Abstract
Osteoarthritis is a common chronic bone and joint disease, which is characterized by degenerative changes and destruction of articular cartilage, secondary hyperostosis. This study aimed to investigate the clinical severity and mechanism of S100A12 in patients with osteoarthritis. Serum samples were obtained from patients with osteoarthritis or normal volunteer in Minhang Branch of Yueyang Hospital of Integrated Traditional Chinese and Western Medicine affiliated to Shanghai University of Traditional Chinese Medicine (Shanghai, China). C57BL/6J mice performed Resection of the medial collateral ligament and medial meniscus as mice model. MC3T3-E1 cells were induced with 100 ng of LPS as vitro model. The serum level of S100A12 was increased in patients with osteoarthritis. Similarly, S100A12 levels of serum and bone tissue from mice model of osteoarthritis were also higher than those of sham group. Over-expression of S100A12 promoted inflammation levels while down-regulation of S100A12 decreased inflammation levels in in vitro model of osteoarthritis. NLRP3 is an important target of S100A12 in pro-inflammation effects of osteoarthritis. NLRP3 was involved in the effects of S100A12 on inflammation in in vitro model of osteoarthritis. S100A12 also accelerated inflammation by NLRP3 in mice model of osteoarthritis. We conclude that serum S100A12 levels was a possible clinical severity, open inflammation of osteoarthritis model by NLRP3 and its receptors may be effective in preventing the development of osteoarthritis.
Introduction
The main clinical symptoms and signs of osteoarthritis (OA) include sudden redness, swelling, heat, and pain of the small joints of the lower extremities. 1 In recent years, with the continuously improved living standards of people, high-protein and high-purine diets have become the daily dining habits of many families. 2 In addition to alcohol consumption, anxiety, fatigue, etc., the incidence of gout has been increasing, with urbanized and young age trend. 3 The prevalence of gout is approximately 0.83%–1.98% among males, and 0.07%–0.72% among females in China.
NLRP3 inflammasome is an important class of receptors in the innate immune system. As a critical regulator of the inflammatory response, NLRP3 can recognize pathogen associated molecular pattern (PAMP). 4 NLRP3 is an important protein in the NLRP3 inflammasome signaling pathway that recognizes pathogens in the immune system of OA. 5 Massive studies have demonstrated that NLRP3 can produce a large number of inflammatory factors in OA or other inflammatory diseases, such as IL-1β, induced inflammatory injury of immune cells or bone cell injury of OA.6,7
S100A12, a newly-discovered pro-inflammatory factor, is involved in the body’s immune defense and inflammatory response and plays key roles in inflammation, which is also able to regulate cell growth and differentiation, inhibit growth, and induce apoptosis. 8 Pro-inflammatory factor S100A12, a member of the calcium-binding protein family, has a high affinity for calcium ions. 9 The stimulation by other inflammatory mediators can change the protein conformation of S100A12, thereby exposing the binding site of target protein, which can interact with specific proteins or peptides to play biological functions. 10 These calcium-binding proteins control critical cellular pathways such as cytoskeleton, cell migration and adhesion, active oxidative defense, wound healing, activation of inflammatory cells, chemotaxis, and antimicrobial effects, which are also involved in the pathophysiological process of metabolic and neoplastic diseases. 11 So, we considered that S100A12 may regulate inflammation factors to prevent osteoarthritis. This study aimed to investigate the clinical severity and mechanism of S100A12 in patients with osteoarthritis.
Material and methods
Clinical patients
Serum samples were obtained from patients with osteoarthritis (n = 24) or normal volunteer (n = 12) in Minhang Branch of Yueyang Hospital of Inte-grated Traditional Chinese and Western Medicine affiliated to Shanghai University of Traditional Chinese Medicine (Shanghai, China). Written in-formed consent was obtained from all participants and the research protocols were approved by the Ethics Committee of the Minhang Branch of Yueyang Hospital of Integrated Traditional Chinese and Western Medicine affiliated to Shanghai University of Traditional Chinese Medicine (No. 201905061781). Diagnosis of osteoarthritis was performed according to criteria of the American College of Rheumatology. X-ray examinations were carried out to divide patients into early, middle, and late KOA stages. Symptoms suggestive of any other chronic inflammatory disease; diabetes; history of corticosteroid treatment; any other form of arthritis; cancer; and family history of osteoarthritis.
