Abstract
Diabetes is not only an endocrine but also a vascular disease. Vascular defects are usually seen as consequence of diabetes. However, at the level of the pancreatic islet, vascular alterations have been described before symptom onset. Importantly, the cellular and molecular mechanisms underlying these early vascular defects have not been identified, neither how these could impact the function of islet endocrine cells. In this review, we will discuss the possibility that dysfunction of the mural cells of the microvasculature—known as pericytes—underlies vascular defects observed in islets in pre-symptomatic stages. Pericytes are crucial for vascular homeostasis throughout the body, but their physiological and pathophysiological functions in islets have only recently started to be explored. A previous study had already raised interest in the “microvascular” approach to this disease. With our increased understanding of the crucial role of the islet microvasculature for glucose homeostasis, here we will revisit the vascular aspects of islet function and how their deregulation could contribute to diabetes pathogenesis, focusing in particular on type 1 diabetes (T1D).
Keywords
Introduction
Pancreatic islets have been charmingly defined as “remarkably sophisticated microorgans.” They are responsible for coordinating the release of glucoregulatory hormones into circulation with changes in blood nutrient levels. This function is enabled by their dense network of capillaries embedded in a 3D scaffold of extracellular matrix (ECM), both essential to maintain endocrine cell mature phenotype, survival, and function. While it has been proposed that defects in the islet vasculature can lead to diabetic phenotypes, 1 it has been difficult to fully ascertain that changes in islet blood perfusion are of pathogenic importance. 2 In this article, after providing a brief overview of the islet microvasculature, we will review studies examining histopathological and functional changes associated with the islet and exocrine vasculature that occur during the development of T1D. We will discuss why close interactions between islet endocrine and vascular cells are needed for proper and timely hormone secretion, and how these may be disrupted in disease states. Our focus will be on pericytes as they are crucial for vascular homeostasis, maintaining vascular stability and integrity. Moreover, given their location at the interface between the blood and the islet parenchyma, pericytes are uniquely positioned to coordinate changes in endocrine cell activity with islet blood fl-ow, potentially modulating islet hormone delivery into circulation.
Short Overview of the Islet Microvasculature
The islet vasculature is considered a major regulator of endocrine pancreas function. 3 Interactions of endocrine cells with the vasculature start at the earliest stages of development, and blood vessels are the sources of developmental signals that induce endocrine cell differentiation. 4 Islets form adjacent to blood vessels, 5 and reducing the islet vasculature by constitutive deletion of the vascular endothelial growth factor A (VEGF-A) in beta cells leads to impaired glucose tolerance and reduced glucose-stimulated insulin secretion. 6 Islets can receive blood through one or two afferent arterioles which then give off numerous capillaries, 7 although recent work by Manami Hara and collaborators has shown that the proportion of islets with and without direct arteriole contact differs greatly between species. In particular, while in mice around 50% of islets has close contact with an arteriole, in humans only around 20% of the islets receive direct contact and arterioles supply instead entire regions of the pancreas containing islets and acinar cells. 8 These results indicate that endocrine and exocrine tissue functions may be more integrated than previously thought, particularly in humans.
Islet capillary tubes are made of a single layer of fenestrated endothelial cells, 5 which are covered by mural cells named pericytes9,10 (Fig. 1). Human 11 and mouse 12 islet capillaries are continuous with those in the acinar tissue, suggesting that islets may exert some control of exocrine tissue functions and vice versa.11,12 Interestingly, we found that the density of pericytes in islets is significantly higher than in the surrounding tissue 13 (Fig. 1). Pericytes form a well-structured network around islet capillaries that may help coordinating the function of endocrine and exocrine tissues within the same pancreas lobule.
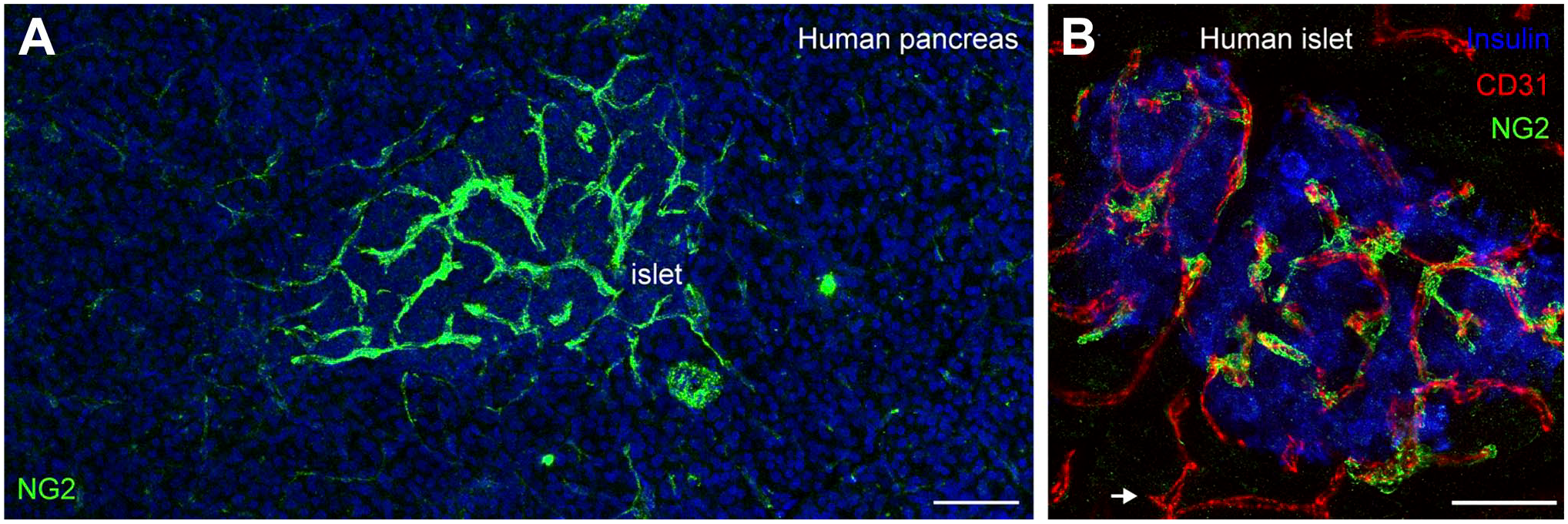
Pericytes’ density and coverage of capillaries in human pancreatic islets. (A) Maximal projection of confocal images showing pericytes (labeled with antibody against neuron-glial antigen 2; NG2; green) in a pancreatic section from a non-diabetic human donor. Islets are enriched in pericytes when compared with surrounding exocrine tissue in the pancreas. (B) Pericytes (NG2; green) associate very closely with islet endothelial cells (labeled with antibody against CD31; red). Insulin expressing beta cells are shown in blue. While islet capillaries are covered with pericytes, acinar capillaries have less pericytes (white arrow). Scale bars = 50 µm (A) and 20 µm (B).
