Abstract
Isolated chicken myoblasts had previously been utilized in many studies aiming at understanding the emergence and regulation of the adult myogenic precursors (satellite cells). However, in recent years only a small number of chicken satellite cell studies have been published compared to the increasing number of studies with rodent satellite cells. In large part this is due to the lack of markers for tracing avian myogenic cells before they become terminally differentiated and express muscle-specific structural proteins. We previously demonstrated that myoblasts isolated from fetal and adult chicken muscle display distinct schedules of myosin heavy-chain isoform expression in culture. We further showed that myoblasts isolated from newly hatched and young chickens already possess the adult myoblast phenotype. In this article, we report on the use of polyclonal antibodies against the chicken myogenic regulatory factor proteins MyoD and myogenin for monitoring fetal and adult chicken myoblasts as they progress from proliferation to differentiation in culture. Fetal-type myoblasts were isolated from 11-day-old embryos and adult-type myoblasts were isolated from 3-week-old chickens. We conclude that fetal myoblasts express both MyoD and myogenin within the first day in culture and rapidly transit into the differentiated myosin-expressing state. In contrast, adult myoblasts are essentially negative for MyoD and myogenin by culture Day 1 and subsequently express first MyoD and then myogenin before expressing sarcomeric myosin. The delayed MyoD-to-myogenin transition in adult myoblasts is accompanied by a lag in the fusion into myotubes, compared to fetal myoblasts. We also report on the use of a commercial antibody against the myocyte enhancer factor 2A (MEF2A) to detect terminally differentiated chicken myoblasts by their MEF2+ nuclei. Collectively, the results support the hypothesis that fetal and adult myoblasts represent different phenotypic populations. The fetal myoblasts may already be destined for terminal differentiation at the time of their isolation, and the adult myoblasts may represent progenitors that reside in an earlier compartment of the myogenic lineage.
T
Studies of avian and mammalian myoblasts have indicated that the myogenic precursors present in developing and mature limb and body wall muscles can be divided into distinct populations on the basis of specific morphological and biochemical traits of the cells identified in culture. These myogenic populations were broadly defined as embryonic myoblasts (appear earlier in embryogenesis), fetal myoblasts (appear later in embryogenesis), and adult myoblasts (satellite cells, present in postnatal and adult muscle) (reviewed in Cossu and Molinaro 1987; Stockdale 1992; Yablonka–Reuveni 1995a). In the chicken, the embryonic myoblasts are most abundant on embryonic Day 5, the fetal myoblasts are most abundant between embryonic Days 8 and 12, and the adult myoblasts appear at the final stages of embryogenesis and are the only myoblasts that persist through adulthood (Stockdale 1992; Yablonka–Reuveni 1995a). The satellite cells are the only well-characterized myogenic precursors in the postnatal and adult muscle and have been thought to be the sole source of the adult myoblasts (Yablonka–Reuveni 1995a, b). Recent studies have raised the possibility that a different class of stem cells is present in the adult muscle that can give rise to various cell types including myoblasts (reviewed in Seale and Rudnicki 2000). The frequency and in vivo myogenic potential of the latter cells are still uncertain, and they are not under consideration in this study.
In our previous studies, the distinctions between adult and fetal chicken myoblasts were based on the detection of different schedules of myosin isoform expression during differentiation in cultures of isolated cells. Myoblasts displaying the adult phenotype were already present in the muscle during late stages of embryonic development (Hartley et al. 1991, 1992). These so-called adult myoblasts were the dominant myogenic precursors in muscles from young or adult chickens, regardless of their proliferative state in vivo (Hartley et al. 1991, 1992; Hartley and Yablonka–Reuveni 1992; Yablonka–Reuveni 1995a). In this study we examined whether distinctions between the adult and fetal populations might already be manifested at an earlier stage of the myogenic program preceding sarcomeric myosin expression. Specifically, we examined the expression patterns of the chicken myogenic regulatory factor (MRF), proteins MyoD and myogenin. The four known MRFs (Myf5, MyoD, myogenin, and MRF4) form the basic helix–loop–helix family of skeletal muscle-specific transcription factors. The MRFs are involved in the specification of the skeletal muscle lineage during early embryogenesis and in the control of cell cycle withdrawal and muscle-specific gene expression (reviewed in Ludolph and Konieczny 1995; Megeney and Rudnicki 1995). The immunocytochemical analysis of MRF protein expression by rodent myoblasts is well documented and has provided a direct means for the phenotypic characterization of muscle precursor cells as they undergo myogenesis in culture and in vivo (Yablonka–Reuveni and Rivera 1994, 1997; Anderson et al. 1998; Cooper et al. 1999). In cultures of rodent satellite cells, MyoD protein expression is typically seen before differentiation coinciding with proliferation, whereas myogenin expression begins together with, or just shortly before, sarcomeric myosin (Smith et al. 1993; Yablonka–Reuveni and Rivera 1994, 1997; Yablonka–Reuveni et al. 1999; Kästner et al. 2000).
