Abstract
We examined the presence of sialyl glycoconjugates in specific granules from murine bone marrow eosinophils. Lectin cytochemistry using Maackia amurensis lectin II (MAL II) specific for sialyl α-2,3 galactose residues demonstrated positive labeling in both immature and mature specific granules. Pretreatment with Clostridium neuraminidase or keratanase II eliminated the positive labeling of MAL II in the specific granules. High iron diamine-thiocarbohydrazide-silver proteinate physical development (HID-TCH-SP-PD) staining, which is specific for sulfated glycoconjugates, also positively labeled immature specific granules lacking crystalloids but not mature granules with crystalloids. Pretreatment with a combination of chondroitinase ABC and keratanase, or a combination of chondroitinase ABC and keratanase II, eliminated the positive labeling obtained with HID-TCH-SP-PD. These results indicate that the sialyl residues detected by MAL II are expressed as terminal sugar residues of keratan sulfate proteoglycan, which appears to be of the corneal type in view of its sensitivity to keratanase and keratanase II.
E
Materials and Methods
Mice
Ten-week-old female (C57BL/6 x DAB/2) F1-BDF1, C57BL/6, and BALB/c mice were purchased from Japan SLC (Shizuoka, Japan).
Tissue Preparation
Bone marrow samples were fixed in half-strength Karnovsky's fixative composed of 2% paraformaldehyde and 2.5% glutaraldehyde in 0.1 M phosphate buffer, pH 7.4 (PBS). They were then dehydrated in a graded ethanol series at −35C. After infiltration with Lowicryl K4M resin (Polysciences; Washington, PA), the specimens were placed in capsules with some fresh resin. Polymerization was performed initially at −35C for 24 hr and then at 20C for another 24 hr under indirect, diffuse UV irradiation using a low-temperature polymerization apparatus TTP 010 (Balzer Union; Lichtenstein). Ultrathin sections were cut with an Ultracut N (Reichert-Jung; Vienna, Austria) ultramicrotome and collected on Formvar-coated gold grids.
High Iron Diamine-Thiocarbohydrazide-Silver Proteinate Physical Development (HID-TCH-SP-PD) Staining (Table 1)
The specimens were stained for 18 hr at room temperature (RT) with a high iron diamine solution consisting of 1.6 ml 40% FeCl3, 120 mg N,N-dimethyl-m-phenylene-diamine-(HCl)2 (Sigma Chemical; St Louis, MO), and 20 mg N,N-dimethyl-p-phenylene-diamine-HCl (Sigma) in 50 ml distilled water, final pH 1.4. The grids were immersed in 1% thiocarbohydrazide (Sigma) in 20% acetic acid solution for 60 min at RT, washed serially with 10% and 5% acetic acid, and finally washed with distilled water. The sections were then stained with 1% silver proteinate (Merck; Darmstadt, Germany) solution for 60 min and finally developed with a physical developer IntenSE M kit (Amersham International; Poole, UK) for 3 min at RT. The sections were finally counterstained with 2% aqueous uranyl acetate for 3 min.
Lectin Staining (Table 1)
Ultrathin sections were incubated in PBS containing 1% BSA (PBS-BSA) for 30 min at RT. The sections were then incubated in biotinylated Maackia amurensis lectin II (MAL II) or Sambucus nigra (SAN) (Vector Laboratories; Burlingame, CA) lectin dissolved in PBS-BSA overnight at 4C. After two washes with PBS, the sections were incubated in 15-nm colloidal gold-labeled streptavidin (1:40) dissolved in PBS-BSA. The sections were then washed twice with PBS, stained with 2% uranyl acetate for 3 min, and then with Millonig's lead for 1 min at RT.
