Abstract
The aim of this study was to establish, using immunolabeling, whether the Kv1.5 K+ channel is present in the pacemaker of the heart, the sinoatrial (SA) node. In the atrial muscle surrounding the SA node and in the SA node itself (from guinea pig and ferret), Western blotting analysis showed a major band of the expected molecular weight, ~64 kD. Confocal microscopy and immunofluorescence labeling showed Kv1.5 labeling clustered in atrial muscle but punctate in the SA node. In atrial muscle, Kv1.5 labeling was closely associated with labeling of Cx43 (gap junction protein) and DPI/II (desmosomal protein), whereas in SA node Kv1.5 labeling was closely associated with labeling of DPI/II but not labeling of Cx43 (absent in the SA node) or Cx45 (another gap junction protein present in the SA node). Electron microscopy and immunogold labeling showed that the Kv1.5 labeling in atrial muscle is preferentially associated with desmosomes rather than gapjunctions.
P
Materials and Methods
Although the guinea pig was the principal species investigated, other species (ferret, mouse) had to be used because of the nature of the antibodies used and the type of experiment carried out. Procedures were carried out under license and in accordance with the regulations of the UK Animals (Scientific Procedures) Act 1986.
SA Node Tissue Dissection
Guinea pigs of either sex weighing 0.2-0.25 kg were sacrificed by stunning and cervical dislocation. Ferrets weighing 1 kg were anesthetized with IP sodium pentobarbital (90 mg/kg). The chest was opened and the heart was excised rapidly and placed in Tyrode's solution at 32–34C. The right atrium was separated from the ventricles and then the left atrium. The separated right atrium was opened by a longitudinal incision in the ventral wall (between the inferior and superior venae cavae) to expose the endocardial surface. The right atrium was trimmed to leave a preparation approximately 10 × 12 mm in the case of the guinea pig SA node and 15 × 15 mm in the case of the ferret SA node. Finally, the preparation, which included the entire SA node and some of the surrounding atrial muscle, was pinned down (endocardial surface up) on silicon rubber. Tyrode's solution contained (in mM): NaCl 93; NaHCO3 20; Na2HPO4 1; KCl 5; CaCl2 2; MgSO4 1; sodium acetate 20; glucose 10; insulin 5 U/ml; equilibrated with 95% O2 and 5% CO2 to give pH 7.4. After dissection, two guinea pig SA node preparations were electrophysiologically mapped at 32C as described previously (Honjo et al. 1999) to locate the leading pacemaker site.
Antibodies
Various primary antibodies were used: (a) a polyclonal antibody against the NH2 terminus of the human Kv1.5 K+ channel raised in rabbit (Mays et al. 1995); (b) a monoclonal antibody (MAb) against residues 252-270 of rat connexin43 (Cx43; gap junction channel protein) raised in mouse (Chemicon; Harrow, UK); (c) a polyclonal antibody against connexin45 (Cx45; another gap junction channel protein) raised in guinea pig (Coppen et al. 1998); (d) an MAb against a multiepitope cocktail to bovine desmoplakins, DPI and DPII (most abundant desmosomal proteins; Buxton and Magee 1992), raised in mouse (Progen; Heidelberg, Germany); and (e) a polyclonal antibody against desmoplakins DPI and DPII, raised in rabbit (Arnemann et al. 1993).
Western Blotting
After the SA node tissue dissection described above (from eight guinea pigs and one ferret), the thin intercaval region between the superior and inferior venae cavae (where the SA node is located) was separated from the posterior wall of the right atrium (including the crista terminalis and atrial appendage). A part of the left ventricular wall was also dissected, and all three types of cardiac tissue (intercaval region, posterior wall of right atrium, left ventricle) were rapidly frozen in liquid N2. The tissues were stored at −80C until the experiment. Whole tissue homogenates were prepared by pulverizing the frozen tissues on dry ice using a mortar and pestle. The powdered frozen tissue was thawed and lysed in SB20 (20% SDS, 0.15 M Tris, pH 6.8) and centrifuged at 4000 × g for 15 min. The supernatant from each tissue sample was separated from the pellets and 4 × Laemlli sample buffer was added to each supernatant sample; 4 × Laemlli sample buffer contained Tris-HCl 10 mM; EDTA 1 mM; glycerol 40%; SDS 2.5%; DTT 1 mM; β-mercaptoethanol 2%; bromophenol blue 0.01%, pH 6.8. Each sample was vortexed and warmed in a water bath at 80C for 15 min. Approximately 10 μl of supernatant from each tissue sample was run on a 12% SDS polyacrylamide gel and electrophoretically transferred to a nitrocellulose membrane. The membrane was blocked with 5% dried skimmed milk in PBS containing 0.1% Tween-20, pH 7.5 (PBS-T). PBS contained (in mM): NaH2PO4 20; Na2HPO4 80; NaCl 100, pH 7.5. After washing in PBS-T, the membrane was incubated with the anti-Kv1.5 primary antibody [diluted 1:5000 in PBS-T, 1% bovine serum albumin (BSA), and 2 mM NaN3] for 0.5 hr at room temperature (RT), overnight at 4C, and then again for 0.5 hr at RT. The membrane was thoroughly washed in PBS-T and incubated with goat anti-rabbit horseradish peroxidase-conjugated secondary antibody (diluted 1:3000 in PBS-T; Dako, High Wycombe, UK) for 1 hr. The membrane was again thoroughly washed and the enzyme activity was revealed using a chemiluminescence system (Supersignal ECL; Pierce, Rockford, IL). An equivalent gel was also run on 12% SDS polyacrylamide gel and stained with Coomassie Blue for total protein estimation in each sample. Gels and ECL films were scanned and the amount of protein in each sample was estimated using a custom-written densitometry program.
Immunofluorescence Double and Triple Labeling
After dissection and, in some cases, mapping of the activation sequence, the SA node preparation, still pinned down on silicon rubber, was photographed. The preparation was embedded at RT in 10% gelatin (porcine, type A; Sigma, Poole, UK) and preheated to 37C in 0.1 M phosphate buffer, which contained (in mM): Na2HPO4 77; NaH2PO4 23. The preparation was raised to allow penetration of the gelatin beneath the epicardial surface of the tissue. The gelatin-embedded tissue was allowed to cool to 4C to solidify the gelatin. The tissue was cut out with a surround of gelatin and all the pins (except a pin that indicated the primary pacemaker site in the electrophysiologically mapped preparations) were removed. The preparation was frozen at −50C in isopentane and stored at −80C until it was ready to be cryosectioned. A part of the liver from two guinea pigs was also removed, rapidly frozen in liquid N2, and stored at −80C until ready to be cryosectioned. Frozen serial sections (~20-μm thick) were cut from the tissue. In three guinea pig SA node preparations, sections were cut at ~1-mm intervals from the superior to the inferior part of the preparation perpendicular to the crista terminalis and through the intercaval region (see Figure 2A). In preparations of two other guinea pig and one ferret SA node, sections were cut at the level of the main branch from the crista terminalis (the level at which the center of the SA node is usually located). After cryosectioning, the sections were mounted on poly-L-lysine-coated glass slides (BDH; Poole, UK) and stored at −80C until use.
