Abstract
Energy production by means of ATP synthesis in cancer cells has been investigated frequently as a potential therapeutic target in this century. Both (an)aerobic glycolysis and oxidative phosphorylation (OXPHOS) have been studied. Here, we review recent literature on energy production in glioblastoma stem cells (GSCs) and leukemic stem cells (LSCs) versus their normal counterparts, neural stem cells (NSCs) and hematopoietic stem cells (HSCs), respectively. These two cancer stem cell types were compared because their niches in glioblastoma tumors and in bone marrow are similar. In this study, it became apparent that (1) ATP is produced in NSCs and HSCs by anaerobic glycolysis, whereas fatty acid oxidation (FAO) is essential for their stem cell fate and (2) ATP is produced in GSCs and LSCs by OXPHOS despite the hypoxic conditions in their niches with FAO and amino acids providing its substrate. These metabolic processes appeared to be under tight control of cellular regulation mechanisms which are discussed in depth. However, our conclusion is that systemic therapeutic targeting of ATP production via glycolysis or OXPHOS is not an attractive option because of its unwanted side effects in cancer patients.
Keywords
Introduction
The bulk of malignant cells in cancer patients are rapidly proliferating differentiated cancer cells, and a small fraction of the malignant cells are undifferentiated cancer stem cells (CSCs). CSCs reside in specific microenvironments or niches where they are maintained in a slowly dividing or quiescent state. Quiescence of CSCs protects them from the cytotoxic effects of chemotherapy and radiotherapy, as these therapeutic strategies preferentially target proliferating cells. CSC protection in their niches causes recurrence of cancer demonstrating the importance to develop therapeutic strategies focused on CSCs.1–8
In this review, we discuss targeting of the energy metabolism of CSCs in their niches as therapeutic strategy. We focus on the synthesis of ATP in glioblastoma stem cells (GSCs) in primary brain tumor patients and in leukemic stem cells (LSCs) in the bone marrow of leukemia patients. ATP synthesis in these stem cell types is the focus of our comparison because we have previously shown that GSC niches in glioblastoma tumors and LSC niches in bone marrow are functionally and morphologically similar.9,10 Therefore, energy metabolism may well be regulated in a similar way.
(Cancer) Stem Cell Niches
There has been confusion about types of hematopoietic stem cell (HSC)/LSC niches and GSC niches. In the past, three types of HSC/LSC niches were reported, endosteal, reticular, and perivascular niches, whereas five types of GSC niches, perivascular, extracellular matrix (ECM), periarteriolar, perihypoxic, and peri-immune niches, have been described. We have performed thorough morphological and histological/histochemical analyses of HSC/LSC niches in human bone marrow and GSC niches in human glioblastoma tumors. On the basis of these studies, we came to the conclusion that both in bone marrow and in glioblastoma tumors, only one type of niche exists, the hypoxic periarteriolar HSC/LSC and GSC niche and that the subtypes that have been described in the past are in fact characteristics of that hypoxic periarteriolar niche.11–13 We have analyzed quantitatively in sections of 16 glioblastoma tumors how many arterioles, capillaries, or venules were associated with markers of GSCs and their niches. We found GSC niches around seven arterioles out of 335 investigated or 2% of arterioles were surrounded by a niche, whereas 924 venules and 8085 capillaries were not associated with GSC niche characteristics. 12
In the subventricular zone (SVZ), neural stem cell (NSC) niches are hypoxic but not periarteriolar (see below). Hypoxia induces and maintains the stemness of CSCs and CSCs are well-protected because of their low proliferation rate and their well-developed DNA repair mechanisms and drug efflux pumps. Receptor-chemoattractant interactions are responsible for stem cell maintenance and retention of stem cells in the niches, such as interactions between C-X-C receptor type 4 (CXCR4) and chemokine stromal derived factor-1α (SDF-1α, also named CXCL12), between receptor CD44 and chemokine osteopontin (OPN),9,10,12–15 and between angiopoietin 1 (Ang1) and its tyrosine kinase receptor Tie2.11,16–18 The stemness of CSCs in the present review is considered to be characterized by the expression of stem cell markers.
GSCs are present in GSC niches in glioblastoma tumors and in the SVZ in brains of glioblastoma patients (Fig. 1). 19 The SVZ is a major niche of normal NSCs. The SVZ is morphologically different from GSC and HSC/LSC niches, as the SVZ is not associated with arterioles, but there is also a close association with blood vessels. 18 The SVZ is hypoxic and the CXCR4-SDF1α and CD44-OPN interactions are also associated with homing of GSCs in the SVZ. Thus, GSCs can escape from the effects of cytotoxic therapy in the SVZ as they are maintained in slowly dividing state and are not removed during surgical resection of the tumor. The origin of GSCs is not exactly known. One hypothesis is that GSCs originate from NSCs in the SVZ and have migrated to form a glioblastoma tumor in the brain at distance from the SVZ.20,21 Another hypothesis is that GSCs arise in the glioblastoma tumor, from which they migrate into the SVZ. 22
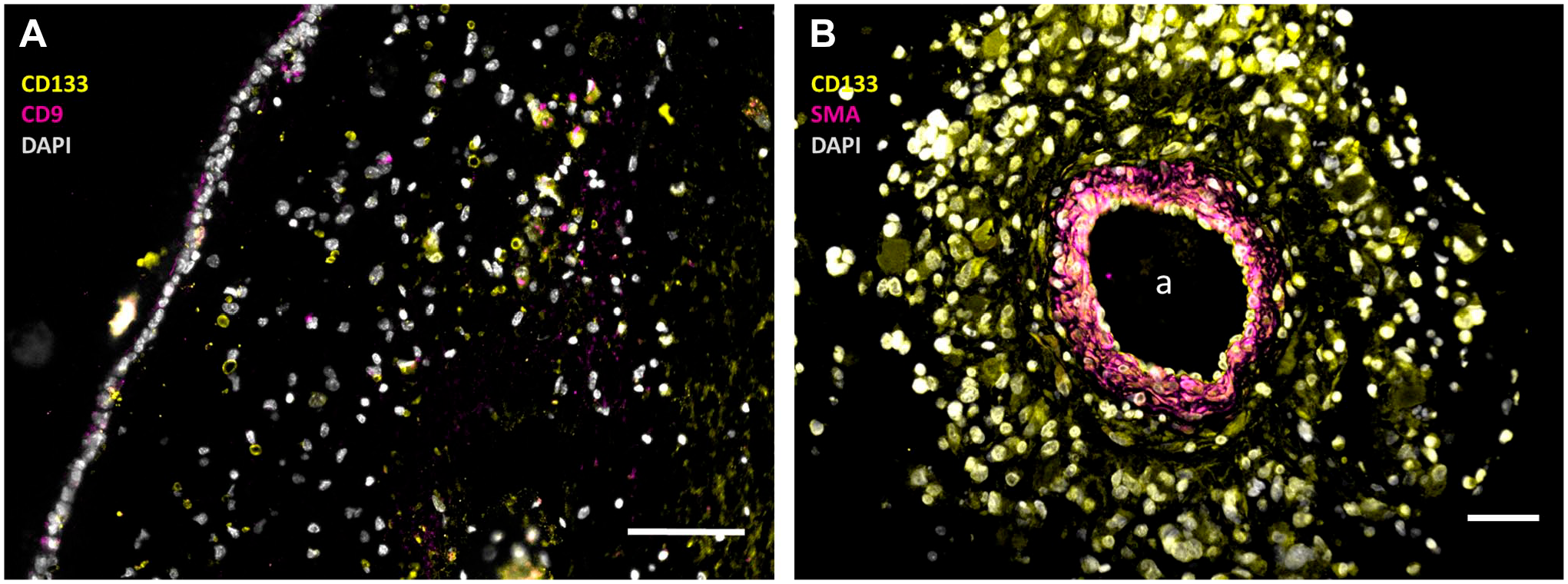
Immunofluorescence images of paraffin sections of a human SVZ (A) and a human glioblastoma tumor (B) showing NSCs and GSCs in the SVZ (A) and GSCs in a periarteriolar niche in the tumor (B). In the SVZ, NSCs are stained in yellow without pink staining (CD133+and CD9−) and GSCs are stained in yellow and pink (CD133+ and CD9+), whereas cell nuclei are stained in white. In the periarteriolar niche in the glioblastoma tumor, GSCs are stained in yellow with white nuclei. The images were constructed on the basis of monochrome images in green, red, and blue after immunofluorescence staining which are shown in Supplemental Figure 1. The pseudocolor images have been constructed to facilitate clear discrimination of NSCs and GSCs by the color-blind readership according to Jambor et al. 23 a, lumen of arteriole. Bars, 100 µm. Images have been prepared on the basis of images in Hira et al.9,19 Abbreviations: SMA, smooth muscle actin; SVZ, subventricular zone; NSCs, neural stem cells; GSCs, glioblastoma stem cells; DAPI, 4′,6-diamidino-2-phenylindole.
