Abstract
We compared the time course of myogenic events in vivo in regenerating whole muscle grafts in MyoD(−/−) and control BALB/c adult mice using immunohistochemistry and electron microscopy. Immunohistochemistry with antibodies to desmin and myosin revealed a striking delay by about 3 days in the formation of myotubes in MyoD(−/−) autografts compared with BALB/c mice. However, myotube formation was not prevented, and autografts in both strains appeared similar by 8 days. Electron microscopy confirmed myotube formation in 8- but not 5-day MyoD(−/−) grafts. This pattern was not influenced by cross-transplantation experiments between strains examined at 5 days. Antibodies to proliferating cell nuclear antigen demonstrated an elevated level of replication by MyoD(−/−) myoblasts in autografts, and replication was sustained for about 3 days compared with controls. These data indicate that the delay in the onset of differentiation and hence fusion is related to extended proliferation of the MyoD(−/−) myoblasts. Overall, although muscle regeneration was delayed it was not impaired in MyoD(−/−) mice in this model.
T
Mice that lack a functional MyoD gene develop normally, with no overt morphological or physiological skeletal muscle abnormalities, and remain viable and fertile (Rudnicki et al. 1992), although the formation of some muscles is delayed and the muscles are smaller (Kablar et al. 1997). In MyoD(2/2) mice, the expression of Myf5 is increased and it was considered that this might act as a compensation for the lack of MyoD during the development of skeletal muscle (Rudnicki et al. 1992). In contrast, although Myf5 knockout mice appear to form normal skeletal muscle in utero, they die perinatally because of severe rib abnormalities that prevent respiratory function (Braun et al. 1992). Double knockout mice lacking both MyoD and Myf5 were devoid of all skeletal muscle (Rudnicki et al. 1993); the mutant pups were born alive but were immobile and died within minutes. Morphological and cytological examination of these mice showed that the spaces that are normally occupied by muscle were filled with loose connective and adipose tissue. These studies show that expression of either Myf5 or MyoD is absolutely essential for skeletal muscle formation in mice.
Skeletal muscle has an exceptional ability to regenerate after mechanical or chemical injury or pathological insult, and regeneration follows a series of clearly defined events, consisting of infiltration of inflammatory cells, satellite cell (myoblast) activation, and myoblast proliferation and fusion (Grounds 1991; Cullen 1997; Grounds and McGeachie 1999). A severe deficit in regenerative capacity has been reported in MyoD(−/−) mice in response to crush injury (Megeney et al. 1996). The crush injury model results in extensive disruption of cell relationships and muscle architecture, in contrast with whole muscle grafts in which there is relatively little damage to the muscle structure and new muscle formation occurs within the scaffolding of the old external lamina (Roberts and McGeachie 1995; Grounds and McGeachie 1999). The whole muscle graft is a highly reproducible model of regeneration and the pattern is similar throughout the length of the graft. We have used this model extensively in our laboratory and have documented (Roberts et al. 1989, 1997) the time course of the regenerative events in skeletal muscle of BALB/c mice (the parental strain of the MyoD(−/−) mice).
Because expression of MyoD (Grounds et al. 1992) and Myf5 (Mouly et al. 1997) are very rapidly upregulated after injury and appear to be essential for skeletal muscle formation during development, it is of particular interest to examine the effect that a lack of either of these genes has on regeneration in adult muscle. This can be readily studied in adult MyoD(−/−) mice but not in Myf5(−/−) mice, which die at birth (Braun et al. 1992). The present study examines regeneration in adult MyoD(−/−) mice using the whole muscle graft model at the light and electron microscopic level, using antibodies to desmin to identify myogenic cells (Kaufman and Foster 1988; Kaufman et al. 1991; Lawson-Smith and McGeachie 1998; Yablonka-Reuveni et al. 1999a) and to myosin heavy chain to identify myoblasts that have fused to form myotubes (Konieczny et al. 1983; Nadal-Ginard et al. 1984; Miller et al. 2000). Myotube formation was also observed using electron microscopy. In addition, the pattern of myoblast replication in grafts during the first 10 days after transplantation was quantitated after staining with antibodies to proliferating cell nuclear antigen (PCNA) (Bravo et al. 1987; Baserga 1991; Lawson-Smith and McGeachie 1998; Yablonka-Reuveni et al. 1999a,b). The amount of PCNA correlates with DNA synthesis during the cell cycle, becoming maximal during S-phase, and it is associated with DNA polymerase-δ (Bravo et al. 1987; Baserga 1991). To examine the relative influence of the (exogenous) host environment compared with intrinsic factors in the myoblasts of the graft, muscles were cross-transplanted between strains (Roberts et al. 1997). The results show that myoblast replication is sustained, leading to a delay in fusion of 2–3 days in regenerating adult MyoD(−/−) muscle, but that fusion is completed by about 8 days and autografts of both strains appear similar (by light microscopy) after this time.
Materials and Methods
Animals
Whole muscle grafts were performed on a total of 38 MyoD(−/−) and 24 BALB/c mice aged 8 weeks (see Table 1). A colony of MyoD(−/−) mice (originally provided by Michael Rudnicki) was established in the Animal Resource Centre at Murdoch University in Perth, Australia. Experiments were conducted in strict accordance with guidelines of the University of Western Australia Animal Ethics Committee and the National Health and Medical Research Council, Canberra, Australia. All animals were housed in individual cages under a 12-hr day/night cycle and allowed access to food and water ad libitum. The null mutation (exon 1 deletion) in MyoD(−/−) mice was confirmed using PCR with primers kindly provided by Dr. Marcia Ontell (Pittsburgh, PA). The BALB/c strain was used as the wild-type control in these studies for two reasons: (a) MyoD(−/−) mice are on a BALB/c-enriched genetic background, and (b) BALB/c muscle exhibits the least efficient regenerative capacity among the different strains investigated to date (Mitchell et al. 1992).
Surgical Procedure for Muscle Grafts
The transplantation procedure for whole muscle grafts has been described in detail previously (Roberts et al. 1989). In brief, the mice were anesthetized with a gaseous mixture of halothane, N2O, and O2. For autografts, the extensor digitoris longus (EDL) muscles were removed from both hindlegs of each mouse and were relocated over the tibialis anterior (TA) muscles of the same leg. Each EDL was sutured proximally to the distal tendon of the quadriceps femoris muscle and distally to the distal tendon of the TA, and the skin closed. Grafts were left for 2–14 days before sample collection and tissue processing (see Table 1).
To examine the relative influence of the (exogenous) host environment compared with (intrinsic) factors within the graft itself, muscles were cross-transplanted between strains. MyoD(−/−) muscles were implanted into both BALB/c and SJL/J hosts (and vice versa) because SJL/L mice show superior regeneration compared with BALB/c mice (Mitchell et al. 1992; Maley et al. 1994, 1995), and it has been demonstrated in cross-transplantation experiments that factors associated with the host environment determine the speed of regeneration in these strains (Roberts et al. 1997). All of the cross-transplanted (allografted) muscles were sampled at 5 days (see Table 1) before rejection of the “foreign” muscle by the host immune system occurred (Roberts et al. 1997).
Tissue Collection and Processing
Mice were sacrificed by cervical dislocation at various times (2–14 days) after grafting. In most cases, hindlegs were fixed immediately in 4% (w/v) paraformaldehyde in PBS for 60 min before transfer to 70% ethanol, in which muscles were dissected out and processed for paraffin sections. Fixed muscles were placed in a Shandon automatic tissue processor, immersed in 70% ethanol, and dehydrated through a series of graded ethanols before being infiltrated and embedded in paraffin. Other grafts were dissected out immediately and snap-frozen in isopentane quenched in liquid nitrogen for immunohistochemistry on frozen sections. In a few cases, muscles were fixed in 2.5% glutaraldehyde in cacodylate buffer for electron microscopy. The numbers of autografted mice sampled at various times after transplantation and processing of grafts for paraffins (P), frozens (F), or electron microscopy (G) are summarized in Table 1.
