Abstract
Gelatin zymography is widely used to detect gelatinase activity, which is performed on unfixed tissue because it is assumed that fixation inactivates enzymes. However, using fixed tissues has several advantages over using fresh tissues for such prevention of tissue decay, thereby preserving the proteins as well as the morphology and structure of the specimens. In this study, we investigated the effects of the four commonly used fixatives (ethanol, acetone, zinc-based fixative (ZBF), and paraformaldehyde (PFA)) on the gelatinolytic activity in mouse brain tissue. Multiple protocols were employed to extract proteins from the fixed brain tissue. Western blotting and in-gel zymography (IGZ) were used to detect the gelatinase proteins and gelatinolytic activity of the extractions, respectively. In situ zymography (ISZ) revealed that ethanol, acetone, ZBF, and short-time PFA fixation did not inhibit gelatinolytic activity. Neither 1% Triton + 1 M NaCl nor 10% DMSO + 1 M NaCl was effective in extracting proteins from ethanol-, acetone-, ZBF-, or PFA-fixed brain tissues. However, 8 M urea + 4% CHAPS effectively extracted gelatinase proteins from ethanol- and acetone-fixed tissues while retaining the gelatinolytic activity. 2% SDS effectively extracted gelatinase proteins from ethanol-, acetone-, and ZBF-fixed tissues while retaining the gelatinolytic activity. Although 2% SDS + heating extracted gelatinase proteins from ethanol-, acetone-, ZBF-, and even long-term PFA-fixed tissues, the gelatinolytic activity was not retained. Our findings suggest that both ISZ and IGZ can be performed on fixed brain tissue, which is anticipated to be an improvement over the conventionally used gelatin zymography methods. (J Histochem Cytochem 71: 481–493, 2023)
Introduction
Matrix metalloproteinases (MMPs) are a family of zinc-dependent endo-peptidases that were first identified for their role in the breakdown of virtually all constituents of the extracellular matrix. Later, it was discovered that these enzymes also cleave intracellular substrates in the nuclear, mitochondrial, vesicular, and cytoplasmic compartments, including the cytoskeletal intracellular matrix. 1 MMPs are produced and secreted in a latent form known as zymogen or pro-MMP, which requires proteolytic cleavage of a propeptide domain to be activated. Due to their critical roles in various physiological and pathological processes, MMPs are constantly the subject of intense research in numerous fields. Among them, gelatinase A (MMP-2, 72 kDa) and gelatinase B (MMP-9, 92 kDa) represent a subgroup of the MMPs, and have type IV collagen, fibronectin, and gelatin as substrates. In the brain, MMP-2and MMP-9 have been the most intensively studied subjects because of their prominent roles in development, injury, and repair, as well as their ease of identification using gelatin zymography. 2
In situ zymography (ISZ) and in-gel zymography (IGZ) are two widely employed techniques for detecting gelatinase activity. ISZ detects and localizes specific protease activities in tissue sections or cultured cells. After incubation with gelatin conjugated to quenched fluorescein, gelatinases proteolyze the substrate, resulting in fluorescence. It is also possible for ISZ to use antibodies to colocalize proteolytic activity with specific antigens. 3 The limitation of this technique is that it may be difficult to distinguish between the various classes of MMPs as well as other proteinases (e.g., serine or cysteine proteases) that can degrade the substrates. 3 Thus, it is essential to use control samples containing appropriate MMP inhibitors. In contrast, IGZ can distinguish between MMP-2 and MMP-9, as well as the pro-form and active-form, though tissue homogenization for the assay prevents localization of enzyme activity. 4
It has long been considered that both ISZ and IGZ must be performed on fresh tissues. Fixatives were thought to inactivate enzymes. For instance, paraformaldehyde (PFA) induces cross-linking between macromolecules in tissues, resulting in irreversible enzyme inactivation. 5 However, we once attempted to use sections fixed with 4% PFA for 10 min as negative controls while performing ISZ on mouse brain cryosections. Surprisingly, the fixation did not abrogate the gelatinase but rather protected it from degradation. This serendipitous discovery led us to believe that the gelatinase may not be as fragile as we had previously believed. Gawlak et al. 6 used thin sections of the alcohol-fixed and polyester wax-embedded brain tissue of the rat to visualize structural details up to the resolution limit of light microscopy in conjunction with immunofluorescent labeling. Porto et al. 7 demonstrated the feasibility of ISZ in PFA-fixed, decalcified, and paraffin-embedded sections of craniofacial tissues from adult animals. Hadler-Olsen et al. 8 also demonstrated that ISZ can be performed on ethanol and zinc-based fixative (ZBF)-fixed paraffin-embedded tissue. However, they were unable to extract gelatinolytic enzymes efficiently from both ethanol- and ZBF-fixed tissue for the IGZ assay.