Quantitative RT-PCR and microarray analysis
Total RNA was isolated from human serum samples or cells samples using Trizol reagent (Thermo Fisher Scientific) and the RNA purity was detected using spectrophotometer. The synthesis of cDNA were performed by the First Strand cDNA Synthesis Kit (Fermentas, Thermo Fisher Scientific). The real-time PCR were carried out on ABI 7500 Fast Real-Time PCR system (Applied Biosystems, USA) by using SYBRGreen PCR Kit (Takara, Dalian, China). These reactions were incubated at 95°C for 5 min, followed by 40 cycles of 95°C for 30 s, 60°C for 40 s, 72°C for 30 s. Relative quantitation of the gene expression was normalized by β-actin mRNA level following the 2−ΔΔCt method and relative to the control group. Primers used at this study: S10012: 5′-ACAGCCTGAGTGTCTTGTTT-3′ and 5′-ACTGATCCTCTGCTCCAGTG-3′; GAPDH 5′-TCACTATCGGCAATGAGCGG-3′ and 5′-TTGGCTACA ACTACAGGGCT-3′.
Total RNA was isolated from mouse using an RNeasy Mini Kit (Qiagen) and microarray were performed using SurePrint G3 Mouse Gene Expression 8 × 60 K (Agilent Technologies). Data were analyzed by Agilent Feature Extraction software (version 10.10.1.1).
ELISA kit analysis
Serum samples were collected at 1000g for 10 min and used to measure Cartilage oligomeric matrix protein (COMP, H211), TNF-α (H052), IL-1β (H002), IL-6 (H007), and IL-18 (H015) levels using ELIST kits according to the manufacturer’s protocol (Nanjing Jiancheng Institute of Biological Engineering). Absorbance values were measured using a microplate reader (Thermolab systems, Helsinki, Finland) at 450 nm.
Animals model for vivo model
The study was approved by the Ethics Committee of Animal Experiments of Minhang Branch of Yueyang Hospital of Integrated Traditional Chinese and Western Medicine affiliated to Shanghai University of Traditional Chinese Medicine. C57BL/6J mice were purchased from Animal laboratory of Shanghai University of Traditional Chinese Medicine. Number of sham group = 6 and number of OA group = 6. Resection of the medial collateral ligament and medial meniscus was performed using a surgical microscope under anesthesia (50 mg/kg pentobarbital sodium). After 10 weeks, mice were analyzed. Under anesthesia (50 mg/kg pentobarbital sodium), mice were sacrificed using carotid.
Histological analysis
The bone tissues were fixed with 4% paraformaldehyde for 24 h. Tissue samples were embedded in paraffin, and the 5-μm – thick sections were stained with Safranin O and Fast Green. Tissue samples were obtained using fluorescence microscopy (Nikon Eclipse TE2000-U, Japan).
Osteoarthritis were assessed using Mankin OA Scoring System: 0–11, changes in articular cartilage structure; 0–8, safranin O staining; 0–2, hypertrophic chondrocytes.
Cell culture and treatment for vitro model
MC3T3-E1 cells were maintained in DMEM supplemented with 10% FBS in a humidified atmosphere of 5% CO2 and 95% O2 at 37°C. S100A12 (5′-CCCACCCAGGTTGGTTTCTA-3′ and 5′-AGGGCTAAGATGAAGCCTGA-3′), siS100A12 (sc-43346, Santa Cruz Biotechnology), S100A12+siNLRP3 (sc-45470, Santa Cruz Biotechnology), siS100A12+NLRP3 (5′-GGCTTCGAATGTTGAGGTAATG-3′), and negative mimics (5′-TTCTCCGAACGTGTCACGT-3′) were transfected into cell using Lipofectamine 2000 (Thermo Fisher Scientific, Waltham, MA, USA) according to the manufacturer’s protocol. After 48 h of transfection, cell was induced with 100 ng of LPS for 24 h.