In leukemia patients, LSCs are present in HSC niches (Fig. 2) where they are protected in a similar way as GSCs in their niches in glioblastoma tumors and the SVZ.
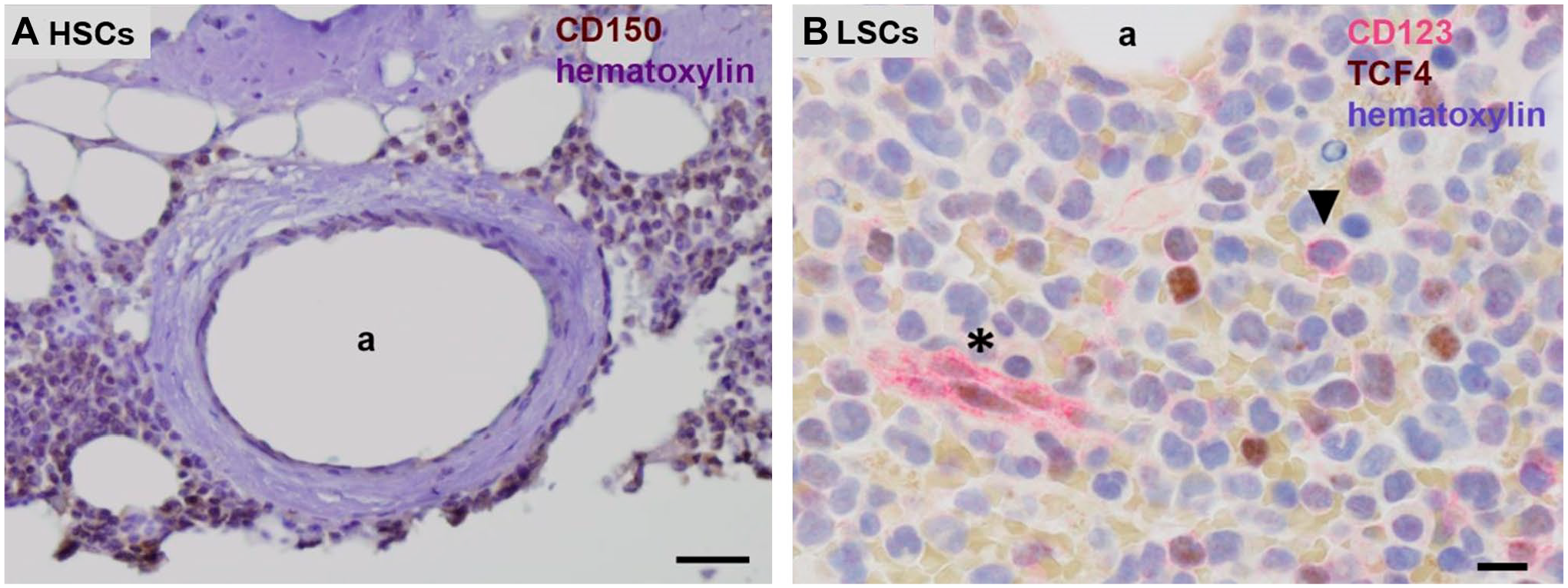
Chromogenic immunohistochemical images of paraffin sections of human bone marrow. (A) Human CD150-positive HSCs (brown) in their periarteriolar niche in healthy bone marrow adjacent to bone and (B) human CD123-positive LSCs (red, arrow head) in a HSC niche in bone marrow of a leukemia patient. Endothelial cells of capillaries are CD123 positive as well (asterisk). Hematoxylin staining in blue. a, lumen of arteriole. Bars, (A) 100 µm, (B) 25 µm. Tissue sections and staining are prepared according to Hira et al. 9 (A) and El Achi et al. 24 Abbreviations: HSCs, hematopoietic stem cells; LSCs, leukemic stem cells.
Isocitrate Dehydrogenase 1/2 Mutations and ATP Synthesis
Besides the similarity of HSC niches in bone marrow that harbor LSCs in leukemia patients and GSC niches in glioblastoma tumors, there is another remarkable similarity between glioblastoma and leukemia and in particular acute myeloid leukemia (AML). Both types of cancer are induced in a considerable percentage of patients by a distinct mutation in the NADP-dependent isocitrate dehydrogenase (IDH) 1 or 2 genes (10–40% of AML patients and 5% of glioblastoma patients). As IDH1 and IDH2 are both involved in energy metabolism including OXPHOS (for recent reviews, see previous studies25–27), the effects of the IDH1 and IDH2 mutations on ATP production in GSCs and LSCs are discussed as well. The mutation occurs in either the IDH1 or IDH2 gene in similar amounts in AML patients and almost uniquely in IDH1 in glioblastoma, 28 although recently a significant number of IDH2 mutations were found in glioblastoma patients in the Hispanic population in the United States and Mexico. 29
ATP Synthesis Pathways
We discuss in the present review ATP synthesis in benign and malignant stem cells in the bone marrow and the brain, HSCs, NSCs, LSCs, and GSCs (Figs. 1 and 2). Two major pathways are available in cells for ATP production (Fig. 3). The most efficient pathway is mitochondrial respiration with the use of oxygen that generates 36-mol ATP per mol glucose, whereas cytoplasmic glycolysis produces 4-mol ATP per mol glucose without the use of oxygen. ATP production by the inefficient glycolytic pathway is a salvage pathway for ATP production in case cells are short of oxygen. In that anaerobic process, lactate is generated from pyruvate and in the early steps of glycolysis, 2-mol ATP per mol glucose are needed to keep glycolysis running, yielding a net 2-mol ATP per mol glucose from glycolysis. When oxygen is available (aerobic conditions), pyruvate is channeled into mitochondria for the more efficient ATP production in the oxidative phosphorylation (OXPHOS) as part of the mitochondrial respiration (Fig. 3).
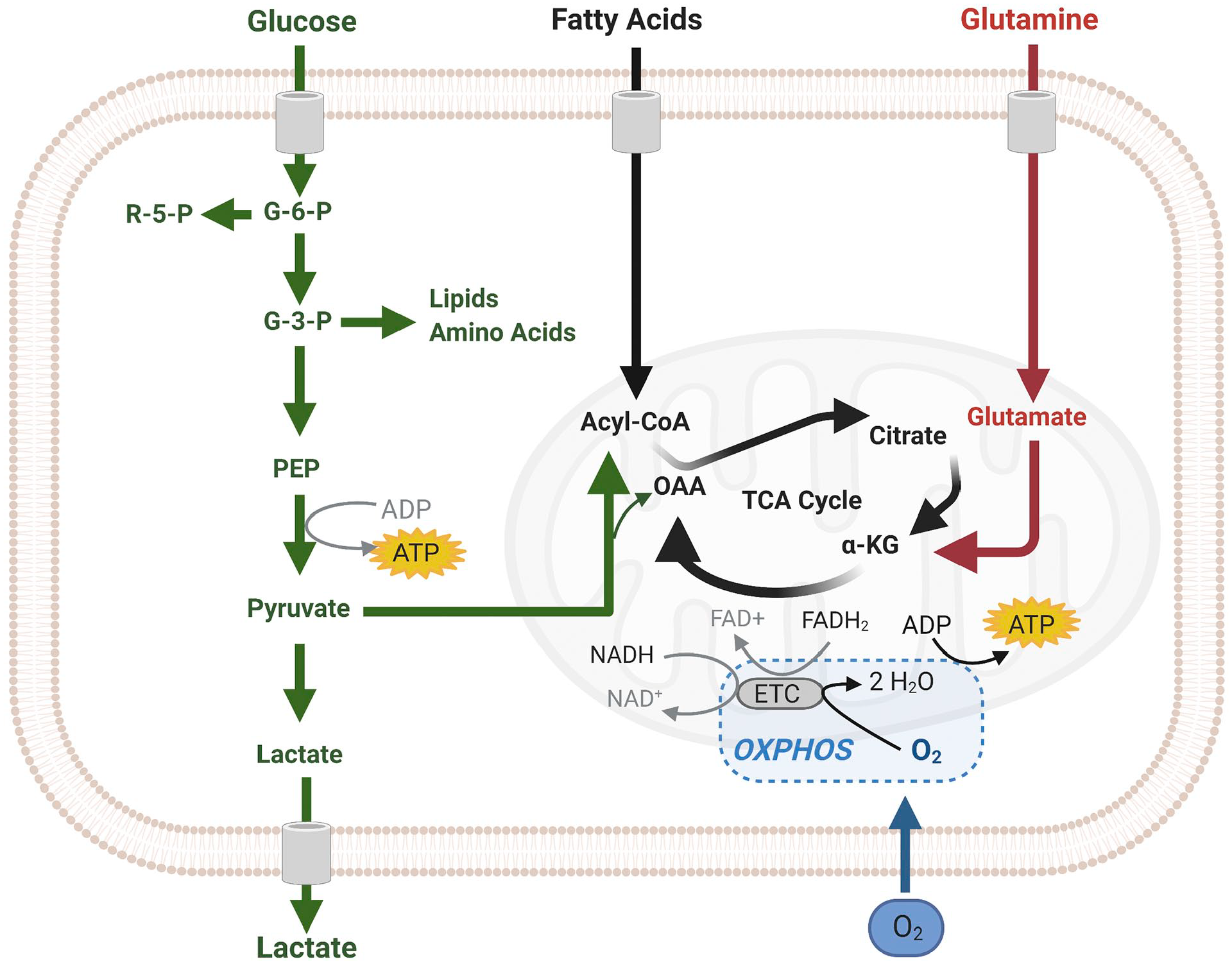
Scheme of cellular ATP synthesis via (an)aerobic glycolysis in the cytoplasm (green) and in mitochondria via OXPHOS (black). The image was created using Biorender.com. Abbreviations: OXPHOS, oxidative phosphorylation; TCA, tricarboxylic acid cycle.