Chicken homologues of all four mammalian MRFs have been described (Lin et al. 1989; Fujisawa–Sehara et al. 1992; Saitoh et al. 1993). However, the status of MRF expression by avian satellite cells has been studied only at the mRNA level employing Northern analysis of cultured cells (Halevy et al. 1994). A direct single-cell analysis of MRF expression by chicken satellite cells is especially desirable in studies on the regulation of myoblast proliferation. The myogenic cells are typically co-isolated with the non-myogenic cells present in the muscle (Yablonka–Reuveni and Nameroff 1987; Yablonka–Reuveni et al. 1987, 1988). These non-myogenic cells can often respond to the same growth factors as the myogenic cells, thus complicating the determination of the effect of the growthpromoting agents on the myogenic cells specifically. Unlike rodent myoblasts, proliferating chicken myoblasts cannot be monitored by desmin expression because only a small number of chicken myoblasts express desmin before terminal differentiation (Yablonka–Reuveni and Nameroff 1986, 1990; Kaufman and Foster 1988). The lack of appropriate immunoreagents for single-cell analysis of chicken myoblasts has affected the progress made in the understanding of satellite cell regulation in poultry.
Here we report on the use of specific antibodies raised against chicken MyoD and myogenin for the analysis of myogenesis in cultured fetal and adult chicken myoblasts. We also report on the utilization of a commercially available antibody raised against the human MEF2A for tracing nuclei of differentiating myoblasts and myotubes in chicken myogenic cultures. MEF2A is a member of the myocyte enhancer factor 2 family of transcription factors. Members of the MEF2 and MRF families act cooperatively during the activation of muscle-specific genes (Molkentin et al. 1995; Molkentin and Olson 1996). We conclude that the adult and fetal myoblasts exhibit different expression schedules of MyoD and myogenin proteins before entering the differentiated, myosin + state. These differences are probably a reflection of the state of the cells along the myogenic program at the time of their isolation.
Materials and Methods
Isolation and Culture of Fetal and Adult Chicken Myoblasts
Fetal and adult myoblasts were isolated from the pectoralis muscle of 11-day-old embryos and 3-week-old chickens, respectively. The source of the embryonated eggs and posthatch chickens (White Leghorn) was detailed previously (Hartley et al. 1992). Single cells were released from the muscles by enzymatic digestion and further enriched for myogenic cells by Percoll density centrifugation essentially as described in our earlier publications (Yablonka–Reuveni et al. 1987; Hartley et al. 1991, 1992). Cells were plated on tissue culture plates precoated with 2% gelatin (Sigma; St Louis, MO), using our standard medium for chicken myoblast cultures [Eagle's minimal essential medium (MEM) containing 10% horse serum, 5% chicken embryo extract, and penicillin and streptomycin at 105 U/liter each). The horse serum (Sigma) was pre-selected for maximal support of growth of chicken myogenic clones. The chicken embryo extract was prepared from 10- and 11-day-old embryos according to previously published procedures, using the entire embryo (O'Neill and Stockdale 1972; Yablonka–Reuveni 1995b).
