Abstract
Lectin-like oxidized low-density lipoprotein receptor-1 (LOX-1) is one of the scavenger receptors that recognizes oxidized low-density lipoprotein as a major ligand. The placenta is a major source of prooxidant during pregnancy, and the level of placental oxidative stress increases rapidly at the end of the first trimester and tapers off later in gestation. In our study, we evaluated placental expression of LOX-1 during different gestational stages in mice and humans. We used immunohistochemistry and ISH to identify LOX-1–expressing cells in murine and human placentas. In both species, higher expression of LOX-1 mRNA during early to midgestational stages compared with late gestation—corresponding to the increased oxidative stress in early pregnancy—was shown by real-time RT-PCR. In murine placenta, we showed that LOX-1–expressing cells were fibroblast-like stromal cells in metrial glands and decidua basalis and that they were glycogen trophoblast cells in the junctional and labyrinth zones. In the human, LOX-1 expression was detected in villous cytotrophoblasts in both first trimester and term placentas. These localization patterns of LOX-1 in murine and human placentas suggest the possible involvement of LOX-1 in high oxidative stress conditions of pregnancy.
P
Lectin-like ox-LDL receptor-1 (LOX-1) is one of the scavenger receptors that is mainly expressed in vascular endothelial cells (Sawamura et al. 1997). It was initially cloned from bovine aortic endothelial cells and from human lung as a novel receptor for ox-LDL (Sawamura et al. 1997). LOX-1 shows strong activity for binding, internalizing, and degrading ox-LDL (Moriwaki et al. 1998b). It is induced by stimuli including angiotensin II (Li et al. 1999), tumor necrosis factor-α (Kume et al. 1998), and advanced glycation end products (Chen et al. 2001), as well as ox-LDL itself (Aoyama et al. 1999). LOX-1 is involved in ox-LDL–induced apoptosis of vascular endothelial cells through intracellular production of reactive oxygen species (Li and Mehta 2000).
LOX-1 is expressed not only in vascular endothelial cells but also in monocyte-derived macrophages, vascular smooth muscle cells, and chondrocytes (Moriwaki et al. 1998a; Yoshida et al. 1998; Draude et al. 1999; Aoyama et al. 2000; Nakagawa et al. 2002). Northern blot analysis of human tissues detected LOX-1 mRNA in vascular-rich organs such as placenta, lung, brain, and liver (Sawamura et al. 1997). Among these tissues, the placenta had the highest expression of LOX-1 mRNA (Sawamura et al. 1997), which indicated a crucial role of LOX-1 in placental function. It is suggested that LOX-1 might be involved in trophoblast invasion in early pregnancy (Pavan et al. 2004; Fournier et al. 2007) and accelerated trophoblast apoptosis in preeclampsia (Lee et al. 2005). Immunohistochemical studies of human placenta showed that LOX-1 localized in extravillous trophoblasts of first trimester placentas (Pavan et al. 2004; Fournier et al. 2007) and in syncytiotrophoblasts of normal and preeclamptic term placentas (Lee et al. 2005). However, localization of LOX-1 in animals, especially the mouse—the best-studied mammalian experimental model system—has not been reported. In this study, we describe the clear localization of LOX-1 in murine placenta, as determined by using immunohistochemistry and ISH, and we compared these results with data for human placentas.
Materials and Methods
Sample Collection
C57BL/6 mice were purchased from Japan Crea (Tokyo, Japan). Mice were bred in the Animal Resource Facility at Kumamoto University under specific pathogen-free conditions. All animal procedures were approved by the Animal Research Committee at Kumamoto University, and all procedures conformed to Regulations for Animal Experiments of Kumamoto University. Adult female mice (2–5 months old) were mated with males in the evening and were monitored, starting the next morning, for the appearance of a vaginal plug. Noon on the day in which a vaginal plug was found was said to be 0.5 embryonic day (E0.5). Placentas were isolated at various developmental stages between days E10.5 and E18.5. For better tissue orientation, endometrial and myometrial components were not dissected away from placental disks during dissection. For study of the adult tissue distribution of LOX-1 mRNA, tissues were obtained from 8-week-old male mice.
Human placental tissues at 6–41 weeks of gestation were obtained from healthy pregnant women. Before collecting any such samples, we obtained signed informed consent, with consent documents written according to the Declaration of Helsinki, from all pregnant women in this study. First trimester placentas (6–12 weeks of gestation) and term placentas (37–41 weeks of gestation) were obtained from legal elective abortions and spontaneous vaginal deliveries, respectively.
RT-PCR
RNA isolation and RT were performed as previously described (Kobayashi et al. 2007). A 1-mg sample of total RNA was used to produce cDNA with an Omniscript RT Kit (Qiagen; Valencia, CA). The sequences of PCR primers used were as follows—mouse LOX-1 (522-bp product): forward primer, 5′-GAGCTGCAAACTTTTCAGG-3′, reverse primer, 5′-GTCTTTCATGCAGCAACAG-3′; human LOX-1 (193-bp product): forward primer, 5′-TTACTCTCCATGGTGGTGCC-3′, reverse primer:,5′-AGCTTCTTCTGTTGTTGCC-3′; glyceraldehyde-3-phosphate dehydrogenase (GAPDH; 392-bp product): forward primer, 5′-GGAAAGCTGTGGCGTTGGCGTGAT-3′, reverse primer, 5′-CTGTTGCTGTAGCCGTATTC-3′. These PCR primers were custom-made by Invitrogen (Carlsbad, CA). The primers for GAPDH were designed to be available for both mice and humans.
The PCR cycle parameters were as follows: 15 min at 95C, three-step cycling (denaturation at 94C for 30 sec; annealing at 55–60C for 30 sec; and extension at 72C for 1 min), and 10 min at 72C using an iCycler Thermal Cycler (Bio-Rad Laboratories; Hercules, CA). Thirty cycles were used for LOX-1; 28 cycles were used for GAPDH. Annealing temperatures for LOX-1 and GAPDH were 55C and 60C, respectively. A 10-ml aliquot of each PCR product was subjected to electrophoresis on a 2% (w/v) agarose gel containing 0.1 mg/ml ethidium bromide.
