Abstract
Intermediate filaments are frequently used in studies of developmental biology as markers of cell differentiation. To assess whether they can be useful to identify differentiating pancreatic endocrine cells, we examined the pattern of expression of nestin, cyto-keratin 20, and vimentin on acetone-fixed cryosections of rat adult and developing pancreas. We also studied vimentin expression in mouse embryonic pancreas at E19. Cytokeratin 20 was found in all pancreatic epithelial cell lineages during the entire development of the rat gland and in the adult animals. Under our experimental conditions, therefore, cytokeratin 20 is not an exclusive marker of rat duct cells. Nestin was detected exclusively in stromal cells either in the adult or developing rat pancreas. Vimentin was observed within cells located in the primitive ducts of rat pancreas starting from E12.5. Their number rapidly increased, reaching its highest level in newborn animals. Vimentin was also spotted in α cells starting from E12.5 but disappeared soon after birth, likely identifying immature or recently differentiated α cells. In addition, vimentin was observed in duct and α cells of mouse developing pancreas showing that its expression in such cells is not an event restricted to the rat. This manuscript contains online supplemental material at http://www.jhc.org. Please visit this article online to view these materials.
Keywords
Intermediate filaments are among these proteins because they exhibit cell type specificity, and their expression is highly coordinated with organ development and cell differentiation. For this reason, intermediate filaments are commonly used as valuable cell markers in developmental biology and oncology. Despite the remarkable work carried out in the past decade (Bouwens et al. 1994; Bouwens and De Blay 1996; Lardon et al. 2002; Yashpal et al. 2004), our knowledge on the fine distribution of the different intermediate filaments in rat developing pancreas is still incomplete. Indeed, the use of a technique particularly suitable for the preservation of intermediate filaments in tissue sections has allowed us to gather an important body of new information that substantially changes our view regarding their pattern of expression. In particular, our results show that, although with a different degree of intensity, all pancreatic epithelial cell lineages in the rat express cytokeratin 20 (CK20) along the entire differentiating process and even in the adult animals. Moreover, in embryos, vimentin seems to be expressed in pancreatic duct-like cells starting from E12.5, and this expression increases over time, reaching its peak soon after birth. Our data also show that even a population of endocrine cells (i.e., α cells) transiently contains vimentin. In addition, experiments on mouse embryos show that the presence of vimentin in developing pancreatic ducts and in some glucagon cells is not a phenomenon restricted to the rat.
Materials and Methods
Antibodies
The following antibodies were used: rabbit anti-human α-amylase (code A-8273), monoclonal mouse anti-vimentin (clone V9), and anti-pan cytokeratin (panCK) from Sigma-Aldrich (St. Louis, MO), mouse monoclonal anti-CK20 (clone Ks 20.8) from Dako (Glostrup, Denmark), mouse monoclonal anti-nestin (clone 2Q178) from Abcam (Cambridge, UK), rabbit anti-glucagon (FL-180, code sc-13091) and goat anti-somatostatin (D-20, code sc-7819) from Santa Cruz (Santa Cruz, CA), guinea pig anti-insulin (code 18-0067) from Zymed (San Francisco, CA), guinea pig anti-pancreatic polypeptide (PP) from Linco Research (St. Charles, MO), goat anti-vimentin (Hartig et al. 1997) kindly provided by Prof. Peter Traub (Max-Plank Institut fur Zellbiologie; Rosenhof, Ladenburg, Germany), donkey cyanine 2 (Cy2)-conjugated anti-mouse IgG (code 715-226-151), donkey Cy2-conjugated anti-rabbit IgG (code 711-226-152), donkey Cy2-conjugated anti-goat IgG (code 705-225-147), donkey Cy5-conjugated anti-mouse IgG (code 715-176-150) from Jackson Immunoresearch Laboratories (West Grove, PA), donkey tetramethyl rhodamine (TRITC)-conjugated antimouse IgG (code AP192R), donkey FITC-conjugated anti-guinea pig IgG (code AP193F), and donkey TRITC-conjugated anti-goat IgG (code AP180R) from Chemicon (Temecula, CA).
Animals and Tissues
All the animals used for this research were maintained and treated in accordance with the current Italian laws on the subject. Five adult male rats (250–300 g), 5 newborn rats, 5 3-day-old rats, 16 pregnant female rats (all Sprague-Dawley rats), and 2 pregnant female mice (C3H-HeN) were used for this study and were purchased from Charles River Laboratories (Calco, Italy). Mice and rats were anesthetized with intraperitoneal injections of chloral hydrate (400 mg/kg) and urethane (800 mg/kg), respectively, and killed by an overdose of the same anesthetics. Embryos were recovered from pregnant rats at different developmental stages starting from E12.5 up to E21.5 and from the pregnant mouse at E19. For our immunofluorescence surveys, we applied the protocol that in our experience best preserves the antigenicity of the intermediate filament network (Carapelli et al. 2004). Pancreas from adult and newborn rats as well as from embryos older than E18 were sampled and frozen by immersion in isopenthane pre-chilled with liquid nitrogen. Embryos younger than E18 were frozen en bloc in the same way.
Sections (as thick as 10 μm) were cut at −23C, mounted on Superfrost slides, air dried, fixed with cold acetone, and kept at −80C until the day of the experiment.
