Abstract
Traditional histological descriptions of the pancreas distinguish between the exocrine and the endocrine pancreas, as if they were two functionally distinct glands. This view has been proven incorrect and can be considered obsolete. Interactions between acinar and islet tissues have been well established through numerous studies that reveal the existence of anatomical and functional relationships between these compartments of the gland. Less attention, however, has traditionally been paid to the relationships occurring between the endocrine pancreas and the ductal system. Associations between islet tissue and ducts are considered by most researchers as only a transient epiphenomenon of endocrine development. This article reviews the evidence that has emerged in the last 10 years demonstrating the existence of stable, close, and systematic relationships between these two pancreatic compartments. Functional and pathophysiological implications are considered, and the existence of an “acinar-duct-islet” axis is put forward. The pancreas appears at present to be an integrated organ composed of three functionally related components of well-orchestrated endocrine and exocrine physiological responses.
Keywords
T
Associations between Ducts and Endocrine Pancreas Are Not Exceptions but the Rule
Depending on the number of endocrine cells clustered together, a rough distinction can be made among scattered single endocrine cells, small buds of endocrine tissue (not penetrated by blood capillaries), and islets of Langerhans.
Single Endocrine Cells
Scattered and isolated endocrine cells within the exocrine parenchyma and the duct epithelial lining have long been recognized (Bensley 1911). The documentation that a consistent number of these cells are located within the epithelial lining of the ducts is, however, more recent (Bendayan 1987). Detailed studies carried out on adult rat pancreas have shown that all four major types of pancreatic endocrine cells [i.e., glucagon-, insulin-, somatostatin- and pancreatic polypeptide- secreting (PP) cells] can be found as single cells intermingled with duct cells (Park and Bendayan 1992; Bertelli et al. 1994). In addition, distal to the confluence of the pancreatic and bile duct systems (i.e., in the common pancreatic-bile duct), serotonin- and cholecystokinin (CCK)-containing cells can also be observed (Park and Bendayan 1992). Remarkably, in diabetic animals CCK- and somatostatin-immunoreactive cells located in the common pancreatic-bile duct double in number (Park and Bendayan 1994). This observation acquires importance because somatostatin has been reported to influence the tonus of Oddi's sphincter (Ahrendt et al. 1992; Binmoeller et al. 1992) and also because islets of Langerhans containing somatostatin-secreting cells have been demonstrated in the submucosal layer of the duodenum close to the Oddi's sphincter (Bendayan and Park 1991,1997), where the epithelium of the common bile duct is also rich in somatostatin-containing cells (Park and Bendayan 1994). In normoglycemic animals, the topographical distribution of the four types of islet cells scattered within the epithelium displays an almost even distribution of insulin- and glucagon-producing cells along the entire duct tree, whereas the incidence of PP and somatostatin-secreting cells gradually increases toward the more distally located portions of the duct system (Park and Bendayan 1992). Most of these endocrine cells are classified as “open type” cells, because they face the duct lumen with a variable area of cell surface (Figure 1). The apical pole of these cells is provided with short microvilli and coated pits, as well as with a rich store of smooth and coated vesicles that suggest an active exchange with the pancreatic juice (Bertelli et al. 1994). In addition, some of these cells display large numbers of secretory granules in their apical pole (Figure 1). Scattered endocrine cells within the epithelial lining of the ducts have been studied in diabetic animals as well (Park and Bendayan 1994). Accordingly, a 50% decrease of insulin-secreting cells has been observed in small pancreatic ducts, whereas these cells virtually disappear from the common bile duct. In contrast, in small pancreatic ducts, glucagon-producing cells increase 4-fold, whereas the number of somatostatin-immunoreactive cells remains substantially unchanged (Park and Bendayan 1994). In some experimental situations of pancreatic regeneration, however, the number of endocrine cells scattered along the ducts increases (Wang et al. 1995; Waguri et al. 1997).
Human pancreas has been much less studied, inasmuch as investigation has been focused on insulin-secreting cells. Interestingly, although in rat pancreas, single insulin-producing cells associated with ducts represent only 1% of the total insulin-producing cell population (Wang et al. 1995), in humans, single and doublet insulin-secreting cells associated with the duct system account for 15% of all beta cells (Bouwens and Pipeleers 1998). In general, “ductal” insulin-producing cells are smaller than those in the islets (Wang et al. 1995; Bouwens and Pipeleers 1998).
Buds of Endocrine Tissue
A second arrangement of endocrine cells associated with the duct system can be found in adult rat pancreas. This arrangement has been referred to as “buds” (Bertelli et al. 1994); these consist of small clusters of endocrine cells predominantly associated with intercalated and intralobular ducts (Figure 2). These clusters of cells are formed of ∼20–25 cells and are continuous with the duct lining. From one edge, they protrude outside the duct; from the other, one or two cells abut on the lumen and are joined to the adjacent duct cells by well-developed intercellular junctions that guarantee ductal integrity. Buds of endocrine cells are composed mainly of insulin-secreting cells; a small number of glucagon- and somatostatin-secreting cells have also been reported in the most peripheral part of the largest buds. Accordingly, cells facing the lumen are always beta cells. The apical pole of these cells displays the same rich store of smooth and coated vesicles that characterizes single endocrine cells, which suggests an active exchange with the luminal content. A similar arrangement, even though not referred to as buds, seems to exist in humans as well (Bouwens and Pipeleers 1998). However, an electron microscopic demonstration that some of these clustered cells face the lumen in human tissue is lacking. Buds of endocrine cells have also been detected in pancreatic tissue of some experimental models of endocrine regeneration (Dutrillaux et al. 1982; Wang et al. 1994), but they are probably common to all models of endocrine neogenesis as an intermediate step leading to the development of islets. Unfortunately, without electron microscopic studies and/or appropriate stainings, buds probably have been overlooked and regarded as only “small islets.”