Details of all grafts performed a
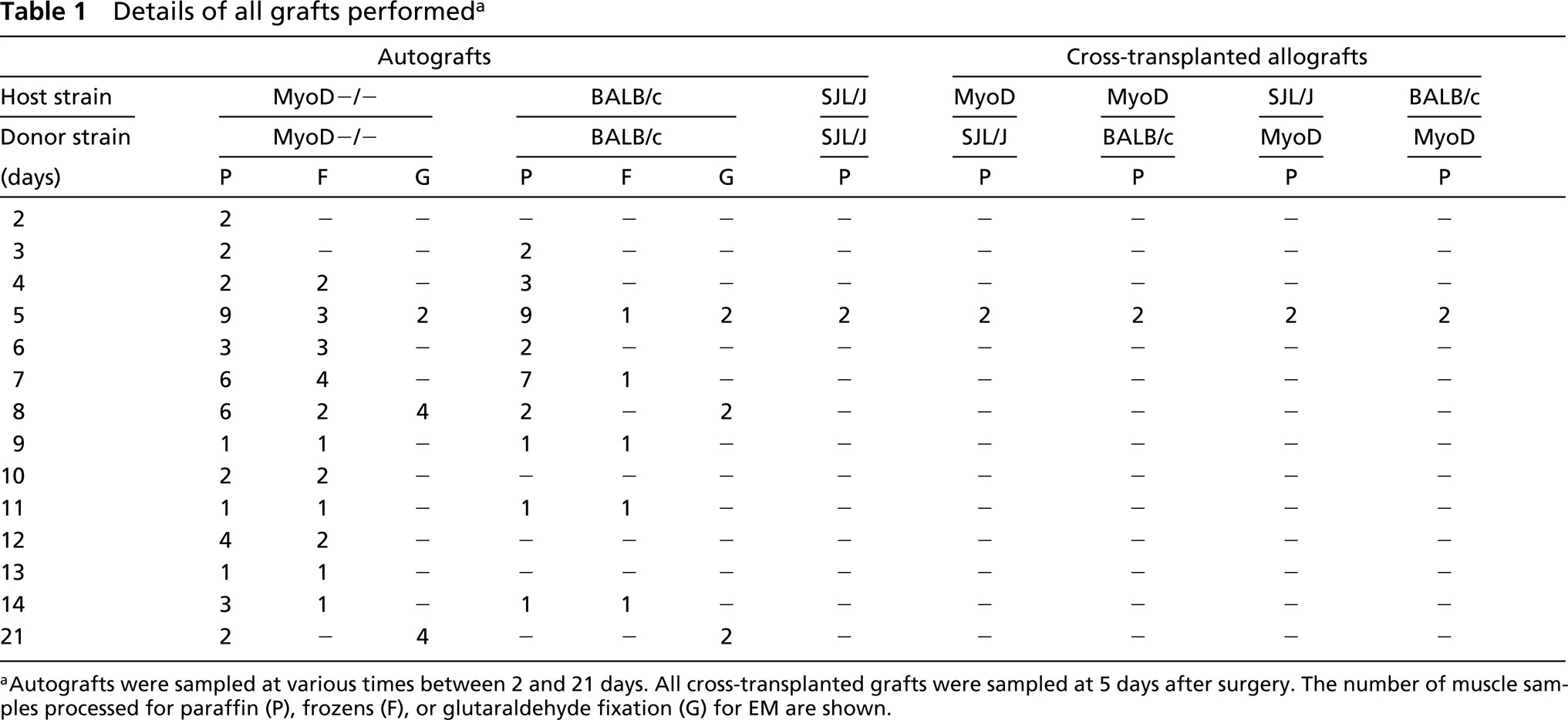

aAutografts were sampled at various times between 2 and 21 days. All cross-transplanted grafts were sampled at 5 days after surgery. The number of muscle samples processed for paraffin (P), frozens (F), or glutaraldehyde fixation (G) for EM are shown.
Immunohistochemistry
Antibodies. The primary antibodies used were a polyclonal rabbit anti-desmin (Biogenex; San Roman, CA), a mouse monoclonal anti-myosin fast heavy chain (Sigma, St Louis, MO; cat. M-4276), a mouse monoclonal anti-PCNA (Nova-Castra Laboratories; Newcastle upon Tyne, UK). Biotiny-lated secondary antibodies used were a donkey anti-mouse IgG (Jackson Immunoresearch Laboratories; West Chester, PA) and donkey anti-rabbit IgG (Jackson Immunoresearch). The biotin conjugates were detected with horseradish perox-idase-conjugated avidin D (Vector Laboratories; Burlin-game, CA).
Immunohistochemical Staining. Desmin and myosin were used to identify myoblasts and myotubes in the muscle grafts.
Desmin immunohistochemistry on paraffin sections required high-temperature antigen retrieval in citrate buffer, pH 6.0 (Lawson-Smith and McGeachie 1998). The slides were allowed to cool in the buffer, endogenous peroxidase was inactivated with 3% H2O2, and nonspecific antibody binding was blocked with 10% (v/v) horse serum. The primary antibody was diluted in 1% BSA–PBS–azide, added to the sections, and incubated at 4C overnight. Sections stained for desmin were sequentially incubated with a biotinylated donkey anti-rabbit and avidin D–peroxidase. Sections were washed three times in PBS between each incubation. Color development was achieved through incubation of the sections with the peroxidase substrate DAB (Pierce: Rockford, IL). Sections were counterstained with hematoxylin and mounted using DPX.
Myosin immunostaining was also carried out on paraffin sections using the antigen retrieval conditions described above. The Histomouse Kit (Zymed Laboratories; So. San Francisco, CA) was used to stain tissue sections for myosin on paraffin sections and for tropomyosin on tissue-cultured cells, because these monoclonals were mouse-derived and specific blocking procedures were required to reduce the background staining due to endogenous mouse immunoglobulins. The staining protocol used was per the manufacturer's instructions, based on the biotin–avidin amplification system with the secondaries conjugated to peroxidase.
Replicating cells in grafts were stained on paraffin sections using the LSAB kit (DAKO; Carpinteria, CA) for PCNA immunohistochemistry per the manufacturer's instructions.
Analysis of Tissues
Histological analysis and cell counts were performed using Image Pro Plus 4.0. The pattern of regeneration in whole muscle grafts is similar throughout its length. Therefore, all counts were based on representative sections from the midregion of each graft. The area of the central necrotic zone, the width of the peripheral regenerating zone, and the area occupied by myofibers before (Day 3) and after (Day 14) were calculated to assess the rate of regeneration in all grafts. In myosin-stained sections, the number of positive myotubes in an entire transverse section were counted and tagged to avoid double counting. PCNA-positive cells were counted in five sequential fields at ×400 magnification along the edge of each graft immediately adjacent to the underlying TA.
Results
Whole Muscle grafts (Autografts)
Histology. The typical histological appearance of a transverse section through a whole EDL muscle autograft is shown in Figure 1, and the sequence of histological events during graft regeneration is well described elsewhere (Roberts et al. 1989; Roberts and McGeachie 1995). During the first week after transplantation, there were marked histological differences in the appearance of MyoD(−/−) compared to BALB/c autografts, and these are summarized in Table 2. At all times examined after 8 days, MyoD(−/−) and BALB/c grafts were histologically indistinguishable at the light microscopic level, with myotubes present throughout the entire graft (Figures 2G and 2H). Although inflammatory cells infiltrated BALB/c grafts earlier (Table 2), after Day 3 morphometric analyses of the central necrotic and peripheral regenerating zone in both MyoD(−/−) and BALB/c grafts did not show any significant difference in the rate of progression of the inflammatory zone of regeneration (data not shown).