Real-time PCR Analysis
To quantify LOX-1 mRNA levels in murine and human placental tissues, real-time PCR was performed with an ABI PRISM 7700 Sequence Detection System (Applied Biosystems; Foster City, CA) as described earlier (Tsujita et al. 2007). A standard curve was constructed by plotting the relative amounts of a serial dilution of placental cDNA. Mouse placentas were taken from at least three animals at each gestational age of E10.5, E11.5, E13.5, E14.5, E15.5, E16.5, E17.5, and E18.5. For human samples, first trimester placentas (6–12 weeks of gestation, n=8) and term placentas (37–41 weeks of gestation, n=5) were compared. For measurements, villi were separated from decidua and other components of first trimester placenta using stereoscopic microscopy. Chorionic villi of central cotyledons were collected from term placentas. All experiments were performed in duplicate. Thermal cycler conditions consisted of 2 min at 50C (with uracil N-glycosylase for prevention of carryover contamination) and 10 min at 95C (for hot start PCR), followed by 40 cycles of 15 sec at 95C and 1 min at 60C. Quantitative values of expression of target genes were determined as relative to endogenous 18S rRNA gene expression.
ISH
cRNA probes for mouse and human LOX-1 were synthesized from PCR-generated cDNA as described previously (Divjak et al. 2002) with minor modifications. PCR amplification of template PCR-generated cDNA (2.5 ml) was performed using PrimeSTAR HS DNA Polymerase (Takara; Kyoto, Japan) with the following primer pairs: T7 mouse LOX-1 primer (544-bp product), 5′-CT
Murine and human placentas were fixed in 4% paraformaldehyde solution at 4C overnight and were embedded in paraffin. Paraffin sections (3 mm) were deparaffinized in xylene and rehydrated in graded alcohols. Sections were treated with 10 mg/ml proteinase K (Roche Diagnostics) for 10 min at 37C and were postfixed in 4% paraformaldehyde, treated with 0.1 N HCl, and acetylated with 0.25% acetic anhydride in 0.1 mol/l triethanolamine (pH 8.0) for 10 min. Sections were dehydrated in graded alcohols and air dried. Samples (50 ng) of cRNA probes were mixed with a 50-fold volume of hybridization buffer containing 50% formamide, 10 mM Tris-HCl (pH 7.5), 200 mg/ml tRNA, 1× Denhardt's solution, 600 mM NaCl, 0.25% SDS, 1 mM EDTA (pH 8.0), and 10% dextran sulfate. The hybridization mixture was heat denatured, applied to dried sections, and covered with Parafilm. Hybridization was performed at 50C for 18 hr in a humidified chamber. After hybridization, Parafilm was removed by a brief washing in 5× SSC (1× SSC: 150 mM NaCl plus 15 mM sodium citrate). Sections were washed in 50% formamide plus 2× SSC for 30 min at 50C. After the sections were washed in TNE (10 mM Tris-HCl, pH 7.5, 0.5 M NaCl, and 0.5 mM EDTA) for 10 min at 37C, they were treated with 10 mg/ml RNase A (Roche Diagnostics) for 30 min at 37C. They were again washed in TNE for 10 min at 37C and were stringently washed sequentially: once in 2× SSC and twice in 0.2× SSC for 20 min at 50C. Sections were washed in buffer 1 (100 mM Tris-HCl, pH 7.5, 150 mM NaCl) for 5 min, were incubated with 1.5% blocking reagent (Roche Diagnostics) diluted in buffer 1, and were incubated for 30 min at room temperature with alkaline phosphatase–conjugated rabbit anti-DIG F(ab') fragment antibody (Roche Diagnostics) diluted 1:500 in buffer 1. After sections were washed twice in buffer 1 for 15 min and once in buffer 2 (100 mM Tris-HCl, pH 9.5, 100 mM NaCl, and 50 mM MgCl2) for 5 min, they were covered with 200 ml of buffer 2 containing nitroblue tetrazolium and 5-bromo-4-chloro-2-indolyl phosphate (Roche Diagnostics) and were placed in humidified chambers. Color development was monitored using a light microscope. Dark blue indicated a positive reaction. After a brief washing in tap water, sections were mounted in permanent aqueous medium (Nichirei; Tokyo, Japan). Counterstaining was not performed. Sections incubated with the sense probe served as negative controls.
Histology and Immunohistochemistry
Formalin-fixed paraffin sections were stained with hematoxylin and eosin. Paraffin sections 3 mm thick were used to localize mouse desmin. Endogenous peroxidase activity in murine placental sections was blocked with 0.3% H2O2 in 100% methanol for 30 min at room temperature, after which sections were incubated for 90 min at room temperature with anti-human desmin rabbit polyclonal antibody (1 mg/ml, RB-9014-P; Lab Vision, Suffolk, UK), which cross-reacts with mouse desmin. An incubation for 30 min at room temperature with goat anti-rabbit Ig-conjugated peroxidase-labeled polymer amino acid (Nichirei) followed. Peroxidase activity was visualized using 3,3′-diaminobenzidine tetrahydrochloride (Dojin Chemicals; Kumamoto, Japan) as the substrate. Meyer's hematoxylin was used as the counterstain. As a negative control, normal rabbit IgG (10 mg/ml; Santa Cruz Biotechnology, Santa Cruz, CA) was used instead of the primary antibody.
For immunohistochemical studies of LOX-1, murine and human placental tissues were fixed with freshly prepared periodate-lysine-paraformaldehyde (PLP) fixative for 6-8 hr at 4C, after which they were washed with a graded series (10–20%) of sucrose in 0.1 M PBS. Fixed tissues were embedded in tissue-embedding media, frozen in liquid nitrogen, and cut into 6-mm-thick sections. Endogenous peroxidase activity in the murine placental sections was blocked as described above, followed by incubation of the sections for 90 min at room temperature with anti-mouse LOX-1 rat monoclonal antibody (JTX-58, rat IgG; 5 mg/ml). Sections were incubated for 30 min at room temperature with goat anti-rat Ig-conjugated peroxidase-labeled polymer amino acid (Nichirei). Visualization of peroxidase activity and counterstaining were performed as described. As a negative control, normal rat IgG (10 mg/ml; Santa Cruz Biotechnology) was used instead of the primary antibody.
To stain human LOX-1, frozen sections of human placental tissues were incubated for 90 min at room temperature with biotinylated anti-human LOX-1 mouse monoclonal antibody (JTX-92, mouse IgG; 6.9 mg/ml). Sections were incubated for 30 min at room temperature with avidin-biotin complex (VECTASTAIN Elite ABC kit; Vector Laboratories, Burlingame, CA). Peroxidase activity was visualized as described above. As a negative control, biotin-conjugated normal mouse IgG (10 mg/ml; Santa Cruz Biotechnology) was used instead of the primary antibody.
