Abstract
The pancreatic islet vasculature comprises microvascular endothelial cells surrounded by mural cells (pericytes). Both cell types support the islet by providing (1) a conduit for delivery and exchange of nutrients and hormones; (2) paracrine signals and extracellular matrix (ECM) components that support islet development, architecture, and endocrine function; and (3) a barrier against inflammation and immune cell infiltration. In type 2 diabetes, the islet vasculature becomes inflamed, showing loss of endothelial cells, detachment, and/or trans-differentiation of pericytes, vessel dilation, and excessive ECM deposition. While most work to date has focused either on endothelial cells or pericytes in isolation, it is very likely that the interaction between these cell types and disruption of that interaction in diabetes are critically important. In fact, dissociation of pericytes from endothelial cells is an early, key feature of microvascular disease in multiple tissues/disease states. Moreover, in beta-cell replacement therapy, co-transplantation with microvessels versus endothelial cells alone is substantially more effective in improving survival and function of the transplanted cells. Ongoing studies, including characterization of islet vascular cell signatures, will aid in the identification of new therapeutic targets aimed at improving islet function and benefiting people living with all forms of diabetes.
The Critical Role of the Microvasculature in Maintaining Islet Health
Pancreatic islets are micro-organs comprising multiple endocrine cell types. The appropriate secretion of insulin and glucagon, endocrine hormones derived, respectively, from islet beta and alpha cells, is critical for the maintenance of glucose homeostasis. This process is tightly controlled and includes paracrine signals from multiple sources, such as the beta and alpha cells themselves, along with somatostatin-secreting delta cells, pancreatic polypeptide–secreting gamma cells, and ghrelin-secreting epsilon cells, which together exert highly coordinated paracrine communication.1–5 A recent, somewhat controversial, study has suggested that non-beta-cell endocrine cells may not be required for normal beta-cell function/glucose metabolism. 6 However, is it well recognized that islets also contain multiple non-endocrine “support” cells, including an extensive microvascular capillary network, as well as other cell types such as resident macrophages and Schwann cells which are critical for normal islet development and endocrine function.7–14
Anatomy of the Islet Vasculature
Microvascular endothelial cells form capillary lumens throughout the body but are morphologically and physiologically heterogeneous.15–17 A specialized arrangement of cell/cell junctional proteins develops through the activity of paracrine interactions between vascular cells, 18 which again differs among microvascular beds from different tissues. This results in heterogeneity, reflecting the organotypic properties of capillaries, which can be continuous, fenestrated, or sinusoidal, depending on the local tissue/organ requirements. 19 In common with other endocrine tissues, the islet endothelium is fenestrated, to facilitate the rapid transfer of nutrients and other circulating factors to endocrine cells, and release of islet hormones into the circulation.
In addition, pancreatic islet microvascular endothelial cells are surrounded by tightly associated mural cells called pericytes. 20 Individual pericytes contact multiple endothelial cells through elongated cell projections13,21 and regulate endothelial cell proliferation, vascular permeability, capillary diameter, and shear flow.20,22–24 Endothelial cells and pericytes both play critical roles during the formation of the developing vasculature and also for maintaining a stable vascular conduit throughout adult life. 25
Islet Vasculature-Derived Signals That Support Islet Health and Function