Desmin Immunostaining of Paraffin Sections
Desmin is an excellent marker for identifying activated myoblasts and myotubes in vivo (Lawson-Smith and McGeachie 1998). Desmin–positive cells were first seen in the periphery of the graft in the regenerating zone close to the TA interface. As regeneration continued, desmin-positive cells were seen lining the contour of necrotic myofibers: these “cuffing cells” (Figure 2D) developed into plump myotubes over time. In BALB/c autografts, desmin-positive myoblasts were present at Day 3 and increased in number after this time. Myotubes were clearly present at Day 4 and were pronounced from Day 5. Typical intense staining of desmin-positive myoblasts and myotubes in BALB/c autografts is shown in Figure 2B.
The pattern of desmin immunostaining was markedly different in MyoD(−/−) autografts. In most grafts up to 7 days, the desmin staining was consistently weak and patchy and the cell boundaries of desmin-positive cells in these grafts were hard to define (Figures 2A and 2C) compared to BALB/c grafts (Figures 2B and 2D). At 7 days, three of the five grafts contained many desmin-positive myotubes and these grafts resembled the Day 5 BALB/c grafts. By Day 8, the number of myotubes in MyoD(−/−) grafts increased to a level similar to BALB/c grafts of the same age.
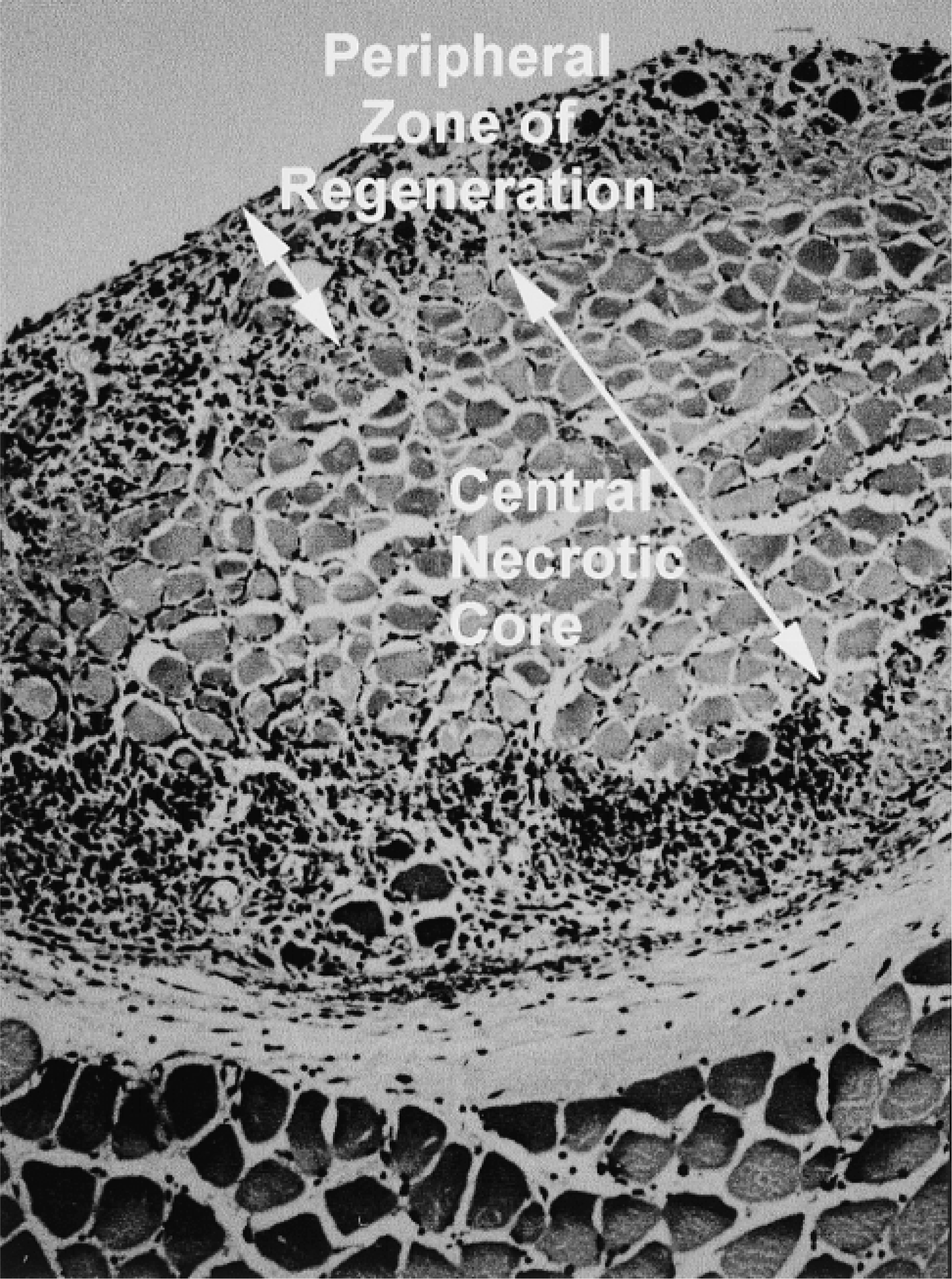
Typical histology of a transverse section through the mid-region of an EDL whole muscle graft and the underlying host TA muscle at 5 days. The peripheral regenerating zone of inflammatory cells and myogenic activity in the graft surrounds the central necrotic core. Note: the same orientation of the graft (at the top) and host (below) is used in all graft photomicrographs.
Myotube Formation in Whole Muscle Autografts
Myosin was used as a marker for the initial stages of myoblast differentiation and subsequent fusion to form myotubes (Figures 2E and 2F). The formation of myosin-positive myotubes was delayed in MyoD(−/−) compared to BALB/c grafts, and the numbers of myosin positive myotubes in typical autografts are summarized in Figure 3. Intensely stained myotubes were present in all four BALB/c grafts at Day 5 (278 myotubes in one typical graft; Figure 2F). In contrast, only very few or no myosin-positive myotubes were seen in all four MyoD(−/−) grafts at Day 5. Similarly, two of the three MyoD(−/−) grafts at Day 6 contained few myotubes (0 and 9 myotubes), although a single MyoD(−/−) graft had significant numbers of myosin-positive myotubes at this time (177 myotubes). At 7 days after grafting, significant numbers of myosin-positive myotubes were seen in most of the five BALB/c grafts. Only two of the five MyoD grafts (Figure 2E) contained many myotubes (229 and 289 myotubes), with the remaining three having 1, 38, and 63 myotubes. At Day 7, the myotubes in both MyoD(−/−) and BALB/c grafts were present mainly in the peripheral regenerating zone, whereas two BALB/c grafts had myotubes in the central regions of the graft. In all grafts examined on Day 8 and later, the distribution of myosinpositive myotubes appeared similar in MyoD(−/−) and BALB/c grafts, although a quantitative analysis was not undertaken.