Diabetes Is a Vascular Disease
Diabetes is a complex immune/metabolic disorder that disrupts the body’s ability to produce or respond to insulin effectively, leading to elevated blood sugar levels. Loss of glucose homeostasis triggers a cascade of events at both cellular and molecular levels, which lead to several complications particularly in the vascular system.14,15 Indeed, vascular complications are the main causes of death and disability in people with diabetes. Macrovascular manifestations include atherosclerosis and medial calcification. The microvascular consequences, such as retinopathy, nephropathy and neuropathy, are major causes of blindness, end-stage renal failure and neuronal dysfunction. 16 These vascular alterations are usually considered complications of diabetes, although there is increasing evidence that vascular dysfunction may precede a range of endocrine diseases. 17 This may be the case of the endocrine pancreas in diabetes, as it is an organ that produces glucoregulatory hormones that have systemic effects, thus requiring properly functioning blood vessels.
Anatomical and Functional Changes of the Islet Vasculature Occur During Pre-Symptomatic Stages of T1D
T1D is caused by the autoimmune destruction of insulin-producing beta cells in the pancreas. 18 Besides marked mononuclear infiltration commonly called insulitis, there are additional pathological abnormalities in endocrine compartments in T1D pancreata, in particular around islet blood vessels. Not only changes in islet blood vessel diameter, density and composition of the extracellular matrix (ECM) have been reported19–23 but also functional defects such as increased vascular leakage24–26 and abnormal blood flow dynamics are observed in pre-symptomatic stages of T1D.27,28These changes in blood flow and tissue perfusion can be detected using common methodologies for non-invasive imaging of the vasculature (e.g., magnetic resonance imaging [MRI] or ultrasound) that already help the diagnostic of different pancreatic diseases (such as pancreatitis 29 or pancreatic tumors 30 ). Because in T1D when symptoms start to develop most critical autoimmune events have already taken place, understanding what goes on during the pre-symptomatic phase could provide an ideal window for therapeutic prevention. 31
Altered Islet Blood Vessel Density and Diameter
Initial studies by Papaccio and collaborators reported a significant and progressive decrease in the density of capillaries in islets upon treatment with the beta cell toxin streptozotocin (STZ),32,33 raising interest in a “microvascular” approach to this disease. 34 Decreased islet vessel density was also recently observed in BALB/c mice treated with multiple low-doses of STZ, 35 as well as in 10-week-old non-obese diabetic (NOD) mice when compared with levels at 5 weeks. 28 Int-erestingly, the morphology of the exocrine microvasculature was preserved. 28 It was also noticed that the degree of microvascular narrowing (stenosis) was higher in islets from STZ-treated mice, which could be reversed with insulin treatment. 35
Canzano and collaborators examined endocrine and exocrine vascular networks in pancreata from organ donors with and without T1D by multiplex immunofluorescence imaging. 19 They found that the average vessel density was lower in islets from control than from T1D subjects, but islet vessels were smaller and more numerous in donors with T1D. 19 Another imaging study using mass cytometry reported that islet vascular density was significantly reduced only in the head of the pancreas in T1D donors, irrespective of the duration of T1D (from 0 to 21 years), but it was reduced in all regions of the pancreas in individuals that had recently been diagnosed (T1D for less than 2 years). 23 Other studies examining pancreata from T1D organ donors have shown that capillaries persist in islets despite loss of beta cells,36–38 indicating that changes in islet vessel density may be more variable in humans, depend on the region of the pancreas examined, as well as on the duration of the disease. Nonetheless, it would be interesting to know if these blood vessels that are still present in T1D pancreata remain functional or whether they just reflect active vascular remodeling or angiogenesis taking place.