IHC and Confocal Microscopy
A TCS 4D Leica (Wetzlar, Germany) confocal microscope or a LSM510 Zeiss with selective multitracking excitation were used to observe double- or triple-labeling experiments carried out on frozen sections. The following double- or triple-staining experiments were performed on all rat samples: vimentin/CK20, glucagon/vimentin, glucagon/CK20, somatostatin/vimentin, somatostatin/CK20, insulin/vimentin, insulin/CK20, PP/CK20, PP/vimentin, amylase/vimentin, amylase/CK20, nestin/glucagon, nestin/vimentin, and nestin/insulin/vimentin. In addition, we carried out glucagon/vimentin or vimentin/pan-CK/glucagon multiple-labeling experiments on mouse embryo sections.
The protocols used on rat sections were as follows: unspecific protein—protein interactions were quenched with 5% BSA in PBS for 10 min before incubating sections with markers of pancreatic endocrine cells (anti-glucagon for α cells, working dilution 1:50 in PBS, or anti-insulin for β cells, working dilution 1:50 in PBS, or anti-somatostatin for δ cells, working dilution 1:200 in PBS, or anti-PP working dilution 1:400 in PBS), or markers of exocrine cells (anti-amylase, working dilution 1:100 in PBS, or anti-CK20, working dilution 1:100 in PBS), or anti-nestin (working dilution 1:100). After rinsing, sections were incubated for 10 min with 10% donkey serum in PBS followed for 1 hr by the appropriate secondary antibody. After washing, goat anti-vimentin antibody (working dilution 1:1000 in PBS) was applied on sections previously incubated with the anti-CK20 or the anti-nestin antibodies and, in some cases, previously incubated with the anti-glucagon antibody. Monoclonal anti-vimentin, anti-CK20 (both antibodies diluted 1:50 in PBS), or anti-nestin antibodies were applied to the remaining sections. On rinsing, slides were once again incubated for 10 min with 10% donkey serum in PBS followed for 1 hr by the appropriate secondary antibody. For triple-labeling experiments, unspecific protein—protein interactions were quenched with 5% BSA in PBS, and sections of rat pancreas were incubated with the following sequence of antibodies: mouse anti-nestin, Cy5-conjugated donkey anti-mouse IgG, guinea pig anti-insulin, FITC-conjugated donkey anti-guinea pig IgG, goat anti-vimentin, and TRITC-conjugated donkey anti-goat IgG. Instead, sections of mouse embryos were incubated with the following sequence of antibodies: goat anti-vimentin, TRITC-conjugated donkey anti-goat IgG, mouse anti-panCK (working dilution 1:100 in PBS), Cy5-conjugated donkey anti-mouse IgG, rabbit anti-glucagon, and Cy2-conjugated donkey anti-rabbit IgG. When double-labeling experiments were performed on mouse sections, the antibodies used were as follows: goat anti-vimentin, TRITC-conjugated donkey anti-goat IgG, mouse anti-panCK, Cy2-conjugated donkey anti-mouse IgG, rabbit anti-glucagon, and Cy2-conjugated donkey anti-rabbit IgG. Between incubations, sections were thoroughly rinsed in PBS, and 10% donkey serum was applied before each secondary antibody. In some cases, computer-assisted three-dimensional (3D) reconstruction through the Z plane of the section was carried out with Zeiss LSM image browser version 4.2.0.93 software. Controls were performed replacing primary antibodies with nonimmune sera.
Results
Adult Rat Pancreas
Adult rat pancreas showed a diffuse immunofluorescence for CK20 that appeared to be widespread to all epithelial cells, although with a different degree of intensity according to cell type (Figure 1). In particular, all duct cells, possibly including centroacinar cells, showed strong CK20 immunoreactivity with thick bundles of fluorescent intermediate filaments (Figures 1A and 1C). Acinar cells, identified by their amylase content, were CK20 immunoreactive as well, but in this case, the labeling outlined a very fine network that appeared stronger just under the apical domain of the cells (Figure 1A). The specificity of the CK20 labeling was confirmed by the disappearance of the fluorescence when the anti-CK20 antibody was not applied to the sections (Figure 1B) and by the pattern of staining that labeled exclusively intracellular cytoskeletal filaments (Figure 1C).
Even endocrine cells were CK20 immunoreactive, but in this case, the intensity of the fluorescence was not uniform. The δ and PP cells (Figures 1D–1G) showed a network of CK20 filaments labeled as strongly as those belonging to the adjacent acinar cells but without any evident polarization. The β cells (Figures 1H and 1J) showed a weaker but almost constant fluorescence, whereas α cells (Figures 1K and 1L) did not exhibit a homogeneous labeling; some cells were provided with relatively strong fluorescence, whereas others were almost devoid of CK20 immunoreactivity.
Once again, the specificity of the CK20 labeling was confirmed by the absence of the fluorescence when the anti-CK20 antibody was not applied (Figure 1I) and by the pattern of staining that labeled exclusively intracellular cytoskeletal filaments in δ (Figure 1E), PP (Figure 1G), insulin (Figure 1J), and glucagon (Figure 1L) cells.
As expected, the pattern of expression of vimentin was limited to the stroma (Bouwens and De Blay 1996) where it labeled blood vessels, nerve fibers, and elongated fibroblast-like cells as previously reported. Nestin was detectable within few elongated stromal cells scattered in the interacinar space or within cells coexpressing vimentin associated with intra-islet blood capillaries (see Online Supplemental Figure SF1).
