Abstract
In this study we examined the expression of nestin in islets, the exocrine part, and the big ducts of the adult human pancreas by immunofluorescent double staining. Two different anti-nestin antisera in combination with various pancreatic and endothelial markers were employed. Nestin-immunoreactive cells were found in islets and in the exocrine portion. All nestin-positive cells co-expressed the vascular endothelial markers PE-CAM-1 (CD31), endoglin (CD105), and CD34 as well as vimentin. Endocrine, acinar, and duct cells did not stain for nestin. We also demonstrated that in the area of big pancreatic ducts, nestin-positive cells represent small capillaries scattered in the connective tissue surrounding the duct epithelium and do not reside between the duct cells. We detected nestin-expressing endothelial cells located adjacent to the duct epithelium where endocrine differentiation occurs. We have shown that nestin is expressed by vascular endothelial cells in human pancreas, and therefore it is unlikely that nestin specifically marks a subpopulation of cells representing endocrine progenitors in the adult pancreas.
N
In several in vitro differentiation assays using embryonic and adult stem cells, nestin has been used as a selection marker for neuronal (Lee et al. 2000) and pancreatic endocrine (Lumelsky et al. 2001) precursor cells. In the adult pancreas, nestin cells were initially described as a specific subpopulation of cells located in islets with possible stem-cell function (Hunziker and Stein 2000; Zulewski et al. 2001). Furthermore, it has been reported that nestin cells reside in the pancreatic ducts, suggesting their role as possible endocrine progenitors (Zulewski et al. 2001). Isolation of the nestin-expressing cells from rat and human islets and their in vitro differentiation into pancreatic endocrine and exocrine cells led to the suggestion that nestin-positive cells had a role as multipotent pancreatic stem cells (Zulewski et al. 2001). Conversely, recent studies have demonstrated that nestin-positive cells do not serve as endocrine precursors either during pancreas development in mice, rat, and human (Lardon et al. 2002; Piper et al. 2002; Selander and Edlund 2002) or in a regenerating model of adult rat pancreas (Lardon et al. 2002). Furthermore, it was shown that nestin is exclusively expressed in mesenchymal cells surrounding the developing pancreatic epithelium (Lardon et al. 2002; Selander and Edlund 2002). Using mainly morphological characterization, it was demonstrated that nestin is mostly expressed in stellate cells aligning endothelial cells in the normal rat pancreas and that under regenerating conditions nestin is additionally expressed in endothelial cells of neocapillaries (Lardon et al. 2002).
The aim of this study was to thoroughly investigate nestin immunoreactivity in the adult human pancreas by using double fluorescence staining to further clarify the type of cells expressing nestin.
Materials and Methods
Human Materials
Human adult pancreases from heart-beating cadaveric non-diabetic donors were procured at European hospitals associated with the Eurotransplant Foundation (Leiden; The Netherlands) and with the beta cell bank of the JDRF Center, a European multicenter program involving beta-cell transplantation in type 1 diabetic patients. The data reported in this study are based on tissue samples taken from the body of two different pancreata. The donors were female with ages 45 years and 30 years.
Fixation and Tissue Preparation
Materials used for histological analysis were fixed either in 4% paraformaldehyde (PFA) in Ca2+- and Mg2+-free PBS (CMF–PBS) for 4 hours at room temperature (RT) or overnight in formalin at 4C before paraffin embedding or cryoprotection, respectively. Paraffin sections of 4 μm were cut with a microtome and stored at RT. Frozen tissue was generated by transferring the fixed specimen in 30% sucrose (Merck; Darmstadt, Germany) in CMF-PBS at 4C for 1 day (cryoprotection) followed by embedding in Tissue tek (Sakura Finetek Europe; Zoeterwoude, The Netherlands) and storage at − 80C. Cryosections of 6-8-μm thickness were cut on a cryostat, mounted on poly-lysine-coated slides (Menzel-Gläser; Braunscheig, Germany), and stored at − 80C. All pictures shown in this study were done on formalin-fixed frozen sections.
Immunohistochemistry
Cryosections were dried at RT for 20 min, washed in CMF and blocked with 10% donkey or goat non-immune serum (Jackson ImmunoResearch Laboratories; West Chester, PA) for 30 min. For paraffin sections, the slides were dewaxed in xylene and rehydrated through a descending ethanol series. Antigen retrieval was accomplished by microwave treatment (two times for 5 min at 600 W in 0.01 mol/liter citrate buffer, pH 6.0) followed by three washes in CMF–PBS. The dilution of the blocking sera and the antibodies was carried out by using CMF–PBS supplemented with 0.3% bovine serum albumin (Sigma, St Louis, MO), 0.3% Triton X-100 (Sigma), and 0.05% saponin (Sigma). After blocking of nonspecific binding, the primary antibodies were applied at the proper concentration (Table 1) for 2 hr at RT followed by three washes in CMF–PBS. Sections were then incubated with the secondary antibodies (Table 2) together with 100 ng/ml 4-diamidino-2-phenylindole (DAPI; Merck) for 30 min at RT and washed three times in CMF–PBS. Slides were coverslipped with mounting medium (KPL) for fluorescence microscopy. Images were collected by using an Olympus BX51 microscope (Olympus Optical; Tokyo, Japan) equipped with a Hamamatsu chilled color 3CCD camera (Hamamatsu Photonics; Solna, Sweden), and images were processed using the IMAGE-Pro 4.5 PC software package (Media Cybernetics; Silver Spring, MD). No specific signal was detected when the primary antibodies were omitted (data not shown).