Vascular alterations that occur in the pancreas at different stages of T1D are accompanied by changes in gene expression. Transcriptome analysis of islets excised using laser capture microdissection directly from frozen pancreatic tissue sections obtained from organ donors with and without T1D showed that several of the most upregulated gene sets were related to the vasculature and angiogenesis. 38 Using publicly available single-cell ribonucleic acid (RNA) sequencing data from human islets (from the Human Pancreas Analysis Program), 39 we recently found that the levels of several genes involved in tissue response to hypoxia, vascular remodeling and endothelin-1 signaling were significantly altered in islet cells from single Aab+ organ donors (GADA+). 37 In summary, significant remodeling and adaptations at the level of the islet microvasculature occur in T1D pancreata at early stages.
Abnormal Composition and Thickness of the Islet ECM
Changes in islet vessel density and diameter are accompanied by changes in the perivascular density of connective tissue and thickness of the basement membrane. At autopsy, human islets from single Aab+ and T1D donors demonstrate striking changes in the density of fibroblasts (Figs. 2 and 3). 37 Studies have shown that several components of the islet ECM are significantly altered in diabetes, including laminins, collagens, heparan sulfate/heparan sulfate proteoglycans, and hyaluronan, as well as the enzymes that degrade these molecules. 40 Some islets from T1D organ donors with short disease duration may even contain amyloid deposits. 41
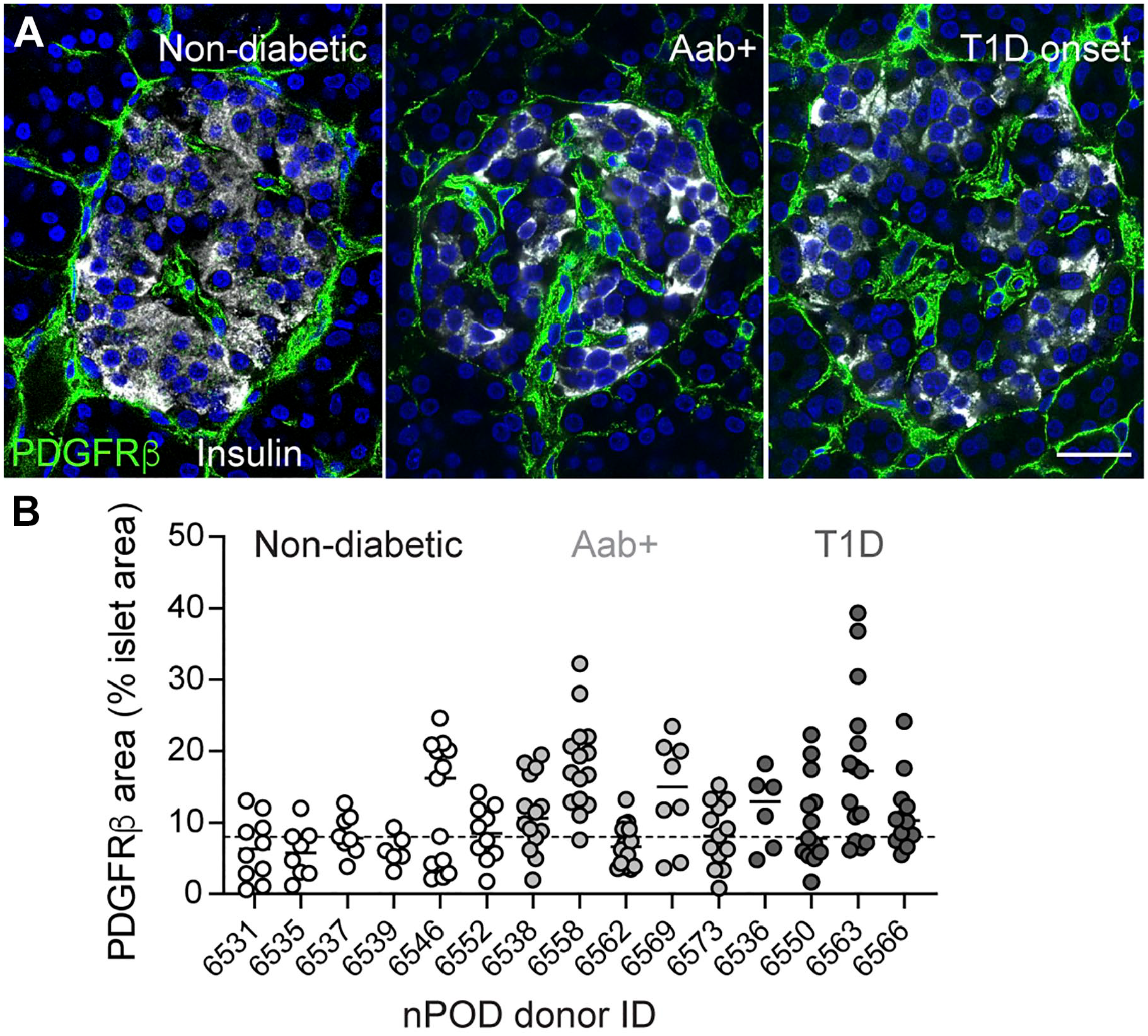
Changes in the density of PDGFRβ-positive stromal cells in human islets at different stages of T1D. (A) Confocal images of islets in pancreatic sections from age-matched non-diabetic, single autoantibody positive (GADA+; Aab+) and a type 1 diabetic donor (T1D duration = 0 years) immunostained for insulin (gray) and platelet-derived growth factor receptor beta (PDGFRβ; green). PDGFRβ is expressed by pericytes and fibroblasts (stellate cell marker). (B) Quantification of the % of islet area immunostained with PDGFRβ in different islets from different nPOD donors. Aab+ donors were all GADA+ (single Aab+) and T1D donors all had diabetes for less than 4 years. Dashed horizontal line indicates average density in islets from non-diabetic donors. Scale bar = 20 µm.
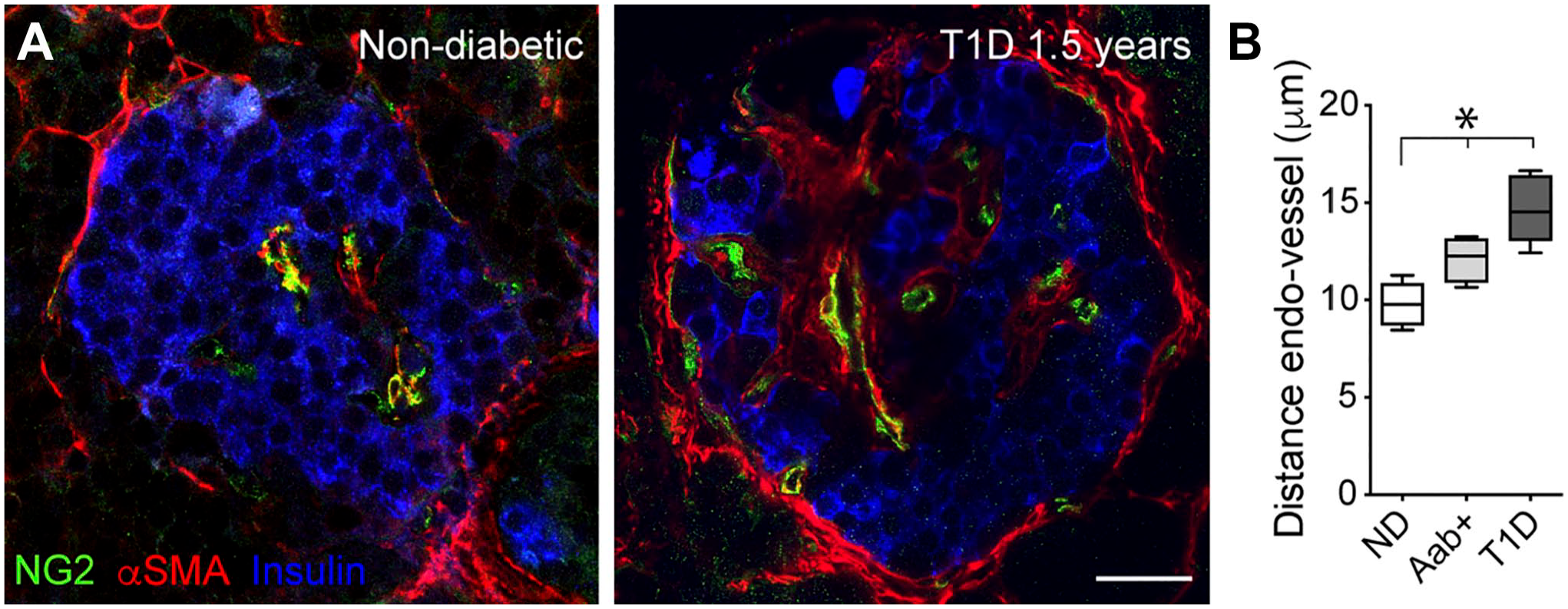
Greater separation of endocrine cells from blood vessels during the development of T1D. (A) Confocal images of islets in pancreatic sections from a non-diabetic donor and a type diabetic donor (T1D duration = 1.5 years) immunostained for insulin (blue), NG2 (green), and αSMA (red). αSMA is expressed by mural cells (pericytes and smooth muscle cells) and also by myofibroblasts (activated stellate cells). Perivascular accumulation of myofibroblasts can be seen in islets from T1D donors. (B) Increased separation of endocrine cells from blood vessel lumen in islets from non-diabetic (ND), single Aab+ and T1D donors. Aab+ donors were all GADA+ (single Aab+) and T1D donors all had diabetes for less than 4 years. Scale bar: 20 µm.
