Abstract
Successful embryo implantation depends on the ability of the trophoblast cells to invade the endometrium and the receptivity of the endometrium. Unlike tumor invasion, trophoblast invasion is spatio-temporaly restricted. Transforming growth factor (TGF)-β is a key inhibitory factor in the invasion of early trophoblast cells. Smad ubiquitination regulatory factor 2 (Smurf2), a HECT type E3 ubiquitin ligase, is an important regulator of the TGF-β signaling pathway, targeting TGF-β receptors and various Smads for proteasome-mediated degradation. In this context, we wished to determine whether Smurf2 has a physiological role during embryo implantation, especially in trophoblast invasion. We examined the spatio-temporal expression of Smurf2 in human placental villi and the function of Smurf2 in trophoblast cell migration and invasion in a model system involving a human extravillous trophoblast cell line, HTR-8/SVneo. Results from RT-PCR and immunohistochemical studies showed that expression of Smurf2 in placental villi was the highest during the first trimester and decreased as the pregnancy progressed. Overexpression of Smurf2 in HTR-8/SVneo cells reduced TGF-β type I receptor levels, and enhanced cell migration and invasion. Conversely, RNA interference—mediated downregulation of Smurf2 resulted in a significant increase in TGF-β type I receptor protein levels. However, the levels of Smad2, another potential target of Smurf2, remained unchanged. In conclusion, the present study suggests that Smurf2 promotes trophoblast cell migration and invasion, and this function may involve downregulation of TGF-β type I receptor.
Transforming growth factor (TGF)-β is involved in a wide range of cellular processes, including growth, differentiation, apoptosis, migration, invasion, and self-renewal (Massague and Chen 2000; Chang et al. 2002). A TGF-β ligand exerts its function by binding and activating cell surface type II and then type I receptors (Massague 1998). Upon phosphorylation by the type II receptor (TGF-βR II), the type I receptor (TGF-βR I) subsequently phosphorylates and activates Smad2 or Smad3, the intracellular signaling molecules for TGF-β (Miyazawa et al. 2002). Phosphorylated Smad2 or −3 is released from the receptors and forms oligomeric complexes with common-partner Smad, Smad4, and translocates into the nucleus to regulate the transcription of target genes. TGF-β, produced mainly by the decidua and to a minor extent by the placenta, is one of the putative inhibitors of trophoblast invasion during the first trimester of human pregnancy (Irving and Lala 1995).
Selective proteolytic degradation by the ubiquitin—proteasome pathway plays an important role in numerous biological processes (Hershko and Ciechanover 1998). Protein ubiquitination is catalyzed by the sequential action of three enzymes: a ubiquitin activation enzyme (E1), ubiquitin conjugation enzymes (E2), and ubiquitin ligases (E3) (Hershko and Ciechanover 1998). E3 determines the specificity of the substrate, and was thought to be the primary regulator of in vivo protein ubiquitination (Xu et al. 2008). There are more than 500 E3s in mammalian cells, which fall into two categories, HECT domain E3s and RING finger and U-box E3s (Chen and Matesic 2007; Wiesner et al. 2007).
Smad ubiquitination regulatory factor 2 (Smurf2) is a C2-WW-HECT-domain E3 that plays a pivotal role in regulating TGF-β signaling by specifically targeting Smad2 and TGF-β type I receptor (Kavsak et al. 2000; Lin et al. 2000) for ubiquitin-mediated degradation (Zhu et al. 1999; Kavsak et al. 2000; Wiesner et al. 2007). Smurf2 is highly expressed in esophageal squamous cell carcinoma, which is correlated with a lower level of Smad2 phosphorylation and inhibited cell survival (Fukuchi et al. 2002). Smurf2 is also able to inhibit TGF-β signaling, which otherwise stimulates the proliferation of fibroblasts, resulting in a senescent phenotype (Zhang and Cohen 2004). We have previously cloned the partial coding sequence of Smurf2 in the rhesus monkey, which shares 99% identity with its human homolog (Yang et al. 2007). We have also detected strong signals of Smurf2 mRNAs and proteins in the trophoblastic column and trophoblastic shell of the placental villi of the rhesus monkey, suggesting that Smurf2 may play a role in trophoblastic cell invasion and placentation (Yang et al. 2007).
In this study, continuing our previous work, we first investigated the expression of Smurf2 in human placenta villi throughout pregnancy and the effect of overexpression of Smurf2 on the invasion of a human EVT cell line, HTR-8/SVneo. Our results clearly demonstrated that overexpression of Smurf2 significantly increased the invasion and migration of HTR-8/SVneo cells. Furthermore, overexpression of Smurf2 down-regulated the expression of TGF-β type I receptor, but not of Smad2. Knocking down Smurf2 by RNAi significantly upregulated the protein expression of TGF-β type I receptor.