Enzyme Degradation (Table 1)
For neuraminidase treatment, ultrathin sections were treated with 1 U/ml Clostridium neuraminidase (Sigma) in 0.1 M acetate buffer (pH 5.0) with 0.04 M CaCl2 for 18 hr at 37C according to the method of Roth et al. (1984). For keratanase II treatment, ultrathin sections were treated with 0.1 U/ml keratanase II (Seikagaku; Tokyo, Japan) in 0.05 M sodium acetate buffer (pH 6.5) for 18 hr at 37C according to the method of Ito et al. (1996). For keratanase treatment, ultrathin sections were treated with 2 U/ml keratanase (Pseudomonas sp.) (Seikagaku) in 0.05 M Tris-HCl buffer (pH 7.4) for 18 hr at 37C according to a modification of the method of Nakazawa et al. (1975) and the manufacturer's protocol. For chondroitinase ABC (Seikagaku) treatment, ultrathin sections were treated with 2 U/ml protease-free chondroitinase ABC in 0.2 M Tris-HCl (pH 8.0) for 18 hr at 37C according to the method of Goode et al. (1991). Control sections were incubated in the same buffer alone for the same period. Afterwards, sections were washed in distilled water three times for 10 min and then stained with MAL II lectin or HID-TCH-SP-PD as described above.
Electron Microscopy
Electron microscopic observation was performed with a Hitachi H-7000 transmission electron microscope at 75 kV accelerating voltage.
Morphometry
Morphometric analysis of colloidal gold labeling on the eosinophil-specific granules was performed using electron micrographs (magnification x 15,000). We measured the areas of the granules and colloidal gold particles using NIH Image (Version 1.58) imaging software from the National Institutes of Health (Bethesda, MD) on a personal computer and calculated the labeling intensity of colloidal gold on the granules. The granules cut in the middle were randomly chosen and measured in approximately 50 cells.
Statistical Analysis
Differences among groups were analyzed statistically by the Mann-Whitney test.
Results
Expression of Sialyl α-2,3 Galactose Residues in Murine Eosinophil-specific Granules
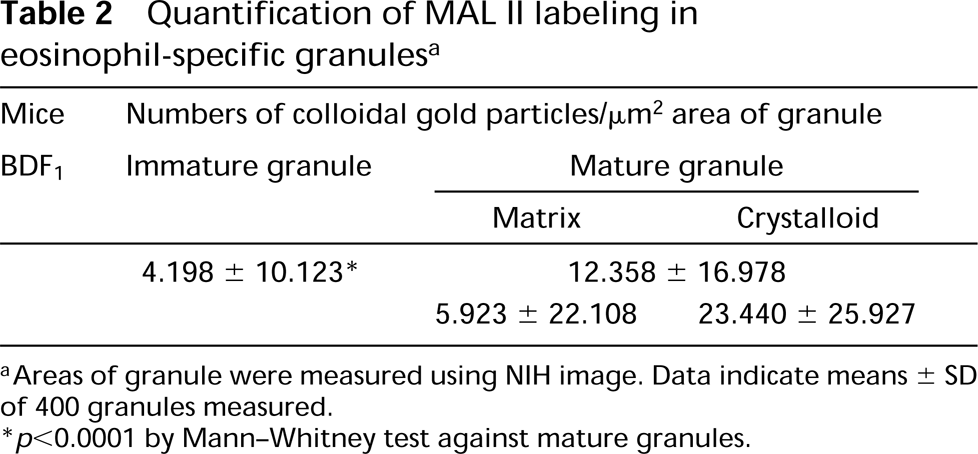
To examine whether eosinophil-specific granules express sialyl glycoconjugates, we performed lectin cytochemistry with MAL II lectin specific for sialyl α-2,3 galactose or SAN lectin specific for sialyl α-2,6 galactose. MAL II lectin positively labeled immature and mature specific granules (Figures 1A and 1B). The labeling was higher in mature granules than in immature granules (Table 2). The positive labeling was completely eliminated by pretreatment with Clostridium neuraminidase, not only in the specific granules but also other areas of eosinophils (Figure 1C). No labeling was observed with SAN lectin (not shown).
