Abstract
Dystroglycan is a cell surface protein which, in muscle, links the extracellular matrix protein laminin-2 to the intracellular cytoskeleton. Dystroglycan also binds laminin-1 and the binding occurs via the E3 fragment of laminin-1. Recently, it was found that dystroglycan is expressed in developing epithelial cells of the kidney. Moreover, antibodies against dystroglycan can perturb epithelial development in kidney organ culture. Therefore, dystroglycan may be an important receptor for cell–matrix interactions in non-muscle tissues. However, information about the tissue distribution of dystroglycan is limited, especially in adult tissues. Here we show that dystroglycan is present in epithelial cells in several non-muscle organs of adult mice. Dystroglycan is enriched towards the basal side of the epithelial cells that are in close contact with basement membranes. We suggest that dystroglycan is involved in linking basement membranes to epithelial and muscle cells. Dystroglycan may be important for the maintenance of tissue integrity.
I
Several proteins of the DGC are muscle-specific, but Northern blot analyses have shown that dystroglycan is expressed in a wide variety of non-muscle tissues (Ibraghimov–Beskrovnaya et al. 1992,1993). Moreover, dystroglycan also binds laminin-1 (Ibraghi-mov–Beskrovnaya et al. 1992), which is not expressed in muscle (Schuler and Sorokin 1995). Binding of dystroglycan to laminin-1 appears to occur via the E3 fragment of laminin-1 (Gee et al. 1993; Brancaccio et al. 1995), and this fragment has been shown to be involved in branching epithelial morphogenesis (Klein et al. 1988; Sorokin et al. 1992; Kadoya et al. 1995; Streuli et al. 1995). Hence, dystroglycan might play important roles during embryonic development of non-muscle tissues (Ekblom 1996). Evidence for this proposal is available from studies on the embryonic kidney. Dystroglycan was found to be expressed in the basement membrane region of developing epithelia, and antibodies against dystroglycan perturbed kidney development in vitro (Durbeej et al. 1995).
It is therefore obvious that dystroglycan may be an important receptor for cell–matrix interactions in non-muscle tissues. A targeted disruption of the dystroglycan gene in mice resulted in early embryonic lethality. In dystroglycan null mice, Reichert's membrane is disrupted, suggesting a role for dystroglycan in basement membrane assembly (Williamson et al. 1997). However, information about the tissue distribution of dystroglycan is limited. Although some results are available for embryonic tissues (Durbeej et al. 1995; Schofield et al. 1995; Yotsumoto et al. 1996; Williamson et al. 1997), very little is known about the distribution in normal adult tissues. Dystroglycan mRNA is abundant in many adult tissues (Ibraghimov–Beskrovnaya et al. 1992,1993), although it is possible that this signal is derived from smooth muscle present in these tissues. Therefore, a main goal of this study was to identify the cell types that express dystroglycan.
Materials and Methods
Immunofluorescence
Mouse tissues from adult male C57 bl/6J hybrid mice and mammary glands from pregnant NMRI hybrid mice were frozen in Tissue Tek. The pregnant uteri were isolated from C57bl/129Sv hybrid mice, fixed in 4% paraformaldehyde, and embedded in paraffin. Human kidney biopsy specimens were kindly provided by Dr. Erik Larsson at the Department of Pathology, Uppsala University, Sweden. Sections of 5-day excisional wounds of mice were kindly provided by Dr. Sabine Werner at Max-Planck Institute for Biochemistry, Martinsried, Germany. Cryostat sections of 8 μm (unfixed, fixed in 4% paraformaldehyde, or fixed in methanol) and paraffin sections of 7 μm were washed in PBS and blocked in 3% BSA in PBS for 10 min. The sections were subsequently incubated with the following antibodies: affinity-purified sheep polyclonal antibody specific for fusion protein B (anti FP-B), which recognizes both α- and β-dystroglycan (Ibraghimov–Beskrovnaya et al. 1992) and AP83, which is an affinity-purified rabbit polyclonal antibody raised against the C-terminal 15 amino acids of β-dystroglycan (Ervasti and Campbell 1991; Williamson et al. 1997). This antibody specifically detected 43-kD β-dystroglycan in Western blot analysis (see Results). Stainings with anti FP-B and AP83 gave similar results in all tissues analyzed (except for kidney and small intestine; see Results). Furthermore, we used rabbit antiserum ISH-1 raised against EHS laminin reacting equally well with all three chains of mouse laminin-1 (Klein et al. 1988); guinea pig polyclonal antibody directed against human placental laminin (isoform specificity of this antibody is unknown) (Durham and Snyder 1995); rat monoclonal antibody (MAb) 200 against the E3 domain of laminin α1-chain (Sorokin et al. 1992); rat MAb 8G11D10 against laminin α2-chain (Schuler and Sorokin 1995); and rat MAb GoH3 recognizing the α6-integrin subunit (Sonnenberg et al. 1990). Bound antibodies were visualized using secondary antibodies conjugated with FITC or Cy3 (Jackson ImmunoResearch Laboratories; West Grove, PA). Slides were examined under a Zeiss microscope equipped with epifluorescence optics.

