Abstract
Pancreatic β cell regeneration remains poorly understood, yet stimulation of adult β cell neogenesis could lead to therapies for type 1 and type 2 diabetes. We studied the effect of embryonic stem (ES) cell transplantation on pancreas regeneration following β cell injury. Female Balb/c nude mice were treated with streptozotocin to induce hyperglycemia and received an ES cell transplant 24 hr later beneath the renal capsule. Transplantation of ES cells prevented hyperglycemia in a subset of mice, maintaining euglycemia and mild glucose tolerance up to 5 weeks. Pancreata of euglycemic mice showed histological evidence of β cell regeneration and expression of pancreas and duodenum transcription factor-1 (PDX-1) and neurogenin 3 (Ngn3) in ductal epithelium. Cell tracing analysis indicated that significant β cell neogenesis from progenitor cells occurred between 2 to 3 weeks following injury in ES cell-transplanted mice but not in sham-transplanted animals. Significantly, whereas pancreas-localized ES cells or their derivatives were adjacent to sites of regeneration, neogenic pancreatic epithelia, including Ngn3+ cells, were endogenous. In conclusion, transplanted ES cells can migrate to the injured pancreas. Transplantation is associated with enhanced endogenous regeneration characterized by expression of Ngn3 and increased β cell differentiation from endogenous progenitor cells. This manuscript contains online supplemental material at http://www.jhc.org. Please visit this article online to view these materials.
Keywords
T
Although adult β cell regeneration from ductal epithelium recapitulates aspects of fetal β cell development, one important distinction to date has been the absence until very recently (Xu et al. 2008) of any clear evidence of neurogenin 3 (Ngn3) expression, an essential protein for all fetal islet cell development (Gradwohl et al. 2000), in the adult pancreas (Bonner-Weir et al. 2004). Previously, we found that β cell damage can induce transplanted embryonic stem (ES) cells to undergo embryonic-like pancreatic endocrine and exocrine neogenesis in vivo from PDX-1+ ductal epithelium between 15 and 28 days after transplantation (Kodama et al. 2008). We also found that transplanted ES cells localized to the injured but not to the normal pancreas (Takeshita et al. 2006). In this study, we show that a subset of renal capsule-transplanted ES cells migrating to the pancreas did not differentiate into β cells by themselves, but improved glycemic control in mice following β cell loss.
Materials and Methods
Animals and Streptozotocin (STZ) Treatment
Animal experiments were performed in compliance with the guidelines of the Institute for Laboratory Animal Research at the National Cancer Center Research Institute. Female Balb/c nude mice (CLEA; Tokyo, Japan), aged 7 weeks, were used for all experiments. Blood glucose measurements were performed on whole venous blood collected from the tail vein using the Freestyle Flash Blood monitoring system (Nipro; Tokyo, Japan) according to the manufacturer's instructions. The basic experimental regimen consisted of a single 300-μl intraperitoneal injection of STZ (Sigma Aldrich; St. Louis, MO) within 15 min of dissolution in freshly prepared 20 mM cold citrate buffer (pH 4.5); 240 mg/kg was used to induce hyperglycemia, and 175–200 mg/kg was used to induce β cell damage with low incidence of overt hyperglycemia. Non-STZ-treated mice received 300 μl citrate buffer alone. Twenty-four hr after STZ treatment, ES cells were transplanted beneath the left renal capsule as described previously (Kodama et al. 2008). PBS was dispensed under the renal capsule for sham transplantation. At subsequent time points, the animals' glucose homeostasis was monitored, and mice were sacrificed for immunohistochemical analyses. Intraperitoneal glucose tolerance testing was performed as described by the Animal Models of Diabetes Complications Consortium (www.amdcc.org). Mice were fasted for 7 hr before intraperitoneal injection with 2 mg/kg glucose solution (Sigma). In vivo imaging analysis of transplanted ES cells was conducted in a cryogenically cooled IVIS system (Xenogen; Alameda, CA) as described previously (Takeshita et al. 2006). Tumor growth was monitored weekly by whole-body bioluminescent imaging.
Culturing and In Vivo Transplantation of ES Cells
The firefly luciferase-expressing ESJ1 cell line, a J1 cell clone of 129SV male origin, was established previously (Kodama et al. 2008). The SK7 cell line, derived from an Institute of Cancer Research strain, was a gift from Professor Shoen Kume (Kumamoto University). SK7 cells were transfected with the pLucNeo plasmid (Promega; Madison, WI) and a stable, luciferase-expressing clone was isolated for use in this study. ESJ1 cells were cultured under feeder-free conditions, whereas SK7 cells were cultured on a layer of inactivated mouse embryonic fibroblasts. Both ES cell lines were maintained as described previously (Kodama et al. 2008). For transplantation, ES cells were trypsinized and dispersed to a single-cell suspension, then counted and washed in PBS. Two μl of cell suspension containing 1 × 105 or 1 × 106 ES cells was transplanted beneath the left renal capsule.
Pancreas Tissue Extraction and Luciferase Assay
The pancreas tissues were dissected, snap frozen, and pulverized under liquid nitrogen. An equal volume of tissue lysis buffer (125 mM Tris-phosphate, pH 7.8, 10 mM dithiothreitol, 10 mM EDTA, 50% glycerol, 5% Triton-X, 0.2 mM phenylmethylsulfonyl fluoride, 1 μg/ml aprotinin, 1 μg/ml leupeptin) was added, and incubated on ice for 20 min. Five μl of supernatant was mixed with 100 μl of luciferase assay reagent (PicaGene; Toyo, Inc., Tokyo, Japan), and the light unit of the luciferase activity was assayed using a luminometer (MiniLumat LB 9506; EG and G Berthold, Vilvoorde, Belgium). The data were expressed as the relative luciferase activity, which is the chemiluminescence elicited from pancreas tissue extract divided by the background chemiluminescence of the lysis buffer alone. The analyses (carried out in triplicate) were repeated a minimum of two times, and the mean ± standard deviations were plotted.
