Abstract
This study aimed to evaluate the effects of general control non-derepressible 2 (GCN2) on osteoarthritis (OA) in vivo and in vitro. First, anterior cruciate ligament transection (ACLT)-induced rat model and interleukin (IL)-1β-induced ATDC5 chondrocyte were established. Hematoxylin and eosin staining and safranin O/fast green staining were employed for analyzing the histological changes in the rat cartilage. In addition, immunohistochemistry, quantitative real-time polymerase chain reaction, enzyme-linked immunosorbent assay, western blot, and immunofluorescence staining were employed for examining cartilage degeneration-, inflammation-, autophagy-, and NLR family pyrin domain containing 3 (NLRP3) inflammasome-associated genes expression. Moreover, 2,7-dichlorodihydrofluorescein acetoacetic acid probe was utilized for examining the intracellular reactive oxygen species. In addition, 5-ethynyl-2′-deoxyuridine assay and flow cytometry were applied for detecting chondrocyte proliferation and apoptosis IL-1β-treated ATDC5 chondrocytes. GCN2 overexpression ameliorated articular cartilage degeneration and inflammation but promoted chondrocyte autophagy in ACLT-induced OA rats. Similarly, we demonstrated that the upregulation of GCN2 could promote chondrocyte proliferation, suppress chondrocyte apoptosis, attenuate chondrocyte inflammation and extracellular matrix degradation, and promote chondrocyte autophagy. Moreover, GCN2 overexpression could inhibit the activation of NLRP3 inflammasome in IL-1β-induced ATDC5 chondrocyte. Furthermore, 3-methyladenine neutralized the protective and autophagy-promoting effects of GCN2 overexpression on ATDC5 chondrocytes. GCN2 could attenuate inflammation and cartilage degeneration, promote chondrocyte autophagy, and inhibit NLRP3 inflammasome activation in OA.
Introduction
Osteoarthritis (OA) is a chronic degenerative disease that currently affects approximately 58 million adults all over the world.1,2 Both the prevalence and incidence of OA are expected to increase with the increasing life expectancy. OA mainly affects the knee joint. Moreover, OA is mainly featured with cartilage degeneration, but other pathological changes at the whole-joint level are also involved, including joint inflammation, osteophyte formation, and subchondral bone remodeling.3–6 A variety of symptomatic treatments are proposed to alleviate the symptoms of OA but cannot retard the progression of this disease.7,8 Therefore, an urgent need exists to search for a new molecular target that can retard OA progression.
The integrated stress response (ISR) is a homeostatic mechanism mediated by eukaryotic translation initiation factor 2α that enables eukaryotic cells to sense and respond to diverse stress signals, such as amino acid starvation and endoplasmic reticulum stress.9,10 General control non-derepressible 2 (GCN2) is one of the four known sensors of the ISR. 9 In recent years, GCN2 has been verified to participate in memory formation and immune response in addition to modulating the amino acid starvation response.11,12 Many researchers have proved that the downregulation of GCN2 may alleviate myocardial impairment.13,14 Interestingly, the GCN2-mediated amino acid starvation-sensing mechanism induces intestinal inflammation in mouse colitis models, suggesting that GCN2 may regulate immune responses, especially inflammation. 12 Moreover, the study by Xia et al. 9 has proved that GCN2 is a potential target for modulating inflammation. However, the function of GCN2 in OA has not yet been determined.
In the present study, the main objective was to explore the potential function and mechanism of GCN2 on OA in vivo and in vitro. We identified that GCN2 could attenuate inflammation and cartilage degeneration, promote chondrocyte autophagy, and inhibit NLR family pyrin domain containing 3 (NLRP3) inflammasome activation in OA. The elucidation of this mechanism will contribute to the development of novel and effective therapies for OA treatment.
Materials and Methods
Establishment of OA Rat Model and Treatment Protocol
The experimental protocol was approved by the Shandong Provincial Third Hospital. Male Sprague Dawley rats (8 weeks old, 300 ± 20 g) were obtained from Jinan Pengyue Experimental Animal Breeding Co., Ltd. Rats were assigned to one of the four treatment groups (Sham, OA, OA + Ad-NC, and OA + Ad-GCN2) using a randomized stratification scheme before the anterior cruciate ligament transection (ACLT) surgery. The anterior cruciate ligaments of the right knee joint (excluding rats in the Sham group) were transected as previously described 15 under 5% pentobarbital sodium anesthesia. Rats in the Sham group underwent surgery, but did not undergo ACLT. After ACLT surgery, rats were injected with penicillin for 3 consecutive days to prevent infection. The experimental protocol was approved by and performed in accordance with protocols from the Animal Care and Use Committee of our hospital. To identify the effect of GCN2, recombinant adenovirus with GCN2 overexpression was injected into the intra-articular cavity of rats three times weekly (30 μl of 1010 PFU/ml). The cartilage was removed following euthanasia at 8 weeks postsurgery.
Quantitative Real-time Polymerase Chain Reaction (qRT-PCR)
Total RNA was extracted from cell culture and articular cartilage samples utilizing TRIzol (Invitrogen, Carlsbad, California, USA) and reversed into complementary DNA with EasyScript Reverse Transcriptase (TransGen Biotech, Beijing, China). qRT-PCR was performed on BioRad CFX96 real-time system using SYBR Green PCR Master Mix (Toyobo, Osaka, Japan). The gene-specific primers utilized are as follows:
Rat GCN2 F: 5′-TTCAGGTGCAAACCCGACTT-3′
R: 5′-GTTCATCAGCGTCCCACTCT-3′
Mouse GCN2 F: 5′-CATCTGGCCTTCACTGCTGA-3′
R: 5′-AGAGCAGTACCAACCATGCC-3′
Rat Collagen Ⅱ F: 5′-GATGTATGGAAGCCCTCGTCC-3′
R: 5′-CCTTTGGCCCTAATTTTCCACT-3′
Rat ADAMTS-5 F: 5′-GCCCACCTAACGGCAAATCT-3′
R: 5′-AGGACACCTGCGTATTTGGG-3′
Rat MMP3 F: 5′-TGGACCCTGAGACCTTACCA-3′
R: 5′-TCGCCAAAAGTGCCTGTCTTT-3′
Rat MMP13 F: 5′-CAAGCAGCTCCAAAGGCTAC-3′
R: 5′-TGGCTTTTGCCAGTGTAGGT-3′
Rat TNF-α F: 5′-ATGGGCTCCCTCTCATCAGT-3′
R: 5′-GCTTGGTGGTTTGCTACGAC-3′
Mouse TNF-α F: 5′-ACCCTCACACTCACAAACCA-3′
R: 5′-ATAGCAAATCGGCTGACGGT-3′
Rat IL-6 F: 5′-AGAGACTTCCAGCCAGTTGC-3′
R: 5′-TGCCATTGCACAACTCTTTTC-3′
Mouse IL-6 F: 5′-CAACGATGATGCACTTGCAGA-3′
R: 5′-TCTGTGACTCCAGCTTATCTCTTG-3′
Rat β-actin F: 5′-CAGCCTTCCTTCCTGGGTATG-3′
R: 5′-CTGTCAGCAATGCCTGGGTA-3′
Mouse β-actin F: 5′-CCAGCCTTCCTTCTTGGGTAT-3′
R: 5′-GGGTGTAAAACGCAGCTCAG-3′.