Embryonic Rat Pancreas
CK20 immunoreactivity was evident in the dorsal pancreatic rudiment since E12.5, the first gestational age studied in this investigation (Figure 2). As previously reported (Bouwens and De Blay 1996), CK20 labeled all pancreatic epithelial cells that at this stage have just entered the protodifferentiated phase and do not show phenomena of cytodifferentiation with the exception of a few α and β cells (Pictet and Rutter 1972; Le Bras et al. 1998). As the growth of the pancreatic rudiment proceeded, CK20 fluorescence persisted in the entire pancreatic epithelium but with some differences that allowed endocrine, acinar, and duct cells to be distinguished. Whereas duct cells showed very strong fluorescence, endocrine cells showed less intense staining, and acinar cells were provided with weak labeling that outlined a fine polarized network of filaments (see Online Supplemental Figure SF2). Duct cells, therefore, were easily identifiable because they lined primitive ducts and were highly CK20 immunoreactive. Indeed, nascent islets were characterized by a relatively strong CK20 fluorescence, which always appeared more intense at the rim of the islets (Figure 2 and see Online Supplemental Figure SF2), where possibly a mantle of differentiating duct cells persist longer as previously reported (Bouwens et al. 1994; Bouwens and De Blay 1996).
Vimentin was detectable within cells of the dorsal (Figures 2A and 2B) and ventral pancreatic rudiments shortly after the beginning of their development (E12.5 and E13.5, respectively). The number of these cells progressively increased with time (Figure 2C), reaching its highest peak in newborn rats when a large subset of the duct cells contained vimentin (Figures 2D and 2E) and rapidly decreasing soon after. Amylase-containing acinar cells were always CK20 immunoreactive from the beginning of their cytodifferentiation, but they never showed vimentin fluorescence.
