Abstract
In this study, we describe pancreatic cell ontogeny in renal capsule–transplanted embryonic stem cells (ES) after injury by streptozocin (STZ), showing pancreatogenesis in situ. Seven-week-old female BALB/c nude mice were treated with either a single 175- or 200-mg/kg STZ dose, a regimen that induces substantial β-cell damage without overt hyperglycemia, and transplanted 24 hr later with 1 × 105 ES. Immunohistochemistry was performed on ES tissue at 15, 21, and 28 days after transplantation using antibodies against stage- and lineage-specific pancreatic markers. After 21 days, PDX-1+ pancreatic foci first appeared in the renal capsule and expressed both amylase and endocrine hormones (insulin, glucagon, and somatostatin). These foci increased in size by day 28 because of acinar and duct cell proliferation, whereas endocrine cells remained non-dividing, and made up 2–4% of ES tumor volume. PDX-1, Nkx6.1, Ngn3, and ISL-1 protein localization patterns in pancreatic foci were comparable with embryonic pancreatogenesis. A prevalence of multihormonal endocrine cells, a characteristic of adult β-cell regeneration, indicated a possible divergence from embryonic islet cell development. The results indicate that β-cell damage, without overt hyperglycemia, induces a process of fetal-like pancreatogenesis in renal capsule–transplanted ES, leading to β-cell neogenesis.
Keywords
D
Adult pancreas regeneration on the other hand is less well characterized, partly because of the variety of models studied and their resultant neogenic responses (Lehv and Fitzgerald 1968; Rosenberg et al. 1983; Gu and Sarvetnick 1993; Fernandes et al. 1997; Guz et al. 2001; Lipsett and Finegood 2002; Jensen et al. 2005; Trucco 2005). However, the occurrence of transitional cells in the regenerating adult pancreas, in particular of the endocrine lineage (Gu et al. 1994; Fernandes et al. 1997; Guz et al. 2001), that are uncommon during normal pancreas development (Herrera 2000) may be a point of divergence between these related biological processes. Unlike the hepatocytes of the liver (Michalopoulos and DeFrances 1997), the regenerative ability of the β cells of the adult rodent pancreas is clearly limited (Bonner-Weir et al. 1981). Recently, work has indicated that this may be because of a lack of stem (responder) cells in the adult pancreas, whose numbers are fixed before e12.5 (Stanger et al. 2007). We were therefore prompted to study whether glucose-independent regenerative stimuli after β-cell injury could induce pancreas neogenesis in exogenous stem cells.
Streptozocin (STZ) specifically and directly kills β cells within 24 hr by necrosis (Chang and Diani 1985) at high doses but induces a secondary killing of β cells by host immune cells that is emphasized during multiple low-dose STZ (MLDS) treatment. The latter treatment bears closer similarity to diabetes etiology than single high-dose STZ- induced hyperglycemia (Like and Rossini 1976).
In this study, we show that, after STZ injury without concomitant overt hyperglycemia, both exocrine and endocrine pancreas cell neogenesis occurs in a subset of transplanted embryonic stem cells (ES) residing at the transplantation site (the renal capsule). Thus, a regenerative response seems to be captured in naive pluripotent stem cells at a remote site that recapitulates major aspects of normal embryonic pancreatogenesis.
Materials and Methods
Animals and STZ Treatment
Animal experiments were performed in compliance with the guidelines of the Institute for Laboratory Animal Research at the National Cancer Center Research Institute. Female BALB/c nude mice (CLEA; Tokyo, Japan), 7 weeks of age, were used for all experiments. Blood glucose measurements were performed on whole venous blood collected from the tail vein using the Freestyle Flash Blood Monitoring System (Nipro; Tokyo, Japan) according to the manufacturer's instructions. For induction of β-cell damage by STZ (Sigma Aldrich; St. Louis, MO), mice received a single 300 μl IP injection of STZ within 15 min of dissolution in freshly prepared 20 mM cold citrate buffer (pH 4.5) at either 175 or 200 mg/kg body weight. Non–STZ-treated mice received 300 μl citrate buffer alone. For renal capsule transplantation, 1 × 105 ES were pipetted beneath the left renal capsule 24 hr after STZ treatment. Briefly, mice were anesthetized by exposure to 1–3% isoflurane, and a 1.5-cm cut through the skin and muscle of the left flank dorsal to the spleen was made. The wound was washed with 1 ml PBS containing penicillin/streptomycin (Invitrogen; Carlsbad, CA), and the kidney was externalized. A small lateral cut was made in the kidney membrane using a scalpel, and 2 μl of cell suspension, or PBS alone for sham transplantation, was dispensed beneath the film. The kidney was replaced in the abdominal cavity, and the incision was closed using surgical wound clips. Intraperitoneal glucose tolerance testing (IPGTT) was performed as described by the Animal Models of Diabetes Complications Consortium (www.amdcc.org). Mice were fasted for 7 hr before IP injection with 2 mg/kg glucose solution (Sigma-Aldrich). IPGTT was performed on day 15 after STZ treatment.
Culturing and In Vivo Transplantation of ES
The firefly luciferase-expressing ESJ1 cell line, a J1 cell clone of 129SV male origin, was established as described previously (Yamamoto et al. 2003). ES were maintained in “feeder-free” culture on gelatin-coated tissue culture plates in DMEM (Invitrogen) containing 20% FBS (Invitrogen), 1× β-mercaptoethanol (Sigma; 100× 7 μl 100% stock in 10 ml PBS), 1× non-essential amino acids (Invitrogen), 1× nucleosides [Invitrogen; 100×: adenosine (8 mg/ml), guanosine (8.5 mg/ml), cytidine (7.3 mg/ml), uridine (7.3 mg/ml), thymidine (2.4 mg/ml)], 1000 U/ml ESGRO/LIF (Chemicon; Temecula, CA), and 1× penicillin/streptomycin (Invitrogen). After thawing, ES were passaged twice before transplantation. For transplantation, ES were trypsinized and dispersed to a single-cell suspension, counted, and washed in PBS. Cell number was adjusted to 5 × 107 cells/ml, and 2 μl of cell suspension was transplanted beneath the left renal capsule.
Immunofluorescence and Immunohistochemical Analyses
After death of the mice by cervical dislocation, ES tumor tissues were preserved in 10% formalin solution before being embedded in paraffin wax and sectioned. Sections were deparaffinized in xylene and rehydrated through a graded ethanol series. Antigen retrieval was performed by boiling sections for 5 min in 10 mM citrate buffer followed by cooling for 30 min to room temperature. Blocking was carried out using Image-iT FX Signal Enhancer (Invitrogen) for 30 min at room temperature. Primary antibodies were applied overnight at 4C. AlexaFluor secondary antibodies (donkey, 1:1000; Invitrogen) were applied for 30 min at room temperature. All antibodies were diluted in ChemMate Antibody Diluent (Dako; Kyoto, Japan). Immunofluorescence-stained sections were mounted with Vectashield mounting medium (Vector Laboratories; Burlingame, CA) containing 4′,6-diamidino-2-phenylindole, HCl (DAPI) to visualize nuclei. The following primary antibodies and dilutions were used: anti-insulin (rat, 1:500; MAB1417; R&D Systems, Minneapolis, MN), anti-glucagon (goat, 1:200; sc-7779; Santa Cruz Biotechnology, Santa Cruz, CA), anti-PDX-1 (rabbit, 1:200; 07-696; Upstate, Charlottesville, VA), anti-α-amylase (rabbit, 1:500; A8273; Sigma Aldrich), anti-somatostatin (rabbit, 1:500; SA-1267; BIOMOL, Plymouth Meeting, PA), anti-Ki67 (rabbit, 1:500; VP-K451; Vector Laboratories), anti-Nkx6.1 (mouse, 1:2000 TSA; gift from Dr. Palle Serup, Hagedorn Institute, Gentofte, Denmark), anti-ISL-1 (rabbit, 1:200; gift from Dr. Helena Edlund, University of Umea, Umea, Sweden), anti-Ngn3 (mouse, 1:2000 TSA; DSHB, University of Iowa, Iowa City, IA), and anti-cytokeratin 7 (mouse, 1:200; sc-23876; Santa Cruz Biotechnology). For staining with mouse primary antibodies, the MOM kit from Vector Laboratories was used, followed by TSA using the NEN TSA Fluorescence Systems kit according to the manufacturer's guidelines (PerkinElmer; Wellesley, MA). Hematoxylin and eosin staining was performed using standard methods. Control mouse embryonic tissue sections (e11.5 and e14.5) were purchased from GenoStaff (Tokyo, Japan). Fluorescence microscopy was performed using a Nikon Eclipse E1000 epifluorescence microscope and ×20, ×40, and ×100 (oil immersion) objectives, equipped with a QICAM FAST1394 CCD camera. Original images were acquired using MetaMorph V6.1r4 software (Universal Imaging Corp.; Nihon Medical Devices, Tokyo, Japan) and overlaid using Adobe Photoshop CS2 V9.0 (Softbank BB Corp, Tokyo, Japan). For evaluation of percent of ES tumors occupied by pancreatic foci, ImageJ software (V 1.31v; available at http://rsb.info.nih.gov/ij/download.html) was used to draw a closed perimeter around foci from multiple tissue sections and measure the sum of the areas occupied by these foci relative to the area of a closed perimeter drawn around the whole ES tumor tissue.