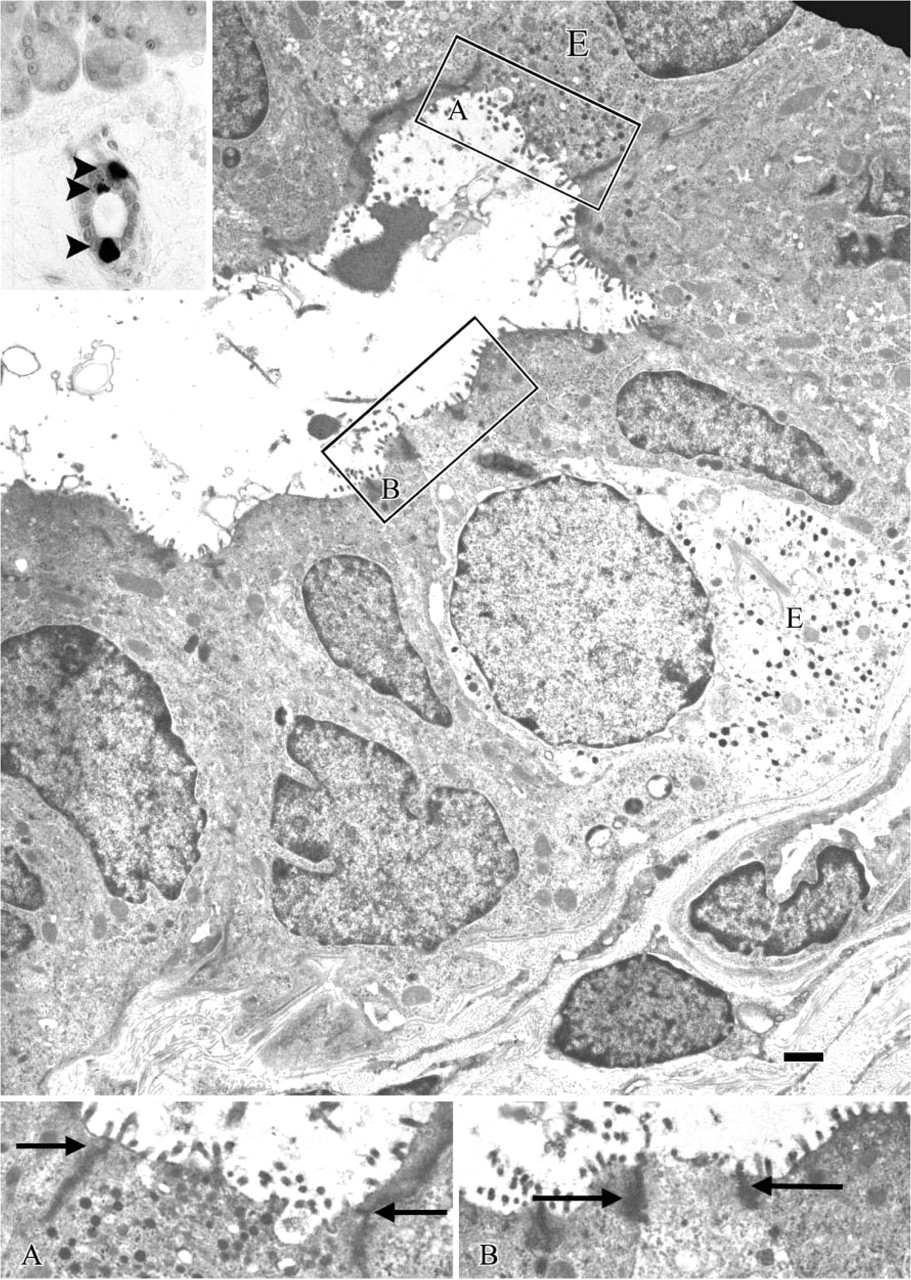
Electron micrograph of a small duct from a rat pancreas. Two endocrine cells (
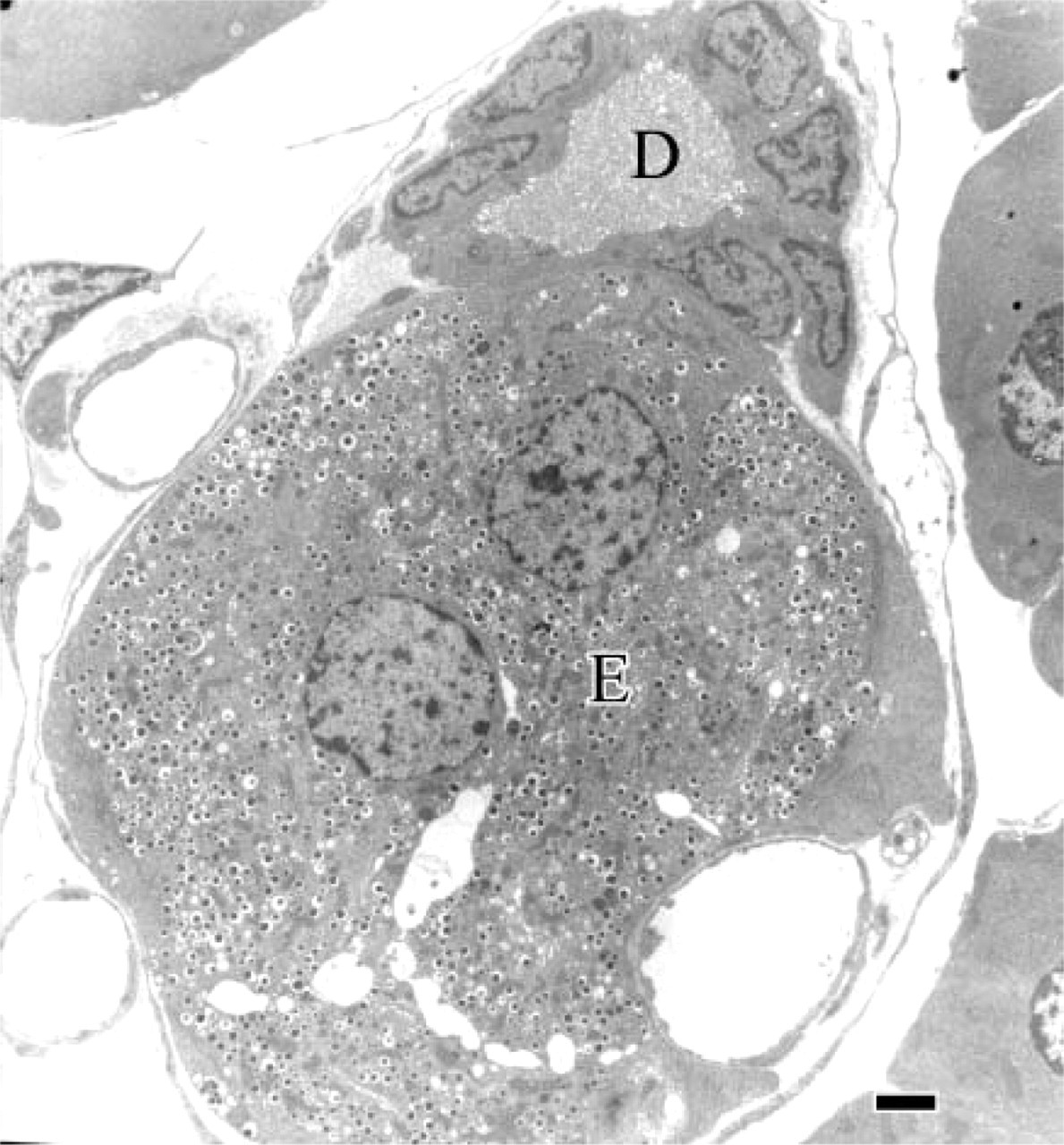
Electron micrograph of an intralobular duct from adult rat pancreas. The duct is provided with a small bud of endocrine cells (E). D, duct lumen. Bar = 2 μm.