Materials and Methods
Tissue Collection
Informed patient consent was obtained in all cases. Collecting and processing of human placenta tissues were performed in accordance with the permission of the ethical committee of Peking University Third Hospital and Xuanwu Hospital Capital Medical University. Normal placenta tissues of the first trimester (6–9 weeks of gestation), second trimester (23–26 weeks of gestation), and term were collected from the Department of Obstetrics and Gynecology, Peking University Third Hospital and Department of Obstetrics and Gynecology, Xuanwu Hospital Capital Medical University. Placentas of the first trimester were from women who underwent legal abortion. The placentas of the second trimester were from women who had normal pregnancies but had to stop gestation for some reason. Term placentas were from uncomplicated, normal term pregnancies. At least three placentas were collected for each time point. The villi of placentas of the first and second trimester were washed twice in cold PBS and divided into two parts. One part was fixed in 4% paraformaldehyde for 24 hr at 4C and kept in 70% ethyl alcohol until processing and paraffin embedding; the other part was immediately placed in dry ice and stored at —80C for future RNA extraction. The villi of term placentas were fixed in 4% paraformaldehyde for 24 hr at 4C and processed for paraffin embedding. This work was approved by the Institute of Zoology, Chinese Academy of Sciences.
Cell Culture
HTR-8/SVneo cells are first-trimester human trophoblasts immortalized following transfection with a gene encoding the simian virus large T antigen (generously provided by Dr. Benjamin K. Tsang of Ottawa Health Research Institute, Canada). They share various phenotypic similarities with the non-transfected parent HTR-8 cells and exhibit a high proliferative index. HTR-8/SVneo cells were grown in RPMI 1640 medium supplemented with 10% FBS and 1% penicillin-streptomycin (Invitrogen; Gaithersburg, MD), and cultured in 95% air and 5% CO2 at 37C. For treatment, cells were exposed to 10 ng/ml TGF-β1 (PEPROTECH EC Ltd.; London, United Kingdom) for 24 hr.
Plasmid Construct and Transfection
pCMV5B-Flag-tagged Smurf2 was from Dr. Jeffrey L. Wrana (University of Toronto). Full-length Smurf2 (∼2.3 kb) was digested from pCMV5B-Flag-tagged Smurf2 and subcloned into pcDNA3.1/zeo(+) vector. Cells (4.0 × 105) were plated in 60-mm dishes, and 30–50% confluence was achieved before transfection. Following the manufacturer's instructions, pcDNA3.1/Zeo (+)-Smurf2 or pcDNA3.1/Zeo (+) empty vector was transfected into cells with Effectene Transfection Reagent (QIAGEN, Inc.; Valencia, CA) (Wang et al. 2006). Zeocin (200 mg/ml; Invitrogen) was applied to select Zeocin-resistant cells.
Small Interfering RNA (siRNA) Experiment
Three siRNAs (siRNA-1, siRNA-2, siRNA-3) specifically targeting Smurf2 mRNA were synthesized by Borui Biotechnology Corp. (Guangzhou, China). The sequences of these three siRNAs are: 1, cca gua ucu aaa cca uau a dT dT dT dT ggu cau aga uuu ggu aua u; 2, caa cga cau cga aau uac a dT dT dT dT guu gcu gua ucu uua aug u; 3, gaa cca auu gaa aga cca a dT dT dT dT cuu ggu uaa cuu ucu ggu u. Control siRNAs with fluorescence were also provided by this company. HTR-8/SVneo cells were transfected with 50 nM siRNA using Lipofectamine 2000 transfection reagent (Invitrogen), following the manufacturer's recommendations. Transfected cells were then cultured and assayed at 48 hr post-transfection to check the downregulation of Smurf2 by Western blotting.
Total RNA Extraction and RT-PCR
Total RNA was extracted using TriZol reagent as advised by the manufacturer (Invitrogen), and RNA concentrations were determined spectrophotometrically at 260 nm. Two μg of RNA was reverse transcribed using Superscript II reverse transcriptase (Invitrogen) with oligo (dT) as primers. PCR was performed using the following primers: Smurf2, sense: 5′ GTT GTG ATG GGT TCT GAT TC3′; antisense: 5′CAC CAA TGG CAA AAG GCT 3′; GAPDH, sense: 5′AGC CAC ATC GCT CAG ACA C3′; antisense: 5′TGG ACT CCA CGA CGT ACT C3′; Annealing temperatures for Smurf2 and GAPDH were 57C and 55C, respectively. Amplification cycles were 32 for Smurf2 and 23 for GAPDH.