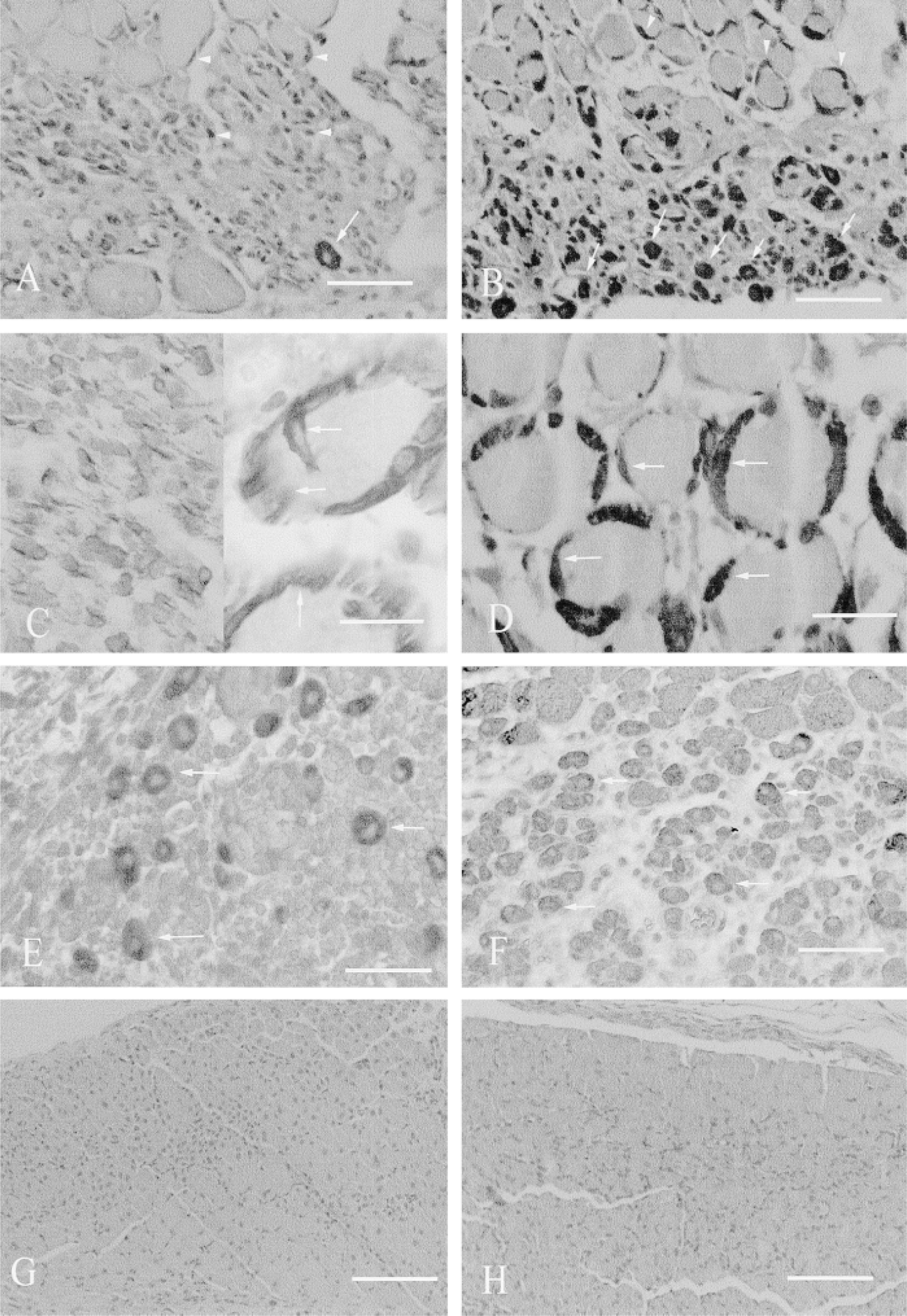
Histology of MyoD(−/−) and BALB/c autografts, with desmin (
EM Studies
Electron microscopy was carried out to observe myoblasts and myotube formation in vivo in MyoD(−/−) and BALB/c Day 5 autografts (Figures 4A and 4B), partially because of difficulties in distinguishing between myoblasts and young myotubes in transverse sections by desmin immunostaining in the MyoD(−/−) grafts. In transverse sections, large numbers of apparently mononucleated “cuffing” cells were seen lying within the contour of the persisting basement membrane of myofibers in MyoD(−/−) autografts (data not shown). Examination of longitudinal sections (LS) clearly defined these as unfused mononucleated cells in the 5-day grafts (Figure 4A). Extensive observations at high power revealed no evidence of sarcomeric organization (indicative of differentiation) in these mononucleated cells but, on the basis of their location and appearance, it appeared likely that they were indeed myoblasts. Large numbers of cells with mitotic figures were noted in these MyoD(−/−) autografts compared with the BALB/c grafts (data not shown). In contrast, in LS sections of BALB autografts at Day 5, mutinucleated myotubes were conspicuous adjacent to surviving myofibers (Figure 4B), in areas where no necrotic sarcoplasm remained, and in areas where many inflammatory cells were present (not shown).
Histological summary of the progression of regeneration in autografts a
aAfter 8 days, grafts from both strains appeared equivalent by light microscopy.
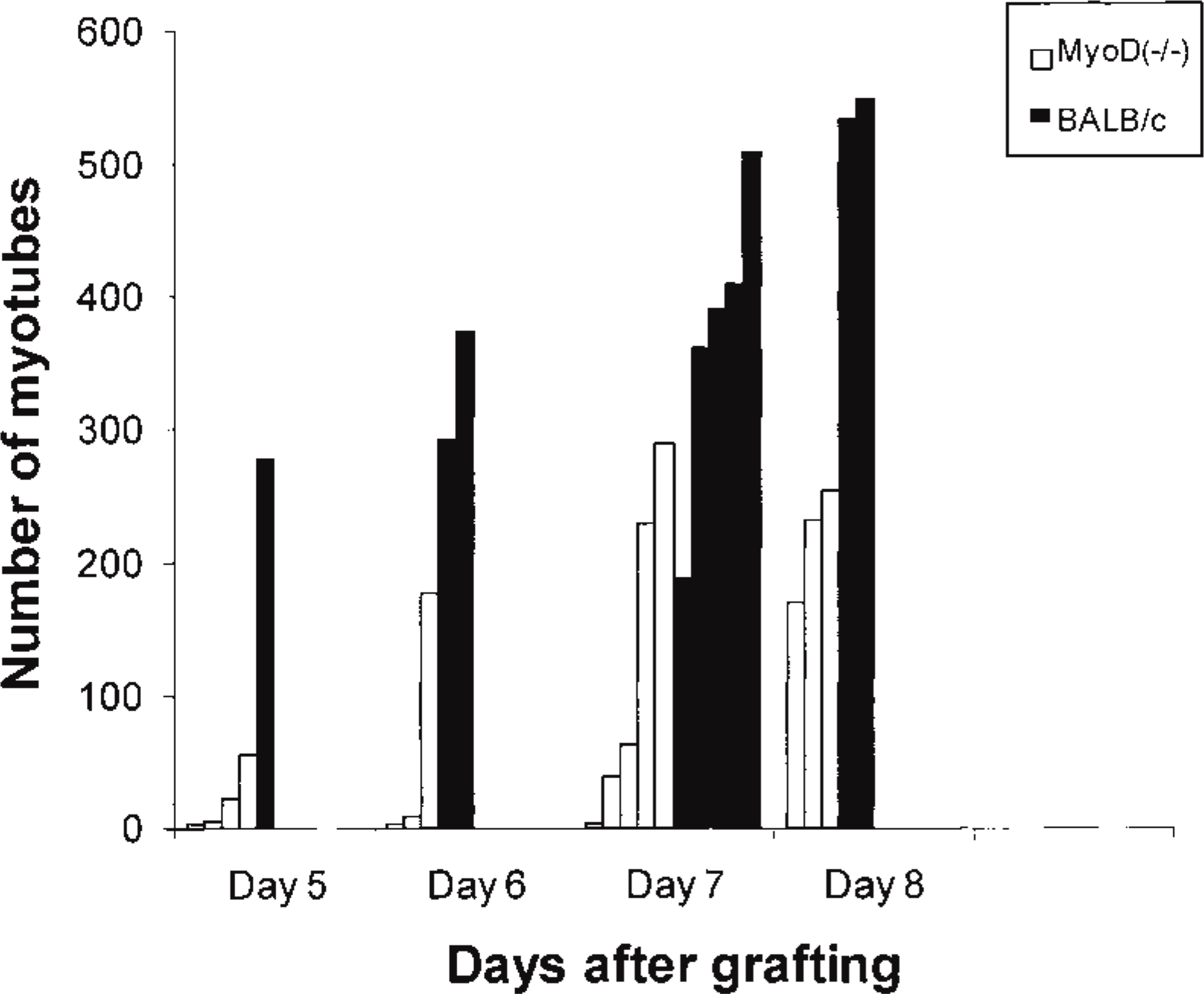
Numbers of myosin-positive myotubes in transverse sections of MyoD(−/−) and BALB/c autografts are shown at various times after transplantation. Each vertical column represents a single graft. Only one BALB/c graft was analyzed in detail because this was typical of all four grafts.