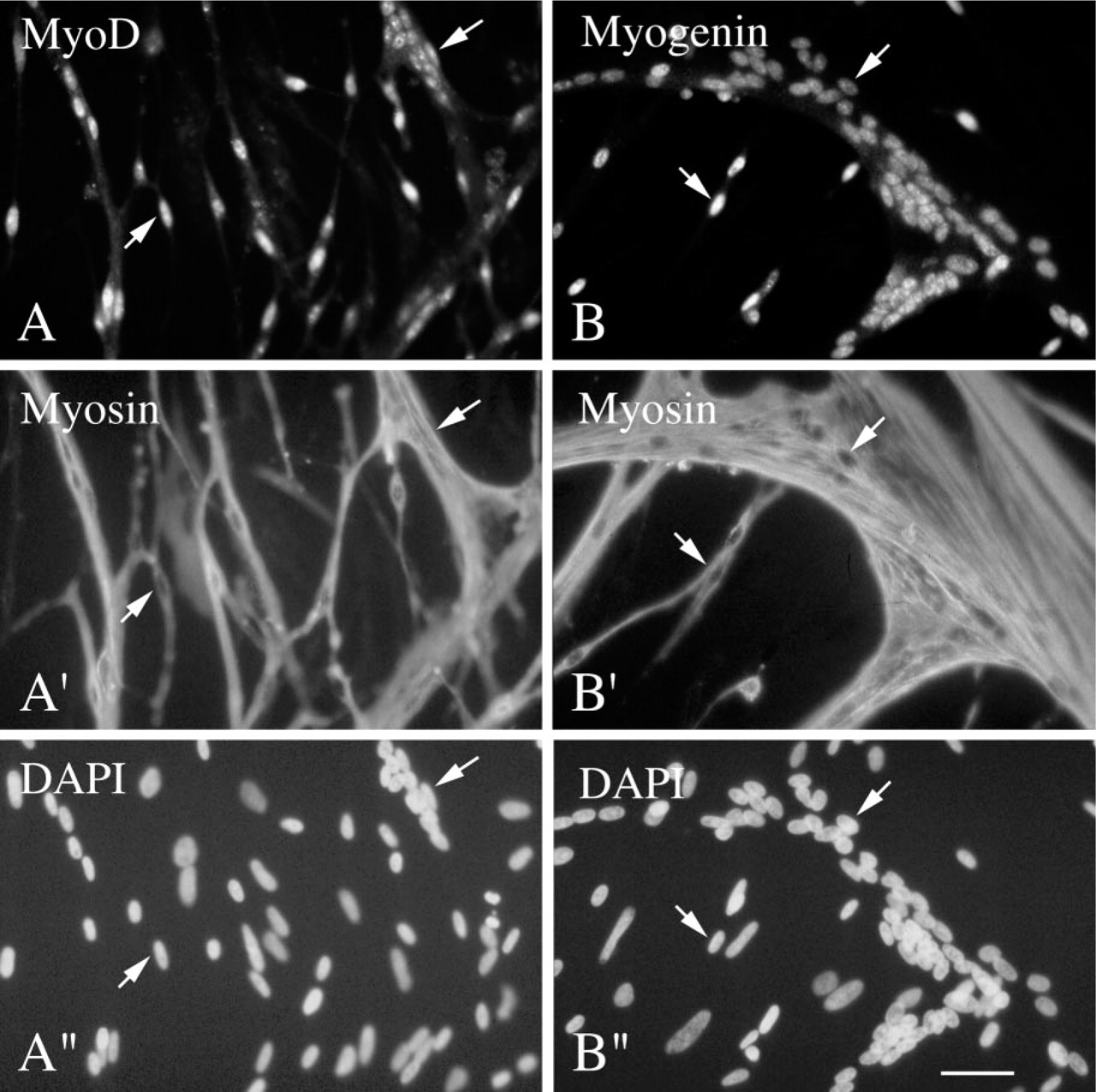
Immunofluorescent micrographs of myogenic cultures reacted with antibodies against MyoD and myosin (left column) or myogenin and myosin (right column). Cells were isolated from 11-day-old chicken embryos and cultured for 3 days in growth medium. Cultures were reacted via double immunofluorescence with a rabbit polyclonal antibody against MyoD along with a mouse monoclonal antibody against sarcomeric myosin (
Primary Antibodies
The antibodies against chicken MyoD and myogenin were generated by immunizing rabbits with the hexahistidine (6His)-tagged full-sized chicken proteins made in E. coli. Proteins eluted from NiTA resin in 6.4 M urea/200 mM histidine, pH 7.4, were mixed with adjuvant and injected subdermally every 2–3 weeks until the titer was satisfactory. The specificity of the antibodies was originally tested by immunoprecipitation of proteins made in the reticulocyte lysate cell-free translation system (Paterson and Roberts 1981). The use of the anti-chicken MyoD was briefly mentioned in earlier studies on the emergence of myogenic cells during chicken development (George–Weinstein et al. 1997; Link and Nishi 1998).