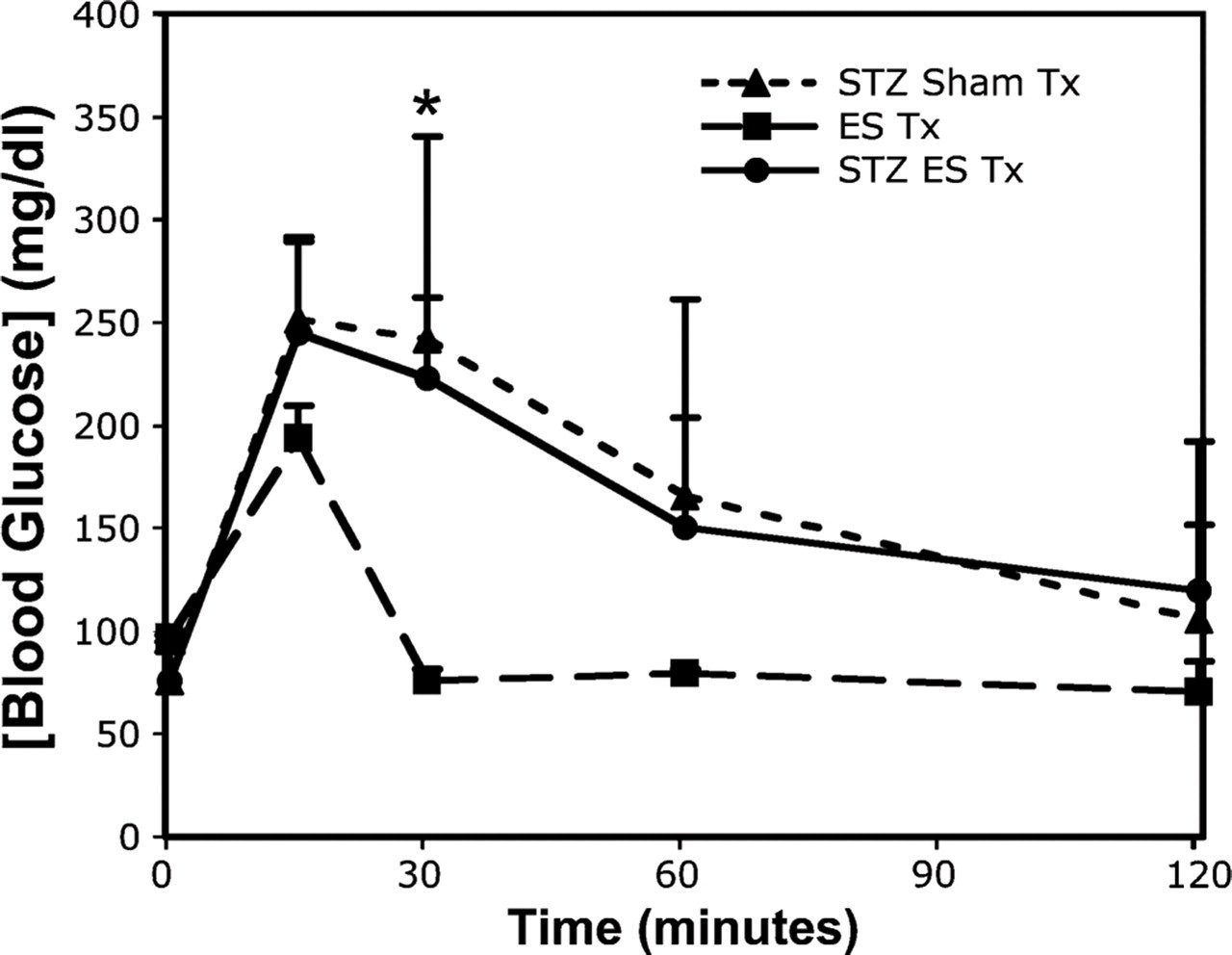
Impaired glucose tolerance in mice at day 15 after a single-dose 175- or 200-mg/kg streptozocin (STZ) treatment or citrate buffer only. Intraperitoneal glucose tolerance testing (IPGTT) was performed on three groups of mice: STZ with embryonic stem cell (ES) transplantation (STZ ES Tx, n=15), citrate buffer with ES transplantation (ES Tx, n=5), and STZ with sham transplantation (STZ Sham Tx, n=10). ∗p<0.01, Student's t-test, two-tailed.