Endothelial cells produce several classes of molecules capable of supporting beta-cell proliferation, identity, and function.10,13,26 Secreted growth factors including hepatocyte growth factor27,28 and cellular communication network 2, formerly known as connective tissue growth factor, 29 support beta-cell proliferation under physiological conditions of beta-cell expansion, such as pregnancy. Endothelin-1, a potent vasoactive peptide, also enhances insulin release via direct beta-cell effects 30 and/or indirect effects on the alpha cells. 31 Endothelial cells also exert positive effects on beta cells through actions of cell–cell (e.g., connexins) or cell–matrix signaling (cadherins), among others.9,10,13
Islet pericytes also secrete paracrine signals, including bone morphogenetic protein 4 and nerve growth factor (NG2), which induce the expression of key beta-cell genes and promote beta-cell function, identity, and survival.32–34 The role of islet pericytes in health and in the context of type 1 diabetes (T1D) is reviewed by Gonçalves et al. 35 in this special collection. Endothelial cells and pericytes are intimately associated, sharing a specialized extracellular matrix (ECM) basement membrane (BM) along the abluminal surface of vessel walls. The islet BM comprises ECM molecules, including laminins, type IV collagen, heparan sulfate proteoglycans, and nidogen.36–41 Both endothelial cells 36 and pericytes 34 contribute to BM deposition (Fig. 1A). These BM components are necessary for islet health and function.10,13 In vitro studies show that exposure to ECM improves survival of human and rat islets and enhances the endocrine commitment in pancreatic cells differentiating from human pluripotent stem cells (hPSCs).42–45 Moreover, the interaction between pancreatic ECM components and endocrine cells of the islet is critical for optimal production/release of insulin.36,46–48
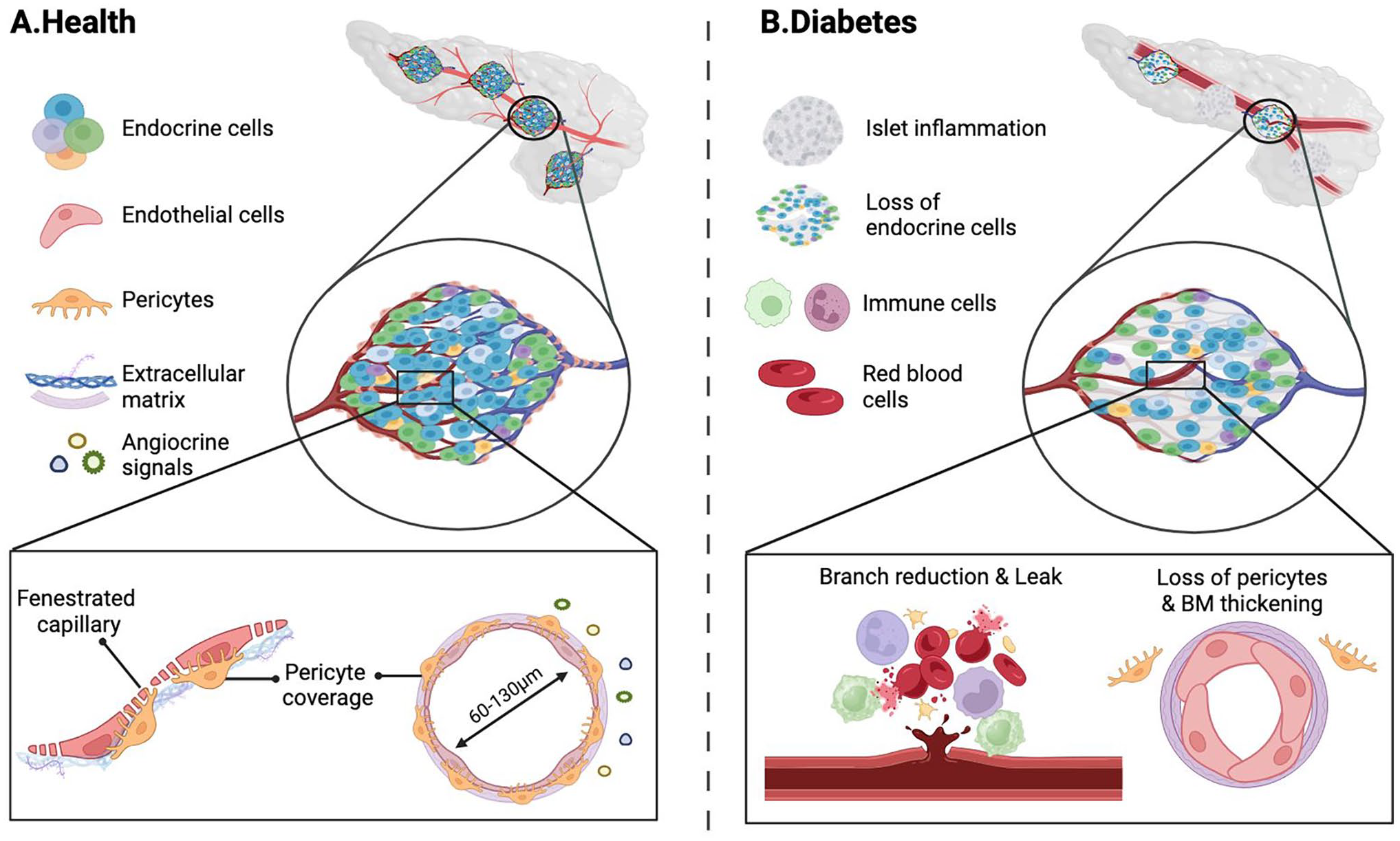
Schematic of the islet vasculature in health or in diabetes. (A) The islets contain a dense network of capillaries, 60–130 μm in diameter. The endothelium is fenestrated allowing exchange between the circulation and endocrine cells of the islets. The fenestrated endothelium is supported by pericytes, which regulate local blood pressure, support vessel wall to prevent leak, and maintain vascular integrity. Together, the endothelial cells and pericytes secrete extracellular matrix components such as laminins, collagens, and fibronectin, as well as paracrine signals such as NG2, HGF, endothelin-1, which regulate islet development and function. (B) In diabetes, the vascular branches are reduced, basement membrane is thickened (due to deposition of ECM components), diameter is changed, and pericyte coverage is lost leading to leak of blood and immune cells, altered mechanical and paracrine signaling which lead to inflammation and fibrosis. Schematic is generated by biorender.com.
Islet Vascular Heterogeneity
Single-cell gene expression and proteomic studies on islet microvascular cells 49–51 have revealed that individual vascular cell types produce distinct secreted factors and miRNAs, including but not limited to the molecules described above. These distinct secretomes likely contribute to highly localized functions and these data have provided a new perspective on the complex nature of the relationships between the microvascular cell types within the pancreatic islet, and how these interactions support islet endocrine function. However, studying such relationships between islet vascular cell types and/or between vascular and endocrine cells in vitro has been hampered by the fact that endothelial cells are rapidly lost to necrosis and apoptosis during standard islet isolation procedures. 52 Nevertheless, strategies such as the use of live pancreatic slices have allowed the in situ characterization of islet vasculature53–58 and demonstrated that while human islets have lower endothelial cell density compared with mice islets,59–62 they are supported by contractile pericytes that regulate blood flow locally to fine-tune glucose homeostasis. 20 The unique phenotype/signature of the islet vasculature appears, therefore, to be critical for normal health and function of islet endocrine cells. Changes to this signature under conditions where the islet vasculature is compromised, including islet transplantation or diabetes as described below and in the work by Mateus Goncalves et al., 35 are understudied and could yield novel therapeutic targets to improve islet function and health.