Western blot
Tissues or cells samples were lysed in RIPA buffer containing a protease inhibitor. Protein concentrations were determined using a bicinchoninic acid (BCA) protein kit. Total protein was performed using a 10% SDS-PAGE, and the resolved proteins were transferred to polyvinylidene difluoride (PVDF). Immunoblotting was performed overnight at 4°C with primary antibodies to S100A12 (ab272713, 1:1000, abcam), NLRP3 (sc-518122, 1:1000, Santa Cruz Biotechnology), MMP-9 (ab76003, 1:1000, abcam), and GAPDH (ab181602, 1:10,000, abcam) antibodies, which were blocked with 5% bovine serum albumin (BSA). Membranes were treated for 1 h at room temperature with goat anti-rabbit peroxidase-labeled secondary antibody (sc-2004, 1:5000, Santa Cruz Biotechnology). Western blots were prepared using the enhanced chemiluminescence kit (Thermo Fisher).
Statistical analysis
Numerical data were recorded as the mean ± standard deviation (SD). Statistically significant differences were considered to be p < 0.05. One-way analysis of variance (ANOVA) were made group comparisons for multiple group two-tailed t-tests were made Group comparisons for two group.
Results
Clinical severity of serum S100A12 levels in patients with osteoarthritis
We first determined the levels of S100A12 in CT-screened osteoarthritis patients (Figure 1(a)). Serum S100A12 levels in patients with osteoarthritis were markedly higher than those of normal group (Figure 1(b)). In addition, S100A12 levels of serum and bone tissue from mice model of osteoarthritis were also markedly higher than those of sham group (Figure 1(c) and (d)). These results showed that S100A12 may be involved in the disease progression of osteoarthritis.
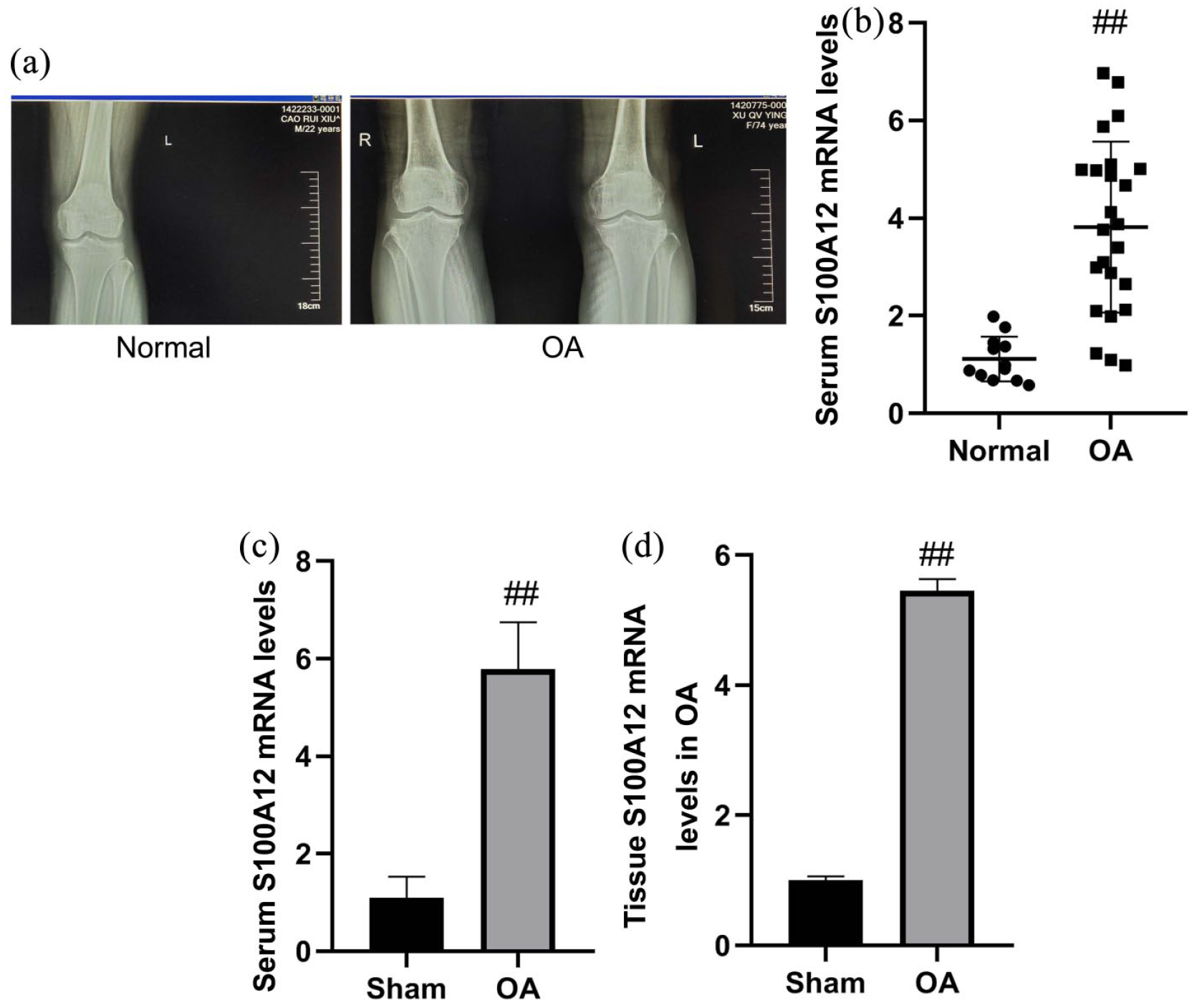
Clinical severity of serum S100A12 levels in patients with osteoarthritis. CT for patients with osteoarthritis (a), serum S100A12 levels in patients with osteoarthritis (b), serum and bone tissue of S100A12 levels in mice model of osteoarthritis (c and d).