Islets of Langerhans
What about islets of Langerhans? Are islets of Langerhans surrounded exclusively by acinar cells, or do they maintain connections with the duct tree? How extensive is their relationship with the duct system? Even though corroboration with more modern techniques is required, systematic association between islets and ducts was also reported in a pioneering study carried out on guinea pig pancreas (Bensley 1911). Further, light and electron microscope evidence of contacts between islets and centroacinar/ductular cells has been reported since 1986 (Leeson and Leeson 1986), but only a recent detailed study has revealed the extent of such relations (Bertelli et al. 2001). In rat pancreas, at least three islets out of four have multiple connections with the ductal system. Moreover, almost all the islets displaying definite relations with the duct system are in contact with centroacinar and/or small intralobular ducts (Figure 3A). These connections mainly involve glucagon- and somatostatin-secreting cells, because they are located toward the periphery of the islets. However, in some cases (6% of the islets), ducts penetrate the islets, making extensive contacts with the beta cell core (Figure 3B). Are these contacts an exclusive arrangement for the rat pancreas, or is this feature shared by the human pancreas as well? A three-dimensional computerized reconstruction of islet and duct distribution within human pancreatic lobules has been carried out, showing that half of the islets are located in close proximity to the duct tree. Yet only 1.3% of the islets demonstrated a clear attachment to the ducts (Watanabe et al. 1999). The use of conventional histological staining, instead of high-resolution immunohistochemistry with cell markers that clearly identify duct and islet cells, leads to an underestimation of the number of duct-islet contacts and overlooks most of the connections occurring between islets and centroacinar/ intercalar/intralobular duct cells. Such connections in rat tissue account for 93% of all the contacts (Bertelli et al. 2001). On the other hand, additional evidence seems to confirm that islets and ducts are connected in adult human pancreas. Indeed, during isolation of human islets, contamination by variable proportions of duct cells (20–60%) seems to be the rule (Keymeulen et al. 1998; Pavlovic et al. 1999). During the isolation procedure, the islets of Langerhans remain attached to those duct cells to which they are related in situ and these cells are considered contaminating elements. Finally, it is well known that the first technical step employed to isolate islets consists of the ductal retroinjection of collagenase. This technique results in a better recovery of clean islets, as compared with the chopped-tissue collagenase digestion or with the use of other pathways for collagenase delivery (i.e., vascular perfusion) (Wollheim et al. 1990). An investigation carried out with retroinjections of India ink into the duct tree (replacing and mimicking collagenase delivery) has demonstrated that in all animal species examined (monkey, dog, pig, rat), as well as in human, the pattern of staining distribution was similar as far as islets of Langerhans were concerned. The staining was systematically found around and even within the islet core, corroborating the concept of close and frequent relationships between the islets and the duct tree (Van Suylichem et al. 1992). All this indicates that such connections are a common feature in mammal pancreas.
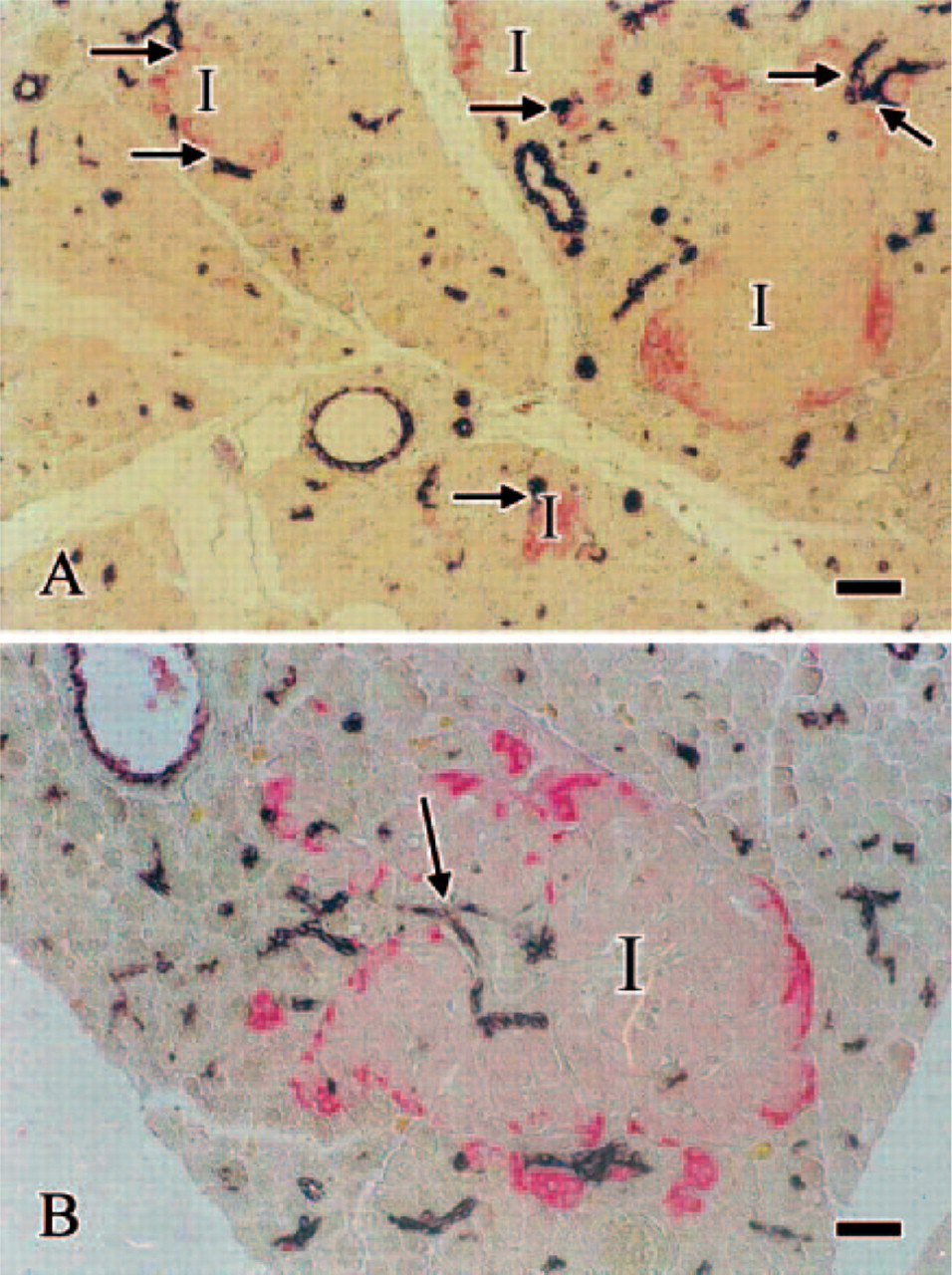
Double immunohistochemical reaction carried out with anti-cytokeratin 20 (duct marker, brown) and anti-glucagon (red) antibodies on a section of rat pancreas. The peripheral mantle of glucagon cells outlines the profile of the islets of Langerhans (I). (
Functional Implication of Endocrine-Duct Associations
It is well known that pancreatic exocrine secretions are partly under the control of the islets of Langerhans (Chey 1993). Historically, the first evidence of an islet influence on exocrine secretions was recognized by Jarotzky (1899). He reported the existence of peri-insular “halos” consisting of acinar cells surrounding the islets and displaying particular histological features. These peri-insular acini, in contrast to the tele-insular ones located far from the islets, are composed of larger cells containing many more zymogen granules (Bendayan and Ito 1979; Bendayan 1985). In addition, early observations of tissues of diabetic patients further supported the concept that islet hormones influence the exocrine pancreas. Pancreas from diabetic patients frequently displays signs of exocrine insufficiency, with reduction of bicarbonate and amylase output in response to secretin/CCK (Vacca et al. 1964; Balk et al. 1975; Domschke et al. 1975; Frier et al. 1976,1978; Sato et al. 1984; el Newihi et al. 1988). On the other hand, pancreatic exocrine deficiency caused by chronic pancreatitis is frequently associated with impaired glucose tolerance (Bank et al. 1975;) or overt diabetes (Nyboe Andersen et al. 1982; Kalthoff et al. 1984; Domschke et al. 1985; Cavallini et al. 1992). In spite of this, the currently more accepted view taken to explain the relationship between endocrine tissue and pancreatic ducts is that these contacts are merely an epiphenomenon of islet development. In other words, it is thought that after the first period of postnatal life, the main tide of endocrine differentiation from duct cells comes to an end, leaving the last differentiated endocrine cells as single cells or as small buds still connected to the ducts from which they arose. The fact that most of the mature islets of Langerhans maintain multiple connections with the duct system compels reconsideration of this position and demands further investigation to unveil the functional interactions between the two pancreatic entities. Connections between the endocrine tissue and the duct system increase the interactions among the different compartments of the gland (i.e., endocrine, acinar, and duct) via a previously unexpected number of pathways that will be emphasized below. These interactions highlight the complexity of the integrated pancreatic functions.