Islet Vascularization for Transplantation
Islet transplantation is a promising long-term therapy for T1D, in which donor islets are infused into the portal vein of the recipients. The islet survival posttransplant is challenged by poor islet engraftment as a result of delayed vascularization and instant blood-mediated inflammatory response (IBMIR) as a result of islet infusion into the portal vein. This results in an estimated 60–80% loss of transplanted islet mass within days after infusion. 63 Survival of transplanted islets depends on multiple factors, with a critical factor underlying this process being the paucity of vasculature within transplanted islets and slow revascularization of islet grafts.64–67 Similar challenges are reported following transplantation of hPSC-pancreatic in the abdomen, which avoids IBMIR, but the lack of rapid and robust vascularization significantly jeopardizes cell engraftment, leading to insufficient insulin secretion. 68 Recent studies, described further in section “Treating the Diabetic Islet through a Microvascular Lens,” have highlighted that the incorporation of vascularization strategies significantly improves pancreatic cell survival posttransplantation, beta-cell function, and overall restoration of glycemic control in hyperglycemic models.69–74
Disturbed Islet Vasculature Morphology in T2D
Generalized Microvascular Disease in Diabetes
Loss of glycemic control in diabetes has profound detrimental effects on the microvasculature throughout the body and this contributes to diabetic complications in multiple organs.75–79 While, as noted above, vascular beds across organs are highly specialized, key similarities in diabetes-induced vascular defects across diverse organs can be observed. These include dysfunctional vascular permeability, increased ECM deposition or altered stiffness, and extracellular fibrosis which are observed in diabetic retina, 80 kidney, 81 brain, 82 and during impaired wound healing. 83
Islet Microvascular Pathology and Inflammation in T2D
Studies of human pancreas specimens from donors with T1D 35 and T2D show that vascular morphology is strikingly abnormal. In T2D, islet capillaries are thickened and fragmented, leading to an apparent increase in vascular density59,84,85 (Fig. 1B). However, it is not possible to determine from these autopsy pancreas samples whether fragmentation of capillaries corresponds to impaired perfusion or function. Therefore, most of our knowledge in this area comes from animal models. While microvascular eye and kidney disease have been classically considered to be end-stage complications of diabetes, in the islet, microvascular abnormalities appear to occur early in the course of diabetes. This early pathology is consistent with a role of islet vasculature pathology/dysfunction in contributing to defects in insulin release and/or compromised beta-cell survival that define the progression of T2D. In support of this, islet vessel density is decreased and pericyte area is increased in obese but nondiabetic ob/ob 86 and mice with transgenic expression of the human form of islet amyloid polypeptide (hIAPP). 87 The ob/ob mouse also shows a marked dilation and increased blood flow within islet capillaries which may occur due to increased activity of endothelial nitric oxide synthase (eNOS).86,88 Moreover, islet endothelial cells from obese hIAPP transgenic mice 87 or db/db mice on a C57BL6/J background (which are resistant to the development of classical diabetic microvascular complications) have an activated inflammatory and pro-adhesive phenotype. 89 This inflamed, pro-adhesive phenotype is also seen in islet vasculature of models of established diabetes including the Goto-Kakizaki (GK) rat 90 and C57BL/Ks.db/db mouse, 91 while islets from the Zucker diabetic fatty (ZDF) rat model of T2D exhibit increased levels of thrombospondin-1 (an anti-angiogenic factor important in endothelial-pericyte interactions) and increased expression of vascular endothelial growth factor (VEGF) expression, 92 potentially in an attempt to compensate for capillary loss. Indeed, in vivo studies have demonstrated that under hyperglycemic conditions, islet endothelial cell proliferation decreases, apoptosis increases.93,94 Furthermore, ultrastructural studies using electron microscopy of islet capillaries from diabetic db/db mice revealed a decrease in capillary density and fibrosis, 95 with the ZDF rat model showing endothelial hypoplasia. 92 Together, these data clearly establish the islet vasculature as a key site of pathology in T2D, with loss of supportive factors and induction of pro-inflammatory mediators that could impair islet endocrine cell function and heath via a variety of mechanisms.