Immunofluorescence and Immunohistochemical Analyses
Tissues were preserved in 10% formalin solution, embedded in paraffin wax, and sectioned. Sections were deparaffinized and rehydrated, and antigen retrieval was performed by autoclaving sections for 5 min in antigen recovery buffer (Sigma). Antibody staining was performed as described previously (Kodama et al. 2008). The following primary antibodies and dilutions were used: anti-insulin (rat, 1:500; R and D Systems, Minneapolis, MN; #MAB1417), anti-PDX-1 (rabbit, 1:200; Upstate, Charlottesville, VA; #07–696), anti-PDX-1 [mouse, 1:2000 tyramide signal amplification (TSA); Developmental Studies Hybridoma Bank (DSHB), University of Iowa; #F6A11], anti-Ki67 (rabbit, 1:500; Vector Laboratories, Burlingame, CA; #VP-K451), anti-Ngn3 (mouse, 1:2000 TSA; DSHB, University of Iowa; #F25A1B3), and anti-α-amylase (1:500; Sigma, #A8273). For detection of BrdU+ cells, the BrdU In Situ Detection Kit (BD Biosciences; Tokyo, Japan) was used, and fluorescence detection was performed by TSA using the NEN TSA fluorescence systems kit according to the manufacturer's guidelines (PerkinElmer; Wellesley, MA). For staining with mouse primary antibodies, the M.O.M. kit from Vector laboratories was used, followed by TSA. Fluorescence microscopy was performed using a Nikon Eclipse E1000 epifluorescence microscope and 20× and 40× objectives, equipped with a QICAM FAST1394 CCD camera. Original images with original fluorescent colors were acquired using MetaMorph V6.1r4 software (Universal Imaging Corp.; Sunnyvale, CA) and overlaid using Adobe Photoshop CS2 V9.0. For measurement of tissue section area, and histomorphometry, ImageJ software (V 1.31v) (available from http://rsb.info.nih.gov/ij/download.html) was used. For morphometric analyses of BrdU+ β cells, sections were costained for insulin and BrdU. The number of BrdU+ β cells was expressed as a percentage of the total number of β cells for each tissue section. For each animal, 3 × 3-μm-thick whole pancreas tissue sections, each separated by 30 μm, were examined.
Fluorescence In Situ Hybridization (FISH)
The digoxigenin (DIG)-labeled Y-chromosome probe was purchased from Chromosome Science Lab, Inc. (Sapporo, Japan), and FISH was performed on 3-μm tissue sections according to the probe manufacturer's guidelines. Briefly, sections were deparafinized and rehydrated, then denatured for 10 min in 2 × saline sodium citrate (SSC) in a microwave oven. Sections were partially digested using pepsin (Sigma) dissolved in 0.1NHCl for 20min at 37C, then dehydrated before applying the probe solution. After denaturation at 90C for 10 min, hybridization was performed in a humidified chamber at 37C for 16 hr. Washing was performed using 50% formamide/2 × SSC. Immunological detection was performed using anti-DIG primary antibody (Dako; Glostrup, Denmark) and AlexaFluro 488 secondary antibody. Sections were counterstained with 4',6-diamidino-2-phenylindole.
Tracing of β Cell Replication
To ascertain the relative contribution of cell replication vs progenitor differentiation, mice were given BrdU (BD Biosciences) by a single intraperitoneal injection on two consecutive days (days 2 and 3) following injury and ES cell or sham transplantation. Then on days 5, 12, and 21, the mice were sacrificed (n=3 per group at each time point), and pancreata were examined histomorphometrically for BrdU+/insulin+ double-positive cells as described above. The total number of β cells, as well as the number of BrdU+/insulin+ cells on each section, were counted and averaged for each mouse.
Statistical Analysis
Two-tailed Student's t-tests were performed, with p<0.05 or p<0.01 considered to be significant. Values are given as mean ± SD or ± SEM.
Results
ES Cell Transplantation Maintains Long-term Euglycemia in STZ-treated Mice
A single high dose of 240 mg/kg STZ caused hyperglycemia within 1 week (blood glucose concentration >200 mg/dl) in 7-week-old female Balb/c nude mice, confirming previous findings (Kodama et al. 2008). When mice (n=16) received 1 × 105 undifferentiated ESJ1 cells by transplantation beneath the left renal capsule 24 hr after STZ treatment, one third (6/16) of the ESJ1-transplanted animals remained euglycemic (blood glucose concentration <200 mg/dl) from day 3 (when glucose measurements were first made) until sacrifice on day 35 (Figure 1A). During the experimental period, the euglycemic mice remained healthy, with positive weight gain (data not shown) and absence of polyuria, and they maintained mild glucose intolerance (Figures 1B and 1C). The remaining ES cell-transplanted mice progressed to severe hyperglycemia, as did the sham-transplanted (PBS instead of ES cells) group. However, the progression to severe hyperglycemia (>400 mg/dl) was significantly (p<0.05) delayed in the ES cell transplant group (Figure 1D). At sacrifice, pancreata of euglycemic animals appeared normal, with no macroscopic ES tumors, unlike results seen previously following intraperitoneal ES cell transplantation (Takeshita et al. 2006).