Western blot analysis of β-dystroglycan expression in various mouse tissues. Five hundred μg (Lanes 1–4) or 250 μg (Lane 5) of total microsomes prepared from adult mouse kidney (Lane 1), liver (Lane 2), stomach (Lane 3), uterus (Lane 4), and skeletal muscle (Lane 5) were separated on a 3–12% linear SDS-PAGE gel and transferred to PVDF membranes. Filters were probed with AP83 to detect β-dystroglycan. Molecular mass markers in kD are indicated to the right.
In Situ Hybridization
In situ hybridization was performed using the previously described deoxyribonucleotide probe, a 45-mer against dystroglycan mRNA (Durbeej et al. 1995). Control sections hybridized with the same amount of labeled probe plus unlabeled probe in excess did not show any specific binding.
Western Blot Analysis
Total microsomes were prepared from various adult tissues in the presence of the following protease inhibitors: aprotinin (1 μM), leupeptin (1 μM), pepstatin A (1 μM), benzamidine (1 mM), and PMSF (1 mM) (Ervasti and Campbell 1991). Microsomes were separated on a 3–12% linear gradient SDS-polyacrylamide gel and transferred to PVDF membranes (Millipore; Bedford, MA). The membranes were probed with AP83 to detect β-dystroglycan. Bound antibodies were vizualized using 4-chloro-1-naphthol and H2O2.
Results
β-Dystroglycan Expression in Adult Mouse Tissues
By immunoblotting with antibody AP83 raised against β-dystroglycan, we detected a 43-kD band representing β-dystroglycan in all adult tissues examined (Figure 1). The lower molecular weight band at around 29 kD seen in Figure 1 (Lane 3) is a breakdown product containing the C-terminus of β-dystroglycan. This was shown by using other C-terminal dystroglycan antibodies (data not shown).
Expression of Dystroglycan in Skeletal Muscle
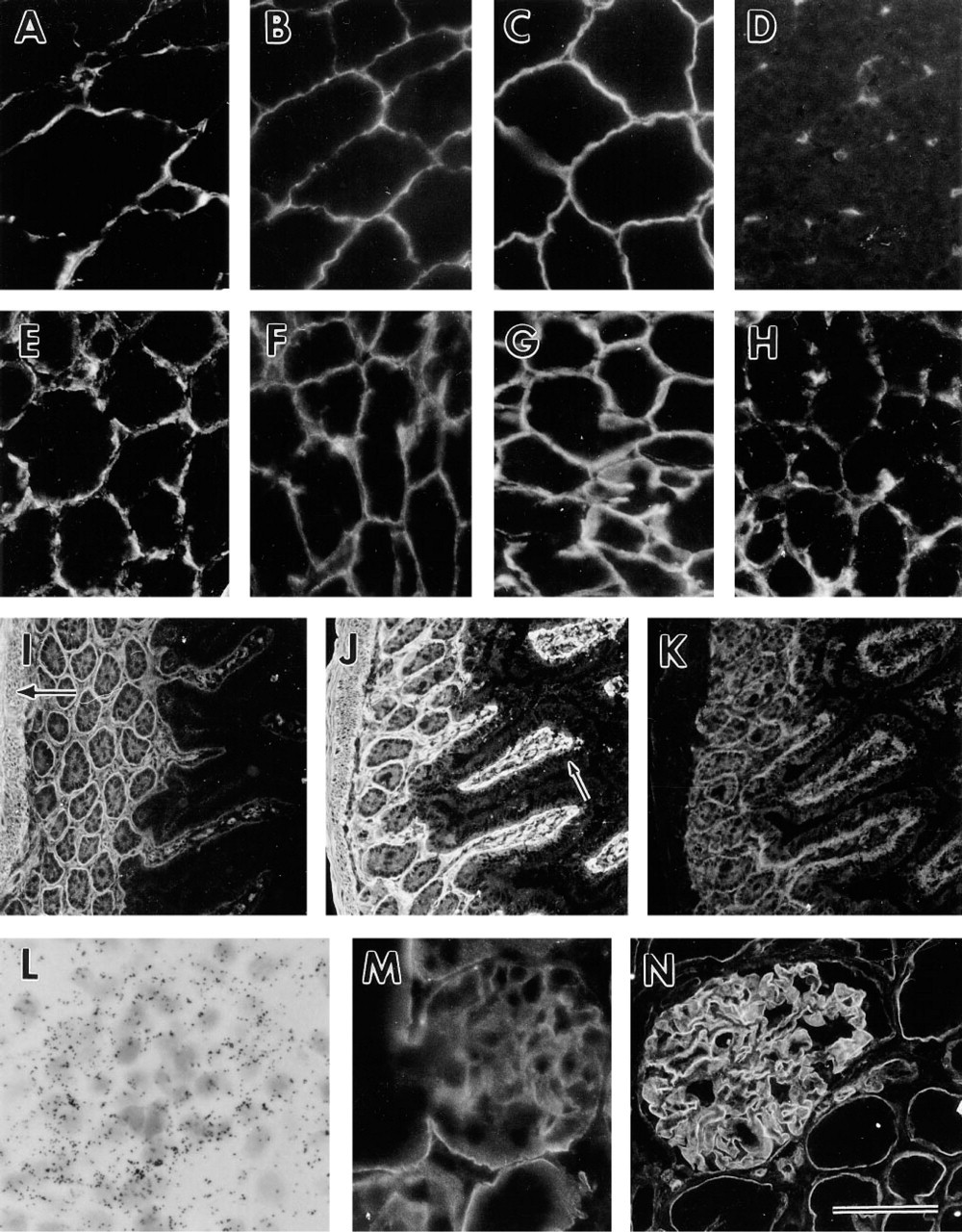
The antibodies to detect α/β-dystroglycan (anti FP-B) and β-dystroglycan (AP83) have been described earlier (see Materials and Methods). As expected, they both stained the sarcolemmal membrane (Figures 2A and 2B). In muscle, dystroglycan is also localized to the neuromuscular and myotendinous junctions (Ohlendieck et al. 1991). The antibody 8G11D10 against the laminin α2-chain has been described previously (Schuler and Sorokin 1995) and reacted with the sarcolemmal membrane (Figure 2C). The antibody GoH3 against the integrin α6-subunit, (Sonnenberg et al. 1990) did not stain the sarcolemmal membrane but did stain the endothelial cells of capillaries between the muscle fibers (Figure 2D).