Perivascular accumulation of connective tissue and altered ECM composition can contribute to endocrine cell dysfunction in different ways: (1) by disrupting the islet cytoarchitecture,42–46 potentially interfering with intercellular communication between different endocrine cells (Fig. 2); (2) by separating endocrine cells from blood vessels (Fig. 3), potentially compromising exchanges and interfering with vascular responses42,45,47; or (3) by compromising the function of the ECM as a physical barrier for leukocyte migration into the tissue. Indeed, changes in composition of the islet ECM can directly impact inflammation and infiltration of immune cells. 40 The glycosaminoglycan hyaluronan, for instance, accumulates around the islet microvasculature early in T1D. 20 Islet hyaluronan deposits form before insulitis and coincide with sites of immune-cell infiltrates. 21 Degradation of the ECM, as well as of the glycosaminoglycan heparan sulfate, by proteinases and heparanase, respectively, is a requirement for leukocytes to migrate from the vasculature to sites in the underlying tissue. 22 Thus, immune cell infiltration is not only affected by the expression of adhesion molecules by endothelial cells (e.g., ICAM-1 48 , 49 ) but also by changes in the ECM.
Given the differences in vascular architecture, density, and composition of the ECM between mouse and human islets,50–53 some diabetes-associated histopathological changes of the ECM are not modeled well in mice. 52 Nevertheless, mouse models of T1D show alterations in the ECM at the ultrastructural level. For instance, in BALB/c mice treated with STZ, the islet-exocrine interface was noticeably widened, with collagenous fibrils proliferating around islet capillaries. In these regions, marked edemas were present. 35
Abnormal Islet Blood Perfusion
Remodeling of the islet vasculature and of the ECM should disturb islet blood flow and perfusion. Indeed, studies have shown that islet blood flow is disturbed during conditions of impaired glucose tolerance and overt diabetes, but it is not known if these disturbances are of pathogenic importance. 54 Researchers have reported both increases and decreases in islet blood flow in animal models of T1D. It seems that at early stages of deranged metabolism blood flow increases in islets but, as the disease progresses, it is then followed by a decrease in blood perfusion. 2 The initial increase in islet blood flow may result from an excessive production of nitric oxide by inducible nitric oxide synthase, as studies have shown that islet blood flow is almost 2-fold higher in STZ-treated mice, but not in animals that lack iNOS (iNOS-deficient mice). 55 Similar results have been observed in female (diabetes-prone) NOD mice. 27
More recently, the group of Richard Benninger has applied contrast-enhanced ultrasound measurements to monitor pancreatic blood flow before disease onset in different mouse models of T1D. 28 They found altered blood flow dynamics (characterized by higher basal blood flow and decreased blood volume) before disease onset, when animals were still normoglycemic but glucose intolerant. Interestingly, stronger blood perfusion alterations correlated with a faster progression to diabetes, and they were also predictive of the success of anti-CD4 therapy. 28
Alterations in vascular parameters, such as transient vasoconstriction, vasodilation, increased blood flow, and vascular leakage, are precede to inflammation, and help to orchestrate the influx of diverse cell types. 56 As a consequence of vasodilation (which in some systems may be preceded or even triggered by a transient vasoconstriction), it was suggested that the increased islet blood flow could be of crucial importance for the recruitment of inflammatory cells into the islet parenchyma during the development of T1D. 57
Increased Vascular Leakage and Insulitis
During the progression of T1D, vascular swelling and leakage that precede insulitis have been used as in-dicatives of islet inflammation. Sandler and Jansson showed in 1985 that capillaries in islets from animals treated with STZ exhibited increased vascular permeability before animals became diabetic. 58 More recently, several groups have applied non-invasive imaging techniques (e.g., MRI and ultrasound) to monitor vascular changes that accompany inflammation. Denis et al. 56 used MRI and magnetic fluorescent nanoparticles to follow islet inflammation in vivo in mouse models of T1D. Already at 4-weeks, nanoparticles extravasated from leaky vessels into both endocrine and exocrine pancreas in female BDC2.5/NOD mice. Extravasated nanoparticles were taken up by cells with a macrophage-like phenotype. Turvey et al. 24 also detected in real-time vascular leakage and its association with insulitis in 6- to 8-week-old female NOD and BDC2.5/NOD mice. Similar results were obtained in BALB/c mice treated with STZ, and considerable leakage of the probe into the islet interstitium was observed at early stages of the disease. 59
Insulitis and vascular leakage were also monitored in vivo using contrast-enhanced ultrasound combined with sub-micron nanobubbles 60 or nanodroplets (which can be vaporized) that leak out of vessels and accumulate in inflamed tissues. Probe leakage and accumulation could be detected in islets from 4-week-old NOD mice, along with mild insulitis that was already present at these very early stages. 60 Ramirez et al. 25 later reported that there was a significant correlation between the extent of leakage and the time at which diabetes arises in NOD mice: the greater the leakage the faster the onset.
MRI and magnetic nanoparticles have also used in humans to demonstrate that nanoparticles accumulated in the pancreas of individuals within 6 months of T1D diagnostic. 61 This MRI signal reflects nanoparticle leakage from the vasculature coupled with uptake by macrophages. 62 MRI studies have also shown that mean pancreatic volume index was 30% lower in T1D patients than in control subjects 61 and revealed a pronounced regional heterogeneity of the levels of pancreatic inflammation. 62 This variability of distribution of pancreatic inflammation in individual T1D subjects is consistent with the heterogeneity reported of the distribution of insulitis in NOD mice.63,64
In summary, structural and functional vascular defects in mouse and human islets can be detected before the onset of symptoms (during stages 0–2 of T1D). 31 The cellular and molecular mechanisms underlying these alterations have not yet been identified. Importantly, vessel diameter, ECM production, blood flow regulation, and vascular leakage are important functions controlled by the mural cells of the microvasculature—pericytes. Studies have shown that the morphology of islet vascular cells –pericytes and endothelial cells—is also altered during disease progression, which can then impact their function. Abundant literature exists on islet endothelial cells and their functional defects in diabetes, which is nicely reviewed by Rebecca Hull and collaborators in an accompanying article within this same collection. We know much less about the role of pericytes in islets. Below we will summarize studies examining the function of islet pericytes under physiological and pathophysiological conditions.