Islet Influence on Pancreatic Duct Secretion
The main secretory product of the pancreatic duct cells consists of a bicarbonate-enriched fluid. Secretion of this fluid is stimulated by nutriments, secretin, and CCK, but the contribution of the latter differs, depending on animal species. Bicarbonate secretion is related mainly to centroacinar and terminal duct cells (Case and Argent 1993), areas that were found to make contact with the endocrine tissue. Interestingly, in its interaction with the endocrine tissue, the duct tree penetrates the islet core with tiny ducts and contacts the periphery of the islets mainly through centroacinar cells and small intralobular ducts (Bertelli et al. 2001). It is likely, therefore, that a remarkable (yet-to-be-defined) proportion of bicarbonate-secreting cells are under the paracrine influence of islet cell secretion. The tissue distribution of the islets, buds, and single endocrine cells throughout the pancreatic parenchyma probably allows insulin and other islet hormones to reach duct cells via a true endocrine pathway as well. Small islets attached or located close to the ducts have been reported to contribute with their efferent vessels to the peri-ductular vascular plexus (Ohtani 1983). Furthermore, in rat and human pancreas, buds and single insulin-producing cells are frequently observed associated with small ducts and it is thought that they discharge insulin into the same capillary network that supplies the duct cells (Park and Bendayan 1992; Bertelli et al. 1994; Bouwens and Pipeleers 1998). Unfortunately, studies specifically designed to investigate the effects exerted by islet secretion on duct cells are few and mainly limited to the four major islet hormones. Moreover, the usual criteria chosen to examine duct response to a given hormone have always been restricted to the volume of fluid secretion and to the concentration of bicarbonate in pancreatic juice. However, hormones may also act in more subtle ways. Recent evidence, for instance, suggests an insulin involvement in the modulation of Ca2+ concentration in pancreatic juice (Ankorina-Stark et al. 2002). On the whole, our knowledge of the islet influence on duct cell physiology is fragmentary and largely incomplete. Nevertheless, this matter deserves a brief overview.
Four Major Islet Hormones. Insulin has a direct stimulatory action on fluid secretion. In isolated perfused rat pancreas, insulin significantly increases secretin-stimulated pancreatic juice secretion, but not protein secretion (Hasegawa et al. 1993). Anti-insulin antibodies, on the other hand, reduce spontaneous fluid secretion and suppress the increase evoked by a meal or by secretin/CCK infusion (Lee et al. 1990). In addition, insulin has been shown to dramatically increase the activity of the Na+/Ca2+ exchanger expressed by duct cells (Ankorina-Stark et al. 2002). Insulin probably acts directly on duct cells through saturable insulin-binding sites, as demonstrated by light and electron microscope autoradiography (Sakamoto et al. 1984).
The overall effect of glucagon on pancreatic acinar and duct secretions is, instead, inhibitory. However, the real physiological importance of this action is far from being established (Chey 1993). In isolated rat pancreas, glucagon significantly inhibits secretin-stimulated pancreatic juice secretion (Hasegawa et al. 1993). In conscious rats, glucagon reduces caerulein-evoked pancreatic secretion in terms of the volume of pancreatic juice, bicarbonate, and protein output (Biedzinski et al. 1987). In dog pancreas, glucagon mainly inhibits water and bicarbonate secretion in response to a meal (Singer et al. 1978). However, results of studies conducted on human subjects are contradictory (Fontana et al. 1975; Clain et al. 1978). Presently, it is impossible to establish whether these effects are due to the direct binding of glucagon to duct cells or to a modulating action exerted on the pancreatic nervous system. It is even possible that glucagon influences duct secretion by binding to secretin receptors, because the two hormones belong to the same “family” (Case and Argent 1993). Unfortunately, studies on the expression of glucagon receptors on duct cells are lacking.
Somatostatin most probably has a direct inhibitory effect on duct cells. It inhibits secretin-induced pancreatic juice secretion in rat perfused pancreas (Hasegawa et al. 1993) and reduces cyclic AMP increase in isolated guinea pig interlobular pancreatic ducts and in cultured duct epithelial monolayers (de Ondarza and Hootman 1995). Studies specifically designed to identify somatostatin receptors in duct cells have not been carried out.
PP has an inhibitory effect on enzyme secretions, whereas less-convincing evidence supports a similar action on bicarbonate and fluid secretion (Langlois et al. 1989). At any rate, this action seems to be exerted via neural pathways (Kohler et al. 1991; Masuda et al. 1994).
Non-classical Islet Hormones. In addition to the four classical islet hormones, islets of Langerhans secrete other bioactive peptides that exert true endocrine effects. In the last two decades, the number of known islet hormones has grown significantly. Among them, we can mention amylin, adrenomedullin (AM), calcitonin gene-related peptide (CGRP), C-peptide, pancreastatin, secretoneurin, ghrelin, cortistatin, orexin A, peptide YY, resistin, urocortin III, and the corticotrophin-releasing factor (CRF). They have been intensely studied, mostly in search of a role in the intra-islet regulation of hormone release. Their possible involvement in the modulation of duct activity has been overlooked. Table 1 summarizes the little information currently available.
AM, CRGP, and amylin belong to the same hormone family and bind with different affinity to the same strictly related receptors (Poyner et al. 2002). Currently, there is no solid information on AM, CRGP, or amylin binding sites in pancreatic duct cells.
The biological action of C-peptide, a 31-amino-acid peptide cleaved off from pro-insulin, has not been recognized until recently. Even though its receptor has not been identified, a true endocrine function is now attributed to C-peptide, because its binding to cell membranes activates signaling pathways (Wahren 2004).
Ghrelin and cortistatin bind to the growth hormone secretagogue receptor (GHS-R). However, cortistatin also binds to somatostatin receptors. GHS-R has been shown by RT-PCR to be present in the islets but not in acinar cells (Volante et al. 2002). The presence of GHS-R on pancreatic duct cells has not been investigated.