The rabbit polyclonal antibody against MEF2A was purchased from Santa Cruz Biotechnology (Santa Cruz, CA). The use of this antibody to detect MEF2A in extracts of C2 cultures via immunoblotting was described in Molkentin et al. (1996). By analyzing mouse- and rat-derived myogenic cultures via immunofluorescence, we showed that the anti-MEF2A reacts with nuclei of mononucleated cells that enter the myogenin + state and with all nuclei in myotubes (Yablonka–Reuveni and Rivera 1997; Yablonka–Reuveni et al. 1999; Kästner et al. 2000).
The monoclonal antibody against sarcomeric myosin (MAb MF20; Bader et al. 1982) was obtained from the Developmental Studies Hybridoma Bank (University of Iowa; Ames, IA). The antibody, originally prepared against chicken skeletal muscle myosin, recognizes all isoforms of chicken sarcomeric myosin heavy chain (Bader et al. 1982) and reacts with all differentiated myoblasts and myotubes in cultures of fetal and adult chicken myoblasts (Hartley et al. 1991).
Immunolabeling of Cultured Cells
Single- and double-immunofluorescence analyses of methanol-fixed cultures were performed as previously described (Yablonka–Reuveni and Rivera 1994). The cultures were rinsed with MEM at room temperature (RT), fixed for 10 min at 4C with ice-cold 100% methanol, air-dried for 10–20 min, and then kept overnight at 4C in sterile Tris-buffered saline containing normal goat serum (TBS-NGS; 0.05 M Tris, 0.15 M NaCl, 1% normal goat serum, pH 7.4). Cultures were rinsed with Tris-buffered saline containing Tween-20 (TBS-TW20; 0.05 M Tris, 0.15 M NaCl, 0.05% Tween-20, pH 7.4). The cultures were then reacted with the primary antibodies for 1 hr at RT, followed by an overnight incubation at 4C. This was followed by a rinse with TBS-TW20 and a 1–2-hr incubation at RT with the secondary antibodies (Organon–Technika Cappel; Downingtown, PA) diluted 1:500 with TBS-NGS. A rhodamine-conjugated goat anti-rabbit IgG was used for the rabbit polyclonal antibodies against MyoD, myogenin, and MEF2A, and a fluorescein-conjugated goat anti-mouse IgG was used for the mouse MAb against sarcomeric myosin. The two secondary antibodies were added together in double-immunofluorescence studies. The cultures were further stained with DAPI to allow detection of all nuclei (Hartley et al. 1991), rinsed with TBS-TW20, and mounted in VECTASHIELD mounting medium (Vector Laboratories; Burlingame, CA). For controls, we used pre-immune rabbit sera. Culture of chicken fibroblasts did not react with the antibodies.