Because endocrine cells differentiate from precursor cells located in the epithelial lining of the primitive ducts, we decided to verify whether vimentin-immunoreactive duct-like cells could be transitional cells in the process of differentiation into mature endocrine cells. The β, δ, and PP cells, although always containing a well-developed network of CK20 intermediate filaments, never showed vimentin immunoreactivity at any time point, even in newborn animals when vimentin-containing duct cells were at their highest number (Figure 3). In contrast, in addition to a network of CK20 filaments that was present throughout the entire development of the pancreas (Figure 4A), α cells also showed vimentin immunoreactivity starting from the earliest gestational age studied (E12.5). At this stage of development, almost all vimentin-containing cells within the dorsal pancreatic bud were α cells, and even the opposite (i.e., α cells showing a vimentin network) applied almost constantly (Figures 4B and 4C). At E13.5, the ventral pancreatic bud showed the same pattern of staining as well. With time, however, α cells seemed to progressively lose vimentin filaments (Figures 4D–4F). Nevertheless, in newborn animals, clusters of vimentin-expressing glucagon cells (Figure 4G) could be still found, but the number of these cells decreased very quickly, and after a few days from birth, they were virtually absent (data not shown). Because nestin has been reported to coassemble with vimentin intermediate filaments (Steinert et al. 1999), we wondered if vimentin-containing α cells could contain nestin as well. Despite all our efforts, however, we were unable to spot any nestin-immunoreactive α cells (see Online Supplemental Figure SF3). Indeed, nestin expression seemed restricted to mesenchymal cells and presumptive developing blood vessels.
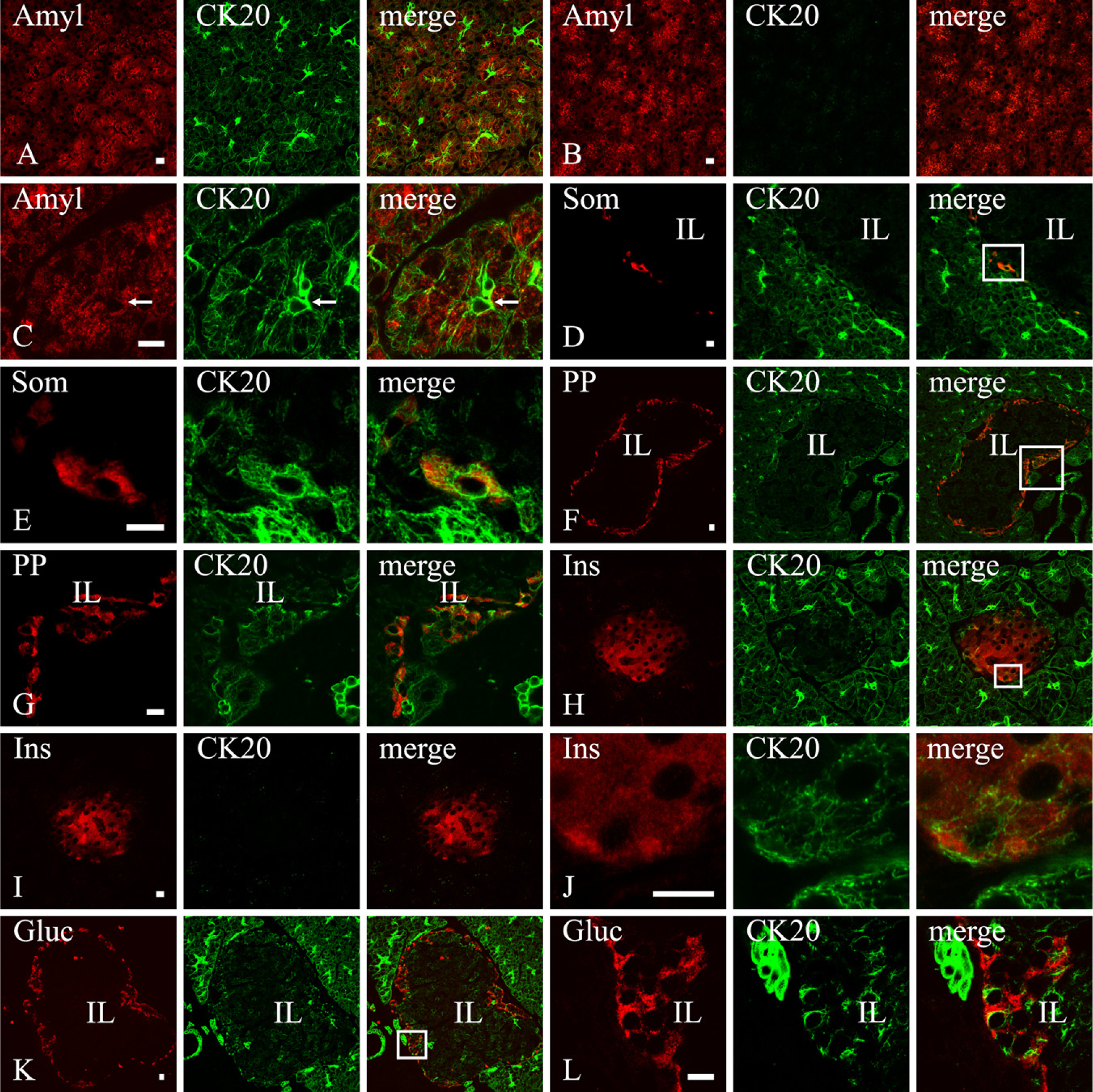
Cytokeratin (CK)20 expression in the adult rat pancreas. Confocal microscopy of double labeling experiments with anti-CK20 and anti-amylase (
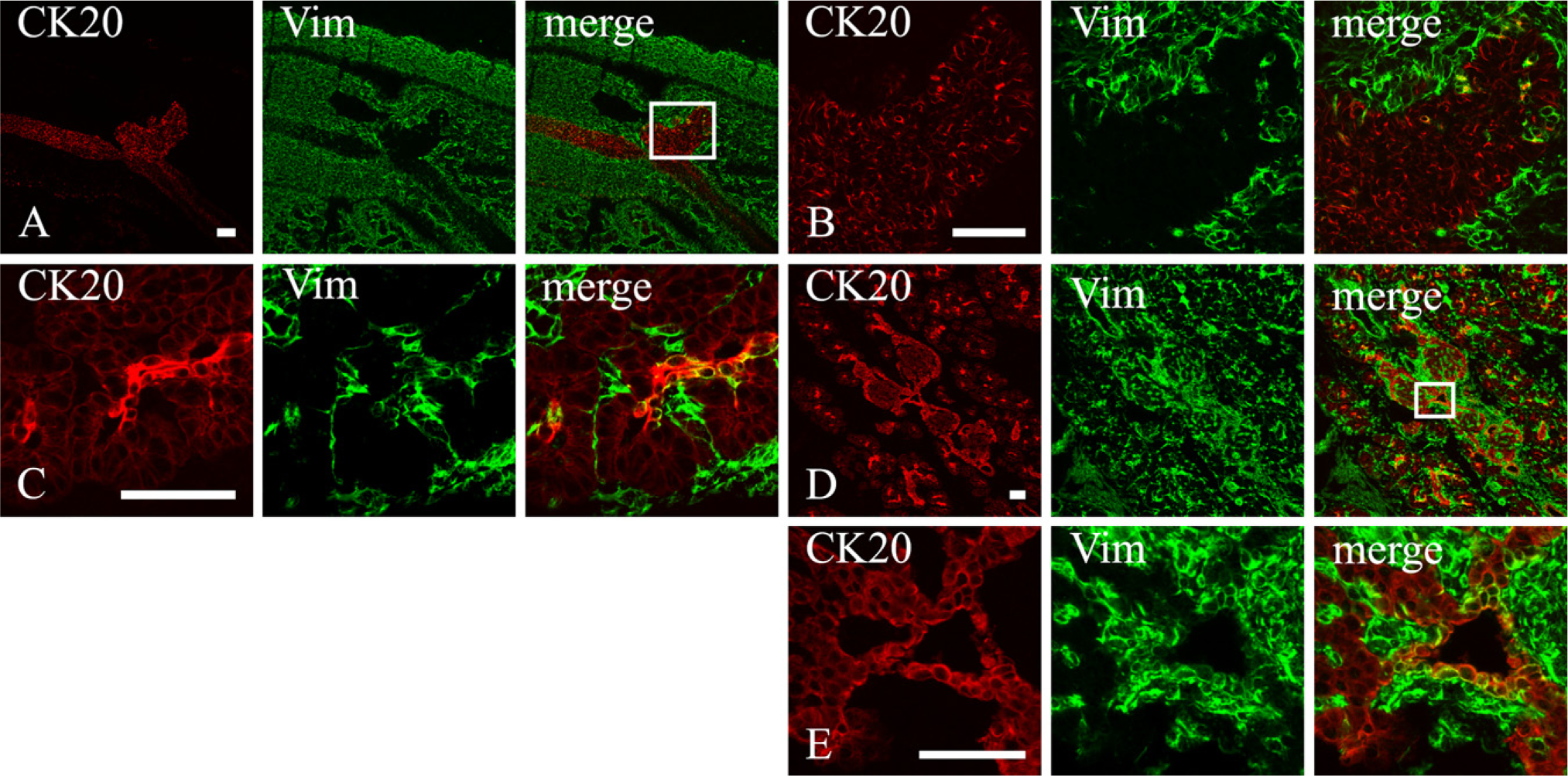
CK20 (red) and vimentin (green) expression in the developing rat pancreas at different gestational ages. (
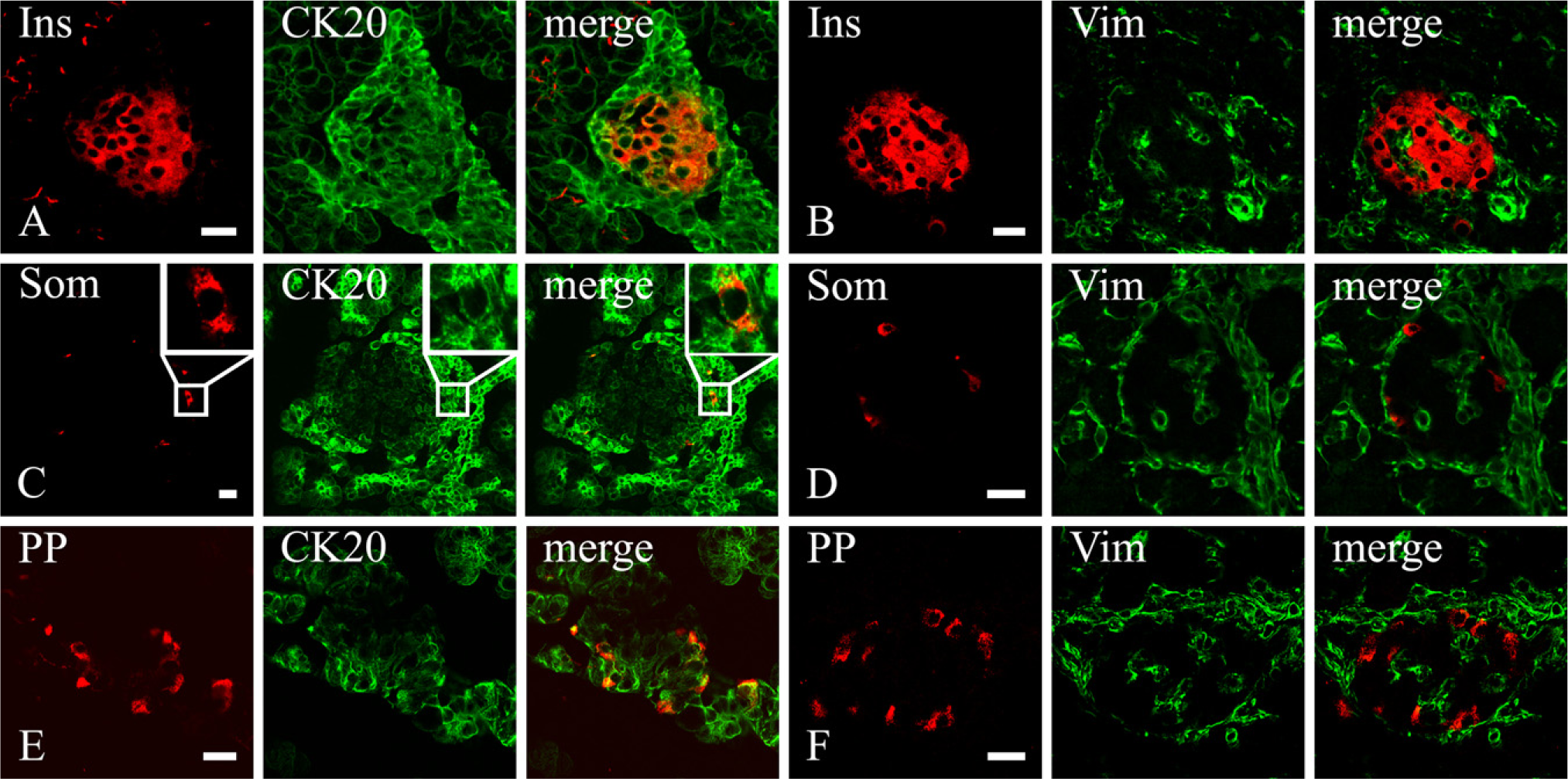
CK20 (
Embryonic Mouse Pancreas
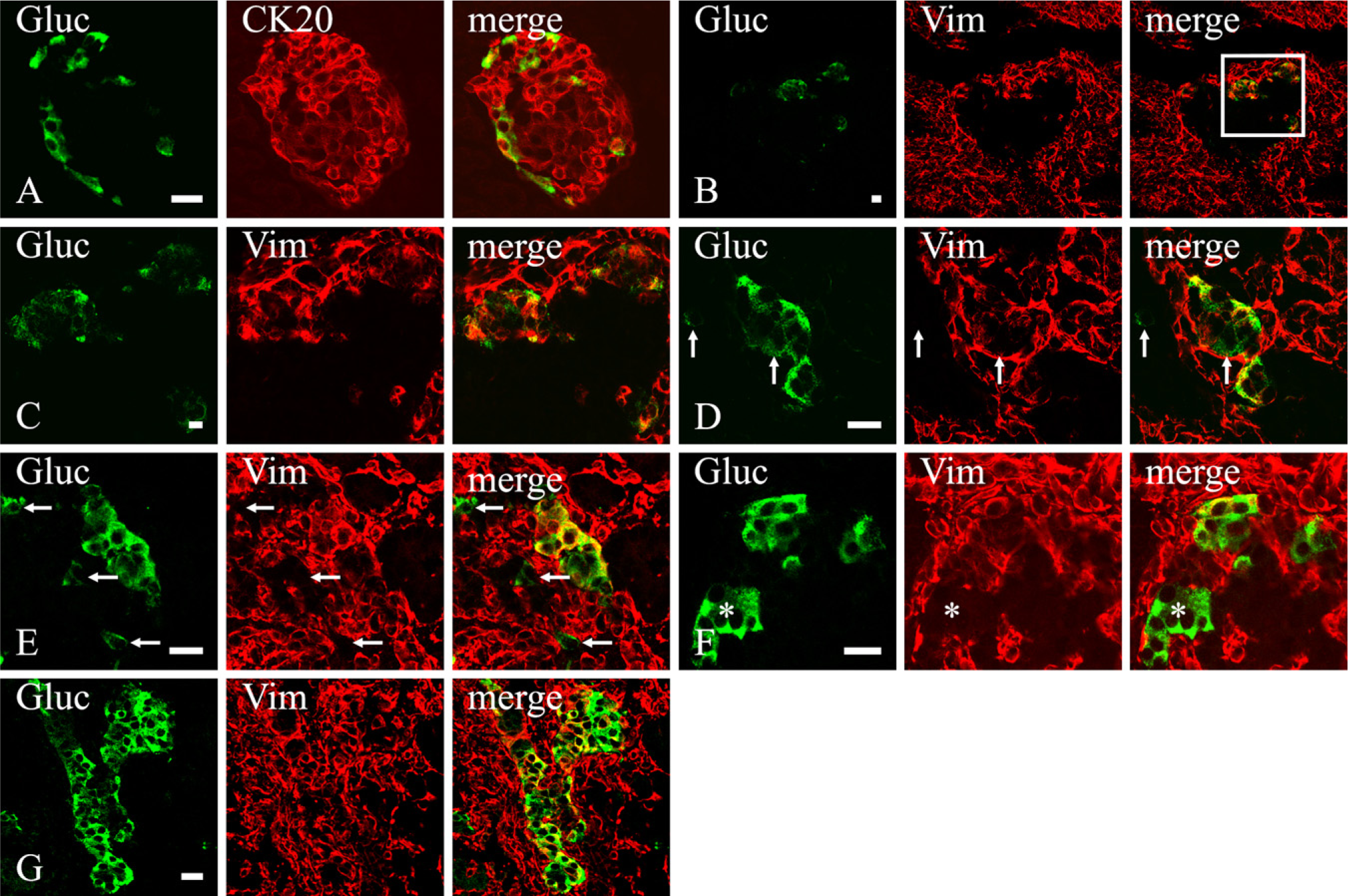
To verify whether the expression of vimentin within duct and α cells in the developing rat pancreas was a phenomenon restricted to this species, we carried out some experiments on sections from mouse embryos. Experiments were limited to the assessment of vimentin immunoreactivity in pancreatic epithelial cells at E19. Pancreatic epithelium was marked by its cytokeratin content. Among epithelial cells, ducts were identified by their morphology and α cells by their glucagon content. At this age, all recognizable ducts coexpressed vimentin and CKs (Figures 5A–5D). The specificity of the labeling was assessed by the following factors: (a) absence of the fluorescence when primary antibodies were not applied; (b) pattern of staining that labeled exclusively intracellular cytoskeletal filaments (Figures 5B–5D); and (c) presence of two distinct networks of cytoskeletal filaments as they appeared by a 3D reconstruction of the section (Figure 5C) or by scanning it through XZ and YZ planes (Figure 5D).