The sections were fixed by immersing the slides in methanol at −20C for 5 min and were washed three times with PBS, pH 7.4, over 0.5 hr. PBS contained (in mM): Na2HPO4 7.7; NaH2PO4 2.3; NaCl 150. The sections were blocked with 5% non-fat dried milk and 10 % normal donkey serum in PBS, either overnight at 4C or 1 hr at RT. The sections were then briefly washed in PBS. For double labeling, the sections were incubated with the primary antibodies (anti-Kv1.5, anti-Cx43, mouse anti-DPI/II, and rabbit anti-DPI/II diluted in 0.25% saponin, 10% normal donkey serum, and 6.15 mM NaN3 in PBS) for ~2-4 hr at 37C. In one experiment, sections were triple labeled. The sections were incubated with anti-Cx45 primary antibody (diluted in 1% BSA in PBS) for 2 hr at RT, followed by a mixture of anti-Kv1.5 and anti-Cx43 primary antibodies (diluted in 10% normal donkey serum and 0.25% saponin in PBS) for 2 hr at 37C. After incubation in the primary antibodies, the sections were washed several times in PBS over 0.5-1 hr and incubated with secondary antibodies conjugated to fluorescent markers (diluted either in 1.5% normal donkey serum and 1% BSA or 1% BSA in PBS) for 1 hr at RT. Secondary antibodies conjugated to FITC, TRITC, Cy3, and Cy5 were used. Sections were washed several more times in PBS over 0.5-1 hr and mounted with either Vectashield (H-1000; Vector Laboratories, Peterborough, UK) or Citifluor (AF1; Agar Scientific, Standsted, UK) mounting medium. Finally, coverslips were sealed with nailpolish.
Anti-Kv1.5 antibody was used at 1:250, antiCx-43 antibody at 1:1000, anti-Cx45 antibody at 1:50, rabbit anti-DPI/II antibody at 1:100, and mouse anti-DPI/II antibody was purchased in a ready-to-use form. All secondary antibodies were obtained from either Chemicon (Temecula, CA) or Dako (Carpinteria, CA). Anti-rabbit or anti-mouse secondary antibody conjugated to TRITC or FITC was used at 1:100 and anti-guinea pig secondary antibody conjugated to Cy3 or anti-mouse secondary antibody conjugated to Cy-5 was used at 1:500.
Immunolabeled sections were examined by confocal laser scanning microscopy (Leica TCS 4D or Leica TCS SP) equipped with argon, krypton, and helium-neon lasers, which allowed excitation at 488-, 568-, and 633-nm wavelengths for the detection of FITC, TRITC/Cy3, and Cy5, respectively. The images recorded were single optical sections and, in the case of double or triple labeling, the images were recorded sequentially. The images were saved and were processed using Corel Photo-Paint and Corel Draw software (Corel; Ottawa, ONT, Canada).
In most experiments, sections were double or triple labeled as described above. With all primary antibodies, it was checked that single labeling produced the same pattern of labeling.
Postembedding Immunogold Labeling for Electron Microscopy
For immunogold labeling, samples were freeze-substituted and embedded at low temperature in Lowicryl K4M (Carlemalm et al. 1981). NF1 mice weighing 25 g were sacrificed by cervical dislocation. Mouse atria were rapidly excised, immersed in 15% glycerol in PBS for 5 min, and plunge-frozen in N2 slush (liquid N2 cooled below its boiling point to approximately −205C). The frozen samples were freeze-substituted in methanol at −70C for 7 days. Infiltration was at −30C for 1-hr periods each in 1:1 and 2:1 mixtures of Lowicryl K4M:methanol, followed by pure Lowicryl (Agar Scientific) overnight. After a further incubation in pure resin for 1 hr, the samples were embedded in fresh resin in gelatin capsules and polymerized with UV light at −30C for 16 hr, and then at RT for up to a further 72 hr, using a Balzers FSU 010 low-temperature embedding unit. Ultrathin sections were prepared using a Reichert ultramicrotome and incubated at RT overnight with anti-Kv1.5 (1:250) or anti-Cx43 (1:500) antibodies. To reduce background labeling, the anti-Kv1.5 antibody was made up in PBS containing 5% non-fat dry milk and 10% normal serum as blocking agent. The anti-Cx43 antibody was used in PBS containing 1% BSA. The sections were then immersed in 10-nm gold/goat anti-rabbit or anti-mouse secondary antibodies (BioCell; Cardiff, UK) diluted 1:50 in PBS for 1 hr (Slot and Geuze 1984), washed, and examined in a Philips EM301 electron microscope.
Results
Detection of Kv1.5 K+ Channel Protein by Western Blotting Analysis
Western blotting analysis was used to detect the presence of the Kv1.5 K+ channel protein and to test the specificity of the anti-Kv1.5 antibody. Figure 1 shows Western blotting analysis of tissue samples prepared from different regions of guinea pig and ferret heart. The antibody detected a single or double band at the expected molecular weight (~64 kD) in the left ventricle, right atrium, and intercaval region (where the SA node is located) from guinea pig, and in the left ventricle and right atrium from ferret (see arrow in Figure 1A). In some experiments, there were other bands labeled corresponding to proteins with molecular weights of ~30 and ~130 kD (the latter can be seen in some lanes in Figure 1A). The former is probably a proteolytic product of Kv1.5, because it was much more prominent if tissue was not thawed into a high SDS solution, whereas the latter could be a dimer of Kv1.5. No bands were detected in guinea pig left ventricle in a control experiment in which the primary antibody was omitted (not shown). Densitometry of blots (e.g., Figure 1A) and parallel Coomassie-stained gels (not shown) was performed, and Kv1.5 labeling was normalized for protein loading and expressed as a fraction of that seen in the right atrium. Mean results from eight guinea pig hearts are shown in Figure 1B. The amount of Kv1.5 K+ channel protein in the SA node was not significantly different (ANOVA) from that in the right atrium and left ventricle (Figure 1B).
Detection of Kv1.5 K+ Channel Protein by Immunofluorescence Labeling
This series of experiments was carried out on guinea pig tissue. Figure 2A shows a diagram of a guinea pig SA node preparation. The SA node is located in the intercaval region (between the entrances of the superior and inferior venae cavae into the right atrium) and is bounded by the crista terminalis (a thick bundle of atrial muscle) on one side and by the atrial septum on the other. To the right of the crista terminalis (left in Figure 2A) is the right atrial appendage. The preparation was electrophysiologically mapped, and the isochrones in Figure 2A represent the time for the action potential to spread from the leading pacemaker site (asterisk) in the SA node. The leading pacemaker site was in the intercaval region, as expected. To study the expression of the Kv1.5 K+ channel protein in detail, confocal microscopy and immunofluorescence labeling were used. Sections were cut perpendicular to the crista terminalis (at level of arrows in Figure 2A) through the right atrial appendage, the crista terminalis, and the intercaval region (where the SA node is located).