Prompted by earlier results indicating ES cell homing to the injured pancreas (Takeshita et al. 2006), we examined pancreata for evidence of ES cell migration from the renal capsule. Luciferase enzymatic activity was detected in tissue from STZ-injured but not STZ-untreated transplant recipients 4 weeks after luciferase-transgenic ES cell transplantation (Figure 1E). A similar result was also found with an allogenic luciferase-transgenic ES cell line, SK7 (data not shown). These data indicated that STZ injury promotes ES cell migration from the renal capsule into the pancreas, which seems to be associated with recovery from high-dose STZ following ES cell transplantation.
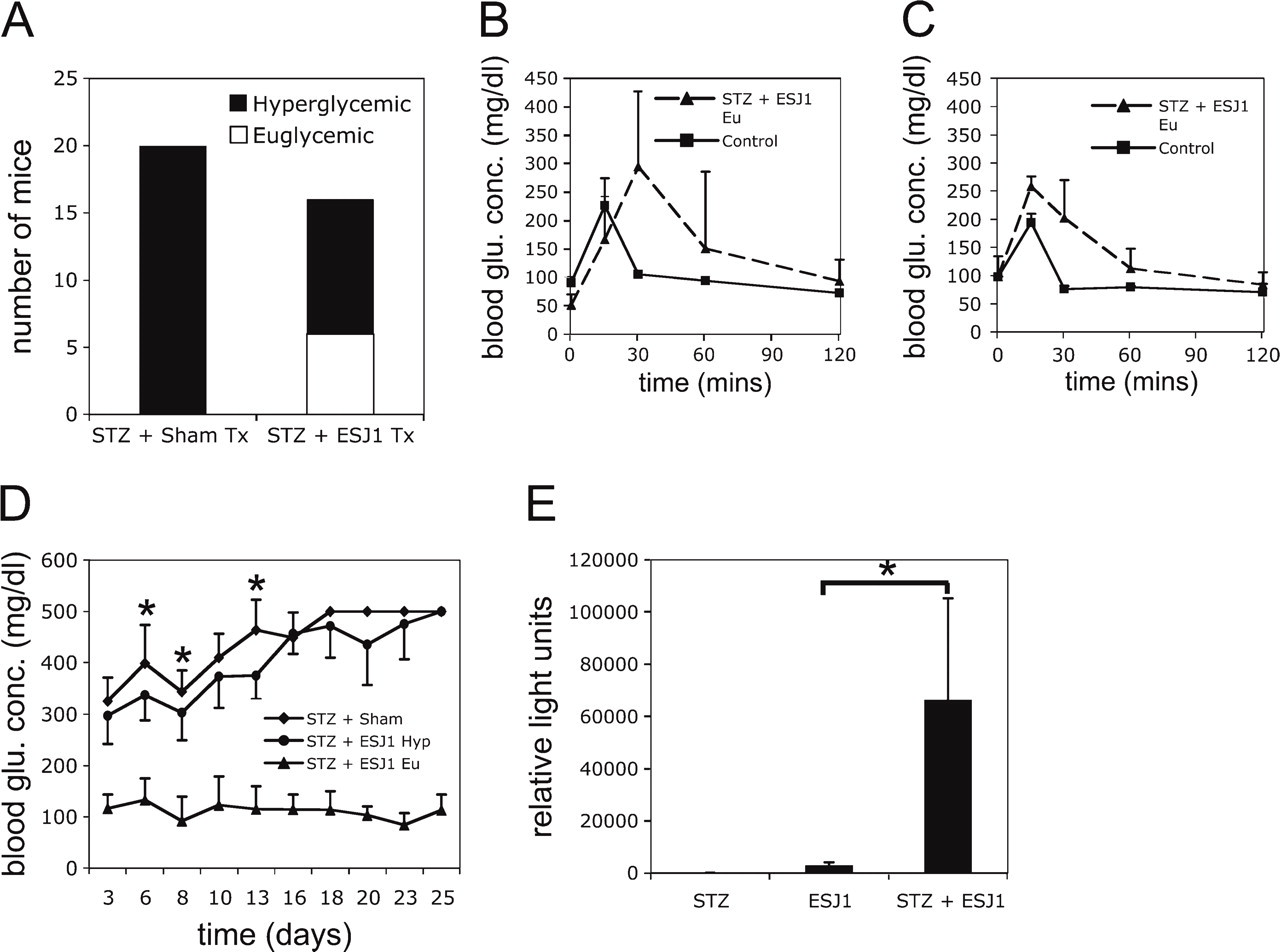
Embryonic stem (ES) cell transplantation prevents hyperglycemia in streptozotocin (STZ)-treated mice. (
Pancreas Regeneration in Euglycemic Mice From PDX-1+ Ductal Epithelium
We compared ES cell-transplanted and sham-transplanted pancreata immunohistochemically. Among the recovered animals, evidence of regeneration was identified, involving both large intralobular ductal epithelium, as well as smaller ductal cells, and characterized by expression of the mature β cell and pancreatic stem cell marker PDX-1 (Figure 2). PDX-1 expression in adult rodent pancreas ductal epithelium is commonly associated with endocrine regeneration (Bonner-Weir et al. 2004). Some ducts displayed near-uniform expression of PDX-1 throughout the duct wall (Figures 2B and 3A). Although the number of β cells in the vicinity of these PDX-1+ ducts was comparably low, most were individual cells or small clusters of cells found in the connective tissue surrounding these ducts, i.e., not embedded in islets or embedded in exocrine parenchyma, suggesting that they were derived from adjacent PDX-1+ duct cells (Figure 3). These β cells were non-dividing (ki67-; data not shown).