Distribution of cells positive for MyoD or myogenin in cultures of fetal myoblasts
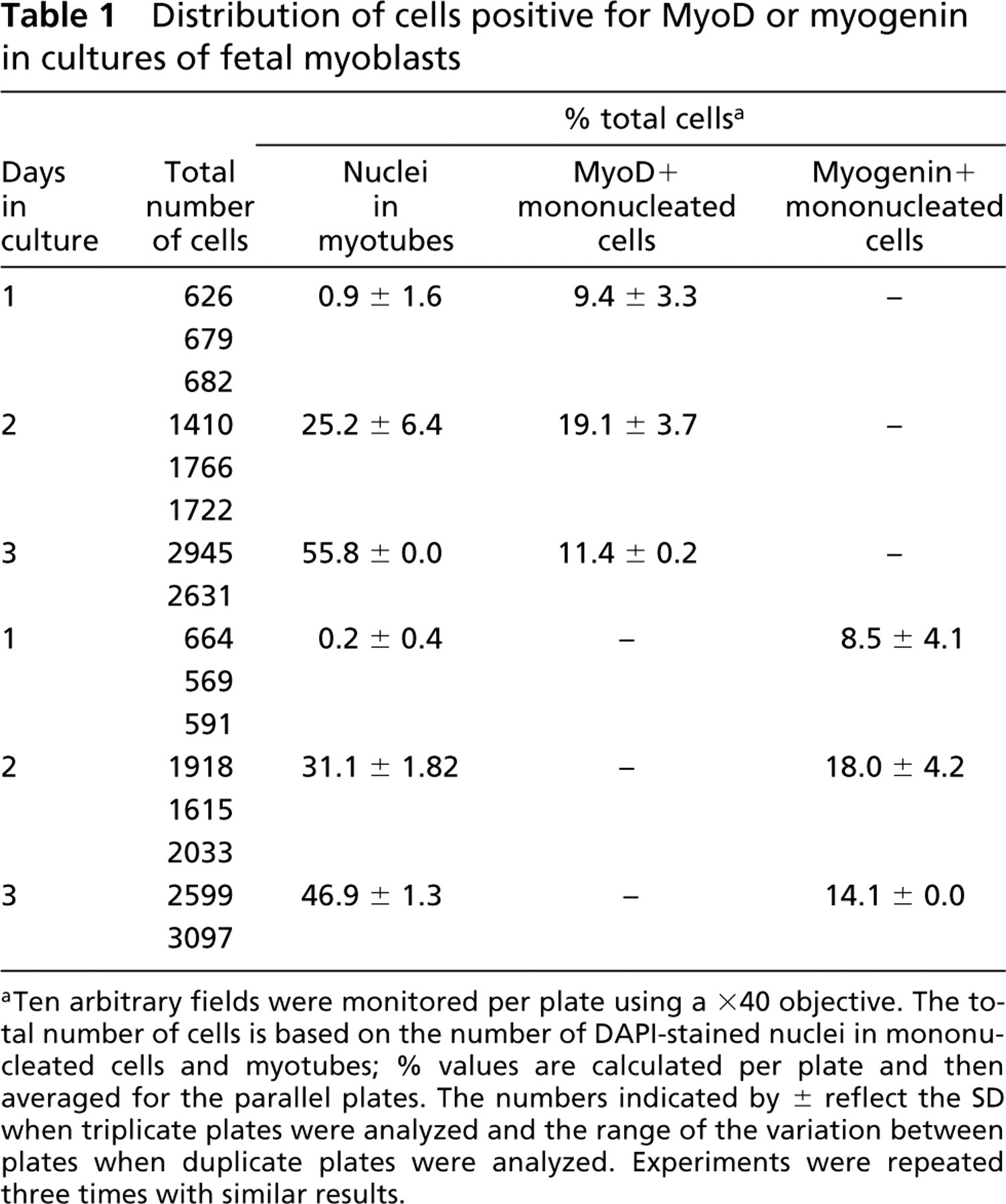
aTen arbitrary fields were monitored per plate using a ×40 objective. The total number of cells is based on the number of DAPI-stained nuclei in mononucleated cells and myotubes; % values are calculated per plate and then averaged for the parallel plates. The numbers indicated by ± reflect the SD when triplicate plates were analyzed and the range of the variation between plates when duplicate plates were analyzed. Experiments were repeated three times with similar results.
Quantification of Positive Cells
Quantification of the number of cells that fused into myotubes and the number of mononucleated cells reacting with the different antibodies was performed by monitoring a minimum of 10 arbitrary fields per culture plate using a ×40 objective. In some instances, indicated in the Results, more than 10 fields were monitored to ensure that at least 500 DAPI-stained nuclei were analyzed per plate. The values of DAPI-stained nuclei included nuclei in mononucleated cells and myotubes. Unless otherwise noted, the frequency of nuclei in myotubes or of mononucleated cells positive for different antibodies was first determined for each plate and then averaged for the parallel plates, as shown in Results. Error values reflect the range of variation between samples when duplicate cultures were analyzed and the SD when triplicate plates were analyzed.
