Abstract
Recent studies have shown that cells from bone marrow (BM) can give rise to differentiated skeletal muscle fibers. However, the mechanisms and identities of the cell types involved remain unknown. We performed BM transplantation in acid α-glucosidase (GAA) knockout mice, a model of glycogen storage disease type II, and our observations suggested that the BM cells contribute to skeletal muscle fiber formation. Furthermore, we showed that most CD45+:Sca1+ cells have a donor character in regenerating muscle of recipient mice. Based on these findings, CD45+:Sca1+ cells were sorted from regenerating muscles. The cell number was increased with granulocyte colony-stimulating factor after cardiotoxin injury, and the cells were transplanted directly into the tibialis anterior (TA) muscles of GAA knockout mice. Sections of the TA muscles stained with anti-laminin-α2 antibody showed that the number of CD45+:Sca1+ cells contributing to muscle fiber formation and glycogen levels were decreased in transplanted muscles. Our results indicated that hematopoietic stem cells, such as CD45+:Sca1+ cells, are involved in skeletal muscle regeneration.
S
It has now been shown by single cell transplantation experiments that hematopoietic stem cells (HSCs) can give rise to progeny that reconstitute the blood and integrate into regenerating myofibers (Camargo et al. 2003; Corbel et al. 2003). However, whether the HSCs themselves or only a subset of HSC derivatives are the cells involved in this process remains to be determined. Definitive identification of the BMDCs that integrate into muscle fibers is required, because determination of the nature of the precise population of cells involved in this process will be critical in guiding future research in this field.
Here, we identified the HSC derivatives that have the capacity to incorporate into muscle fibers. These derivatives were isolated by fluorescence-activated cell sorter (FACS)-based fractionation of cells from damaged muscle, followed by direct transplantation into acid α-glucosidase (GAA) knockout mice, which is a mouse model of glycogen storage disease type II (GSD-II). GSD-II is an autosomal recessive lysosomal storage disease caused by a deficiency of GAA (Hers 1963). The disease is characterized by massive glycogen accumulation in the skeletal muscles.
The results of this study using transplantation into GAA knockout mice showed that HSCs contribute to muscle fiber formation.
Materials and Methods
Mice
C57BL/6J (wild-type) mice were purchased from Nihon Clea (Tokyo, Japan). GAA knockout mice and green fluorescent protein (GFP) mice were maintained in a pathogen-free environment. The GAA knockout mice were a kind gift from Dr. Raben (National Institutes of Health, Bethesda, MD). The GFP mice express GFP ubiquitously from the cytomegalovirus enhancer-chicken β-actin hybrid promoter (a kind gift from Dr. Okabe, Osaka University, Osaka, Japan). All animal experiments were approved by the Kyoto Prefectural University of Medicine Animal Care Committee.
Western Blotting Analysis
Soluble protein (50 μg/lane) was electrophoresed through denaturing SDS-16% polyacrylamide gels and electrotransferred onto nitrocellulose membranes. The membranes were blocked with 5% non-fat milk, incubated with a 1:1000 dilution of rabbit, anti-human GAA polyclonal antibody (Genzyme; Framingham, MA), washed, and probed with a 1:5000 dilution of horseradish peroxidase–conjugated anti-rabbit IgG derived from donkey (Amersham Pharmacia; Piscataway, NJ), and visualized using an enhanced chemiluminescence (ECL) detection system (Amersham Pharmacia).
BM Transplantation (BMT)
Previous studies have shown the ability of whole BM to give rise to skeletal muscle fibers. To evaluate BMT, whole BM cells were transplanted into lethally irradiated GAA knockout mice. BM cells were harvested from 8- to 10-week-old male wild-type and transgenic mice ubiquitously expressing GFP for Western blotting analysis and immunofluorescence analysis, respectively. The marrow of 8- to 10-week-old recipient GAA knockout mice was ablated by lethal irradiation (9 Gy), after which each mouse received 5 × 107 nucleated unfractionated BM cells by intravenous injection.
Muscle Injury and Muscle Regeneration
To induce muscle regeneration, a dose of 25 μl of 10 μM cardiotoxin (Sigma; St. Louis, MO) was injected directly into the tibialis anterior (TA) muscle as described previously (Polesskaya et al. 2003). At specified intervals after injury, mice were euthanized, and their hindlimb skeletal muscles were removed.
Recombinant human granulocyte colony-stimulating factor (G-CSF) (300 μg/kg/d, IP; Chugai Pharmaceutical, Tokyo, Japan) was given once a day on Days 0–4.