Expression of Dystroglycan in the Digestive Tract
We then analyzed dystroglycan expression in pancreas, small intestine, liver, trachea, kidney, mammary gland, testis, uterus, and skin with anti FP-B and AP83. In all tissues, except where indicated, the two antibodies gave similar results. However, only pictures of anti FP-B staining are shown.
Expression of Dystroglycan in Trachea
In the trachea, α/β-dystroglycan was expressed in the basement membrane region of the surface epithelium as well as in the smooth muscle. The laminin α2-chain was co-expressed with dystroglycan, whereas no laminin α1-chain could be seen. Strong staining of the integrin α6-subunit was seen on the basal side of the epithelium. Both α/β-dystroglycan and the integrin α6-sub-unit were detected over the entire cell surface of the surface epithelium (data not shown).
Expression of Dystroglycan in Kidney
In the embryonic kidney, expression of dystroglycan has previously been shown. Dystroglycan mRNA is produced by epithelial cells of the ureter, developing tubules, and developing glomeruli, and α/β-dystroglycan is localized at the basal side facing the basement membrane of these epithelial cells (Durbeej et al. 1995). In adult mouse kidney, dystroglycan mRNA was expressed in epithelial podocytes (Figure 2L). By using anti-FP-B antibodies reacting with both α- and β-dystroglycan, the strongest staining in the kidney was seen in the glomerular basement membrane. α/β-Dystroglycan was also expressed in Bowman's capsule and in the basement membrane region of the proximal and distal tubules (Figure 2M). The polypeptides could not be detected in the basement membrane region of the collecting ducts (data not shown). Antibody AP83 reacted only with the glomerular basement membrane and not with the tubule basement membranes in adult mouse kidney (data not shown). By using FP-B antibodies on sections of adult human kidneys, a similar staining was observed as in adult mouse kidneys, i.e., staining of the glomerular basement membrane and the basement membrane region underlying proximal and distal tubule cells (Figure 2N). Antibody AP83 against β-dystroglycan reacted only with the glomerular basement membrane in adult human kidney (data not shown).