It was generally assumed until recently that cancer cells use glycolysis irrespective the presence of oxygen as a consequence of dysfunctional or impaired mitochondria. However, in most tumors, the use of glycolysis for ATP production independently of the presence of oxygen is an essential part of a “selfish” metabolic reprogramming as was explained recently by Vaupel and Multhoff. 30 Therefore, we have investigated at the electron microscopical (EM) level the morphology of mitochondria in differentiated glioblastoma cells and GSCs (Fig. 4). The morphology of mitochondria from the level of individual mitochondrion ultrastructure to the level of the entire mitochondrial network in a cell reflects mitochondrial function. 31 Ultrastructurally, the morphology of cristae can be highly variable in accordance with the metabolic state of mitochondria32,33 and processes that cause their functional decline, such as aging. 34 Generally, more numerous, tightly packed cristae support more efficient OXPHOS because the cristae establish inner mitochondrial compartments and accommodate protein complexes of the electron transport chain and ATP synthase. 35 These protein complexes in turn impact the shape of mitochondrial cristae.36–38 Regarding the mitochondrial network, mitochondrial fusion that produces highly elongated mitochondria supports efficient OXPHOS, whereas mitochondrial fission that results in smaller and rounded mitochondria promotes glycolysis and greater biosynthesis of anabolic precursors.39,40 It appeared that in all glioblastoma cells investigated be it differentiated cells or GSCs, the morphology of mitochondria is deviant from normal cells. Mitochondria in glioblastoma cells are generally small, with very dense matrices and disorganized cristae, which is in line with the assumption that glycolysis is used by cancer cells for ATP production because of defective mitochondria. Similar defective mitochondrial ultrastructure in line with a glycolytic phenotype was reported also by Vallejo et al. 41 for adult and pediatric glioblastoma. Therefore, we are investigating at present the activity of the electron transport chain in mitochondria of glioblastoma cells at the ultrastructural level using enzyme histochemical methods, such as that for demonstrating cytochrome c oxidase activity. 42
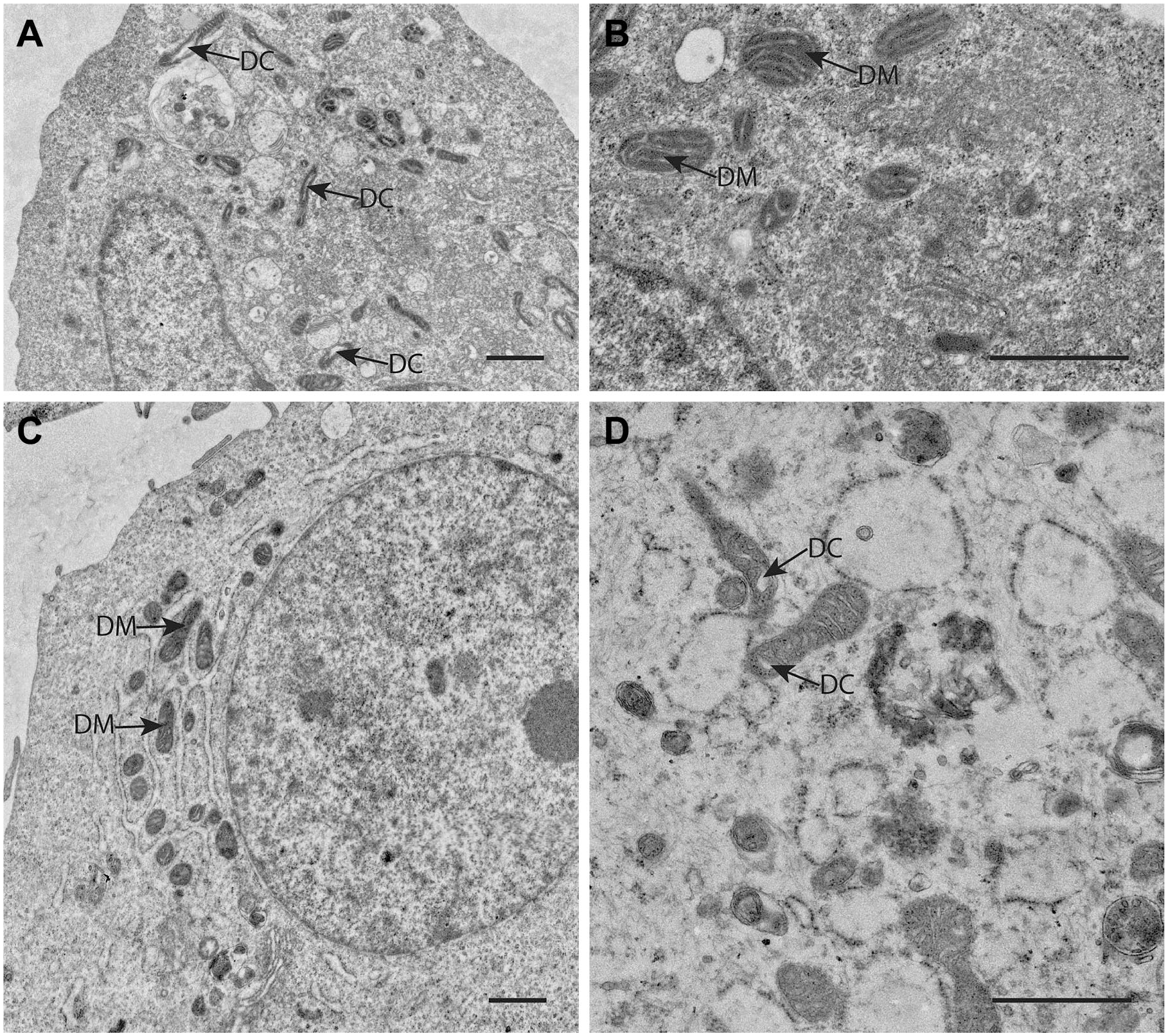
Electron microscopical (EM) images of mitochondria in human GSCs (A, NCH421 and B, NCH644 primary patient GSCs) and in human differentiated glioblastoma cells (C, U87 cancer cells and D, NIB140 primary patient cancer cells) showing the deviant morphology of mitochondria with relatively few and often dilated cristae (DC) and very electron-dense matrices (DM) in both GSCs and differentiated glioblastoma cells indicating that glycolysis rather than OXPHOS is used by these cells for ATP production. Bars, 1 µm. NCH421 and NCH644 cells are a kind gift of Prof. Dr Christel Herold-Mende, University of Heidelberg, Germany. 43 Cell preparations and EM imaging procedures were performed as described in Bogataj et al. 44 Abbreviations: DC, dilated cristae; GSCs, glioblastoma stem cells; OXPHOS, oxidative phosphorylation.