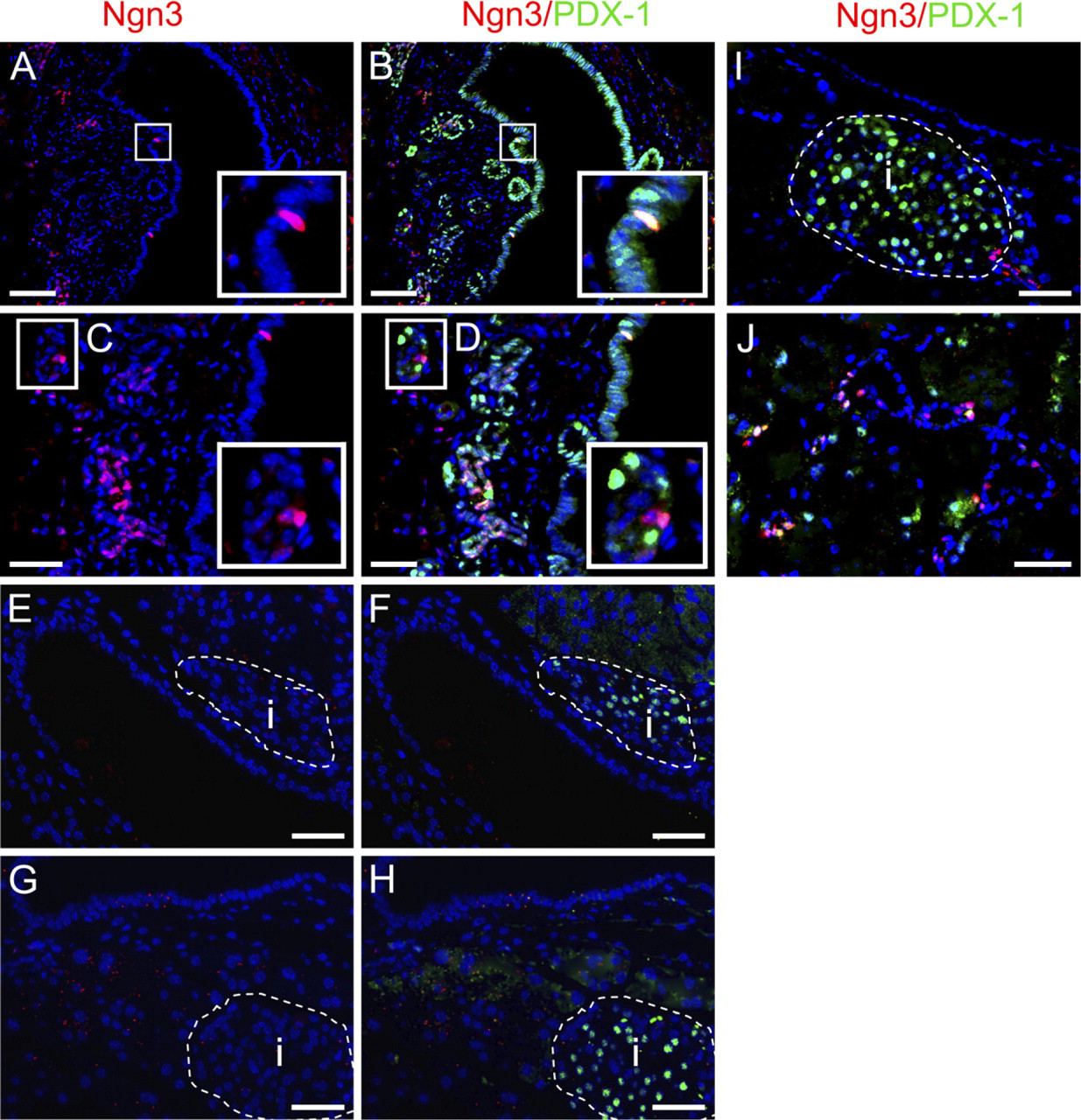
Neurogenin 3 (Ngn3) and pancreas and duodenum transcription factor-1 (PDX-1) expression in STZ-treated ES cell-transplanted mice. Nuclear-localized PDX-1 expression was detected in the duct cells of STZ + ES cell-transplanted mice (
A key distinction between adult pancreas regeneration and fetal pancreas development to date has been the absence and absolute requirement, respectively, of Ngn3 protein expression. In the present study, however, we detected Ngn3 protein expression in isolated cells within the wall of large ducts coexpressing PDX-1 (Figures 2A and 2B) and also as clusters, both PDX-1+ and PDX-1-, within smaller ducts. In addition, Ngn3+ duct cells were found associated with established islets (Figure 2I). There was an overlap with PDX-1 expression among the Ngn3+ cells, a pattern resembling the secondary transition stage of fetal endocrine pancreas development (Pictet and Rutter 1972). The presence of Ngn3+ cells 5 weeks after STZ treatment is an indication of prolonged β cell neogenesis. No overlap between Ngn3 and insulin expression was detected (data not shown).