Expression of Dystroglycan in Reproductive Organs
Expression of Dystroglycan in Skin
α/β-Dystroglycan was expressed pericellularly in all epidermal layers of the skin except for the stratum corneum (Figure 3K). This is in contrast to the integrin α6-subunit, which was present pericellularly only in the most basal cells resting on the basement membrane (Figure 3L). Weak staining of α/β-dystroglycan could also be detected on the basal side of the cells resting on the basement membrane (Figure 3K). The integrin α6-subunit, on the other hand, was strongly expressed at the basal cell surface in contact with the basement membrane (Figure 3L). The difference in dystroglycan and integrin α6 expression was also illustrated in a 5-day mouse excisional wound. Dystroglycan was expressed pericellularly in the proliferating epidermal keratinocytes (Figure 3M), whereas the integrin α6-subunit was located pericellularly in the most basal cells and was also enriched at the basal cell surface (Figure 3N). Dystroglycan was also located over the entire cell surface, with an enrichment towards the basal side of epithelial cells in sweat glands and hair follicles (data not shown).
Discussion
Dystroglycan is a novel laminin binding protein involved in muscle cell attachment to the extracellular matrix (Henry and Campbell 1996). Dystroglycan might also be important for kidney epithelial morphogenesis in vitro (Durbeej et al. 1995). Because dystroglycan mRNA is expressed in various adult and fetal tissues (Ibraghimov–Beskrovnaya et al. 1992,1993), dystroglycan function may not be restricted to muscle and kidney. Dystroglycan null mice die at Day 6.5 of embryonic development. In these mice, Reichert's membrane is disrupted, suggesting a role for dystroglycan in basement membrane assembly at early stages of development (Williamson et al. 1997). The early embryonic lethality of dystroglycan null embryos does not permit analyses of the role of dystroglycan in organ development or in maintenance of tissue architecture. In a first step elucidating the function of dystroglycan in adult non-muscle tissues, we have here analyzed the expression pattern of dystroglycan in various adult organs. We found that dystroglycan is focally expressed on the basal side of epithelial cells in several tissues analyzed, suggesting that dystroglycan is a ubiquitous cell surface receptor involved in linking epithelial cells to basement membranes in adult tissues. In several epithelia analyzed, dystroglycan was also seen pericellularly. In addition, Belkin and Smalheiser (1996) have reported the presence of dystroglycan in cell-cell contacts, where it was shown to be co-localized with E-cadherin. A pericellular expression of dystroglycan was particularly evident in all epidermal layers of the skin. Weak staining could also be seen along the basal surface of the basal epidermal cells. The basal epidermal cells are attached to a basement membrane by many hemidesmosomes but it remains to be determined whether dystroglycan is a component of hemidesmosomes. We further found dystroglycan associated with some but not all blood vessels. Dystroglycan appeared to be associated with smooth muscle, but clearly the presence of dystroglycan in vasculature needs to be further explored.
On the basis of our findings, dystroglycan is expressed in muscle and epithelial cells in the adult organism. In addition, dystroglycan is localized to the neuromuscular and myotendinous junctions (Ohlendieck et al. 1991), to various regions and cell types within the central nervous system (Gorecki et al. 1994; Montanaro et al. 1995; Tian et al. 1996), and to the Schwann cell outer membrane of peripheral nerves (Yamada et al. 1994). These expression data suggest a functional diversity of dystroglycan.
Dystroglycan specifically binds the E3 fragment of the laminin α1-chain and to E3-like fragments of the laminin α2-chain (Gee et al. 1993; Brancaccio et al. 1995; Pall et al. 1996). The laminin α1-chain is co-expressed with dystroglycan in the basement membranes of kidney proximal tubules, mammary gland, and testis only. The E3 fragment of laminin-1 has previously been shown to be involved in lactational differentiation of the mammary gland. A function blocking anti-α6-integrin antibody had no effect on milk protein expression, whereas β1-integrin obviously was important for casein expression. These data suggest that either other integrin α-chains and/or other receptor types are involved in lactational differentiation (Streuli et al. 1995). The presence of dystroglycan in mammary gland raises the possibility that dystroglycan is involved in mammary gland differentiation.