S100A12 induced inflammation in in vitro model of osteoarthritis
To investigate the function of S100A12 in in vitro model of osteoarthritis, we regulated S100A12 expression using S100A12 plasmid or si-S100A12 plasmid. As a result, S100A12 plasmid markedly increased the expression level of S100A12, and observably induced the levels of TNF-α, IL-1β, IL-6, and IL-18 in vitro (Figure 2(a)–2(e)). By contrast, siS100A12 plasmid observably decreased the expression level of S100A12, and observably suppressed the levels of TNF-α, IL-1β, IL-6, and IL-18 in vitro (Figure 2(f)–2(j)). These data suggested that S100A12 might be a potential therapeutic strategy for osteoarthritis.

S100A12 open inflammation in vitro model of osteoarthritis. S100A12 expression (a), TNF-α, IL-1β, IL-6, and IL-18 levels (b–e) in vitro model of osteoarthritis by over-expression of S100A12; S100A12 expression (f), TNF-α, IL-1β, IL-6, and IL-18 levels (g–j) in vitro model of osteoarthritis by down-regulation of S100A12.
S100A12 induced NLRP3 inflammasome in in vitro model of osteoarthritis
Genotyping result showed that NLRP3 expression was memorably up-regulated in the regulation gene of S100A12 over-expression (Figure 3(a) and (b)). NLRP3 was a key target of S100A12 in in vitro model of osteoarthritis (Figure 3(c)). Over-expression of S100A12 memorably induced the protein expression of S100A12, NLRP3 and MMP-9 in vitro (Figure 3(d)–(g)). Down-regulation of S100A12 memorably suppressed the protein expression of S100A12, NLRP3, and MMP-9 in vitro (Figure 3(d), 3(h)–(j)). IF showed that over-expression of S100A12 memorably induced the protein expression of NLRP3 in vitro (Figure 3(k)). This result suggested that S100A12-mediated NLRP3 inflammasome at inflammation response of OA.