Pericytes’ Physiological Functions in Pancreatic Islets
The vasculature is a physical barrier between the blood and the tissue parenchyma. The ratio of pericytes to endothelial cells appears to be related to the degree of tightness of the vascular barrier: the greater the ratio, the better the endothelial barrier. 65 Pericyte-to-endothelial cell ratio varies mostly from 1:1 to 1:10, and highest ratios are usually found in tissues with high metabolic activity. In the brain and retina, pericyte-to-endothelial cell ratio is 1:1, and pericytes are important components of the blood-brain barrier and blood-retina barrier. 66 In the lungs, pericyte-to-endothelial cell is 1:7–1:9 and pericytes are involved in optimal gas exchanges. 66 In the kidney, the ratio is 1:2.5, and changes in renal vascular permeability amplify renal structural and functional disorders. 67 We have estimated pericyte-to-endothelial cell ratio in mouse and human islets to be between 1:3 and 1:2, respectively.9,52 These data suggest that the islet endothelial barrier has an intermediate leakiness 1 : not as tight as the microvasculature of the retina or brain but tighter than that of the lungs.
Pericytes have been extensively studied in the brain given their crucial role for maintaining the blood–brain barrier. 68 Pericytes provide structural support and control key microvascular functions such as angiogenesis, transendothelial permeability, and blood flow. 69 They are also capable of synthetizing most of the structural and adhesive proteins associated with the ECM. 65 Recently, they have also been shown to be capable of modulating of inflammation and the local immune cell repertoire in different tissues, including the islet. 70 Below we will summarize studies that have examined the physiological functions of islet pericytes. Most of them have focused on the trophic support they provide for beta cells but being vascular cells, they may have more dynamic effects and affect endocrine cell secretory capacity indirectly by improving islet vascular function and regulating blood flow.
Pericytes as Source of Trophic Factors for Beta Cells
Seminal studies by Limor Landsman and coworkers have shown that pericytes are not only part of the islet microenvironment but they are crucial for beta cell proliferation, maturity and function.10,71,72 After birth, pericytes constitute a major part of the mesenchymal cell population in the pancreas, 71 which have been shown to support the proliferation of pancreatic progenitors and of differentiated beta-cells. 71 Mouse islets incubated with pericyte-conditioned medium showed a doubling in the number of beta cells, indicating that factors secreted by cultured pancreatic pericytes stimulate proliferation of primary adult beta-cells. 71 Interestingly, blocking β1 integrin signaling inhibited the stimulatory effects of pericyte-conditioned medium on beta cell expansion, further supporting the essential role of the ECM for beta cell proliferation. 73
Pancreas pericytes are the source of trophic factors that are important for beta cell function and insulin se-cretion. Accordingly, systemic (diphtheria toxin-based) depletion of pericytes in adult mice leads to glucose intolerance, impaired glucose-stimulated insulin secretion, and reduced expression of genes associated with a mature beta-cell phenotype. 10 One pericyte-secreted ligand is bone morphogenetic protein 4 (BMP4), which has been shown to support beta cell function, in a process that depends on the transcription factor Tcf7l2. 72 BMP4 starts to be produced by pancreatic pericytes at the middle of the postnatal period, contributing to the functional maturation of beta cells that progressively occurs in both humans and mice. 74 Pericytes are also capable of synthetizing different components of the islet vascular basement membrane. 75 Given that the composition of the islet vascular niche greatly affects beta cell differentiated status and function, 73 it is possible that some of the trophic effects of pericytes on beta cells may underlie some changes in the ECM.
Pericytes are also the source of nerve growth factor (NGF) which is an important trophic factor for nerves. 76 A study by Houtz and collaborators showed that NGF release by pericytes rapidly increased upon elevated glucose concentrations. NGF-activated TrkA receptors expressed by islet beta cells, promoting remodeling of the actin cytoskeleton and insulin granule exocytosis. 76 In summary, during development and through adulthood, islet pericytes are crucial for beta cell proliferation, maintenance of a mature phenotype and for proper function. It is thus clear that pericytes are pivotal components of the islet niche, and in vitro strategies to make functional islets should take this important player into account.
Pericyte-Mediated Control of Islet Blood Vessel Diameter and Flow
Pericytes have been shown to regulate blood flow in different tissues, including the kidney, 77 the brain,78,79 and the heart. 80 In these tissues, pericytes act as local gates that manage blood flow distribution to meet the metabolic demands of that tissue. In the brain, for instance, capillary dilation occurs before arteriole dilation, and it occurs in regions in the vessel where pericytes are located. 81 Contractile pericytes are strategically placed at capillary junctions and changes in their activity direct blood flow to specific tissue regions. 82 Pericytes respond to molecules derived from endothelial cells and astrocytes (e.g., metabolites of arachidonic acid), mediating the functional hyperemic response: the increase in blood flow that occurs upon neuronal activation. 83
Do Pericytes Have Similar Functions in Islets and Also Participate in the Regulation of Islet Blood Flow?
To be able to regulate blood flow, islet pericytes must express contractile proteins such as alpha smooth muscle actin (αSMA), which is a major constituent of the contractile apparatus in mural cells. 84 We found that a subset of pericytes in mouse and human islets expresses αSMA9,85 (Fig. 3), being potentially contractile. Also, in the brain, expression of αSMA is limited to a subpopulation of pericytes in the post-arteriole transitional region. 82 Several other studies also found SMA-expressing cells covering capillaries in mouse and human islets.10,86,87 Capillaries in human islets may even contain more smooth muscle cells than mouse islet capillaries. 2 αSMA levels expressed by islet pericytes are much lower than those present in smooth muscle cells that wrap islet feeding arterioles. 9
Importantly, these contractile (αSMA-positive) pericytes are innervated by sympathetic nerve fibers that reach mouse and human islets,13,87 and respond to sympathetic agonists. Using living pancreas slices from mice and non-diabetic human donors, we observed that sympathetic adrenergic input (exogenous [e.g., norepinephrine or phenylephrine administration] and endogenous [using tyramine, which is a sympathomimetic that stimulates the release of endogenous norepinephrine] 88 ) increases pericyte cytosolic calcium levels ([Ca2+]i) and reduces capillary diameter and local blood flow.9,13,85 By contrast, activating beta cells by increasing extracellular glucose concentration decreases [Ca2+]i in pericytes, dilating islet capillaries, and increasing local blood flow.9,37 We concluded that pericytes control islet capillary diameter and blood flow under different metabolic states: upon activation of beta cells with high glucose, pericytes relax and blood vessels dilate, allowing an increase in blood flow in the islet; upon activation of the sympathetic nervous system, pericyte [Ca2+]i levels increase and blood vessels constrict, leading to a decrease in local blood flow. 9
But Do These Changes in Local Blood Flow Affect Plasma Levels of Islet Hormones and Glucose Metabolism?