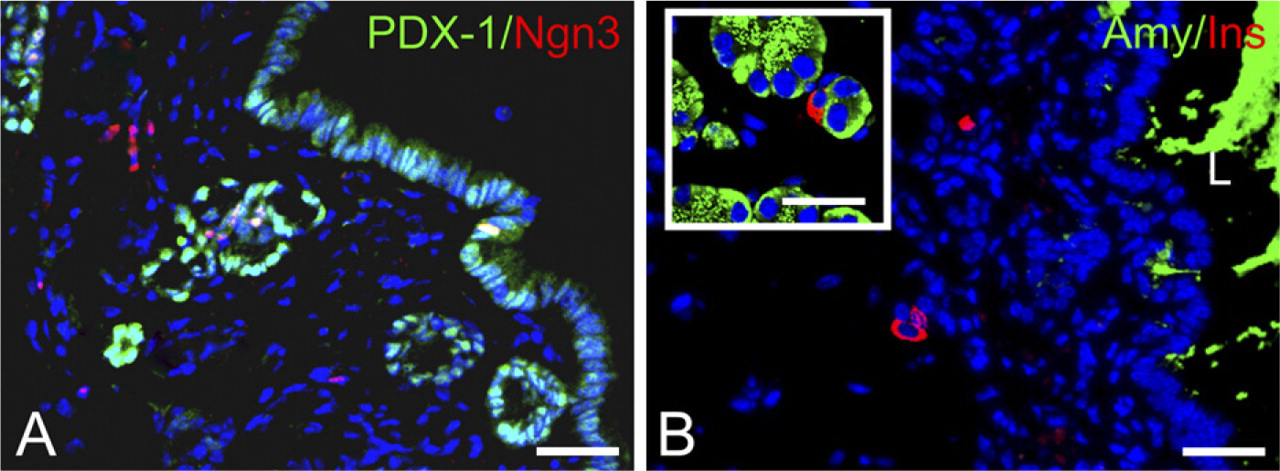
β cells at sites of regeneration. Adjacent to PDX-1- and Ngn3-expressing ductal epithelium, isolated and small clusters of insulin+ cells were found in STZ + ES-transplanted mice. Amylase staining was weak, other than in ductal lumen (
Neogenic Endocrine Progenitor Tissue Is Derived From an Endogenous Source
Identification of the fate of ES cell derivatives following migration to the pancreas is a key step toward understanding their role in the regenerative response to β cell injury. To distinguish male (ES-derived) from female (endogenous) cells and to identify the origin of PDX-1/Ngn3-expressing ductal epithelial cells, Y-chromosome-specific interphase FISH was performed on tissue sections from regenerating pancreata. Typically, the pancreas-localized ES cells existed as small clusters adherent to the interstitium and adjacent to sites of neogenesis. Contrary to our expectations based on former data (Kodama et al. 2008), the ES cells themselves were never identified as components of normal ductal, exocrine, or endocrine tissue within the pancreas. However, areas of regeneration (PDX-1+/Ngn3+ ductal tissue) and ES cell-derived cells were closely associated, a typical example of which is shown in Figure 4. The data suggest that the exogenous cells have a role to play in stimulating the endogenous response. Hence, sex-specific FISH analysis demonstrated that infiltrating ES cell derivatives do not comprise the metaplastic pancreatic epithelium. Importantly, in mice that became hyperglycemic following STZ treatment and ES cell transplantation, ES cell infiltration into the pancreas was rare or not detected (data not shown), whereas all of the recovered mice analyzed had readily detectable ES cell clusters in the pancreas. Together this suggests that ES cell homing from the renal capsule to the pancreas indirectly stimulates β cell regeneration after high-dose STZ.
ES Cell Transplantation Induces β Cell Neogenesis From Endogenous Progenitors
We devised an experiment to estimate the relative contribution of the processes of β cell replication vs progenitor cell differentiation toward the injury response in ES cell-transplanted and sham-transplanted mice. STZ was administered at 200 mg/kg, which in our hands typically induces overt hyperglycemia: >200 mg/dl blood glucose concentration for three consecutive readings in <50% of animals (Kodama et al. 2008). Hence, most of the animals were euglycemic at the time of sacrifice (see supplementary Table 1). The immunohistochemically detectable nucleotide analog, BrdU, was administered shortly after STZ injury/ES cell transplantation, and then the numbers of β cells that were BrdU+ at an early (day 5) and two later time points (days 12 and day 21) were counted. We made two rational assumptions: (a) Any given β cell could only divide once during a 3-week experimental period; and (b) the β cell replication component of BrdU+/Ins+ cells would be the same during all three time point analyses, because β cell progenitors are stimulated to divide within a 1-week period after injury (Xu et al. 2008). On the basis of these assumptions, we postulated that any increase in the number of BrdU+ β cells on day 5, day 12, and day 21 would indicate β cell neogenesis by progenitor cell differentiation. The results of this experiment were quite clear. A slight increase, on average, in the rate of β cell replication in the ES cell-transplanted vs sham-transplanted animals was found at day 5 (Figure 5), although it was not statistically significant. At day 12, there was no detectable difference in the overall number of BrdU+ cells compared with day 5, although the marginal difference between ES cell-transplanted and sham-transplanted animals remained. However at day 21, we detected the first indication of progenitor-derived β cell neo-genesis: a significant (p<0.01) rise in the fraction BrdU+ β cells in the ES cell-transplanted group compared with this group at day 5 and day 12. There was also a significant (p<0.01) difference between ES cell-transplanted and sham-transplanted animals at day 21 (Figure 5). The data are compatible with a wave of β cell differentiation from progenitor cells in the ES cell-transplanted group that were stimulated to divide during the first week following pancreas injury. Although this experiment was ended at day 21, it is likely that the margin of progenitor-derived cells would have increased further, based on immunohistochemical detection of Ngn+/PDX-1+ duct cells at day 35 following pancreas injury (Figure 2).