Urocortin III and CRF belong to the same family of hormones and bind with high affinity to CRF receptors. Both peptides increase CCK-stimulated protein secretion, but this effect seems to be mediated by cholinergic intrapancreatic neurons (Guzman et al. 2003). In spite of this, CRF receptors appear to be located at least on the acinar compartment of the pancreas (Kanno et al. 1999).
Peptide YY, a 36-amino-acid peptide, is probably the best studied of the non-classical islet hormones. It decreases bicarbonate and protein content of pancreatic juice in unstimulated rats (Robinson et al. 1996) and strongly inhibits nutrient-, hormonal-, and vagally stimulated bicarbonate and fluid secretion (Tatemoto 1982; Lluis et al. 1987). This action is probably mediated, at least in part, by peptide YY direct binding to Y1 receptors (Grandt et al. 1995), inasmuch as this receptor has been detected in centroacinar and intralobular duct cells (Jackerott and Larsson 1997).
Other Islet Metabolites as Candidates to Influence Duct Cells. Islets of Langerhans can influence duct secretion through further molecules. It has become evident in the last decade that there is a local renin- angiotensin system (RAS) in the pancreas (Leung and Carlsson 2001). Any RAS requires a local source of substrate (i.e., angiotensinogen) and a series of enzymes capable of transforming such a substrate into its octapeptidic, metabolically active form (i.e., angiotensin II). In the pancreas, these requirements are fulfilled (Leung and Chappell 2003). Angiotensinogen (Agt) mRNA has been detected in rat pancreas (Lau et al. 2004). Agt is detectable in glucagon-secreting cells (Regoli et al. 2003) and in a subset of PP-producing cells (E. Bertelli, unpublished data). Moreover, an angiotensin II (Ang II)-generating system has been reported in isolated rat islets of Langerhans (Lau et al. 2004). Ang II appears to be involved in the mechanism of insulin release (Lau et al. 2004), but Ang II receptors (AT1 and AT2) have been assigned to the duct epithelium as well (Leung et al. 1997). AT1 receptors have also been shown in a human pancreatic duct cell line (CFPAC-1) whose bicarbonate secretion is influenced by Ang II (Chan et al. 1997; Cheng et al. 1999). Interestingly, captopril, an angiotensin-converting enzyme (ACE) inhibitor, reduces secretin-induced bicarbonate output in conscious dogs (Howard-McNatt and Fink 2002). Ang II, therefore, might also be involved in the regulation of bicarbonate secretion by duct cells.
Islet hormones and pancreatic exocrine secretion
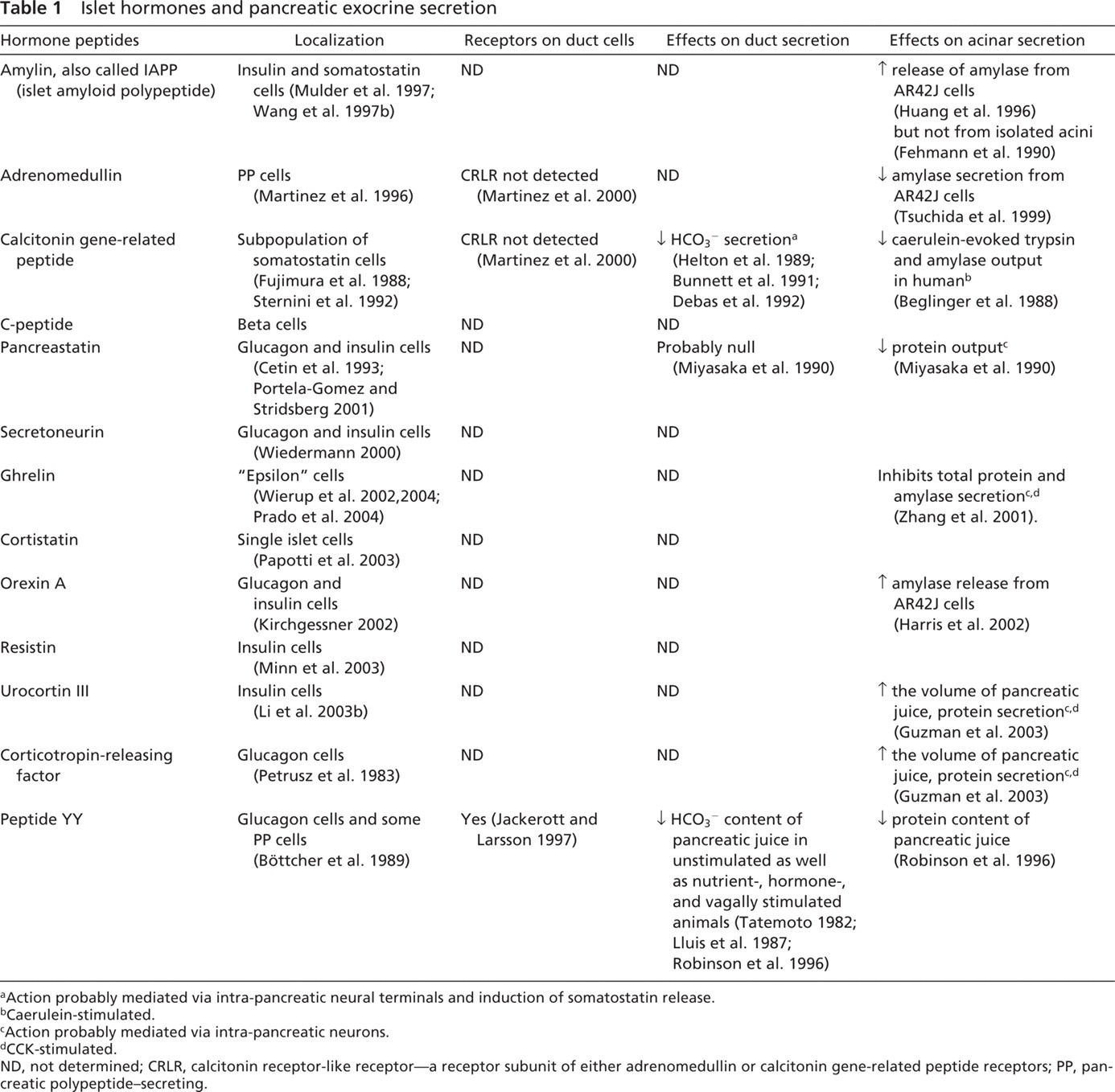
Action probably mediated via intra-pancreatic neural terminals and induction of somatostatin release.
Caerulein-stimulated.
Action probably mediated via intra-pancreatic neurons.
CCK-stimulated.
ND, not determined; CRLR, calcitonin receptor-like receptor—a receptor subunit of either adrenomedullin or calcitonin gene-related peptide receptors; PP, pancreatic polypeptide-secreting.
The metabolism of arachidonic acid in islets of Langerhans results in the production of prostaglandins and leukotrienes via the cyclooxygenase and lipoxygenase pathways, respectively (Metz et al. 1983; Persaud et al. 2004). The exact role of eicosanoids in pancreatic duct secretion is unknown (Case and Argent 1993). However, inasmuch as secretin-stimulated bicarbonate output is inhibited by leukotriene C4 and blocked by indomethacin, it can be hypothesized that an important role is played by endogenous eicosanoids in bicarbonate secretion (Konturek et al. 1988; Beauchamp et al. 1992).