Most of what we know about the impact of islet blood flow on glucose metabolism is based on chronic ablation of islet blood vessels or on systemic manipulations that can create indirect effects (e.g., intravenous administration of angiotensin II or angiotensin receptor blocker). 89 The impact on glucose metabolism of changing blood flow specifically in the islet had not been addressed. To specifically manipulate blood flow in the islet, we used optogenetics to alter pericyte activity as these are excitable cells,90,91 in combination with islet transplantation into the anterior chamber of the mouse eye.13,92 We found that activation of pericytes with blue light in intraocular islet grafts contracted capillaries and diminished blood flow. These alterations had metabolic consequences: pericyte activation decreased insulin and increased glucagon plasma levels, producing hyperglycemic effects. Our findings thus indicate that pericytes mediate vascular responses in the islet that are required for adequate hormone secretion and glucose homeostasis. 13
We then assessed whether this same mechanism is used by the sympathetic nervous system to control islet hormone secretion and glucose metabolism. To mimic sympathetic nervous input, we used phenylephrine which is a specific agonist of α1-adrenergic receptors.9,13 Phenylephrine, administered as eye drops, increased [Ca2+]i levels in pericytes in intraocular grafts and decreased islet blood flow. Importantly, these changes in flow were linked to a decrease in plasma insulin levels and an increase in glycemia. These metabolic outcomes are similar to the effects that activating the sympathetic nervous system produces in humans. 88 Impaired glucose homeostasis was also observed in an elegant study by Sarah Stanley and collaborators where specific activation of pancreatic efferent sympathetic neurons using chemogenetics and adeno-associated viruses led to glucose intolerance. 93
Interestingly, interfering with sympathetic nervous input to the pancreas delays the onset of T1D.94,95 Gustaf Christofferson and Matthias Von Herrath observed that in a mouse model of T1D (expression of lymphocytic choriomeningitis virus–glycoprotein (RIP-LCMV-GP) under rat insulin promoter) inhibiting α1-adrenergic receptors or ablating sympathetic nerves chemically or surgically delayed the onset of diabetes. 94 The development of hyperglycemia in STZ-treated mice was almost completely prevented by treating the animals with the α1-adrenoreceptor antagonist prazosin. 95 Manipulating sympathetic input could have directly affected immune cell infiltration into the pancreas, as indeed most cell types in both innate and adaptive immune systems express adrenergic receptors, 96 or indirectly by affecting blood flow in the pancreas which in turn can affect immune cell infiltration. 57
In summary, these data indicate that pericyte-mediated control of islet blood flow impacts glucose metabolism. 13 Sympathetic nerves may act on pericytes to change blood flow, affecting the hormonal output of islets and impacting immune cell infiltration both direct and indirectly.
Immunoregulatory Functions of Pericytes
Besides controlling transendothelial permeability, pericytes have also been shown to actively participate in immunosurveillance in different tissues. 97 Studies have shown that islet endothelial cells express costimulatory markers and may function as antigen-presenting cells and mice and humans.98,99 Like endothelial cells, in response to inflammatory mediators, pericytes in different tissues can also express adhesion molecules and chemoattractant factors that control leukocyte migration in vivo.100,101
Pericytes in the pancreas may also have similar functions. Burganova and collaborators have shown that they are the primary source of interleukin-33 (IL-33). 70 Importantly, knocking out pericytic IL-33 not only turned male (but not female) mice glucose intolerant due to impaired insulin secretion, but it also reduced the number of T lymphocytes and dendritic cells in islets. 70 Future studies investigating the role of pericytes as active components/mediators of immune responses in islets will be needed to decipher their role in shaping islet inflammation and its impact on glucose homeostasis.
Pathophysiological Changes Associated With Pericytes and Blood Flow Control
Pericytes have important physiological functions in islets as described above. Importantly, pericyte coverage of islet capillaries drops drastically in individuals with long-standing T2D (for more than 5 years; pericyte-to-endothelial cell ratio drops from ~30% to <10%). 9 In T1D, we also a found a decrease in the pericyte-to-endothelial cell ratio, and several capillaries in T1D islets lacked pericytes or were covered by pericytes with an abnormal morphology. 37 These data suggest that, under diabetic conditions, islets may lose a source of trophic factors that maintain beta cell mature phenotype and function, as well as the capacity to control their own blood flow. These changes can lead to inadequate plasma insulin levels, further deteriorating glycemic control. However, because longitudinal studies are not possible in humans, it is still not known what happens to islet pericytes during the development of diabetes. Moreover, important aspects of the vasculature and the local microenvironment of the human islet are not modeled well in mice.50–53 It has thus been difficult to study what happens to islet pericytes during diabetes progression. We would like, in particular, to understand whether pericytes contribute to the increased density of connective tissue and abnormal ECM present in islets in diabetes?
Pericytes have enormous plasticity due to their pluripotent and postnatal undifferentiated nature. 102 Studies have shown that they can adopt various phenotypes and transform, for instance, into myofibroblasts in fibrotic diseases in different tissues.103–108 However, regarding the pancreas, studies had focused on pancreatic stellate cells as they have been shown to play a role in pancreatic fibrosis. 109 Indeed, upon activation by inflammatory cytokines 110 and oxidative stress, 111 stellate cells can switch from a quiescent state 112 and transform into αSMA-positive myofibroblast-like cells with an increased capacity to synthetize ECM components (collagens, proteoglycans, structural glycoproteins and hyaluran). Stellate cells are always found in single-cell RNAseq analysis of human pancreatic islets. They can be identified by expression of their marker gene PDGFRB 113 and COL1A1. 37 Also based on their gene profiles, stellate cells can be subdivided into quiescent, standard activated and immune-activated, and, interestingly, pericytes are included within the quiescent stellate cell population. 113 We have recently shown that a subcluster of islet stellate cells expresses pericyte identity genes (e.g., CSPG4, PDGFRB, RGS5, and ACTA2) 37 and the pericyte markers neuron-glial antigen 2 (NG2) and PDGFRβ. 13,114 Mesenchymal stem cells derived from human islets express different pericyte markers such as CD146, NG2, αSMA, and PDGRFβ, as well as mesenchymal cell markers (e.g., CD105 and CD-90). 115 These studies support that a mesenchymal stellate cell-like population resides in islets and pericytes may be among them.
