Abstract
Uropathogenic Escherichia coli invade the urothelial umbrella cells by using the zipper mechanism. However, the details of the early events of this invasion, such as the formation of the phagocytic cup, are not yet well understood. We show here, using thin section electron microscopy and immunogold labeling, that the plasma membrane curves around the bacterial surface in the phagocytic cup. There exists a uniform gap between the bacterium and the urothelial membrane, and actin filaments are present in the phagocytic cup. We suggest that the action–reaction between the mechanical forces generated by pilus retraction of the bacterium and the actin polymerization in the urothelial cell plays a role in maintaining the phagocytic cup. This manuscript contains online supplemental material at http://www.jhc.org. Please visit this article online to view these materials.
Keywords
U
The details of the invasion process of UPEC, especially of the early stages, however, are far from well understood (Pizarro-Cerda and Cossart 2006). First, it has been difficult to visualize the invasion using transmission electron microscopy (EM) in the mouse model (Fukushi et al. 1979), because the UPEC invasion itself is a relatively rare event (Mysorekar and Hultgren 2006). Second, cultured bladder cancer cells have been the only in vitro system used to study the UPEC invasion (Martinez et al. 2000; Martinez and Hultgren 2002; Duncan et al. 2004; Eto et al. 2007), but these cells do not closely mimic the true urothelium, i.e., they do not form the rigid urothelial plaques—a key feature of the urothelial apical surface (Sun 2006). Third, the urothelial umbrella cells have an unusual cytoskeleton structure very different from other epithelial cells. It was found that the actin filaments are completely absent from the regions underneath the apical membrane of the umbrella cells (Romih et al. 1999; Veranic and Jezernik 2002; Acharya et al. 2004). In addition, there exists a chicken wire network of intermediate filaments below the apical surface (Veranic and Jezernik 2002), and the apical urothelial plaques are connected to the intermediate filaments through small bridges of unknown protein compositions (Staehelin et al. 1972). This raises the question of whether the actin filaments play the key role, as in the case of the zipper mechanism in general (Finlay and Cossart 1997; Rottner et al. 2005), in the formation of the phagocytic cup induced by UPEC.
We describe an EM study of the phagocytic cup induced by UPEC in the mouse model. We show that the attachment of UPEC can overcome the rigidity of the urothelial plaques to form a smooth phagocytic cup that curves around the bacteria but maintains a uniform distance from the bacterial body. In addition, actin filaments are indeed present in these cups, likely recruited to the cup by the mechanical stress induced by the UPEC attachment.
Materials and Methods
Bacterial Strains and Antibodies
UPEC strain J96 was purchased from ATCC (Rockville, MD; catalog number 700336) and grown at 37C in Luria-Bertani broth shaking at 270 rpm overnight, followed by another 48 hr of static growth to induce the growth of the type 1 pili. The expression of type 1 pili was verified by mannose-sensitive agglutination using 1% baker's yeast in PBS. The bacteria were harvested by centrifugation at 1200 × g for 20 min at room temperature and resuspended in PBS for inoculation.
The primary antibodies used for antigen detection included mouse monoclonal anti-UPIII antibody AU1 (Liang et al. 2001), rabbit polyclonal anti-E. coli antibody (Biodesign International; Saco, ME), and mouse monoclonal anti-chicken gizzard actin antibody C4 (Lessard 1988) (Cedarlane Laboratories; Burlington, NC). Gold particle–conjugated secondary antibodies used to detect the primary antibodies included the 12-nm gold particle–conjugated goat anti-mouse IgG and 18-nm gold particle–conjugated goat anti-rabbit IgG (Jackson ImmunoResearch laboratories; West Grove, PA).
Inoculation of Bacteria
Female C3H/HeN mice 7–18 weeks old (Harlan; Indianapolis, IN) were anesthetized by intraperitoneal injection (0.15 mg/15 g body weight) of 20 mg/ml ketamine and 3 mg/ml xylazine in 0.9% NaCl and inoculated by transurethral catheterization with a 50-μl suspension of bacteria at a concentration of ∼2 × 109 cfu/ml. The mice were killed 24 hr later (McTaggart et al. 1990), and their urinary bladders were removed for further processing (see below). A total of eight mouse bladders were used for our experiments. All experiments on animals received prior approval from the institutional animal care and use committee of New York University School of Medicine and conformed to the standards set by the appropriate regulatory agencies.