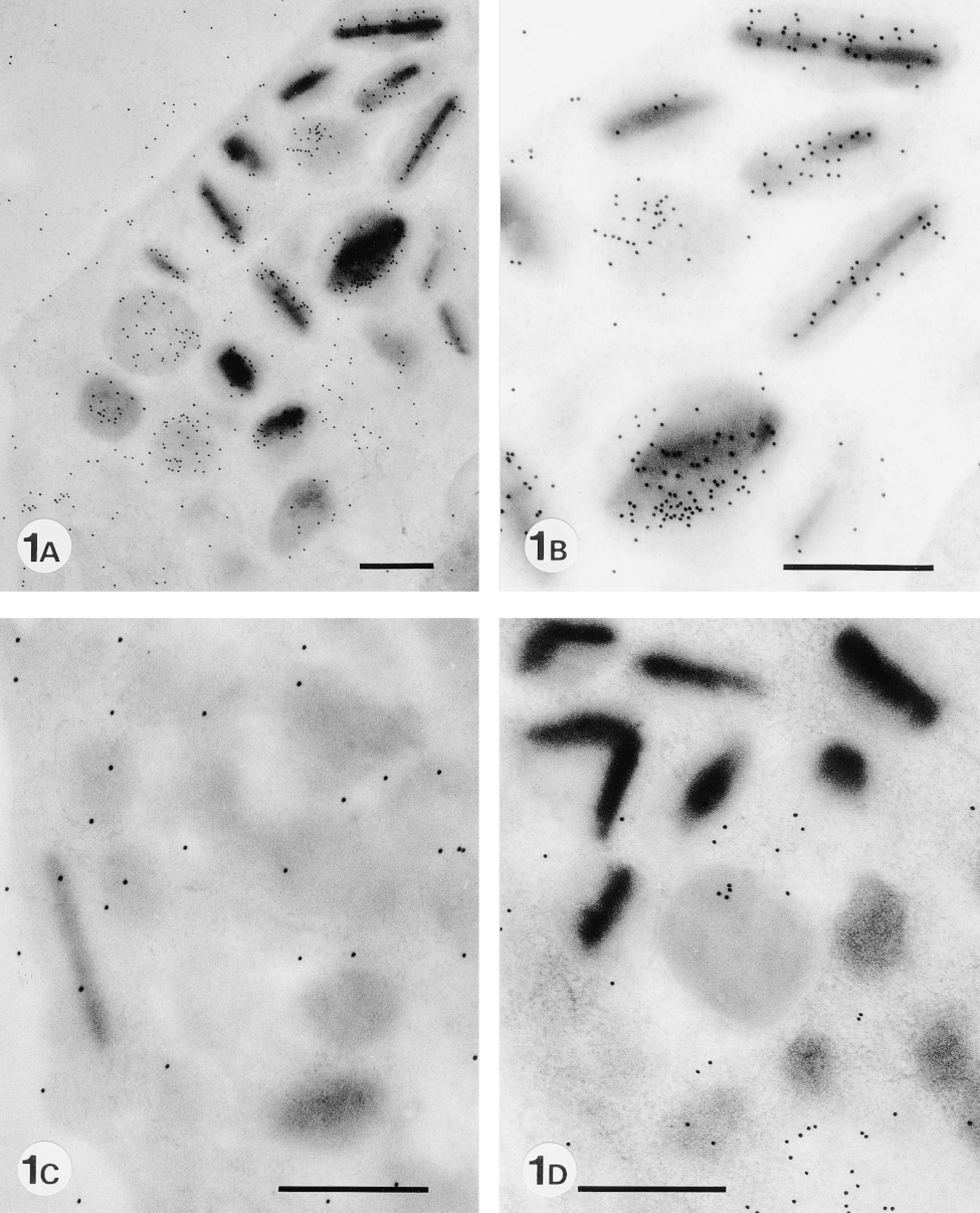
Expression of sialyl residues in murine eosinophil-specific granules with and without enzyme treatment. (A) MAL II lectin staining of eosinophil-specific granules. (B) A higher magnification of A. (C) MAL II lectin staining after pretreatment with Clostridium neuraminidase. Almost no labeling is observed. (D) MAL II lectin staining after pretreatment with keratanase II. Almost no labeling is seen except in cytoplasm. Bars = 0.5 μm.