The laminin α2-chain was co-expressed with dystroglycan in several basement membranes. These data suggest that dystroglycan might bind laminin α2 at sites where they are co-expressed. In muscle, dystroglycan and laminin α2-chain are co-expressed in the sarcolemma. An animal model for autosomal recessive muscular dystrophy is the dy mouse. Homozygous dy/ dy mice have drastically reduced levels of laminin α2-chain mRNA and more than a 95% reduction of the protein. The low levels of the laminin α2-chain apparently lead to defects in the muscle basement membrane, and the mice display a dystrophic phenotype. Aside from having a progressive muscle disease these mice are smaller and die of an unknown cause between 2 and 6 months (Sunada et al. 1994; Xu et al. 1994). The laminin α2-chain is also co-localized with dystroglycan in trachea, salivary gland, mammary gland, and testis. Thus far, consequences of the absence of laminin α2-chains in other organs have not been studied.
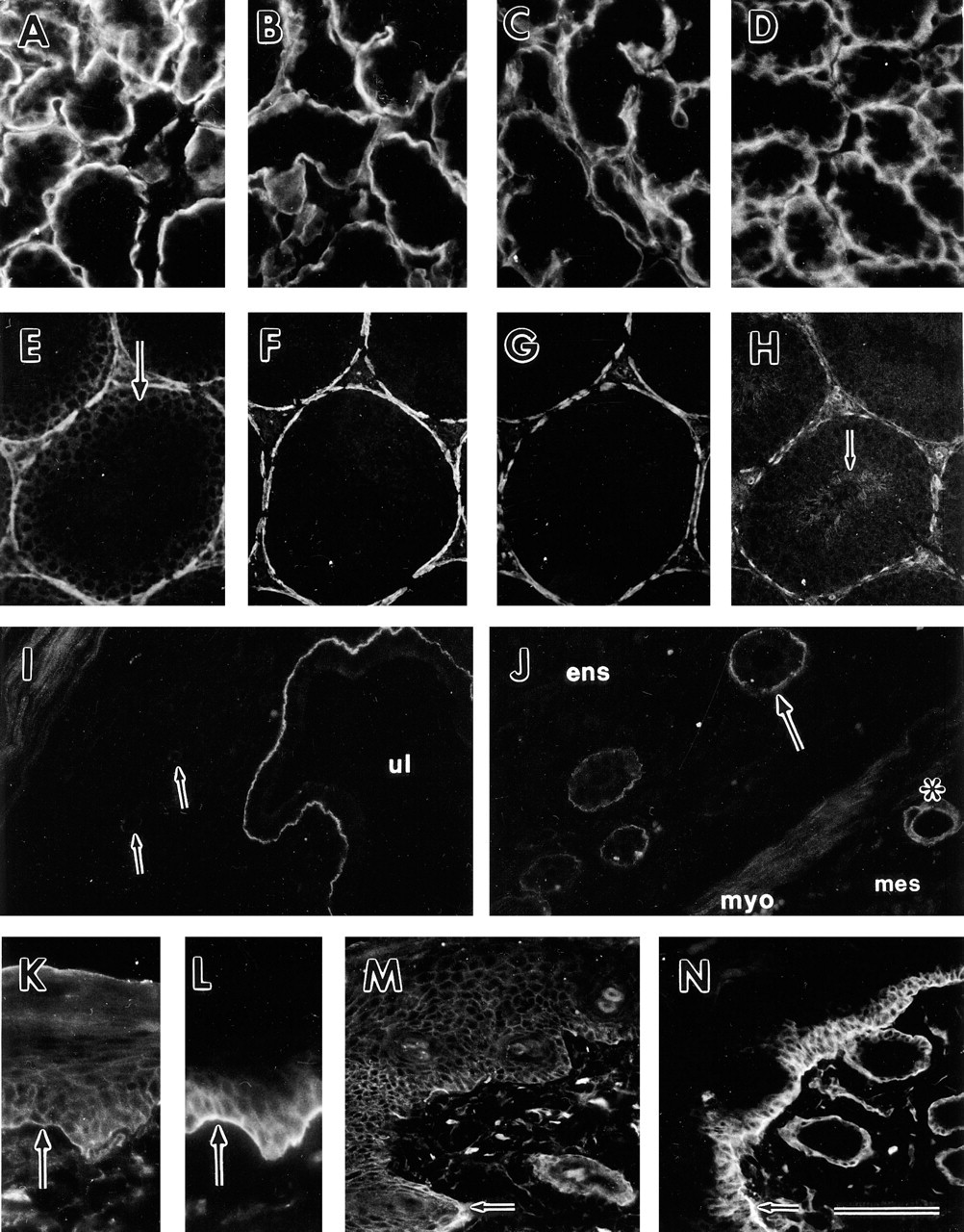
Interestingly, we could show that dystroglycan is prominently expressed in the glomerular basement membrane region of the kidney. Our results on the expression of dystroglycan in glomerular basement membrane apparently differ from previously published results (Miner and Sanes 1996), indicating the absence of dystroglycan in this location. In this study we used two different antibodies, an antibody against a fusion protein of both α- and β-dystroglycan and an antibody against β-dystroglycan. Both of these reacted with the glomerular basement membrane regions in both mouse and human kidneys. The presence of dystroglycan in the glomerular basement membrane is further supported by in situ hybridization analysis showing that epithelial podocytes produce dystroglycan mRNA. Neither the laminin α1- nor the laminin α2-chain (Durbeej et al. 1996; Miner et al. 1997; Sorokin et al. 1997) is expressed in the mature glomerular basement membrane, suggesting the presence of other laminin α-chains interacting with dystroglycan. Indeed, the recently described laminin α5-chain (Miner et al. 1995) is produced by the glomerular podocytes (Durbeej et al. 1996) and the protein is localized to the glomerular basement membrane (Miner et al. 1997; Sorokin et al. 1997). In addition to the glomerular basement membrane, a number of other basement membranes are devoid of laminin α1- and α2-chains. Several previous studies have suggested a more widespread distribution of the laminin α1-chain in mature tissues (Engvall et al. 1990; Virtanen et al. 1995), whereas other investigators have shown a more restricted distribution (Ekblom et al. 1990; Miner et al. 1997; Sorokin et al. 1997). The