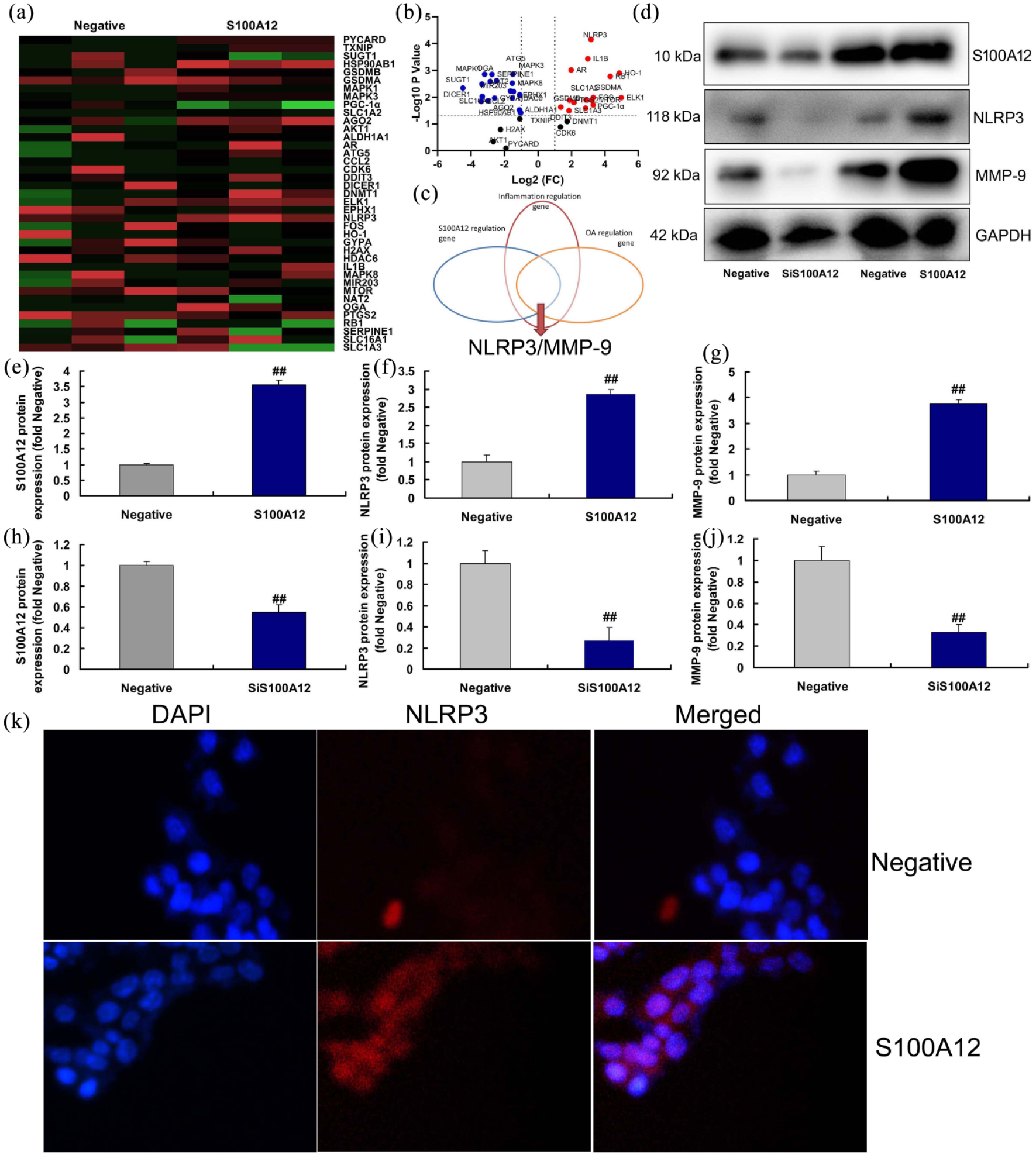
S100A12 induced NLRP3 inflammasome in vitro model of osteoarthritis. Heat map and volcano figure (a and b), analysis result (c), Western blot for S100A12/NLRP3/MMP-9 protein expression (d), statistics for S100A12/NLRP3/MMP-9 protein expression (e–g) in over-expression of S100A12; statistics for S100A12/NLRP3/MMP-9 protein expression (h–j) in down-regulation of S100A12, NLRP3 protein expression (IF, k).
NLRP3 was involved in the effects of S100A12 on inflammation in in vitro model of osteoarthritis
Over-expression of NLRP3 significantly induced the protein expression of NLRP3 and MMP-9, significantly increased the levels of TNF-α, IL-1β, IL-6, and IL-18 in vitro by si-S100A12 plasmid (Figure 4(a)–(g)). Down-regulation of NLRP3 significantly suppressed the protein expression of NLRP3 and MMP-9, significantly decreased the levels of TNF-α, IL-1β, IL-6, and IL-18 in vitro by S100A12 plasmid (Figure 4(h)–(n)).

NLRP3 participated in the effects of S100A12 on inflammation in vitro model of osteoarthritis. Statistics for NLRP3/MMP-9 protein expression (a and b), western blot for NLRP3/MMP-9 protein expression (c), TNF-α, IL-1β, IL-6, and IL-18 levels (d–g) in over-expression of NLRP3; statistics for NLRP3/MMP-9 protein expression (h and i), western blot for NLRP3/MMP-9 protein expression (j), TNF-α, IL-1β, IL-6, and IL-18 levels (k–n) in down-regulation of NLRP3.