Transmission EM
For immunoelectron microscopy, mouse bladders and bacteria pellets were fixed for 4 hr at 4C in a freshly prepared solution containing 3% paraformaldehyde, 0.1% glutaraldehyde, 4% sucrose, and 0.1 M sodium cacodylate buffer (pH 7.4). Dehydration of the samples in graded methanol was carried out as follows: 30% methanol for 15 min at 0C, 50% methanol for 15 min at −20C, 70% methanol for 15 min at −35C, 80% methanol for 15 min at −35C, 95% methanol for 15 min at −35C, and three changes of 100% methanol for 15 min each at −35C. Infiltration of Lowicryl K4M resin (Polysciences; Warrington, PA) was carried out at −35C in the mixtures of methanol and K4M resin at the ratio of 1:1 for 1 hr, 1:2 for 1 hr, 1:3 for 1 hr, pure resin for 1 hr, and pure resin for overnight. Samples were finally embedded in BEEM capsules (Electron Microscopy Sciences; Hatfield, PA) filled with K4M resin and polymerized under UV light (360 nm) for 48 hr at −35C, followed by another 12 hr at room temperature.
For immunogold labeling, thin sections of 50–70 nm on formvar-coated nickel EM grids were first floated on a drop of PBS (pH 7.4) containing 1% BSA, 0.05% Triton X-100, and 0.05% Tween 20 for 5 min; the grids were then transferred to a drop of a dilution buffer (PBS with 1% BSA and 0.05% Tween 20) that contained primary antibodies and were incubated for 3 hr. Afterward, the grids were washed with PBS and transferred to a drop of gold particle–conjugated secondary antibodies in the dilution buffer for 1 hr. The working dilution ratios of the antibodies were 1:1 for AU1, 1:25 for anti-actin antibody, 1:300 for anti-E. coli antibody, and 1:30 for the secondary antibodies. Grids were stained with uranyl acetate and lead citrate, airdried, and imaged using a Philips CM12 electron microscope (FEI; Eindhoven, The Netherlands). A total of 300 micrographs were taken, and only selected images are presented here.