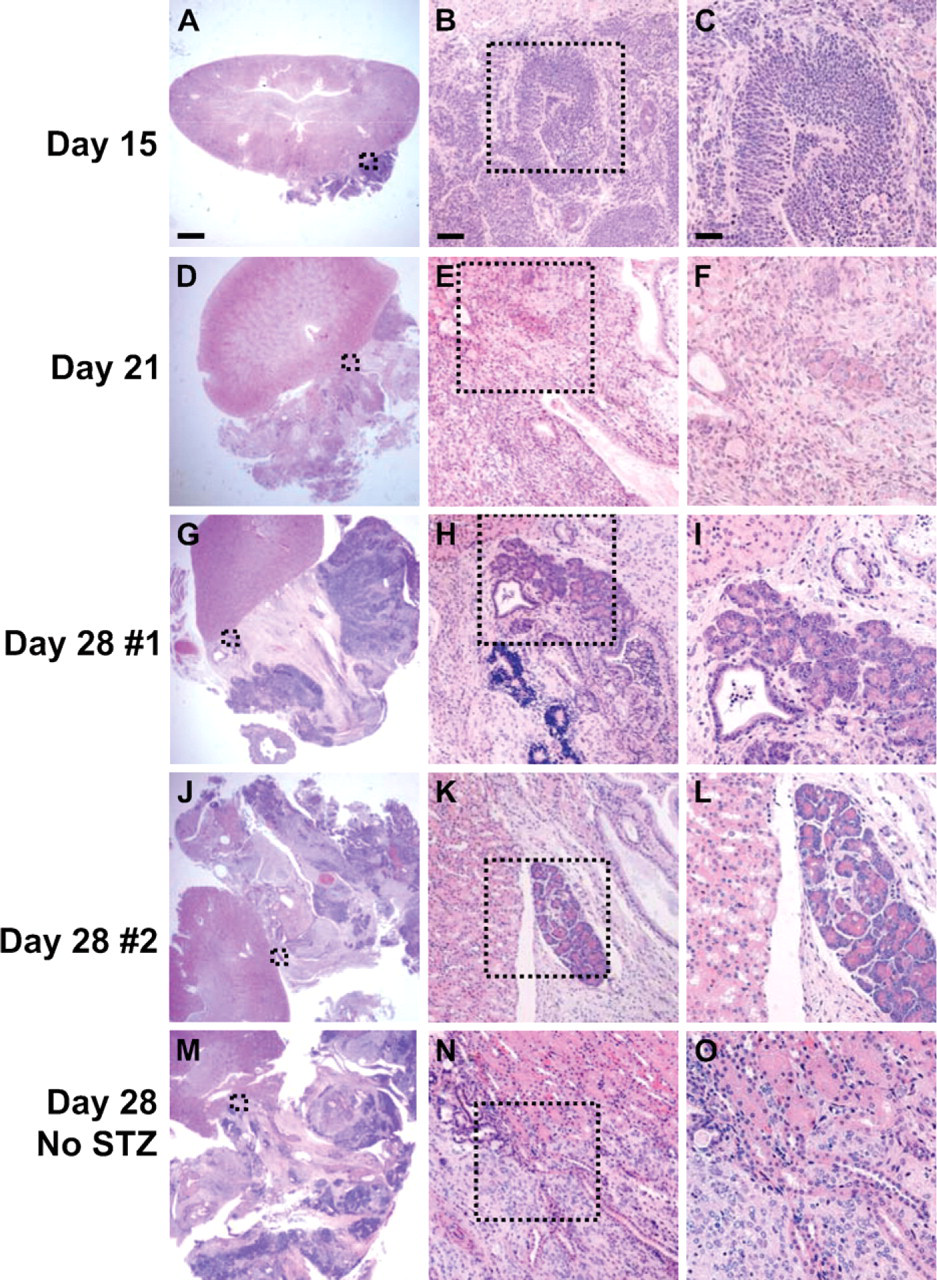
Histology of hematoxylin and eosin (H&E)-stained pancreatic foci in ES tumors grown under renal capsule of STZ-injured mice. Foci were detectable at days 21 and 28 (two examples shown) after ES transplantation but not at day 15 or day 28 in non–STZ-injured animals (arbitrary regions shown in both cases). Boxed area in left panels is enlarged in center panels and again in right panels. Pancreatic foci were almost invariably located proximal to the kidney. Bars:
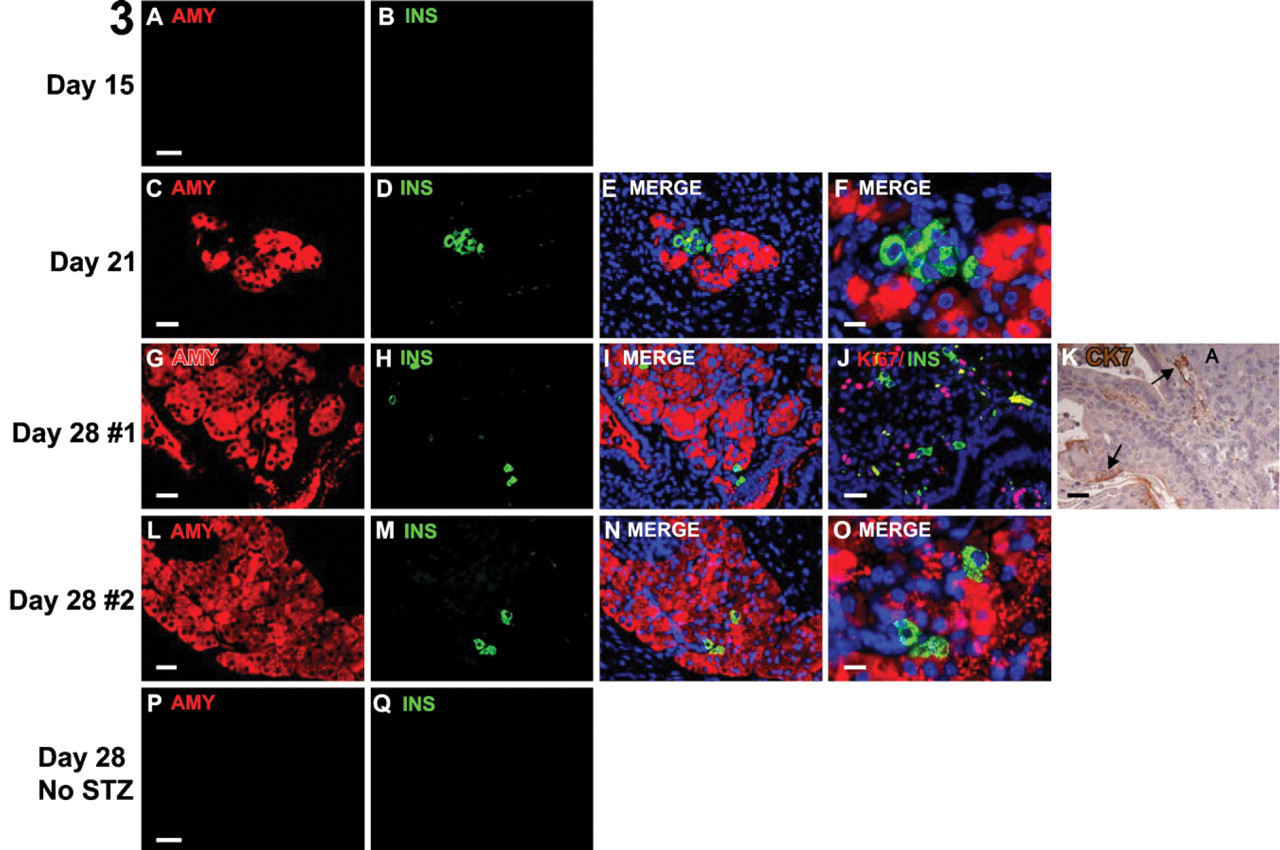
Immunohistological analysis of pancreatic foci. Shown are adjacent tissue sections to those shown in Figure 2 with the same foci stained for amylase (red-cytoplasm) and insulin (green-cytoplasm). Day 15 (
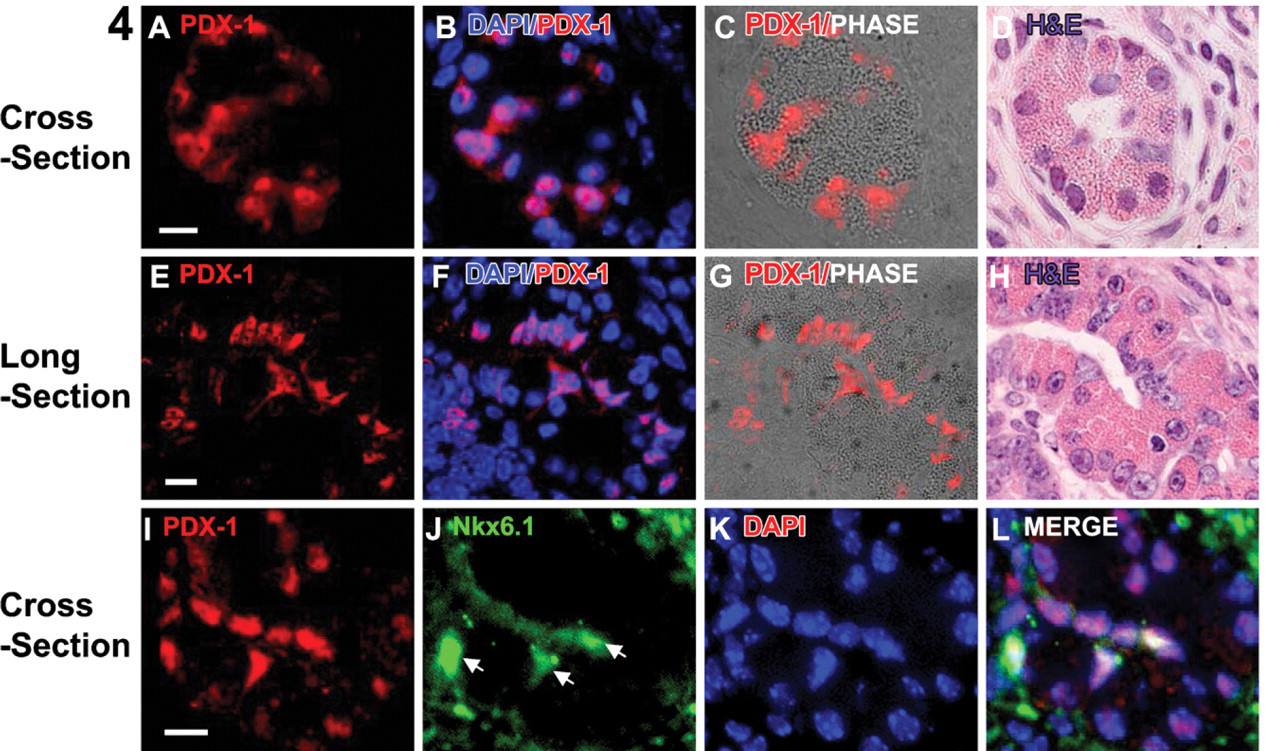
The first morphologically and biochemically distinguishable pancreatic cells in early-stage pancreatic foci. Cross- (
Morphometric analysis of pancreatic foci derived from renal capsule–transplanted ES

ES, embryonic stem cells; Tx, transplantation.