S100A12 accelerated inflammation in mice model of osteoarthritis by NLRP3
We further validated the NLRP3 function in the effects of S100A12 in inflammation of osteoarthritis model. S100A12 recombinant protein could significantly induced the protein expression of NLRP3 and MMP-9, significantly promote bone cell fibrosis (HE staining), significantly increase COMP, osteophytes area, and modified Mankin osteoarthritis score, and significantly enhanced the levels of TNF-α, IL-1β, IL-6, and IL-18 in mice model of osteoarthritis (Figure 5). NLRP3 inhibitor significantly suppressed the protein levels of NLRP3 and MMP-9, significantly prevented bone cell fibrosis (HE staining), significantly inhibited COMP, osteophytes area, and modified Mankin osteoarthritis score, and significantly reduced the levels of TNF-α, IL-1β, IL-6, and IL-18 in mice model of osteoarthritis by S100A12 recombinant protein (Figure 5).
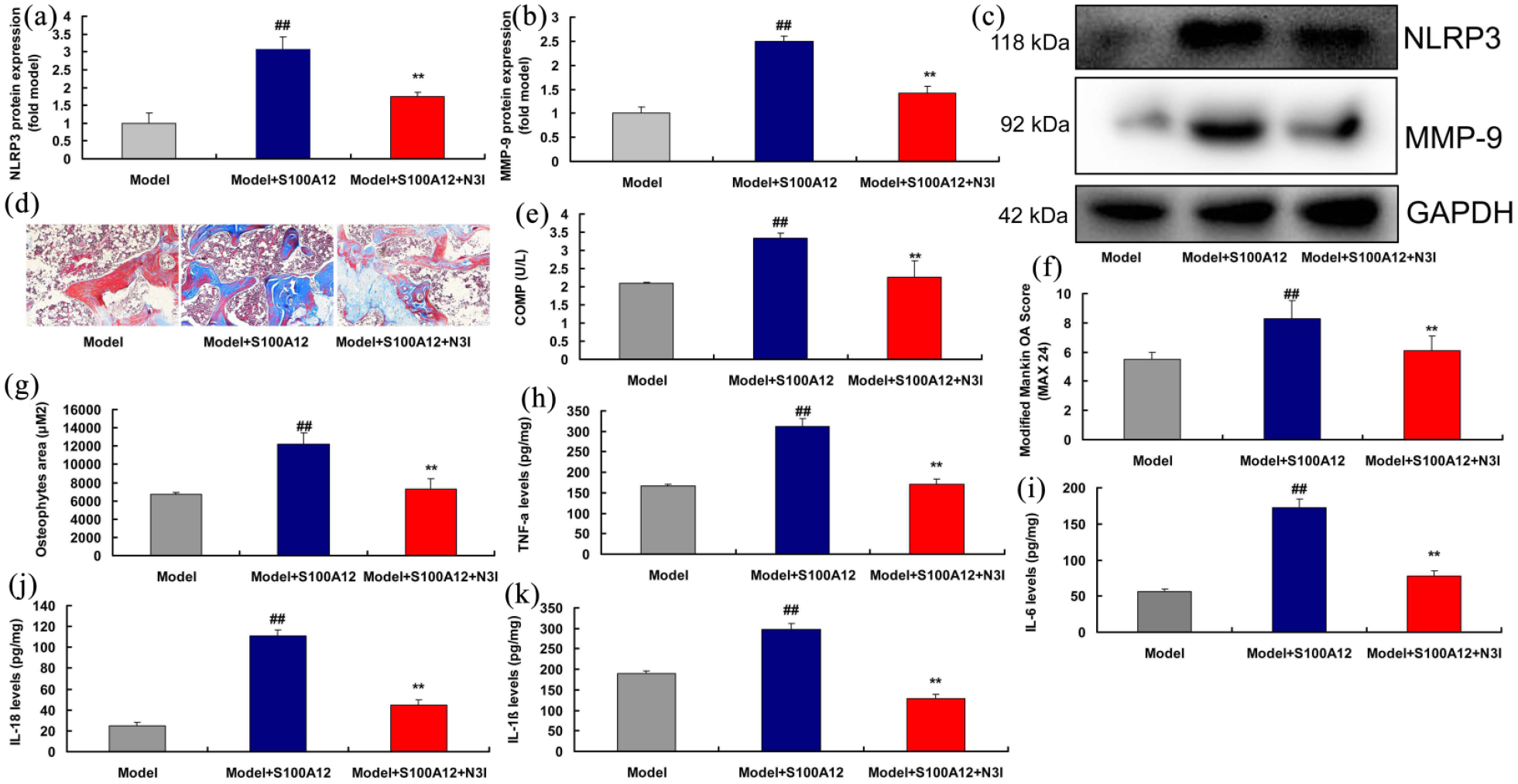
S100A12 accelerated inflammation in mice model of osteoarthritis by NLRP3. Statistics for NLRP3/MMP-9 protein expression (a and b), western blot for NLRP3/MMP-9 protein expression (c), bone tissue (HE staining, d), COMP (e), osteophytes area (f), modified Mankin osteoarthritis score (g), and TNF-α, IL-1β, IL-6, and IL-18 levels (h–k).