Histology and Immunohistochemical Assay
The knee joints were fixed for 48 hr with 10% neutral formalin buffer at room temperature, followed by decalcification for 4 weeks in ethylene diamine tetraacetic acid and dehydration using ethanol in a conventional gradient. Subsequently, the samples were embedded into paraffin, sectioned at a thickness of 5 µm, and then stained with hematoxylin and eosin (H&E) or safranin O/fast green. Two blinded observers used the Osteoarthritis Research Society International (OARSI) scoring system for assessing the degeneration of articular cartilage as previously described. 16 For immunohistochemical staining, the paraffin sections of rat cartilage samples were incubated in 3% hydrogen peroxide to block the endogenous peroxidase activity. The primary antibodies (GCN2, no. K002634P, 1:100, Solarbio, Beijing, China; Collagen II, no. ab34712, 1:50, Abcam, Cambridge, UK; ADAMTS-5, no. ab231595, 1:100, Abcam; MMP13, no. 18165-1-AP, 1:100, Proteintech, Wuhan, China; LC3, no. 14600-1-AP, 1:50, Proteintech) were added to the tissue sections and then incubated overnight at 4C. After washing, the sections were incubated in secondary antibody for 1.5 hr at room temperature. Finally, the tissue section was stained with 3, 3′-diaminobenzidine tetrahydrochloride (Sigma, Saint Louis, Missouri, USA), counterstained with hematoxylin, and mounted.
Cell Culture and Treatment
The ATDC5 chondrocyte cell line was gained from Cobioer Biosciences (Nanjing, China). The cells were maintained in DMEM/Ham’s F12 (Sigma) supplemented with 10% FBS and 100 U/ml penicillin/streptomycin at 37C with an atmosphere of 5% CO2 and 95% humidity. ATDC5 cells were treated with 10 ng/ml interleukin (IL)-1β (MedChemExpress, Princeton, New Jersey, USA; HY-P7097) for 24 hr at 37C. Moreover, ATDC5 chondrocytes were pre-exposed to GCN2 overexpression vector (GeneCopoeia, Guangzhou, China) and 3-methyladenine (3MA, 10 μM, HY-19312, MedChemExpress) for 24 hr followed by treatment with IL-1β.
5-Ethynyl-2′-Deoxyuridine (EdU) Assay
The EdU assay was carried out by using a Cell-Light EdU DNA Cell Proliferation Kit (Ribobio, Guangzhou, China) as per the instructions of manufacturer. In brief, the treated ATDC5 chondrocytes (2 × 105 cells/well) were seeded into 96-well plates and cultured with EdU for 120 min. After that, ATDC5 chondrocytes were fixed with 4% paraformaldehyde, permeabilized, and stained with 4′,6-diamidino-2-phenylindole (DAPI) at a concentration of 10 μg/ml for 20 min. At last, the positive ATDC5 chondrocytes were photographed and counted utilizing a Eclipse Ni-U fluorescence microscope (NIKON, Tokyo, Japan).
Flow Cytometry Analysis
Cell apoptosis was assessed utilizing a fluorescein isothiocyanate (FITC)-Annexin V/propidium iodide (PI) Apoptosis Detection Kit (Vazyme Biotech, Nanjing, China). Shortly, ATDC5 cells were resuspended and coincubated with FITC-Annexin V/PI for 20 min in the absence of light. The apoptosis in each group was examined and analyzed by flow cytometry with a BD FACSCalibur instrument (BD Biosciences, San Jose, California, USA).
Measurement of Reactive Oxygen Species (ROS)
2,7-Dichlorodihydrofluorescein acetoacetic acid (DCFH-DA) is a cell-permeable probe that examines intracellular ROS. ATDC5 chondrocytes were stained with DCFH-DA (10 mM, Invitrogen) for 30 min at 37C free of light, and then resuspended in PBS. The fluorescence intensity was observed by using an Eclipse Ni-U fluorescence microscope (NIKON).
Enzyme-linked Immunosorbent Assay (ELISA)
According to the manufacturer’s protocol, mouse tumor necrosis factor-α (TNF-α) ELISA kit (no. PMTA00B, R&D, Minneapolis, Minnesota, USA) and mouse IL-6 ELISA kit (no. PM6000B, R&D) were utilized for assessing TNF-α and IL-6 levels in ATDC5 cells.
Western Blot Analysis
ATDC5 cells were lysed in radio-immunoprecipitation assay lysis buffer (Beyotime, Jiangsu, China), followed by centrifugation at 12,000 rpm for 10 min at 4C to harvest the supernatants. Equal amounts of protein (50 μg) and marker (5 μl, Solarbio) were run on an SDS-PAGE and transferred onto polyvinylidene difluoride (PVDF) membranes following electro-blotting. Subsequently, the PVDF blotting membrane was incubated in primary antibodies (GCN2, no. K002634P, 1:500, Solarbio; Bcl-2, no. K003505P, 1:1000, Solarbio; Bax, no. K008076P, 1:1000, Solarbio; cleaved caspase-3, no. 9661, 1:500, Cell Signaling, Danvers, Massachusetts, USA; TNF-α, no. ab183218, 1:500, Abcam; IL-6, no. ab290735, 1:1000, Cell Signaling; Collagen II, no. ab34712, 1:1000, Abcam; ADAMTS-5, no. ab231595, 1:1000, Abcam; MMP3, no. ab52915, 1:1000, Abcam; MMP13, no. 18165-1-AP, 1:1000, Proteintech; LC3, no. 14600-1-AP, 1:1000, Proteintech; Beclin 1, no. ab210498, 1:1000, Abcam; NLRP3, no. ab270449, 1:500, Abcam; ASC, no. ab307560, 1:500, Abcam; pro-caspase-1, no. ab179515, 1:500, Abcam; caspase-1, no. 89332, 1:500, Cell Signaling; β-actin, no. K200058M, 1:3000, Solarbio) overnight at 4C followed by horseradish peroxidase (HRP)-conjugated secondary antibody conjugated to Goat Anti-rabbit IgG H&L (HRP; no. SA00001-2, 1:5000, Proteintech) for 60 min at room temperature. Finally, protein bands were detected with an enhanced chemiluminescence reagent (Beyotime) with β-actin as the internal control.
Immunofluorescence Staining
First, 4% polyformaldehyde was added to fix ATDC5 cells for 20 min at room temperature. Next, the cells were permeabilized and blocked with 0.3% Triton X-100 and 5% bovine serum albumin for 120 min at room temperature. Subsequently, the cells were incubated overnight at 4C with the specific primary antibodies (LC3, no. 14600-1-AP, 1:50, Proteintech; NLRP3, no. ab270449, 1:50, Abcam) and then incubated with the goat anti-rabbit IgG H&L (Alexa Fluor 488; no. ab150077, 1:500, Abcam) or goat anti-rabbit IgG H&L (Alexa Fluor 555; no. ab150078, 1:500, Abcam) for 30 min at room temperature. Finally, the sections were counterstained with DAPI in mounting media, and then the fluorescence single images were captured under an Eclipse Ni-U fluorescence microscope (NIKON).