Real-time PCR
Total RNA was extracted using an RNeasy RNA purification system (Qiagen; Hilden, Germany) in accordance with the manufacturer's instructions. Real-time PCR analysis of Wnt5a mRNA was performed using a QuantiTect SYBR Green RT-PCR kit (Qiagen) in accordance with the manufacturer's instructions and processed on an ABI prism 7300 Sequence Detection System (Applied Biosystems; Foster City, CA), in which the mixture was heated to 50C for 30 min for reverse transcription and denatured at 95C for 15 min, followed by 40 cycles of 94C for 15 sec, 58C for 30 sec, and 72C for 30 sec. The following primers were used: Wnt5a (5′-TTTGGCAGGGTGATGCAAAT-3′ and 5′-GCGGTAGCCATAGTCGATGTT-3).
Preparation of Mononuclear Cells From Muscle for Transplantation
Cardiotoxin (CTX)-injured skeletal muscles were dissected, minced, and treated with 0.2% type II collagenase (Worthington Biochemical; Lakewood, NJ) for 40 min at 37C. Muscle slurries were filtered through 100-μm nitrex mesh (BD Biosciences; Bedford, MA). Mononuclear cells were washed twice with DMEM supplemented with 5% FBS and suspended at a concentration of 2–3 × 106 cells/ml. Staining was performed for 30 min on ice using the following antibodies: CD45-FITC or CD45-APC (BD Pharmingen; San Diego, CA) and Sca1-PE (Invitrogen; Eugene, OR).
Primary antibodies were diluted 1:200. Stained cells were analyzed with a FACSAria flow cytometer (Becton Dickinson; San Jose, CA). We used only propidium iodide–negative fractions for further experiments. Sorted cells were transplanted by local injection into TA muscle of GAA knockout mice.
Immunofluorescence Analysis and Periodic Acid-Schiff Staining
After transplantation, we analyzed the TA muscles from engrafted recipients for the presence of GFP+ cells by immunofluorescence analysis. TA muscles were snap frozen in isopentane, and serial sections 10 μm thick were cut on a cryostat, fixed in cold acetone, and stained overnight at 4C with a primary antibody against the basal lamina marker laminin-α2 (rat anti-mouse, 1:400; Alexis Biochemical, Lausen, Switzerland). After short washes in PBS, the sections were incubated with secondary antibody (goat anti-rat Alexa555; Molecular Probes, Eugene, OR) in blocking solution for 2 hr. The same sections were also stained with periodic acid-Schiff (PAS) according to standard methods.
Results
Contribution of BM Cells to Muscle Fibers In Vivo
Whole BM cells, derived from wild-type mice, were transplanted into lethally irradiated GAA knockout mice by intravenous injection. After 1 month, GAA protein was detected by Western blotting analysis in recipient mice (Figure 1). We assessed the contribution of BM cells from donor mice to skeletal muscle fibers by double immunofluorescent staining of GFP and laminin-α2. Some donor-derived GFP+ muscle fibers were shown stained for laminin-α2 expression in TA muscles of recipients (Figure 2). These findings indicated that BM cells generated skeletal muscle fibers in recipient mice.
Migration of BM-derived Cells to Regenerating Muscle
To clarify which types of donor cell contribute to muscle fibers, we analyzed the surface marker expression of recipient muscles. Recent studies have shown that stem cells derived from skeletal muscle express the hematopoietic markers CD45 and Sca-1 (Kawada and Ogawa 2001; McKinney-Freeman et al. 2002). We analyzed cells that expressed CD45 and Sca-1 in recipient muscle and found that the majority (>70%) of these cells were positive for GFP (Figure 3). It was proposed that CD45+:Sca-1+ cells migrated to the regenerating muscle derived from BM.
G-CSF Treatment Increases HSCs After Cardiotoxin-induced Injury
The contribution of HSCs to muscle is a response to tissue damage that was previously missed when studied in the absence of overt muscle injury (Wagers et al. 2002). In contrast, damage induced by delivery of toxins, such as cardiotoxin or notexin, has been shown to result in an increased contribution to skeletal muscle (Fukada et al. 2002; Camargo et al. 2003; Corbel et al. 2003; Abedi et al. 2007). CD45+:Sca-1+ cells showed a 30-fold expansion in number after cardiotoxin-induced injury. To be clinically useful, it is important to isolate and expand myogenic progenitor cells to improve the efficacy of cell transplantation in generating normal skeletal muscle. G-CSF has been reported to enhance tissue regeneration in myocardial infarction (Adachi et al. 2004). We examined whether G-CSF mobilized the CD45+:Sca-1+ cells into regenerating muscles. Five-day treatment with G-CSF significantly increased CD45+:Sca-1+ cells in regenerating muscles (Figures 4A–4C).