For routine EM, mouse bladders and bacteria pellets were fixed at room temperature for 2 hr with 2.5% glutaraldehyde in 0.1 M sodium cacodylate buffer (pH 7.4) and postfixed with 1% aqueous osmium tetroxide for 2 hr. After dehydration sequentially in graded ethanol, the samples were embedded in Eponate 12 (Ted Pella; Redding, CA).
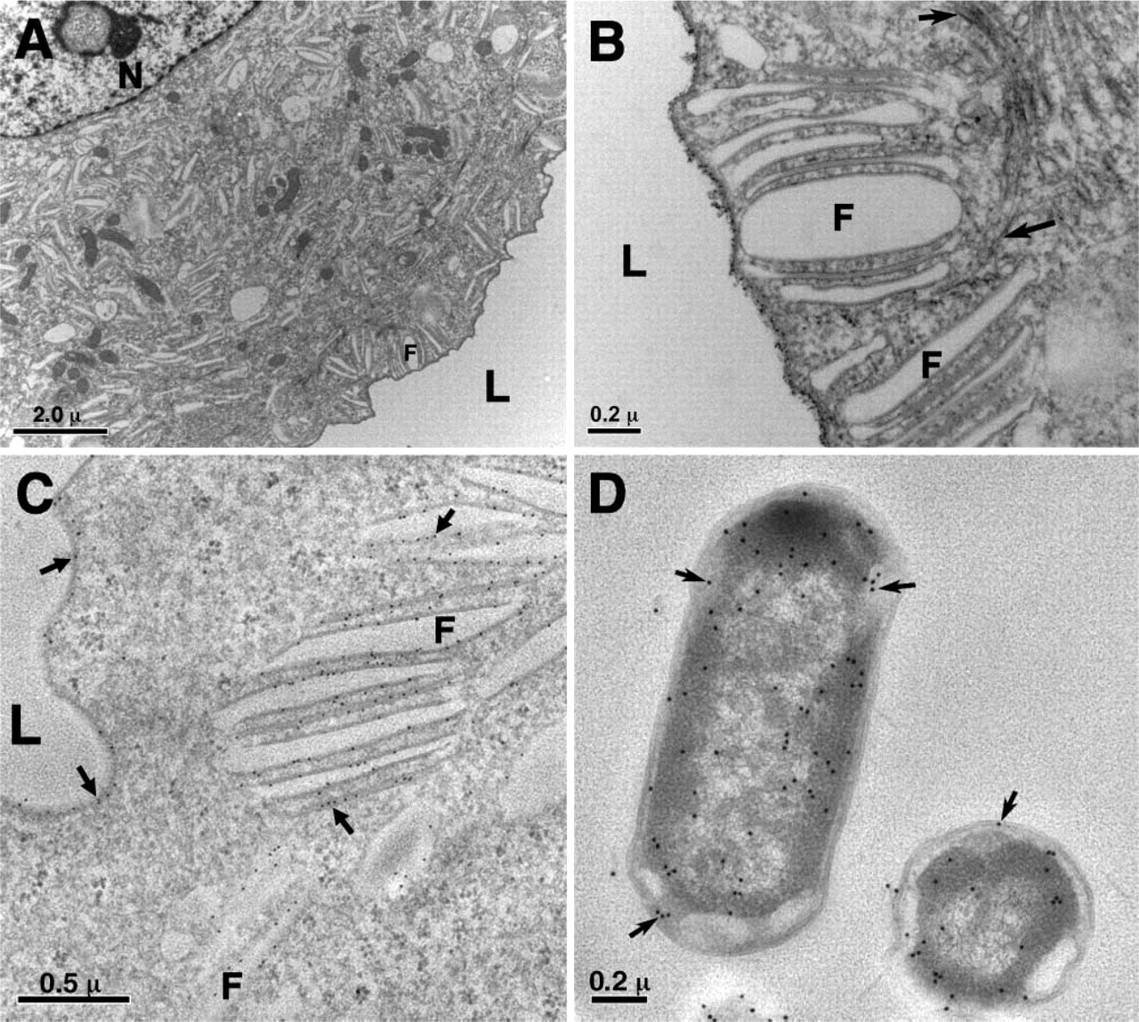
Unique morphologies of urothelial umbrella cells and immunogold labeling. (
Results
Morphologies of UPEC-induced Phagocytic Cups
We carried out experimental UTIs using the well-established mouse model of UPEC invasion (McTaggart et al. 1990; Mulvey et al. 1998). Mouse urothelium not only expresses uroplakins that are almost identical to the human proteins (Wu et al. 1994), but also, like the human urothelium, its UP Ia harbors the high mannose moiety that can serve as the receptor of UPEC adhesin FimH (Zhou et al. 2001; Xie et al. 2006). Hence the mouse urothelium provides a unique opportunity for studying UTIs often not available for other pathogen–host systems (Lecuit and Cossart 2002; Cossart and Sansonetti 2004). As a control, we first examined mouse urothelial surface by standard fixation for EM (Figures 1A and 1B). Our protocol preserved the morphologies of the urothelial surface—the thin-section EM showed that the urothelial apical surface was covered with the concaved urothelial plaques and that there were numerous fusiform vesicles of the same AUM structure in the cytoplasm. We used monoclonal anti-UP III antibody AU1 to immunogold label the urothelial plaques and the fusiform vesicles (Figure 1C). AU1 is highly specific in labeling AUM (Liang et al. 2001), allowing us to monitor the membrane during any morphology changes. For bacteria, we used the uropathogenic type 1–piliated E. coli J96, a strain isolated from a pyelonephritis patient and commonly used in UPEC studies (Hull et al. 1981; Svanborg Eden et al. 1983; Hung et al. 2002; Duncan et al. 2004). Under culture conditions optimized for pili growth, the bacteria harbored many pili of varying lengths (Supplemental Figure SF1; see also Hahn et al. 2002). We were also able to find a commercially available anti-E. coli antibody that could immunogold label these bacteria (Figure 1D).