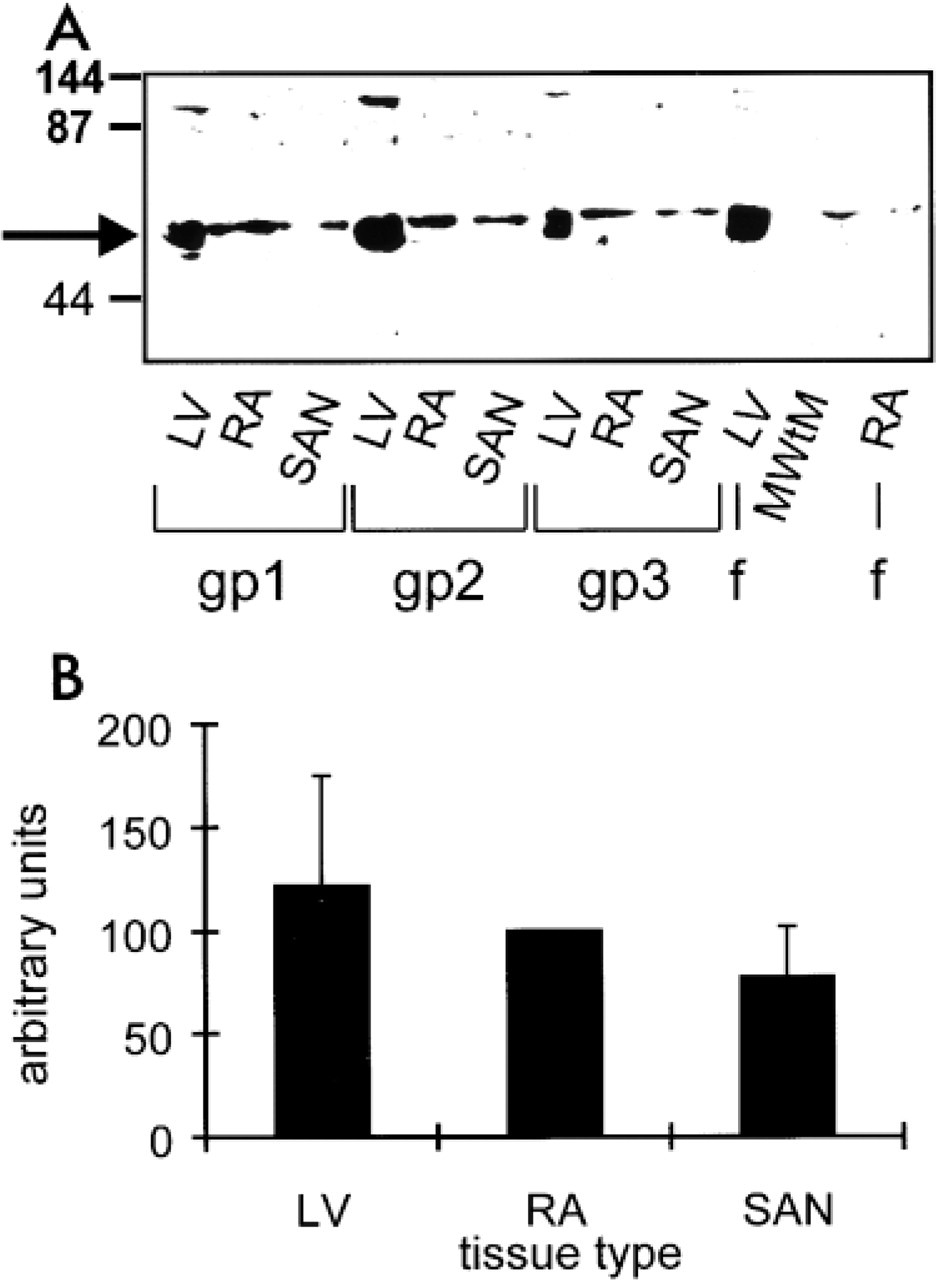
Western blotting analysis of tissue samples prepared from different regions of heart. (
Figures 3A and 3B show Kv1.5 and Cx43 labeling in the atrial muscle of the crista terminalis. Cx43 is a gap junction protein located throughout much of the heart to provide electrical coupling between cells. In the guinea pig and other species, Cx43 is absent in the SA node (ten Velde et al. 1995; van Kempen et al. 1995). In the present study, absence of Cx43 labeling was used as a marker for the SA node. As expected, Cx43 labeling was detected in the atrial muscle of the crista terminalis (Figure 3B). Kv1.5 labeling was also detected in the atrial muscle (Figure 3A). Throughout the atrial muscle, Kv1.5 labeling appeared in clusters (often as lines of labeling). Similar Kv1.5 labeling was seen in atrial tissue from five guinea pigs. Inspection of Figures 3A and 3B shows that the patterns of labeling of Kv1.5 and Cx43 at the level of confocal microscopy were similar. In Figure 3C, the Kv1.5 and Cx43 confocal micrographs are superimposed; this shows that the two labels were closely associated and, in some cases, co-localized, i.e., there are green (Cx43 label alone) and red (Kv1.5 label alone) spots adjacent to one another and some yellow spots (co-localization of two labels). Because in atrial muscle Cx43 is located in gap junctions at intercalated discs, these findings suggest that Kv1.5 is also present at the intercalated disc.
Figure 4A shows Kv1.5 and Cx43 labeling at the leading pacemaker site (as identified electrophysiologically; asterisk in Figure 2A) in the SA node. As expected, Cx43 signal was largely absent from the tissue (a small amount of Cx43 labeling in green can be seen in Figure 4A) but, in contrast, Kv1.5 labeling was detected (red labeling in Figure 4A). The pattern of Kv1.5 labeling in the SA node was different from that in the atrial muscle. Whereas in the atrial muscle Kv1.5 labeling was in clusters (Figure 3A), in the SA node Kv1.5 labeling was punctate (Figure 4A). A similar pattern of Kv1.5 labeling was seen in SA node tissue from five guinea pigs.
In the intercaval region, towards the interatrial septum, Cx43 labeling was again detected (not shown). In this region, Kv1.5 labeling was also detected (not shown). The pattern of Kv1.5 and Cx43 labeling was similar to that in the crista terminalis. The two labels were closely associated at the level of resolution afforded by confocal microscopy, and appeared in clusters.
Figure 2B summarizes the distribution of Kv1.5 and Cx43 labeling in different regions of the guinea pig SA node. The result shown is typical of three preparations, two of which were electrophysiologically mapped. The black zone represents the SA node, in which punctate Kv1.5 labeling was detected but Cx43 labeling was not detected. The width of the black zone varies from the superior to the inferior part of the preparation. The dark gray zone is the region of the crista terminalis and the intercaval region, in which both Kv1.5 and Cx43 labeling was detected. The extent of the black zone in Figure 2B, in which there was punctate Kv1.5 labeling but no Cx43 labeling, is outlined in Figure 2A (dotted line). The leading pacemaker site (asterisk) was located in this zone.