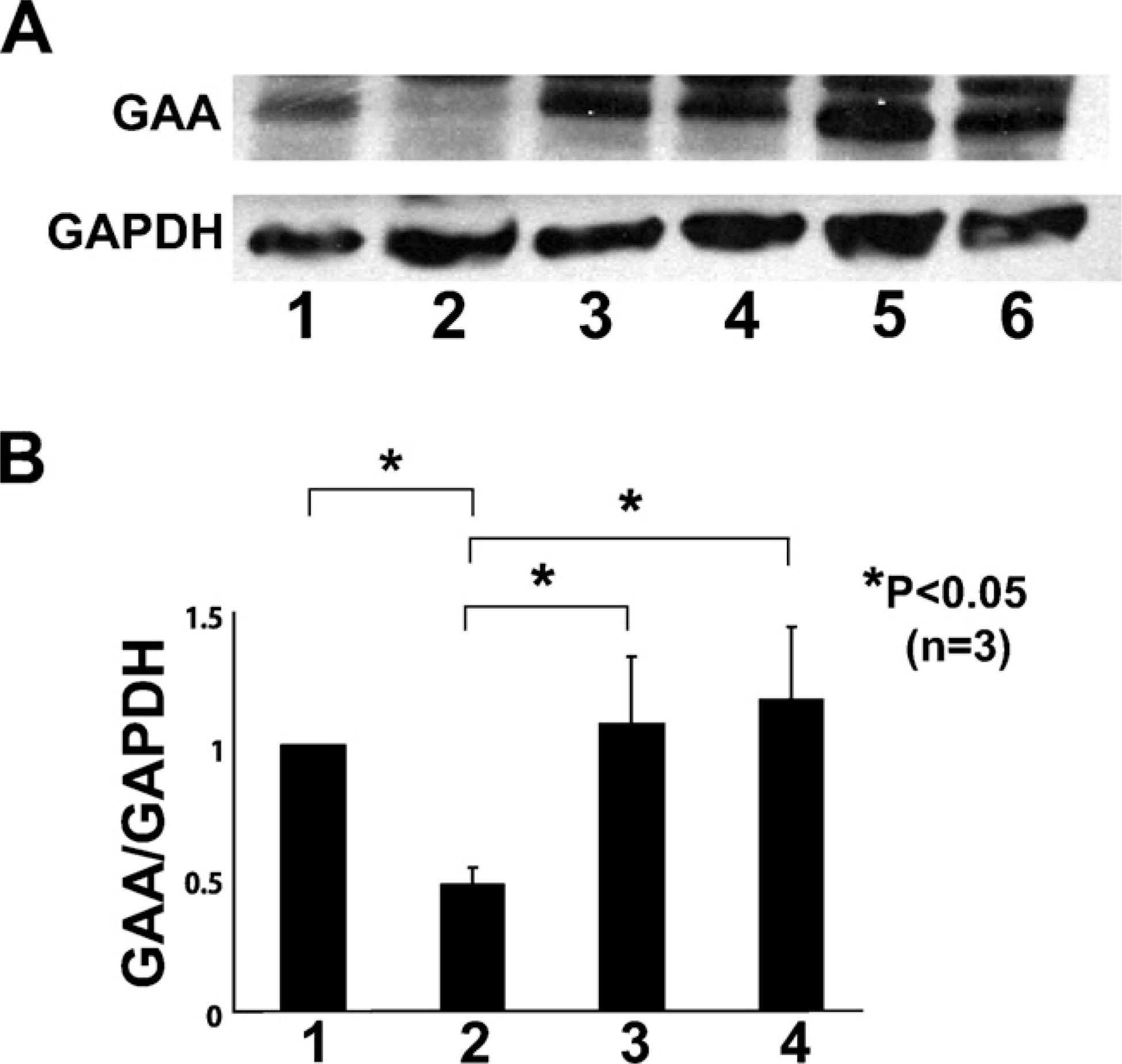
Expression analysis of α-glucosidase (GAA) in tibialis anterior (TA) muscle after whole bone marrow transplantation (BMT) or intramuscular injection of muscle-derived hematopoietic stem cells (HSCs; CD45+:Sca-1+ cells) into TA muscle. (
In addition, we analyzed Wnt5a mRNA expression in regenerating muscles by real-time PCR. Gene expression was assayed for 5 days after CTX muscle injury with and without 5-day treatment with G-CSF injection. Wnt5a was strongly expressed in CTX-injured muscles followed by 5-day treatment with G-CSF (Figure 4D).
HSCs Generate Skeletal Muscle in GAA Knockout Mice
To investigate the process of settlement of donor hematopoietic cells into skeletal muscles, the HSC fraction derived from injured skeletal muscles of GFP-transgenic mice was transplanted into TA muscles of GAA knockout mice. Thirty days after transplantation, we observed GFP+ muscle fibers surrounded by intact basal laminal membranes in the TA muscle (Figure 5). GAA protein was also detected by Western blotting analysis in recipient mice (Figure 1). This result suggested that transplanted CD45+:Sca-1+ cells generated the muscle tissues.