In some experiments, in addition to labeling vimentin and CK intermediate filaments, we also stained α cells with an anti-glucagon antibody. Consistent with findings in rat embryos, at this late gestational age, most glucagon cells of mouse pancreas were not vimentin immunoreactive. However, a few of them contained a double network of CK and vimentin filaments (Figure 5E).
Discussion
A major goal in the studies on pancreatic regeneration aimed to restore an appropriate number of β cells would be the identification of islet precursor cells capable of proliferating and differentiating into endocrine cells. Even though this issue is still highly debated (Bonner-Weir and Sharma 2006; Dhawan et al. 2007; Zhao et al. 2007; Xu et al. 2008), these cells are supposed to persist in the pancreatic ducts of adults because, under several experimental conditions, the gland has been shown to undergo regenerative phenomena leading to the formation of small endocrine clusters (Wang et al. 1994,1995; Bouwens and Rooman 2005; Kauri et al. 2007; Xu et al. 2008). To understand whether this regeneration (i.e., islet neogenesis) is caused by the activation of hidden stem cells that follow to some extent the same pathway of differentiation occurring in the embryonic pancreas, it is crucial to achieve knowledge of the precise sequence of events and proteins that are expressed in the developing pancreas. With this in mind, we analyzed the pattern of expression of some intermediate filaments that could be used as valuable markers for the phenotypic identification of differentiating and/or transitional cell types.