No immunofluorescence in the atrial muscle or the SA node was detected when the primary antibodies were omitted (not shown).
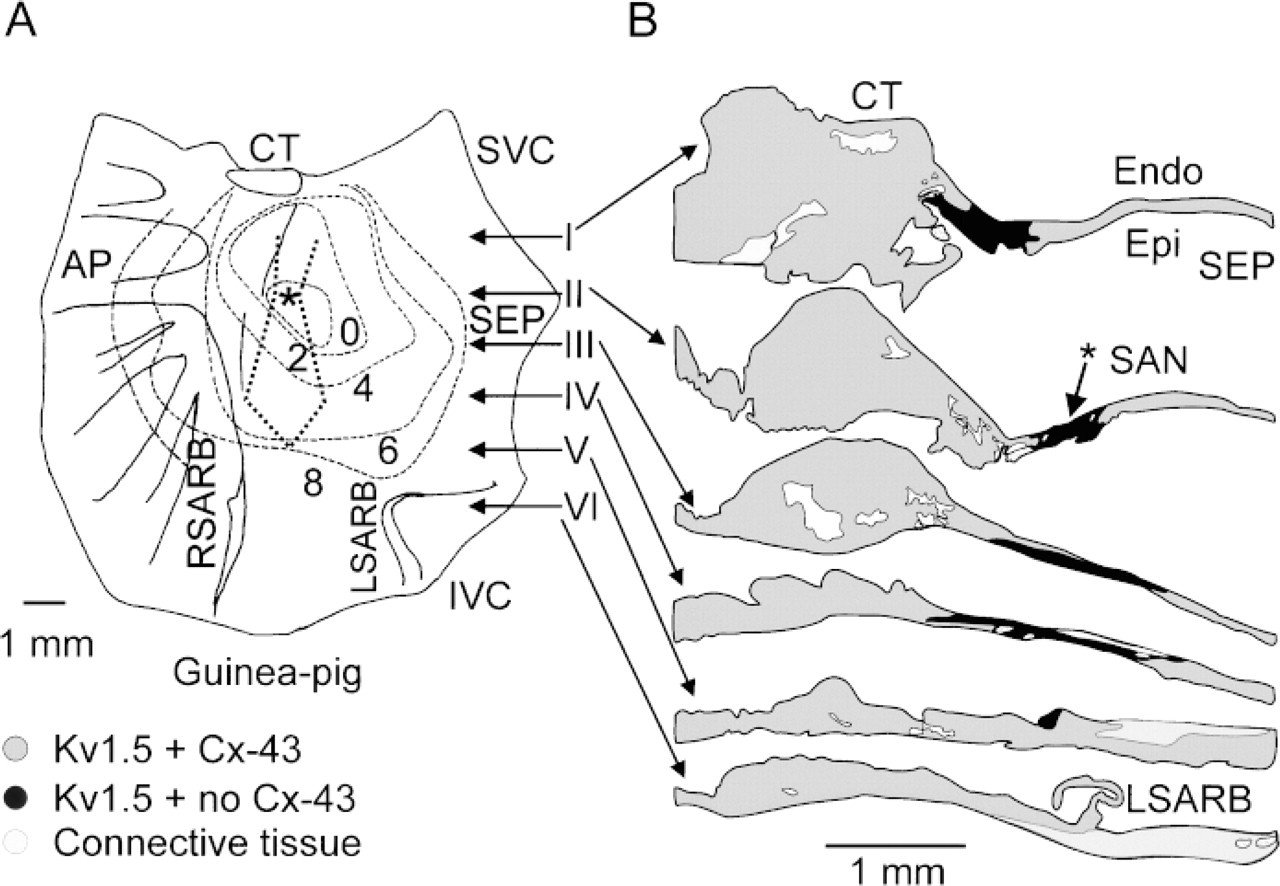
Summary of distribution of Kv1.5 and Cx43 immunofluorescence labeling in different regions of guinea pig SA node. (
Is Kv1.5 Co-localized with Another Gap Junction Protein in SA Node?
In atrial muscle, Kv1.5 labeling was closely associated with Cx43 labeling (Figures 3A and 3B), whereas in the SA node, although Cx43 labeling was mainly absent, Kv1.5 labeling was still observed (Figure 4A). Other connexins, e.g., Cx45, are known to be present in the SA node (Coppen et al. 1999), and it is possible that Kv1.5 is associated with these connexins rather than Cx43. To answer this question, triple immunofluorescence labeling with rabbit anti-Kv1.5, mouse anti-Cx43, and guinea pig anti-Cx45 antibodies was carried out. The ferret rather than the guinea pig SA node had to be used because the anti-Cx45 antibody was raised in guinea pig. Figure 5 shows the distribution of Kv1.5 (Figure 5A), Cx45 (Figure 5B), and Cx43 (Figure 5C) labeling in the ferret SA node. As in the guinea pig SA node, Cx43 labeling was largely absent but Kv1.5 labeling was detected (Figure 5C). The pattern of Kv1.5 labeling in the ferret SA node (Figure 5A) was similar to its distribution in the guinea pig SA node (Figure 4A): punctate. In the ferret SA node, Cx45 labeling was detected and was also punctate (Figure 5B). Figure 5D, in which the Kv1.5 and Cx45 confocal micrographs are superimposed, shows that the Kv1.5 and Cx45 labeling was rarely associated. There are many green (Kv1.5 label alone) and red (Cx45 label alone) spots with different distributions and few yellow (co-localization of two labels) spots. No immunofluorescence was detected when the primary antibodies were omitted (not shown).
Is Kv1.5 Co-localized with Desmosomal Proteins?
In the SA node, because Kv1.5 labeling was only sometimes spatially associated with Cx45 labeling, it is possible that Kv1.5 is associated with a connexin other than Cx43 and Cx45. However, it would be surprising if Kv1.5 was actually located within gap junctions. A more likely explanation is that, in atrial muscle, Kv1.5 is present in another structure alongside Cx43-containing gap junctions at the intercalated disc. Desmosomes are also present at the intercalated disc in atrial muscle. Guinea pig tissue was double labeled with anti-Kv1.5 and anti-DPI/II antibodies (DPI/II are abundant desmosomal proteins; Buxton and Magee 1992). When the experiment was first attempted, the anti-Kv1.5 and anti-DPI/II antibodies were added simultaneously. Although there was good DPI/II labeling, there was no detectable Kv1.5 labeling. This suggested that the DPI/II labeling was blocking Kv1.5 labeling (perhaps because the antibody binding sites are physically close). The labeling procedure was subsequently revised and the Kv1.5 antibody was added first and then ~4 hr later the anti-DPI/II antibody was added; in this case, both Kv1.5 and DPI/ II labeling was observed. Figures 3D and 3E show Kv1.5 and DPI/II labeling in the atrial muscle of the crista terminalis. Throughout the atrial muscle, DPI/II labeling, like Kv1.5 labeling, was in clusters. Figure 3F, in which the Kv1.5 and DPI/II confocal micrographs are superimposed, shows that the Kv1.5 and DPI/II labeling was co-localized. There are few red (Kv1.5 label alone) and green (DPI/II label alone) spots and many yellow (co-localization of two labels) spots. Figures 4B and 4C show Kv1.5 and DPI/II labeling in the SA node. DPI/II labeling in the SA node was punctate (Figure 4B), unlike DPI/II labeling in the atrial muscle (Figure 3E). Figure 4C shows that in the SA node the Kv1.5 and DPI/II labeling was also co-localized. There are few red (Kv1.5 label alone) and green (DPI/II label alone) spots and many yellow (co-localization of two labels) spots. In the experiment shown in Figures 4B and 4C, an adjacent section was double labelled for Kv1.5 and Cx43, and in the region shown in Figures 4B and 4C, Cx43 labeling was largely absent (although Kv1.5 labeling was present; not shown). This confirms that the tissue shown in Figures 4B and 4C is SA node. In the atrial muscle and intercaval region, no labeling was detected when the anti-DPI/II antibody was omitted (not shown).