Nowadays, it is realized that proliferating cells such as differentiated cancer cells predominantly use glucose for ATP production in the cytoplasmic glycolysis instead of OXPHOS independently of the presence of oxygen (the so-called Warburg effect or aerobic glycolysis) because glycolysis enables synthesis of building blocks for macromolecules needed by proliferating cells.30,45,46 Moreover, ample NADPH production is also facilitated in this way for reductive biosynthetic reactions.45,47
Quiescent undifferentiated CSCs have a modest need for building blocks and excessive NADPH production because of their slow proliferation rate and, thus, the question arises what metabolic pathways are used by normal stem cells such as HSCs and NSCs and by CSCs such as LSCs and GSCs for ATP production in their hypoxic niche microenvironment. We discuss the cell biological regulation mechanisms of these metabolic pathways as far as these are presently known. These cell biological regulation mechanisms are explained briefly in texts in Boxes that are separate from the main body of text on ATP production in HSCs, NSCs, LSCs, and GSCs.
Energy Metabolism of HSCs
HSCs are resident in niches in human bone marrow which are hypoxic and periarteriolar and express the chemoattractants SDF1α and OPN and their receptors CXCR4 and CD44, respectively, which are essential for the homing of the slowly dividing or quiescent HSCs.9,10 HSC niches are present in red bone marrow around arterioles adjacent to endosteum and trabecular bone. HSC niches are hypoxic despite the fact that they are uniquely found around arterioles, but arterioles are transport vessels and not exchange vessels like capillaries and thus HSC niches do not receive oxygen from the arteriolar lumen.12,13 HSCs produce low levels of intracellular reactive oxygen species (ROS). 11 HSCs are at the top of the pyramid of blood cells above hematopoietic progenitor cells (HPCs) and differentiated red and white blood cells, including megakaryocytes that fragment into thrombocytes, also named platelets. In healthy humans, one trillion (1012) differentiated red and white blood cells are released into the circulation each day. Under normal conditions, only relatively few HSCs are involved in the formation of new mature red and white blood cells. The vast majority of that trillion red and white blood cells originate from rapidly dividing HPCs which are located in HPC niches in the center of the red bone marrow around the sinusoids and the central vein and are continuous with HSC niches in the periphery of red bone marrow around arterioles. 11 The newly formed differentiating red and white blood cells are released into the sinusoids and leave the bone marrow via the central vein. The HSCs in their hypoxic periarteriolar niches at the periphery of the bone marrow are a life-long reserve to ensure the availability of stem cells during the entire life of individuals and supply HPCs in times of depletion of red and/or white blood cells in cases of blood loss. 48 The HPC niches are also hypoxic but intracellular levels of ROS in HPCs are higher than those in HSCs.11,49–52
Metabolism including ATP synthesis of HSCs, HPCs, and hematopoietic stem and progenitor cells (HSPCs) is probably the best studied metabolic aspect in stem cells because of the therapeutic relevance of metabolism on the ex vivo expansion of phenotypically and functionally defined HSPCs harvested from umbilical cord blood and peripheral blood for transplantation purposes in malignant and non-malignant diseases.48,53–55 The term HSPCs is used for the stem-like cells that are harvested from umbilical cord blood, bone marrow aspirate, and peripheral blood for these purposes. 56 The general consensus is that HSCs are anaerobic glycolytic, whereas during the proliferation phases of HPCs, the energy metabolism is mixed glycolytic and OXPHOS, mainly OXPHOS during the differentiation phase and again glycolytic in the maturation phase.54–58 Anaerobic glycolysis in HSCs is determined by high levels of the transcription factors hypoxia-inducible factor-1 (HIF-1) and HIF-2 (see Box 1).9,48 Moreover, the SDF-1a-CXCR4 axis regulates OXPHOS activity in HSCs as well in a thus far unknown manner. 59
Hypoxia-inducible Factors (HIFs).

Abbreviations: ARNT, aryl hydrocarbon receptor nuclear translocator; GSCs, glioblastoma stem cells.
Both HIFs are master switches in cells to adapt to hypoxic conditions 67 and are induced by peroxisome proliferator-activated receptors (PPARs; see Box 2), which are nuclear receptors that act as transcription factors. 69 In fact, intracellular PPAR levels in HSCs are the highest measured in any cell type in the human body. 70 Inhibition of PPARs promotes ex vivo expansion of HSPCs. 54 Moreover, the transcription factor Meis1 (see Box 3), that is expressed in HSCs, transactivates HIF-1α expression by a Meis1-binding motif in the first intron of the HIF-1α gene.55,67 The transcription factor forkhead box O3 (FOXO3; see Box 4) plays a role in many metabolic events in cells, among others protection against ROS, and is a key regulator of HSC quiescence and maintenance of the HSC pool. 48 Finally, autophagy (see Box 5) is also critically important to maintain low mitochondrial metabolic activity and quiescence of HSCs.71,72 Autophagy and stem cell fate are under the control of transcription factor FOXO3 in both normal stem cells and CSCs. 71
Peroxisome Proliferator-activated Receptors (PPARs).

Abbreviations: FAO, fatty acid oxidation; HSCs, hematopoietic stem cells; HIF, hypoxia-inducible factor; STAT3, signal transducer and activator of transcription 3; NSCs, neural stem cells; LSCs, leukemic stem cells; GSCs, glioblastoma stem cells; PML, promyelocytic leukemia protein.
Myeloid Ecotropic Viral Integration Site 1 (Meis1).

Abbreviations: AML, acute myeloid leukemia; HSCs, hematopoietic stem cells; HIF, hypoxia-inducible factor; ROS, reactive oxygen species.
Forkhead Box O3 (FOXO3).

Abbreviations: OXPHOS, oxidative phosphorylation; ROS, reactive oxygen species; GSCs, glioblastoma stem cells; CSCs, cancer stem cells; HSCs, hematopoietic stem cells; NSCs, neural stem cells; LSCs, leukemic stem cells.
Autophagy.

Abbreviations: HSCs, hematopoietic stem cells; NSCs, neural stem cells; HPCs, hematopoietic progenitor cells; NPCs, neural progenitor cells; GSCs, glioblastoma stem cells; LSCs, leukemic stem cells; FDA, Food and Drug Administration.
A number of seemingly contradictory aspects is apparent in the energy metabolism of HSCs in their niches. 48 A major issue is that HSCs are anaerobic glycolytic but cannot survive without mitochondria.48,100 This riddle was at least partly solved by demonstrating that PPARs induce both cytoplasmic anaerobic glycolysis and mitochondrial fatty acid oxidation (FAO; Fig. 3).54,70,73 The latter is equally important for maintaining a viable population of HSCs.70,101,102 OXPHOS is inhibited in HSC mitochondria at the level of pyruvate conversion into acetyl-CoA by pyruvate dehydrogenase after its phosphorylation by pyruvate dehydrogenase kinase (PDK).48,70,103 When PDK is inhibited, quiescence of HSCs is lost and levels of ROS are elevated. Thus, pyruvate entry into the mitochondria has a strong impact on HSC fate. 48 This finding shows that the presence of mitochondria does not mean that all their metabolic pathways are active.48,59,69,70,101,102 HSC quiescence can also be interrupted by signaling from differentiated blood cells, for example, by the chemokine (C-C motif) ligand-6 (CCL-6) secreted by eosinophils. 104
It has to be realized that the cellular mechanisms of HSCs as described here are not necessarily relevant for the in vivo situation in human red bone marrow. The studies have been performed either on mouse bone marrow or human ex vivo HSPCs harvested from umbilical cord blood or peripheral blood. For example, low levels of oxygen have been measured in HSC niches in mouse bone marrow. 105 Nevertheless, we think it is safe to conclude that HSCs in their hypoxic periarteriolar niches adjacent to endosteum and trabecular bone are anaerobic glycolytic and only use particular functions of mitochondria, for example FAO, whereas other functions such as OXPHOS are shut down, to meet their metabolic needs as quiescent long-living cells. How HSCs exactly use mitochondria remains to be investigated. 48 HSCs contain relatively many partially inactive mitochondria not only to meet their metabolic needs but also to be able to rapidly switch on mitochondrial activity in case HSCs need to become HPCs. 48
In conclusion, HSCs are dependent on anaerobic glycolysis for their energy supply, whereas FAO is essential for their stem cell fate.