Mechanisms Leading to Islet Microvascular Defects in T2D
Hyperglycemia and Oxidative Stress
Dysregulated metabolism of glucose and other nutrients (e.g., lipids) seen in T2D contributes to diabetic microvascular complications across tissues and organs, including direct effects on endothelial cells.96,97 When exposed to high glucose, endothelial cells exhibit increased expression of inflammatory adhesion molecules and increased permeability and production of advanced glycation end (AGE) products.98,99 AGE production has been widely shown to impair islet endocrine cell function and survival100,101 (recently reviewed in the work by Anastasiou et al. 102 ), and feeds back to have negative effects upon the islet vasculature, resulting in endothelial-to-mesenchymal transition and islet fibrosis in a mouse model. 103 Furthermore, in vitro studies of cultured islet endothelial cells show that exposure to a T2D milieu (high glucose, palmitate, TNFα, and insulin) is sufficient to induce the expression of pro-inflammatory cytokines and cell adhesion molecules, and results in an endothelial secretome that is no longer able to support beta-cell function. 10
Dysregulation of Growth Factors
Maintaining vascular integrity requires a group of growth factors that when lost results in disruption of tight junctions, apoptosis of vascular cells and mobilization of pericytes with concomitant differentiation into pro-fibrotic phenotype. 104 Among the growth factors or receptors critical for maintenance of pericyte and endothelial cell adherence eNOS, endothelin-1 and VEGF-A are dysregulated in models of T2D.86,89,90 In particular, VEGF dysregulation results in a variety of vascular defects including disrupting the fenestrated structures within the islet microvasculature.105,106 VEGF is dysregulated in the diabetic ob/ob mouse 86 and within the islet vasculature of ZDF rats, which increased microvascular permeability leading to intra-islet hemorrhages. 92
Islet Amyloid
Aggregation of the beta-cell peptide islet amyloid polypeptide (IAPP) into islet amyloid is a feature observed in the majority of people with T2D.107,108 The hIAPP aggregation is well known to be toxic to beta cells 109 (reviewed in the work by Raleigh et al. 110 ) and also induces an inflammatory response in macrophages/dendritic cells.111,112 It has long been appreciated that islet amyloid deposition is closely associated with islet capillaries113–115 (Fig. 2A), but its impact on islet endothelial cells has only recently been investigated. Capillary morphology in amyloid-laden islets is selectively disturbed both in T2D humans59,85 and hIAPP transgenic rodent models87,116 (Fig. 2). Amyloid-laden islets have decreased capillary density,85,87 and remaining capillaries that are adjacent to amyloid deposits exhibit increased diameter, selective upregulation of the cell adhesion molecule vascular cell adhesion protein 1 and increased number of NG2-expressing pericytes. 87 Aggregated hIAPP induces a toll-like receptor 2/4–dependent pro-inflammatory reaction in cultured islet endothelial cells, 87 demonstrating that the inflammatory effects seen in vivo may include a direct action on endothelial cells. However, work using brain microvascular pericytes117,118 also suggests that hIAPP may have detrimental effects on both vascular cell types. Bulk islet RNAseq data also suggest that hIAPP aggregation alters gene expression in both endothelial cells and pericytes, both in transgenic mice overexpressing hIAPP and islets from T2D donor. 119
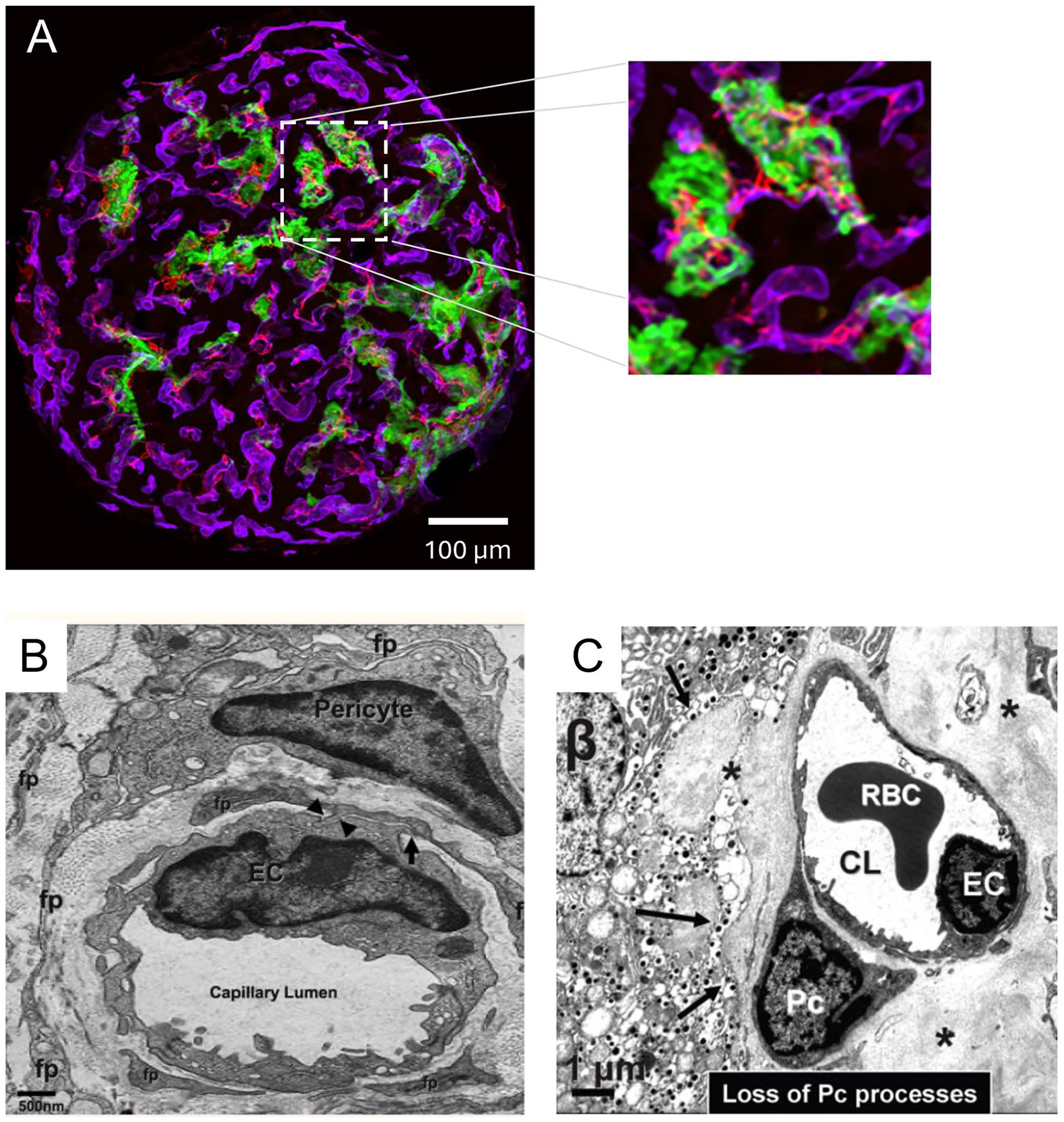
Effect of hIAPP deposition on islet microvasculature. (A) Confocal maximum intensity projection (captured on Nikon A1R confocal of an islet from an hIAPP transgenic mouse (A), with labeling for amyloid deposition (green; thioflavin S staining), microvascular endothelial cells (via IV injection of DyLight-649-conjugated Lycopersicon esculentum lectin) and pericytes (anti-NG2 AB5320, EMD Millipore and visualized with Alexa Fluor-568-conjugated goat anti-rabbit IgG); scale bar = 100 µm. Inset illustrates the close association between amyloid deposits and the islet vasculature, making it ideally placed to disrupt islet vascular homeostasis. (B) Electron micrograph showing normal microvascular morphology from a rat islet including endothelial cell (EC), pericytes and foot processes (FP) protruding from pericytes to contact endothelial cells (arrows/arrowheads). (C) Electron micrograph showing disturbed vascular morphology in a transgenic HIP rat. The deposition of hIAPP as amyloid (*) occurs between beta cells (β, arrows) and islet vasculature. In this example, the endothelial cell (EC) and pericyte (Pc) are still present but there is a notable reduction in pericyte foot processes, a hallmark of vascular instability (CL = capillary lumen; RBC = red blood cell). Panels B and C are reproduced, with permission, from Hayden et al. 116