Distribution of myosin+ cells in the MyoD+ or myogenin+ cell populations in cultures of fetal myoblasts

aTen arbitrary microscopic fields in individual plates were analyzed using a ×40 objective. Only mononucleated cells were analyzed. Myotubes are always positive for the myosin (MF20) antibody.
Results
Immunostaining of Cultured Cells with Antibodies Against MyoD, Myogenin, and Myosin
Figure 1 shows micrographs of myogenic cultures from 11-day-old chicken embryos, stained via double immunofluorescence with the polyclonal antibody against MyoD (Figure 1A) or the polyclonal antibody against myogenin (Figure 1B), along with the monoclonal antibody against myosin (Figures 1A′ and 1B′, respectively). Parallel micrographs of DAPI-stained nuclei are shown in Figures 1A″ and B″, respectively. Micrographs are shown for culture Day 3 when both single cells and multinucleated myotubes are present. The nuclei in some of the mononucleated cells and in all myotubes are positive for MyoD and myogenin.
Distribution of Cells Expressing MyoD and Myogenin in Fetal Cultures
The quantification of MyoD+ and myogenin + cells in myogenic cultures from 11-day-old chicken embryos is summarized in Table 1. The cultures, collected every 24 hr, were scored for the total number of cells, the number of cells that fused into myotubes (nuclei in myotubes), and the number of mononucleated cells labeled with each antibody. By Day 1 the cultures were essentially devoid of myotubes and about 9% of the mononucleated cells were already positive for MyoD or myogenin. By Day 2 there was a robust increase in the number of cells that fused into myotubes (25–31% of total cells) and this parameter further increased by Day 3 (47–56% of total cells). As in Day 1 cultures, the number of mononucleated cells positive for MyoD or myogenin was similar in Day 2 cultures (19% and 18%, respectively) and in Day 3 cultures (11% and 14%, respectively). A double-staining analysis with the MAb against myosin in combination with the anti-MyoD or anti-myogenin Ab demonstrated that most of these MyoD+ or myogenin + mononucleated cells were negative for myosin (Table 2).
Distribution of Cells Expressing MyoD and Myogenin in Adult Cultures
The quantification of MyoD+ and myogenin + cells in myogenic cultures from 3-week-old chickens is summarized in Table 3. The cultures were co-reacted with the anti-MyoD or anti-myogenin Ab along with the MAb against myosin. The immunostaining images of positive cells were identical to those shown in Figure 1 for the fetal cultures. The cultures, collected every 24 hr, were scored for the total number of cells, the number of cells that fused into myotubes, and the number of mononucleated cells that were singly or doubly labeled with the antibodies. By Day 1 the cultures were essentially devoid of myotubes or mononucleated cells positive for any of the antibodies examined. By Day 2 the cultures demonstrated a significant increase in the number of MyoD+ or myogenin+ mononucleated cells. These positive cells were mostly negative for myosin, indicating that they had not yet completed the differentiation process. Furthermore, by culture Day 2 there was a higher number of MyoD+/myosin– cells compared to myogenin+/myosin– cells (37% vs 21%, respectively), indicating that the cells first become positive for MyoD and then for myogenin. The Day 2 cultures were still devoid of myotubes but some myosin + cells began to emerge, adding up to about 3% of the total cells. By culture Day 3 there was a robust increase in the number of cells that fused into myotubes, but a pool of MyoD+/myosin– or myogenin+/myosin– mononucleated cells was still evident in the Day 3 cultures. The number of all myosin+ mononucleated cells in the Day 3 cultures was low and was similar to that present in Day 2 cultures. As in the fetal cultures, the majority of the MyoD+ or myogenin+ cells in the adult cultures were negative for myosin.
Distribution of positive cells in cultures of adult myoblasts reacted by double immunofluorescence with antibodies against MyoD and myosin or myogenin and myosin

aTwenty arbitrary fields were monitored for the Day 1 plates and 10 fields were monitored for the Day 2 and Day 3 plates using a ×40 objective. The total number of cells is based on the number of DAPI-stained nuclei in mononucleated cells and myotubes; % values are calculated per plate and then averaged for the duplicate plates. The numbers indicated by ± reflect the range of the variation between the duplicate plates. Experiments were repeated three times with similar results.