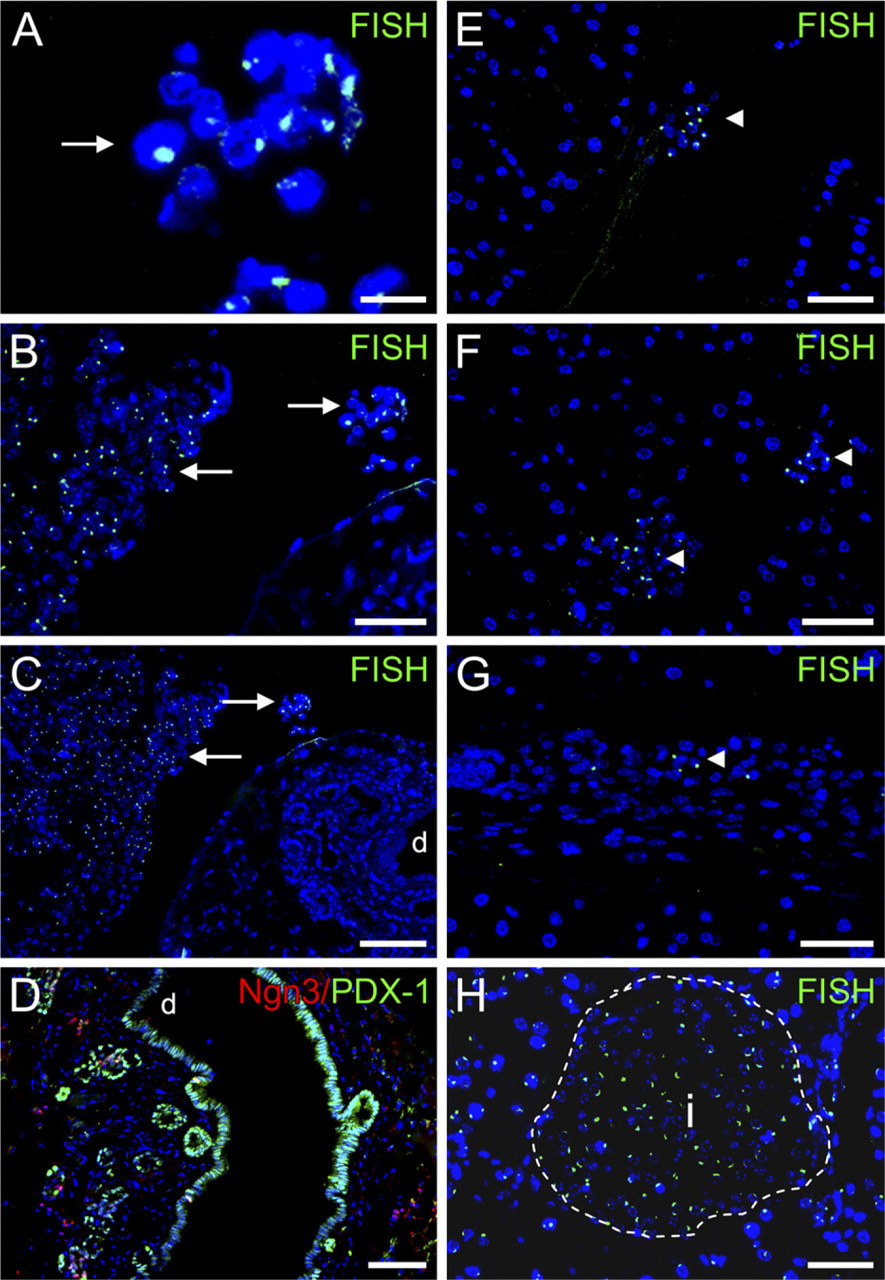
Neogenic pancreatic endocrine tissue is derived from an endogenous source. Y-chromosome-specific interphase fluorescence in situ hybridization was used to distinguish donor (male, Y+; green nuclear signal) from host (female, Y-) pancreatic tissue. PDX-1+ and Ngn3+ ductal epithelium and all endocrine cells that were examined (>8000) were Y-chromosome-negative. However, sites of regeneration were in proximity to transplanted cells. (
Discussion
In this study, we report that in a subset of STZ-treated mice that received renal capsule-transplanted ES cells, Ngn3 expression, enhanced β cell neogenesis, and resistance to hyperglycemia were observed. These regenerative responses were associated with migration of transplanted cells to the injured pancreas.
The islets of the adult pancreas are fully developed shortly after birth. Thereafter, adjustments to the pool of β cells in response to fluctuations in metabolic demand appear to arise from self-duplication (Dor et al. 2004). However, this mechanism does not wholly account for pathological β cell death, in which there appear to be alternative routes for β cell replenishment that are collectively understood to be weak (Rood et al. 2006). Pancreatic β cell regeneration appears to include a rapid response (Levine and Itkin-Ansari 2008), which is likely to be β cell replication, and a delayed response that involves duct cell metaplasia (Bonner-Weir et al. 1981; Xu et al. 2008). STZ-induced β cell death resembles the specific β cell loss during diabetes and typically results in a characteristically weak regenerative response with incomplete recovery of lost cells. To our knowledge, this response to STZ has never previously been associated with expression of Ngn3. Caution, however, is warranted in the use of rodent models, because STZ may additionally damage the surviving cells, hampering the regenerative response, at least its cell replication component, leading to an underestimation of the importance of cell duplication in diabetic individuals (Nir et al. 2007). In comparison with the delayed response seen in a previous study involving STZ-treated rodents (Bonner-Weir et al. 1981), our present findings suggest that ES cell transplantation caused an amplification or prolongation of the endogenous response to β cell damage, exposing sites of regeneration and indirectly causing or enhancing Ngn3 protein expression at these sites. Conversely, in the absence of transplanted ES cells, Ngn3 protein expression was not detectable, indicating that it may otherwise be very transient, at undetectable levels, or perhaps may not be expressed at all.