Immunohistochemistry
Immunohistochemistry was performed using the Histo-stain-Plus Kit (Zhongshan Corp.; Beijing, China) as recommended by the manufacturer. Briefly, sections (5 μm) were deparaffinized in xylene and rinsed in ethyl alcohol and a graded series of ethyl alcohol in water. Antigen retrieval was performed by placing the slides in boiling citric acid buffer (10 mM citrate sodium and 10 mM citric acid) for 15 min. The tissue sections were then incubated at room temperature with 3% H2O2 in methanol for 15 min to quench endogenous peroxidase. After being blocked by normal goat serum for 30 min, the sections were sequentially incubated with anti-Smurf2 (Upstate Biotechnology; Lake Placid, NY) at 4C overnight and biotinylated secondary antibody for 30 min before detection with DAB (Zhongshan Corp.). Sections were rinsed with distilled water, counterstained with hematoxylin, and mounted with histomount (Zymed Laboratories, Inc.; San Francisco, CA). Controls were performed by replacing the primary antibody with normal IgG or omitting the secondary antibody. No specific staining was observed in control sections.
Matrigel Invasion and Cell Migration Assays
Invasion of HTR-8/SVneo cells was measured in Matrigel-coated (Becton Dickinson; Franklin Lakes, NJ) transwell inserts (6.5 mm; Costar, Cambridge, MA) containing polycarbonate filters with 8-μm pores. The inserts were coated with 50 μl of 1 mg/ml Matrigel matrix according to the manufacturer's recommendations. Briefly, 1 × 105 cells in 200 μl RPMI 1640 medium without FBS were plated into the upper chamber, whereas 800 μl of medium with 10% FBS was added to the lower well. Cells were cultured at 37C for 24 hr. Non-invaded cells on top of the transwell were scraped off with a cotton swab. The filters with invaded cells attached were washed with PBS, fixed in methanol for 10 min, and stained with hematoxylin and eosin. The number of invaded cells in the lower well was counted under a light microscope in 10 random fields at a magnification of X200. Mean cell numbers in each field were used for statistical analyses. Each experiment was performed in triplicate. The cell migration assay was performed using techniques similar to those used in the cell invasion assay, except that the filter in the transwell was not coated with Matrigel.
Western Blotting
Cells were washed twice with cold PBS, and lysis buffer (50 mM Tris-HCl, pH 8, 150 mM NaCl, 1% NP-40, 50 mM PMSF) including protease inhibitors (100X, 1 μg/ml of leupeptin, aprotinin, and pepstatin) was added for protein extraction. Quantification of proteins was determined by ultraviolet spectrophotometer (Bechman DU530; Fullerton, CA), and 50 μg of protein was loaded onto a 10% SDS-PAGE gel. After migration, proteins were electrotransferred to a nitrocellulose blotting membrane (Pall; Pensacola, FL). The membrane was incubated with blocking buffer [3% BSA in Tris-buffered saline Tween-20 (TBST)] for 1 hr at room temperature, washed, and incubated with one of the following primary antibodies: anti-Smurf2 (Upstate); anti-Smad2 (Cell Signaling; Danvers, MA); actin (I-19) (Santa Cruz Biotechnology; Santa Cruz, CA), anti-TGF-βR I (Santa Cruz Biotechnology), and anti-actin (Santa Cruz Biotechnology) overnight at 4C, washed with TBST for 3 × 15 min at room temperature, incubated for 1 hr at room temperature with horseradish peroxidase-conjugated secondary antibodies, and washed three times with TBST. Protein—antibody complexes were visualized using the Enhanced Chemiluminescence System (Pierce, Rockford, IL).
Statistical Analysis
Densitometric readings for the immunohistochemstry were performed by computer-aided laser scanning densitometry (Personal Densitometer SI; Molecular Dynamics, Sunnyvale, CA). For each peroid of pregnancy, three placentas were used, and immunohistochemical experiments were repeated at least three times. To make the statistical significance of the quantitative difference credible, at least three slides from three placentas of each group were examined for density scanning. In each compartment on a slide, at least 50 areas were randomly selected. Densitometric readings of intercellular substance which were assigned to background were subtracted. Values are expressed as means ± SEM for each group. Differences were examined by ANOVA (one-way ANOVA, defining p<0.05 as being statistically different), using the Statistical Package for Social Science (SPSS 10.0 for Windows package release; SPSS, Inc., Chicago, IL).
Quantification of the bands from RT-PCR and Western blotting was determined by MetaView Image Analyzing System (Version 4.50; Universal Imaging Corp., Downingtown, PA), and each band was normalized to its corresponding control. Statistical analysis was performed using SPSS 10.0 program. Results are expressed as means ± SEM. Statistical analysis was performed by two-way ANOVA and posttest, or t-test; p<0.05 was considered as statistically significant.