Primary antibodies
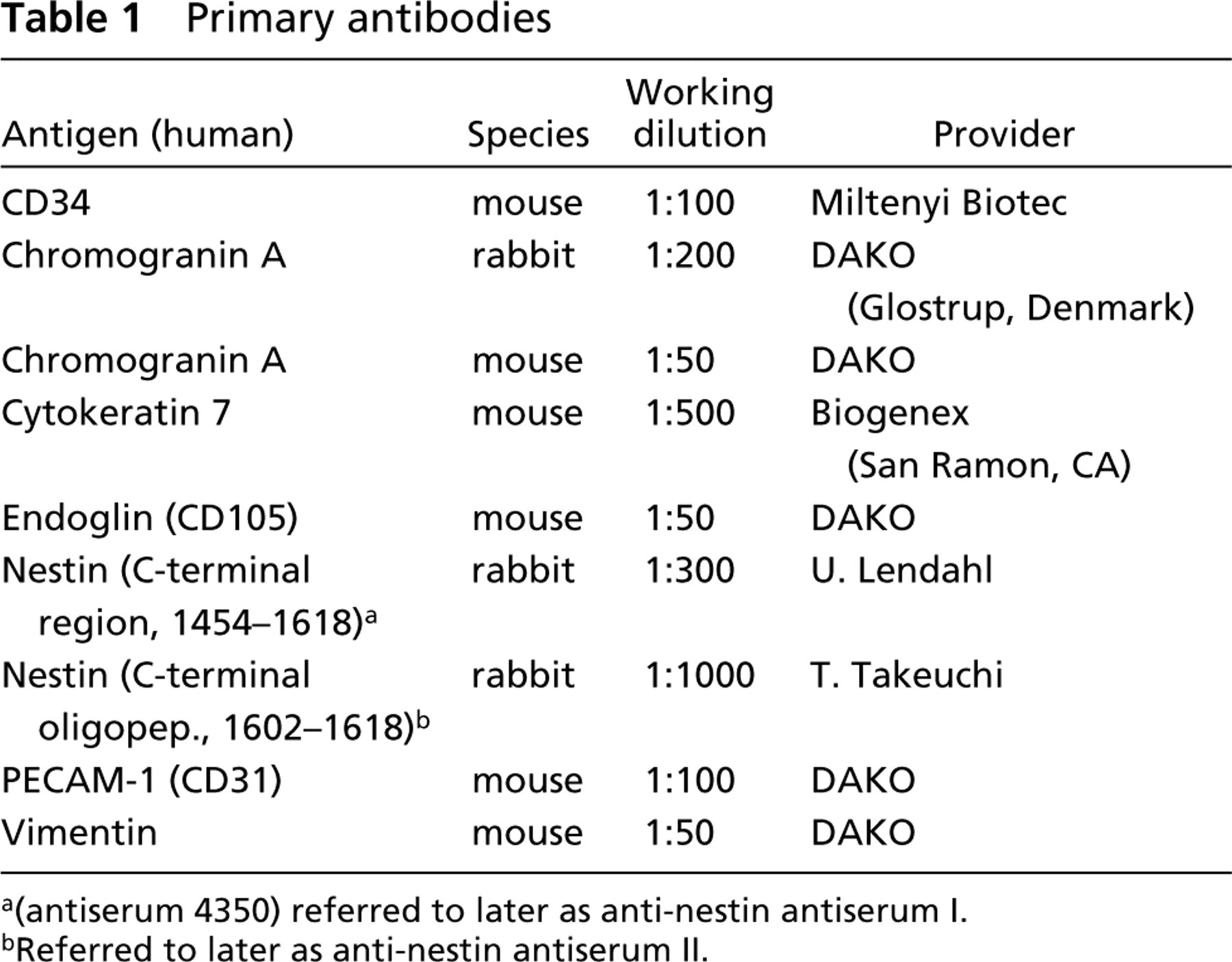
a(antiserum 4350) referred to later as anti-nestin antiserum I.
bReferred to later as anti-nestin antiserum II.
The two anti-nestin antisera (Grigelioniené et al. 1996; Sugawara et al. 2002) used in this study showed a comparable staining pattern on formalin-fixed tissue. On adjacent sections they stained the same type of cells (data not shown). Nevertheless, in our hands the U. Lendahl antiserum 4350 (anti-nestin antiserum I) gave the best results, whereas the antiserum from T. Takeuchi (anti-nestin antiserum II) showed a weaker staining intensity for nestin in the islets and the exocrine part, but was equally good in detecting nestin cells in the connective tissue surrounding the large ducts. The specific anti-nestin antisera used for the stainings in this study are indicated in the figure legends.
By testing the antisera on differently fixed tissues we could observe that especially formalin-fixed, frozen and, to a certain extent, paraffin-fixed tissue showed the most convincing results concerning staining intensity as well as the specific filamentous staining pattern of nestin. Using PFA-fixed frozen and paraffin pancreas from the same specimen the nestin immunoreactivity was moderate to very weak and revealed a more unspecific pattern throughout the whole pancreas.