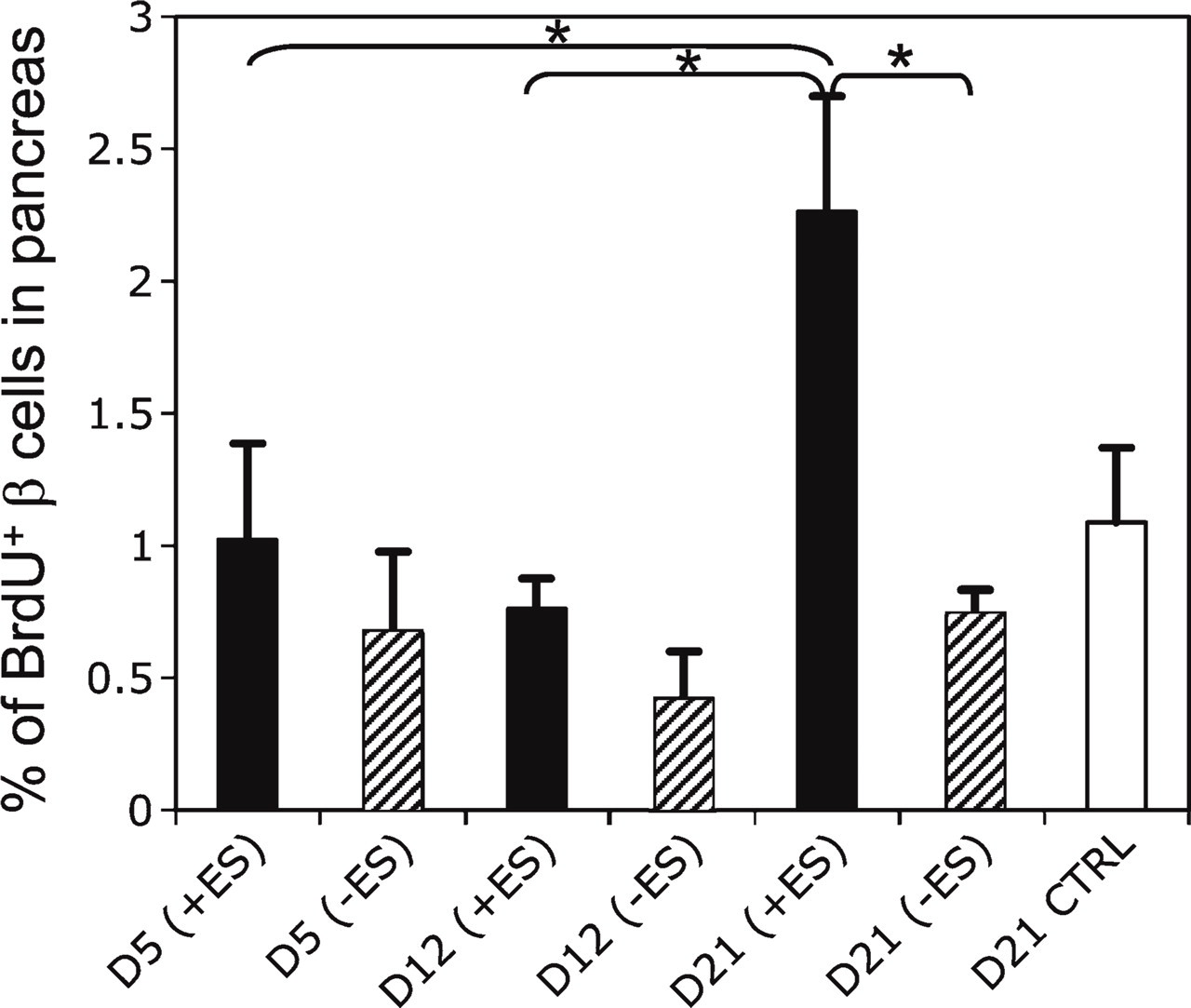
Lineage analysis of β cell neogenesis. A single moderate dose (200 mg/kg) of STZ was given to mice on day 0, and transplantation (ES cell or sham) was performed on day 1. BrdU was administered to mice from three different groups [STZ + ES cell transplant (black bars, n=9), STZ + sham transplant (hatched bars, n=9), and non-treated controls (white bars, n=3)] on days 2 and 3 of the study. BrdU/insulin double-positive cells were counted and expressed as a percentage of total insulin+ cells in tissues from STZ-treated animals sacrificed at days 5, 12, and 21. For non-treated controls, morphometry was performed on day 21 only. Day 5 values represent the rate of β cell neogenesis by mitosis of preexisting β cells. Day 12 and day 21 values represent the combined β neo-genesis from preexisting β cells and that from progenitor differentiation. A significant rise in the rate of BrdU+ β cells in the STZ + ES transplant group at day 21, compared with days 5 and 12 and compared with sham-transplanted mice, is compatible with the onset of a wave of β cell differentiation in this group between days 12 and 21, from progenitor cells that were stimulated to divide shortly after injury. Data are given as mean ± SEM, ∗ p<0.01.