Statistical Analysis
All data are presented as mean ± SD. The significance of differences between groups was assessed using the t-tests and one-way ANOVA using GraphPad Prism 8.0 (GraphPad Software, San Diego, CA). Differences with a p<0.05 were considered statistically significant.
Results
GCN2 Overexpression Ameliorated ACLT-induced Degeneration of Rat Articular Cartilage
The data of qRT-PCR and immunohistochemical staining manifested that GCN2 was downregulated in OA rats (Fig. 1A and B). Then, the function of GNC2 on OA was explored by using adenovirus with GCN2 overexpression. Figure 1C illustrated that the injection of Ad-GCN2 significantly reversed the downregulation of GCN2 caused by ACLT surgery. In addition, H&E staining results manifested that the increased chondrocyte hypertrophy and matrix damage occurred in OA cartilages (Fig. 1D and E). On the contrary, the pathological damage in OA cartilages was significantly neutralized by GCN2 overexpression (Fig. 1D and E). Subsequently, safranin O/fast green staining was used to investigate the degree of tissue lesion (Fig. 1F). The considerable cartilage degeneration and loss of fiber layers induced by ACLT surgery were markedly alleviated by the overexpression of GCN2 (Fig. 1F). Moreover, the increased OARSI score in OA rats was remarkedly reversed by GCN2 overexpression (Fig. 1G). Based on the results of immunohistochemical staining, the downregulated expression of Collagen II, as well as the upregulated expression of ADAMTS-5 and MMP13, which were observed in OA rat articular cartilage, was significantly neutralized by GCN2 overexpression (Fig. 2A). In addition, qRT-PCR data further confirmed that the increased Collagen II level, as well as the decreased ADAMTS-5, MMP3, MMP13, TNF-α, and IL-6, was found in OA rat articular cartilage (Fig. 2B–G). After injection of Ad-GCN2, expression of these genes was markedly decreased (Fig. 2B–G). Besides, we also demonstrated that GCN2 overexpression notably neutralized the downregulated expression of LC3 in OA rat articular cartilage (Fig. 2H). Taking together, the aforementioned results demonstrated that GCN2 overexpression could ameliorate ACLT-induced degeneration of rat articular cartilage.
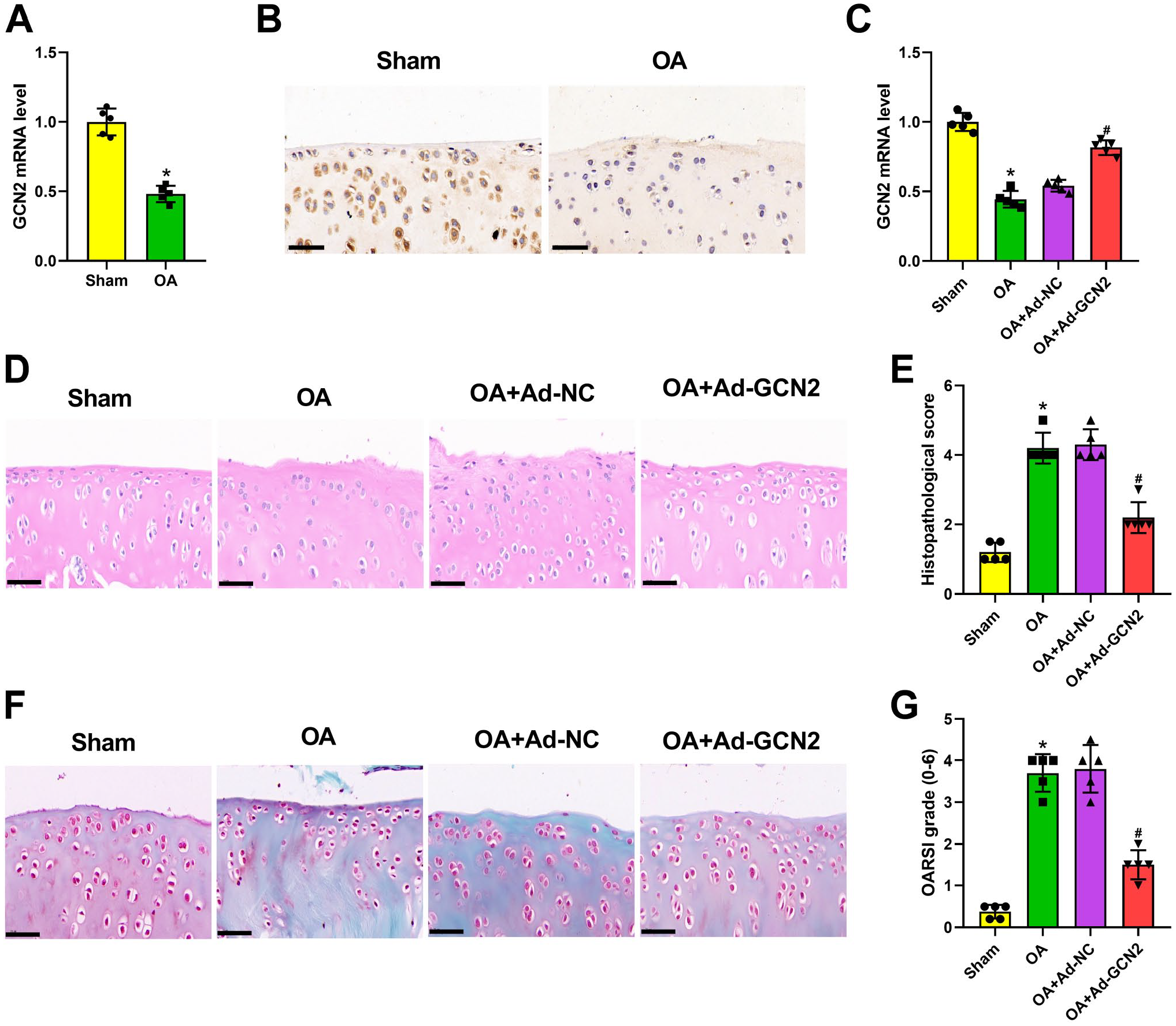
General control non-derepressible 2 (GCN2) overexpression ameliorated osteoarthritis (OA) cartilage lesions in rats. GCN2 expression in articular cartilage tissues isolated from Sham and anterior cruciate ligament transection (ACLT) surgery rats was detected using quantitative real-time polymerase chain reaction (qRT-PCR; n=5 rats per group; Panel A). GNC2 mRNA expression was decreased in cartilage tissues of OA rats. GCN2 expression in articular cartilage tissues isolated from Sham and ACLT surgery rats was measured using immunohistochemical staining (n=5 rats per group; Panel B). GNC2 expression was decreased in cartilage tissues of OA rats. After ACLT surgery and the injection of adenovirus with GCN2 overexpression or its negative control, qRT-PCR was used for detecting GCN2 level in articular cartilage tissues of rats (n=5 rats per group; Panel C). After injection of adenovirus with GCN2 overexpression, GNC2 mRNA expression was increased in cartilage tissues of OA rats. The histological changes of rat cartilage tissues were analyzed by hematoxylin and eosin staining (n=5 rats per group; Panel D), histopathological score (n=5 rats per group; Panel E), safranin O/fast green staining (n=5 rats per group; Panel F), and Osteoarthritis Research Society International scoring system (n=5 rats per group; Panel G). GCN2 overexpression significantly attenuated the pathological damage of OA cartilages, cartilage degeneration, and fiber-layer loss caused by ACLT surgery. *p<0.05, #p<0.05. (“Bars B, D and F = 50 μm.”) . Ad-GCN2= adenovirus with GCN2 overexpression; Ad-NC= adenovirus with negative control.