Electron Microscopy and Immunoelectron Microscopy
Electron microscopic observation was performed as described previously (Komohara et al. 2005). For immunoelectron microscopy, frozen sections of PLP-fixed murine placentas were incubated overnight at 4C with anti-mouse LOX-1 monoclonal antibody (JTX-58). The sections were washed with PBS, after which they were incubated for 2 hr at 4C with peroxidase-conjugated anti-mouse IgG [(F(ab')2] (Amersham; Buckinghamshire, UK). Peroxidase activity was visualized in the same way as noted above. The samples were processed as previously described (Komohara et al. 2005). Ultrathin sections were evaluated using a Hitachi H-7500 electron microscope (Tokyo, Japan) without counterstaining.
Results
Expression of LOX-1 mRNA in Murine Organs
Using RT-PCR, we examined the expression of LOX-1 mRNA in various organs of the mouse (Figure 1A). The placenta had the highest level of LOX-1 mRNA expression. Lung and aorta showed moderate expression, and expression in other organs was weak or absent. To compare the level of LOX-1 mRNA expression in the placenta at various developmental stages, placental tissues from E10.5 to E18.5 were examined by means of real-time RT-PCR. Expression of LOX-1 mRNA peaked at midgestation (E14.5–E15.5) and decreased thereafter (Figure 1B).
Compartment-specific Expression of LOX-1 mRNA in Murine Placenta
Because strong LOX-1 mRNA expression was observed during midgestational days, placental tissue at E14.5 was used to examine the localization of LOX-1 protein and mRNA. The fundamental morphology of murine placenta is completed by midgestation, with the tissue being compartmentalized into four distinct regions: metrial glands (MG), decidua basalis (DB), junctional zone (JZ), and labyrinth zone (LZ) (Georgiades et al. 2002). The MG and DB belong to the maternal side, whereas the LZ and JZ belong to the fetal side. To examine the compartment-specific expression of LOX-1 mRNA, we separated pregnant uterus and placenta at E14.5 into four parts: myometrium, MG + DB, JZ + LZ, and yolk sac + amnion. RT-PCR analysis showed stronger expression of LOX-1 mRNA in the MG + DB and LZ + JZ compared with the other two parts (Figure 2).
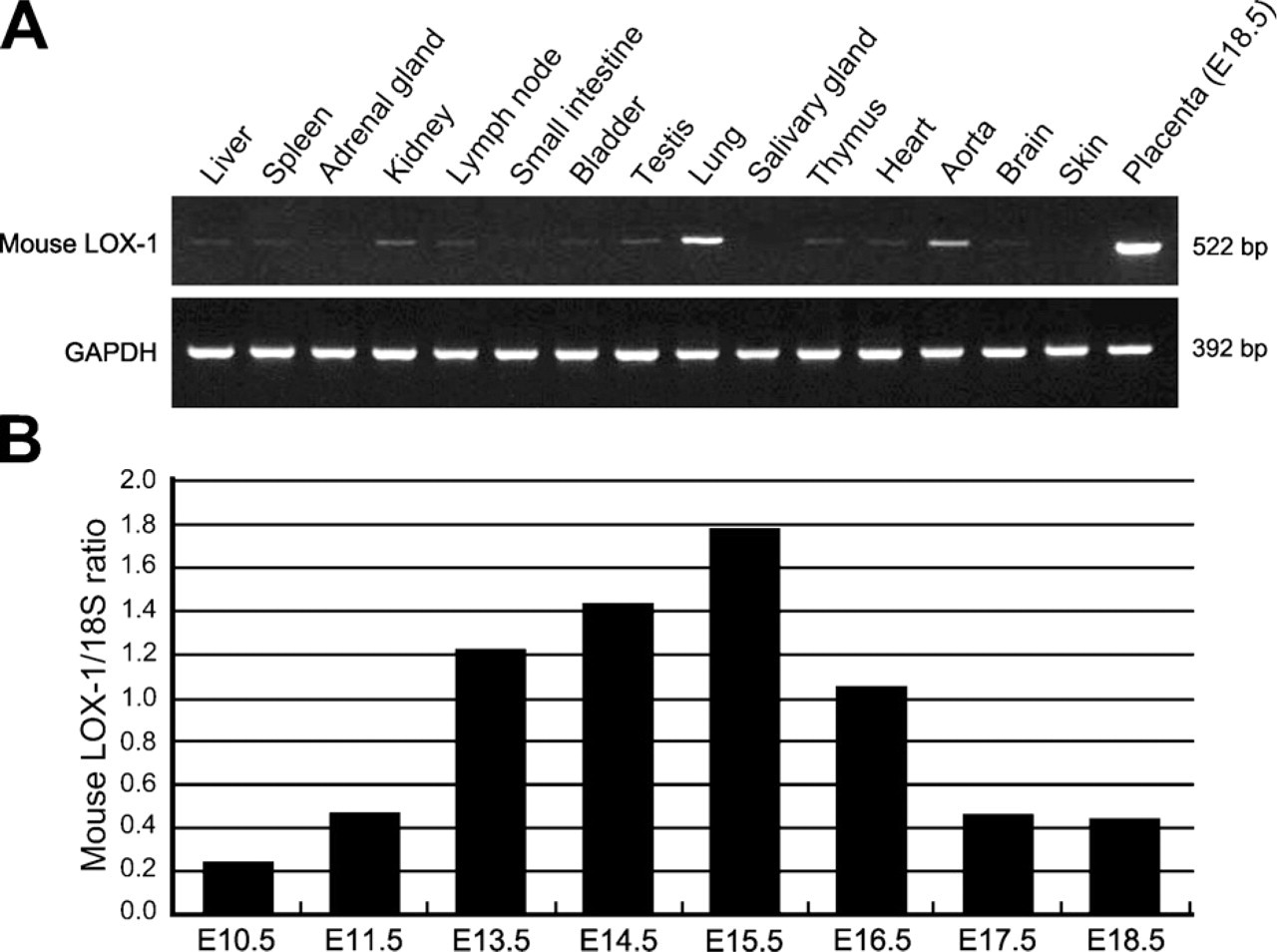
Expression of lectin-like oxidized low-density lipoprotein receptor-1 (LOX-1) mRNA in different murine organs (
Localization of LOX-1 Protein and mRNA in Different Compartments of Murine Placenta
At E14.5, the four compartments of the murine placenta have distinct histological features (Figure 3A). To clarify the precise localization of LOX-1 expression in each compartment, we used immunohistochemistry and ISH. In agreement with results of RT-PCR analysis, LOX-1 protein was detected in these compartments, with distinct localization patterns. Diffuse LOX-1 staining was observed in the MG and the outer part of the DB (Figure 3B). Strong cord-like staining in the JZ and faint diffuse staining in the LZ were also seen (Figure 3B). No positive staining was observed in a negative control section (Figure 3C). ISH showed clear colocalization of mRNA with the protein in all compartments (Figure 3D). An ISH control section, with the sense probe, showed no positive signal (Figure 3E).