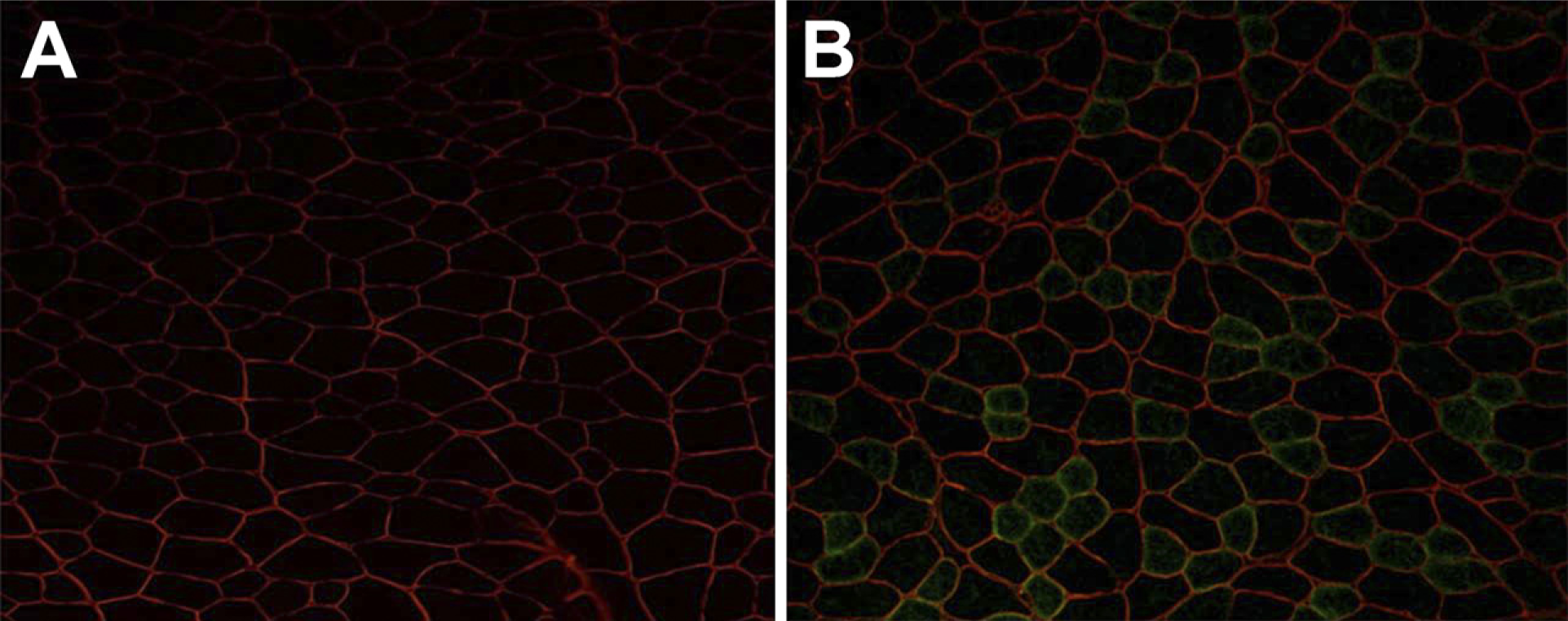
Bone marrow (BM) cells contribute to skeletal muscle fiber formation. Unfractionated green fluorescent protein (GFP)+ BM cells were transplanted into lethally X-irradiated GAA knockout mice. At 1 month after transplantation, cryostat sections of TA muscles were stained with anti-laminin-α2 antibody. Double staining for GFP (green) and laminin-α2 (red) on transverse sections of TA muscles of non-transplanted GAA knockout mice (
Efficiency of Transplantation With CD45+:Sca-1+ Cells
To assess the efficiency of transplantation with CD45+:Sca-1+ cells, we evaluated PAS-stained sections of TA muscles from GAA knockout mice (Figure 6A) as controls and GAA knockout mice with direct transplantation of CD45+:Sca-1+ cells (Figure 6B). Glycogen levels decreased in GAA knockout mice with direct transplantation of CD45+:Sca-1+ cells.