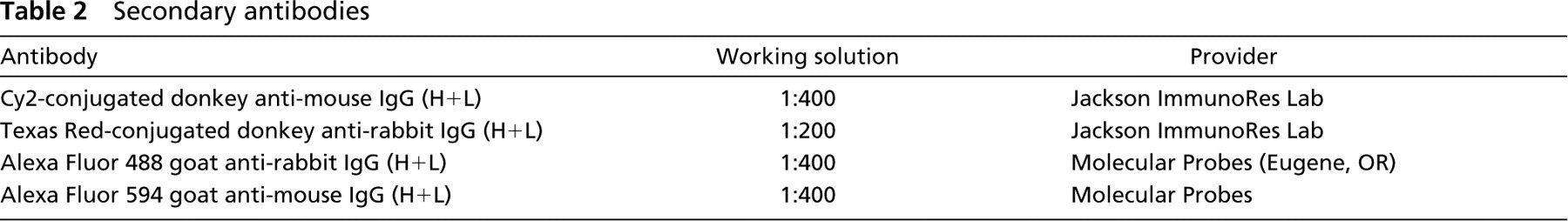
Secondary antibodies
Results
Nestin Expression in Pancreatic Islets of Langerhans
We first examined for nestin localization in pancreatic islets. Nestin immunoreactivity could be found in a subpopulation of cells in and around the islets. The cells expressing nestin were either single or more often organized in small longitudinal groups of cells (Figures 1A, 1E, and 1I). The numbers of nestin cells and their localization within and around the islet varied. Some islet sections did not show any nestin immunoreactivity. No co-expression of nestin and chromogranin A, a marker of endocrine cells, could be detected (Figure 1H). Further immunofluorescent double staining revealed that, in the pancreatic islets, nestin-positive cells co-expressed the vascular endothelial markers PECAM-1 (platelet/endothelial cell adhesion molecule-1), endoglin, and CD34 (Figures 1A–1C, 1E–1G, and 1I–1K). Notably, the staining pattern of nestin and PECAM-1 was very similar. We found no evidence to suggest that nestin-positive/PECAM-1-negative cells were present. Nestin also co-localized with endoglin and CD34, although not all endoglin and CD34 immunoreactive cells stained positive for nestin (Figures 1E–1G and 1I–1K). We observed that in islets nestin was co-localized with vimentin, which mainly marks cells of mesenchymal origin, but we also found several vimentin-positive/nestin-negative cells next to the double-labeled cells (data not shown). Figures 1C and 1D illustrate that the nestin/PECAM-1 double-stained cells mark a distinct subpopulation of the vi-mentin-expressing cells on the adjacent islet stained section. Chromogranin A immunoreactivity on adjacent sections was used to demonstrate the islet area of the nestin staining (Figures 1D, 1H, and 1L).
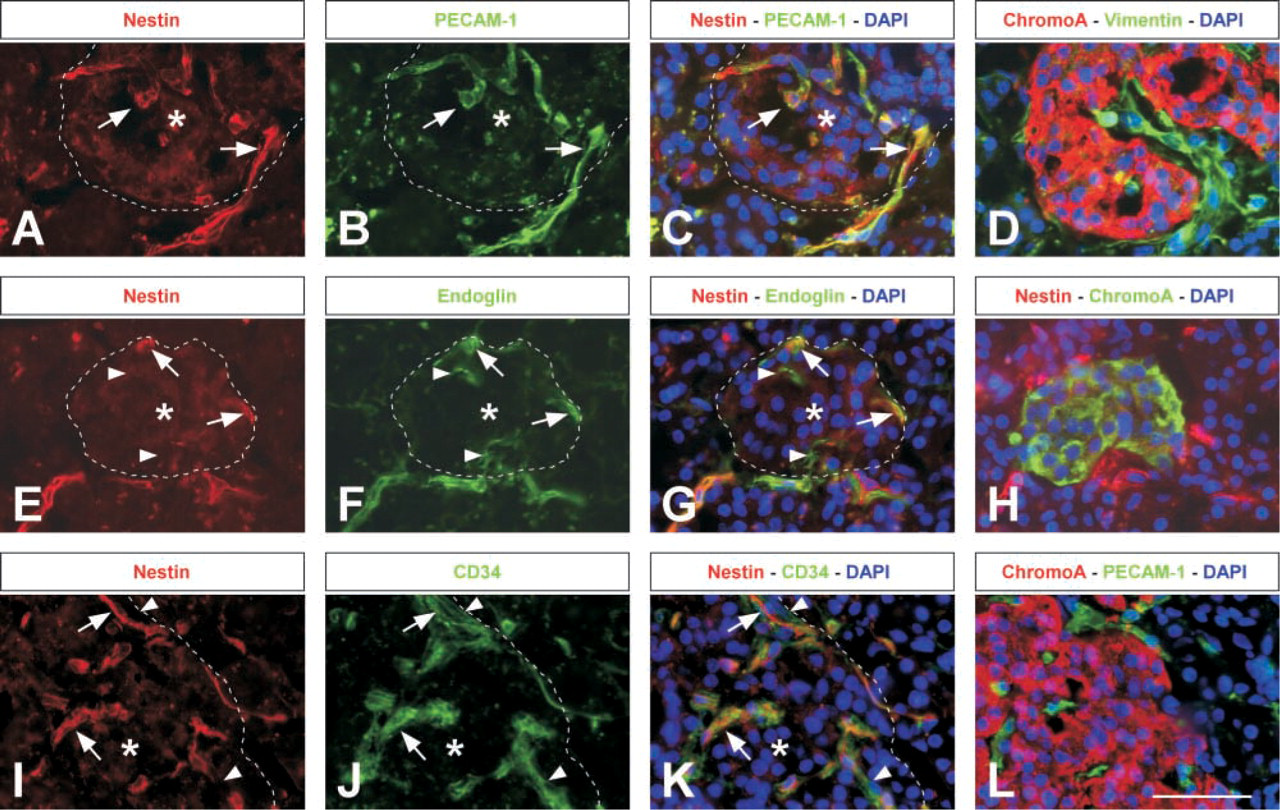
Immunohistochemical detection of nestin in the endothelial cells of human pancreatic islets. (
Expression of Nestin in the Exocrine Part of the Pancreas
To further investigate nestin immunoreactivity in the pancreas, we performed a closer examination of the exocrine part. Nestin was co-localized neither with the acinar marker amylase (Figures 2A and 2D) nor with cytokeratin 7 (CK7), a marker of pancreatic duct cells (Figures 2L–2N). We could find nestin expression frequently in capillary-like structures as well as in some single cells scattered in the exocrine region and often located next to lobules and acini (Figures 2A, 2E, 2I, and 2L). All nestin-positive cells co-expressed PECAM-1 (Figures 2A–2C), CD34 (Figures 2I–2K), vimentin (Figures 2E–2G), and endoglin (data not shown). Occasionally, we could detect endoglin (data not shown) as well as CD34-expressing cells and very rarely PECAM-1 (data not shown)-positive cells that did not stain for nestin in the exocrine part of the pancreas (Figures 2I–2K). In contrast, vimentin was detected in a broader population of cells, which did not express nestin (Figures 2E–2G).