Cellular Localization of LOX-1 Protein and mRNA in Different Compartments of Murine Placenta
In the mouse MG at midgestation, the major cellular components are granulated metrial gland (GMG) cells, also known as uterine natural killer (NK) cells, and fibroblast-like stromal cells. These two cell types are closely associated, with the fibroblast-like cells surrounding large, plump GMG cells (Figure 4A). In immunohistochemical studies, LOX-1 was localized in the fibroblast-like cells but not in GMG cells (Figure 4B). ISH clearly differentiated these two cell types: LOX-1 mRNA was detected in the cytoplasm of the fibro-blast-like stromal cells surrounding the GMG cells (Figure 4C). The expression pattern of LOX-1 mRNA in the fibroblast-like cells was in agreement well with that of desmin protein, a marker for this type of cell (Figure 4D). Ultrastructural observation confirmed the intimate association of the fibroblast-like cells and the GMG cells (Figure 4E). Immunoelectron microscopy confirmed dense labeling of the cell membrane of fibroblast-like stromal cells for LOX-1 (Figure 4F).
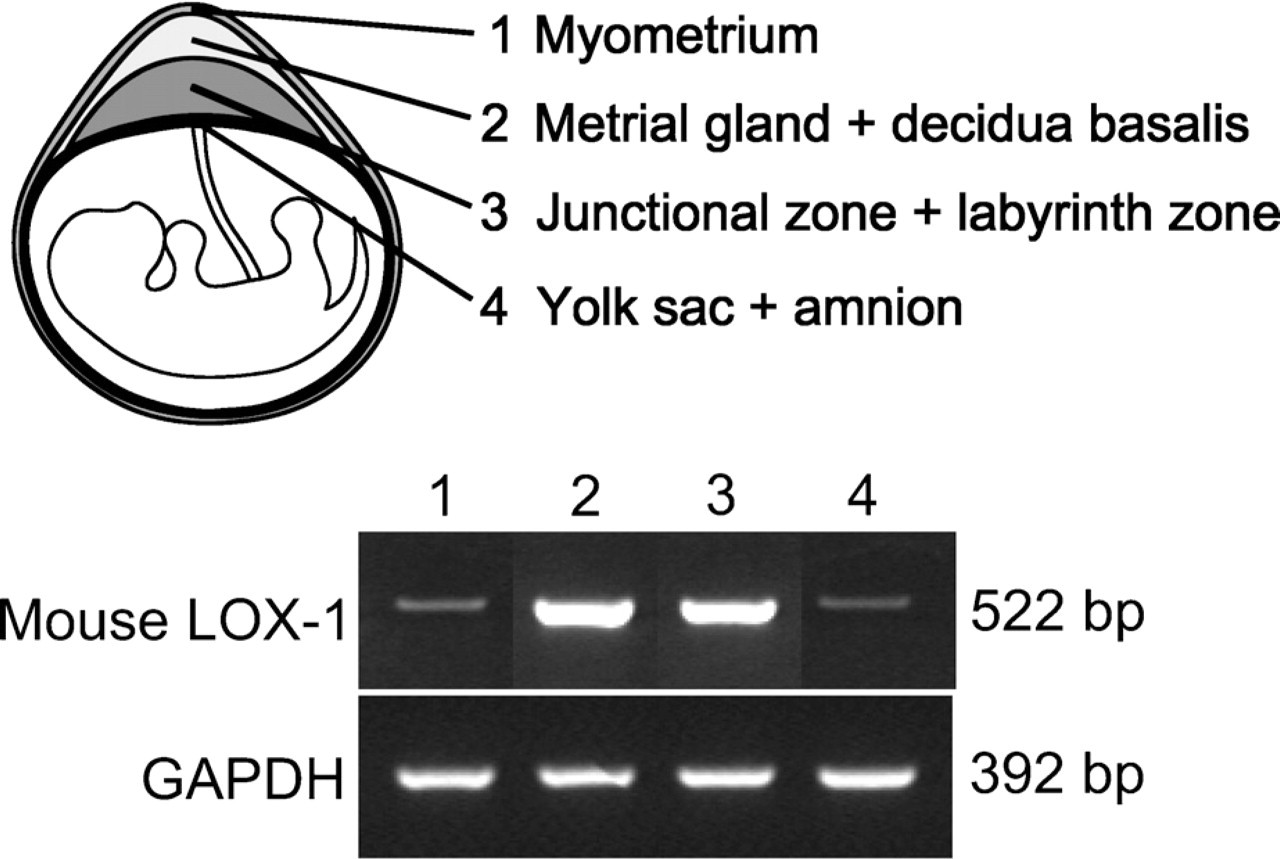
Expression of LOX-1 mRNA in different compartments of murine placenta. For RT-PCR, the placenta (E14.5) was divided into four parts: myometrium, metrial gland (MG) + decidua basalis (DB), junctional zone (JZ) + labyrinth zone (LZ), and yolk sac + amnion. The MG + DB (Lane 2) and the JZ + LZ (Lane 3) showed strong mRNA expression.
In the DB, the fibroblast-like cells formed a mesh-work around the GMG cells (Figure 5A), but in a rather looser fashion than in the MG. The loose arrangement made it easier to determine that LOX-1–expressing cells were fibroblast-like stromal cells (Figure 5B). Electron and immunoelectron microscopy showed positive LOX-1 staining on plasma membranes of these fibroblast-like cells (Figures 5C and 5D).