Results
Expression of Smurf2 in the Human Placental Villi Was the Highest During the First Trimester, as Compared With the Second and Third Trimesters
To study the spatio-temporal expression of Smurf2 during placentation, human placenta villi from first, second, and third trimesters were collected, and immunohistochemical and RT-PCR analyses were performed. As shown in Figure 1, immunohistochemical assay showed that Smurf2 proteins were localized mainly in the nuclei of cytotrophoblast cells and column trophoblast cells, as well as cytoplasm of syncytiotrophoblast cells in human placenta at gestational weeks 6–9 and 25 and at full term. Some mesenchymal cells inside the placental villi also exhibited positive staining for Smurf2. Control sections using normal IgG to replace the primary antibodies, or omitting the secondary antibodies, showed no specific staining (data not shown). Densitometric quantification of immunostaining of Smurf2 showed that immunostaining of Smurf2 proteins in villous cytotrophoblast cells and syncytiotrophoblast cells from the first trimester was significantly more intense than that of proteins from the second and third trimesters (Figure 1G). Moreover, as shown by RT-PCR analyses, expression of Smurf2 mRNA in placental villi from the first trimester was much higher as compared with that of the second trimester (Figure 1H).
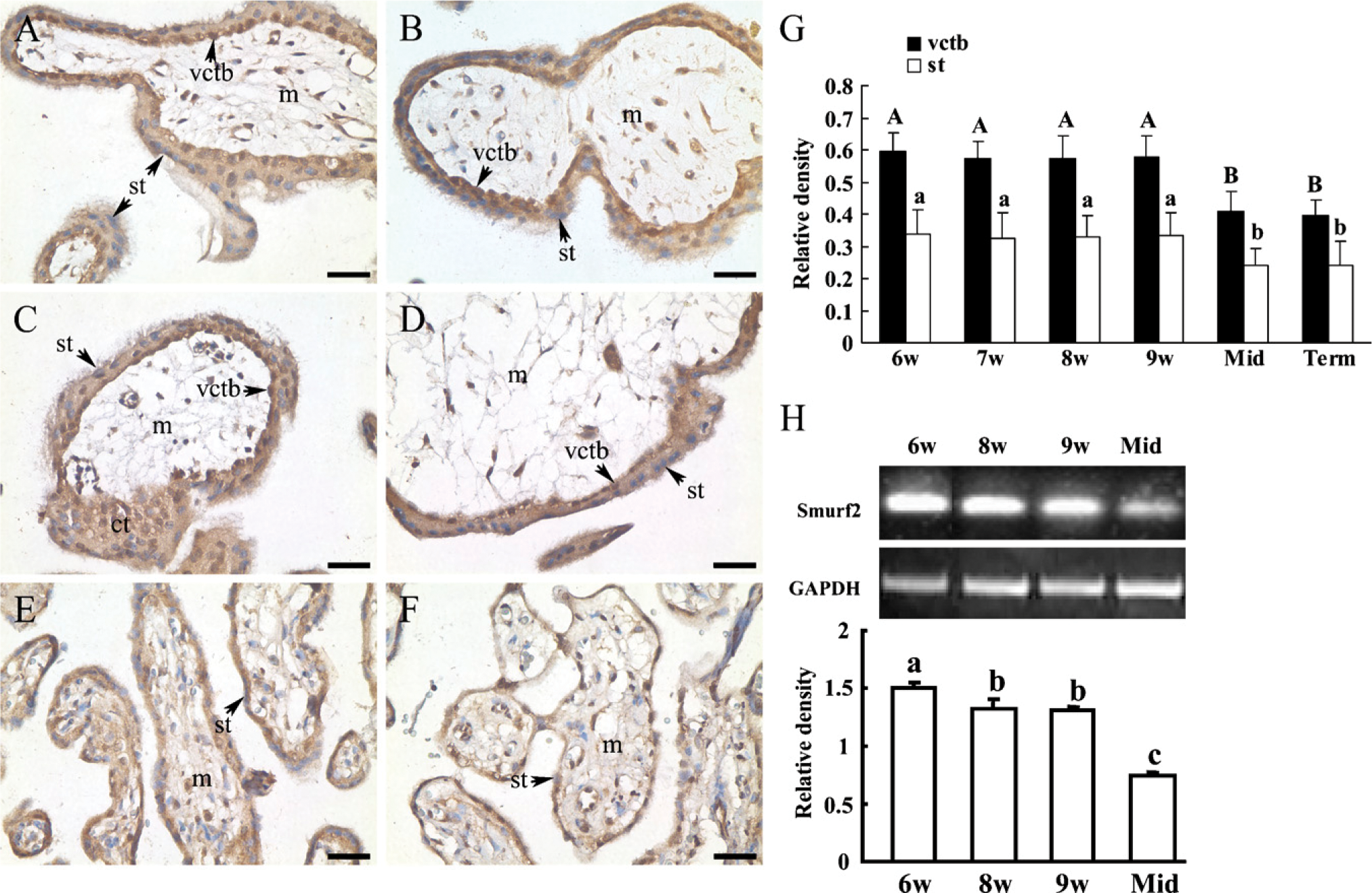
Expression of Smad ubiquitination regulatory factor 2 (Smurf2) in the human placental villi at different stages of pregnancy. Immunohistochemical assay to show the distribution of Smurf2 protein in human placenta at gestational weeks 6 (
Overexpression of Smurf2 Enhanced the Migration and Invasion of HTR-8/SVneo Cells
High levels of Smurf2 in early trophoblasts implies a role of Smurf2 in the high invasiveness of placental trophoblast cells during early pregnancy. To test this hypothesis, Smurf2 was overexpressed in HTR-8/SVneo cells, a human invasive EVT cell line. Because migration is a necessary step in the invasion cascade, we studied the effect of overexpression of Smurf2 on cell invasion and migration by using a Matrigel invasion assay and a transwell cell migration assay. As shown in Figures 2A, 2B, and 2E, overexpression of Smurf2 markedly increased the invasion of HTR-8/SVneo cells. Further studies using a transwell cell migration assay showed that overexpression of Smurf2 significantly enhanced the migration of HTR-8/SVneo cells (Figures 2C, 2D, and 2F).