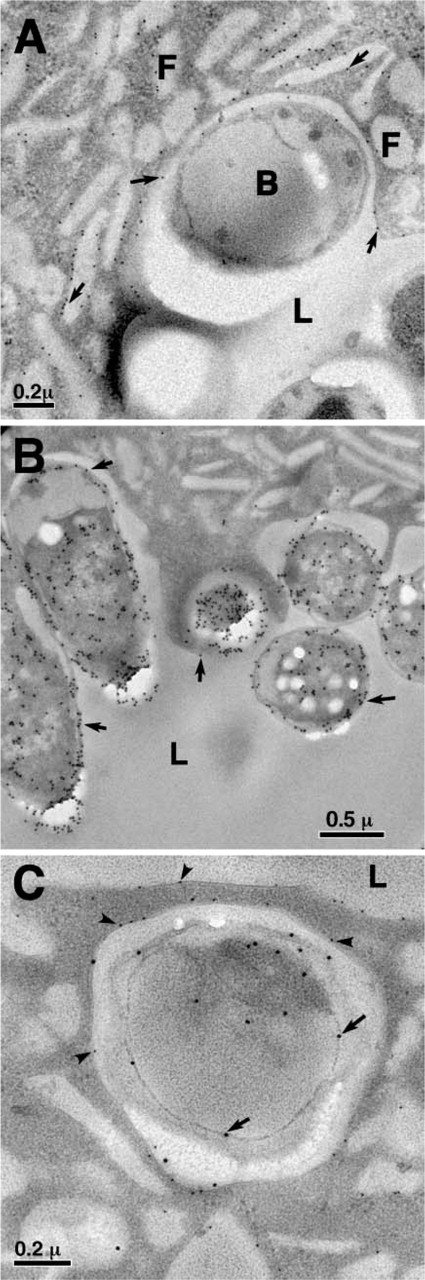
Morphologies of the uropathogenic E. coli (UPEC)-induced phagocytic cups. (
Many micrographs of the phagocytic cups from thin-section EM were collected, and Figure 2 shows several typical images. The first striking feature of these phagocytic cups was that the apical plasma membrane of the umbrella cell lost the scallop-shaped form of the individual urothelial plaques. Instead, the plasma membrane now followed the shape of the attached bacteria, curving around the bacteria. Immunogold labeling using AU1 antibody showed that the apical membrane of the urothelial cell under the bacteria was still composed of uroplakins (Figures 2A and 2C). In addition, each bacterium had its own phagocytic cup even when there was a cluster of bacteria localized at the same surface area (Figures 2B and 3B).
The second striking feature of the UPEC-induced phagocytic cups was that there was always an almost uniform gap between the urothelial cell plasma membrane and the bacterial outer membrane (Figures 2 and 3). The width of this gap, estimated from the analysis of >200 phagocytic cups, is ∼50–150 nm. Re-examination of the thin-section images in early literature on experimental UTI showed this gap to be a common feature of the urothelial phagocytic cup (Fukushi et al. 1979; McTaggart et al. 1990). This gap was