CK20 (
CK20, widely used as a marker of pancreatic duct cells in adult rodent pancreas (Bouwens 1998), has also been reported to be expressed in islet cells of the developing pancreas (Bouwens and De Blay 1996). Its presence in hormone-containing cells has been interpreted as a marker of transitional cells (Bouwens et al. 1994; Wang et al. 1995; Peters et al. 2000; Bouwens 2004) to differentiate from duct to islet cells. Our findings showed that CK20 expression in the rat pancreas is broader than previously thought, because under our experimental conditions, it is detectable in all epithelial cells either in the developing or in the adult organ. It is therefore evident that the simple detection of CK20 should not be considered in itself definitive proof for duct cell identification or, when located within hormone-containing cells, as differentiating cells. On the other hand, based on the different intensity and distribution of the labeling, CK20 still seems useful to determine major cell phenotypes (duct, acinar, or endocrine). Interestingly, some endocrine cell types (δ and PP cells) seem to maintain a well-developed CK20 intermediate filament network even in adult rats.
Our results differ substantially from what have been previously reported. This is likely because of the different IHC approach that we carried out (cryosections fixed with cold acetone), which is optimal to preserve intermediate filament antigenicity. However, it is well known that fixation with cold acetone (Larsson 1988) extracts small molecules (i.e., insulin). It is therefore possible that cells containing low levels of insulin (e.g., recently differentiated cells) may have skipped their identification.
The pattern of nestin expression is not a clear issue in pancreatic developmental biology. Indeed, almost all cell types present in the pancreatic rudiment (i.e., mesenchymal cells, endothelial cells, pancreatic stellate cells, acinar cells, endocrine cells, duct cells, and unidentified cells within the islets of Langerhans) have been reported at least once as containing nestin (Lardon et al. 2002; Selander and Edlund 2002; Klein et al. 2003; Delacour et al. 2004; Yashpal et al. 2004). Even in the adult pancreas, nestin has been reported in different cell types (Hunziker and Stein 2000; Zulewski et al. 2001; Lardon et al. 2002; Klein et al. 2003). Our findings closely match those of groups that found nestin in mesenchymal cells of the developing pancreas or in cells of mesenchymal origin in the adult animals (Lardon et al. 2002; Selander and Edlund 2002; Klein et al. 2003). In particular, we found nestin associated with islet blood capillaries, in developing blood vessels, and in fibroblast-like cells of embryonic pancreas. Remarkably, in our survey, nestin was only present in cells coexpressing vimentin. Coexpression with vimentin is a control that should always be considered because nestin is not capable of self-assembling but always requires vimentin, α-internexin, or, possibly, desmin to copolymerize with (Steinert et al. 1999; Herrmann and Aebi 2000). Hence, based on the inconsistency of previously published results and on our own results, the reliability of nestin as a marker of pancreatic epithelial cells should be considered doubtful.
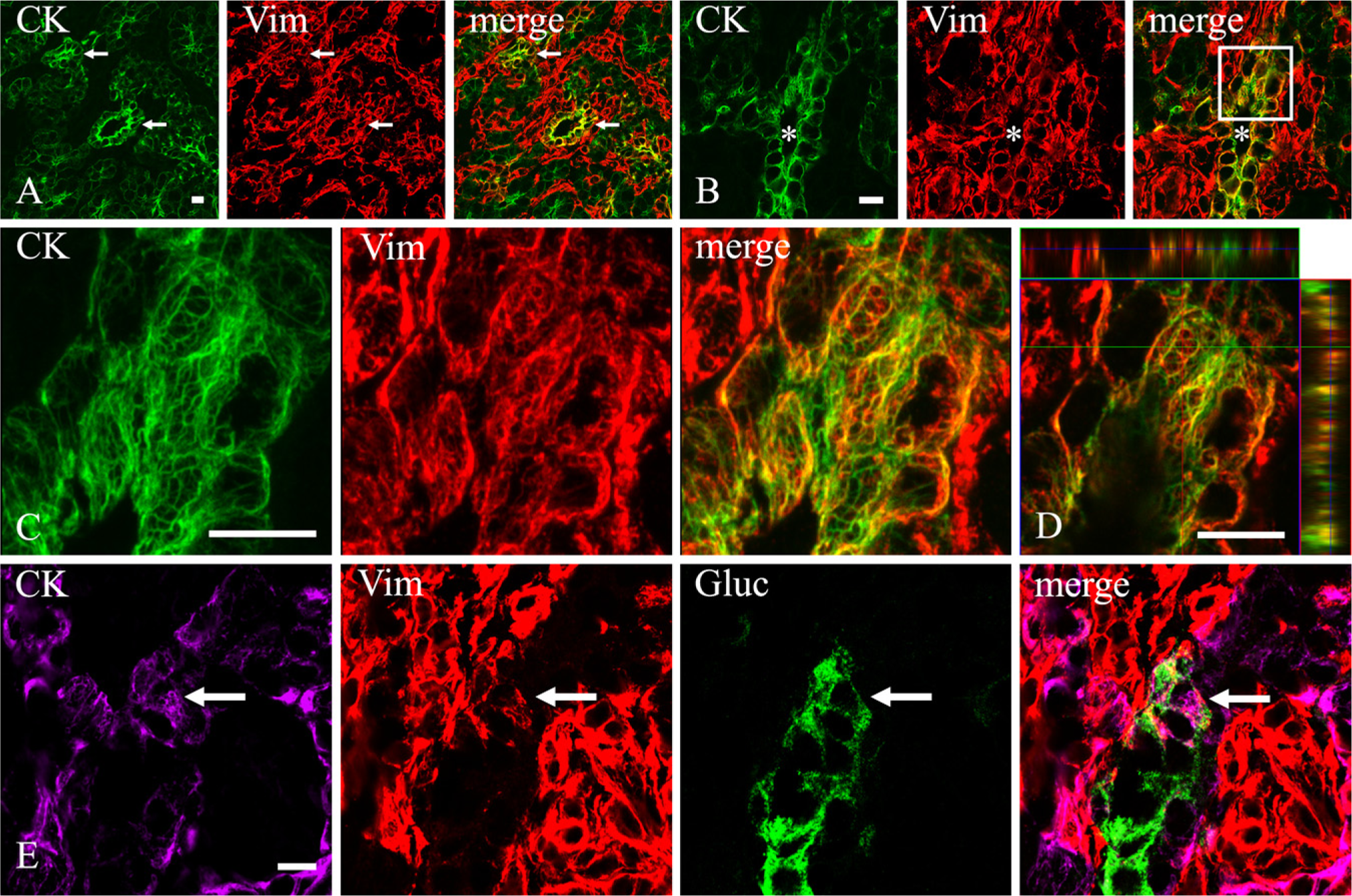
Vimentin expression within duct and α cells in mouse developing pancreas (E19). (
Vimentin is known to be expressed in all mesenchymal or mesenchymal-derived cells. In addition, in embryonic rat pancreas, between E17 and E20, vimentin has been transiently found in duct cells but never in endocrine or in acinar cells (Bouwens and De Blay 1996). In contrast, we were able to observe coexpression of vimentin and CK20 in some duct-like cells starting from E12.5 and reaching its highest labeling intensity in newborn animals. Moreover, we confirmed that, at least at late gestational ages, vimentin is expressed in mouse pancreatic ducts as well. The presence of vimentin in rodent developing pancreatic ducts is quite an interesting phenomenon because vimentin is seldom expressed in epithelial cells under physiological