Localization of Sialyl Residues in Eosinophil-specific Granules to Keratan Sulfate
To examine whether sialyl residues are linked to keratan sulfate, the sole proteoglycan that contains sialic acids (Toda and Seno 1970; Choi and Meyer 1975), MAL II lectin staining was performed after treatment with neuraminidase-free keratanase II. As shown in Figure 1D, the enzyme treatment completely eliminated the labeling of MAL II lectin in the specific granules.
HID-TCH-SP-PD staining showed various degrees of positive labeling in crystalloid-free immature granules (Figure 2A), consistent with the pattern of positive labeling with HID-TCH-SP in human and rabbit eosinophil specific granules reported by Parmley et al. (1982). The positive HID-TCH-SP-PD staining was slightly decreased by pretreatment with keratanase (Figure 2B), or keratanase II (not shown). This staining was not completely eliminated but was greatly decreased by pretreatment with chondroitinase ABC specific for chondroitin sulfate and dermatan sulfate (Figure 2C). Finally, staining was completely eliminated by a combination of chondroitinase ABC and keratanase (Figure 2D), or a combination of chondroitinase ABC and keratanase II (not shown). These results suggest that keratan sulfate in the specific granules is of the corneal type (Figure 3), because corneal-type keratan sulfate I is sensitive to both keratanase and keratanase II, whereas keratan sulfate II is sensitive to keratanase II but resistant to keratanase.
Specificity of stainings and glycosidases a

aHID-TCH-SP-PD, high iron diamine-thiocarbohydrazide-silver proteinate physical development; MAL II, Maackia amurensis lectin II; SNA, Sambucus nigra.