Analysis of Apoptosis
Cell death by apoptosis in ES tumor sections was assayed using the DeadEnd Fluorometric TUNEL System (Promega; Tokyo, Japan) according to the manufacturer's instructions.
Statistical Analysis
Two-sample unequal variance two-tailed Student's t-test was performed for comparisons of glucose tolerance between normal and STZ-injured animals, with p<0.05 considered significant.
Results
Pancreatic Foci Are Induced After β-Cell Damage by STZ
In a preliminary study, we found that a single administration of 175, 200, 225, or 240 mg/kg STZ to female BALB/c nude mice 7–8 weeks of age (n=20 each dose) resulted in 0%, 10%, 90%, or 100% hyperglycemia, respectively. The BALB/c genetic background displays unusually high resistance to STZ-induced DNA damage, which is directly related to poly (ADP-ribose) polymerase (PARP) activation and NAD depletion (Cardinal et al. 1999). Because we aimed to induce substantial β-cell damage without overt hyperglycemia, we treated mice with a dose of 175 or 200 mg/kg STZ before ES transplantation. All mice maintained non-fasting blood levels <150 mg/dl and displayed 1 g or greater net weight increase over the experimental period (≥15 days; data not shown). β-cell damage by STZ was assessed by IPGTT at day 15 after STZ administration, where citrate buffer was given to control mice. Impaired glucose tolerance was detected in both sham-transplanted (n=10) and ES-transplanted (n=15) groups, with both groups treated with STZ, whereas ES-transplanted mice treated with citrate buffer alone (n=5) maintained normal glucose tolerance (Figure 1). Definitive pancreatic foci, comprising both endocrine (PDX-1+/hormone+) and exocrine (amylase+) cells, were identified in ES tumors at days 21 and 28 but not at day 15 (Figures 2 and 3). Almost invariably, these foci occurred proximal to the kidney cortex (Figure 2), and each ES tumor analyzed from STZ-treated animals at both day 21 and day 28 contained multiple pancreatic foci comprising ∼1–2% and 2–4%, respectively, of the total ES tumor volume (Table 1). Importantly, pancreatic foci were not detected in ES tumors from mice without β-cell damage (citrate buffer treated; Figures 2 and 3; Table 1).
Each pancreatic focus characteristically contained a single or multiple luminal epithelial ducts from which lobulated acinar-like structures emerged. Scattered within and around the acinar cells were small isolated clusters of hormone-positive cells (Figure 3). Day 28 foci were 5–20 times larger overall than those at day 21, resulting from acinar and ductal, but not endocrine, cell expansion (determined by Ki67 expression; Figure 3J). Although small clusters of two to six endocrine cells were found at both day 21 and day 28, at day 21, the endocrine cells were grouped around the edges of acini, but by day 28, they became scattered throughout the exocrine parenchyma. The endocrine cell ratio was ∼10% of the acinar cell number at day 21 but decreased to ∼1% by day 28, apparently as a result of acinar cell proliferation. There was no evidence of apoptotic β cells (0/33) at either day 21 or day 28.
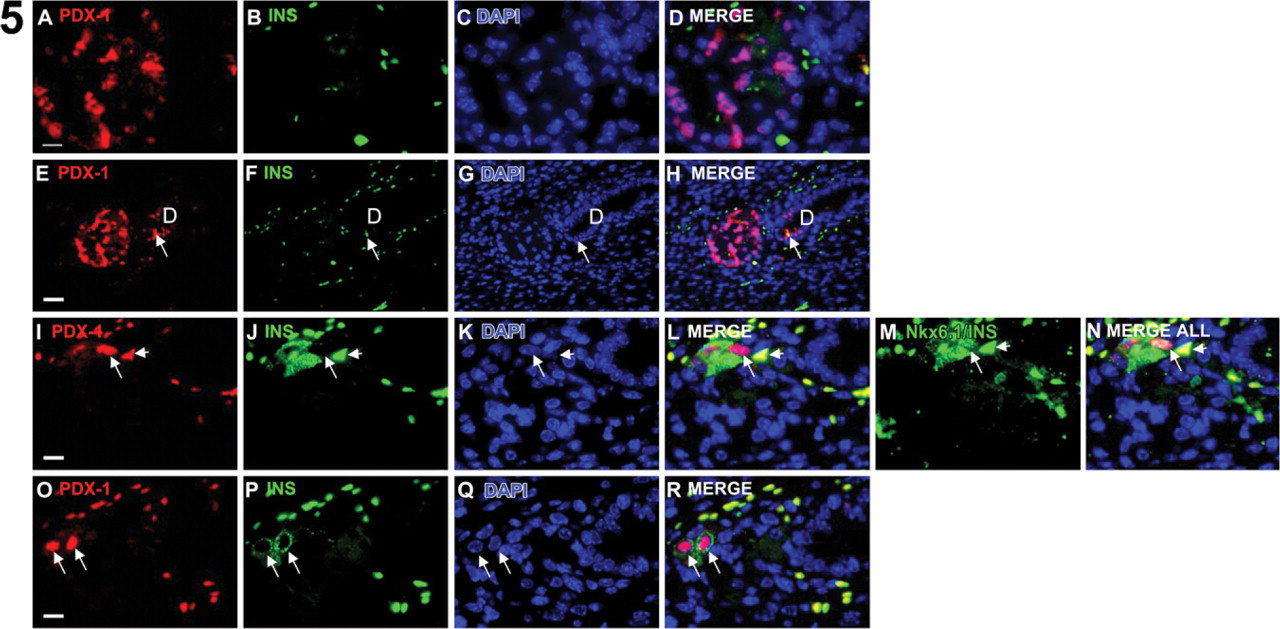
Emergence of Nkx6.1/PDX-1/insulin endocrine cells in early pancreatic foci is accompanied by a loss of PDX-1 expression in non-endocrine cells. Shown are three near-adjacent sections (
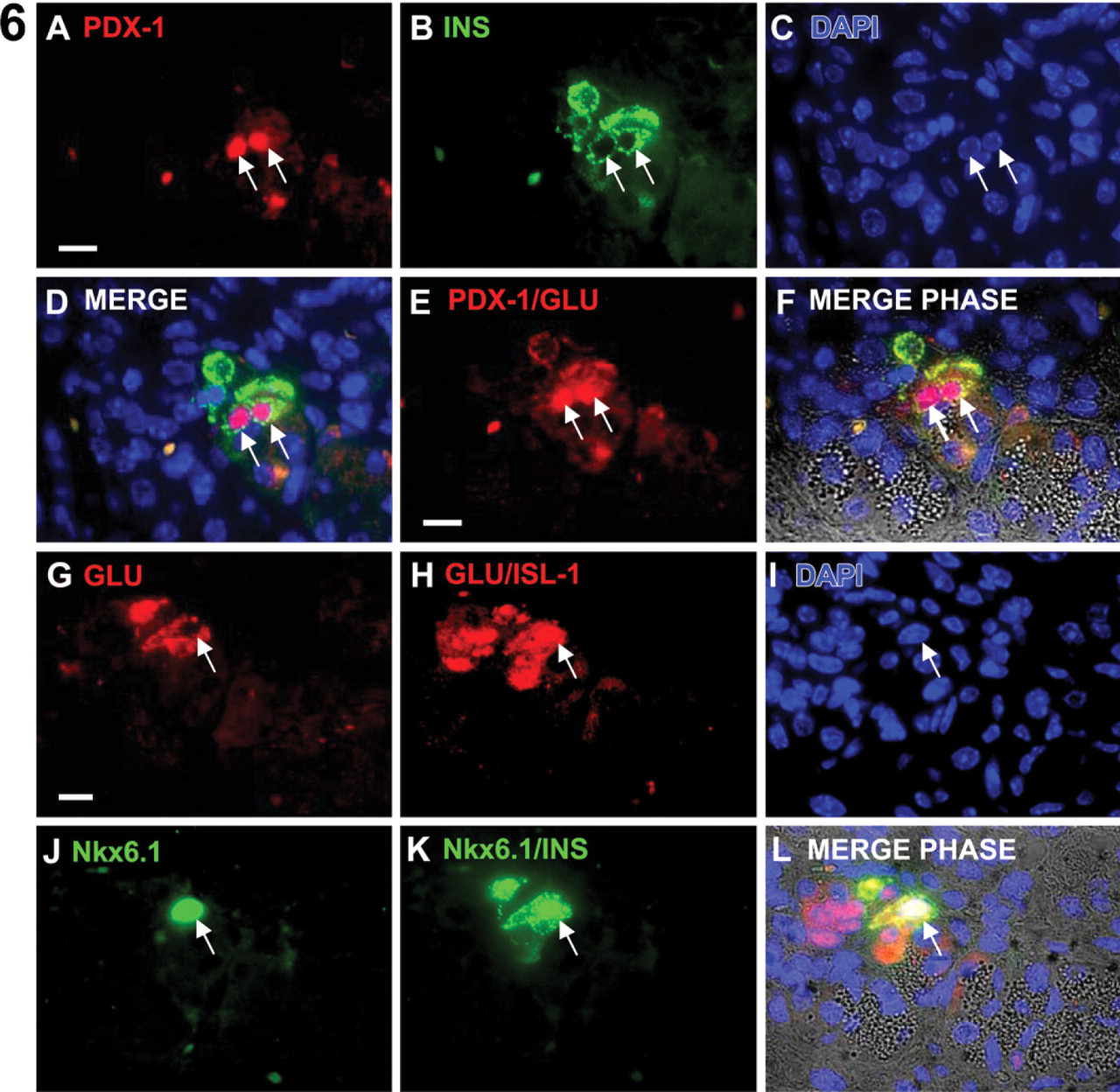
Immature endocrine cells coexpress insulin and glucagon as well as multiple islet-specific transcription factors. Two serial tissue sections from a day 21 pancreatic focus are shown (
Early Development of Pancreatic Foci