ECM Deposition and Fibrosis
Increased deposition of ECM (which includes, but is not limited to, amyloid deposition) and fibrosis is a hallmark of diabetic vasculature in multiple tissues. In the islet, morphological changes observed in pancreatic sections from T2D subjects include capillary thickening and an increase in BM deposition59,120 (Fig. 1B). This is often associated with fragmentation and increased vessel density as compared with nondiabetic subjects.59,84,120 In islets from the T2D ob/ob mouse model, microvessels are dilated and have a BM nearly double in thickness. 86 However, this effect is not shared by the db/db mouse. 86
As mentioned, both endothelial cells and pericytes synthesize ECM components and both likely contribute to increased ECM deposition in T2D. In addition, however, when pericytes become activated by cytokines or growth factors, they can differentiate into a mobile pro-fibrotic phenotype.24,54,121 Pericytes trans-/de-differentiation may contribute to the loss of their contractile functionality and alteration of blood flow within the islets. In addition, the pericyte–myofibroblast transition can activate inflammation of endothelial cells122–124 and results in an increase in ECM production as well as altered ECM components (e.g., fibrillar collagen and smooth muscle actin).124,125 The thickening observed within the islet vasculature in diabetes is correlated with areas of beta-cell loss 120 and likely impedes the normal paracrine communication between beta cells and endothelial cell/pericytes that is required for normal beta-cell function and survival.
Destabilization of Islet Microvasculature—An Underappreciated Component of Islet Pathology in Diabetes
The diverse microvascular defects observed across organs and tissues during the progression of T2D share the unifying themes of oxidative stress, inflammation, dysregulation of growth factors, apoptosis of endothelial cells and pericytes, pericyte detachment, pericyte myofibro-transformation, and tissue fibrosis. One of the key features of microvascular pathology when glycemic control is lost is a disruption of the association between endothelial cells and pericytes. 82 Activated pericytes can lose attachment to endothelial cells, and the loss of this connection is one of the earliest observed cellular defects in diabetes126,127 (Fig. 1B). Disruption of endothelial/pericyte homeostasis can be initiated by several mechanisms, including inflammation, generation of reactive oxygen species, AGE production, apoptosis, cell detachment, mobilization, differentiation, and alterations in ECM deposition,24,128 many of which are observed in the diabetic islet. Mechanistically, dysregulation of cell-to-cell junctional molecules in both endothelial cells and pericytes likely underlie destabilization of the islet vasculature in diabetes. In non-islet tissues, these proteins are well known to govern cell contact and transmission of intercellular signals. Conversely, disruption of these signals under diabetic conditions can contribute to migration of pericytes away from endothelial cells and promote increased vascular permeability. 129 While the mechanisms underlying the destabilization of islet microvasculature are not yet fully understood, the loss of pericyte–endothelial interactions in the islet microvasculature, whether through pericyte apoptosis or migration/differentiation, likely contributes to the dysregulation of capillary dilation instability, pathological angiogenesis, and rarefaction.20,54,82,130–132 Loss of vessel integrity and vascular remodeling may contribute to a reduction in beta-cell function through multiple mechanisms including disruption of paracrine signaling, 133 increased islet inflammation,134,135 and inflammation-related fibrosis. 90
Treating the Diabetic Islet Through a Microvascular Lens
The specific molecular mechanisms governing normal interactions between endothelial cells and pericytes in the islet and how these are disturbed under diabetic conditions are poorly understood. Whether reversing or preventing this loss of interaction can lead to new and improved diabetes therapies is currently understudied and is an active area of ongoing research. However, as discussed below (and summarized in Table 1), there is evidence, both from studies utilizing medications already approved for clinical use in T2D treatment and from modifications to islet transplantation procedures in animal models, that strategies which ameliorate islet vasculature inflammation, ECM deposition, and/or destabilization may be an effective approach to improve islet function and survival.