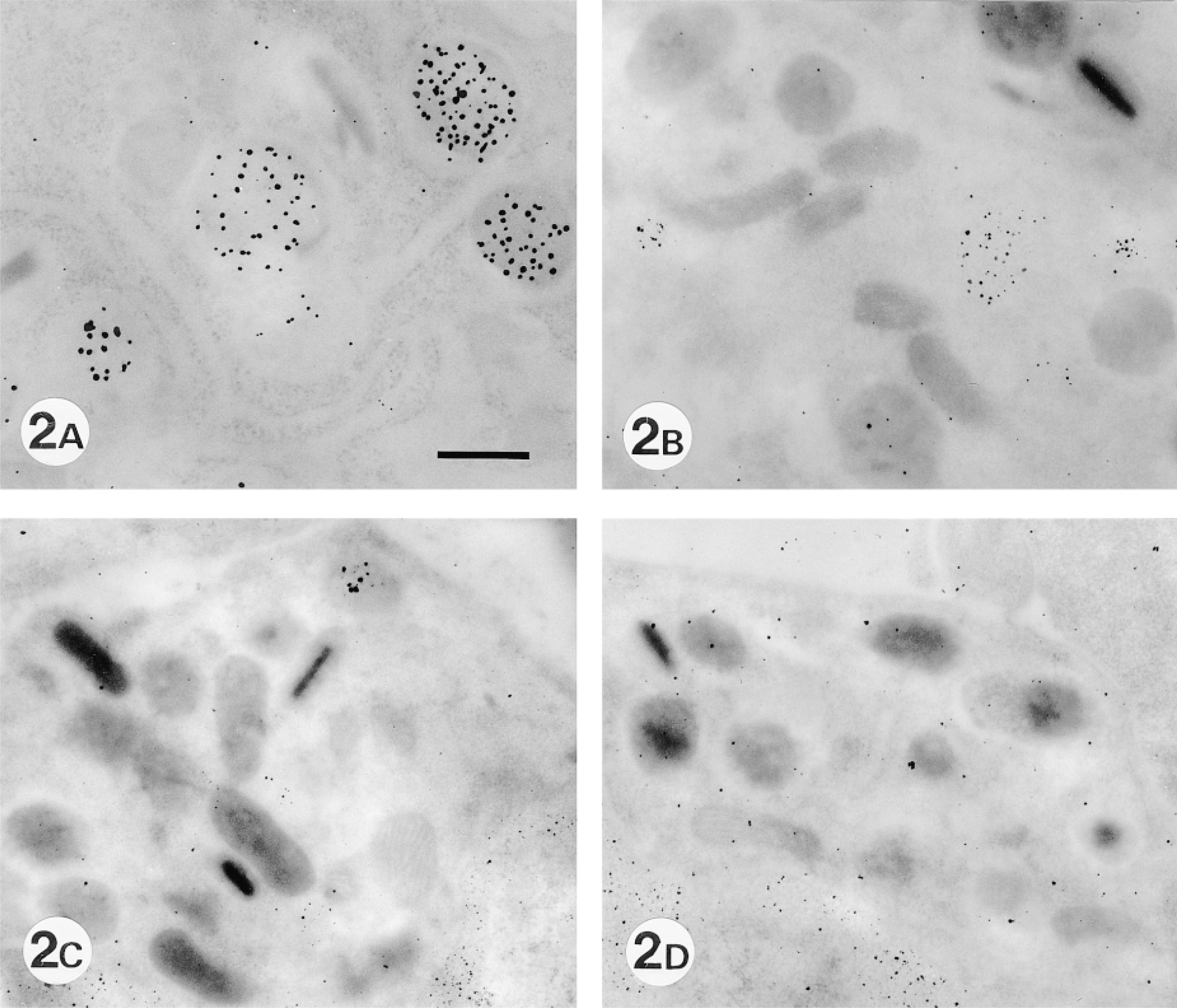
Localization of sialyl residues in eosinophil-specific granules with and without enzyme treatment. (A) HID-TCH-SP-PD staining of eosinophil-specific granules. Various degrees of labeling in crystalloid-free immature granules are seen. (B) HID-TCH-SP-PD staining after pretreatment with keratanase. Much labeling is still observed. (C) HID-TCH-SP-PD staining after pretreatment with chondroitinase ABC. Labeling is greatly decreased (D) HID-TCH-SP-PD staining after pretreatment with chondroitinase ABC and keratanase. No labeling is observed. Bar = 0.5 μm.
Discussion
The present studies, using a combination of lectin cytochemistry with MAL II specific for sialyl α-2,3 galactose residues and enzyme degradation methods, demonstrated that sialyl residues are expressed in eosinophil-specific granules. The sialyl residues were shown to be linked to sulfated proteoglycan using a combination of HID-TCH-SP-PD staining and enzyme degradation. These results confirm the presence of keratan sulfate in eosinophil-specific granules.
The nucleotide sugar CMP-N-acetylneuraminic acid is synthesized in the nucleus (Kean 1991), and glycosylation reactions occur in the lumen of the endoplasmic reticulum and Golgi apparatus (Hirschberg and Snider 1987; Verbert et al. 1987). This explains the difference in MAL II lectin cytochemical staining between pretreatment with neuraminidase and with keratanase II. Neuraminidase was able to eliminate all positive labeling of MAL II in other areas as well as the specific granules, whereas keratanase II eliminated the positive labeling of MAL II that was restricted to specific granules.