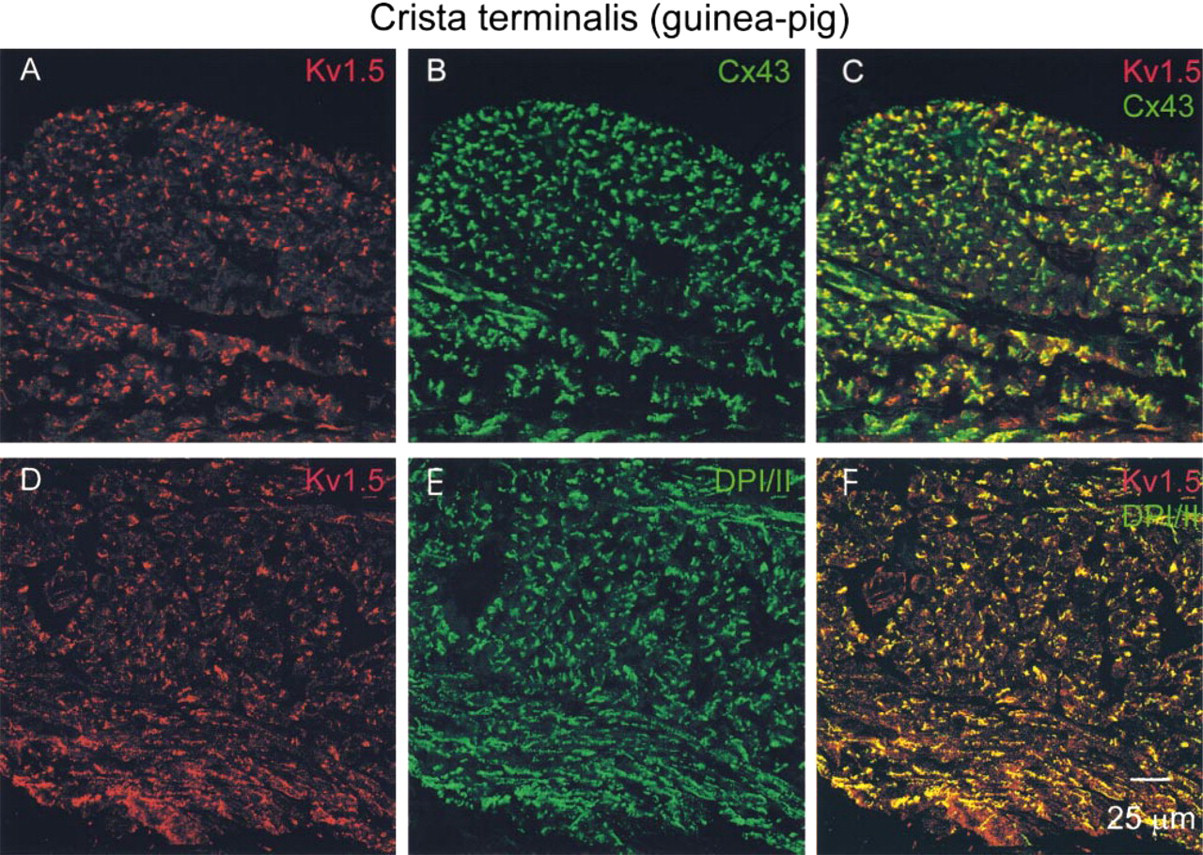
Confocal micrographs showing Kv1.5, Cx43, and DPI/II labeling in atrial muscle of crista terminalis of guinea pig. Kv1.5 (
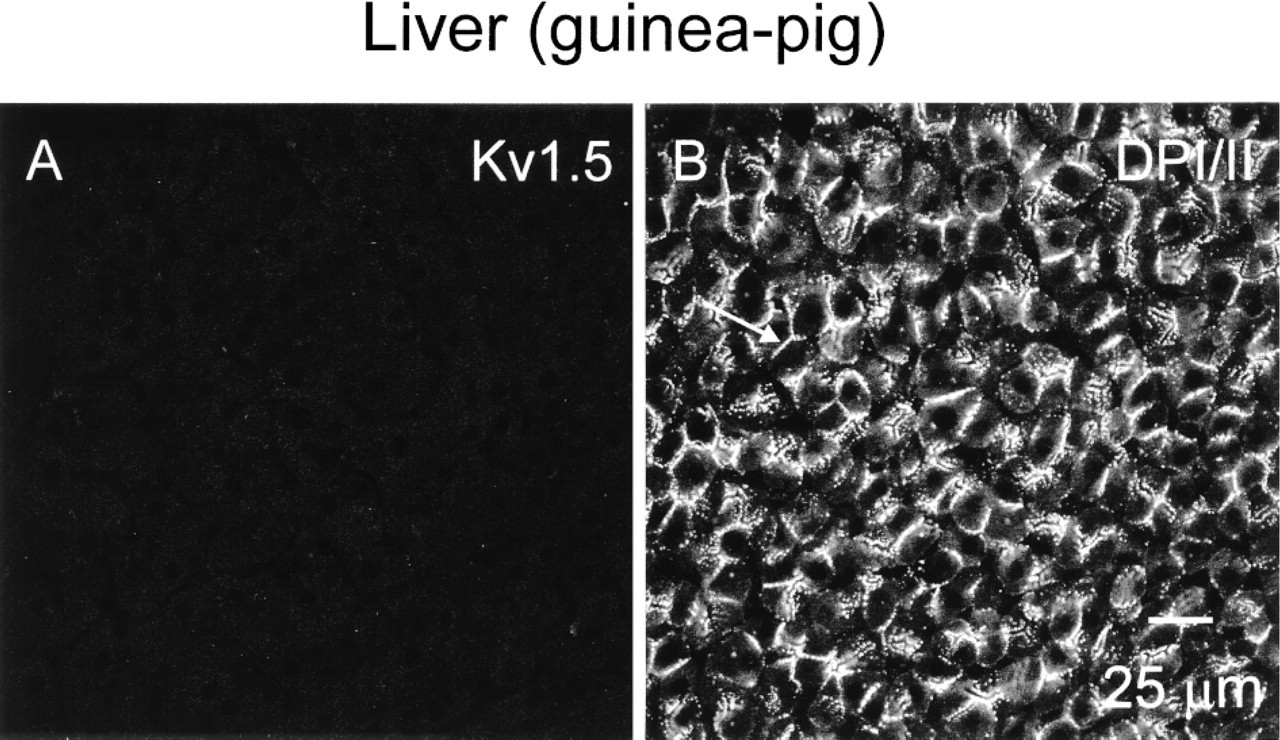
It could be argued that the anti-Kv1.5 antibody binds nonspecifically to desmosomes. To test this possibility, guinea pig liver (in which desmosomes are known to be abundant and the presence of voltage-dependent K+ channels is unlikely) was double labelled for Kv1.5 and DPI/II. Figure 6 shows pronounced DPI/II labeling in guinea pig liver (Figure 6B) but no Kv1.5 labeling (Figure 6A). In the liver, no labeling was detected when the anti-Kv1.5 and anti-DPI/II antibodies were omitted. These results suggest that the Kv1.5 antibody detects Kv1.5 rather than binding nonspecifically to desmosomes.