Discussion
OA is a common chronic bone and joint disease, which is characterized by degenerative changes and destruction of articular cartilage, secondary hyperostosis. In terms of clinical manifestation, it is mainly characterized by repeated and progressive pain and swelling of the joint, along with joint deformities and movement disorders. 12 The etiology of OA is not yet fully understood. 13 In recent years, accumulative studies have shown that various cytokines play important roles in the occurrence and development of OA, which are closely associated with the functional changes of synovium, articular cartilage, and subchondral cartilage. A large number of cytokines and proteins are involved in the pathological changes of OA. 14 This experiment determined that Serum S100A12 levels in patients with osteoarthritis were higher than those of normal group. Feng et al. 15 showed that S100A12 was associated with brain inflammation in severe traumatic brain injury. These results indicate that S100A12 might be a potential treatment target against OA, and its mechanism needs to be further study. However, this paper used MC3T3-E1 cells for vitro model, and which was an insufficient for this experiment, we will use more cell model to checking our conclusion. This experiment was not executed any power calculation for estimation of sample size selected, which was the limitation of the study.
S100A12 has a high serum response to various inflammatory reactions, such as rheumatoid arthritis, ulcerative colitis, and various types of tumors, which is associated with most inflammatory diseases.16,17 In the present study, S100A12 open inflammation in vitro model of osteoarthritis. Moreover, these results indicate that S100A12 mediated inflammation might be a potential target against OA.
A large number of studies have indicated that NLRP3 and Caspase-1 are the critical transition points for the activation of inflammasome. 18 Inflammatory factors, such as IL-1β, are the end-products after activation of inflammasome, which are the main key points of research on the prevention and treatment of metabolic diseases, such as acute gouty arthritis.19,20 Our results revealed that S100A12 induced NLRP3 inflammasome in vivo and vitro model of osteoarthritis. NLRP3 participated in the effects of S100A12 on inflammation in vitro model of osteoarthritis. Kim et al. 21 reported that S100A12 activated NLRP3 inflammasome in airway epithelial cells. This suggests that the inhibition of S100A12-induced NLRP3 inflammasome is a potential target against OA.
The content of MMP-1, MMP-2, MMP-3, MMP-9, MMP-13, and MMP-14 in the serum and joint fluid of OA patients is significantly higher than that of the normal control group.22,23 The imbalance can cause the excessive degradation of collagen, proteoglycan, and elastic fibers in the articular cartilage, thereby resulting in the destruction and degeneration of articular cartilage, which is the basic link of OA.24,25 We found that S100A12 induced MMP-9 protein expressions in vivo and vitro model of osteoarthritis. Jiang et al. 26 indicated that S100A12 induced MMP-2 and MMP-9 expression in HASMCs. These results confirmed that S100A12 induced MMP-9 expression by NLRP3 inflammasome in OA to promote inflammation reactions.
Conclusions
In conclusion, we found that S100A12 expression was up-regulated in patients with osteoarthritis and promoted inflammation of osteoarthritis via activation of MMP-9 protein expressions by NLRP3 inflammasome in mice model or vitro model. In conclusion, our results demonstrate that S100A12 played a powerful role in inflammation of osteoarthritis, and providing a potential target for the regulation of inflammation and bone cell impairment of osteoarthritis.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
The research protocols were approved by the Ethics Committee of the Minhang Branch of Yueyang Hospital of Integrated Traditional Chinese and Western Medicine affiliated to Shanghai University of Traditional Chinese Medicine (No. 201905061781).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Nation Natural Science Foundation of Changning (Grant No. 20194Z002).
Informed consent
Written informed consent was obtained from all participants.