Additional EM was carried out on 8- and 21-day autografts from both strains to specifically observe the myotubes and numbers of potential satellite cells. The myotubes in Day 8 MyoD(−/−) grafts had many closely packed nuclei that were a little “disorganized” (Figure 4C), and these myotubes resembled the (newly formed) myotubes in Day 5 BALB/c grafts (Figure 4B). In the 8-day MyoD(−/−) grafts, although many unfused cells (presumably myoblasts) were present, none was observed closely apposed to the surface of new myotubes or in a classical satellite cell position (beneath the basement membrane) in either MyoD(−/−) or BALB/c autografts. It was considered that such satellite cells might not be evident until the newly formed myofibers had matured further, and therefore grafts from both strains were also sampled at 21 days for EM analysis. Myotubes in 21-day grafts from both strains had highly organized contractile apparatus (Figures 4D and 4E) and appeared of similar size in terms of both diameter and length. In the four MyoD(−/−) grafts examined, mononuclear cells in the classical satellite cell position under the basement membrane (Figure 4F) appeared more frequent compared with the two BALB/c grafts examined. A comprehensive quantitation was not undertaken, but counting of all satellite cells in 10 grid squares (one grid square is 55 × 55 nm) in single sections (11 nm thick) revealed five and 11 satellite cells in the two MyoD(−/−) grafts (from different animals) and two and two satellite cells in the two BALB/c grafts (from the same animal). There was no marked difference between the two strains in the number of unfused mononuclear cells lying outside the basement membrane. All grafts had extensive interstitial connective tissue and many endothelial cells in intimate contact with myotubes. Nerve endplates were obvious in both MyoD(−/−) and BALB/c grafts, indicating innervation of the grafts.
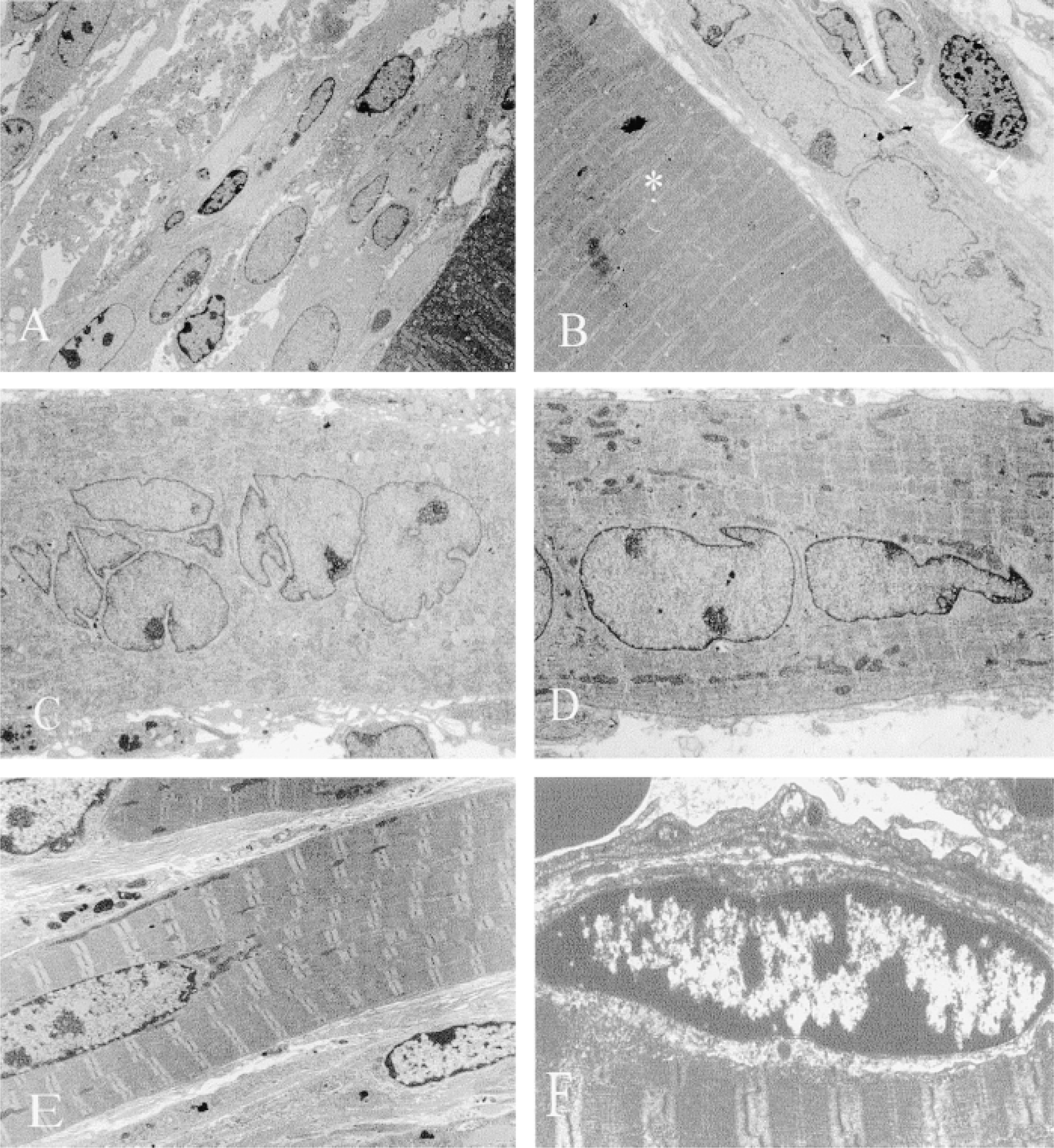
Electron microscopy of MyoD(−/−) and BALB/c autografts. (
Cell Proliferation in Grafts
The mononuclear cells labeled by PCNA immunohistochemistry fell into two broad groups: (a) those that were within or closely associated with persisting or new myofibers, which were considered to be myoblasts (Figure 5A); and (b) other cells that included macrophages, fibroblasts, endothelial cells, and unidentified cells, some of which may have been myogenic (Figure 5A). It is acknowledged that some of the cells located in and closely apposed to the basement membrane of necrotic myofibers may also have been macrophages, but for the purposes of this study this was not considered to be significant. When PCNA-stained sections were examined in parallel zones of both strains of autografts (as described earlier), marked differences in the distribution of PCNA-positive cells were noted, and this was especially apparent in Day 5 grafts. In regions of MyoD(−/−) grafts where little necrotic muscle remained, there were few or no myotubes and many PCNA-positive cells (Figure 5B). In contrast, in BALB/c grafts where little necrotic tissue remained, myotube formation was conspicuous and few PCNA-positive cells were present (Figure 5C).
The number of replicating myoblasts (identified as described above) in grafts at 3–14 days after transplantation was quantitated and expressed as a percentage of the total number of proliferating cells using PCNA immunohistochemistry (Figure 6). The peak in myogenic cell replication (50% of total replicating cells) in BALB/c grafts occurred by 6 days, after which time replication declined markedly. In MyoD(−/−) grafts, extensive myogenic cell replication was seen from 3 days to 8 days. The decrease in replication observed in BALB/c grafts after 6 days was not seen until 9 days in the MyoD(−/−) grafts. After 8 days in the MyoD(−/−) grafts, the level of replication decreased rapidly to that observed in BALB/c grafts (Figure 6).