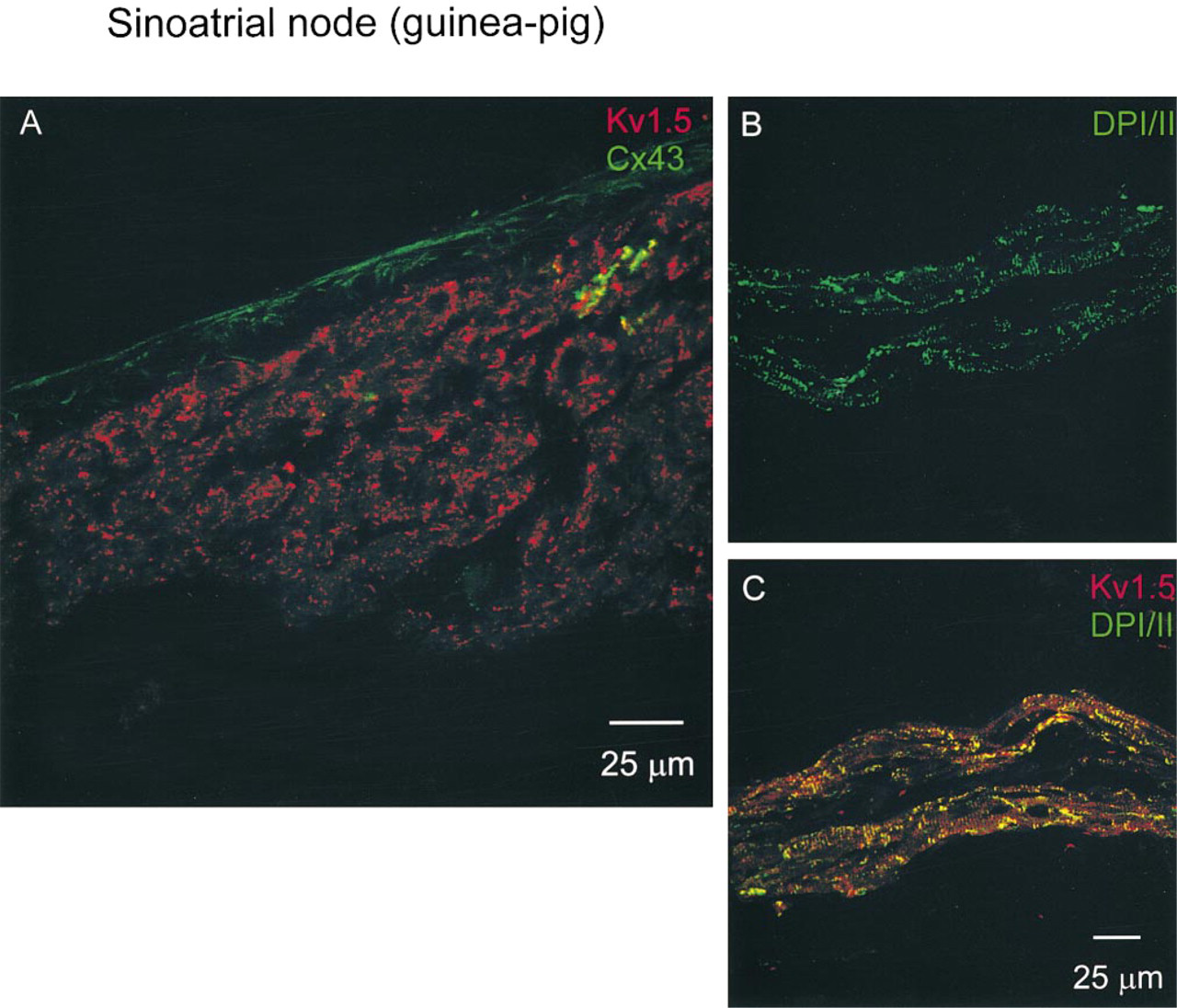
Confocal micrographs showing Kv1.5, Cx43, and DPI/II labeling in guinea pig SA node. (
DPI/II in guinea pig tissue was also studied using another antibody to DPI/II raised in rabbit (not shown). DPI/II labeling was once again observed throughout the atrial muscle and SA node. In the atrial muscle, DPI/II labeling again appeared in clusters, whereas in the SA node, in which Cx43 labeling was not detected, DPI/II labeling was punctate. Because both the anti-Kv1.5 and anti-DPI/II antibodies were raised in rabbit, association of Kv1.5 and DPI/II labeling could not be studied. In the atrial muscle and intercaval region, no immunofluorescence was detected when the rabbit anti-DPI/II antibody was omitted.
Taken together, these results suggest that Kv1.5 must be located at or close to desmosomes.
Subcellular Immunolocalization of Kv1.5
To detect the precise site of binding of the anti-Kv1.5 antibody at high resolution, we carried out immunogold labeling of ultrathin sections of freeze-substituted, Lowicryl-embedded mouse atria. Experiments had to be carried out on mouse tissue because attempts to process guinea pig tissue did not result in adequate tissue preservation. Electron microscopic examination of sections labeled with anti-Kv1.5 antibody demonstrated high levels of labeling at the intercalated disc membrane (Figures 7A and 7B). The gold label was not uniformly distributed within the disc, however, but was preferentially associated with desmosomes (Figures 7C and 7D). Lower levels of labeling occurred along the membranes of fasciae adherentes junctions and non-junctional regions of the discs, with no labeling (above background) at gap junctions. As previously reported (Yeh et al. 1998; Severs 1999), the anti-Cx43 antibody gave highly specific labeling of gap junctions, with no labeling of other junction types or other cell structures (Figure 7E). No labeling was obtained when the primary antibodies were omitted.