After E12.5, clusters of glycogen trophoblast cells with clear cytoplasm were noted among spongiotrophoblasts in the JZ (data not shown). Cord-like clusters of glycogen trophoblasts in the JZ were strongly positive for LOX-1 protein (Figure 5E) and mRNA (Figure 5F). After E14.5, some glycogen trophoblasts crossed the giant cell layer and migrated into the DB, where they were still positive for LOX-1 but at a weaker intensity (Figure 5E). Spongiotrophoblasts and giant trophoblasts showed no positive staining (Figures 5E and 5F). Electron microscopy showed that these glycogen trophoblast cells had a characteristic pale vacuolated cytoplasm (Figure 5G), and LOX-1 was localized along the cell membrane (Figure 5H). In the LZ, weak staining of LOX-1 protein (Figure 5I) and weak mRNA expression (Figure 5J) were shown in labyrinthine cytotrophoblasts.
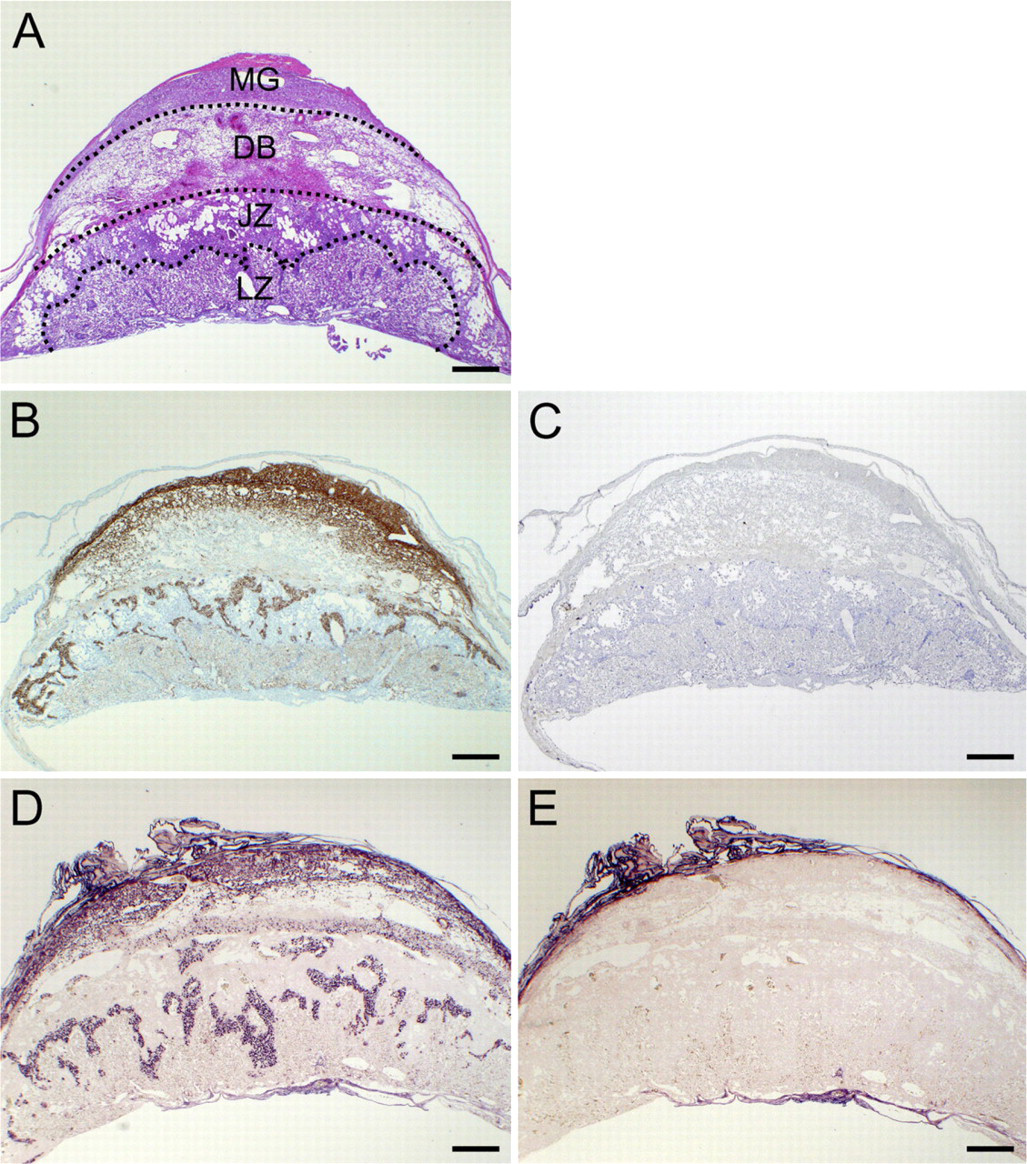