Studies Investigating Effects of Clinically Approved Drugs on Islet Microvasculature Using Animal Models.
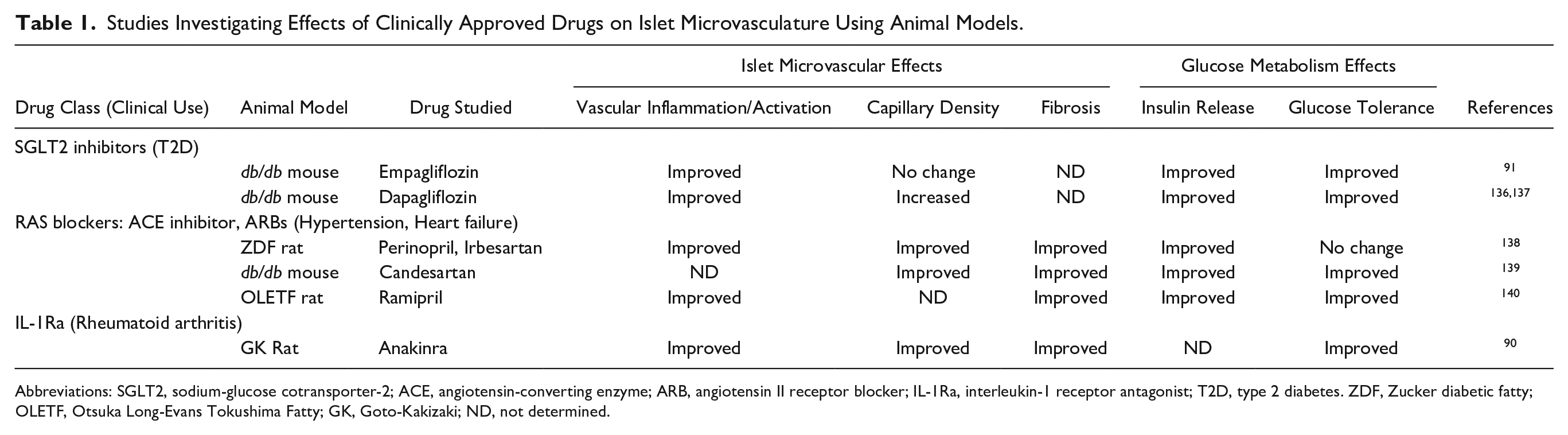
Abbreviations: SGLT2, sodium-glucose cotransporter-2; ACE, angiotensin-converting enzyme; ARB, angiotensin II receptor blocker; IL-1Ra, interleukin-1 receptor antagonist; T2D, type 2 diabetes. ZDF, Zucker diabetic fatty; OLETF, Otsuka Long-Evans Tokushima Fatty; GK, Goto-Kakizaki; ND, not determined.
Repurposing Existing Therapies to Treat the Islet Microvasculature
Several studies in animal models suggest that existing therapies used in patients with T2D may have beneficial effects on the islet vasculature. For example, normalizing blood glucose with sodium-glucose co-transporter 2 inhibitors is highly effective in reducing endothelial cell inflammation and dysfunction, including in the islets.91,136 This is associated with improved beta-cell function,91,136,137 which may in part result from the improved endothelial phenotype. In fact, in clinical trials, this class of drugs have protected against diabetic microvascular and cardiovascular disease.141,142 Using agents that target the renin-angiotensin system (RAS), by inhibiting either the angiotensin-converting enzyme or the angiotensin II type 1 receptor, is a common approach for treatment of hypertension, a common comorbidity in T2D. A large body of literature supports a role for RAS-targeting compounds in slowing the progression of diabetic kidney disease and decreasing the incidence of T2D.143–145 Use of these agents in animal models of T2D has been shown to be effective in reducing the severity of vascular defects within the islet, including fibrosis, and is again associated with an improvement in insulin release.138–140 Anti-inflammatory strategies may also be beneficial. In the GK rat model of T2D, blocking the activity of pro-inflammatory cytokine interleukin-1 (IL-1) with an IL-1 receptor antagonist reduced the expression of multiple markers of endothelial cell activation and inflammation, reduced vascular fibrosis, and improved islet function. 90 Of note, clinical studies investigating the effect of blocking IL-1 signaling on glucose control in T2D have yielded mixed results. Some,146,147 but not all studies,148,149 showed a beneficial effect, suggesting blockade of IL-1 alone may not be sufficient to improve beta-cell function in people living with T2D.