TGF-βR I, but not Smad2 Was Downregulated by Overexpression of Smurf2
To determine the substrate of Smurf2 during human trophoblast invasion, HTR-8/SVneo cells were stably transfected with Smurf2 expression plasmid or empty vector, and Western blotting was performed to detect the levels of the candidate substrates. Smurf2 was proved to be overexpressed in HTR-8/SVneo cells stably transfected with Smurf2 plasmids (Figures 3A and 3B). TGF-βR I was significantly downregulated in Smurf2-overexpressed cells (Figures 3A and 3D). However, levels of Smad2 in cells transfected with Smurf2 or control vector exhibited no significant differences (Figures 3A and 3C). We also included TGF-β1 in this study to further test whether TGF-β1 has a role in the down-regulation of TGF-βR I by Smurf2. The results showed that the downregulation of TGF-βR I by Smurf2 was independent of the stimulation by TGF-β1 (Figure 3A).
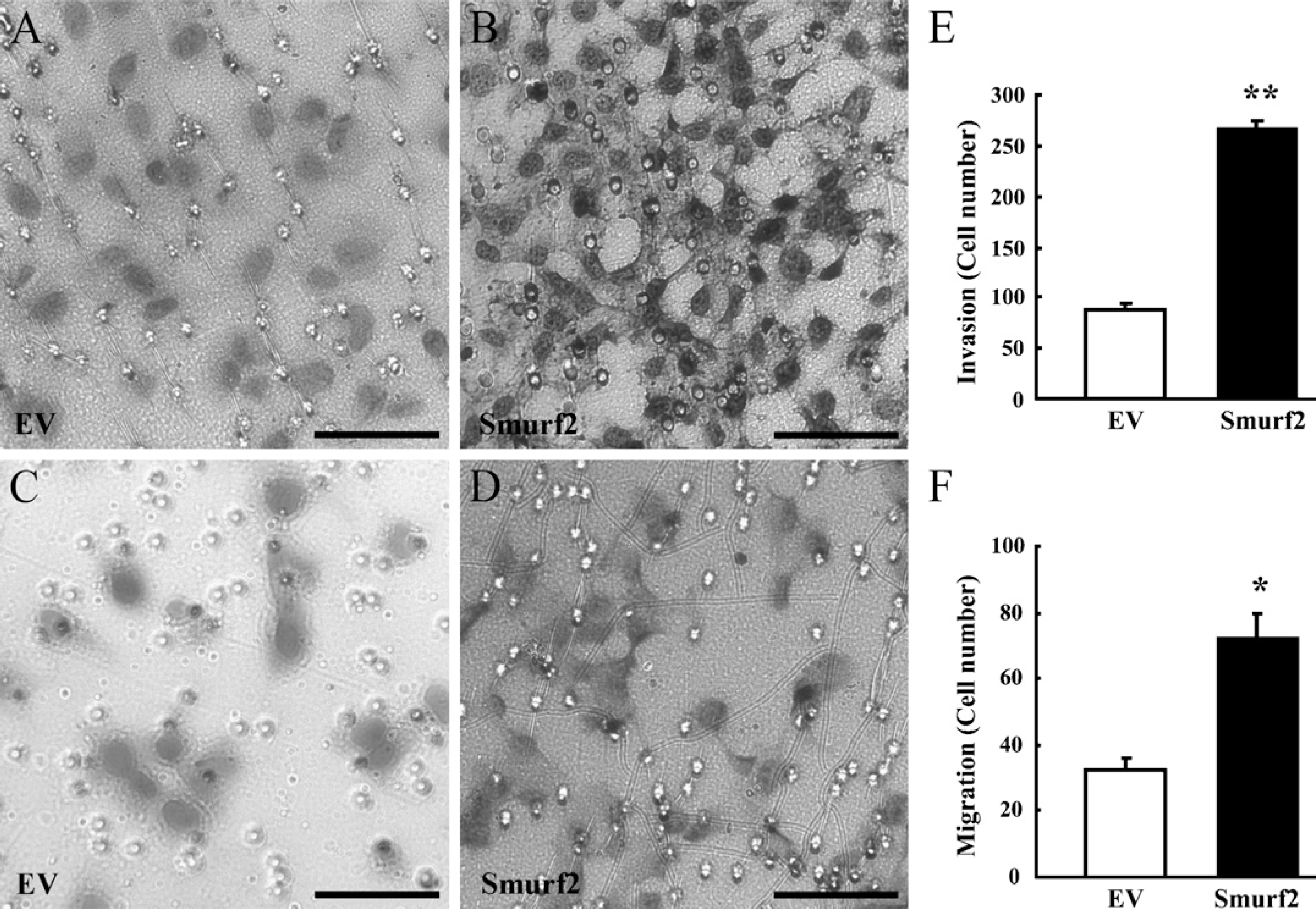
Overexpression of Smurf2 enhanced the migration and invasion of HTR-8/SVneo cells, a human invasive extravillous trophoblast (EVT) cell line. (
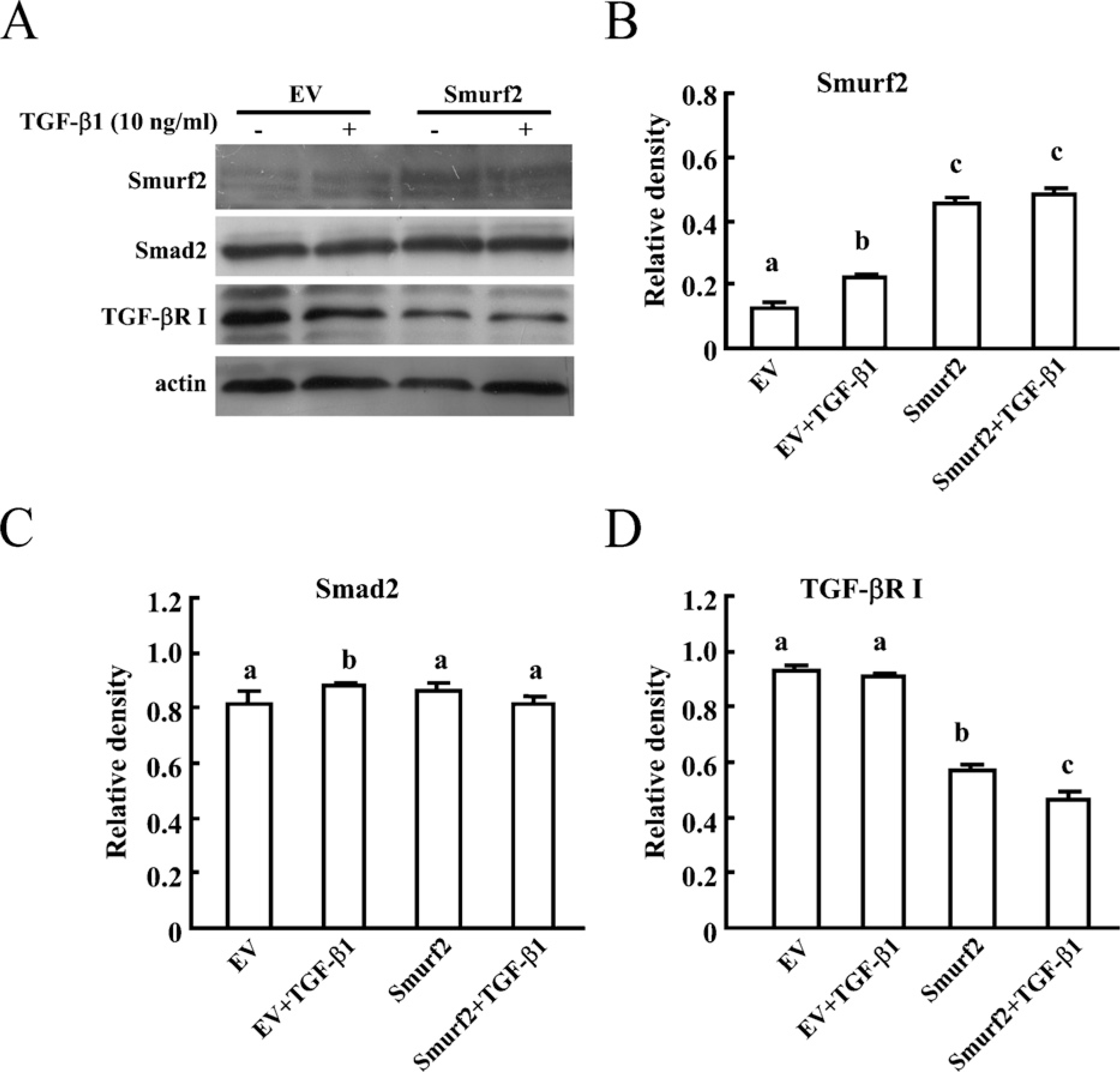
TGF-βR I, but not Smad2 was downregulated by Smurf2 overexpression. (
TGF-βR I Was Rescued by Smurf2 RNAi in HTR-8/SVneo Cells
To further prove the regulatory effect of Smurf2 on the expression of TGF-βR I, an RNAi experiment was performed in HTR-8/SVneo cells. Two out of three fragments of Smurf2 siRNA showed significant downregulation of Smurf2 levels (Figures 4A and 4B). Expression of TGF-βR I was significantly upregulated in cells transfected with Smurf2 siRNAs, as compared with cells transfected with control siRNA (Figures 4A and 4D). However, Smad2 levels were not significantly changed (Figures 4A and 4C).