Cross-transplantation Studies
In cross-transplantation studies (Table 1), BALB/c and SJL/J EDL muscles were allografted into MyoD(−/−) hosts to see whether the rate and extent of myotube formation in “normal” grafts was affected by the MyoD-deficient environment. Five days after grafting these were directly compared to autografts of the same species (e.g., SJL/J cross-transplants were compared with SJL/J autografts of the same age). Histological comparison revealed no marked differences between these cross-transplants and the equivalent autograft controls in terms of the infiltration of mononuclear cells and the number and distribution of myotubes (data not shown). Where MyoD(−/−) grafts were allografted into BALB/c or SJL/J hosts to see whether the normal host environment would modify regeneration in the null graft, the time of myotube formation within the MyoD(−/−) graft was not affected (data not shown), and these grafts were indistinguishable from MyoD(−/−) autografts at 5 days, when no or few myotubes were present.
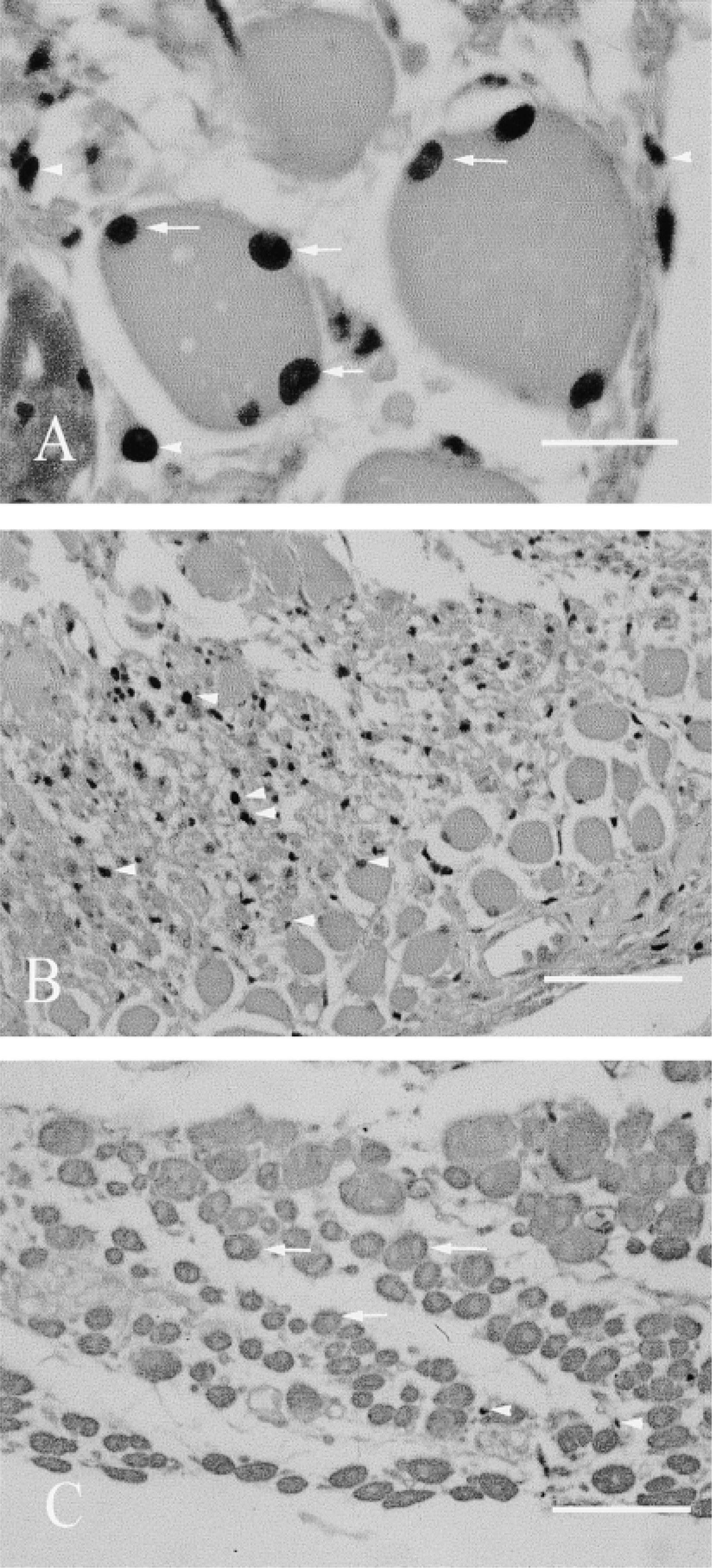
PCNA immunohistochemical staining shows two types of positively stained nuclei. (
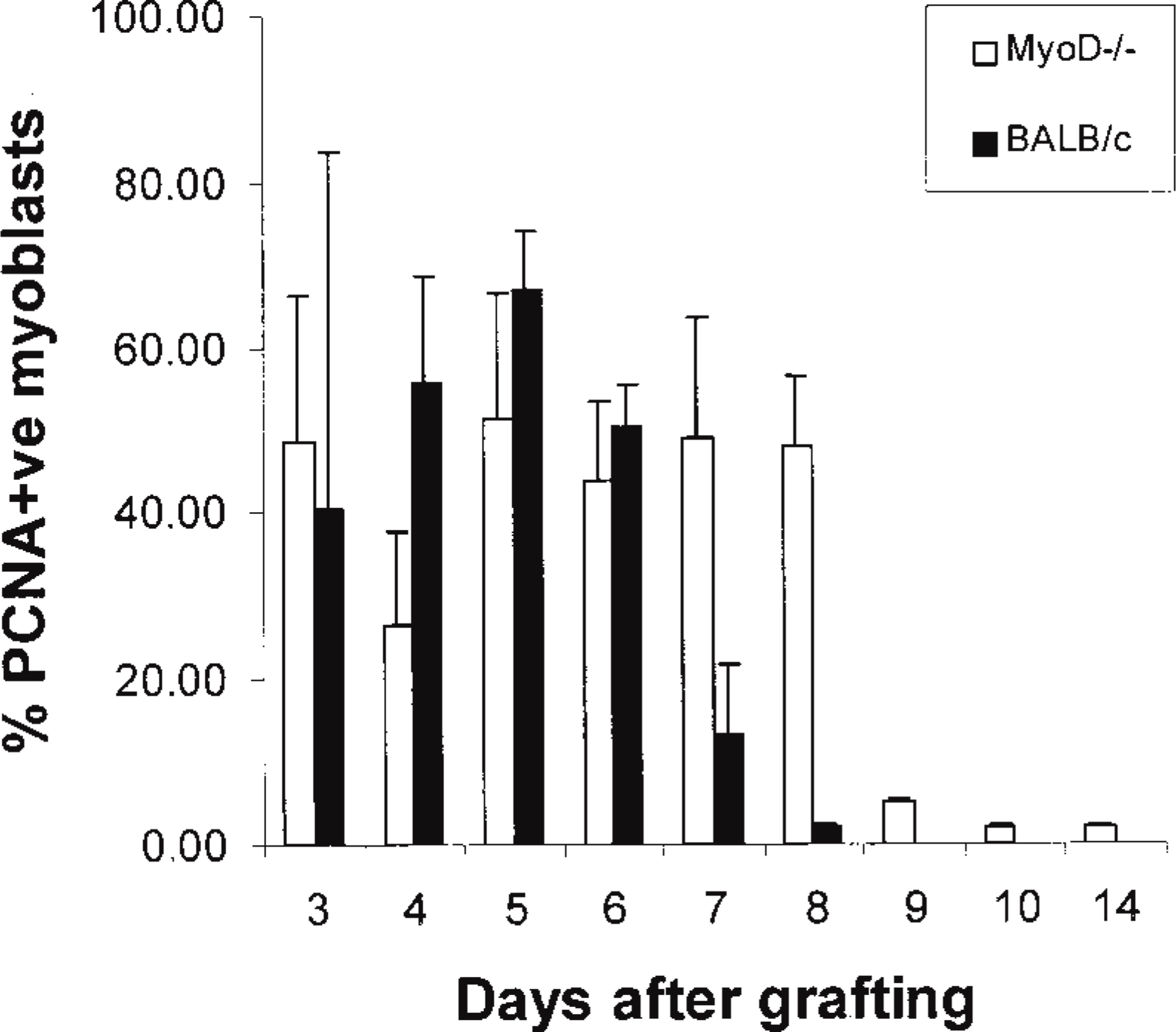
Quantitation of myogenic cell replication in whole muscle grafts using PCNA immunohistochemistry. The total number of PCNA-positive (replicating) cells was counted. The number of PCNA-positive myogenic cells, i.e., intimately associated with myofibers (see Figure 5A), was calculated as a percentage of the total number of PCNA-labeled cells.