At day 21, signs of early foci development were apparent. Small foci containing ductules with histological characteristics of acinar cells—large cytoplasm with eosinophilic zymogen granules—were visible with nuclei uniformly PDX-1weak (Figure 4). In the most primitive foci, the homeodomain transcription factor, Nkx6.1, was coexpressed with PDX-1 in cells that lacked any hormone expression, suggesting that these cells were endocrine progenitor cells (Figures 4I–4L). Staining of sequential tissue sections of the same foci at day 21 revealed association of polarized PDX-1+ ductal cells (Figures 5E–5H), adjacent/evaginating PDX-1weak acinar tissue (Figures 5A–5D), and rare endocrine cells within the ducts (Figures 5E–5H, arrow). In subsequent tissue sections, PDX-1 disappeared from the majority of cells in the acinar bundle and the ductal cells, and the first clear PDX-1+/insulin+ cells appeared at the acinar cell periphery (Figures 5I–5R). These endocrine cells coexpressed the β-cell transcription factor Nkx6.1 (Figure 5J).
Endocrine Cell Development in Pancreatic Foci
Hormone coexpression was predominant in endocrine cells of ES-derived pancreatic foci. In more developed foci, PDX-1 was downregulated in the majority of the acinar-like cells, after which both insulin and glucagon protein were detectable. Surprisingly, coexpression of glucagon and insulin was dominant, with these multi-hormonal cells also expressing the islet cell markers PDX-1, Nkx6.1, and ISL-1 (Figure 6). Although foci were larger at day 28 than at day 21 (Figures 2 and 3), coexpression of insulin and glucagon persisted in the majority of endocrine cells at day 28, despite the obvious increase in foci size. As for day 21 foci, hormone coexpression at day 28 was accompanied by expression of PDX-1, Nkx6.1, and ISL-1 (Figure 7). The ISL-1+ cells were more numerous than Nkx6.1+ cells at day 21 (Figure 6), whereas at day 28, the number of ISL-1+ and Nkx6.1+ cells were equivalent (Figure 7). In the embryonic pancreas during normal mouse development, a similar pattern of ISL-1/Nkx6.1 expression as at day 21 and day 28 was seen at e11.5 and e14.5, respectively (Figure 8).
Multiple Hormone-expressing Cells
Somatostatin and insulin expression was detected simultaneously in day 21 foci, and there was minor overlap of expression of both proteins (Figure 9); weak expression of somatostatin was detectable in insulin+ cells but not vice versa (Figures 9A–9C). At day 28, this overlap was more apparent because the overall number of endocrine cells, although scattered, was increased, and the majority of insulin+ cells coexpressed somatostatin (Figures 9D–9F). Somatostatin expression levels, however, were lower in these multihormone-expressing cells than in somatostatin mono-positive cells (Figures 9D–9F, arrows). Surprisingly glucagon, somatostatin, and insulin triple hormone coexpression was common among the endocrine cells at day 28 (Figure 10). As for all of the endocrine cells identified, these multihormonal cells were non-dividing (Ki67-; Figure 10A). Although multi-hormonal endocrine cells were predominant at day 28, hormone mono-positive insulin+/PDX-1+ cells and glucagon+/PDX-1- cells were also detected in the same foci (Figure 10), suggesting that some terminal endocrine cell differentiation occurred by day 28.
Cell Proliferation and Expression of the Pro-endocrine Gene Ngn3
Ki67 staining of pancreatic foci at day 21 showed that endocrine cells were non-dividing (data not shown). At day 28, endocrine cells remained non-dividing, whereas many of the acinar, duct, and other hormone cells were Ki67+ (Figures 3 and 10). Ngn3 is the earliest pro-endocrine cell lineage marker expressed in the embryonic pancreas (Jensen et al. 2000b). Ngn3 protein was detected at day 21 and day 28, and both Ngn3+/PDX-1+ and Ngn3+/PDX-1- cells were observed (Figure 11). All Ngn3+ cells were negative for endocrine hormones and Nkx6.1 and ISL-1 (data not shown). At day 28, individual Ngn3+/PDX-1+ cells were associated with clusters of Ki67+ cells within the acinar parenchyma (Figure 11I), surrounded by hormone+ cells (data not shown). An absence of overlapping Ngn3 and ISL-1 expression is consistent with our staining data for the embryonic pancreas (Figures 8G–8I).
