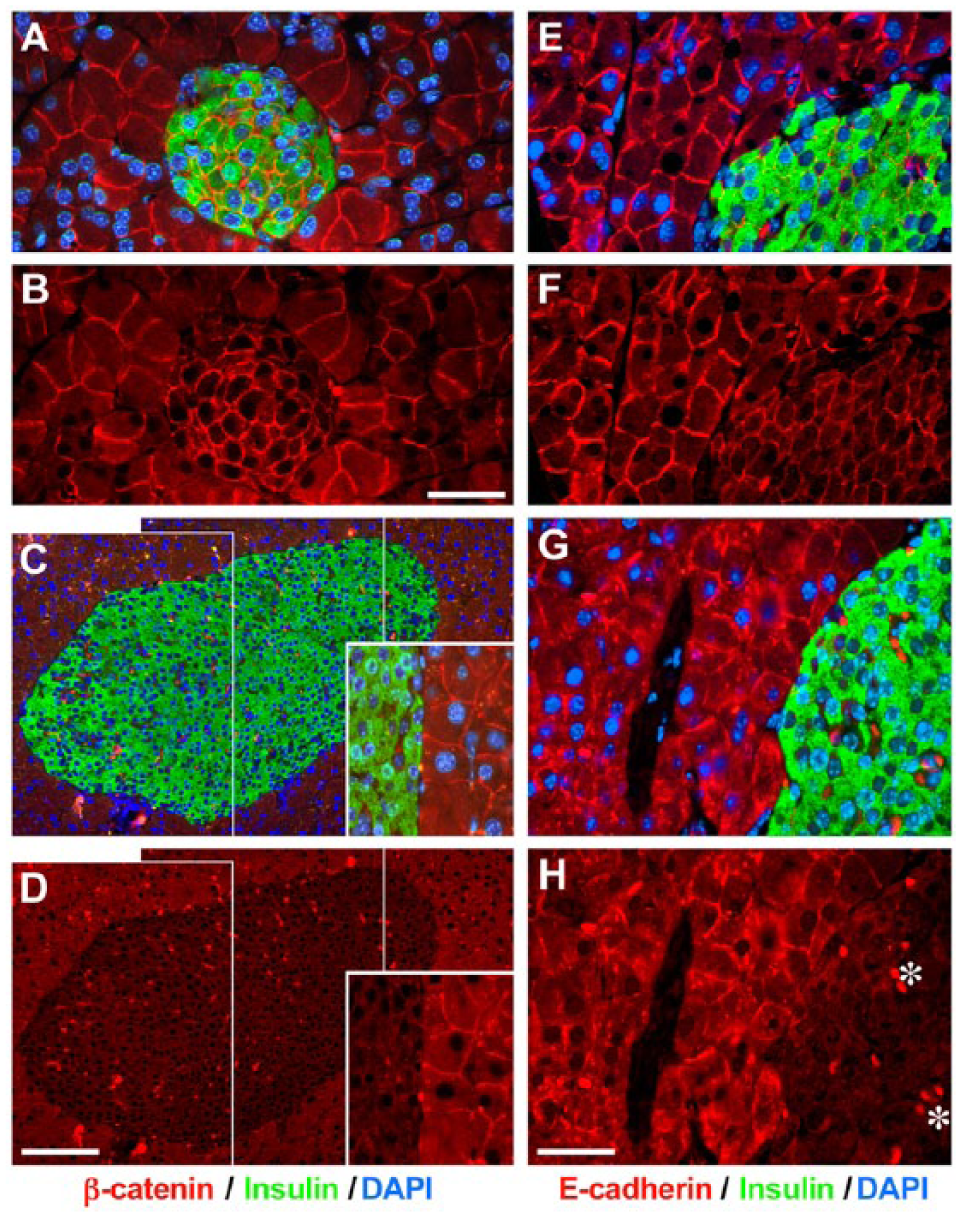
Altered β-catenin and E-cadherin expression pattern in MMTV-hEpCAM transgenic pancreas. Immunofluorescence staining for β-catenin (red) and insulin (green) in wild type (A, B) and MMTV-hEpCAM pancreas (C, D). Typical β-catenin-specific immunoreactivity restricted to cell–cell contacts can be appreciated in wild type tissue, with prominent staining in β-cells easily identifiable in the red channel (B). This expression pattern is altered in MMTV-hEpCAM pancreata, which display a significant reduction in β-catenin staining intensity in islet cells (C, D), and its loss of confinement to the cell–cell boundaries, with spotted redistribution to intracellular regions (C and D, insets). Immunoreactivity specific for E-cadherin is readily detected at the cell–cell boundaries in acinar and islet cells in wild type pancreas (E, F). In contrast, MMTV-hEpCAM pancreata exhibit a significant redistribution of E-cadherin from cell–cell contacts to the cytosol (G, H), revealing a pattern similar to that observed for β-catenin-specific immunoreactivity. Occasional strong red spots located in vascular spaces are due to the autofluorescence of red blood cells (examples marked in H by *). Scale (B) 50 µm; (D) 150 µm; (H) 50 µm.