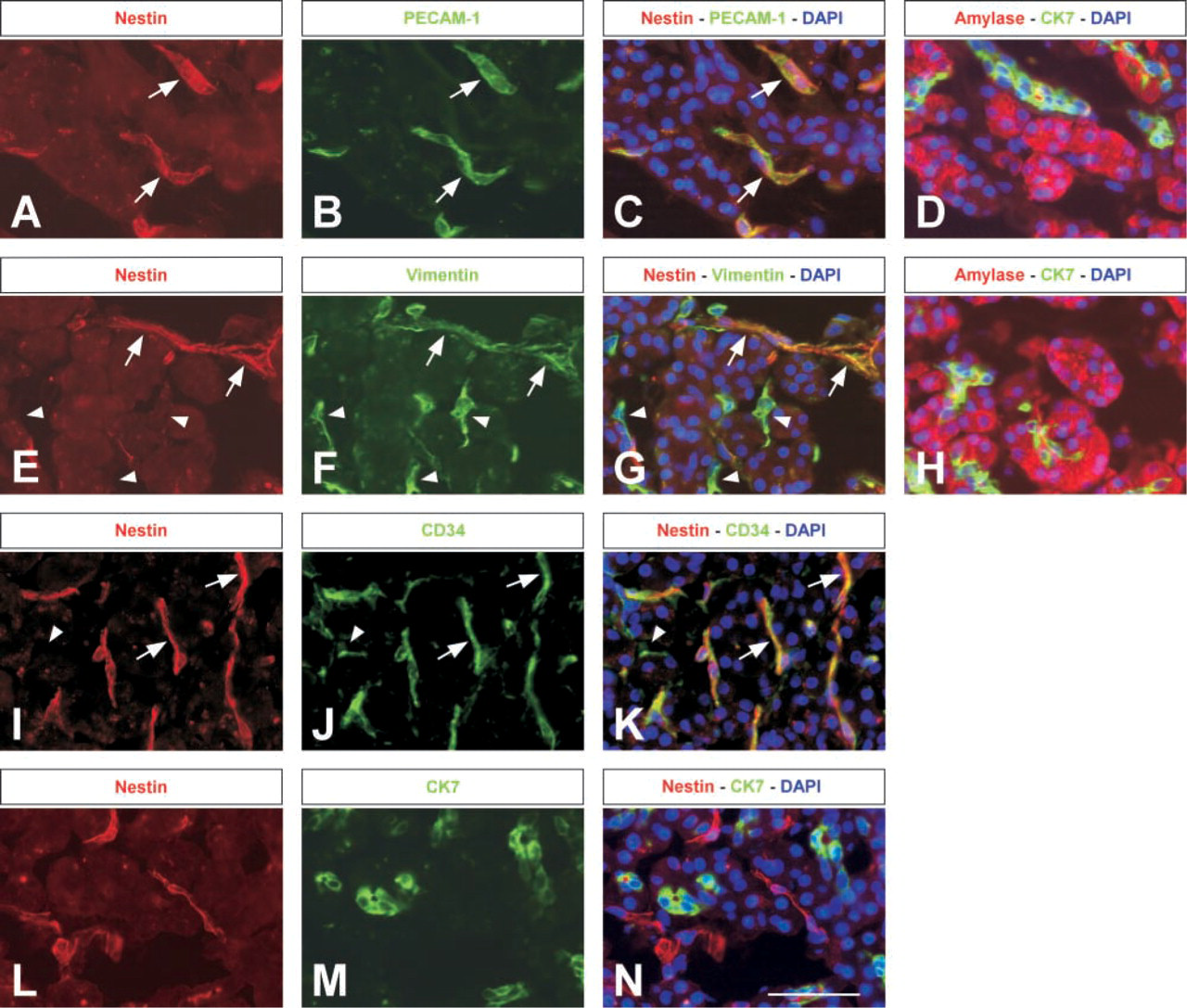
Nestin immunoreactivity in the endothelial cells of the human exocrine pancreas. (
Nestin Expression in the Large Ducts
In the large interlobular pancreatic ducts, the duct epithelium is surrounded by a thick layer of connective tissue, bordering the exocrine lobules. This is illustrated by double staining for vimentin and amylase (Figure 3M). On the adjacent sections we examined nestin expression. Using anti-nestin antiserum I, we detected nestin immunoreactivity in single cells and in small cell groups that were dispersed in the connective tissue and in several cases were very close to the duct epithelium (Figures 3A, 3E, and 3I). Similar to our previous observations in the exocrine and endocrine part of the pancreas, the nestin-positive cells co-expressed PECAM-1 (Figure 3) as well as endoglin, CD34, and vimentin (data not shown). Using a second anti-nestin antiserum (antiserum II), we obtained comparable results as with antiserum I (Figures 3I–3K). Endothelial cells of larger vessels in the periphery of the connective tissue did not show any nestin immunoreactivity (data not shown). Double immunostaining on consecutive sections revealed no co-expression of nestin and the duct marker CK7. However, in several cases nestin-positive cells were in close proximity to the duct epithelium and sometimes they were immediately adjacent to it (Figures 3D, 3H, and 3L). We could not detect any nestin expression in cells integrated in the duct epithelium or delaminating from it.
Next we investigated if nestin is transiently expressed in duct regions that give rise to endocrine cells. For that reason we examined areas where developing endocrine cells occasionally bud off from the epithelium in the large ducts. Double immunofluorescence staining showed that in these local regions chromogranin A-immunoreactive (endocrine) cells were mostly seen as a second cell layer on the peripheral side of the CK7-positive duct epithelium but still attached to it (Figures 4A–4C). Some cells co-expressed both markers, often in an arrangement where chromogranin A was expressed only or to a stronger extent distally, whereas CK7 immunoreactivity was found more proximal to the epithelium. Analysis of nestin and chromogranin A immunoreactivity on consecutive sections showed that nestin was not expressed in the maturing cell types but was frequently located in cells of the connective tissue neighboring the areas of budding endocrine cells (Figures 4D–4F). These adjacent nestin-positive cells co-expressed PECAM-1 (data not shown).
We also detected nestin in later stages of the duct-endocrine budding process, where it was expressed either in cells aligning endocrine aggregates still attached to the duct or was found in large, already vascularized clusters placed in the outside edge of the connective tissue (data not shown).