Using fixed tissues has several advantages over using fresh tissues. For instance, it prevents tissue decay, thereby preserving the proteins as well as the morphology and structure of the specimens without the use of a cryogenic refrigerator. This is important because cryopreservation of a large number of clinical samples is challenging. Therefore, this study aimed to systematically investigate the effects of the four commonly used fixatives, that is, ethanol, acetone, ZBF, and PFA, on gelatinolytic activity in mouse brain tissue. Moreover, several protocols were used to extract proteins from these fixed brain tissues for western blotting (WB) and the IGZ assay.
Methods
The animal experiments were performed under the compliance with the protocol approval by the Animal Care and Use Committee at the University of Missouri, which is in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals. Adult male C57Bl/6J mice (The Jackson Laboratory, Bar Harbor, Maine, USA), 6 to 9 weeks old and weighing 20–28 g were used in this study. Animals were housed in a 12-hr light/dark cycle and permitted food and water intake ad libitum.
Induction of Traumatic Brain Injury (TBI)
A controlled cortical impact (CCI)-induced TBI model was produced with an electromagnetic impactor device as previously described.9,10 Briefly, mice were anesthetized with isoflurane, 5% for induction and 2% for maintenance. They were then placed in a stereotaxic frame (Benchmark Deluxe; MyNeurolab, St. Louis, MO). Rectal temperatures were maintained at 37 °C±0.5 °C with animals placed on a thermostat-controlled heating pad (HS-3x2.5 Heater; Cell MicroControls, Norfolk, VA). Following a midline skin incision, a 5.0-mm diameter craniotomy was performed over the left lateral skull using a motorized drill mounted on the stereotactic arm. The EM impactor with a 3.0-mm-diameter tip was centered at 2.7 mm lateral to midline and 3.0 mm anterior to lambda at an angle of 15° to deliver a CCI for a depth of 2.5 mm below the dura with a velocity of 0.5 m/s and dwell time of 100 min. After injury, the impact site was covered with a plastic skull cap and the incision was sutured. Antibiotic ointment was applied to the skin and the mice were allowed to recover fully. All efforts were made to minimize animal suffering.
ISZ on Brain Cryosections
Twenty-four hours after TBI, mice were sacrificed by inhaling of an overdose of 5% isoflurane and perfused with saline. The brains were dissected and embedded in optimal cutting temperature compound (OCT) on dry ice/ethanol slurry. Then, the brains were cut into 10-µm-thick coronal sections on a freezing microtome, transferred to poly-
Test Extraction Efficiency
Totally, 24 intact mouse brains were used to test the efficacy of different extraction methods on different fixatives of fixed brain tissues. Each mouse brain was equally separated into 5 parts, weighted, and then fixed for 24 hr in either 70% ethanol at −20 °C, acetone at −20 °C, ZBF at 4 °C, or 4% PFA at 4 °C as the fixed tissues, or stored at −80 °C as fresh tissue, respectively. Then, the fixed tissues were rinsed in PBS 3 times, 5 min for each. Five extraction buffers or protocols were used to extract proteins: (1) 1% Triton and 1 M NaCl in ddH2O; (2) 10% dimethyl sulfoxide (DMSO) and 1 M NaCl in ddH2O; (3) 8 M Urea and 4% CHAPS (3-[(dimethylammonio]-1-propanesulfonate) in ddH2O; (4) 2% sodium dodecyl sulfate (SDS) (2% SDS and 40 mM Tris in ddH2O, PH 8.2); (5) SDS with heating: tissue was homogenized in 2% SDS lysis buffer, and then the mixture was incubated at 100 °C for 20 min and 60 °C for 2 hr. Extraction buffers were added a ratio of 1:6 (6 µl of extraction buffer per 1 mg of tissue), and the samples were homogenized with a pestle in a microcentrifuge tube. After staying on ice for 30 min, the homogenates were centrifuged at 17,000 × g for 10 min at 4 °C. The supernatants were collected, and the protein concentration was determined by bicinchoninic acid (BCA) assay according to the BCA Kit instruction manual (Sigma-Aldrich, BCA).