Discussion
The CD326 antigen, formerly referred to as KSA antigen (Varki et al. 1984; Edwards et al. 1986; Spurr et al. 1986; Bumol et al. 1988) and later renamed EpCAM (Epithelial Cell Adhesion Molecule) due to its function in regulating cell–cell interactions (Litvinov et al. 1994a; 1994b; 1997), is a glycoprotein that has been shown to regulate cell proliferation in both normal and transformed epithelia (Cirulli et al. 1998; Balzar et al. 1999; Schnell et al. 2013a). Because of these multiple functions, EpCAM has been receiving significant attention for both diagnostic and therapeutic purposes (van der Gun et al. 2010; Schnell et al. 2013a).
Building on our previous work on the function of EpCAM in epithelial cells (Litvinov et al. 1994a, 1994b, 1997; Cirulli et al. 1998; Balzar et al. 1999; Winter et al. 2007), we have generated a transgenic mouse model that allows for the overexpression of human EpCAM under the control of the MMTV-LTR promoter, known to drive robust gene expression in multiple ductal and secretory epithelia (Choi et al. 1987; Wagner et al. 2001; Moraes et al. 2007; Yang et al. 2010). Owing to the notion that the pancreatic epithelium develops from a multipotent ductal primordium, giving rise to all epithelial lineages (ductal, acinar, and endocrine) (Pan and Wright 2011), this transgenic model (MMTV-hEpCAM) allowed us to investigate the effects of hEpCAM overexpression in the pancreatic epithelium. Intriguingly, although significant hEpCAM transgene expression is observed throughout the acinar and ductal compartments of the pancreas of transgenic mice induced with dexamethasone at weaning age, the enlarged pancreatic islets do not express hEpCAM in adult (12-week-old) MMTV-hEpCAM transgenics (Fig. 2J). At present, although we do not know at which stage of development the effects of the transgene impacts on epithelial cell expansion and results in the generation of these strikingly large islet clusters, it is reasonable to speculate that the increase in islet cell size observed in adult transgenics may result from a hEpCAM-mediated expansion of ductal progenitors, where the MMTV promoter is known to be most active (Choi et al. 1987; Wagner et al. 2001; Moraes et al. 2007; Yang et al. 2010). This possible interpretation is further supported by the notion that the MMTV promoter can drive strong gene expression in ductal epithelia during embryonic life as early as E13.5 (Sakamoto et al. 2012). Consequently, it is possible that, once the endocrine differentiation program is initiated in embryonic islet progenitors, the MMTV promoter activity declines and the expression of the hEpCAM transgene is turned off. This, in turn would also allow for proper endocrine differentiation to occur, as demonstrated by the unaltered expression pattern of PDX-1 in islet β-cells (Fig. 5). Interestingly, despite the significant increase in islet cell mass, these MMTV-hEpCAM transgenics remained normoglycemic (data not shown). Based on the recent demonstration that E-cadherin positively regulates insulin secretion (Parnaud et al. 2015), our observation that the expression pattern of both β-catenin and E-cadherin is altered in islets from MMTV-hEpCAM transgenics (Figure 6C, 6D, 6G, 6H) points to a possible defect in insulin secretion in these mice. However, due to the enlarged total islet mass, insulin secretion may still be present at sufficient levels to ensure euglycemia. Furthermore, the relative increase in α-cell numbers in the islets of MMTV-hEpCAM transgenics may also contribute to counter-regulatory mechanisms of glucose homeostasis. Future studies will need to be designed to fully investigate the metabolic phenotype of this animal model.