Discussion
Using two different anti-human nestin antisera and various established endothelial markers, we show by double staining that nestin is expressed in endothelial cells of the adult human pancreas. Nestin-positive cells could be found in islets of Langerhans, the exocrine region, and scattered in the connective tissue surrounding large interlobular pancreatic ducts. In all cases, nestin immunoreactivity was co-localized with the endothelial markers PECAM-1, endoglin, and CD34, as well as vimentin. In large blood vessels nestin was expressed at low levels or was absent. Both nestin antisera gave a comparable immunoreactivity pattern. Occasional discrepancies in the staining intensity might be explained by the varying affinity of the antisera.
Apart from the larger blood vessels, we observed that the expression pattern of nestin was very similar to PECAM-1 in the different parts of the pancreas. PECAM-1 is a cell adhesion molecule which is strongly expressed by vascular endothelial cells of normal tissue and in proliferating blood vessels of a variety of tumors (Parums et al. 1990; Scholz and Schaper 1997). A few PECAM-1-positive cells that did not express nestin were observed. These cells could be leukocytes, which have been shown to express PECAM-1 (Liao et al. 1995). Nestin also co-localized with endoglin, which is a component of the transforming growth factor (TGF)-beta receptor complex (Cheifetz et al. 1992). It has been shown that endoglin is expressed on endothelial cells of normal and especially proliferating blood vessels and is implicated in endothelial cell function (Gougos and Letarte 1990; Kumar et al. 1996). Finally, we found that nestin is co-expressed with CD34, a glycosylated transmembrane protein, which has been reported to be expressed on vascular endothelial cells (Fina et al. 1990) and human hematopoietic stem cells (Civin et al. 1984). Nestin-positive cells always expressed both endoglin and CD34. However, in comparison to PECAM-1 we observed a higher number of endoglin- and CD34-expressing cells, which did not co-localize with nestin. These cells varied in staining intensity and had often a stroma-like appearance. It has been reported that, in some tissues, endoglin and CD34 mark not only vascular endothelial cells but also fibroblasts and leukocytes, which could explain the additional occurrence of endoglin-positive/nestin-negative and CD34-positive/nestin-negative cells (Nakayama et al. 2000; Balza et al. 2001; O'Connell et al. 1992). However, it can neither be excluded that a minute number of PECAM-1, CD34, and endoglin-positive endothelial cells of small blood vessels do not express nestin nor that nestin is expressed in a small subpopulation of non-endothelial cells that are also positive for the tested endothelial markers. Nestin has recently been shown to be expressed in several fibroblast cell lines (Steinert et al. 1999).