Discussion
In recent years, many studies have shown that BMDCs are involved in the regeneration of skeletal muscle fibers (Brazelton et al. 2003; Corbel et al. 2003; Palermo et al. 2005). In these experiments, the investigators used one of several cell tracking methods (i.e., transgenic mice expressing fluorescent proteins, male donors to female recipients, or lacZ transgenic mice) to follow the fate of the donor marrow cells in the recipient mice (Camargo et al. 2003). GAA knockout mice were adopted as a new cell tracking method in this study. In the TA muscle of the transplanted knockout mice, GAA was detectable by Western blotting, and GFP-positive muscle fibers were observed. Glycogen levels were reduced in PAS-stained sections of TA muscles. The type of marrow cells responsible for this phenomenon remains unknown. Although some researchers showed a mesenchymal origin of marrow-derived muscle fibers, others have suggested that hematopoietic cells are responsible for regeneration of muscle fibers (Ferrari et al. 1998; Weissman 2000; Blau et al. 2001; LaBarge and Blau 2002). Our observations indicated that CD45+:Sca-1+ cells were responsible for the appearance of GFP+ muscle fibers in recipient mice, which showed that hematopoietic cells were responsible for regeneration of muscle fibers. The cells were also responsible for the appearance of GAA. Recent studies have shown that stem cells derived from skeletal muscle exhibiting hematopoietic potential express the hematopoietic markers CD45 and Sca-1 (Kawada and Ogawa 2001; McKinney-Freeman et al. 2002). Wnt proteins constitute one of the most important families of signaling molecules in development, and these proteins also have vital roles in adult tissues—for example, in the regulation of cell proliferation and motility, generation of cell polarity, and specification of cell fate. Wnt signaling has been implicated in the control of differentiation of HSCs. Culture experiments with either mouse or human HSCs indicated that the Wnt family may act as growth factors for progenitor cell populations (Brandon et al. 2000). Others have reported that satellite cells can be derived from CD45+ cells on exposure to growth factors of the