unlikely an artifact of the dehydration-fixation process, because it was also present in images taken by the quick-freeze/deep-etch method (Mulvey et al. 1998), which did not involve dehydration-fixation in sample preparation (Heuser 1989).
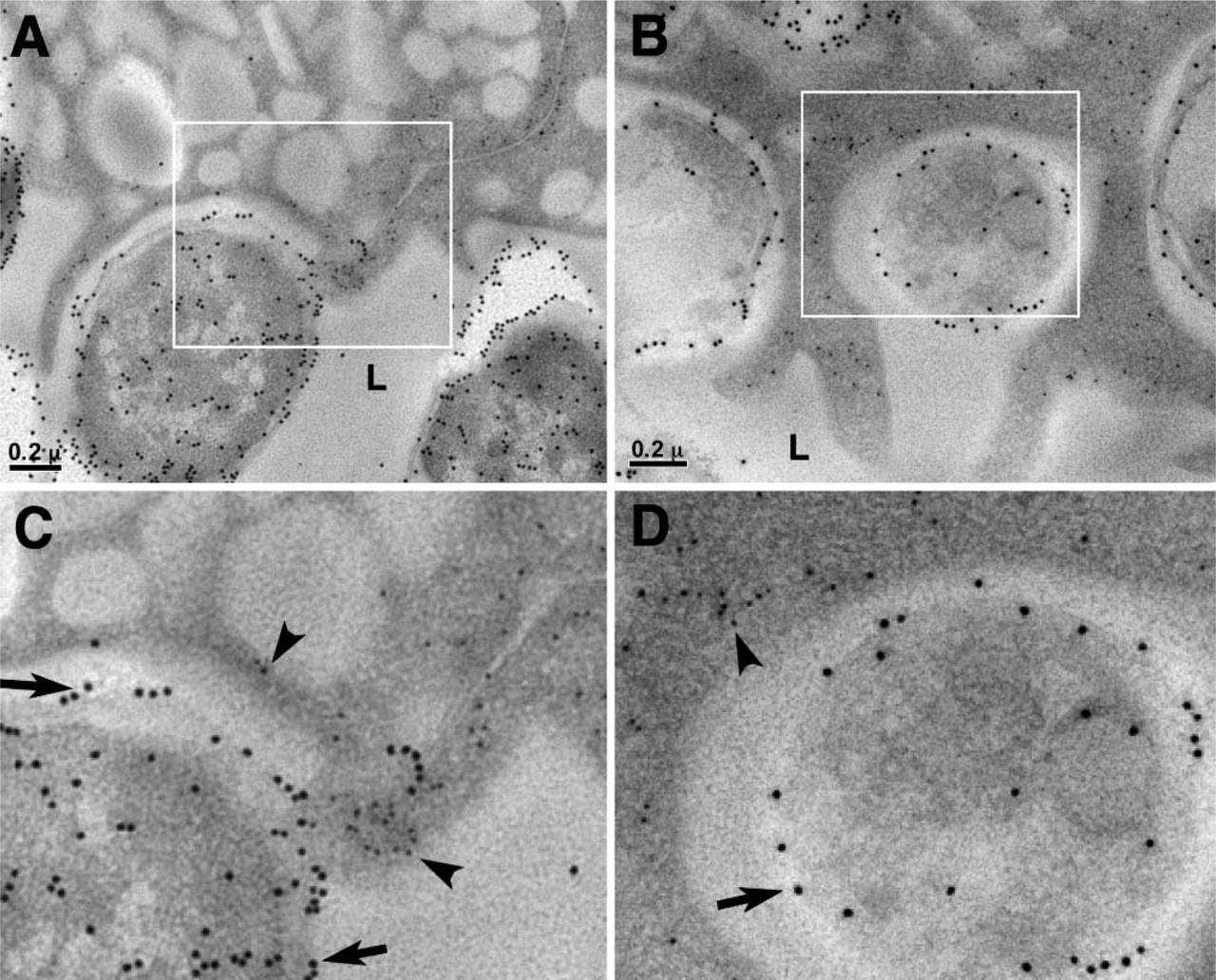
Actin filaments in the phagocytic cups. (
Actin Filaments in the Phagocytic Cups
We further addressed the question of whether actin filaments were present in the phagocytic cups. We again used the method of immunogold labeling for detection. After trying various commercially available antibodies, we found that the monoclonal mouse anti-chicken gizzard actin IgG1 antibody C4 (Lessard 1988), recently used successfully in labeling actin filaments in intestine (Schwarz and Humbel 2007), provided good labeling for actin filaments in mouse urothelia (Figures 3 and 4). Using this antibody, we found in the control mouse umbrella cells actin filaments only at the cell junction regions and in the basal regions where the cell membrane interdigitates with that of the cells underneath the umbrella cells (Figure 4), but not anywhere near the regular apical membrane. This was consistent with previous assessments that there were no actin filaments at the apical surface under the cell plasma membrane (Romih et al. 1999; Veranic and Jezernik 2002; Acharya et al. 2004).