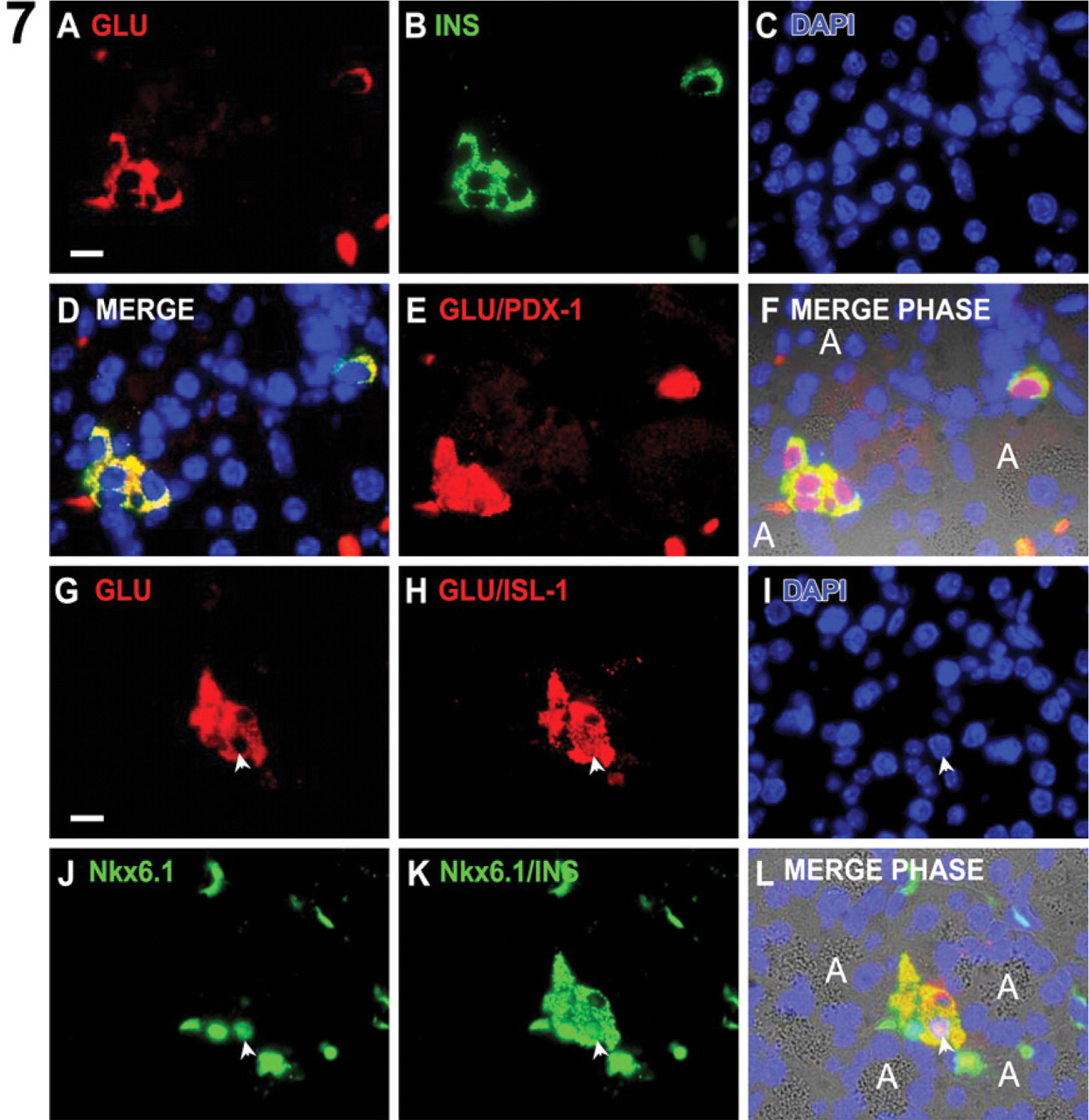
Endocrine cell characteristics in day 28 pancreatic foci. Hormone expression was more dense in day 28 endocrine cells, and the overall number of them was greater than at day 21. However, the majority of day 28 endocrine cells still coexpressed both insulin and glucagon, indicating immaturity. Two serial tissue sections from a day 28 pancreatic focus are shown (
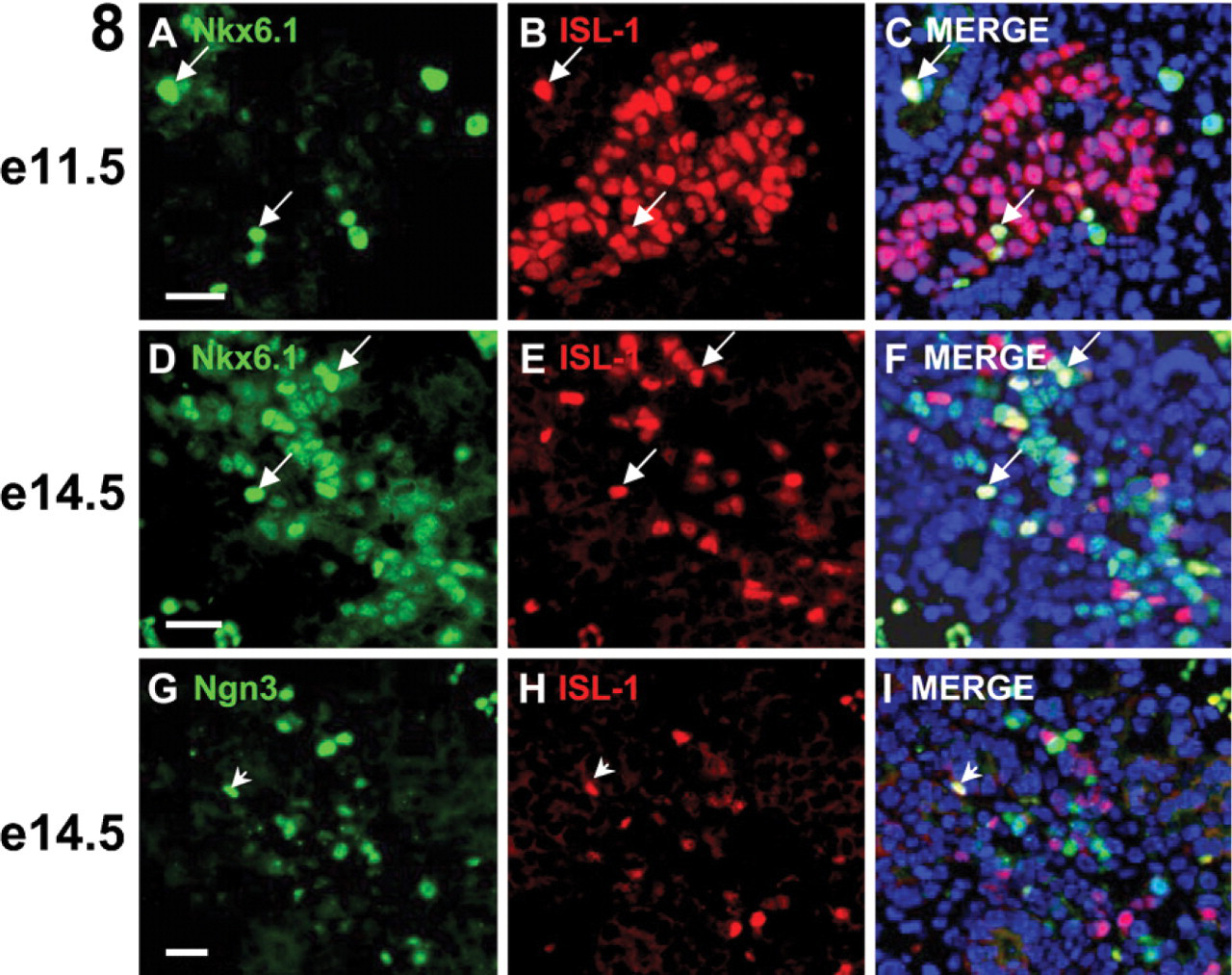
Expression of pancreatogenic transcription factors during normal mouse development. An e11.5 tissue section (
Discussion
Embryonic pancreatogenesis is complex, requiring signaling from non-endodermal (Kim et al. 1997) sources and mesenchymal stimulation (Rose et al. 1999), and it is exceedingly difficult to produce insulin+ cells through spontaneous differentiation (Kahan et al. 2003; Micallef et al. 2005). ES were reported to be able to produce insulin-expressing cells only when combined with rudiments of embryonic pancreas (but not liver or telencephalon) under the renal capsule in vivo, and this morphogenesis did not apparently occur in ES when transplanted alone (Brolen et al. 2005). In this study, we showed that pancreatogenesis can occur in ES transplanted under the renal capsule of mice 1 day after β cells have been depleted by STZ.