Pericytes as Activated Stellate Cells or Myofibroblasts
Many cells in the pancreas would be vulnerable to pro-inflammatory stressful conditions, but vascular cells (endothelial, smooth muscle cells/pericytes, and adventitial fibroblasts) exhibit remarkable sensitivity to these stressors. 116 Pericytes, in particular, seem to be highly vulnerable to injury due to the effects of multiple metabolic toxicities including excess of reactive oxygen species (ROS), pancreatic inflammation and glucose toxicity. 117 Studies have shown that a metabolic unstable environment, rich in pro-inflammatory cytokines, is present in the pancreas before disease onset, which could contribute to an “activation” of islet pericytes as described for pancreatic stellate cells.109,118 Indeed, although diabetes is characterized by chronic hyperglycemia, blood glucose levels are already above normal in prediabetes, although still below diabetes thresholds. 119 Interestingly, intermittent high glucose, but not constant elevated glucose, increases the expression by retinal pericytes of pro-inflammatory cytokines (such as macrophage chemoattractant protein 1 [MCP-1]) as well as of key mediators of ER stress and cell death (e.g., ATF4 and CHOP). 120 Pericytes could thus be involved in the inflammatory process that is present in the exocrine pancreas early in T1D. Indeed, tissue inflammation is among the abnormalities described in the exocrine pancreas when autoantibodies are already detectable but hyperglycemia is not. 121 Additional alterations include interstitial fibrosis, adipogenesis, atherosclerosis and acinar loss with atrophy in diabetes. 117 In a case report that examined the pancreas from a single Aab+ individual (GADA+), there was abundant inflammation in the exocrine pancreas (not so much in endocrine compartment) with areas of chronic pancreatitis and acinar to ductal metaplasia, and with other areas of atrophy and fatty infiltration. 122 The origin of this exocrine inflammation in T1D is still not clear, neither pericytes’ role in it.
“Injured pericytes” are also present in islets in mouse models of T1D, as indicated by their swollen mitochondria and dilated rough endoplasmic reticulum. 35 Abnormal shape and foot process loss are additional features of pericytes in islets from diabetic mice. 35 Altered pericyte morphology and vascular remodeling are also observed in BALB/c mice treated with STZ and in NOD mice at 7–8 weeks. 123 Remodeling includes an increase in pericyte density and accumulation on the walls of the feeding arteriole at lesioned sites, with marked increase in NG2 surface expression. 123 Also in different rodent models of T2D, abnormal pericyte density and shape have been observed. In ob/ob and db/db mice, pericytes ensheathing dilated microvessels exhibit hypertrophic processes. 124 Nakamura et al. 125 also noticed in db/db islets hypertrophy of pericytes and an abundance of actin-like microfilaments in their cytoplasm. Beautiful transmission electron microscopy studies by Hayden and coworkers have shown that pericytes at the islet-exocrine interface undergo proliferation, migration, and transformation into αSMA-positive profibrotic myofibroblasts in rodent models of hyperinsulinemia, insulin resistance and hypertension.126,127 Hypertrophied pericytes associate closely with collagen fibrils, 47 as well as with perivascular amyloid aggregates in transgenic mice expressing human islet amyloid polypeptide when exposed to a high fat diet. 128
Studies described above are suggestive of a potential change in the phenotype of islet pericytes and of their involvement in the excessive production of ECM. To demonstrate that islet pericytes could indeed turn into myofibroblasts, we used a transgenic mouse model of beta cell expansion and chronic hyperinsulinemia (AktTg mice), 129 which also exhibits increased deposition of ECM in perivascular regions in islets. 52 We examined the islet microvasculature in these animals and found that not only islet pericytes proliferated extensively, but they also switched their phenotype and turned into profibrotic myofibroblasts. 52 Indeed, by crossing them with transgenic mice that express a fluorescent reporter in mural cells in peripheral tissues (using the NG2/Cspg4 promoter), we found pericyte de novo expression of myofibroblast markers (e.g., periostin) and enhanced production of ECM proteins fibronectin and collagen type I. 52 Changes in the islet pericyte phenotype and ECM accumulation were associated with diminished islet blood perfusion and impaired capillary responses to vasoactive stimuli. These vascular alterations had metabolic consequences: when corrected for the increased beta cell mass, in vivo glucose-stimulated insulin secretion was impaired in AktTg mice.52,130
Impaired Islet Vascular Responses Associated With an Abnormal Pericyte Phenotype
Given their important physiological functions in islets, alterations in pericyte morphology and phenotype under pathophysiological conditions can have functional consequences. In a mouse model of neuroendocrine tumors (Men1 mice), 131 where pericyte remodeling and changes in their density resemble those described in T1D, 123 impaired vascular responses of islet and tumor arterioles were observed. 131 These included diminished vasodilation in response to elevated glucose levels and impaired glucose-induced increase in blood flow, while vascular responses to nitric oxide synthase inhibitor L-NAME were preserved.
In a recent study using nPOD tissue, we compared the phenotype and function of pericytes in islets from non-diabetic organ donors with those in individuals at different stages of T1D: single Aab+ (GADA+; “stage 0”) and T1D donors with relatively short disease duration (from 0 to 4 years; stage 3). Using living pancreas slices to monitor pericyte [Ca2+]i levels and vasomotion ex vivo, we found that islet pericyte and capillary responses to elevated glucose and norepinephrine were significantly impaired in islets from single Aab+ and T1D donors. 37 A potential switch in the phenotype of islet pericytes toward myofibroblasts could have been responsible for these functional defects as αSMA-positive cells, more abundant in islets from Aab+ and T1D donors, expressed pericyte markers PDGFRβ, CD146, and NG2 (Fig. 4).