Wnt family (Polesskaya et al. 2003). Our results were consistent with these findings. FACS was used to subfractionate HSCs with CD45 and Sca-1 antibodies, the purity of which was 95% after sorting. Our experiments did not exclude the possibility that mesenchymal cells may also show differentiation to muscle fibers as they do in vitro. Some researchers have shown that BMDCs, including HSCs, give rise to muscle-specific stem cells (i.e., satellite cells) and fuse under physiological conditions to form mature myofibers. In this study, these cells were not cultured to exclude the possibility of contamination of mesenchymal cells. Whether all BMDCs follow the course remains unclear, and it is possible that a large proportion of cells contribute directly to myofibers and that both mechanisms coexist in the same tissue. Further cell lineage studies using genetic markers will be needed to define the intermediate cell types that may be involved in the blood-to-muscle pathway and the factors that mediate their fusion with muscle fibers.
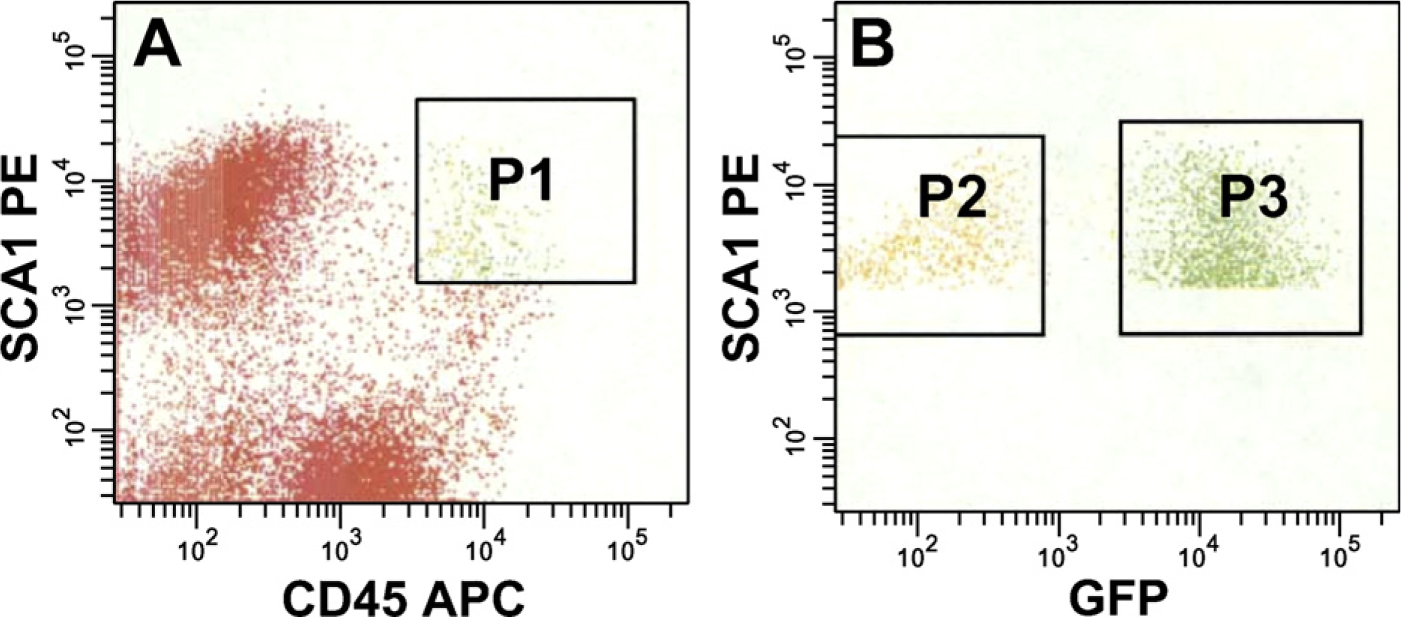
Migration of BM-derived cells to regenerating muscle. (
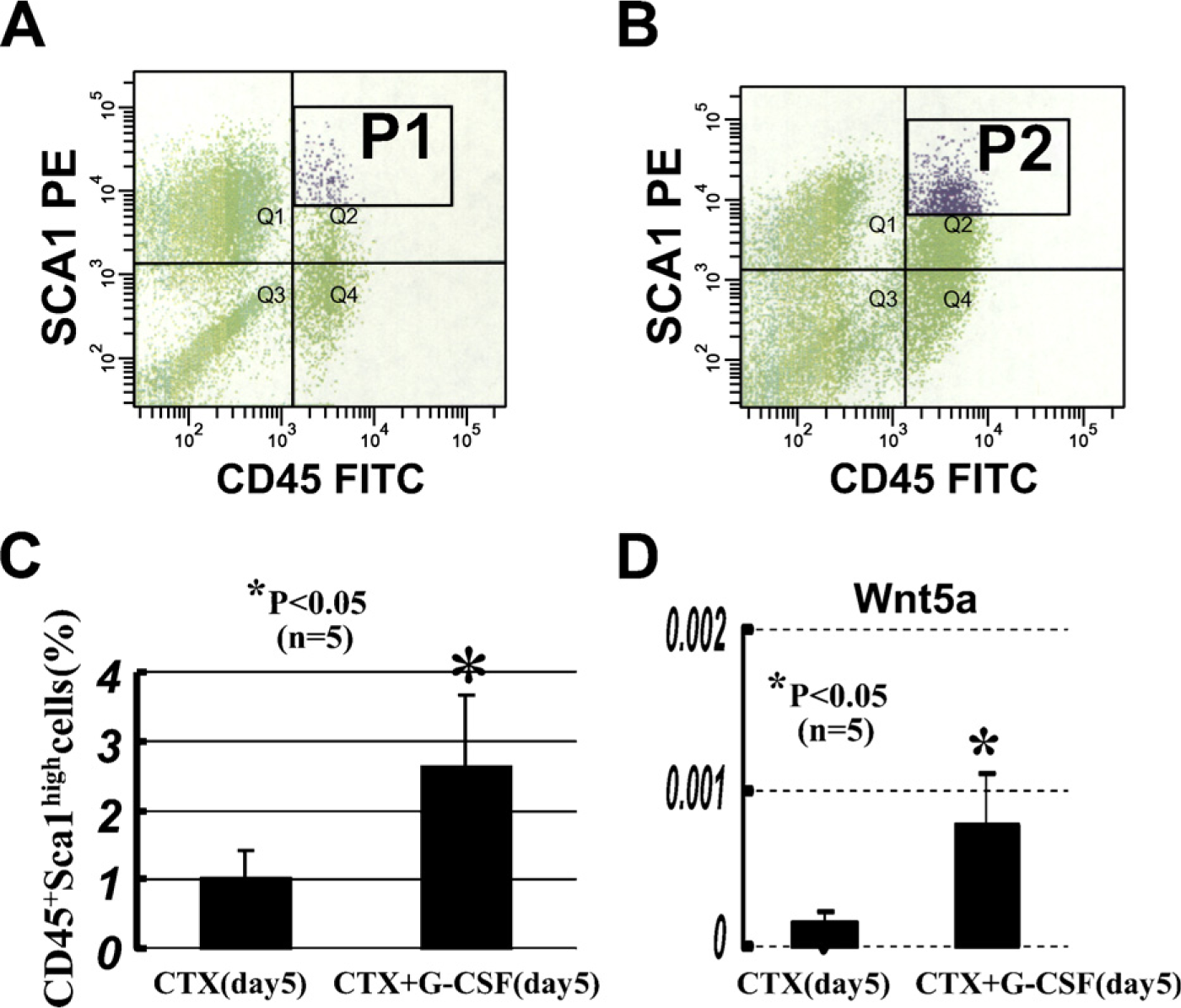
Fluorescence-activated cell sorter (FACS) analysis for infiltrating CD45+:Sca-1+ cells of cardiotoxin (CTX) injury (
CD45 is considered a lineage-restricted pan-hematopoietic marker that is not expressed on satellite cells (Asakura et al. 2002) or on any other non-hematopoietic cell type. Sca-1 is also a protein expressed in murine HSCs (Nakauchi et al. 1999). In addition, Sca-1–expressing cells isolated from muscle exhibit a high degree of plasticity (Torrente et al. 2001; Tamaki et al. 2002). HSCs express several surface markers, such as Sca-1 and c-kit (Goodell et al. 1997; Gussoni et al. 1999). Moreover, freshly isolated muscle side population (SP) cells express Sca-1 but not c-kit (Gussoni et al. 1999). We used CD45+:Sca-1+ cells isolated according to cell surface marker expression, which are not equivalent to SP cells (Morita et al. 2006). These cells are an attractive source for cell-based therapy for muscular disorders because they are thought to be disseminated to all muscles in the body via the circulation.