Energy Metabolism of NSCs
Energy metabolism of quiescent NSCs and the drastic alterations in this metabolism associated with the conversion of NSCs into neural progenitor cells (NPCs) are very much the same as those of HSCs and HPCs, as is shown here.
The developing brain is mainly glycolytic and uses FAO, and this energy metabolism shifts toward the use of glucose rather than lipids during adolescence and becomes more and more OXPHOS dependent. 106 In the adult brain, OXPHOS is the major source of ATP in neurons, whereas astrocytes remain glycolytic and produce lactate that is exported into the ECM to be consumed by neurons that take up lactate and convert it to pyruvate to fuel OXPHOS.86,106,107 However, certain areas of the human brain remain glycolytic and contain quiescent NSCs such as the SVZ that lines the lateral ventricles, the subgranular zone (SGZ) between the granule cell layer and the hilus of the dentate gyrus of the hippocampus, and a few areas in the hypothalamus.106,108–111 FOXO3 regulates glycolysis in NSCs and plays a significant role in the maintenance of the stem cell fate of NSCs in combination with the low oxygen levels in SVZ and SGZ in a similar way as in quiescent HSCs.86,108 It also stabilizes HIF-1α and thus regulates the activity of HIF-1 and possibly of HIF-2. 86 Intracellular levels of promyelocytic leukemia protein (PML; see Box 6) that is involved in the formation of nuclear bodies in NSCs as well as in HSCs are also important for their quiescence. Loss of PML induces loss of quiescence and upregulation of proliferation, whereas loss of PML has been found to be associated with decreased levels of PPARγ in HSCs.73,86
Promyelocytic Leukemia Protein (PML).

Abbreviations: FAO, fatty acid oxidation; PPAR, peroxisome proliferator-activated receptor; FOXO3, forkhead box O3; HSCs, hematopoietic stem cells; NSCs, neural stem cells; LSCs, leukemic stem cells; GSCs, glioblastoma stem cells; mTOR, mammalian target of rapamycin.
In rodents, NSCs of the SVZ are needed permanently throughout life to differentiate into neurons and astrocytes that migrate toward the olfactory bulb, whereas in humans, this renewal of the neuronal circuits that are responsible for smell hardly occurs or does not occur at all.106,118,119 In humans, it has been observed that NSCs become NPCs that proliferate and differentiate as a consequence of ischemic stroke and that NPCs migrate to the damaged brain tissue for repair.108,120–123
Quiescent NSCs in the SVZ and SGZ are anaerobic glycolytic and need FAO,106,108 exactly like HSCs. The FAO inhibitor etomoxir causes depletion of both quiescent NSCs and HSCs indicating the importance of FAO for their stem cell fate.86,124–126 When NSCs become activated, for example, by an ROS spike and become NPCs, mitochondria switch to OXPHOS127–129 and upregulate the redox state,128,130–132 whereas glycolysis and FAO are downregulated and lipogenesis is elevated.106,108 Upregulation of the redox state involves higher ROS production and more active defense mechanisms against ROS, which is orchestrated by increased expression of the transcription factor erythroid 2-related factor 2 (Nrf2; see Box 7) that senses oxidants and regulates antioxidant defense. 133 Besides the spike of ROS that can activate NSCs into NPCs, branched chain amino acids, the endogenous neuroprotective bile acid tauroursodeoxycholic acid, interferon γ, and signal transducer and activator of transcription 3 (STAT3; see Box 8) have been reported recently to activate human NSCs into NPCs in vitro or in vivo in rodents.134–137 Whether these inducers of switches of NSCs into NPCs are active in vivo in humans is not known yet.
Erythroid 2-related Factor 2 (Nrf2).

Abbreviations: ROS, reactive oxygen species; HIF, hypoxia-inducible factor; HSCs, hematopoietic stem cells; HPCs, hematopoietic progenitor cells; HSPCs, hematopoietic stem and progenitor cells; CSCs, cancer stem cells.
Signal Transducer and Activator of Transcription 3 (STAT3).

Abbreviations: OXPHOS, oxidative phosphorylation; ROS, reactive oxygen species; GSCs, glioblastoma stem cells.
Finally, the role of glutamine/glutamate in NSCs and NPCs is not exactly known. Recently, a novel methodology to analyze dynamically cell metabolism in time was reported with the example of an extracellular shot of glutamine (15 mM) to both human NSCs and NPCs in vitro. Analysis of metabolites in the cells in time showed that NSCs and NPCs responded in a similar way to the extracellular glutamine shot and it mainly involved levels of amino acids, indicating that the glutamine did not affect energy metabolism very much. 148 It may well be that glutamine/glutamate play their role in tumorigenesis. 149
In conclusion, NSCs are dependent on anaerobic glycolysis for their energy supply, whereas FAO is essential for their stem cell fate. Thus, NSCs and HSCs function in very similar ways for their energy supply.
Energy Metabolism of LSCs
Similarly to their HSC counterparts, LSCs need a hypoxic environment. Therefore, hijacking the hypoxic periarteriolar HSC niches in bone marrow is an attractive option for LSCs.9,10 Metabolic pathways that provide metabolites needed for anabolic cell growth are upregulated in LSCs 150 and mitochondria play an essential role in these pathways in LSCs as has recently been reviewed by Panuzzo et al. 26 Whereas HSCs depend on anaerobic glycolysis for generation of ATP, LSCs produce ATP mainly via OXPHOS.26,151,152 OXPHOS is more efficient as energy producer than glycolysis and sustains LSC energy requirements for survival. 153 Damage by ROS that are generated by OXPHOS is resisted by antioxidative mechanisms. 151 Raffel et al. 152 showed by quantitative proteomics that in comparison with HSCs, LSCs have a profoundly altered mitochondrial metabolism, both at the transcriptional (mRNA) level and at the posttranslational (protein) level. Not only OXPHOS was distinctly different but also FAO, lipid synthesis, and amino acid metabolism.26,152 Glutamine and glutamate are efficiently converted into α-KG via upregulated glutaminase and glutamate dehydrogenase activities.26,154 This pathway produces NADPH which is essential for reduction of glutathione.26,47,155,156 Inhibition of FAO or amino acid uptake reduces OXPHOS activity in LSCs indicating that both pathways supply substrate for OXPHOS.26,154,157 Recently, it was found that the adrenomedullin-calcitonin receptor-like receptor axis 158 and spleen tyrosine kinase 159 both induce OXPHOS activity in LSCs. Inhibition or knock down of both proteins specifically targets LSCs in AML.158,159
Reactive aldehydes that are produced during FAO are effectively detoxified by upregulated fatty aldehyde dehydrogenase, more specifically named aldehyde dehydrogenase 3a2 (ALDH3A2), in LSCs.160,161