Another metabolite with unconfirmed effects on pancreatic duct secretion is nitric oxide (NO). NO is synthesized by NO synthases (NOS), using l-arginine as a substrate. Islets of Langerhans express either constitutive NOS or inducible NOS (Eizirik et al. 1996; Salehi et al. 1996; Henningsson et al. 2000), and NO is thought to be involved in the regulation of glucose-stimulated insulin release (Henningsson et al. 2002). NO has controversial effects on bicarbonate pancreatic duct secretion. Specific inhibitors of NOS reduce secretin-induced fluid and bicarbonate output in rats (Jyotheeswaran et al. 2000), but this action has not been demonstrated in humans (Konturek et al. 1997). Moreover, it is still unknown whether NO inhibition of bicarbonate secretion in rats is due to a direct action on duct cells, because it also modulates insulin release (Smukler et al. 2002).
The last molecule that should be considered is vascular endothelial growth factor (VEGF). VEGF is expressed by islet cells in mouse, rat, and human pancreas (Christofori et al. 1995; Kuroda et al. 1995; Rooman et al. 1997; Hansel et al. 2004). Insulin-secreting cells, in particular, appear to be responsible for islet VEGF immunoreactivity (Kuroda et al. 1995; Fan and Iseki 1998). Low levels of VEGF mRNA have been shown in immortalized human pancreatic ductal epithelial (HPDE) cells as well (Liu et al. 1998; Li et al. 2004a). Receptors for VEGF, Flt-1 and Flk-1 have been found in the endothelial cells of the islets of Langerhans (Christofori et al. 1995). Neuropilin-1 and neuropilin-2 have also been shown to bind to VEGF, in addition to their natural ligand, semaphorin (Soker et al. 1996,1998). Neuropilins have been detected in islet cells (Cohen et al. 2002; Fukahi et al. 2004; Hansel et al. 2004) and are overexpressed in pancreatic tumors (Fukahi et al. 2004; Li et al. 2004a). Interestingly, Flk-1 has also been located in rat pancreatic ducts (Rooman et al. 1997), and neuropilin mRNA has been found in HPDE cells (Li et al. 2004a). VEGF is mitogenic for pancreatic ductal epithelium, and islet-secreted VEGF could influence population dynamics of pancreatic ducts as previously suggested (Rooman et al. 1997).
In summary, we can say that as studies on islets of Langerhans progress, the more it appears that the islet acts as a complex biochemical unit that synthesizes a remarkable number of molecules having a large spectrum of bioactive properties. In addition to the four classical islet hormones, we can now count at least thirteen additional “non-classical” hormones and several other molecules that are discharged into the surrounding tissue to act in a paracrine way on neighboring cells and/or to reach the bloodstream. These substances, therefore, can reach the ducts via paracrine and/or endocrine pathways. However, their actual role and involvement in the modulation of duct secretion are far from being established.
Pancreatic Ductal Influence on Islet Physiology
Because most of the islets are in contact with the ducts, the only mandatory requirement for a given islet hormone to influence the duct epithelium is the presence of a specific hormone receptor on the duct cells. Less obvious is the possibility that duct cells, in turn, influence islet physiology. Constitutively or upon activation, duct cells produce a number of factors that indeed can exert several subtle actions on islet cells. In particular, duct cells can secrete growth factors, cytokines, and eicosanoids that might affect islet cell function.
Growth Factors. Transforming growth factor (TGF)-α is a member of the epidermal growth factor (EGF) family that is implicated in phenomena of duct cell proliferation that precede islet cell differentiation (Wang et al. 1993). Its receptor (EGF-R) is broadly expressed in the pancreas, but mostly by islet cells (Hormy and Lehy 1994). TGF-α is detectable in ducts of normal rat pancreas (Wang et al. 1997a) and is overexpressed by duct and acinar cells in chronic pancreatitis (Korc et al. 1994). TGF-α is also overexpressed by duct cells upon ligation of the main pancreatic duct (Wang et al. 1997a). Interestingly, under the same circumstances, TGF-α overexpression by duct cells is accompanied by gastrin expression, which is known to be involved in islet differentiation (Wang et al. 1993). Gastrin receptors are expressed by glucagon-secreting cells in the normal pancreas (Saillan-Barreau et al. 1999) and by duct cells upon duct ligation (Rooman et al. 2001). Overexpression of gastrin and TGF-α by duct cells has a role in islet cell differentiation from progenitor cells located within the duct epithelium, as well as in islet cell proliferation (Wang et al. 1993,1995). The prevalent view is that TGF-α operates by inducing metaplastic proliferation of pancreatic ducts, and gastrin is believed to complete the differentiation program (Wang et al. 1993). However, an action of TGF-α as a cofactor promoting proliferation of preexisting islet cells or islet cell precursors embedded within the islets of Langerhans could also be hypothesized as responsible not only for duct expansion, but also for islet cell proliferation under duct ligation conditions (Wang et al. 1995). This does not seem to occur in transgenic mice overexpressing gastrin alone (Wang et al. 1993).
Neurotrophin-4 (NT-4) is a nerve growth factor-related peptide that binds preferentially to the TrkB receptor (Klein 1994). Whereas the TrkB receptor is detectable in glucagon-producing cells (Shibayama and Koizumi 1996; Schneider et al. 2001), NT-4 is strongly expressed by duct cells, suggesting the possible existence of an NT-4/TrkB-based duct-islet axis (Schneider et al. 2001). Activation of TrkB receptors in glucagon-secreting cells with the alternative ligand brain-derived neurotrophic factor (BDNF) affects glucagon secretion by isolated mouse islets of Langerhans (Hanyu et al. 2003).
Betacellulin (BTC) is a member of the EGF family that binds to the EGF receptors erbB1 and erbB4 (Dunbar and Goddard 2000). It promotes islet cell differentiation and proliferation (Mashima et al. 1996; Demeterco et al. 2000; Li et al. 2001,2003a). Immunohistochemistry on human pancreas has identified BTC and its receptors in islet and duct cells (Miyagawa et al. 1999), suggesting possible reciprocal influence under particular circumstances. Because exogenous administration of BTC enhances islet regeneration (Li et al. 2001,2003a), a role for endogenous BTC in islet and duct cell proliferation can also be envisioned.