In an earlier study, we found that IP-transplanted ES homed to the pancreas and produced exocrine and endocrine cells locally (Takeshita et al. 2006). However, it was unclear whether attachment to the pancreas was necessary for this effect. Furthermore, there was variability in the pattern of cell homing—ES tumors occasionally formed at other tissue sites such as the liver. Finally, the growth of ES tumors with pancreatic foci was 10-fold less efficient after IP transplantation compared with renal capsule transplantation. The data presented in this study showed that pancreas β-cell induction can occur in undifferentiated stem cells transplanted beneath the renal capsule of mice with β-cell damage and raises the possibility that transplantation of a non-teratogenic stem cell type could have a therapeutic effect in individuals with impaired β-cell function.
In the mouse pancreas, after early expression throughout the pancreas anlagen, there is a dramatic decrease in the relative number of PDX-1–expressing cells between e10.5 and e11, remaining low until after e13.5, when it begins to increase again (Ohlsson et al. 1993). This temporal decrease in PDX-1 expression is thought to result from in-growth of the exocrine parenchyma and dispersion of PDX-1 positive cells, which coincides with a 20-fold relative increase in the number of insulin-producing cells between e13.5 and e15.5, a period known as the secondary transition (Pictet and Rutter 1972). In this study, PDX-1+ pancreatic foci expressed amylase and endocrine hormones 3 weeks after transplantation into STZ-treated mice, and after 4 weeks, these foci increased in size up to 20-fold, increasing both exocrine and endocrine cell numbers, although disproportionately. This effect was caused by acinar and duct proliferation (β-cell apoptosis was not evident), whereas neogenic endocrine cells were likely derived from precursor differentiation rather than by endocrine cell expansion. In the mouse embryo, endocrine cells are generally non-dividing except for a brief period of expansion before and after birth (Jensen et al. 2000a; Dor et al. 2004), whereas from e15.5, when they first appear, acinar cells remain proliferative through to birth to provide the large body of exocrine tissue needed (Slack 1995).
In the developing mouse pancreas, the basic helix-loop-helix transcription factor Ngn3 is expressed transiently during the secondary transition and marks the earliest pro-endocrine fated cells detectable; this transcription factor is essential for islet cell development, although it is downregulated before any islet hormone expression is detectable (Gradwohl et al. 2000; Jensen et al. 2000a; Schwitzgebel et al. 2000; Gu et al. 2002). In this study, Ngn3 protein was detected in hormone-negative, proliferating cells, surrounded by exocrine and endocrine cells. This pattern resembles a core of dividing progenitor cells maintained undifferentiated by notch-induced Hes-1 expression (Apelqvist et al. 1999), concordant with the secondary transition (e13.5–e15.5) period when definitive islet cell differentiation is underway.
During pancreas development, the homeodomain transcription factor Nkx6.1 is coexpressed with PDX-1 from e9.5 in most pancreatic bud epithelial cells (Pedersen et al. 2005), and after onset of the secondary transition expression, becomes restricted to the β-cell lineage (Oster et al. 1998) although it is necessary for normal development of both β and α cells (Sander et al. 2000; Henseleit et al. 2005). In the adult pancreas, its expression is confined to β cells (Brolen et al. 2005). As during fetal development, Nkx6.1 expression was detectable before hormone expression, and in more developed foci, its expression was mainly restricted to insulin-expressing cells. In contrast to fetal development (Herrera 2000), however, multiple hormone-expressing cells coexpressing PDX-1 and Nkx6.1 were detectable. The prevalence of such cells suggests a divergence from the normal embryonic β-cell neogenesis pathway in the adult in response to STZ injury. A comparison between endocrine cell ontogeny in ES-derived pancreatic foci and embryonic pancreas development is given in Table 2.