Localization of LOX-1 protein and mRNA in murine placenta at E14.5. Four distinct compartments were easily distinguished by means of hematoxylin and eosin staining (
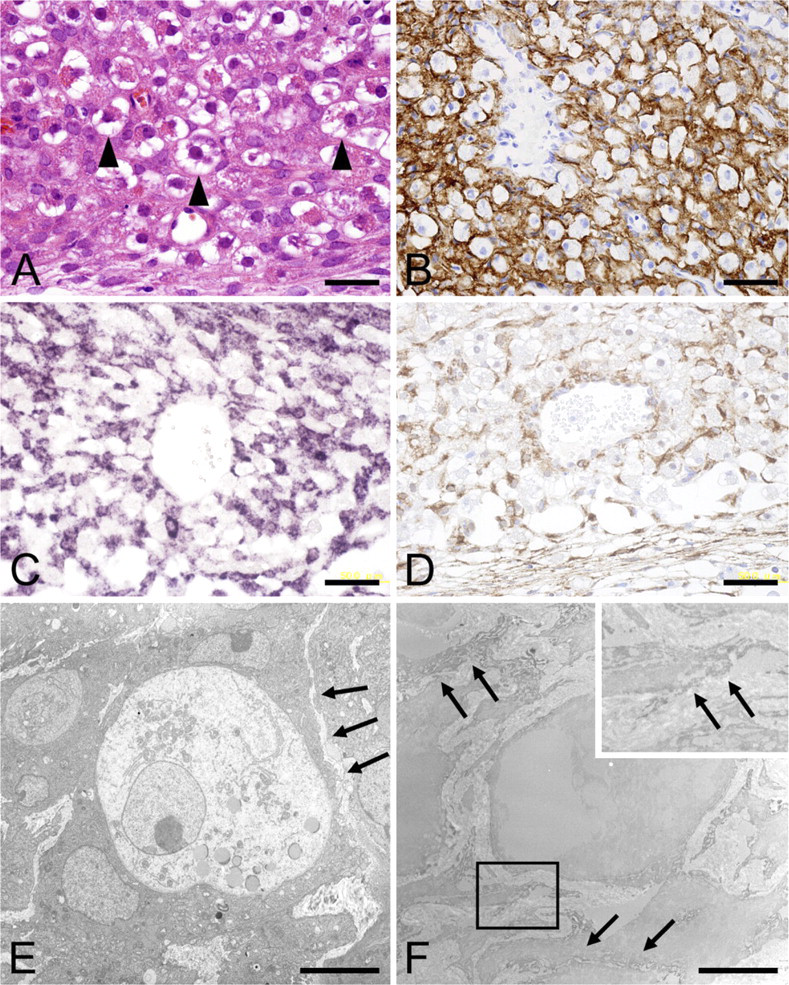
LOX-1–expressing cells in the MG of murine placenta at E14.5. (
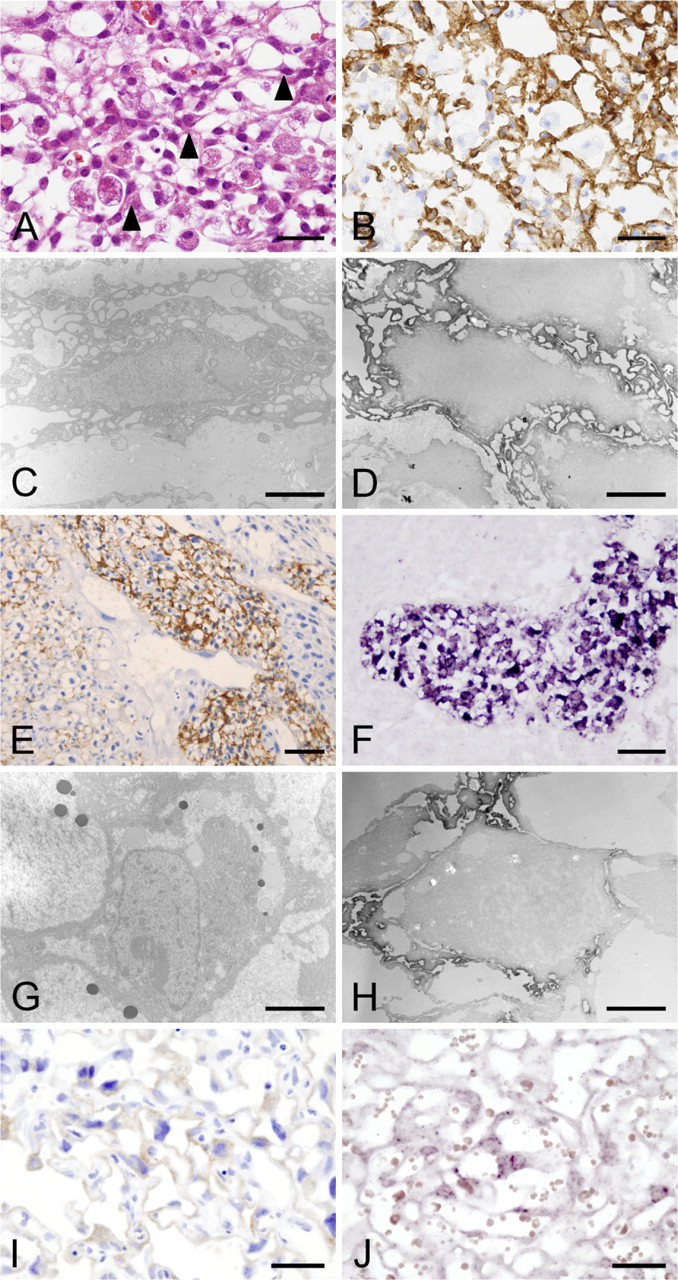
LOX-1–expressing cells in the DB, JZ, and LZ of murine placenta at E14.5. (
From these data, we concluded that, in the maternal compartment, LOX-1 localized in fibroblast-like stromal cells, whereas in the fetal compartment, LOX-1 was strongly expressed in glycogen trophoblast cells and weakly expressed in labyrinthine cytotrophoblasts.
Expression of LOX-1 mRNA in Human Placenta
To compare the expression levels of LOX-1 mRNA in first trimester and term human placentas, we performed real-time RT-PCR. Significantly stronger expression of LOX-1 mRNAwas found in first trimester placenta compared with term placenta (Figure 6).