Leveraging Islet Microvascular Stability to Improve Islet Cell Replacement Therapies
Islet microvessels are also important in improving beta-cell replacement therapies via islet transplantation (mainly T1D but also potentially T2D). For example, reconstructing the islet vasculature and ECM which are digested during islet isolation from a donor’s pancreas or engineering a vasculature for co-transplantation with islets can significantly improve islet survival and function posttransplant69,72–74,150 (Fig. 3). Management of inflammation, angiocrine signaling, and pericyte/endothelial cell interactions is also likely to positively impact the survival of islets following transplantation and provide strategies critical for the development of bioengineered islet substitutes to treat diabetes. Of note, incorporating non-pancreatic accessory cells in islet grafts could improve graft survival posttransplantation. These accessory cells are mostly obtained form the placental tissues such as the amniotic epithelial cells and umbilical vein perivascular cells and have angiogenic or immunomodulatory capacities. 151 However, an optimal islet engraftment will likely require formation of neobasement membrane as the BM is damaged by collagenase digestion procedures commonly used during islet isolation. 38 A challenge shared in transplanting both hPSC-pancreatic cells and donor islets is poor survival of the transplanted cells/islets as a result of delayed and inefficient connection with the host.68,152,153 Failure of neovascular growth and anastomoses with host vasculature likely arises due to a combination of factors including ischemic shock, inflammation at the site of transplant, as well as the heterogeneity of endothelial cells between pancreatic islets and those in the host transplant site (e.g., liver or omentum), inefficient pericyte recruitment or a paucity of growth factors necessary to maintain pericyte/endothelial junctions and loss/mobilization of pericytes with trans-differentiation of pericytes into myofibroblasts, and lack of appropriate BM/ECM. Moreover, in adult life, vasculogenesis (the formation of new blood vessels) is extremely limited; 154 hence, revascularization relies on angiogenic sprouting from the transplanted site. Therefore, diseases, such as diabetes, that have significant vascular disease involvement can significantly jeopardize the angiogenic potential of the host, and there is evidence that an immunosuppressive regimen, coupled with current methods of cell therapy, also dampens vascularization. 155 Therefore, if revascularization is left to the host, it could have unexpected and variable outcomes (Fig. 3B and C). Therefore, reconstructing the islet vasculature is needed to boost in vivo engraftment and endocrine islet function. However, studies by many groups demonstrated that single endothelial cells cannot give rise to a stable vascular network posttransplantation156,157 (Fig. 3C–E), further highlighting the imperative role of pericytes and ECM in promoting vessel architecture, function, and stability. Our recent work and that of others demonstrated that survival of implanted islets and hPSC-derived pancreatic cells is improved with the addition of co-transplanted preformed microvessels that contain both endothelial cells and pericytes at a physiological ratio.73,74 The microvessels secrete ECM 158 and angiocrine signals159,160 that support sprouting angiogenesis,73,74 vessel remodeling, and vascular stability. 161 These features allowed the microvessels to support the engraftment of donor islets or hPSC-pancreatic progenitors in diabetic mice, by rapidly perfusing with the host and rescuing hypoxia in pancreatic cells, and to reconstitute the intra-islet vasculature support glucose-stimulated insulin secretion long term.73,74 These features are not achievable when single endothelial cells are used, which further confirms the importance of pericytes and their attachment to the endothelial cells for lumen integrity and vascular function. 73 In addition to including preformed microvessels with transplanted islets, other cells and tissues including mesenchymal stromal cells, isolated pericytes, or segments of arteries,162,163 these strategies and others reviewed in the work by Aghazadeh et al. 150 could boost islet function posttransplantation likely by providing growth factors or ECM components beneficial for islet health. Moreover, there is a large literature exploring the use of naturally occurring ECM or fabricated biomaterials to support islet transplantation; review of this work is beyond the scope of this article, but readers are directed to the following excellent reviews on the topic.164–167 These studies create consensus that more than one feature of the vasculature is important for developing a stable and functional vascular network, and to support successful beta-cell replacement therapy for T1D.