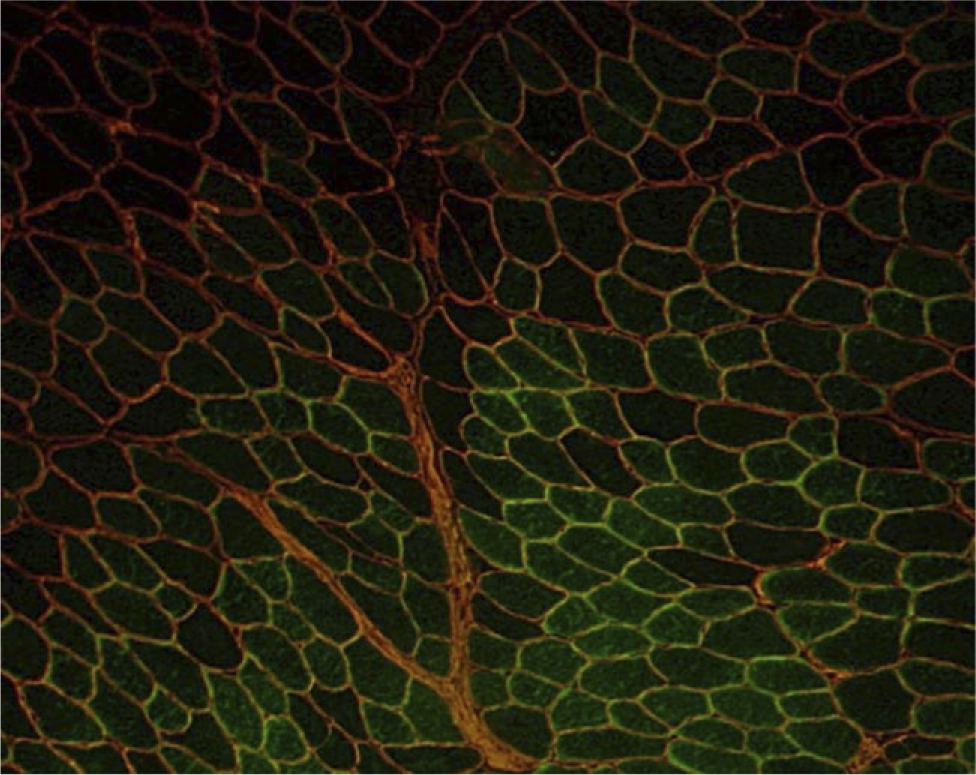
HSCs contribute to skeletal muscle fiber formation. GFP+ CD45+:Sca-1+ cells were transplanted into TA muscles of GAA knockout mice. At 1 month after transplantation, GFP+ muscle fibers (green) surrounded by intact basal laminal membranes (red) in a representative TA transverse section. GFP+ fibers observed in GAA knockout mice injected intramuscularly with CD45+:Sca-1+ cells were found to be 14.0 ± 3.0%. GFP+ fibers are reported as means ± SD of three sections separated by at least 100 μm (total fibers; n = 830–1050 for three independent experiments).
The results of this study using GAA knockout mice indicated that muscle-derived HSCs double-positive for Sca1 and CD45 are directly involved in the regeneration of muscle fibers. This myogenic activity is detectable from single cells injected directly into the TA muscle of GAA knockout mice. The observation that the derivatives of muscle-derived HSCs yield GFP+ myofibers is in good agreement with recent reports showing that individual transplanted muscle-derived HSCs can contribute to muscle regeneration (Camargo et al. 2003; Corbel et al. 2003). The identity and relationship between HSCs in BM and muscle-derived HSCs remain to be elucidated, but muscle-derived HSCs have been shown to adopt a myogenic fate in response to muscle injury (Polesskaya et al. 2003). The expression of GAA was also confirmed by Western blotting. In PAS-stained sections, muscle glycogen levels decreased in the TA muscles of treated GAA knockout mice. Thus, muscle-derived stem cells are of great interest as a potential source of myogenic progenitor cells that may be useful for cell transplantation. Enzyme replacement and adeno-associated virus vector-mediated gene therapies have been developed as effective therapeutic strategies for treating GSD-II. Further studies using subsets of HSC derivatives, such as CD45+:Sca-1+ cells, are needed to examine the therapeutic alternatives for GSD-II.