Distribution of MEF2A+ Cells in Chicken Myogenic Cultures
Figure 2 shows micrographs of myogenic cultures from 11-day-old chicken embryos stained via double immunofluorescence with the polyclonal antibody against MEF2A along with the monoclonal antibody against myosin. Micrographs are shown for culture Day 3 when both single cells and multinucleated myotubes are present. MEF2A+ nuclei were typically seen in myosin+ mononucleated cells and in all myotubes. The double immunostaining images of positive cells in cultures of adult myoblasts were the same as those shown in Figure 2.
Discussion
Generation of antibodies against chicken MyoD and myogenin has enabled us to characterize the adult and fetal myoblasts before they fully differentiated into cells expressing sarcomeric myosin. Compared to cultures of fetal myoblasts that displayed a rapid expression of MyoD and myogenin combined with a rapid fusion into myotubes, adult cultures displayed a lag in MyoD and myogenin expression combined with a delayed fusion into myotubes. Furthermore, the MyoD + or myogenin + cells appeared at a similar frequency at each time point analyzed in the fetal cultures, suggesting that the fetal myoblasts express both MRF proteins. On the other hand, in the adult cultures there was a distinct lag in the onset of expression of myogenin compared to MyoD, indicating that the cells first express MyoD and then myogenin. In concordance with earlier studies discussed below, we interpret the findings on the different kinetics of MyoD+ and myogenin + cells by cultured adult and fetal myoblasts as evidence for the different states of adult and fetal myoblasts along the myogenic lineage.
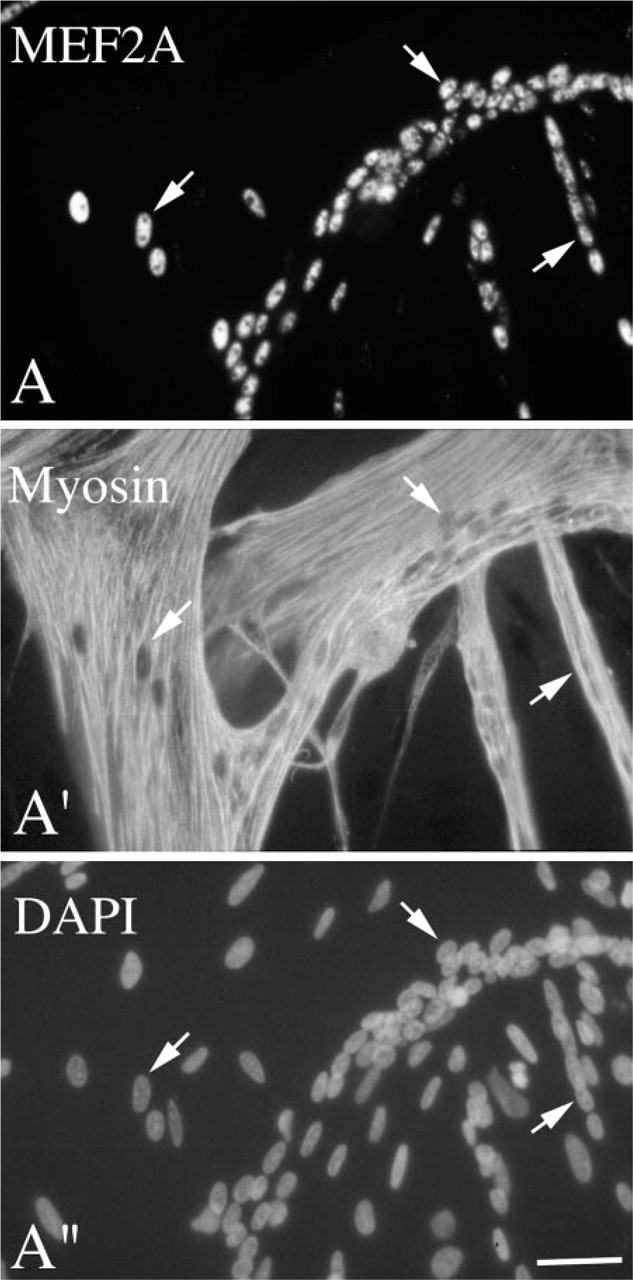
Immunofluorescent micrographs of myogenic cultures reacted with antibodies against MEF2A and myosin. Cells were isolated from 11-day-old chicken embryos and cultured for 3 days in growth medium. Cultures were reacted via double immunofluorescence with a rabbit polyclonal antibody against MEF2A (
Clonal analyses of individual chicken myoblasts have suggested that the majority of the fetal myoblasts are in a “committed cell compartment” destined to divide only several times before terminal differentiation. In addition, a small proportion of the myoblasts from this fetal stage was identified as stem cells. These so-called stem cells undergo multiple cell divisions, able to produce self-renewed stem cells along with committed cells (Quinn et al. 1984, 1985, 1989; Nameroff and Rhodes 1989). The latter studies further concluded that the number of stem cells increases in advanced stages of development. Myoblasts isolated from the adult chicken have been thought to reside primarily in the stem cell compartment because they mostly give rise to large myogenic clones (Yablonka–Reuveni et al. 1987). The age-dependent difference between the frequency of committed cells vs stem cells in the cell preparation was also reflected in the rapidity of formation of myotubes, which was delayed in cultures from late chick embryos and postnatal animals (Quinn et al. 1989; Hartley et al. 1991, 1992). It should be noted that the term “stem cells” used by Quinn and colleagues refers to cells at an earlier state of the myogenic lineage (capable of self renewal and giving rise to the committed myoblasts). These so-called stem cells are not in any way equivalent to the recently identified tissue stem cells mentioned in the Introduction that might be able to give rise to various cell types including myoblasts.