discrepancy now appears to be resolved because it has been demonstrated that antibody 4C7 (Engvall et al. 1990), used by many investigators to detect laminin α1-chain, instead detects the laminin α5-chain (Tiger et al. 1997). Therefore, the laminin α5-chain is widely distributed in mature tissues and is co-localized with dystroglycan in a number of tissues. It will now be interesting to determine whether the laminin α5-chain also binds α-dystroglycan.
The presence of α/β-dystroglycan in the tubule basement membranes of the kidney is unclear. Tubule basement membrane regions clearly reacted with anti FP-B antibodies (detecting both α- and β-dystroglycan), whereas antibody AP83 against β-dystroglycan failed to react with the basal side of epithelial cells of the tubules in both mouse and human kidneys. A similar observation was noted in the small intestine. Antibodies directed to both α- and β-dystroglycan recognized the basal side of the surface epithelium of the villi. However, AP83 (against β-dystroglycan) did not stain the basal side of the surface epithelium. These data indicate either that α-dystroglycan is present without β-dystroglycan in the tubule basement membrane and the basement membrane region of the villi or that β-dystroglycan is present but can not be detected because masking of the epitopes in these locations. A non-overlapping distribution of α- and β-dystroglycan has also been shown in rat cerebellum (Tian et al. 1996). Therefore, it is possible that α-dystroglycan is present without β-dystroglycan in certain locations.
In muscle, α/β-dystroglycan is a part of the DGC, a complex of proteins that anchors dystrophin to the sarcolemma. The major part of the other proteins in the DGC complex appears to be muscle-specific, but a few are ubiquitously expressed (Adams et al. 1993; Bönnemann et al. 1995; Lim et al. 1995). It will now be important to study the nature of the DGC complex in epithelial cells. We have recently shown that a shorter isoform of dystrophin, Dp140, is exclusively expressed in a subset of embryonic kidney epithelial cells (Durbeej et al. 1997). Therefore, Dp140 may interact with β-dystroglycan in kidney epithelium. However, Dp140 expression is restricted to embryonic kidney epithelium, suggesting that other intracellular dystrophin-like proteins should exist in other epithelia.
Footnotes
Acknowledgements
Supported by grants from the Swedish Cancer Fund, Barncancerfonden, and Knut and Alice Wallenberg Stiftelsen. MDH was partially supported by the Iowa Cardiovascular Interdisciplinary Research Fellowship (HL07121) and KPC is an investigator of the Howard Hughes Medical Institute.
We thank Anne-Mari Olofsson for expert technical help.