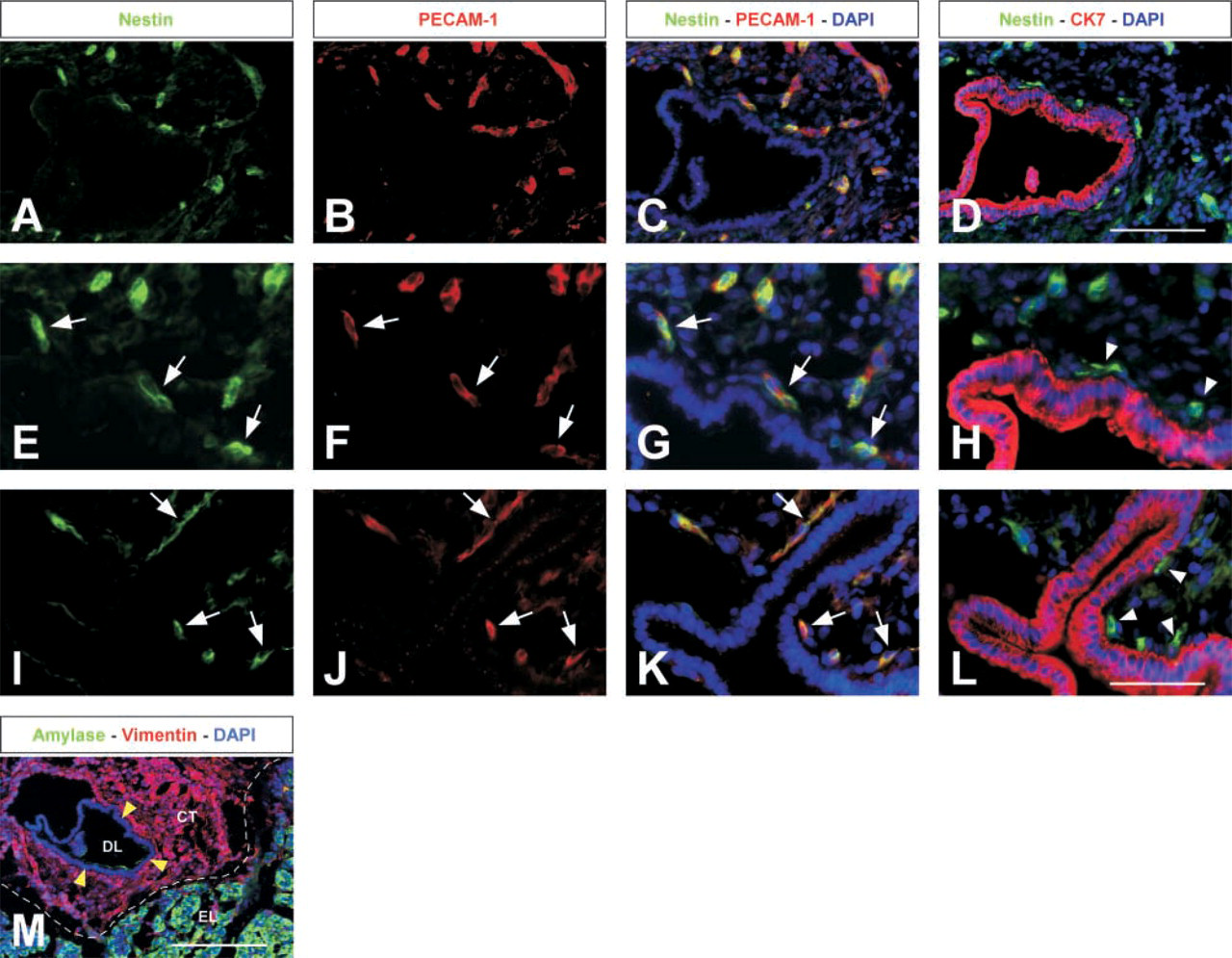
Nestin expression in endothelial cells of the large human pancreatic ducts. (
The endothelial localization of nestin was further substantiated by staining for vimentin, a class III inter-mediate filament protein, which is mainly expressed in cells of mesenchymal origin, such as endothelial cells, fibroblasts, and smooth muscle cells (Osborn et al. 1984). We discovered that nestin was expressed in a subpopulation of vimentin-positive cells. Compared to the expression pattern of the endothelial markers it appears that nestin co-localizes only with vimentin in endothelial cells. Fibroblasts of connective tissue showing strong immunoreactivity for vimentin did not express nestin. Interestingly, it has been reported that proper polymerization of nestin filaments requires vimentin (Eliasson et al. 1999; Steinert et al. 1999).