The present analysis has identified a molecular distinction between fetal and adult chicken myoblasts that is displayed by the cells even before they enter the phase of differentiation and distinct expression of muscle-specific sarcomeric myosins previously reported (Hartley et al. 1991, 1992). The presence of primarily committed cells in the fetal population may be reflected by the rapid expression of both MyoD and myogenin. The stem cell nature of the adult myoblasts might be linked to the lag in acquiring the MyoD + state. This possibility is further supported by our studies of satellite cells from mice lacking MyoD. The MyoD–/– satellite cells remained proliferative for a longer time and their transition into the myogenin+ state was drastically reduced in cultures of isolated myofibers (Yablonka–Reuveni et al. 1999). Hence, an ability of the cell to delay or escape MyoD expression might be important for its capacity to remain proliferative for a longer time. The role of MyoD in enforcing cell cycle withdrawal and differentiation has been previously demonstrated in studies of myogenic cell lines (Halevy et al. 1995). Comparing growth aspects of adult and fetal chicken myoblasts, we showed that the rate of differentiation of the adult myoblasts could be accelerated when the cells were maintained in serum-poor medium but that the differentiation of fetal myoblasts could not be influenced (Yablonka–Reuveni 1995b). These findings further indicate that the myogenic state of adult myoblasts is “plastic” and refractive to influences of extracellular agents, whereas the fetal myoblasts are already at the state that can not be further accelerated towards differentiation.
We also demonstrate in the present study that a commercial antibody against human MEF2A protein reacts with differentiated chicken myoblasts. The pattern of MEF2A expression in myosin + chicken myoblasts and myotubes is identical to its temporal expression pattern in rodent myoblasts (Yablonka–Reuveni and Rivera 1997; Yablonka–Reuveni et al. 1999; Kästner et al. 2000).
In conclusion, the availability of suitable antibodies has enabled us to demonstrate that the transition along the MyoD–myogenin–MEF2A pathway in adult chicken myoblasts follows the same protein expression program as seen in mouse and rat satellite cells (Yablonka–Reuveni and Rivera 1994, 1997; Yablonka–Reuveni et al. 1999; Kästner et al. 2000). The immunoreagents described in the study can now be utilized in studies on the regulation of myoblast proliferation and the effect of growth factors and cytokines on the phenotypic compartments during myogenesis of avian myoblasts.
Footnotes
Acknowledgements
ZY-R was supported by grants from the National Institutes of Health (AG13798), the Cooperative State Research Service–United States Department of Agriculture (Agreement No. 99-35206-7934) and the United States–Israel Binational Agricultural Research and Development Fund (Agreement No. IS-3093-99).
We thank Priscilla Natanson and Anthony Rivera for excellent technical support.