Recent evidence demonstrates that EpCAM can be cleaved by PS-2 and TACE, leading to the generation of a short intracellular fragment (EpICD) that, in a complex with FHL2 and β-catenin, translocates to the nucleus to interact with Lef-1 and activates target genes such as c-myc (Maetzel et al. 2009; Chaves-Pérez et al. 2013). This proteolytic process also generates a large extracellular domain (EpEx) that can further activate homophilic EpCAM–EpCAM interactions. Thus, it has been shown that EpEx provides neighboring cells with a paracrine signal for further cleavage that amplifies short-range EpCAM-mediated pro-growth signaling (Denzel et al. 2009). Based on these observations, it is possible that, despite the lack of hEpCAM transgene expression in mature islet cells, adjacent acinar and/or ductal cells expressing the transgene may release significant levels of the cleaved extracellular (EpEx) domain, which, in turn, acts on the endogenous mouse EpCAM (highly homologous to hEpCAM) expressed by islet cells (data not shown). Consistent with the demonstration that hEpCAM overexpression causes β-catenin redistribution from sites of cell–cell adhesion to intracellular compartments to regulate signaling (Litvinov et al. 1997; Balzar et al. 1999; Winter et al. 2007; Maetzel et al. 2009; Gostner et al. 2011; Chaves-Pérez et al. 2013), our observations of the altered β-catenin expression pattern in both acinar cells and islet cells (Fig. 6) provides indirect evidence for a possible paracrine signaling on the islet tissue from nearby acinar or ductal cells. This is a possibility that we are actively investigating using a recombinant EpEx domain of hEpCAM (Vercollone and Cirulli, unpublished).
An additional intriguing finding in these enlarged MMTV-hEpCAM transgenic islets is that the architectural organization of islet cell types exhibit an altered distribution, with α-cells and δ-cells invading the islet core that is normally occupied primarily by β-cells (Fig. 4). We interpret this disrupted cell type segregation as possibly due to the reduced β-catenin expression and redistribution in islet cells (Fig. 6). Thus, as a result of EpCAM signaling on E-cadherin-mediated cell–cell adhesion, whether direct or indirect from adjacent acinar cells expressing the hEpCAM transgene, it is possible that the strength of E-cadherin-mediated cell–cell adhesion between islet cell types is significantly decreased (Litvinov et al. 1997; Balzar et al. 1999; Winter et al. 2007; Maetzel et al. 2009; Gostner et al. 2011; Chaves-Pérez et al. 2013). This, in turn, obliterates differences in calcium-dependent and calcium-independent adhesive properties of distinct islet cell types that we and others have previously demonstrated to function as critical driving forces for islet cell type segregation within islets (Rouiller et al. 1990; 1991; Cirulli et al. 1993; 1994; Esni et al. 1999).
In conclusion, we report a striking pancreatic phenotype induced by the in vivo overexpression of hEpCAM under the control of the MMTV-LTR promoter. Although this promoter is not specific for pancreatic cell lineages, its predominant activity in ductal epithelia allowed for a robust transgene expression in pancreatic ductal cells. Based on the notion that ductal domains comprise putative islet progenitors (Lynn et al. 2007; Seymour et al. 2007), the resulting phenotype in our MMTV-hEpCAM transgenic mice is likely due to a significant expansion of islet progenitors before their endocrine specification. The effects of hEpCAM overexpression that we observed are consistent with the functional involvement of this glycoprotein in the modulator of epithelial cell adhesion and growth. Thus, collectively, our data provide important in vivo validation of hEpCAM signaling properties and offer unique opportunities to further explore the function of this glycoprotein in the regulation of islet development and function. Accordingly, studies are currently under way to test hEpCAM functions in specific pancreatic cell lineages to determine if it can be targeted to promote islet cell expansion and/or regeneration. These are two areas of research that hold significant implications for the future development of cell replacement therapies to cure diabetes.
Footnotes
Acknowledgements
We are grateful to Dr. Laura Crisa (University of Washington, Department of Medicine, Seattle, WA) for insightful discussions and comments on the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Work in the Cirulli lab was supported by JDRF Research Grants #1-2005-1084 and #1-2004-13, and by the WA State Life Sciences Discovery Fund Program Grant #4553677.