The frequency of BMDCs contributing to skeletal muscle fibers is generally reported to be low. In CTX injury, inflammatory cell infusion activates muscle regeneration. Muscle repair after CTX injury involves entry of inflammatory cells, particularly macrophages, followed by activation and fusion of satellite cells. Growth factors from macrophages may be needed to trigger satellite cell repair of muscle. Our study showed that CD45+:Sca-1+ cells are increased by G-CSF treatment. Because of its ability to mobilize granulocytes, G-CSF has been suggested to enhance the accumulation of inflammatory cells into the skeletal muscle (Duhrsen et al. 1988; Molineux et al. 1990; Lane et al. 1995). The relationship between G-CSF and Wnt signaling is unclear. Importantly, a large proportion of CD45+:Sca-1+ cells isolated from regenerating muscle seemed to represent a significant source of myogenic progenitors during regenerative myogenesis, and these cells were expanded by G-CSF.
In conclusion, our results indicated that G-CSF treatment enhances muscle-derived HSCs in regenerating muscles, and muscle-derived HSCs are involved in skeletal muscle regeneration using transplantation into GAA knockout mice.
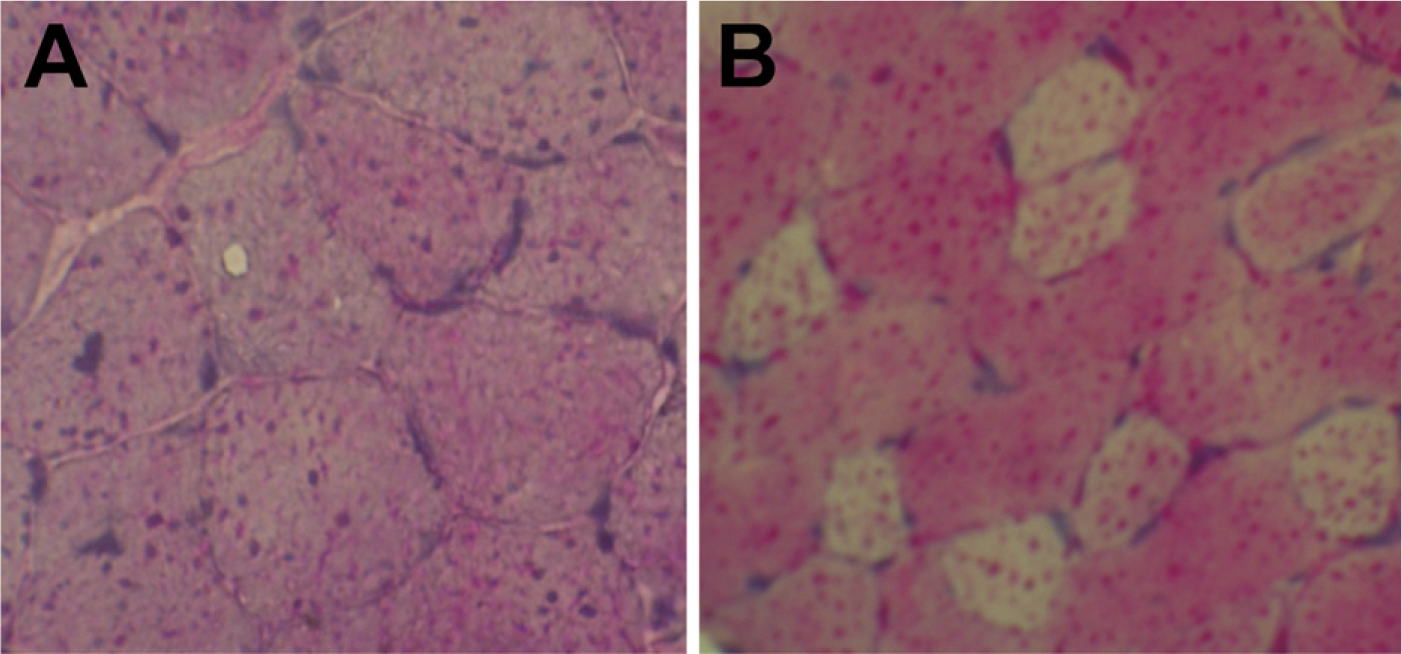
Periodic acid-Schiff–stained sections of TA muscles of GAA knockout mice (
Footnotes
Acknowledgements
We thank Drs. Okabe and Raben for providing GFP transgenic mice and GAA knockout mice, respectively, and Kenichi Fukazawa (Becton Dickinson) for support with flow cytometry.