Discussion
Myotube Formation Is Delayed During Regeneration of Whole MyoD(−/−) Muscle Grafts
The whole muscle graft is a highly reproducible model of regeneration, and this study demonstrates a striking delay in the timing, but not an inhibition, of myoblast fusion to form myotubes in vivo in skeletal muscle deficient in MyoD. Long-term grafts of MyoD(−/−) and control BALB/c muscle appeared very similar at the light and EM levels. The delayed formation of hypaxial muscle during embryogenesis also indicates that a similar delay in fusion may occur in developing MyoD(−/−) muscles (Kablar et al. 1997). Our observations highlight the importance of observing skeletal muscle regeneration at a number of timepoints, especially during the very early events. Observations limited to only a single time may well lead to incorrect assumptions. For example, at early times there is an obvious delay in the formation of myotubes and, if no later times were considered, it might be concluded that there was a total inhibition of the regenerative process. Conversely, examination of grafts only after 8 days would indicate no difference in the regenerative process between MyoD(−/−) and control muscles.
Is Myotube Formation Impaired in MyoD(−/−) Muscles?
Previous studies in MyoD(−/−) muscle regenerating after crush injury (Megeney et al. 1996) report a severe impairment of regeneration at both 4.5 and 14 days, in marked contrast to the results in whole muscle grafts. This may be largely accounted for by the nature of the injury, because the model used to study myogenesis in adult muscle can influence the pattern of regeneration observed (Lefaucheur and Sebille 1995). For example, whereas there is no overall difference in the appearance of whole muscle autografts between SJL/J and BALB/c mice (Roberts et al. 1997), a dramatic difference in the capacity for myotube formation between these strains is seen where the tissue architecture is severely disrupted in minced grafts (Grounds and McGeachie 1990) or crush-injured muscles (Mitchell et al. 1992). This appears to be due to the nature of the ECM substrate, which can have a striking influence on the efficiency of myotube formation. This is very marked in BALB/c mice, in which myotube formation is severely impaired (relative to SJL/J mice) on interstitial-type ECM substrates in vitro (Maley et al. 1995) and in vivo (Grounds 1990; Mitchell et al. 1992; Grounds et al. 1998), compared with environments rich in basement membrane components (Maley et al. 1995), e.g., whole muscle grafts (Roberts and McGeachie 1995; Roberts et al. 1997). Because the background strain for the MyoD(−/−) mice is BALB/c, this sensitivity to the ECM substrate may well exaggerate the delayed myotube formation in crushinjured MyoD(−/−) muscle (Megeney et al. 1996) compared with the excellent regeneration seen in whole muscle grafts in the present study. The extent to which this sensitivity to interaction with the ECM might influence myogenesis in MyoD(−/−) muscles remains to be further investigated.
The question arises of whether myotube formation by MyoD(−/−) myoblasts (apart from being delayed) is also impaired in vivo. This was the conclusion of the crush injury study by Megeney et al. (1996) but does not seem to be the case in the whole muscle grafts. In the MyoD(−/−) whole muscle grafts at Day 8, when many myotubes were present throughout the graft, our EM studies showed many “unfused” cells that were probably myoblasts. Although this might have been a consequence of the relatively recent onset of myotube formation, there appeared to be more than in Day 5 BALB/c grafts at a comparable stage of early fusion. However, this is probably accounted for by a surfeit of myoblasts produced by the extended phase of proliferation in the MyoD(−/−) grafts. The 21-day MyoD(−/−) grafts showed increased (up to fourfold) numbers of satellite cells compared to BALB/c grafts. Again, this may simply be due to the sustained myoblast proliferation that occurred in MyoD(−/−) grafts. The increase observed here (more than double) correspond roughly with the reported 2.5-fold difference in the proportion of satellite cells (% expressed per total number of sub-laminar nuclei) in 4-month-old mdx:MyoD(−/−) mice (8.3%) compared with agematched parental mdx (3.3%) and MyoD(−/−) mice (2.9%) (Megeney et al. 1996).
The severe pathology resulting from MyoD deficiency in dystrophic mdx mice (Megeney et al. 1996) is more difficult to account for. The endogenous “focal” injury in dystrophic muscles consists of segmental necrosis in small clusters of adjacent myofibers and, because regeneration occurs within the basement membrane of these damaged myofibers, this appears to resemble more closely the situation in whole muscle grafts (rather than crush-injured tissue). Therefore, other factors, perhaps associated with the dystrophic process or with the reduced amount of muscle formed during development in MyoD-deficient mice (Kablar et al. 1997), presumably must exacerbate the effects of the delayed myoblast fusion and contribute to the pathology in the MyoD(−/−)/mdx mice over time.
Sustained Replication of MyoD(−/−) Myoblasts
Comparison of cell replication in MyoD(−/−) and control BALB/c whole muscle autografts showed a high level of PCNA immunostaining in MyoD(−/−) grafts in areas that, in normal grafts at this time, are associated with myotube formation. The delay in fusion of MyoD(−/−) myoblasts shown here and in other studies (Megeney et al. 1996; Sabourin et al. 1999; Yablonka–Reuveni et al. 1999a) appears to be the result of sustained proliferation of myoblasts. Such sustained proliferation would also account for the observations made here and by others (Megeney et al. 1996) that myoblasts accumulate in regenerating MyoD(−/−) muscle.
Tissue culture studies in our laboratory confirm an extended phase of myoblast proliferation and delayed myotube formation in primary cultures of MyoD(−/−) muscle (data not shown). This agrees with the detailed tissue culture studies from other laboratories showing that MyoD(−/−) myoblasts continue to proliferate even after the addition of fusion media, and it is suggested that the inefficient withdrawal from the cell cycle of MyoD(−/−) myoblasts is the basis of the delay in differentiation and fusion (Sabourin et al. 1999; Yablonka-Reuveni et al. 1999b).
Although it might be considered that enhanced myoblast proliferation would result in “superior” new muscle formation, this does not generally seem to be the case. It was not seen in the present study nor in two other studies of muscle regeneration. The local injection of hepatocyte growth factor (HGF) after freeze injury resulted in increased numbers of myoblasts, but overall myotube formation and regeneration were inhibited in a time- and dose-dependent manner (Miller et al. 2000). Similarly, after contusion injury in rats, pulsed ultrasound promoted satellite cell proliferation in the damaged muscle but this had no effect on the overall regeneration of the treated muscle (Rantanen et al. 1999). The observations that enhanced myoblast proliferation does not necessarily result in improved regeneration have important implications for the design of potential strategies to enhance regeneration in clinical situations.