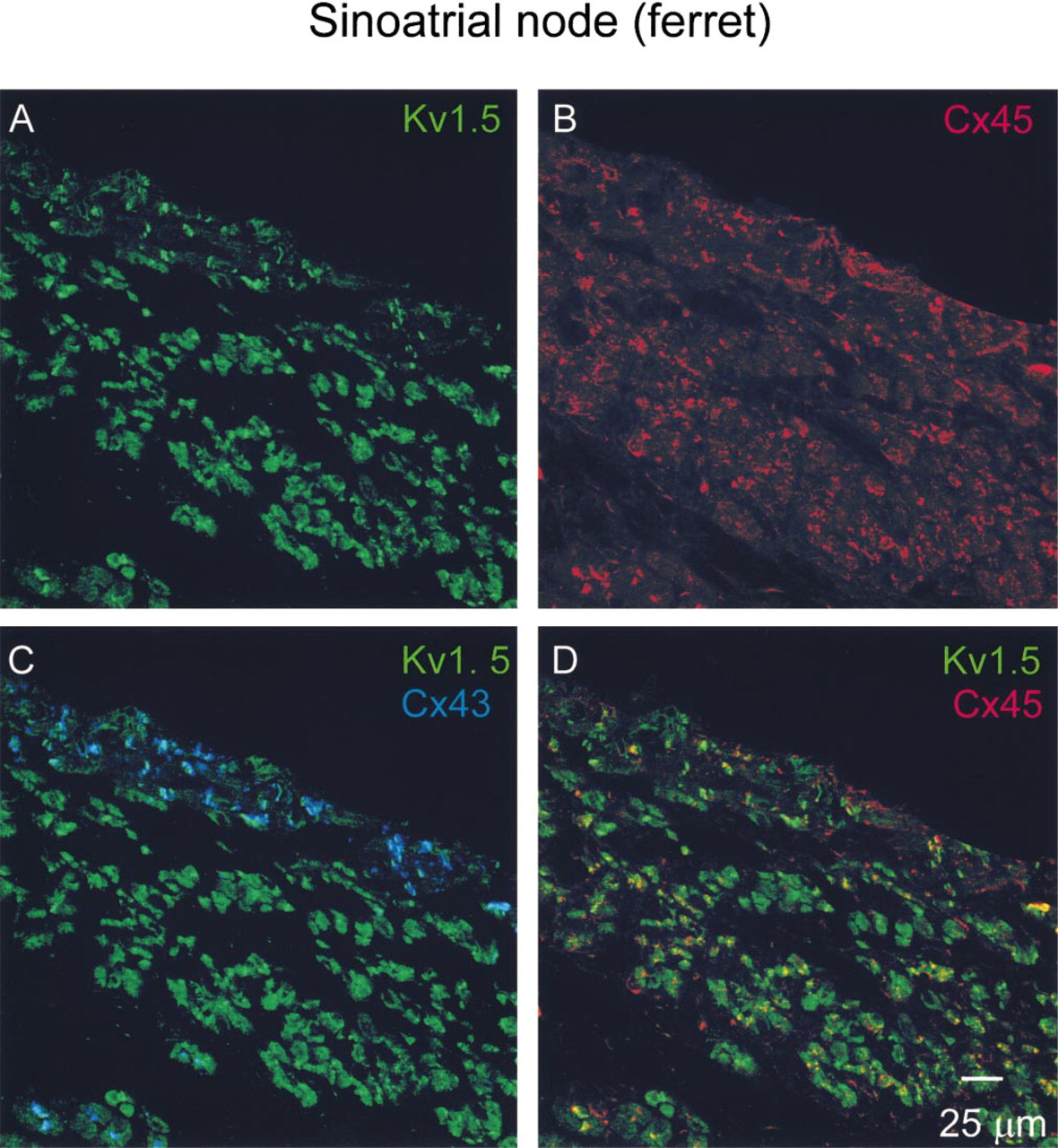
Confocal micrographs showing Kv1.5, Cx45, and Cx43 labeling in a triple-labeled section of ferret SA node. Kv1.5 (
Confocal micrographs showing Kv1.5 (
Discussion
In this study, at the confocal microscopic level, immunolabeling of Kv1.5 revealed Kv1.5 immunofluorescence labeling in SA node in close association with DPI/II and occasionally Cx45 labeling and in atrial muscle in close association with DPI/II and Cx43 labeling and, at the electron microscopic level, immunogold labeling in atrial muscle preferentially associated with desmosomes and other parts of the intercalated disc membrane rather than gap junctions.
Validity of Data
In Western blotting experiments, labeling of a protein of the expected molecular weight was observed (Figure 1). In Western blotting, immunofluorescence, and immunogold experiments, no labeling was observed in the absence of the anti-Kv1.5 antibody. In a previous study using the same antibody as used in the present study, Mays et al. (1995) obtained labeling of mouse L-cells transfected with Kv1.5, but not of sham-transfected cells, in both Western blotting and immunofluorescence experiments. In the same study, no labeling was observed when the antibody was preabsorbed with the peptide against which the antibody was raised (Mays et al. 1995). Finally, in the study of Mays et al. (1995), the same pattern of labeling at the intercalated disc in human atrium and ventricle was observed using a second antibody (raised against the S1-S2 linker region rather than the NH2 terminal of the human Kv1.5 K+ channel). It was not possible to use the second anti-Kv1.5 antibody in the present study because it works only with human tissue (Mays et al. 1995). Barry et al. (1994) also observed strong labeling at the intercalated disc (as well as weaker labeling in the lateral plasma membrane) of rat atrial and ventricular cells using another anti-Kv1.5 antibody. In this study, we have demonstrated that it is unlikely that the Kv1.5 antibody binds nonspecifically with desmosomes, because Kv1.5 labeling was not detected in guinea pig liver, whereas pronounced DPI/II labeling was observed (Figure 6). These findings suggest that the antibody used does bind specifically to Kv1.5 in heart tissues to give a reliable indication of its presence.
Subcellular Location of Kv1.5
In atrial muscle, the intercalated disc is an extensive and irregular structure containing gap junctions, desmosomes and fasciae adherents (Severs 1995). This explains the clustering of Cx43 and DPI/II labeling in atrial muscle (Figures 3B and 3E). In the SA node, there are no intercalated discs (James et al. 1966; Rubenstein et al. 1987; Severs 1989). There are gap junctions, desmosomes, and fasciae adherentes in the SA node, but they are smaller and fewer in number than those in atrial muscle and are not as regularly arranged as in atrial muscle (Bleeker et al. 1980). This explains the punctate labeling of DPI/II, not previously reported, in the SA node (Figure 4B). The co-localization of Kv1.5 and DPI/II immunofluorescence labeling in both atrial muscle and SA node suggests that Kv1.5 is present at or close to desmosomes in both tissues. Co-localization of Kv1.5 with DPI/II can explain the different pattern of Kv1.5 labeling in atrial muscle and SA node (in clusters and punctate, respectively; Figures 3A and 4A). It is interesting that the pattern of labeling of Kv1.5 in the SA node is similar to the pattern of labeling of Kv1.5 in the ventricles of newborn human and rat (Mays et al. 1995).