circumstances (Carapelli et al. 2004). Its appearance and the increase of its expression in late gestational ages could be related to the pancreatic expression of transforming growth factor (TGF)-β signaling components (Kim and Hebrok 2001). In particular, TGF-β receptor II is present in the pancreatic rudiment of the mouse from E12.5, and it becomes progressively more expressed in developing ducts up to E18.5 (Crisera et al. 1999). The upregulation of vimentin expression is one of several biological effects exerted by TGF-β1 (Carey and Zehner 1995), and it has recently been shown that a TGF-β1 response element is present within the activator protein complex-1 region of the vimentin promoter (Wu et al. 2007). It is therefore possible that signaling through TGF-β receptor II may be one of the factors inducing activation and regulation of vimentin expression in duct cells. Which biological asset vimentin would convey to developing pancreatic ducts is not clear at this time. However, in the last decade, important and unexpected roles for vimentin have been shown in a wide variety of biological events. These events range from the simple structural reinforcement of the cell to adhesion, migration, signaling, and even DNA regulation and cell differentiation (Benes et al. 2006; Ivaska et al. 2007). Therefore, it is reasonable to hypothesize an involvement of vimentin in some phenomena likely occurring in developing pancreatic ducts as well.
After its finding in embryonic rat pancreatic ducts (Bouwens and De Blay 1996), vimentin was not used as a marker of ducts undergoing processes of islet morphogenesis until recently, when it was reported in pancreatic ducts of adult rats that are subtotally pancreatectomized (Ko et al. 2004) or that develop tubular complexes (Wang et al. 2005). Our results, widening the window of time for vimentin expression in rat pancreatic ducts and showing that vimentin is also expressed in mouse duct cells, reinforce the idea of using vimentin as a marker of ducts undergoing islet morphogenetic events. This is also supported by the observation that vimentin is present in α cells located close to primitive ducts and around the nascent islets. The finding of vimentin-containing α cells beside vimentin-expressing ducts strengthens the idea that such ducts are actually generating islet cells. Indeed, coexpression of glucagon and vimentin in the same cells strongly suggests the temporary existence of transitional forms of immature glucagon-secreting cells still show the arrangement of duct intermediate filaments. In support of this hypothesis is also the observation that, in early embryos (E12.5), virtually all α cells contain vimentin, whereas, in later gestational ages, the amount of vimentin-negative glucagon-containing cells (mature α cells) rapidly increases, outnumbering vimentin-immunoreactive α cells. Despite this, it is to be noted that soon before and after birth, these transitional cells can be easily found as if a wave of newly differentiating α cells occurred at that age. Indeed, the dynamic of islet cell growth has been previously studied, and it is characterized by a rapid increase in the α cell population during late rat fetal life (McEvoy and Madson 1980), mainly because of the addition of newly differentiated cells rather than to the proliferation of preexisting α cells (Kaung 1994).
The absence of other transitional forms of immature endocrine cell types containing vimentin (other than α cells) might suggest that vimentin expression is restricted only to previously α-committed precursor cells. However, the large number of vimentin-immunoreactive duct cells in late gestational ages does not support this hypothesis, and it is quite possible that even non-α endocrine cells may arise from vimentin-containing precursors after losing vimentin. Lineage tracing studies will probably be needed to clarify this point.
Footnotes
Acknowledgements
This study was supported by intramural funds from the University of Siena (PAR Servizi 2005–2006) to E.B. and L.F.
We thank Prof. G. Lungarella for the use of the Zeiss confocal microscope.