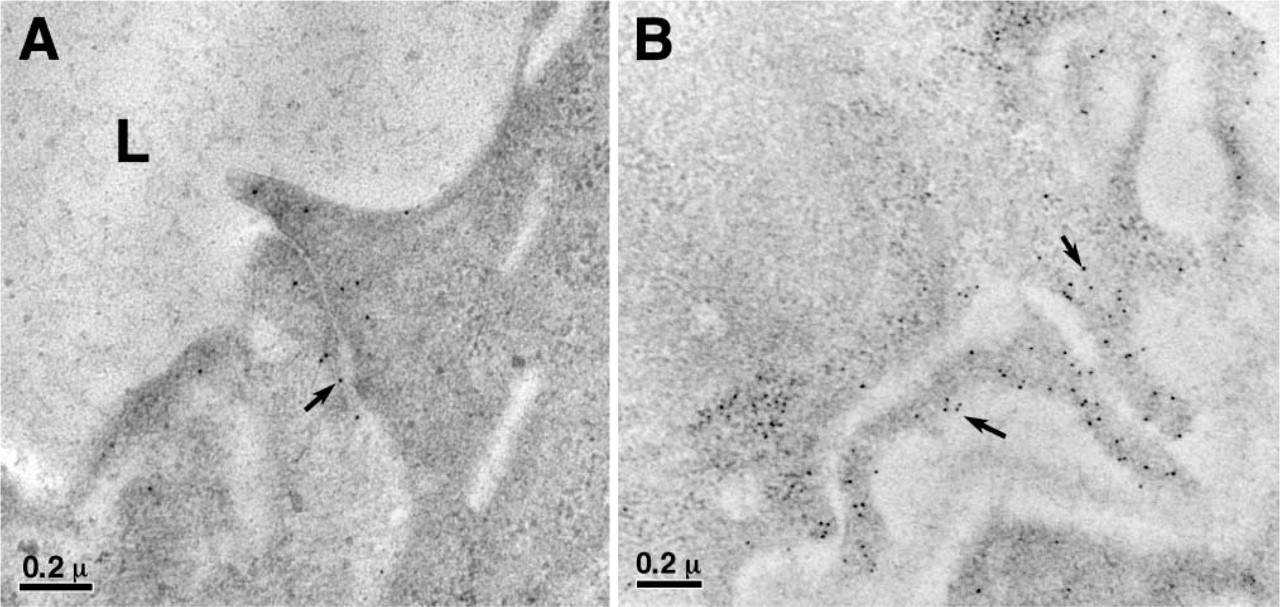
Immunogold labeling of actin filaments in the control mouse bladder urothelium. Actin filaments were immunogold labeled (arrows) with anti-actin antibody C4 and were found only at the regions of the tight junctions of the umbrella cells (
Unlike the regular apical urothelial surface, we found that actin filaments were present in all the phagocytic cups induced by J96 (Figure 3). These filaments were all localized to the regions in the phagocytic cups with dark contrast in the EM images, presumably densely packed with actin filaments. We therefore can conclude that the actin filaments are indeed involved in the urothelial cytoskeleton rearrangement occurring on the attachment of the UPEC. In cultured cell systems, it was found that phosphoinositide 3 kinase, focal adhesin kinase, α-actinin, and vinculin are involved in the signaling pathway of the UPEC invasion (Martinez et al. 2000). We have not yet been able to find suitable antibodies to immunogold label these proteins in the mouse model. It may require other methodologies to confirm these molecules' participation in UPEC-induced cytoskeleton rearrangement in urothelium.
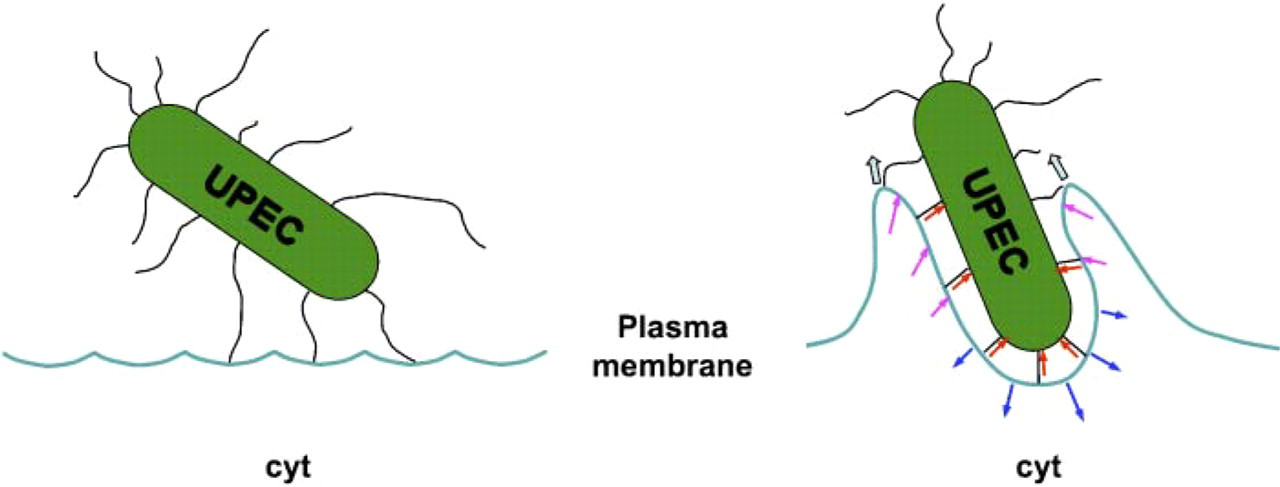
The forces at the UPEC-induced phagocytic cup. At the bottom of the cup, the force generated by the pilus retraction (red arrows) is balanced by the retractile force generated by the actin polymerization (blue arrows) to maintain the gap between the bacterial body and the urothelial cell plasma membrane. While at the leading edge of the cup, the force generated by the pilus retraction will have the same direction as the force generated by the actin polymerization (magenta arrows), leading to the rise and closure of the phagocytic cup (wide arrows). Cyt, cytoplasm.