Activin A, B, and AB are three members of the transforming growth factor-beta (TGF-β) superfamily. They are constituted by the assembly of βA and βB subunits that can give rise to βA-βA homodimers (activin A), βB-βB homodimers (activin B), and βA-βB heterodimers (activin AB). Both subunits have been detected by RT-PCR in ducts and islet cells (Zhang et al. 2002). However, the βA subunit was detected only in glucagon-producing, centroacinar, and duct cells, whereas the βB subunit was shown in somatostatin-secreting and duct cells (Zhang et al. 2002; La Rosa et al. 2004). Thus, whereas all activins can be formed in duct cells, in endocrine cells, activin A is expressed predominantly by glucagon-secreting cells and activin B by somatostatin-producing cells. However, recent observations challenge this, inasmuch as both activins A and B have been shown in insulin-producing cells (Tsuchida et al. 2004). Expression of activin receptor subunits has been demonstrated in islets and ducts (Ueda 2000; Zhang et al. 2002; Tsuchida et al. 2004). Activin A is involved in islet cell differentiation and regeneration (Furukawa et al. 1995; Mashima et al. 1996; Demeterco et al. 2000; Ueda 2000; Li et al. 2004b), whereas all activins stimulate insulin release (Totsuka et al. 1988; Florio et al. 2000; Tsuchida et al. 2004). Even though current data on the sites of expression of activins and their receptors are somewhat confusing, increased expression of activins in normal pancreatic tissue, as well as in streptozotocin-treated or partially pancreatectomized animals (Zhang et al. 2002), supports interactions between ducts and islets on an activin-based axis.
Cytokines. Ducts can also influence islet physiology through the secretion of cytokines. Even though under normal conditions cytokine secretion by duct cells does not seem to occur at detectable levels, it does take place under experimental conditions and could acquire a remarkable importance in pathology and in islet transplantation.
The involvement of duct cells in the inflammatory processes occurring in diabetes has been reported (Papaccio et al. 1994a,b). In particular, inflammatory infiltration in ducts located close to the islets has been shown in the early stages of experimental diabetes (Papaccio et al. 1994b) and mononuclear infiltration occurs in small ducts in non-obese diabetic mice (Papaccio et al. 1994a). Duct cells display a strong major histocompatibility complex (MHC) class II immunore-activity (Papaccio et al. 1994a,b), suggesting an active response to inflammatory events. Upon interleukin-1β (IL-1β) challenge, duct cells have been shown to secrete tumor necrosis factor (TNF)-α (Movahedi et al. 2004). Moreover, IL-1β together with interferon (IFN)-γ stimulate inducible NOS expression in duct cells and production of NO (Pavlovic et al. 1999; Pipeleers et al. 2001). IL-1β, in turn, can be produced by islet cells, infiltrating immune cells (Heitmeier et al. 2001), and even duct cells (Vosters et al. 2004). In the latter case, IL-1β expression is triggered by CD40 binding, which also leads to the expression and secretion of TNF-α (Vosters et al. 2004). IL-1β inhibits insulin release in rat islets through the induction of NO formation (Corbett et al. 1993a), and its action is potentiated by TNF-α (Mandrup-Poulsen et al. 1987). On the other hand, TNF-α itself is able to suppress basal and glucose-stimulated insulin secretion and proinsulin mRNA transcription in HIT-T15 pancreatic cells (Tsiotra et al. 2001).
Other cytokines (i.e., monocyte chemoattranct protein 1, ENA78, and IL-8) are produced by duct cells (Saurer et al. 2000). Even though their direct action on islet cell secretion has not been reported, they deserve to be mentioned as possible mediators in events leading to islet graft rejection (Piemonti et al. 2002). Indeed, as previously suggested (Pavlovic et al. 1997), duct cells located close to islet cells may also actively participate in the local immune reactions that occur in autoimmune diabetes. They can express MHC class II (Papaccio et al. 1994a,b; Pavlovic et al. 1997) and CD40 (Vosters et al. 2004); they can release NO (Pavlovic et al. 1999), which, in turn, damages or affects beta cell function (Corbett et al. 1993b; Eizirik and Pavlovic 1997); and, secreting chemoattractant factors, they can contribute to the recruitment of immune cells (Saurer et al. 2000).
Other Possible Mechanisms for Ductal Influence on Islet Physiology (Acinar-Islet Units?). It has been reported that in vivo infusion of secretin has a modulating action on islet hormone secretion. However, data differ depending on animal species and experimental models. In dogs, secretin appears to inhibit glucagon release (Santeusanio et al. 1972; Hisatomi and Unger 1983; Koizumi et al. 1986), whereas increased secretion has also been reported (Otsuki et al. 1981). In the perfused isolated pancreas, secretin stimulates insulin and somatostatin release (Ipp et al. 1977; Hisatomi and Unger 1983). Yet, as far as insulin is concerned, this effect is not observed in the conscious animal (Koizumi et al. 1986). In rats, secretin induces glucagon secretion (Otsuki et al. 1981; Szecowka et al. 1982), but at physiologic doses, it seems to be ineffective on glucagon (Ferrer et al. 2001) as well as on insulin release (Otsuki et al. 1981; Szecowka et al. 1982; Ferrer et al. 2001). In humans, secretin has been reported to be unsuccessful in altering either insulin or glucagon secretion (Shima et al. 1978; Brodows and Chey 1980), but increased insulin release has also been reported (Glaser et al. 1988). Studies carried out in vivo and in vitro on mice islets have shown that secretin increases basal and stimulated insulin release (Ahren and Lundquist 1981; Kofod et al. 1988a,b) as well as glucagon secretion, even at high glucose concentrations (Ahren and Lundquist 1986; Kofod et al. 1988b). In spite of the secretin response achieved in isolated islets (Kofod et al. 1988a,b), which should rule out neuronal mediation, autoradiographic studies have so far failed to detect secretin receptors in islets of Langerhans (Ulrich et al. 1998). Thus, secretin may influence islet hormone secretion binding to lower affinity-related receptors or by other indirect mechanisms. Presently, a new indirect mechanism can be put forward based on duct-islet contacts and could be named the “acinar-duct-islet axis.” In accord with this model, the flow of exocrine secretion (either acinar or ductal) can be monitored by those endocrine cells that face the ductal lumen. As mentioned earlier, an intense endocytotic activity, testified to by the presence of a rich store of smooth and coated vesicles in the apical pole of those endocrine cells that face the ductal lumen, probably reflects intense internalization. Alternatively, duct cells may monitor the content of pancreatic juice or influence adjacent endocrine cells upon secretin binding. Indeed, pancreatic duct cells are provided with well-developed primary cilia with specific sensory functions (Bertelli and Regoli 1994; Pazour and Witman 2003). Interestingly, the tip of these organelles displays variable morphological features suggesting different sensory capabilities (Bertelli and Regoli 1994). At any rate, duct cells or endocrine cells facing the lumen can exert their influence on neighboring islet cells according to a model proposed 15 years ago and still considered quite valid (Pipeleers 1987). Gap junctions among endocrine cells could allow the spreading of eventual signaling events through a larger population of endocrine cells (Meda et al. 1986; Meda 1996). An additional consideration can be made following this line of reasoning. Inasmuch as islets of Langerhans have preferential relations with the most proximal segments of the duct tree (centroacinar and small-sized intralobular duct), it is highly probable that they contact ducts draining portions of acinar tissue located very close to the islets themselves (i.e., the peri-insular tissue), the very same parts of the acinar tissue which, in turn, are more heavily influenced by islet hormones (acinar-islet units).