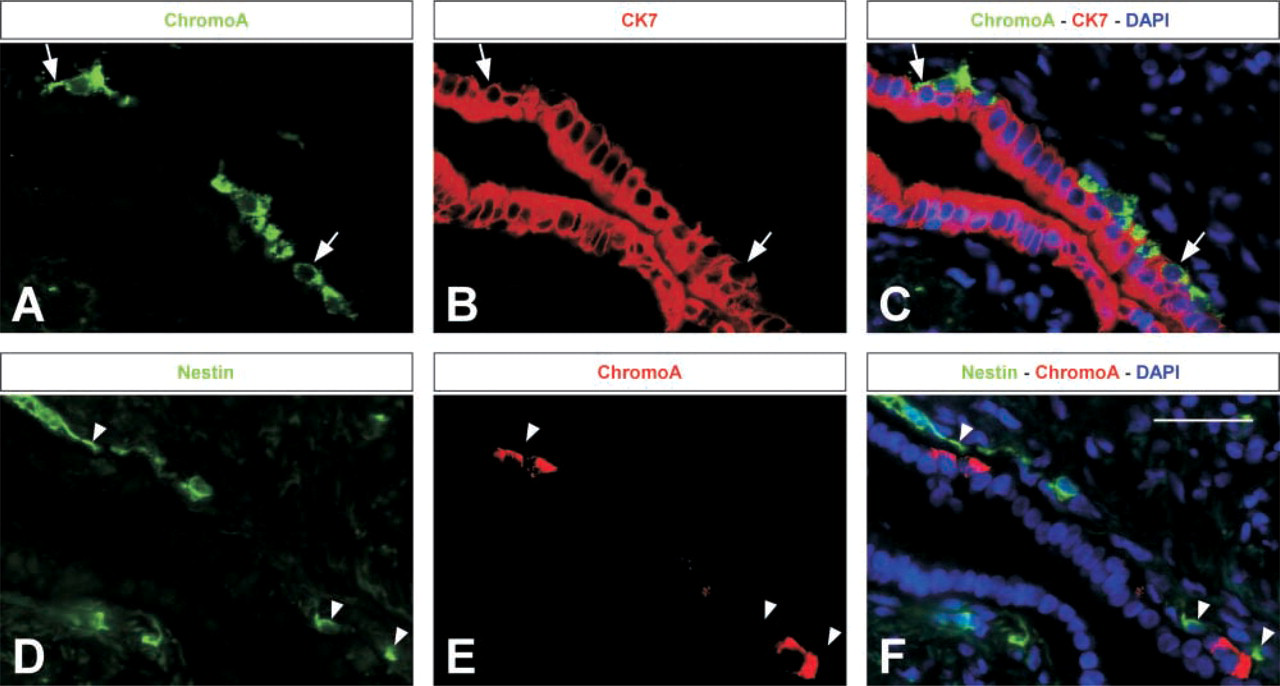
Nestin cells do not give rise to endocrine cells in the large ducts. (
PECAM-1, CD34, and recently endoglin and nestin have been used as promising prognostic markers in a variety of tumor types because of their enhanced expression in activated and proliferating endothelial cells (Tanigawa et al. 1997; Vindigni et al. 1997; Tanaka et al. 2001; Sugawara et al. 2002). It has further been shown that nestin is highly expressed in newly formed endothelial cells of rat embryos but significantly reduced in blood vessels of adult tissue (Mokry and Nemecek 1998). Similarly, Lardon et al. (2002) have recently described that nestin is upregulated in mesenchymal cells during regeneration of the rat pancreas after duct ligation. Using mainly morphological criteria, nestin expression was reported to be expressed in endothelial and stellate cells, a special kind of pericyte, during the process of active angiogenesis. Remarkably, they could detect nestin in normal rat pancreas only at a low frequency and mainly in stellate cells associated with blood capillaries in close proximity to endothelial cells. Because we did not check for further markers of pericytes we cannot rule out that in the human pancreas nestin also co-localizes with that special type of cells. However, the distinct staining pattern of nestin, its consistent co-localization with the tested endothelial markers and its low expression in large blood vessels suggests that nestin preferentially marks endothelial cells of capillaries in the adult human pancreas. In addition, the fairly frequent occurrence of nestin immunoreactivity and the fact that in adult pancreas the turnover of endothelial cells is relatively low (Engerman et al. 1967) suggest further that the vast majority of these nestin-positive capillaries do not represent newly formed vessels.