Discussion
Highly coordinated control of proliferation, migration, and invasion of EVTs is the prerequisite for successful implantation and placentation (Biondi et al. 2006). Incomplete trophoblast invasion and spiral artery remodeling result in spontaneous abortion, intrauterine growth retardation (IUGR), and preeclampsia (Khong et al. 1986; Ball et al. 2006; Kadyrov et al. 2006). Ubiquitin-mediated proteolysis of regulatory proteins plays an important role in many physiological and pathological events (Ciechanover and Schwartz 2002). Our previous studies have demonstrated that the 26S proteasome has a significant role in mouse embryo implantation through regulation of the proteolytic activities of matrix metalloproteinase (MMP)-2 and −9 (Wang et al. 2004). Hass and Sohn showed that pre-eclamptic placenta exhibited ∼30% less proteasomal activity than normal placenta (Hass and Sohn 2003). Ubiquitin-conjugating enzyme UBE2Q2 (UBCi) was also specifically expressed at implantation sites in the rabbit endometrium (Melner et al. 2004,2006). Mice lacking the proteasome activator PA28γ (Murata et al. 1999), ubiquitin-conjugating enzyme UbcM4 (Harbers et al. 1996), ubiquitin ligase CUL7 (Huber et al. 2005) or Fbxw8 (Tsutsumi et al. 2008) exhibited developmental abnormalities such as IUGR and pathological defects in placenta. The above lines of evidence suggest that the ubiquitin—proteasome pathway is essential for embryo implantation and placental development.
In this study, we have demonstrated that another E3 ligase, Smurf2, may be involved in human trophoblast cell invasion via inhibition of TGF-β type I receptor based on the following lines of evidence: First, Smurf2 was highly expressed in the cytotrophoblast cells and EVTs of placental villi during the early stages of pregnancy. Second, stable overexpression of Smurf2 significantly increased the migration and invasion of HTR-8/SVneo cells based on the migration and Matrigel-invasion assays. Third, Smurf2 overexpression significantly down-regulated the expression of TGF-β type I receptor, which attenuates the signaling of TGF-β, one of the putative inhibitors of trophoblast cell invasion. Fourth, correspondingly, the expression of TGF-β type I receptor was rescued in cells transfected with Smurf2 siRNA.
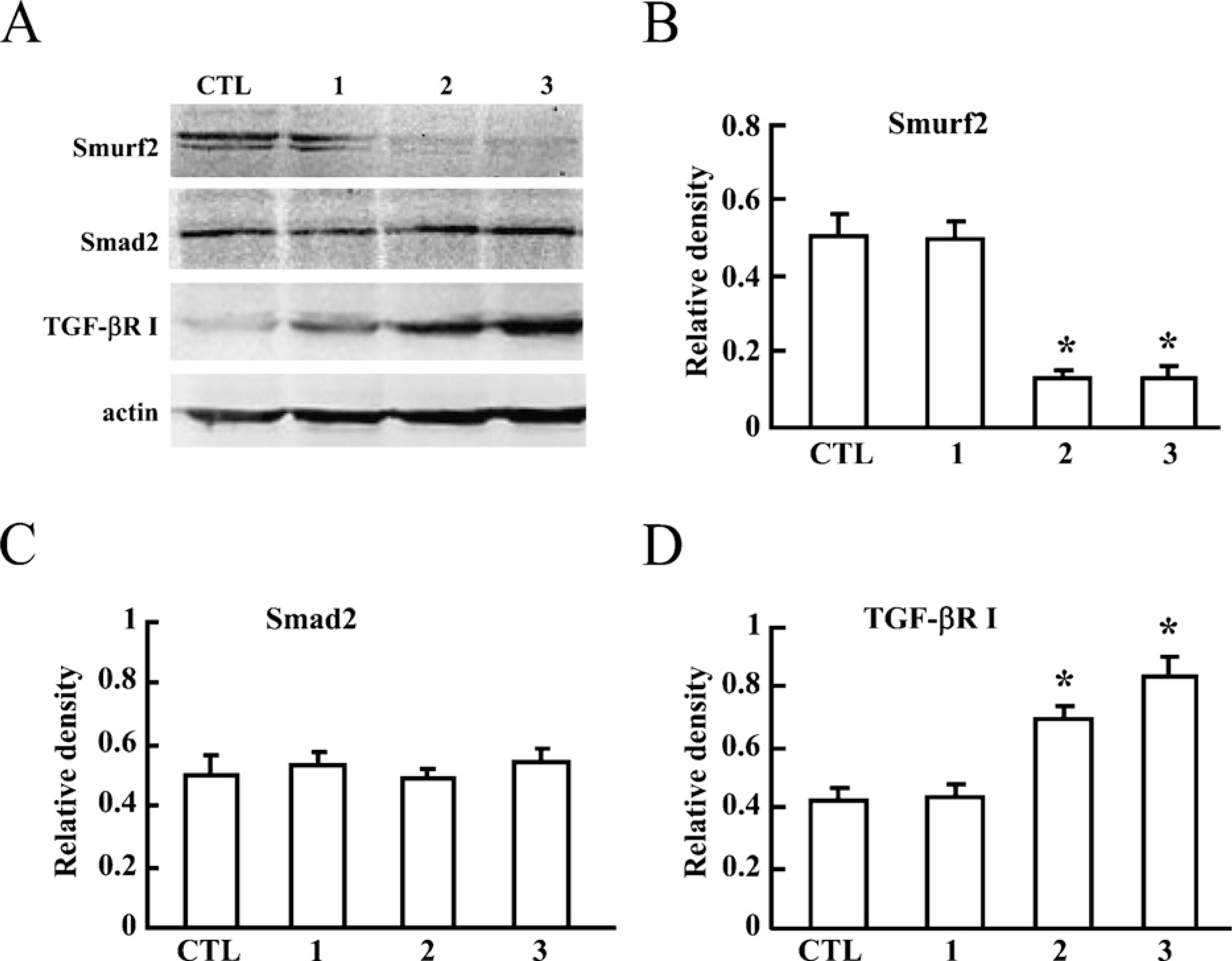
TGF-βR I, but not Smad2 was significantly rescued in EVT cells by knocking down Smurf2. (