Identification of LOX-1–expressing Cells in Human Placenta
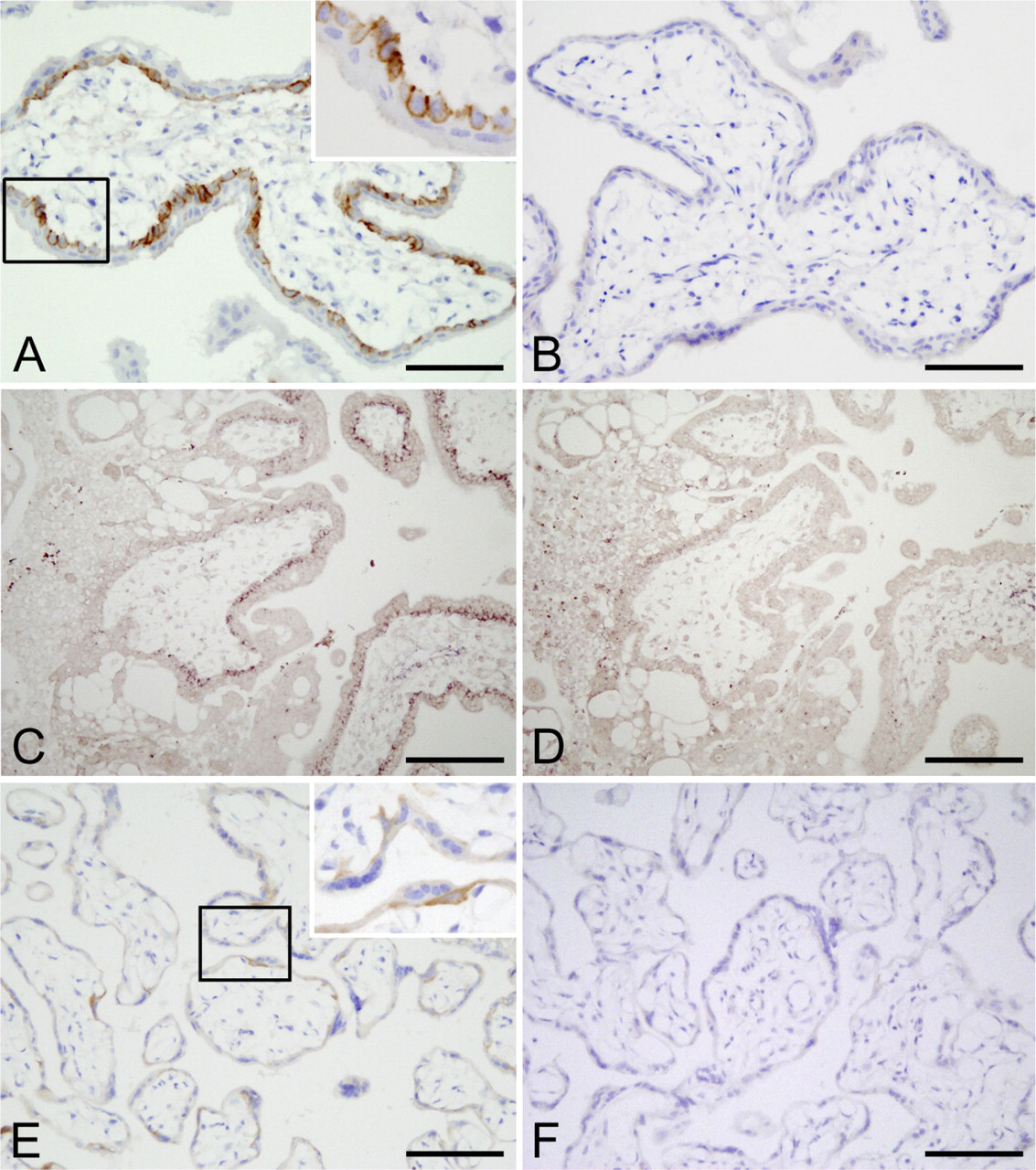
In the first trimester human placenta, strong positive immunoreactivity for LOX-1 was seen along the cell membrane of most villous cytotrophoblasts but not in syncytiotrophoblasts (Figure 7A). No positive staining was observed in a negative control section (Figure 7B). ISH of a first trimester placenta showed a positive reaction for LOX-1 mRNA in villous cytotrophoblasts (Figures 7B and 7C). The localization pattern of LOX-1 did not change in the term placenta. Lox-1 is positive on flattened cytotrophoblasts located beneath the syncytiotrophoblasts (Figure 7E). No staining was observed in a control section (Fig. 7F).
Discussion
Since the discovery of LOX-1 by Sawamura et al. (1997), many reports have suggested its possible involvement in the pathogenesis of atherosclerosis (Mehta et al. 2006). Recently, Mehta et al. (2007) reported that deletion of LOX-1 in atherogenic LDL receptor knockout mice reduced atherosclerosis and proinflammatory and prooxidant signals. LOX-1 expression is enhanced in some proatherogenic settings such as hypertension and hyperlipidemia in vivo and in atherosclerotic lesions (Mehta et al. 2006). LOX-1 is expressed at very low levels in healthy endothelium (Sawamura et al. 1997), and the physiological role of LOX-1 is not well understood. Northern blotting data for normal mice and humans (Sawamura et al. 1997) showed that the placenta is the organ with the greatest expression of LOX-1 mRNA, which indicates that LOX-1 may play an important role in maintaining pregnancy.
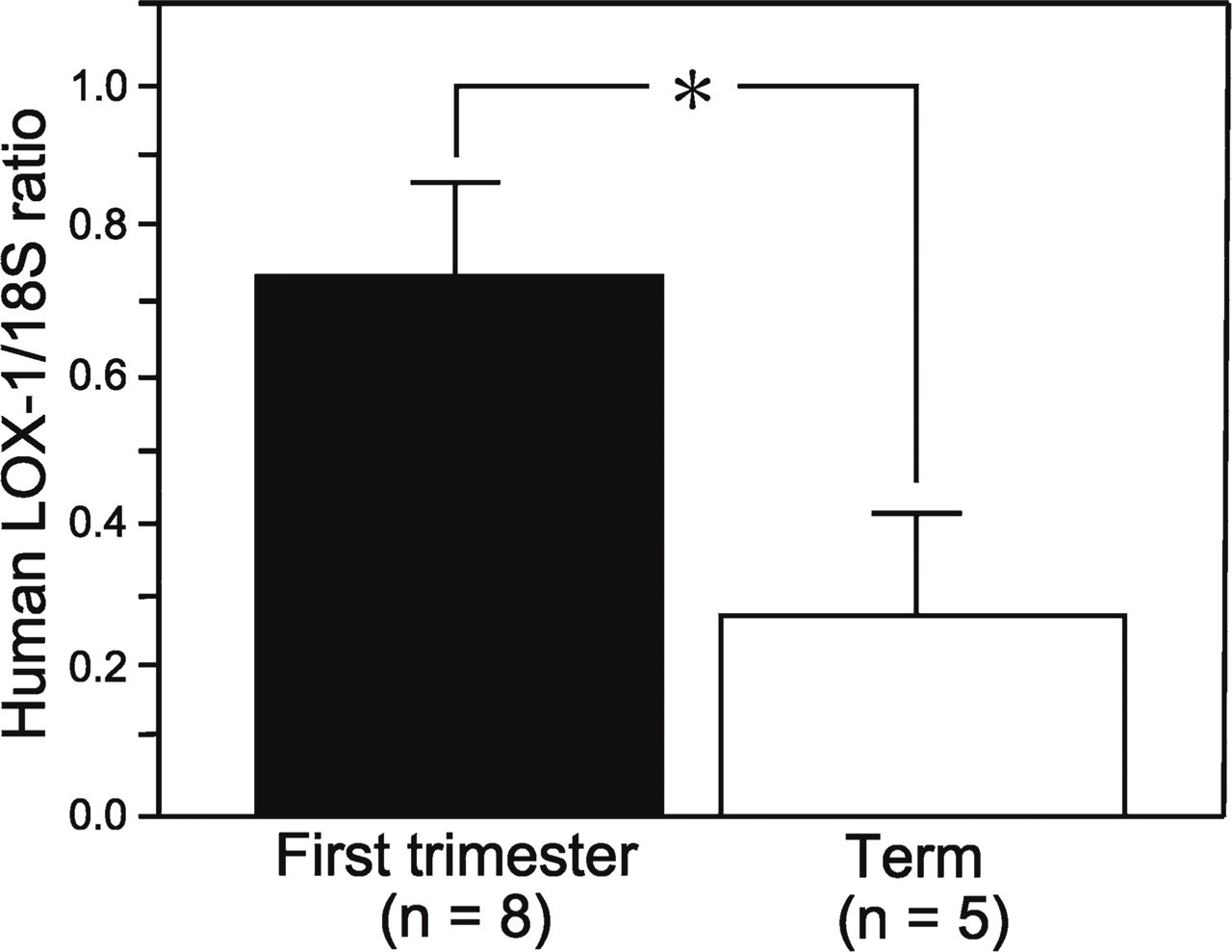
Expression of LOX-1 mRNA in human placenta. Real-time PCR showed significantly greater LOX-1 mRNA expression in the first trimester placenta than in the term placenta (∗p<0.05).