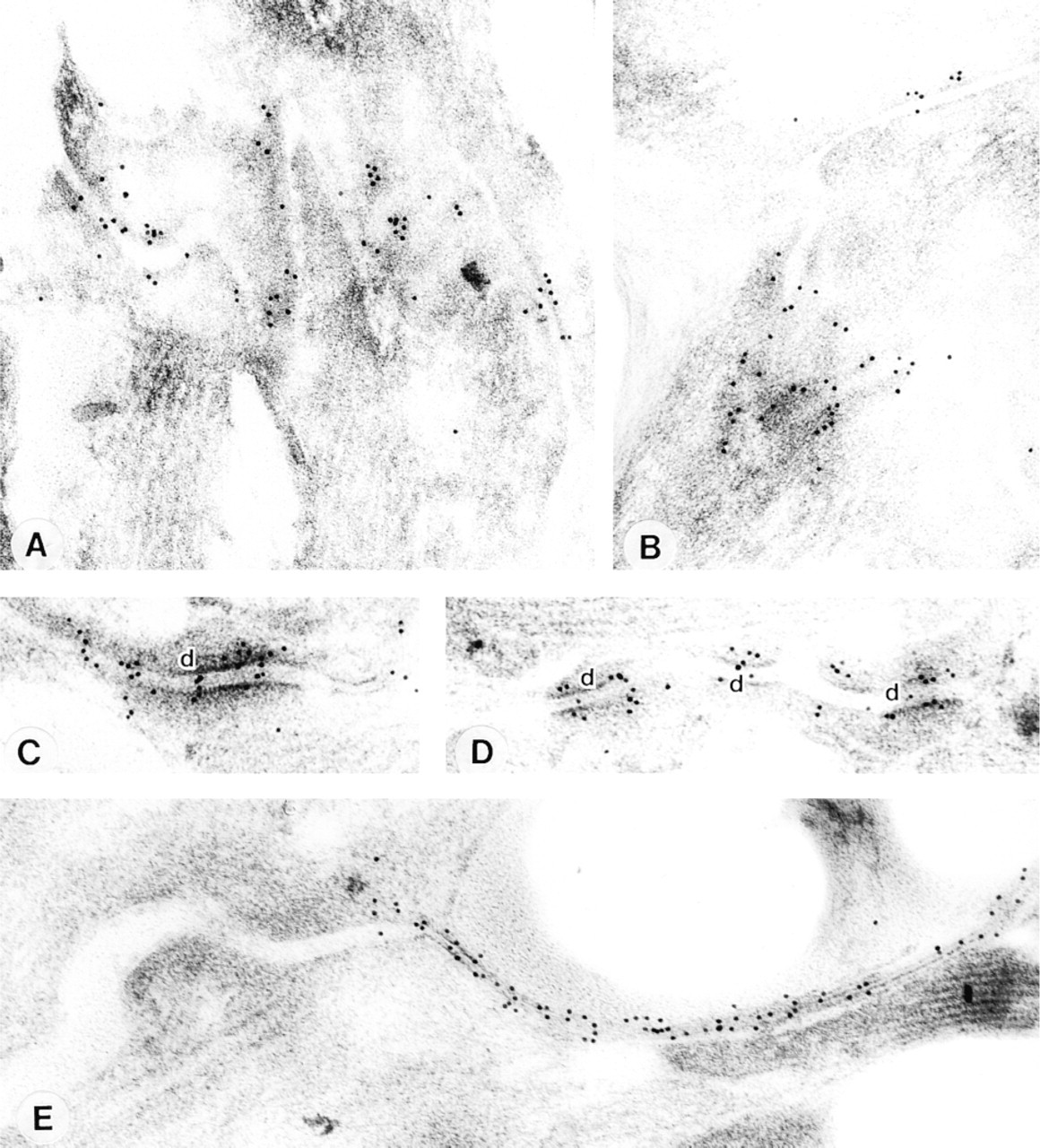
Immunogold localization of Kv1.5 and Cx43 in mouse atrium. Transmission electron microscopy of thin sections of Lowicryl-embedded mouse atrial tissue after immunogold labeling for detection of anti-Kv1.5 (
The immunogold findings at the electron microscopic level demonstrate preferential binding of the Kv1.5 antibody at desmosomes (Figure 7). Although the preferential binding of Kv1.5 at desmosomes is unexpected, the presence of extracellular space between the adjacent membranes at desmosomes is substantial (Figure 7), and therefore the channel might be able to function. Our finding that Kv1.5 labeling was preferentially associated with desmosomes does not exclude the presence of Kv1.5, at lower levels, in other regions of the intercalated disc and lateral plasma membrane. This possibility has been suggested by Mays et al. (1995) and Barry et al. (1994), who obtained strong labeling at the intercalated disc, with weaker labeling in the lateral plasma membrane of rat atrial and ventricular cells, using another anti-Kv1.5 antibody.
A number of ion channels, Kv1.2, Kv2.1, Kv4.2, and rH1 (Na+ channel), apart from Kv1.5, as well as the Na+-K+ pump (unpublished observations) and IP3 have been reported to be concentrated at the intercalated disc (Kijima et al. 1993; Barry et al. 1994; Mays et al 1995; Cohen 1996), and it will be interesting to discover whether any of these proteins are also present in or close to desmosomes. It has been suggested that ion channels are present at the intercalated disc because the intercalated disc may be best suited for anchoring channel proteins to the cytoskeleton or because newly formed channels are first inserted and concentrated at the intercalated disc by the biosynthetic machinery (Mays et al. 1995). Alternatively, it is possible that channels are located at the intercalated disc in assciation with desmosomes, because this location ensures that the proteins are less exposed to mechanical stress.
Possible Physiological Role of Kv1.5 in SA Node
Kv1.5 is responsible for ultra-rapid delayed rectifying K+ current (iK,ur) sensitive to block by 4-AP (e.g., Wang et al. 1993). Honjo et al. (1999) and Lei et al. (unpublished data) obtained evidence of a 4-AP-sensitive sustained outward current in rabbit SA node cells. If the density of Kv1.5 is sufficiently high, it should play a significant role in action potential repolarization. In the SA node, 4-AP (a blocker of iK,ur and ito) results in action potential prolongation (as well as other changes) (Boyett et al. 1998). In models of rabbit peripheral and central SA node action potentials incorporating 4-AP-sensitive sustained outward current at appropriate densities (Zhang et al. unpublished data), block of the current results in a 39% and a 12%, respectively, prolongation of the action potential and a 27% and 8%, respectively, increase in cycle length (unpublished observations). It is possible, therefore, that the Kv1.5 K+ channel by itself plays a significant role in the electrical activity of the SA node. Another possibility is that Kv1.5 forms a heteromultimer with other channel subunits.
Footnotes
Acknowledgements
Supported by the British Heart Foundation, by the Ministry of Education, Science and Culture of Japan, by the Japan Society for the Promotion of Science, and by the NIH (grant number HL49330).
We wish to thank Ms J. Higgins and Mr D. Harrison for technical assistance and Dr S. Jones for a control Western blot experiment. The rabbit anti-DPI/II antibody was a gift from Dr A.I. Magee (University College London).