Smurf2 has been thought to be the E3 ligase for Smad1, Smad2, and TGF-β type I receptor (Chen and Matesic 2007) that will ubiquitinate these proteins and somehow trigger their proteasome-mediated degradation. In our study, based on the overexpression and RNAi study, TGF-β type I receptor, but not Smad2, was observed to be regulated by Smurf2. These results suggest that in this trophoblast cell line, TGF-β type I receptor may be the main target and substrate of Smurf2. Whether there are other potential targets of Smurf2 such as other Smads, whether this conclusion can be extended to human placenta in vivo, and whether TGF-β type I receptor is regulated by Smurf2 via proteasome-mediated degradation are interesting questions that need to be further investigated.
Mechanisms whereby TGF-β inhibits EVT cell invasion has begun to be clarified. Blockage of trophoblast invasion by TGF-β is accompanied by upregulation of the tissue inhibitor of MMPs (TIMP)-1 (Graham and Lala 1991), reduced levels of collagenase type IV and urokinase-type plasminogen activator (uPA) activities, and increased activities of PAI-1, a specific inhibitor of uPA (Graham and Lala 1991; Graham 1997). TGF-β was also involved in cell migration and trophoblast differentiation, which form two other mechanisms of TGF-β-induced inhibition of trophoblast invasion (Graham 1997). In this study, downregulation of TGF-β type I receptor by Smurf2 provides another mechanism involving an E3 ligase Smurf2, whereby TGF-β inhibits trophoblast cell migration and invasion. Furthermore, the high expression of Smurf2 in first-trimester cytotrophoblast cells, which are highly invasive, and the lower expression in second- and third-trimester trophoblast cells, which are less invasive, indicate that Smurf2 is involved in the stringently controlled trophoblast invasion, which shares similarities with invasion of tumor cells but is unlike the infinite invasion of tumor cells. It has been reported that Smurf2 is highly expressed in esophageal squamous cell carcinoma (Fukuchi et al. 2002). In this respect, Smurf2 is involved in trophoblast and tumor cell invasion. However, a very recent study has shown that Smurf2 prevents breast cancer cell migration by inducing degradation of Smurf1 (Fukunaga et al. 2008). Therefore, the function of Smurf2 and its targets and regulatory network in tumorigenesis remain to be further elucidated.
In summary, we provide evidence to demonstrate that Smurf2 is involved in the migration and invasion of HTR-8/SVneo cells. Furthermore, this function may be implemented via targeting TGF-β type I receptor as its substrate.
Footnotes
Acknowledgements
This study was supported by Program 973 (2006CB504006 and 2006CB944008), special foundation of the President of the Chinese Academy of Sciences to H.W., and a grant from the Natural Science Foundation of China (30770323) to H-Y.L.
The authors are grateful to Dr. Jeffrey L. Wrana (University of Toronto) for kindly offering the pCMV5B-Flag-tagged Smurf2 plasmid.