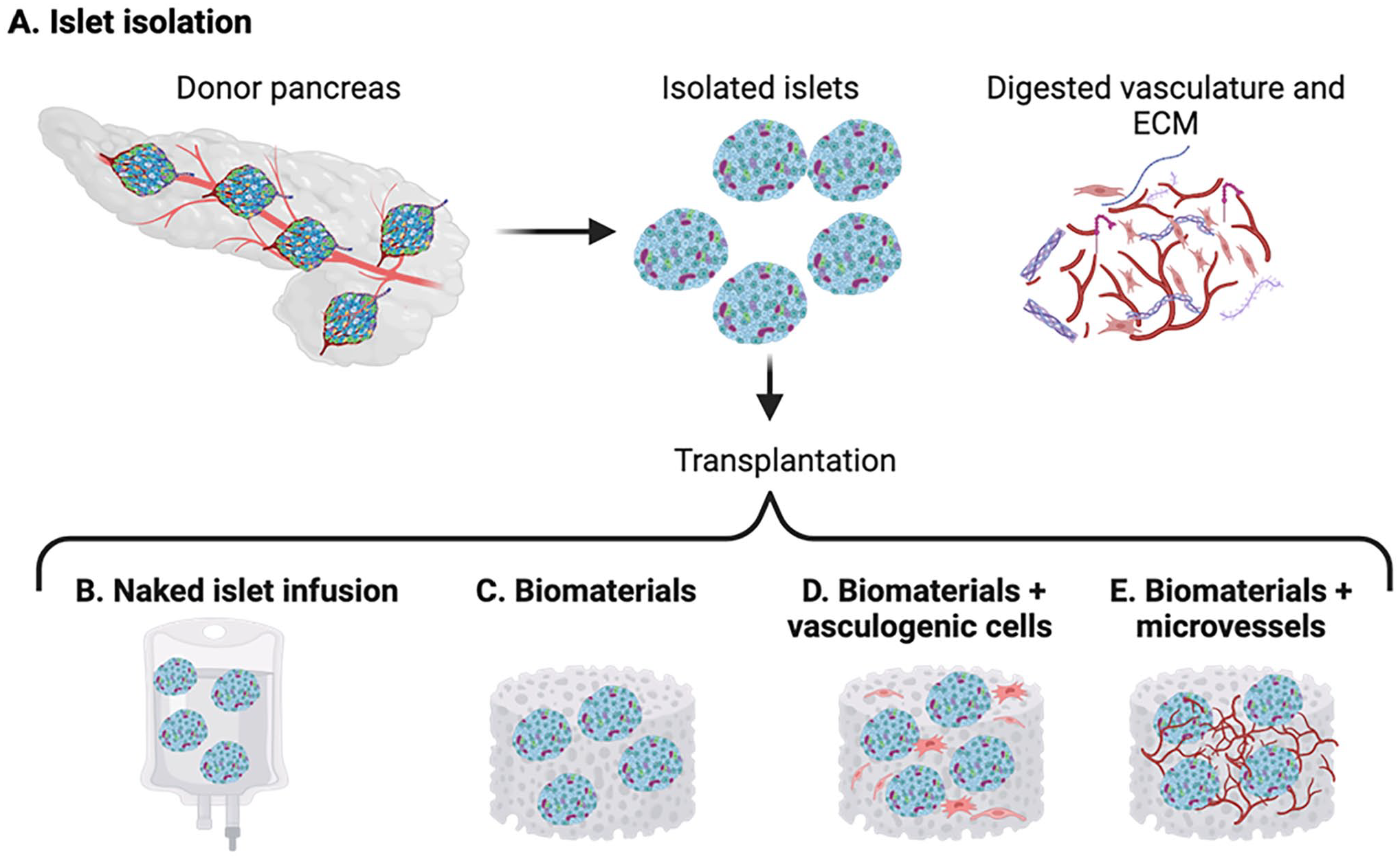
Schematic of current models of islet transplantation. (A) Islets are isolated from a donor’s pancreas, leading to digestion of the vasculature and extracellular matrix. Isolated islets can be transplanted by infusion into the portal vein naked (B), or via biomaterial scaffolds, meshes, hydrogels, or encapsulation devices alone (C), or along with vascular cells such as endothelial cells, mesenchymal stromal cells, fibroblasts (D), or with intact microvessel fragments containing endothelium and perivascular coverage (E).
Summary and Future Directions
The islet microvasculature, comprising endothelial cells and pericytes, is critical for normal islet health and endocrine function. The islet microvasculature exerts this positive effect in at least three distinct ways:
Serving as a conduit for delivery of oxygen and nutrients to the islet endocrine cells that require these for their specialized metabolic functions and to sense the need for release of their hormone products. This conduit also provides the optimal means by which these hormones are released into the bloodstream and thereby to their target tissues.
Producing paracrine signals and ECM components that support both vascular health and support optimal islet function.
Acting as a barrier against inflammation and immune cell infiltration.
In T2D (and T1D, reviewed by Mateus Gonçalves et al. 35 ), the islet microvasculature displays a variety of defects that impact all three of these physiological functions. Loss of glycemic control results in oxidative stress and inflammation that negatively impacts survival of vascular cells. In addition, the two main vascular cell types, endothelial cells and pericytes, alter expression of ECM, inflammatory, and cell–cell junctional proteins in T2D. Morphological features of the microvasculature include vessel dilation, loss of pericyte/endothelial cell contact, increased ECM deposition, fibrosis, and apoptosis of vascular cells. These morphologic and functional defects are common across tissues and organs.
Maintaining the relationship between pericytes and endothelial cells within the islet microvasculature is critical, impacting both vascular homeostasis and endocrine cell function. Understanding how islet pericytes and endothelial cells interact is important for our attempts to improve survival of grafted islets in the treatment of diabetes as well as the development of novel therapies aimed at the treatment of islet microvascular disease and thereby improving beta-cell function and survival within the endogenous pancreas.
Footnotes
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors contributed to the literature review and were responsible for drafting and/or critically editing the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Department of Veterans Affairs, VA Puget Sound Health Care System (Seattle, WA) and VA Merit Review I01-BX004063 (RLHM), by the Seattle Institute for Biomedical and Clinical Research and NIH awards R01-DK088082 (RLHM) and P30-DK017047 (University of Washington Diabetes Research Center), the Tri-Agency-funded Canada Excellence Research Chair in the Islet Microenvironment (CERC-2022-00023) (RLHM), and CIHR award PJT 192021 (YA).