Unlike previously, when ES cells were administered intraperitoneally (Takeshita et al. 2006), ES cells that localized to the pancreas in the present study did not form noticeable tumors; instead, these cells existed as small clusters (see Figure 4). The absence of tumors may indicate that they are in some differentiated state, although it is still possible that some of these pancreas-localized ES-derived cells are tumorigenic. It will be important to characterize the subsets of ES-derived cells that congregate within the pancreas. This will be addressed in a future study. What we can determine to date is that these cells appear to be Ngn3- and PDX-, as well as negative for differentiated islet cell markers such as insulin and glucagon, while many of them are proliferating (Ki671). Identification of these cells may help to explain their role, if any, in pancreas regeneration following STZ-induced injury. In any case, insulin+ cells were not detected among the ES cell derivatives in the pancreas. Although the role of the ES cells remains unclear, a plausible speculation might be that they secrete mitogenic factors. In both β cell replication and neogenesis from progenitor cells, exogenous growth factors could amplify the response. The pancreatic duct cells are known to express receptors for mitogenic factors, which, following activation, would lead to an expanded pool of precursor cells susceptible to endogenous β cell differentiation. Keratinocyte growth factor (Movassat et al. 2003), GLP-1/exendin-4 (Tourrel et al. 2001), ghrelin (Irako et al. 2006), gastrin (Rooman et al. 2001), epidermal growth factor (Rooman and Bouwens 2004), and activin + betacellulin (Li et al. 2004) all appear to promote regeneration in this manner. However, a more complicated involvement of the ES cells in endocrine cell specification and differentiation cannot be ruled out.
Lineage tracing of β cell replication has been applied to the problem of cell ontogeny in the adult pancreas (Dor et al. 2004; Teta et al. 2007). In these studies, it was demonstrated that cell replication is the primary if not only method of β cell neogenesis in the normal and acutely injured adult pancreas. In the present study, we applied a cell tracing approach to distinguish β cell replication and progenitor cell differentiation in our STZ injury/ES cell transplantation model. For the latter route, following progenitor cell expansion, an undefined time lag for β cell differentiation is required. This β cell differentiation time period was estimated to lie between day 12 and day 21 following initial β cell injury, due to the detection of a significant rise in neogenic β cells at day 21 compared with earlier time points in the ES cell-transplanted group. In the case of β cell neogenesis through replication of preexisting β cells, it would appear that only a single round of replication occurred for any β cell during the experimental period based on the findings of Teta et al. (2007), namely, that >1 month refractory period follows adult β cell replication. Hence, although further analysis is required to show a clear effect of ES cell transplantation on β cell replication, the data presented here indicate a significant effect of ES cell transplantation on β cell differentiation from progenitor cells at day 21. In the sham-transplanted animals, no significant β cell neogenesis was observed during the course of the experiment. The apparent absence of β cell differentiation from progenitor cells in sham-transplanted mice is in line with the results of Teta et al. (2007), who demonstrated that acute pancreas injury does not ordinarily induce progenitor-derived neogenic β cells.
A question raised is whether ES cell transplantation can reverse established hyperglycemia. When we transplanted ES cells into mice with established hyperglycemia 15 days following high-dose STZ (n=7), no reversal of hyperglycemia was observed up to day 35, despite a 10-fold higher cell number (1 × 106) than used at 24 hr after STZ (see supplementary Figure 1). This demonstrates that ES cells do not have an independent glucose-lowering ability and that the effect on glycemic control seen in animals following day 1 transplantation appears to require interaction with host cells.
For type 1 diabetes mellitus, replacement of a patient's islets of Langerhans is the only treatment that achieves an insulin-independent, constant normo-glycemic state and avoidance of hypoglycemic episodes (Pileggi et al. 2006). In the future, in vitro-differentiated ES cells may provide an unlimited supply of β cells for transplantation. Before that is possible however, a key test of function for ES-derived β cells is their ability to improve glucose regulation in diabetic animals, often STZ-injured athymic Balb/c nude mice. In light of the results of our study, contaminating ES cells or their derivatives (Shi et al. 2005) need to be considered, not only as a teratoma risk, but also as a potential stimulator of endogenous regeneration, an effect that could be attributed to the transplanted β-like cells.
Collectively, the data presented here indicate that ES cell transplantation is associated with a regenerative response to β cell death by STZ. However, although we believe it unlikely, we cannot rule out the possibility that the results seen are an effect of STZ. The pattern and time scale (around 3 weeks) of the onset of endogenous β cell neogenesis from progenitors are similar to those of ES cell-derived β cell neogenesis in vivo (Takeshita et al. 2006; Kodama et al. 2008). In both cases, endocrine pancreas fetal development is comparable: PDX-1+ ductal epithelium acquires Ngn3 expression and finally leads to endocrine gene expression. Hence, interaction between the injured pancreas and the ES cells may promote reciprocal β cell neo-genesis. A relationship akin to this has been reported following neuronal stem cell transplantation into the hypoxia-ischemia-injured mouse brain (Park et al. 2002).
In summary, we describe for the first time evidence of Ngn3-mediated β cell neogenesis in the adult pancreas following STZ treatment causing resistance to hyperglycemia. This response is correlated with ES cell transplantation, and resembles the developmental ontogeny of fetal endocrine β cells. Further studies are warranted to identify the signaling pathways involved in this regeneration, which will have direct relevance to diabetes therapy.
Footnotes
Acknowledgements
We thank Dr. Takahiro Ochiya for making available to us his laboratory facilities at the Section for Studies on Metastasis (NCCRI), Dr. Fumitaka Takeshita for assistance with animal care, Dr. Yoshinori Ikarashi (National Cancer Center Research Institute) for helpful discussion during the study, and Prof. Shoen Kume (Kumamoto University) for the gift of the SK7 ES cell line. The Ngn3 and PDX-1 monoclonal antibodies were developed by Dr. O.D. Madsen and were obtained from the Developmental Studies Hybridoma Bank developed under the auspices of the National Institute of Child Health and Human Development and maintained by the University of Iowa, Department of Biological Sciences, Iowa City, IA.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