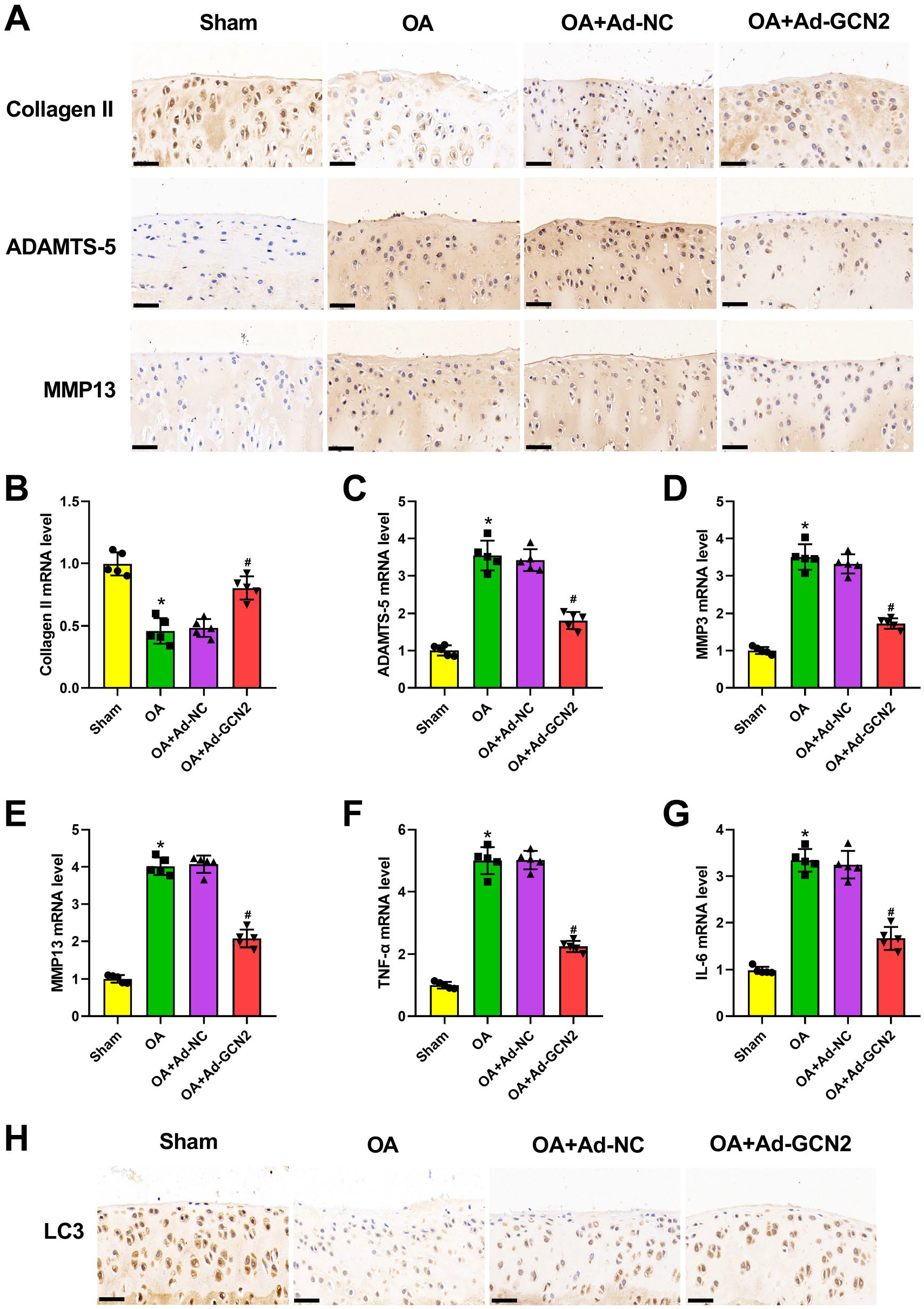
General control non-derepressible 2 (GCN2) overexpression ameliorated articular cartilage degeneration and inflammation and promoted chondrocyte autophagy in osteoarthritis (OA) rats. After anterior cruciate ligament transection surgery and the injection of adenovirus with GCN2 overexpression or its negative control, immunohistochemical staining was utilized for testing Collagen II, ADAMTS-5, and MMP13 levels (n=5 rats per group) in articular cartilage tissues of rats (Panel A). GCN2 overexpression significantly increased Collagen II, and decreased ADAMTS-5 and MMP13 expression in OA rats. Quantitative real-time polymerase chain reaction was applied for assessing Collagen II (n=5 rats per group; Panel B), ADAMTS-5 (n=5 rats per group; Panel C), MMP3 (n=5 rats per group; Panel D), MMP13 (n=5 rats per group; Panel E), tumor necrosis factor-α (TNF-α; n=5 rats per group; Panel F), and interleukin-6 (IL-6; n=5 rats per group; Panel G) mRNA level in articular cartilage tissues of rats. GCN2 overexpression significantly increased Collagen II, and decreased ADAMTS-5, MMP3, MMP13, TNF-α, and IL-6 expression in OA rats. Immunohistochemical staining was utilized for detecting LC3 level (n=5 rats per group) in articular cartilage tissues of rats (Panel H). GCN2 overexpression significantly increased LC3 expression in OA rats.*p<0.05, #p<0.05. (“Bars A and H = 50 μm.”)
GCN2 Overexpression Enhanced Chondrocyte Proliferation and Suppressed Chondrocyte Apoptosis in IL-1β-treated ATDC5 Chondrocytes
As displayed in Fig. 3A and B, GCN2 overexpression significantly reversed the expression of GCN2 in IL-1β-treated ATDC5 chondrocytes. We also performed EdU and flow cytometry to assess the role of GCN2 in IL-1β-treated ATDC5 chondrocytes and demonstrated that the inhibitory proliferation and accelerative apoptosis, which were induced by IL-1β, were significantly reversed by GCN2 overexpression (Fig. 3C and D). Interestingly, western blot data also verified the inhibitory effect of GCN2 overexpression on chondrocyte apoptosis via detecting Bcl-2, Bax, and cleaved caspase-3 (Fig. 3E).