Recently, nestin expression has been described in the developing mouse, rat and human pancreas. In all studies the absence of nestin in the developing pancreatic epithelium was demonstrated and suggested that nestin-positive cells do not serve as pancreatic precursor cells during development (Lardon et al. 2002; Piper et al. 2002; Selander and Edlund 2002). The latter group have further shown in mouse that nestin immunoreactivity was found in the mesenchymal cells surrounding the epithelial structures at early stages. Later in development, nestin-positive cells were dispersed between the epithelial cells of the growing pancreas. In addition, they demonstrated that markers for endocrine progenitor cells were located only in the pancreatic epithelium. Similarly, in the developing rat pancreas, Lardon et al. (2002) have shown the presence of nestin in mesenchymal cells and, judged by their morphological appearance, they suggested that it was endothelial capillaries expressing nestin and surrounding the duct epithelium. Likewise, our results show that nestin immunoreactivity is associated with small cell groups scattered in the connective tissue around the epithelium of large interlobular ducts and in several cases aligning with it. It has earlier been shown in rat pancreas that big ducts are surrounded by a plexus of capillaries enclosing the epithelial tube and proceeding along it for proper blood supply (Egerbacher and Böck 1997). Co-localization with endothelial markers suggests that nestin is expressed by capillary endothelial cells of the periductal vascular system.
In the adult pancreas the duct epithelium is generally believed to be one source to replenish the beta-cell mass during the turnover process of endocrine cells (Bouwens and Pipeleers 1998; Bonner-Weir 2000). We demonstrate in this study that nestin expression was absent in epithelium of pancreatic ducts since it could neither be co-localized with duct cell marker CK7 nor seen in cells residing in the epithelium. In contrast, Zulewski et al. (2001) found in the adult rat pancreas nestin expression also by cells existing in the duct epithelium and partially co-expressing the duct epithelial marker CK19. They further suggested that these nestin-positive cells integrated in the ducts might represent distinct islet progenitors.
To explain this discrepancy we tried to investigate specific nestin expression in regions where endocrine cells develop from the large duct epithelium. Examination of these focal areas revealed that nestin was co-expressed neither with CK7 nor with chromogranin A. Instead, we found nestin immunoreactivity in small capillaries frequently aligning the differentiating cells from the stromal side. The co-expression of both ductal and endocrine markers in some of the differentiating cells developing from the large duct epithelium suggests that CK7 positive duct cells represent the endocrine progenitors. Similar observations have been reported previously by Bouwens and coworkers, who demonstrated further that especially in human pancreas also small intercalated duct cells of the exocrine part give rise to minor units of beta-cells (Bouwens and Pipeleers 1998). In contrast to Zulewski et al. (2001), we could detect nestin neither in the ductules nor in centroacinar cells, but occasionally adjacent to them and co-localized with the endothelial markers (data not shown).
Recently, Lammert et al. (2001) demonstrated in mouse and frog that endothelial signaling to the adjacent endoderm is important for endocrine cell differentiation during pancreas development. The critical role of mesenchymal–epithelial signaling during organ development has also been described in other tissues (Matsumoto et al. 2001; Serluca et al. 2002). Similarly, it could be considered that in the adult pancreas under certain conditions endocrine differentiation from duct cells is initiated by signals from adjoining endothelial cells. How such a focal activation of endo-thelial cells surrounding the duct might be accomplished and whether all or only a distinct subpopulation of CK7-positive duct cells may be sensitive to possible signals from aligning adjacent capillaries is very speculative and requires more investigation.
Thus, in agreement with the studies on the developing pancreas our results support the hypothesis that nestin is not a marker for endocrine precursors of the ducts but nestin cells might indirectly be involved in the endocrine differentiation process by releasing initiation signals to the duct epithelia since they represent adjacent capillaries.
Potential progenitor cells residing in adult pancreatic islets have been discussed as another source for new beta cells (Guz et al. 2001; Petropavlovskaia and Rosenberg 2002). When nestin was first discovered in pancreas it was shown to be expressed in a subset of cells in mouse pancreatic islets and, based on the similarities between neural and pancreatic development, it was proposed that the neural stem cell marker nestin might be a marker for pancreatic progenitors (Hunziker and Stein 2000). Later, Zulewski et al. described nestin-immunoreactive cells in adult rat and human islets (Zulewski et al. 2001). In contrast to our results in human pancreas, where we used the established endothelial markers PECAM-1, endoglin, and CD34 as well as vimentin, Zulweski et al. showed that nestin is not coexpressed in rat islet vascular endothelium utilizing collagen IV as an endothelial marker. By in vitro isolation and subcloning of nestin cells from islets of rats and human, they demonstrated that under specific conditions the isolated cells showed a certain ability to express a variety of pancreatic endocrine, exocrine, and liver markers.
In our study we noted that the occurrence of nestin varied in the different islet areas and that nestin expression was more frequently seen in larger islet sections, where more endothelial cells were present compared to the small islet regions presenting a lower occurrence of capillaries. Usually nestin expression was observed in islets as small groups of endothelial cells lying scattered between the endocrine cells. In certain instances, islets were sectioned in a region where whole nestin-positive capillaries infiltrating the islets could be seen. Nestin immunoreactivity could not be detected in islet sections where vascular endothelium was absent. Our findings suggest that using nestin as a marker for isolation of endocrine progenitors would lead to a specific accumulation of endothelial cells mainly from microvessels and it is not unlikely that potential islet cell progenitors adjoining capillaries might be carried along in the isolation procedure.
Footnotes
Acknowledgements
Supported by grants from the Danish Research Council (Center for Transgenic Animals) and from the Juvenile Diabetes Research Foundation (JDRF Center of Beta Cell Therapy in Europe).
We are grateful to Urban Lendahl and Toshiyuki Takeuchi for supplying us with the anti-nestin antisera.