There is a strong competition in the niches between HSCs and LSCs. In this competition, LSCs have a greater competitive advantage. 151 This advantage is at least partly caused by assistance of mesenchymal stem cells (MSCs) to LSCs. MSCs are present in niches of HSCs and LSCs (Fig. 5A) and assistance of MSCs enables LSCs to keep their leukemic phenotype and chemotherapy resistance as was demonstrated in an in vivo mouse model of AML. LSCs co-opt energy sources and antioxidant defense mechanisms provided by MSCs in the niches to survive chemotherapy. Assistance of LSCs was proven to be provided at the expense of HSCs. 162 It is not described by Forte et al. 162 how the exchange from MSCs to LSCs takes place but extracellular vesicles (EVs),163,164 tunneling nanotubes (TNTs), and/or tumor microtubes165,166 may well play a role in these exchange processes. Furthermore, extracellular ATP provides resistance of LSCs against chemotherapy by a cAMP-mediated mitochondrial stress response. cAMP is produced by the ectonucleotidase CD39 that converts extracellular ATP into cAMP that is then internalized by LSCs. 167
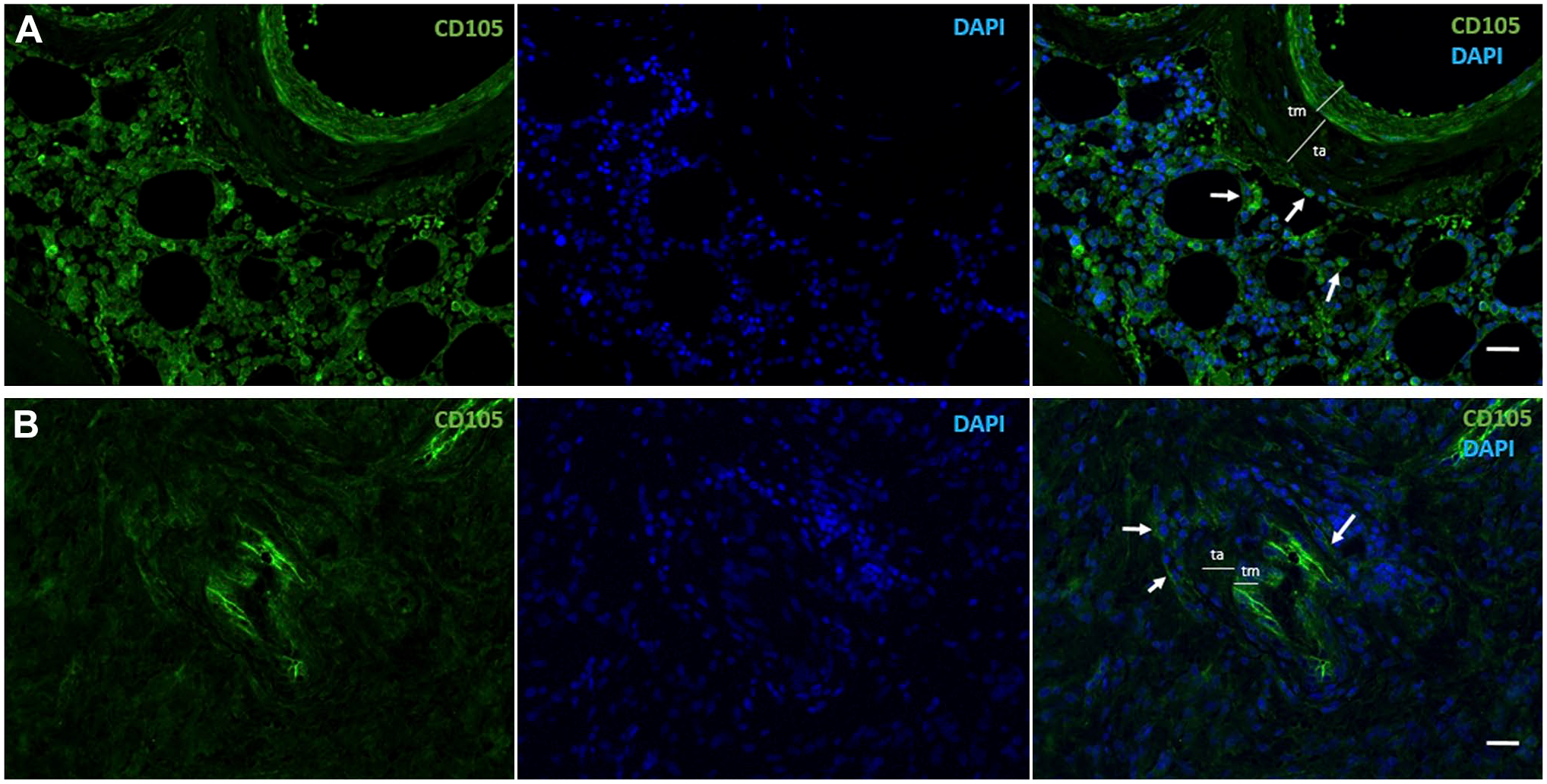
Immunofluorescence images of human CD105-positive MSCs (green, arrows) in a periarteriolar HSC niche in healthy bone marrow adjacent to bone (A) and a periarteriolar GSC niche in a glioblastoma tumor (B). Monochrome images of CD105-positive MSCs in green and DAPI fluorescence (nuclei) in blue and the composition images in green and blue are shown. Smooth muscle cells in the tunica media (tm) of the arterioles are also CD105 positive. ta, Tunica adventitia of the arterioles. Tissue sections and immunofluorescence staining are prepared as described in Hira et al. 9 Bars, 100 µm. Abbreviations: MSCs, mesenchymal stem cells; HSC, hematopoietic stem cell; GSC, glioblastoma stem cell; DAPI, 4′,6-diamidino-2-phenylindole.
All these data have been published recently and indicate that LSCs in hypoxic periarteriolar HSC niches in the bone marrow are different from HSCs with respect to energy metabolism (anaerobic glycolysis versus OXPHOS and altered FAO and amino acid metabolism) and that LSCs in HSC niches are also aided by the microenvironment to survive chemotherapy. This stresses that LSCs have to be removed from HSC niches to optimize the efficiency of anti-LSC therapy, for example, by interfering with their attachment in HSC niches by SDF-1α-CXCR4 with the use of the CXCR4 inhibitor plerixafor.9,10
In a considerable percentage of AML patients (10–20%), cancer cells harbor the IDH1 or IDH2 mutation.25,28,168 The IDH1 and IDH2 mutations occur in similar numbers of AML patients and are mutually exclusive. 28 The effects of IDH1 or IDH2 mutations on the production of ATP [OXPHOS versus (an)aerobic glycolysis] in LSCs do not seem to be profound. The mutations cause metabolic rewiring from aerobic glycolysis to OXPHOS in LSCs and differentiated leukemia cells26,168 independently of their differentiation status. Therefore, we assume that in both IDH1/2-mutated LSCs and differentiated leukemia cells, mitochondrial metabolism including OXPHOS is prevailing. However, as far as we know, this assumption has not been tested experimentally yet.
In conclusion, LSCs depend on OXPHOS for their energy requirements despite the fact that LSCs are hiding in protective hypoxic periarteriolar niches in the bone marrow, whereas FAO and amino acids are needed in the mitochondria to fuel OXPHOS.
Energy Metabolism of GSCs
GSCs play an essential role in glioblastoma tumor development, growth, and aggressiveness. Besides a low proliferation rate that protects GSCs in their hypoxic niches, GSCs possess efficient DNA repair mechanisms and a well-developed drug efflux ABC transporter system.169–171 GSCs express stem cell markers, such as SOX2, NANOG, OLIG2, NESTIN, IDI1, MYC, and MUSHASHI1, and cell surface proteins, such as CD133, CD44, LICAM, and CD15170 and CD9 (Fig. 1) 172 that mediate interactions with their microenvironment of their niches. Furthermore, GSCs interact with differentiated glioblastoma cells and promote malignant progression of glioblastoma. 173 GSCs remain undifferentiated by genetic and epigenetic alterations in signaling pathways such as Notch, bone morphogenetic protein, nuclear factor kappa B, and Wnt.174–177
GSC niches harbor various non-malignant cell types such as immune cells and MSCs (Fig. 5B) that are recruited to the glioblastoma tumor site. The dynamic and complex interactions within niches promote GSC therapeutic resistance and antitumor immune responses. Among the non-cancerous cell types that are present in GSC niches, microglial cells and macrophages prevail. 178 Macrophages are differentiated monocytes from the bone marrow, whereas microglial cells are derived from the yolk sac.179,180 Microglial cells are predominant in newly diagnosed tumors, whereas monocyte-derived macrophages are predominant in recurrent tumors in GSC niches. 181 GSCs recruit macrophages through chemoattractants such as vascular endothelial growth factor, colony stimulating factor 1, SDF-1α, interleukin-6 (IL-6), IL-1b, and the ECM protein periostin, which polarize macrophages to an immune-suppressive and angiogenic phenotype that promotes tumor growth and progression. 182 Not all macrophages are protumoral but for successful application of antitumor macrophages, a better understanding of interactions of GSCs and macrophages is urgently needed. 183
Recently, involvement of cell types and proteins in CSCs and in particular in GSCs has been reviewed, such as a novel cell type, the telocyte, 184 scaffold proteins,185,186 the renin–angiotensin system, 187 and specific proteases and their endogenous inhibitors. 188
MSCs are another cell type that infiltrates glioblastoma tumors and are present in GSC niches, as they are in HSC niches in bone marrow (Fig. 5).9,189 MSCs are important players in glioblastoma because they stimulate proliferation and invasive behavior in differentiated glioblastoma cells and increase the self-renewal capacity of GSCs.9,189–191 MSCs that express CD105 are demonstrated in periarteriolar GSC niches in Fig. 5. Cancer cells, including GSCs, can acquire mitochondria from MSCs in their niches via TNT-mediated intercellular communication. The acquisition of mitochondria increases OXPHOS and ATP production in cancer cells as well as indirectly affects their general metabolism. As a consequence, cancer cells change their invasive and proliferative properties and increase their capacity to develop therapeutic resistance. 192 MSCs also release EVs that contain miRNAs that enhance tumorigenicity of GSCs. 193
Energy metabolism of GSCs is less well studied than that of NSCs, HSCs, and LSCs, but the general consensus is that both IDH1 wild-type and IDH1-mutated GSCs mainly use OXPHOS for the generation of ATP, whereas IDH1wt-differentiated glioblastoma cells use aerobic glycolysis and IDH1-mutated differentiated glioblastoma cells use OXPHOS for ATP production as a consequence of the metabolic rewiring.27,194–197 FAO is essential for GSCs107,198,199 as it is for NSCs, HSCs, and LSCs.