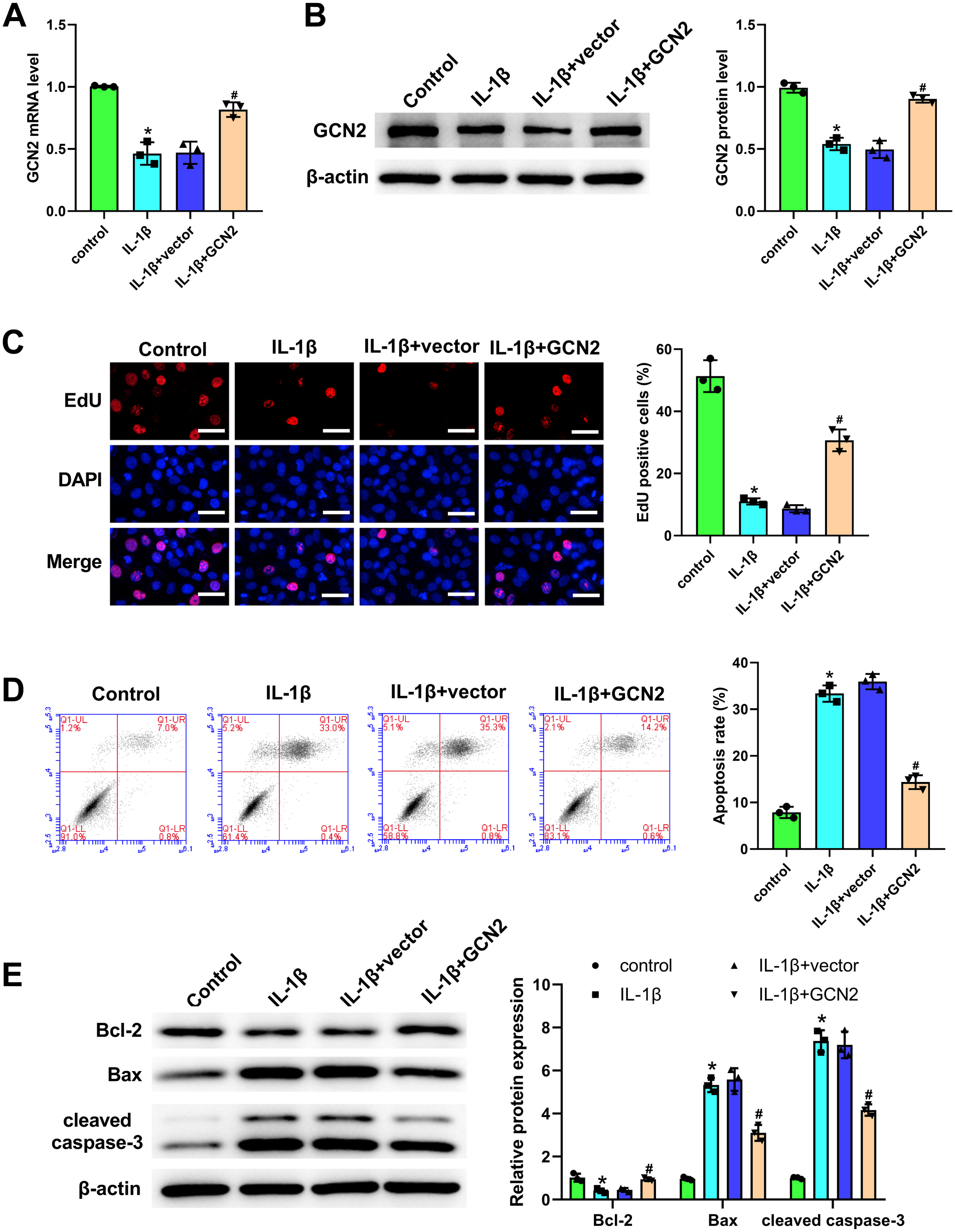
General control non-derepressible 2 (GCN2) overexpression enhanced chondrocyte proliferation and suppressed chondrocyte apoptosis in interleukin (IL)-1β-treated ATDC5 chondrocytes. After the treatment of IL-1β and the transfection of GCN2 overexpression vector or negative vector, quantitative real-time polymerase chain reaction (Panel A) and western blot (Panel B) were employed for assessing GCN2 level in chondrocytes. After transfection with GCN2 overexpression vector, GNC2 expression was increased in IL-1β-treated chondrocytes. 5-Ethynyl-2′-deoxyuridine (EdU) assay was applied for assessing the function of GCN2 on chondrocyte proliferation (Panel C). Positive cells were labeled with EdU (red), and nuclei were stained with 4′,6-diamidino-2-phenylindole (blue). GCN2 overexpression significantly increased the proliferation of IL-1β-treated chondrocytes. Flow cytometry was applied for assessing the function of GCN2 on chondrocyte apoptosis (Panel D). GCN2 overexpression significantly reduced the apoptosis of IL-1β-treated chondrocytes. Western blot was used for exploring the function of GCN2 on apoptosis-associated proteins in chondrocytes (Panel E). GCN2 overexpression significantly increased Bcl-2, and decreased Bax and cleaved caspase-3 expression in IL-1β-treated chondrocytes. The values are mean ± SD of three independent experiments (n=3). *p<0.05, #p<0.05. (“Bar C = 50 μm.”)
GCN2 Overexpression Attenuated Chondrocyte Inflammation and Extracellular Matrix (ECM) Degradation, but Promoted Chondrocyte Autophagy in IL-1β-treated ATDC5 Chondrocytes
The results, presented in Fig. 4A, demonstrate that a high ROS level was observed in IL-1β-induced chondrocytes. After transfection of GCN2 overexpression vector, ROS level was significantly decreased in IL-1β-induced chondrocytes (Fig. 4A). Moreover, both ELISA and qRT-PCR confirmed that the increased expression of TNF-α and IL-6 induced by IL-1β in chondrocytes was notably reversed after transfection of GCN2 overexpression vector (Fig. 4B and C). Meanwhile, we also demonstrated that the downregulated expression of Collagen II, LC3, and Beclin 1, as well as the upregulated expression of ADAMTS-5, MMP3, and MMP13, which were induced by IL-1β in chondrocytes, markedly attenuated via transfection of GCN2 overexpression vector (Fig. 4D and E). In addition, the data of immunofluorescence staining further verified the role of GCN2 overexpression on LC3 in IL-1β-induced chondrocytes. Above all, these results demonstrated that GCN2 overexpression attenuated chondrocyte inflammation and ECM degradation; however, it promoted chondrocyte autophagy in IL-1β-treated ATDC5 chondrocytes.