During pregnancy, the placenta is a major source of prooxidant and endogenous antioxidant synthesis (Walsh 1998). Excessive generation of free radicals depletes antioxidant pools and causes oxidative damage to lipids, proteins, and DNA. The level of placental oxidative stress increases rapidly at the end of the first trimester, when placental vascular development is occurring, and such stress tapers off later in gestation (Jauniaux et al. 2000). Our present finding of higher expression of LOX-1 mRNA during early and mid-gestational stages compared with late gestation in both mouse and human indicates induction of LOX-1 by increased oxidative stress at this gestational stage and active involvement of LOX-1 in the functioning of early gestational stage placenta.
In maternal decidual compartments of murine placenta, namely the MG and DB, the major cellular components are vascular endothelial cells, uterine NK cells (GMG cells), and fibroblast-like stromal cells. It is interesting to note that LOX-1 localized in fibroblast-like cells—the cells that are closely associated with uterine NK cells—but not in vascular endothelial cells. Certain studies indicate the possible role of uterine NK cells in control of trophoblast invasion and remodeling of uterine spiral arteries during the first half of pregnancy (Bulmer and Lash 2005). Intimate association between uterine NK cells and fibroblast-like stromal cells is believed to be important for survival of the uterine NK cells (Stewart 2000). Because ox-LDL binding of LOX-1 in fibroblasts induces expression of various adhesion molecules, such as intercellular adhesion molecule-1 and vascular cell adhesion molecule-1 (Chen et al. 2005), LOX-1 may be involved in strengthening the close association of these cells. The number of uterine NK cells decreases in late pregnancy, and apoptotic uterine NK cells were observed at day 16 in rat placenta, with their number increasing thereafter (Fukazawa et al. 1998). Because LOX-1 functions in the elimination of aged or apoptotic cells (Oka et al. 1998), LOX-1 expression by fibroblast-like stromal cells may also contribute to the elimination of apoptotic uterine NK cells.
(
In the JZ and LZ, LOX-1 expression was restricted to glycogen trophoblast cells. Glycogen trophoblast cells are of undetermined origin that arise in the JZ (Fukazawa et al. 1998). Although these cells were once thought to be differentiated from spongiotrophoblasts (Adamson et al. 2002; Georgiades et al. 2002), more recent evidence suggests that they are distinct from spongiotrophoblasts (Rampon et al. 2005; Bouillot et al. 2006). Because we found no LOX-1 expression in spongiotrophoblasts at the mRNA and protein levels, we believe that LOX-1 is one of the molecules that discriminates between glycogen trophoblast cells and spongiotrophoblasts.
This is the first study to describe the distinct localization of LOX-1 in murine placenta. It is interesting to note that LOX-1 expression is not observed in vascular endothelial cells of murine placenta. Because vascular endothelial cells are a major cell type for LOX-1 expression in the lung, aorta, and other organs (Sawamura et al. 1997), LOX-1 expressed by fibroblast-like stromal cells and glycogen trophoblast cells may possess unique functions in murine placenta.
Because glycogen trophoblast cells invade decidua, they are considered to be analogous to the human invasive extravillous trophoblasts (Malassine et al. 2003; Coan et al. 2006). Pavan et al. (2004) reported that ox-LDL, but not native LDL, inhibited human extravillous trophoblast invasion in a concentration-dependent manner, as evidenced in the trophoblast invasion assay. They also discovered that LOX-1 was the main scavenger receptor responsible for uptake of ox-LDL by cytotrophoblasts (Pavan et al. 2004). Taking these data together, LOX-1 expression by glycogen trophoblast cells may regulate their invasion of the DB. The weaker expression of LOX-1 by invading glycogen trophoblast cells in the DB, which we observed, supports this hypothesis.
In the first trimester placenta of humans, we confirmed the localization of LOX-1 in villous cytotrophoblasts but not syncytiotrophoblasts nor vascular endothelial cells by means of immunohistochemistry and ISH. We also found LOX-1 localization in villous cytotrophoblasts in term placentas. Our result in term placenta is inconsistent with the previous observation by Lee et al. (2005), who observed LOX-1 expressionin syncytiotrophoblasts in normal term and preeclamptic placentas, with stronger staining in the latter. LOX-1 staining in Figure 3 of their report (Lee et al. 2005), however, seems to localize in the cytotrophoblast layer. Inarecent study, Mori et al. (2007) reported that the cytotrophoblast layer in the term placenta becomes thinner by spreading their cell surface over the basal lamina, and 80% of its continuity is still preserved. Our data of LOX-1 localization in term placenta support their observation.
For the murine placenta, we clearly showed that LOX-1–expressing cells were fibroblast-like stromal cells in the MG and DB and glycogen trophoblast cells in the JZ and LZ, as well as labyrinthine cytotrophoblasts in the LZ. LOX-1 does not seem to be essential to maintain placental function, because LOX-1–deficient mice were fertile (Mehta et al. 2007) and had no detectable abnormal pregnancy. However, LOX-1–expressing placental cells may be involved in management of oxidative stress during pregnancy. Further study is needed, however, to clarify the exact role of LOX-1 in pregnancy.
Footnotes
Acknowledgements
This study was supported in part by Grants-in-Aid for Scientific Research (B16390108, C17590797) from the Ministry of Education, Culture, Sports, Science, and Technology of Japan.