Discussion
We present here a careful examination of UPEC-induced phagocytic cups in the mouse UTI model by thin-section EM. Our data showed several striking features of these cups. One of these features is that the apical plasma membrane of the umbrella cell in the phagocytic cup lost the scallop-shaped form of the individual urothelial plaques, and the urothelial plasma membrane instead followed the shape of the attached bacterium (Figures 2 and 3). This suggests that there likely exists a mechanical force that can overcome the rigidity of the urothelial plaques during the formation of the phagocytic cups (see discussion below). Interestingly, each bacterium was found to have its own phagocytic cup (Figures 2 and 3). The corollary of this is that each bacterium will be internalized into a membrane-enclosed vacuole of its own. We did not find flask-shaped caveolae in any of the phagocytic cups, contrary to that in the case of the UPEC invasion model using cultured cells (Duncan et al. 2004), further showing that the urothelium is different from the cultured cells.
Although the gap between the bacteria and urothelial apical cell membrane is electron lucent (Figures 2 and 3), it is likely maintained by the bacterial pili (not visible under the current method of sample preparation; Figure 1D). Because the type 1–piliated bacteria harbor pili of uneven lengths up to 2000 nm in culture (Supplementary Figure SF1), one can assume that they also have pili in different lengths before attaching to the urothelial membrane. These pili are quite rigid; they have to adjust their lengths during the formation of the phagocytic cups to maintain a relatively uniform gap between the bacteria and the host cell membrane. Interestingly, a gap is also observed between the bacteria and the urothelial cellular content after the bacterial internalization (Bishop et al. 2007) and development into the so-called intracellular bacterial communities (Fukushi et al. 1979; Anderson et al. 2004). Hence, the gap between the bacteria and the urothelial cell content may be important for the bacterial invasion of and survival in the urothelial cells.
The presence of actin filaments in both tight junctions and the UPEC-induced phagocytic cups (Figures 3 and 4) suggests some similarities between junctional formation and phagocytosis (Cossart et al. 2003). The tight junctions of the umbrella cells must endure chronic mechanical stresses during the distension and contraction of the bladder in the micturition cycles. The actin filaments in the tight junction regions may be induced by these mechanical stresses, and the actin filaments play a role in strengthening the junction (Fanning 2001). Mechanical forces can also play a role in forming the phagocytic cups. The attachment of UPEC to the urothelial UP Ia receptors by the type 1 pili can create an acute local mechanical stress at the binding site. This mechanical stress may be further enhanced by the pilus retraction (Mulvey et al. 1998; Merz et al. 2000; Maier et al. 2004). The UP Ia molecules then serve as mechanoreceptors to transmit the mechanical signal into a cellular chemical signal, leading to the formation of actin filaments and the phagocytic cup at the bacterial binding site (Bershadsky et al. 2003; Ingber 2003; Yoshigi et al. 2005). Interestingly, it has been shown that the directions of mechanical forces generated by actin polymerization depend on the local curvature of the surface (Upadhyaya et al. 2003). The actin filaments would exert a retractile force at the bottom of the phagocytic cup but a propulsive one at the edge of the cup (Figure 5). At the bottom of the phagocytic cup, on the one hand, the force generated by the pilus retraction is balanced by the retractile force generated by the actin polymerization to maintain the gap between the bacterial body and the urothelial cell plasma membrane. On the other hand, at the leading edge of the cup, these two forces have the same direction to cause the rise and closure of the phagocytic cup, leading to the internalization of the bacterium.
Footnotes
Acknowledgements
This work was supported by National Institutes of Health Grant DK52206.
We thank Drs. Wenbiao Gan, Hongying Huang, Lan Mo, Richard P. Novick, Liyu Tu, Huitang Zhang, and Weiyue Zheng for useful discussions, Dr. Heinz Schwarz for suggestion of antibody C4 for immunogold labeling of actin filaments, and Valicia Burke for critically reading the manuscript.