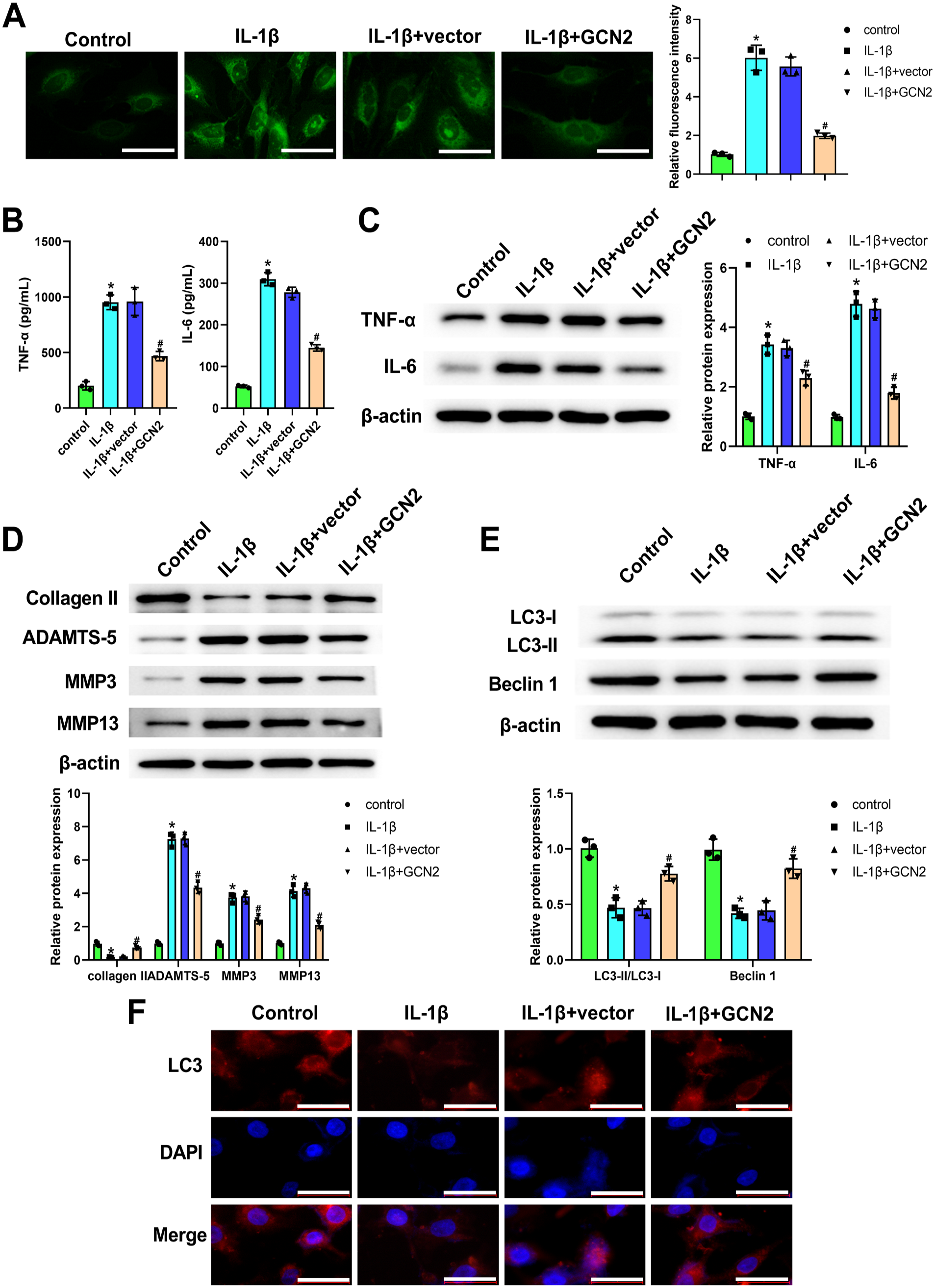
General control non-derepressible 2 (GCN2) overexpression attenuated chondrocyte inflammation and extracellular matrix degradation, but promoted chondrocyte autophagy in interleukin (IL)-1β-treated ATDC5 chondrocytes. After the treatment of IL-1β and the transfection of GCN2 overexpression vector or negative vector, 2,7-dichlorodihydrofluorescein acetoacetic acid (DCFH-DA) probe was used for assessing the reactive oxygen species (ROS) level (Panel A). ROS levels in chondrocytes were analyzed by DCFH-DA fluorescence intensity using a fluorescence microscope. GCN2 overexpression significantly reduced the fluorescence intensity of IL-1β-treated chondrocytes. Enzyme-linked immunosorbent assay (Panel B) and western blot (Panel C) were applied for measuring tumor necrosis factor-α (TNF-α) and IL-6 level in chondrocytes. GCN2 overexpression significantly reduced TNF-α and IL-6 expression in IL-1β-treated chondrocytes. Western blot for detecting Collagen II, ADAMTS-5, MMP3 and MMP13 expression in chondrocytes (Panel D). GCN2 overexpression significantly increased Collagen II, and decreased ADAMTS-5, MMP3, and MMP13 expression in IL-1β-treated chondrocytes. Western blot for detecting LC3 and Beclin 1 expression in chondrocytes (Panel E). GCN2 overexpression significantly increased LC3 and Beclin 1 expression in IL-1β-treated chondrocytes. Immunofluorescence staining was employed for analyzing LC3 level in chondrocytes by using a fluorescence microscope (Panel F). Red represents LC3, and blue represents nuclei. GCN2 overexpression significantly increased LC3 expression in IL-1β-treated chondrocytes. The values are mean ± SD of three independent experiments (n=3). Abbreviation: DAPI = 4′,6-diamidino-2-phenylindole. *p<0.05, #p<0.05. (“Bars A and F = 50 μm.”)
GCN2 Overexpression Inhibited the Activation of NLRP3 Inflammasome in IL-1β-treated ATDC5 Chondrocytes
The results of western blot demonstrated that the expression of NLRP3, ASC, pro-caspase-1, and caspase-1 was significantly elevated in the IL-1β group when compared with those in the control group (Fig. 5A). After transfection of GCN2 overexpression vector, the expression of these genes was markedly reversed in ATDC5 chondrocytes (Fig. 5A). In addition, the results of immunofluorescence staining further verified the inhibitory function of GCN2 overexpression on NLRP3 expression in IL-1β-induced chondrocytes (Fig. 5B). Overall, these results demonstrated that GCN2 overexpression could inhibit the activation of NLRP3 inflammasome in IL-1β-treated ATDC5 chondrocytes.