Early debate over a possible multihormonal ontogeny of islet endocrine cells (Alpert et al. 1988; Herrera et al. 1991; Micallef et al. 2005) has been quelled by lineage ablation/tracing (Herrera 2000) and static gene coexpression analysis (Jensen et al. 2000a), indicating that embryonic islet α and β cells develop from precursors with non-overlapping hormone expression. However, in the adult pancreas, transitional cells have been identified that are absent or rare during embryonic development. Somatostatin+/PDX-1+ and somatostatin+/insulin+/PDX-1+ transitional cell numbers increased in euglycemic STZ-injured pancreatic islets (with negligible β-cell neogenesis from the pancreatic ducts) (Fernandes et al. 1997). Continuous destruction and replacement of islets from ducts in human insulin promoter-interferon γ transgenic adult mice induced multihormonal (insulin+/glucagon+, insulin+/somatostatin+, insulin+/PP+), ductal/exocrine (carbonic anhydrase II+/amylase+), and exocrine/endocrine (amylase+/insulin+) intermediate cells (Gu et al. 1994). The latter was also detectable in e14.5 transgenic, but not normal, embryos (Gu et al. 1994). Together these studies suggest that multihormonal cells are a hallmark of adult regeneration.
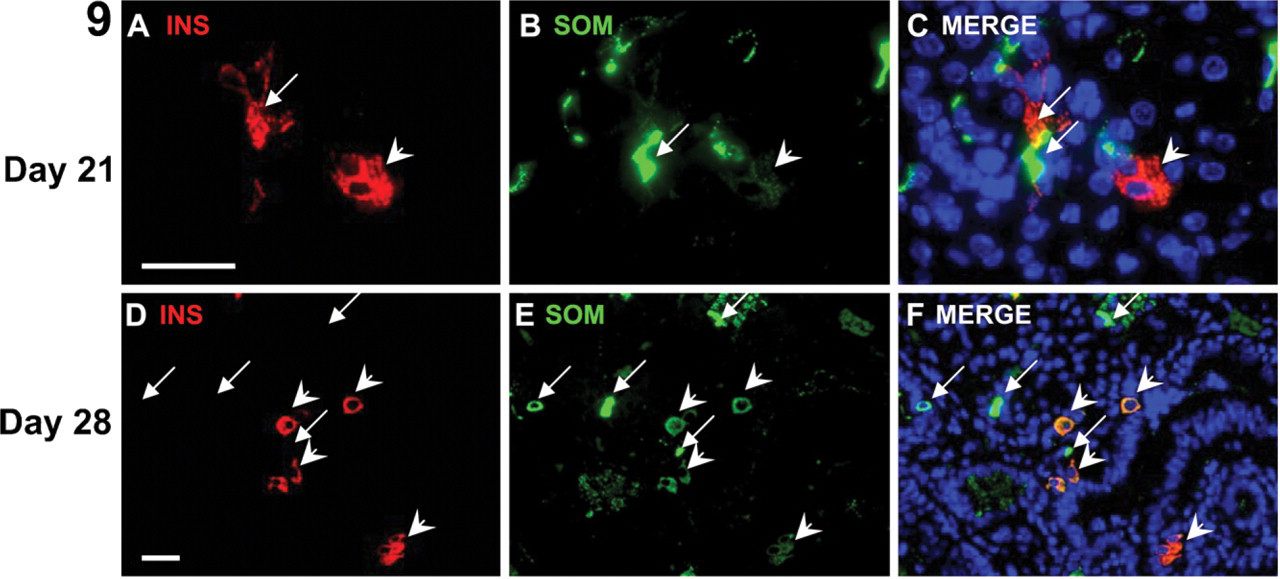
Multihormone expression in endocrine cells. Somatostatin and insulin coexpression in day 21 (
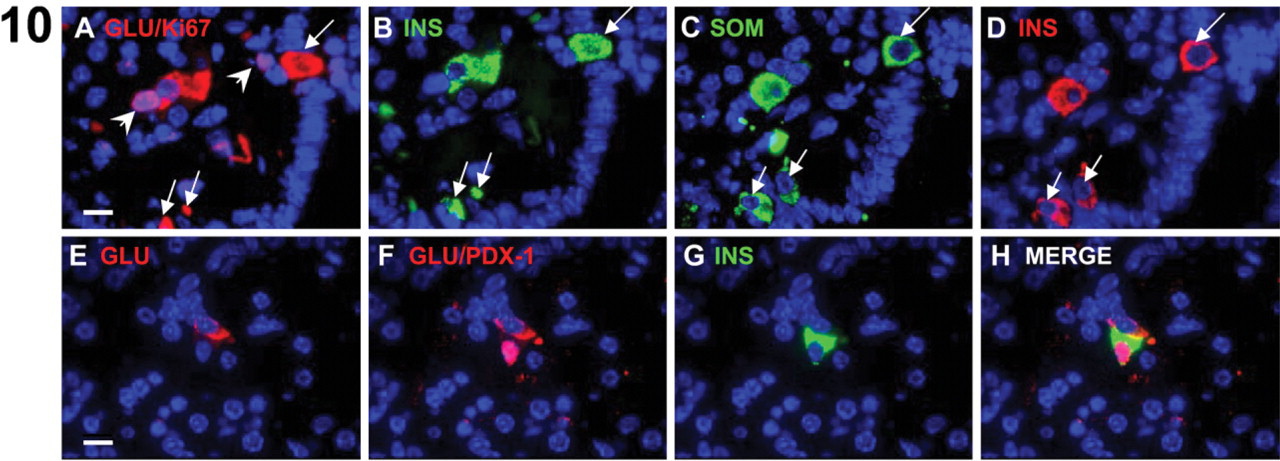
Multihormone expression in endocrine cells. Non-dividing (Ki67-negative) somatostatin, insulin, and glucagon triple-positive endocrine cells are detectable in day 28 foci, along with mono-positive insulin and mono-positive glucagon expressing cells. Two serial sections (
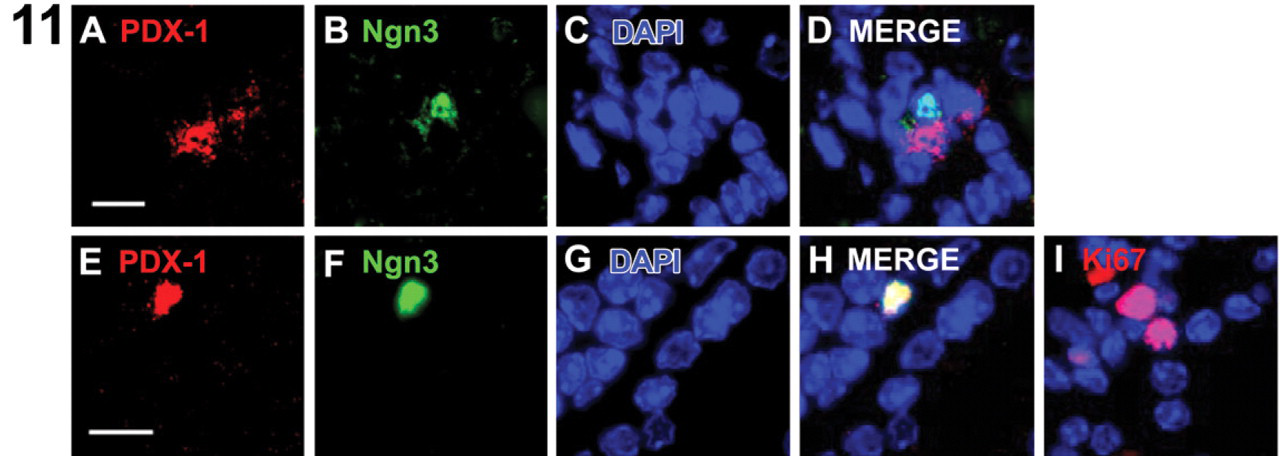
Ngn3 is the earliest pro-endocrine cell marker expressed in the embryonic pancreas. Ngn3 protein was detected in pancreatic foci at both day 21 and day 28. Day 21 (
Comparison of pancreatogenesis in transplanted ES in STZ-injured mice and during fetal development
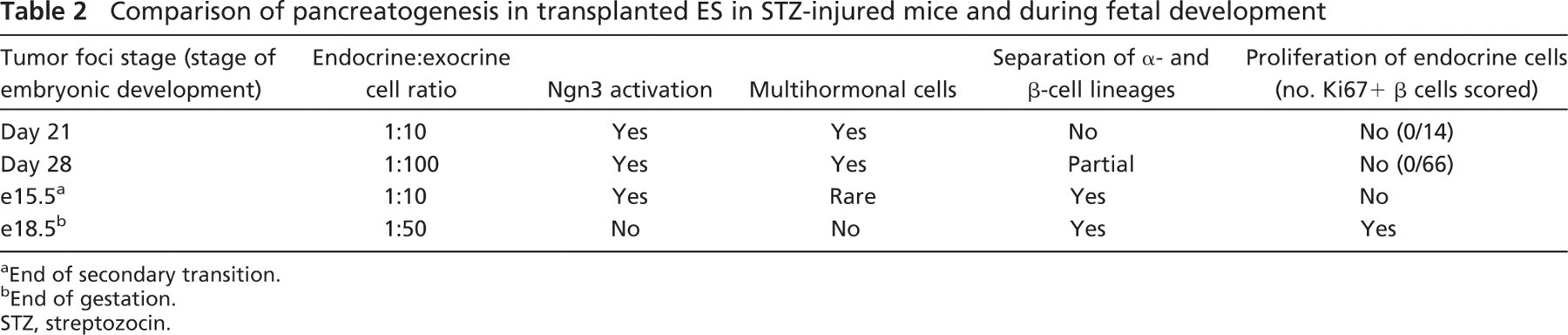
a End of secondary transition.
b End of gestation.
STZ, streptozocin.
The renal capsule is known to support the maturation and survival of fetal islet-like cell clusters (Beattie et al. 1994). In addition, it seems that IP administration of either exendin-4, a glucagon-like peptide analog, or keratinocyte growth factor results in a significant acceleration of β-cell maturation and overall β-cell mass in this system (Movassat et al. 2002, 2003). An important indication from these findings is that renal capsule–transplanted tissue, committed to pancreatic endocrine differentiation, may be susceptible to stimulation by systemic factors.
In conclusion, we identified embryonic-like pancreatogenesis leading to insulin-producing cells in renal capsule–transplanted ES after β cell–specific injury. The data suggest that an inductive environment exists in the renal capsule that seems to be dependent on systemic regenerative stimuli elicited on β-cell destruction. It will be important to examine the effect of elevated blood glucose on endocrine differentiation in this system. Additionally, further studies will be necessary to establish whether other cell types, for example, partially differentiated ES, can undergo pancreatogenesis without teratoma formation, and whether their insulin-producing cell progeny can improve glucose homeostasis in vivo.
Footnotes
Acknowledgements
We thank N. Namatame, Y. Yamamoto, and A. Inoue (Section for Studies on Metastasis, NCCRI) for technical assistance, Professor H. Edlund (University of Umea, Umea, Sweden) for the gift of anti-ISL-1 antiserum, and Dr. P. Serup (Hagedorn Institute, Denmark) for the gift of anti-Nkx6.1 antibody. The anti-Ngn3 monoclonal antibody developed by Professor O. D. Madsen was obtained from the Developmental Studies Hybridoma Bank developed under the auspices of the NICHD and maintained by The University of Iowa, Department of Biological Sciences, Iowa City, IA. This work was supported in part by a Grant-in-Aid for the Third-Term Comprehensive 10-Year Strategy for Cancer Control; Health Science Research Grants for Research on the Human Genome and Gene Therapy from the Ministry of Health, Labor, and Welfare of Japan.