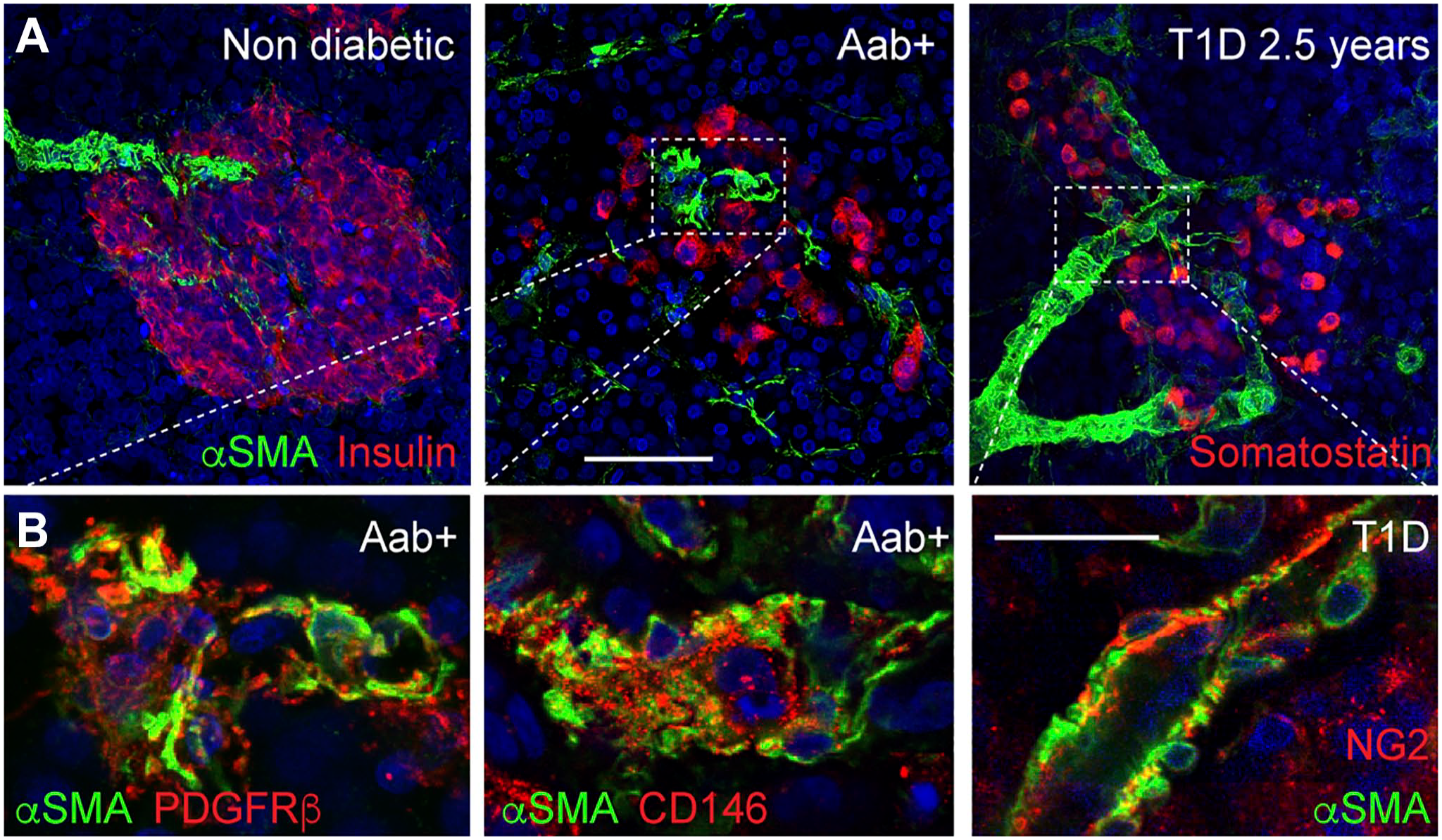
Myofibroblasts (or activated stellate cells) in islets from Aab+ and T1D donors express pericyte markers. (A) Maximal projections of confocal images of islets in fixed pancreas slices from a non-diabetic (ND), a single Aab+ (GADA+) and a T1D organ donor (T1D duration: 2.5 years) immunostained for insulin (ND, Aab+; red) or somatostatin (T1D; red) and αSMA (green). αSMA is expressed by mural cells (pericytes and smooth muscle cells) and by myofibroblasts (activated stellate cells). (B) Zoomed images of regions within rectangles in (A) showing colocalization of alphaSMA with pericyte markers PDGFRbeta, CD146 and NG2. The shape of αSMA-expressing cells is different in islets from Aab+ and T1D donors, and these cells also express other pericyte markers such as PDGFRβ, CD146 and NG2. 13 These data suggest that islet pericytes may have contributed to this cell population, as previously suggested by us. 37
In summary, pericytes are crucial for microvascular homeostasis throughout the body. 132 Their loss or dysfunction may compromise pericytes’ vascular-stabilizing properties. 133 This results in vulnerable capillaries, prone to instability, increased leakage which, ultimately, may facilitate immune cell infiltration. Early islet pericyte dysfunction could thus underlie the vascular abnormalities that occur in the pancreas during the development of diabetes.
Pericytes as Potential Targets of SARS-CoV-2 Viruses
Pericytes have received additional attention during the COVID-19 pandemic as in different tissues they express the host cell receptor for SARS-CoV—angiotensin converting enzyme 2 or ACE2—as well as proteases capable of cleaving the viral envelope proteins and mediate fusion.134–137 Pericyte-like cells integrated into cortical organoids can be infected with SARS-CoV-2 and served as viral replication hubs. 138 In the pancreas, the cellular distribution of ACE2 has been a matter of debate. While numerous reports show that ACE2 is expressed by islet endocrine cells,139,140 other investigators have reported that ACE2 is restricted to the microvasculature in islet and acinar regions of the human pancreas.134,141,142 In collaboration with the group of Alvin Powers and Marcela Brissova, we showed that a subset of pericytes in human islets expresses ACE2, making them susceptible for infection by SARS-CoV-2. 134 In future studies, we will determine whether microvascular/pericyte dysfunction in the pancreas could contribute to the transient loss of glucose homeostasis reported upon infection by SARS-CoV coronaviruses.
Conclusion
This review article places vascular pericytes at the center of diabetes pathophysiology. The endocrine pancreas is an organ that requires properly functioning blood vessels, as it produces glucoregulatory hormones that have systemic effects. Pericytes are integral components of the islet microvasculature. Studies from different laboratories have shown that these mural cells have important physiological functions in islets: not only they contribute for maintaining beta cell mature phenotype and function, but they also participate in ECM synthesis, regulate islet blood perfusion and have immunoregulatory functions. In addition, a subset of pericytes in human islets expresses the host cell receptor for SARS-CoV-2, ACE2, making them susceptible for infection by these viruses. In this scenario, we hypothesize that pericyte dysfunction could be an initial trigger for microvascular pathology that has been described in islets and exocrine tissue at early stages of disturbed glucose homeostasis. Abnormal pericyte function would compromise endothelial cell activity, leading to uncontrolled inflammatory responses, and impacting endocrine cell function in different ways. We believe that a deeper understanding of the mechanisms required to maintain a functional pancreatic vasculature and efficient interactions between pericytes, endocrine cells, and immune cells is necessary to fully comprehend the pathogenesis of diabetes.
Footnotes
Acknowledgements
The authors thank Dr. Ruy Andrade Louzada (University of Miami) for careful revision and discussion of the manuscript. The authors thank the Network for Pancreatic Organ Donors with Diabetes (nPOD), in particular the organ donors, their families, and the nPOD slicing team for producing living pancreas slices that allow the scientific community to conduct functional studies in humans.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors discussed the content and reviewed the article. JA wrote the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by NIH grants K01DK111757 and R01DK133483 (to J.A.), by NIDDK-supported Human Islet Research Network (HIRN, RRID: SCR_014393; ![]() ; UC4 DK104162) New Investigator Pilot Award (to J.A.), and by the Helmsley Charitable Trust for nPOD team science (2018PG-T1D060 and 2112-04895).
; UC4 DK104162) New Investigator Pilot Award (to J.A.), and by the Helmsley Charitable Trust for nPOD team science (2018PG-T1D060 and 2112-04895).