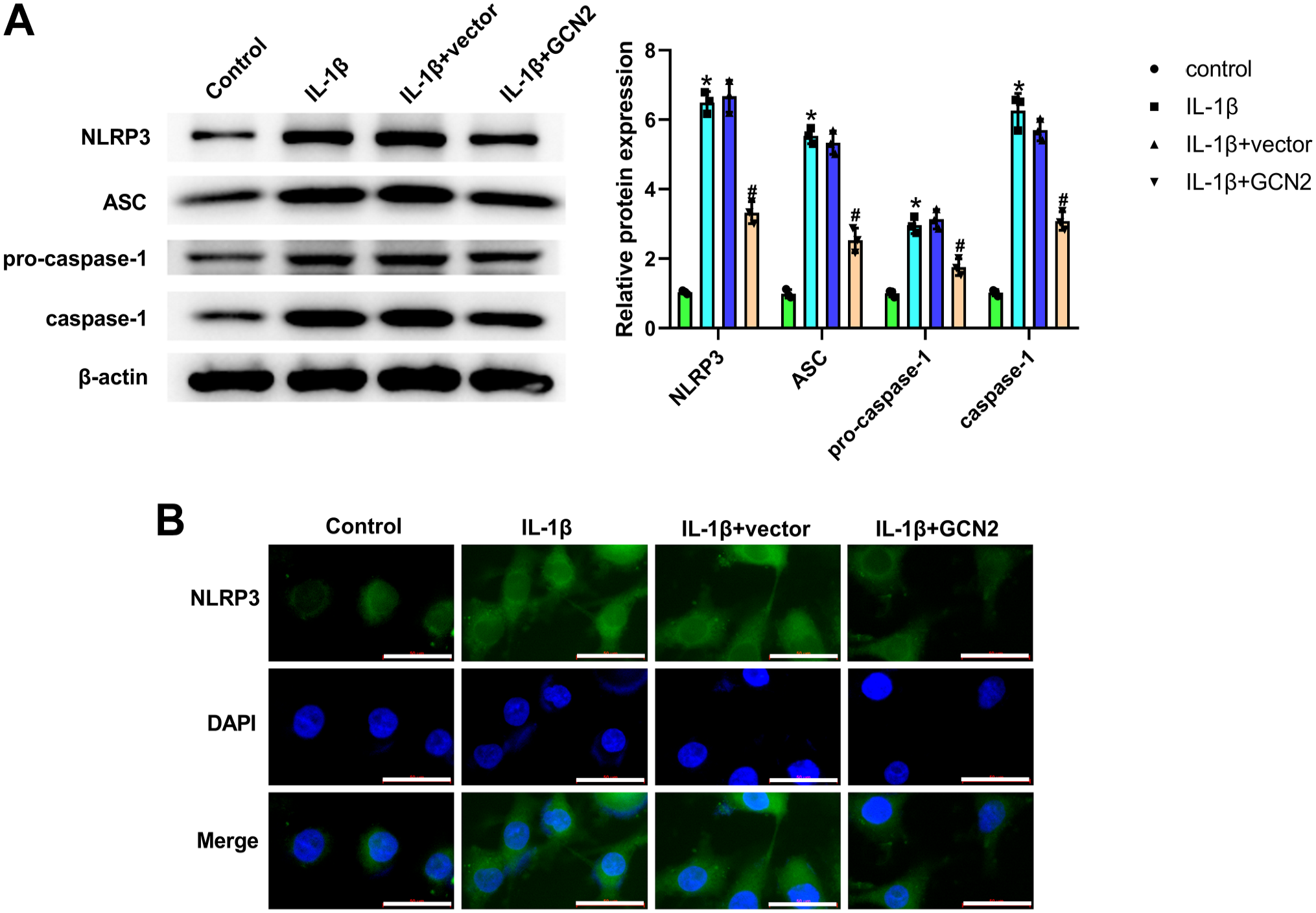
General control non-derepressible 2 (GCN2) overexpression inhibited the activation of NLR family pyrin domain containing 3 (NLRP3) inflammasome in interleukin (IL)-1β-treated ATDC5 chondrocytes. After the treatment of IL-1β and the transfection of GCN2 overexpression vector or negative vector, western blot was utilized for measuring NLRP3 inflammasome-related proteins in chondrocytes (Panel A). GCN2 overexpression significantly decreased NLRP3, apoptosis-associated speck-like protein containing a CARD (ASC), pro-caspase-1, and caspase-1 expression in IL-1β-treated chondrocytes. After the treatment of IL-1β and the transfection of GCN2 overexpression vector or negative vector, immunofluorescence staining was employed for testing NLRP3 level in chondrocytes by using a fluorescence microscope (Panel B). Green represents LC3, and blue represents nuclei. GCN2 overexpression significantly decreased NLRP3 expression in IL-1β-treated chondrocytes. The values are mean ± SD of three independent experiments (n=3). Abbreviation: DAPI = 4′,6-diamidino-2-phenylindole. *p<0.05, #p<0.05. (“Bar B = 50 μm.”)
3MA Neutralized the Protective Effect and Autophagy-promoting Effect of GCN2 Overexpression on ATDC5 Chondrocytes
When compared with the IL-1β + GCN2 group, the level of LC3 and Beclin 1 was significantly reduced in ATDC5 chondrocytes (Fig. 6A). In addition, we discovered that the inhibitory effect of GCN2 overexpression on ROS in chondrocytes was attenuated by 3MA (Fig. 6B). Moreover, the results from ELISA and qRT-PCR demonstrated that 3MA effectively nullified the inhibitory effect of GCN2 expression on inflammatory factors in chondrocytes (Fig. 6C and D). At the same time, the upregulated expression of Collagen II, as well as the downregulated expression of ADAMTS-5, MMP3, MMP13, NLRP3, ASC, pro-caspase-1, and caspase-1, which were caused by GCN2 overexpression vector transfection in chondrocytes, markedly neutralized via the administration of 3MA (Fig. 6E and F). Above all, these results demonstrated that 3MA could neutralize the protective and autophagy-promoting effects of GCN2 overexpression on ATDC5 chondrocytes.
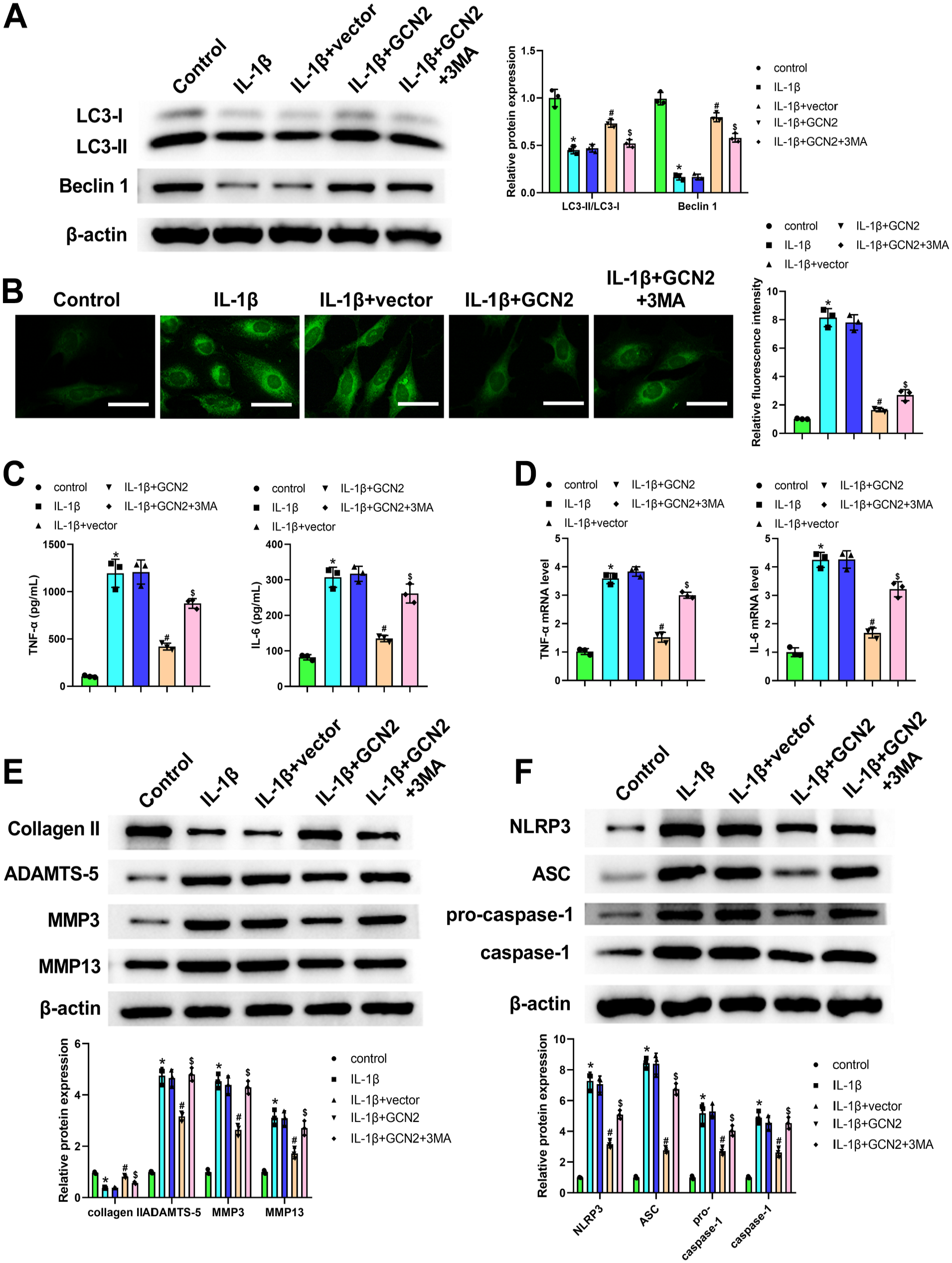
3-Methyladenine (3MA) neutralized the protective effect and autophagy-promoting effect of general control non-derepressible 2 (GCN2) overexpression on ATDC5 chondrocytes. After the treatment of interleukin (IL)-1β and 3MA, and the transfection of GCN2 overexpression vector or negative vector, western blot was applied for detecting LC3 and Beclin 1 expression in chondrocytes (Panel A). The increased LC3 and Beclin 1 expression induced by GCN2 overexpression in IL-1β-treated chondrocytes was attenuated by 3MA. 2,7-Dichlorodihydrofluorescein acetoacetic acid (DCFH-DA) probe was used for assessing the reactive oxygen species (ROS) level (Panel B). ROS levels in chondrocytes analyzed by DCFH-DA fluorescence intensity using a fluorescence microscope. The reduced fluorescence intensity induced by GCN2 overexpression in IL-1β-treated chondrocytes was attenuated by 3MA. Enzyme-linked immunosorbent assay (Panel C) and quantitative real-time polymerase chain reaction (Panel D) were used for measuring tumor necrosis factor-α (TNF-α) and IL-6 level in chondrocytes. The decreased TNF-α and IL-6 level in IL-1β-treated chondrocytes induced by GCN2 overexpression was attenuated by 3MA. Western blot for detecting Collagen II, ADAMTS-5, MMP3, and MMP13 expression in chondrocytes (Panel E). The increased Collagen II expression, and the decreased ADAMTS-5, MMP3, and MMP13 expression, in IL-1β-treated chondrocytes induced by GCN2 overexpression was attenuated by 3MA. Western blot was utilized for measuring NLR family pyrin domain containing 3 (NLRP3) inflammasome-related proteins in chondrocytes (Panel F). The decreased NLRP3, ASC, pro-caspase-1, and caspase-1 expression in IL-1β-treated chondrocytes induced by GCN2 overexpression was attenuated by 3MA. The values are mean ± SD of three independent experiments (n=3).*p<0.05, #p<0.05. (“Bar B = 50 μm.”)
Discussion
This study demonstrated that GCN2 is involved in OA progression both in vivo and in vitro. The expression of GCN2 was downregulated in ACLT-induced rat OA model and IL-1β-induced chondrocytes. We proved that GCN2 could attenuate inflammation and cartilage degeneration, promote chondrocyte autophagy, and inhibit NLRP3 inflammasome activation in OA, revealing that GCN2 is a promising target for OA treatment.