Overall, duct cells are much more active than is commonly believed, inasmuch as they are capable of releasing growth factors and cytokines that exert important paracrine effects. Now that closer relations between ducts and islets of Langerhans have been established, islet cells can be considered as the major target of such paracrine secretions. Growth factors can be mainly involved in mediating signals for islet cell differentiation and proliferation, but in many instances, they may also affect islet secretion (Totsuka et al. 1988; Florio et al. 2000; Hanyu et al. 2003; Tsuchida et al. 2004). The actual existence and importance of a growth factor-based duct-islet axis require further studies that would represent a research area of great potential.
However, even though some cytokines have been shown to influence islet secretion (Corbett et al. 1993a; Tsiotra et al. 2001), they appear primarily as mediators of immunological events (Saurer et al. 2000; Piemonti et al. 2002; Movahedi et al. 2004). In this context, the importance of duct cells in the progression of immunological responses in type I diabetes and in islet graft rejection becomes a matter of increasing interest (Pipeleers et al. 2001; Movahedi et al. 2004; Vosters et al. 2004).
Last but not least, the activity of duct cells can influence islet cells following previously unexpected paths. These might involve islet cells located directly in relation with pancreatic juice (and therefore with the exocrine products of acinar and duct cells) and/or duct cells (Bertelli et al. 2001). On the whole, these direct pathways could be referred to as the “acinar- duct-islet axis.”
Exocrine Secretion of Hormones
It has been known for several years that all major islet hormones are present in pancreatic juice (Carr-Locke and Track 1979; Conlon et al. 1979; Lawrence et al. 1979; Ertan et al. 1981; Sarfati et al. 1986; Bertelli et al. 2001). Interestingly, even though constantly detected, their concentration in pancreatic juice does change upon caerulein and secretin stimulation (Escourrou et al. 1985). Despite repeated reports on the exocrine secretion of islet hormones, the exact role played by such secretion is still unknown. One obvious target could be the intestinal epithelium, which displays insulin receptors on its brush border (Pillion et al. 1985; Buts et al. 1997). Yet alternative possibilities may exist and should be considered. Immunoelectron microscope evidence suggests that duct cells can uptake insulin from the pancreatic juice (Bertelli et al. 2001). However, it is not known whether this phenomenon is a mechanism of reabsorption of hormones that are intended to return to the bloodstream or whether it reflects a more significant, direct effect on duct cells. On the other hand, other non-classical islet hormones may be secreted in the pancreatic juice and exert their action on ducts. This is probably the case for Ang II, because AT receptors have been assigned to the luminal pole of duct cells (Leung et al. 1997). Agt secretion in the pancreatic juice has been systematically observed in rat pancreas (Regoli et al. 2003), and Ang II can be generated via unconventional pathways not involving renin and ACE. Other enzymes are capable of cleaving Agt and release Ang II. Interestingly, two of them (i.e., trypsin and kallikrein) are normal constituents of pancreatic juice (Arakawa et al. 1980; Maruta and Arakawa 1983). A role for luminal Ang II in the regulation of duct secretion is supported by the finding that Ang II influences bicarbonate secretion in CFPAC-1 (Chan et al. 1997; Cheng et al. 1999).
A great body of work remains to be carried out. To explore the roles of hormonal exocrine secretions, the presence of other “non-classical” islet hormones in pancreatic juice, as well as their in vitro action on duct cells, should be carefully investigated.
Islets and Buds: Two Different Endocrine Compartments?
The microangioarchitecture of the pancreatic vascular bed has been the subject of intense investigation. The prevalent idea is that the intra-islet capillary network is arranged in a constant pattern flowing unidirectionally from insulin- to glucagon- and somatostatin-secreting cells (Bonner-Weir and Orci 1982; Stagner et al. 1988; Stagner and Samols 1992). Even though the view of a unique microcirculation pattern has been challenged (Redecker et al. 1992) and definite results are far from having being reached (Brunicardi et al. 1996), the concept that insulin release regulates glucagon-producing cell function (switch-off hypothesis) seems in general correct and has been confirmed through several experimental approaches (Maruyama et al. 1984; Shi et al. 1996; Burcelin and Thorens 2001; Hope et al. 2004). The contrary, however (i.e., a regulation of insulin-secreting cell function by somatostatin and glucagon secretions), might also be effective even if the core-to-mantle islet blood flow were to be confirmed as a rule. Buds of insulin-producing cells associated with the duct tree and single insulin-secreting cells scattered within the duct lining are expected to receive the same blood supply that serves the ducts. It is evident, therefore, that the hormone content of such blood will exert its action either on duct cells or on endocrine cells associated with the ducts (buds and single scattered cells). Few studies on ductular blood supply have been carried out (Lifson and Lassa 1981; Ohtani et al. 1983,1986; Ohtani and Wang 1997). Pancreatic ducts have been reported to receive blood from a direct arterial supply and from connections with the acinar capillary network (Lifson and Lassa 1981; Ohtani et al. 1983). However, interestingly, ducts are also supplied by blood vessels efferent from islets located close to the duct tree (Ohtani 1983; Ohtani et al. 1986; Ohtani and Wang 1997). The size of such islets is variable, but they can be quite large (Ohtani et al. 1986). Thus, according to Ohtani and Wang (1997), the peri-ductular plexus receives blood that has flowed through the core and the mantle of islets and that carries high concentrations of insulin, somatostatin, glucagon, and PP to the endocrine cells associated with the ducts. Therefore, the secretory activity of endocrine tissue associated with and embedded in the duct epithelium, accounting in humans for at least 15% of all insulin-secreting cells (Bouwens and Pipeleers 1998), is assumed to be influenced by this islet- duct portal system and could functionally operate in a very distinct way, compared with the islets of Langerhans, thereby outlining two functionally discrete endocrine compartments. In this context, it deserves to be mentioned that even studies carried out on monkey pancreas support this view, inasmuch as 9% of the islets of Langerhans (usually small and bud-like sized) seem connected via an insulo-insular portal system with larger islets located upstream (Zhou and Gao 1995; Zhou et al. 1996).
In conclusion, it is evident that the question of the pancreatic microcirculation remains a puzzle that is apparently still far from being solved. Accordingly, the importance and the extent of a second endocrine compartment located downstream from the main body of the islets of Langerhans are strictly linked to the existence of insulo-insular or insulo-ductal portal systems and will have to await elucidation of the entire pancreatic microcirculation.
Conclusion
Overall, this review establishes that physical, topographical, and functional relationships exist between the endocrine tissue and the duct system in the pancreas. Because interactions between the endocrine and the acinar tissues have previously been well established and because the duct system is physically and functionally connected to the acinar tissue, we are dealing with an integrated, well-tuned organ, the pancreas, in which all the components are functionally related for well-orchestrated functional responses. The simplistic view of a pancreas as composed of a separated independent endocrine system physically dispersed within an exocrine parenchyma connected to its excretory duct system, the function of which would be limited to the simple transport of the pancreatic juice, becomes from now on an obsolete concept. The interesting additional hypothesis that arises from this and other studies is the mutual functional influence one compartment exerts on the other two. We are now confronted with a pancreatic gland in which an endocrine-exocrine-ductal axis is in place for the benefit of optimal functional performances.
Footnotes
Acknowledgements
This work has been supported by intramural funds from the University of Siena (PAR 2003 quota servizi) to E.B. and by a grant from the Canadian Institutes of Health Research to M.B.