Keratan sulfate proteoglycans are classified into two types on the basis of their linkage to protein (Stuhlsatz et al. 1989) (Figure 3). Keratan sulfate I is linked to protein by GlcNAc → asparagine and is found in the cornea (Seno et al. 1965). The second type, keratan sulfate II, is linked to protein by O-glycosidic linkages between N-acetylgalactosamine and serine or threonine, and is found in cartilage and intervertebral disks (Anderson et al. 1964). These two types of keratan sulfate can be distinguished by their sensitivity to keratanase and keratanase II (Nakazawa et al. 1989). Keratan sulfate I is sensitive to both keratanases, whereas keratan sulfate II is sensitive to keratanase II but resistant to keratanase. Keratanase cleaves the internal β1-4 galactosidic linkages in keratan sulfate and requires at least one N-acetyl-glucosamine 6-sulfate residue adjacent to the galactose residue participating in the galactosidic linkage. Keratanase II cleaves β1-3 glucosaminidic linkages to galactose, as well as those in keratan sulfates to galactose-6-sulfate (Nakazawa et al. 1989). Keratanase contains a small amount of neuraminidase, but keratanase II is negative for contaminating neuraminidase. We next examined the sensitivity to these two enzymes using HID-TCH-SP-PD staining.
Eosinophil-specific Granules Are Sensitive to Both Keratanase and Keratanase II, Suggesting that They Contain Keratan Sulfate I
The distribution of keratan sulfates and their other functions are not clearly understood. Keratan sulfate I (corneal keratan sulfate) appears to play an important role in maintaining the transparency of the tissue in addition to the common role of proteoglycans, which control the concentration gradient of water in the stroma (Nakazawa et al. 1989). In comparison with keratan sulfate, sialic acids have been analyzed much more extensively. They occur mainly as terminal sugar residues, conferring chemical and biological diversity to glycoconjugates. One of the functions of sialic acids is related to their relatively large hydrophilic and acidic features, which exert physicochemical effects on glycoconjugates and their environment (Schauer et al. 1995). Their other function is in molecular and cellular recognition by masking galactose residues to prevent biological recognition or by making sialyl residues serve as recognition sites (Schauer et al. 1995). Sialyl residues expressed in eosinophil-specific granules may cooperate with sulfated residues in the safe storage of highly basic granule proteins by neutralizing them with their acidity, and enzymes by inactivating their activity.
The present study demonstrated that chondroitinase ABC and keratanase (or keratanase II) are necessary for completely eliminating the positive labeling produced by HID-TCH-SP-PD. Parmley et al. (1982) reported that testicular hyaluronidase partially removed the HID-TCH-SP-stained material, and suggested the presence of chondroitin 4- and 6-sulfate and other sulfated glycoconjugates. Metcalfe et al. (1982) demonstrated biochemically that the composition of sulfated glycosaminoglycans from human eosinophils was 70–81% chondroitin 4-sulfate, 9–12% chondroitin 6-sulfate, and 5–12% dermatan sulfate. Because chondroitinase ABC is specific for chondroitin sulfate and dermatan sulfate (Oike et al. 1982), the results of the present study are consistent with their findings. Furthermore, we clearly demonstrated that the remaining sulfated GAG in eosinophils is keratan sulfate.

Carbohydrate structure of keratan sulfates (I and II). Gal, galactose; GlcNAc, N-acetylglucosamine; Man, mannose; Fuc, fucose; Asn, asparagine; NeuAc, sialic acid; Ser, serine; Thr, threonine. From Stuhlsatz et al. (1989), with permission.
HID-TCH-SP-PD staining showed that murine eosinophil-specific granules have a staining pattern similar to those of human and rabbit, and also can be divided into five developmental stages. Parmley et al. (1982) stained human and rabbit developing eosinophils with HID-TCH-SP and divided their specific granules into five developmental stages: precursor granules, labeled in the center of the granule; immature granules Type I, labeled in both the granule center and periphery; immature granules Type II, labeled in the granule perpihery; immature granules Type III, no labeling and lack of a crystalloid core; mature granules, no labeling but a crystalloid core present. Lack of labeling with HID-TCH-SP(-PD) is consistent with the decreased distribution of radiosulfate in mature specific granules (Parmley et al. 1982).