Subchondral bone remodeling, joint inflammation, osteophyte formation, and chondrocyte-mediated ECM degradation are the main features of OA.5,17 Articular cartilage degeneration has long been considered to be a major factor that triggers the development of OA. Therefore, many therapeutic strategies for articular cartilage degeneration have been used to mitigate the progression of OA.18,19 In addition, various inflammatory cytokines play an important role in OA. 20 Inflammatory cytokines, such as TNF-α, IL-1β, and IL-6, are widely recognized for their role in promoting the catabolic processes during OA advancement, which can accelerate the degradation of the major ECM through repressing proteoglycans, aggrecan, and Collagen II.21,22 In addition, the importance of inflammatory cytokines in facilitating enzymatic mediators of cartilage catabolism, such as matrix metalloproteinases (MMPs) and a disintegrin-like and metalloprotease domain with thrombospondin type 1 repeats (ADAMTS) enzymes, highlights the potential of targeting early inflammation for the prevention of OA. 23 ADAMTS-5 (aggrecanase-2) is a vital member of the ADAMTS metalloproteinase family, which participates in cartilage degradation. 24 In our study, GCN2 was lowly expressed in OA. Moreover, we also demonstrated that the upregulation of GCN2 significantly ameliorated ACLT-induced degeneration of articular cartilage and inflammation in rats. Similarly, the upregulation of GCN2 could promote chondrocyte proliferation, suppress chondrocyte apoptosis, and attenuate chondrocyte inflammation and ECM degradation in IL-1β-treated ATDC5 chondrocytes.
Autophagy is a highly conserved process of lysosome-mediated degradation of long-lived proteins and damaged organelles to maintain cellular homeostasis and metabolism, thereby promoting cell survival. 25 Autophagy, a protective mechanism of cells, is reported to participate in a variety of biological functions such as cell differentiation, cell proliferation, and apoptosis.26,27 Yan et al. 28 have proved that autophagy can attenuate OA in mice by suppressing chondrocyte pyroptosis and improving subchondral bone remodeling. Moreover, a study by Bay-Jensen et al. 29 has confirmed that maintaining autophagy level in chondrocytes is a critical way to prevent cartilage degeneration. Our findings demonstrated that GCN2 overexpression significantly promoted chondrocyte autophagy in OA by detecting autophagy-related markers.
In recent years, increasing researchers have focused on the vital functions of NLRP3 inflammasome in immune-associated diseases, including OA.30,31 The NLRP3 inflammasome is composed of NLRP3, ASC, and caspase-1. 32 When stimulated, NLRP3 protein assembled with ASC and pro-caspase-1 into the NLRP3 inflammasome allowing the production of cleaved caspase-1 and subsequently activating IL-1β, which finally mediates tissue inflammation.32–34 An increasing number of studies have reported that the dysfunction of autophagy may cause inflammation and hyperactivation of NLRP3 inflammasomes, thus acting as their main regulator. 35 Interestingly, Ravindran et al. 12 have indicated that GCN2 can control gut inflammation through repressing NLRP3 inflammasome activation. Therefore, we investigated whether GCN2 improved OA through regulating NLRP3 inflammasome, and found that the upregulation of GCN2 inhibited NLRP3 inflammasome activation in OA.
In conclusion, the upregulation of GCN2 could attenuate inflammation and cartilage degeneration, promote chondrocyte autophagy, and inhibit NLRP3 inflammasome activation in OA. The significant effect of GCN2 on OA indicates the potential application of GCN2, in the near future, as a molecular target for OA therapy.
Footnotes
Author Contributions
All authors have contributed to this article as follows: conception and design (LF), collection and assembly of data (LF, ZW, and JL), data analysis and interpretation (YL, WH), manuscript writing (LF), and all authors gave final approval for the manuscript.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
The experimental protocol of our study was performed in accordance with the Guide for the Care and Use of Laboratory Animals. The experimental protocol was approved by the Shandong Provincial Third Hospital.
Availability of Data and Material
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