A number of proteins have been described recently that may serve as therapeutic target to reduce OXPHOS activity, especially in GSCs, such as translocator protein (TSPO), insulin-like growth factor 2 mRNA-binding protein 2 (IGF2BP2 or IMP2), oncostatin M, and glycerol-3-phosphate dehydrogenase (GPDI). 27 TSPO is a transmembrane protein in the outer mitochondrial membrane of glial cells 200 and is highly expressed in glioma. 201 Loss of TSPO results in a shift from OXPHOS toward glycolysis. IGF2BP2 is involved in the maintenance of CSCs 202 by delivering electron transport chain subunit-encoding mRNAs to mitochondria and contributing to complex I and IV assembly. 203 Oncostatin M is a cytokine of the IL-6 subfamily and is expressed in brain by various cell types (neurons, astrocytes, and microglia) and is involved in immunosurveillance in the brain. 204 Its receptor is specifically expressed by GSCs in mitochondria and interacts with complex I to promote OXPHOS. Deletion of the oncostatin M receptor reduces OXPHOS activity, but increases ROS levels and sensitizes GSCs to irradiation. 205 GPDI is expressed by GSCs but not by NSCs. 43 GSCs express GPDI in relation with their quiescence and has been proposed as an attractive therapeutic target in glioblastoma.
Promising inhibitors of mitochondrial activity of GSCs have been described as well.107,144,195,206–210 Mudassar et al. 210 proposed to inhibit OXPHOS to increase the low oxygen levels in hypoxic GSC niches to sensitize GSCs to irradiation by repurposing antimalaria drugs such as atovaquone, ivermectin, proguanil, mefloquine, and quinacrine. Lonidamine (LND) is an antiglycolytic drug with limited clinical effects in cancer patients207,211–213 but LND in a mitochondria-targeting form (Mito-LND) appears to be a selective OXPHOS inhibitor with very low toxicity in mice. 213 These characteristics of Mito-LND makes it an attractive candidate to target CSCs in general and GSCs in glioblastoma.
Verteporfin has been Food and Drug Administration (FDA) approved to treat macular degeneration in the eye. It inhibits OXPHOS at complex III and IV and is specifically effective against GSCs and not to differentiated glioblastoma cells or normal cells. 214 Recently, verteporfin was suggested to be a therapeutic agent for epidermal growth factor receptor (EGFR)-amplified and EGFR-mutant glioblastoma on the basis of a study using cultured glioblastoma cells. 215
The FDA-approved antidiabetic drug metformin and the FDA-approved antimalaria drug chloroquine have been tested in a phase IB clinical trial to investigate whether repurposing of these drugs can optimize standard treatment of IDH1-mutated glioblastoma and other IDH1-mutated cancer types, but failed to induce a clinical response.27,197,216 Alternatively, phenformin is a lipophilic analogue of metformin and may reach higher levels in mitochondria of cancer cells and thus may be more effective in the treatment of IDH1-mutated cancer cells. 196 Metformin and phenformin inhibit complex I of OXPHOS, whereas metformin, phenformin, and chloroquine inhibit α-KG production from glutamine and glutamate by glutamate dehydrogenase. 27 Effects of the FAO inhibitor etomoxir were compared with the effects of a ketogenic diet. It appeared that etomoxir prolonged survival of mice, whereas the ketogenic diet did not affect survival or even reduced survival of the mice. It was also found that IDH1 wild-type and IDH1-mutated cells were not differently affected by a ketogenic diet although IDH1-mutated cells use FAO in a different way than IDH1 wild-type cells. 198 In another study of the effects of a ketogenic diet in vitro and in vivo reported modest effects, if any. 217 Therefore, ketogenic diets for glioblastoma patients have to be applied with care to avoid unnecessary negative effects on the quality-of-life of glioblastoma patients because these diets are difficult to maintain.
Repurposing of anti-GSC drugs was reviewed recently. 218
A different approach was proposed by Hira et al.9,10 to sensitize GSCs to radiotherapy and chemotherapy. GSCs are kept in their hypoxic periarteriolar niches in glioblastoma tumors and the SVZ 19 by SDF-1α-CXCR4 interactions in a similar way as HSCs and LSCs are kept in their hypoxic periarteriolar bone marrow niches. Inhibition of CXCR4 by FDA-approved plerixafor is used successfully to remove LSCs out of the bone marrow niches to render them more sensitive to chemotherapy and HSCs in healthy donors to be harvested in the peripheral blood for stem cell transplantation. 10 It is suggested to remove GSCs from their niches in glioblastoma before radiotherapy or chemotherapy.9,10
Concluding Remarks
The present review of the literature shows clearly that quiescent NSCs and HSCs in their niches behave very similar with respect to energy metabolism. The cells are present in a hypoxic environment and depend on anaerobic glycolysis and FAO. When needed, both stem cell types are activated and become progenitor cell types that leave the NSC and HSC niches and switch to OXPHOS and lipid synthesis and downregulate glycolysis and FAO. In both cell types, similar cell biological regulation mechanisms of their stemness and ATP metabolism are involved (Fig. 6). Moreover, mitochondria are present in both quiescent stem cell types but are only partially active (FAO and not OXPHOS) and when needed can switch rapidly to an altered metabolism in proliferating progenitor cells (OXPHOS and lipid biogenesis but not FAO).
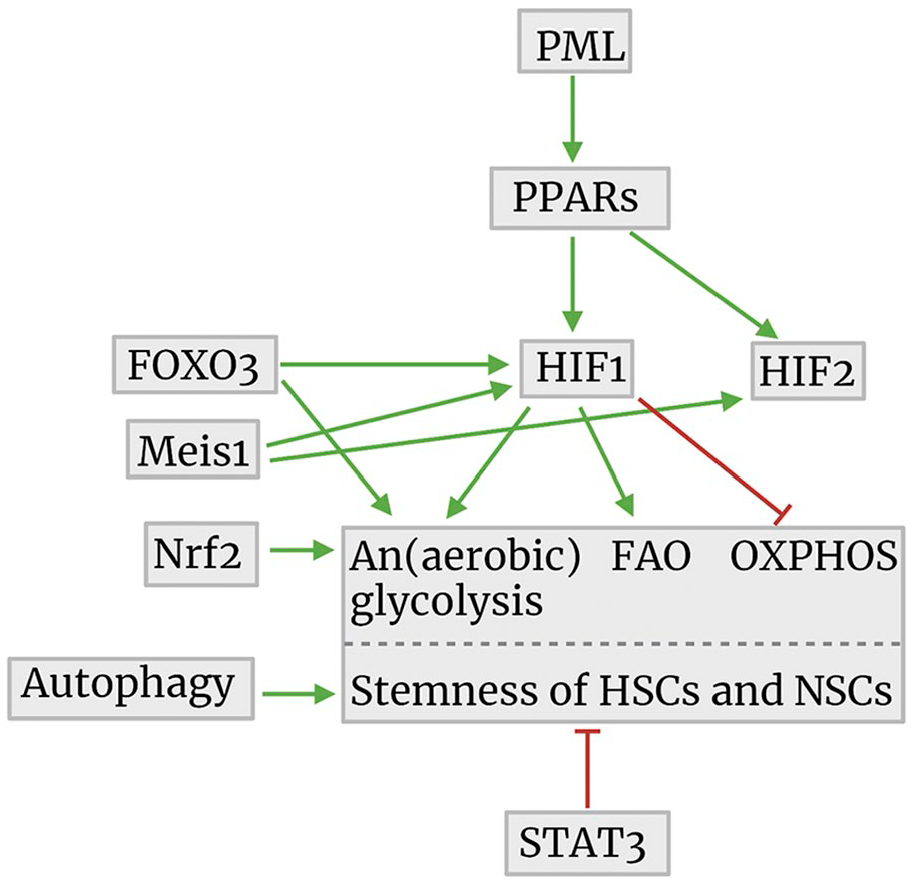
Cell biological regulation mechanisms of ATP synthesis in relationship to the maintenance of stemness of HSCs and NSCs. The image was created using Biorender.com. Abbreviations: PML, promyelocytic leukemia protein; PPARs, peroxisome proliferator-activated receptors; FOXO3, forkhead box O3; FAO, fatty acid oxidation; OXPHOS, oxidative phosphorylation; HSCs, hematopoietic stem cells; STAT3, signal transducer and activator of transcription 3; NSCs, neural stem cells; HIF, hypoxia-inducible factor; Meis1, myeloid ecotropic viral integration site 1; Nrf2, nuclear factor erythroid 2-related factor 2.