Popken-Harris et al. (1998) reported that human eosinophil-specific granules in the process of condensation possess the proform of major basic protein (proMBP) (Barker et al. 1988; McGrogan et al. 1988) around the periphery and have MBP in the central area. The cDNA for MBP encodes a prepromolecule with a total length of 222 amino acids. PreproMBP includes a secretory leader of 15 amino acids, an acidic propiece of 90 amino acids (Shikata et al. 1993; Pop-ken-Harris et al. 1995), and a basic MBP portion of 117 amino acids. The propiece has an estimated pI of 3.9 and confers an overall acidic charge on proMBP. Popken-Harris et al. obtained eosinophils with specific granules by culturing umbilical cord stem cells with interleukin 5. However, the granules were in the process of condensation and did not become the mature specific granules containing a crystalloid core. These authors concluded that the conversion of proMBP to MBP occurs in the granule, and their results support the hypothesis that the propiece of proMBP functions to protect maturing eosinophils from the toxic MBP during granule processing. Furthermore, they pointed out that a lack of HID-TCH-SP labeling in immature granules Type III and mature granules is consistent with the almost total lack of labeling of proMBP in the condensing central area. However, their finding raises the question of whether toxic MBP that loses the propiece in the central area is harmful to specific granules.
Quantification of MAL II labeling in eosinophil-specific granules a
aAreas of granule were measured using NIH image. Data indicate means ± SD of 400 granules measured.
∗ p>0.0001 by Mann-Whitney test against mature granules.
The present study demonstrated that, in contrast to HID-TCH-SP-PD staining, sialyl residues are positive in mature specific granules (Figures 1A and 1B) and that their expression is higher in mature than in immature specific granules (Table 2). HID-TCH-SP-PD method detects sulfated residues by forming electrostatic linkage with ionized sulfated residues in the tissue section. Absence of labeling does not always suggest lack of sulfated residues but may also suggest the presence of cationic substances that neutralize the negative charges of sulfated residues. This concept is consistent with the results of low but positive labeling with autoradiography using 35SO4 2- and lack of labeling with HID-TCH-SP-PD staining in mature granules reported by Parmley et al. (1982). On the other hand, MAL II lectin binds sialyl α-2,3 galactose residues not with electrostatic strength but with more specific affinity. Therefore, sialyl residues can be detected by MAL II lectin as long as the residues are present in the tissue section. A biochemical study has demonstrated that GAGs may form complexes with lysosomal enzymes and inactivate them (Avila and Convit 1976; Avila 1978). Corresponding findings of masking of acid phosphatase reactivity have been reported in mature eosinophil granules (Wetzel et al. 1967; Bass et al. 1981). The present results of the positive labeling of sialyl residues and the negative labeling of HID-TCH-SP-PD in mature granules also support this concept. Sialyl residues would play a similar role to other sulfated GAGs in specific granules. Therefore, they appear to share with other sulfated GAGs the role of complexing with highly basic granule proteins and inactivating them during the maturation of specific granules. The presence of another acidic GAG, keratan sulfate, suggests that the functions of eosinophil-specific granules are mediated by many substances, including keratan sulfate and other sulfated GAGs.
The sialyl residues can be detected by a MAL II lectin in neutral buffer in contrast to sulfate residues in very acidic buffer, suggesting that the molecules can be isolated without denaturation for further analysis with techniques of molecular biology. Eosinophils play an important role in allergy and inflammatory diseases by releasing their granule proteins. Clarification of the mechanism for safe storage of highly basic granule proteins and their release will help the development of therapy for allergy or eosinophil-related diseases. Analysis focusing on these sialyl molecules will contribute to the purpose.
Footnotes
Acknowledgements
Supported in part by the Kodama Foundation for Research in Medical Science.
We wish to thank Mr S. Nonaka for technical assistance and Mr S. Imamura for secretarial assistance.