Other Factors that May Contribute to the Delayed Myoblast Fusion
Differences in the ability of myoblasts to proliferate, differentiate, and fuse in vivo exist among strains (Roberts et al. 1997) and among muscles (Pavlath et al. 1998), and appear to correlate with regenerative capacity. These differences may be due to intrinsic factors in myogenic cells and/or to differences in the host environment related to the efficiency of the inflammatory response, availability and response to growth factors, and composition of the ECM. The list of growth factors that can influence the myogenic program is extensive and includes the fibroblast growth factor (FGF) family, the platelet-derived growth factor (PDGF) family, hepatocyte growth factor (HGF), the transforming growth factor-β (TGF-β) family, leukemia inhibitory factor (LIF), and insulin-like growth factors (IGFs) (Bischoff 1986; Grounds and Yablonka-Reuveni 1993; Kurek et al. 1998; Grounds 1999; Grounds and McGeachie 1999). The balance between growth factors and ECM molecules can dramatically influence myoblast proliferation, differentiation, and fusion. The ECM plays an integral role in deciding cell fate, and myoblasts show a greater capacity for fusion on complex substrates such as Matrigel compared with fusion on individual ECM components such as fibronectin, collagen, and laminin (Grounds and McGeachie 1989; Lyles et al. 1992; Maley et al. 1995; Melo et al. 1996; Grounds 1999). Because SJL/J myoblasts show a superior ability to form myotubes compared to BALB/c and other strains (Maley et al. 1994, 1995), we used conditioned medium from SJL/J fusion cultures to examine the possibility that the delay in fusion of MyoD(−/−) myoblasts is due to a lack of some soluble “activating factors.” The addition of SJL/J-conditioned medium to MyoD(−/−) fusion cultures did not significantly affect myotube formation (data not shown). This indicates either that the delay in MyoD(−/−) myoblast fusion is not the result of a limiting soluble factor or that MyoD(−/−) myoblasts are unable to react to the presence of such fusion factors. These results agree with the mixed culture experiments of Sabourin et al. (1999). Furthermore, the same delay in myotube formation was seen in MyoD(−/−) EDL allografts implanted into either BALB/c or SJL/J hosts, supporting the idea that this delay is intrinsic to the MyoD(−/−) cells and is not influenced by diffusible or systemic factors associated with the host environment.
There are other instances in which myoblasts can fuse in the absence of MyoD. Fusion is well documented in L6, L8, and BC3H1 myogenic cell lines, which express little or no MyoD (Braun et al. 1989). Most notably, during early myotome development two sub-lineages of myogenic precursors exist, one expressing MyoD and the other Myf5 (Cossu et al. 1996). Some express Myf5 but not MyoD while they begin to express myogenin and differentiate (Smith et al. 1994), and some differentiate in the absence of either MyoD or myogenin (Venters et al. 1999). In satellite cells from single adult myofibers in culture, a Myf5+/myogenin+/ MyoD−- state is not observed by RT-PCR (Cornelison and Wold 1997), and it appears that satellite cells can enter the myogenic program via two different pathways by expressing either Myf-5 or MyoD (Cooper et al. 1999). Although myogenin expression is reduced in regenerating MyoD(−/−) skeletal muscle compared to wild-type skeletal muscle (Sabourin et al. 1999), this may relate more to the delayed fusion (and hence delayed myogenin expression) (Yablonka–Reuveni et al. 1999b) and the relative timing of the observations. Although expression of a range of genes relating to cell cycle arrest and differentiation was examined in MyoD(−/−) cells in vitro (Sabourin et al. 1999), and the pathway may involve genes such as p21 and p57 (Zhang et al. 1999), the precise mechanism for the delay in differentiation in MyoD(−/−) cells has yet to be determined.
In normal muscle, MyoD+ cells rapidly exit the cell cycle and differentiate (Halevy et al. 1995), whereas it has been suggested that Myf5+ cells may represent a “stem cell” population in muscle (Yablonka–Reuveni et al. 1999b). It has been proposed that more of these stem cells are present in MyoD(−/−) muscle and that there is a greater propensity for stem cell self-renewal (i.e., formation of satellite cells) rather than progression through myogenesis (Megeney et al. 1996; Sabourin et al. 1999). Our studies showed that myogenesis is not impaired in whole muscle grafts in MyoD(−/−) mice, and the appearance of regenerated muscle (by light microscopy) in this model was similar to controls after Day 8. Although examination of mature grafts (sampled at 21 days) by EM revealed evidence of increased numbers of unfused mononuclear cells, presumed to be myoblasts, the persistence of unfused myoblasts in MyoD(−/−) muscles might simply reflect a problem with the kinetics of myotube formation combined with the excessive numbers of myoblasts present (due to the sustained proliferation), and these “surplus” myoblasts might have no option other than to remain trapped beneath the basement membrane in a satellite cell position. Whether this population of unfused MyoD(−/−) myoblasts has any unusual or stem cell properties requires further investigation.
The Appearance of Desmin-positive Myoblasts Is Altered in MyoD(−/−) Grafts
In MyoD(−/−) grafts, the appearance of desmin-positive myoblasts differed strikingly from BALB/c controls. The desmin staining of MyoD(−/−) myoblasts in vivo was weak and generally ill-defined within the cytoplasm, suggesting that MyoD may play some role in the production or organization of desmin. Decreased or variable desmin immunostaining has been reported previously for MyoD myoblasts in primary cultures (Megeney et al. 1996; Yablonka–Reuveni et al. 1999b) and on cultured myofibers (Yablonka–Reuveni et al. 1999b). Although MyoD(−/−) myoblasts are able to manufacture desmin, the desmin may not be appropriately organized. Tissue culture studies in our lab (data not shown) found that MyoD(−/−) myoblasts did not adhere readily to Matrigel and formed clusters corresponding to clumping of MyoD(−/−) myoblasts observed on isolated myofibers in culture (Yablonka–Reuveni et al. 1999b). A similar clustering of myoblasts on Matrigel is seen with desmin(−/−) myoblasts (Vincent Mouly, personal communication). These observations indicate either a direct or an indirect role for desmin in the delayed fusion process of MyoD(−/−) myoblasts and also suggest a role for interactions with ECM moelcules. Although an altered morphology of MyoD(−/−) myoblasts in vitro has also been reported (Megeney et al. 1996), no such difference was seen by us or by Yablonka–Reuveni (personal communication).
In desmin(−/−) mice, distinct morphological abnormalities, such as non-aligned fibers with sparse filaments as well as abnormal sarcomeric structure, have been observed in embryonic (Capetanaki et al. 1997) and adult skeletal muscle (Lescaudron et al. 1993, 1997; Li et al. 1993, 1994, 1997). Despite this, overall muscle formation and regeneration in embryonic and adult desmin(−/−) mice is reported to be normal (Capetanaki et al. 1997). However, the timing and efficiency of myoblast differentiation and fusion and the expression of differentiation-specific genes (such as tropomyosin and myosin) and the MRFs have not yet been studied. In vitro experiments have demonstrated that myogenesis and expression of the MRFs is inhibited in desmin(−/−) embryonic stem cells (Weitzer et al. 1995) and in transgenic C2C12 cells in which the production of desmin is inhibited by the constitutive production of antisense mRNA targeted to desmin (Li et al. 1994). The correlation between the absence of MyoD and the poor expression of desmin in myoblasts suggests that desmin may play an important role in fusion.
In conclusion, these data demonstrate that myoblasts deficient in MyoD in regenerating adult muscle have sustained proliferation leading to delayed fusion, but that this problem is overcome with time and normal myotubes eventuate. Once myoblasts initiate the differentiation cascade, the rate of myotube formation appears similar to that of wild-type cells. The alternative mechanisms independent of MyoD expression that facilitate fusion remain to be elucidated. These observations suggest that sustained proliferation of myoblasts increases the pool of myoblasts that settle into the satellite cell position. The significance of the observation of unfused “leftover” mononuclear cells and whether these MyoD(−/−) satellite cells might have stem cell properties remain to be clarified.
Footnotes
Acknowledgements
Supported by a grant from the National Health and Medical Research Council, Canberra, Australia.
We would like to acknowledge the considerable technical contribution of Mr Jim Hopley in the processing and sectioning of tissues for autoradiography, immunohistochemistry, and histology. We also thank Mr Mike Archer for all of the electron microscopy and analysis.