GSCs and LSCs appear to be quiescent and keep their stemness in similar ways with FAO and OXPHOS activity for ATP production. So, it seems that inhibition of OXPHOS is a therapeutic option to eradicate GSCs and LSCs to diminish the risk of recurrence of glioblastoma or AML (Fig. 7).
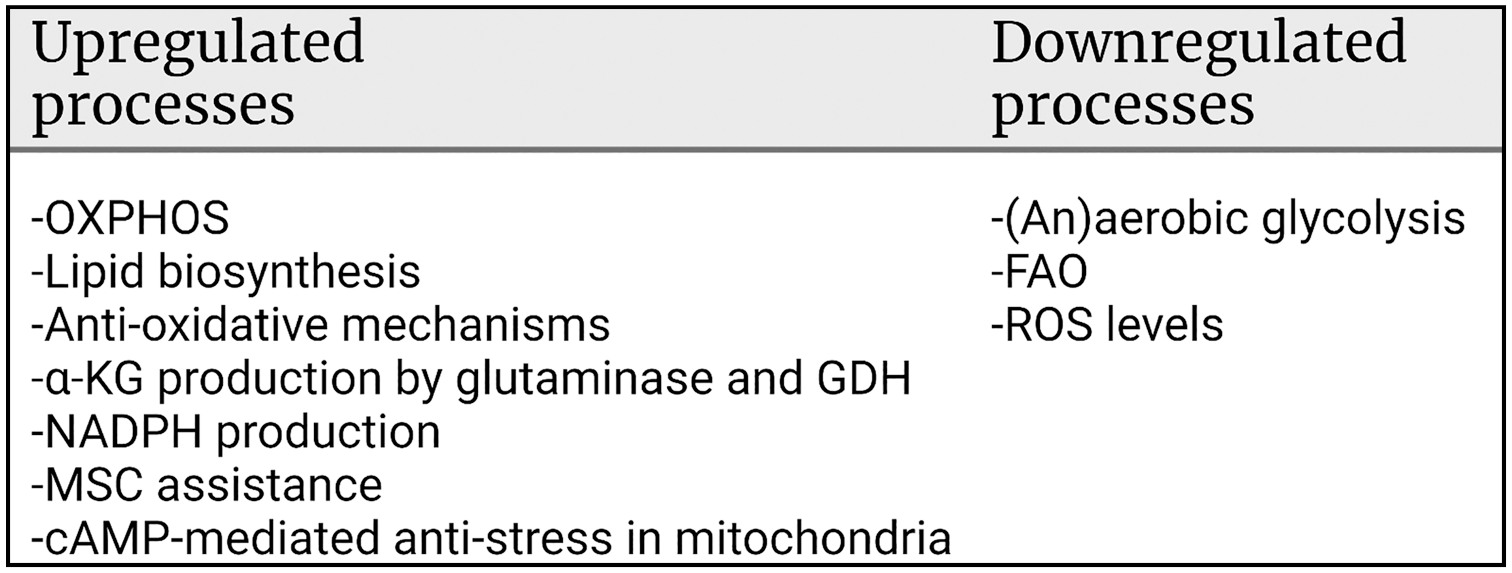
Metabolic regulation mechanisms in relation to the maintenance of stemness of LSCs and GSCs. The image was created using Biorender.com. Abbreviations: OXPHOS, oxidative phosphorylation; MSC, mesenchymal stem cell; FAO, fatty acid oxidation; ROS, reactive oxygen species; LSCs, leukemic stem cells; GSCs, glioblastoma stem cells; GDH, glutamate dehydrogenase; α-KG, α-ketoglutarate.
However, we argued recently against energy metabolism as target for therapy in cancer. 27 First, inhibition of aerobic glycolysis in differentiated cancer cells has proven in a host of clinical trials to be either ineffective or to cause unwanted side effects.144,211–213 Second, cancer is hypothesized to be a redox disease. 219 Therefore, Watson 220 argued that physical exercise prevents cancer as well as diabetes, dementia, and cardiovascular diseases by producing low levels of ROS that are needed for correctly folding proteins in the endoplasmic reticulum. Generation of low levels of ROS reduces the risk of cancer, 221 prolongs survival of cancer patients and delays recurrence of cancer, 222 and improves quality-of-life of cancer patients including glioblastoma patients.223,224 Because ROS are generated during OXPHOS,27,67 we conclude that systemic treatment of cancer patients with OXPHOS inhibitors is not an effective therapeutic option and leads to adverse effects in patients. Therefore, disruption of the interactions between CSCs and their protective niches seems to be a more promising option as therapeutic strategy because CSCs are then mobilized out of the niches resulting in their differentiation and proliferation and ultimately their sensitization to radiotherapy and chemotherapy.9,10 This approach is currently being investigated by using the FDA-approved drug plerixafor that inhibits CXCR4 to remove LSCs from bone marrow niches before chemotherapeutic treatment in AML and multiple myeloma patients and for harvesting HSCs in patients or healthy donors for HSC transplantation (for a recent review, see Hira et al. 10 ). Clinical trials are ongoing at the moment to investigate whether removal of GSCs from their niches before irradiation or temozolomide chemotherapy of glioblastoma patients is an effective therapeutic option. 10
In conclusion, energy metabolism (OXPHOS) in leukemic and GSCs is not a likely therapeutic avenue because OXPHOS is needed for ROS production in healthy cells and tissues of cancer patients as a first-line anticancer barrier.
Supplemental Material
sj-pdf-1-jhc-10.1369_00221554211054585 – Supplemental material for Cell Biology Meets Cell Metabolism: Energy Production Is Similar in Stem Cells and in Cancer Stem Cells in Brain and Bone Marrow
Supplemental material, sj-pdf-1-jhc-10.1369_00221554211054585 for Cell Biology Meets Cell Metabolism: Energy Production Is Similar in Stem Cells and in Cancer Stem Cells in Brain and Bone Marrow by Cornelis J.F. van Noorden, Barbara Breznik, Metka Novak, Amber J. van Dijck, Saloua Tanan, Miloš Vittori, Urban Bogataj, Noëlle Bakker, Joseph D. Khoury, Remco J. Molenaar and Vashendriya V.V. Hira in Journal of Histochemistry & Cytochemistry
Footnotes
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
The authors have contributed to this article as follows: conception and design (CJFvN, VVVH), collection and/or assembly of data (NB, MV, UB, JDK), data analysis and interpretation (CJFvN, BB, MN, MV, RJM, VVVH), initial analysis of energy metabolism in glioblastoma stem cells (AJvD) and hematopoietic stem cells (ST), fluorescence image processing (NB), electron microscopical study and imaging (MV, UB), manuscript writing (CJFvN, BB, MN, MV, RJM, VVVH), final approval of manuscript (CJFvN, BB, MN, AJvD, ST, NB, MV, UB, JDK, RJM, VVVH), and supervision of the entire study (CJFvN, BB, MV, VVVH).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Slovenian Research Agency (Project J3-2526; CJFvN, BB, MN, MV, UB) and the Postdoctoral project Z3-1870 (BB), the European Program of Cross-Border Cooperation for Slovenia-Italy Interreg TRANS-GLIOMA (Program 2017; BB, MN), the IVY Interreg Fellowship (VVVH), and the Fondation pour la Recherche Nuovo-Soldati 2019 (RJM).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