Preparation of TBI Brain Extractions
Twenty-four hours after TBI, mice were sacrificed by an overdose of isoflurane and perfused with saline, followed by dissection of their brains. Coronal brain sections 5.5–0.5 mm away from Lambda were cut into 1-mm-thick coronal sections, and then further cut into 1 × 1 × 1 mm3 blocks. All the tissue blocks were divided into 5 portions. The samples of all experiments derived from difference regions of the brain tissues which were cut into 1 × 1 × 1 mm3 pieces, and separated into 5 portions equably. IGZ confirmed that the content and activity of MMP-9 and MMP-2 were equivalent in 5 portions (Appendix Fig. 1). As descripted above, the samples were weight, kept fresh or fixed for 24 hr and then extracted by the Urea and CHAPS, 2% SDS or SDS with heating respectively. Because the other two methods, Triton and NaCl, DMSO and NaCl were shown to be unable to extract proteins efficiently from the fixed brain tissues, these two methods were not test here. The five samples extracted from the same brain were then diluted to the same concentration as the lowest concentration of extraction sample (from zinc or PFA-fixed tissue), and ready for SDS-Poly acrylamide gel electrophoresis (PAGE) and further gel silver staining, western blot, and IGZ analysis.
Gel Silver Staining
For the SDS-PAGE, protein samples were mixed in a ratio of 3:1 with 4× loading buffer containing 8% SDS, 40% glycerol, 0.04% bromophenol blue, and 250 mM Tris-HCl. For the SDS extracted samples, loading buffer without SDS was used. For each sample, 24 μL of mixture was loaded per well for the 10-well comb, dissolved in 10% SDS-PAGE at constant voltage until the bromophenol blue tracking dye reached the bottom of the gel. The proteins were visualized using a ProteoSilverTM Silver Stain Kit (Sigma-Aldrich, PROT-SIL1) according to the instruction manual.
Western Blotting
Western blots of prepared samples were performed as described previously. 11 Briefly, protein samples are mixed in a ratio of 1:1 with 2× loading buffer. For each sample, 10 μg of total protein was loaded and dissolved in 8% SDS-PAGE at 100 V. Proteins were transferred to 0.45-μm-thick nitrocellulose membranes at 300 mA for 3 hr. Membranes were blocked in Tris-buffered saline (TBS) with 0.1% Tween 20 (TBS-T) containing 2% bovine serum albumin (BSA) for 2 hr at room temperature. Membranes with transferred protein were incubated with antibodies against MMP-9 (mouse anti-MMP-9, Chemicon MAB13415, 1:1000), MMP-2 (Rabbit anti-MMP-2, Chemicon AB808, 1:1000), and beta-actin (1:50000, Sigma-Aldrich, St. Louis, MO) in TBS-T containing 2% BSA overnight at 4 °C. After rinsing with TBS-T, membranes were incubated with goat anti-rabbit IgG-horseradish peroxidase (1:4000, Santa Cruz Biotechnology, Santa Cruz, CA) or goat anti-mouse IgG-horseradish peroxidase (1:2000, Santa Cruz Biotechnology, Santa Cruz, CA) for 1 hr at room temperature. Immunolabeling was detected by chemiluminescence ECL (Thermo Scientific, 32106) /WestPico/femto. Developed blots on x-ray films were digitally scanned and analyzed.
IGZ on Fixed Brain Tissue
Gelatin IGZ was performed as described previously. 9 Briefly, the extractions from unfixed or fixed brain tissues were mixed in a ratio of 3:1 with 4× loading buffer. The protein samples were then separated by electrophoresis in a 10% SDS-PAGE gel containing 0.1% gelatin under nonreducing conditions until the 37 kD band of marker ran out of the gel. The gels were renatured in 1× 2.5% Triton-x-100, followed by incubation in developing buffer at 37 °C for 2–5 days. Conditioned media of Gelatin-Sepharose 4B-enriched HT1080 cell, containing both MMP-2 and MMP-9, was used as positive controls. Gels were stained with Coomassie blue and digitally scanned.
Statistical Analysis
All data are presented as the mean ± the standard error of the mean (SEM). The one-way ANOVA followed by Tukey’s Multiple Comparison Test was applied to test the difference between groups. Differences were considered significant at p<0.05 for all analyses.
Results
ISZ on Unfixed and Fixed Brain Sections
To induce gelatinase expression, a CCI-induced TBI model was established in this study, which has been shown to elevate levels of both inactive latent MMP-9 (proMMP-9) and active MMP-9 (actMMP-9) in the lesioned cortex 24 hr after trauma. 9 ISZ was used to detect gelatinolytic activity in the peri-contusional cortex of the unfixed and fixed brain sections. Compared with unfixed snap-frozen sections, the ethanol-, acetone-, ZBF-, and PFA-fixed sections not only demonstrated gelatinolytic activity but also expressed much more intense and sharper signals, indicating that 10 min of fixation with ethanol, acetone, ZBF, or PFA preserved gelatinolytic activity on TBI brain sections (Fig. 1). All the activities in the unfixed or fixed section were inhibited by the broad-spectrum MMP inhibitors 1,10-phenanthroline and EDTA (metal ions chelating agent), but not by a non-MMP protease inhibitor cocktail (PIC), indicating that they were MMP gelatinolytic activities.
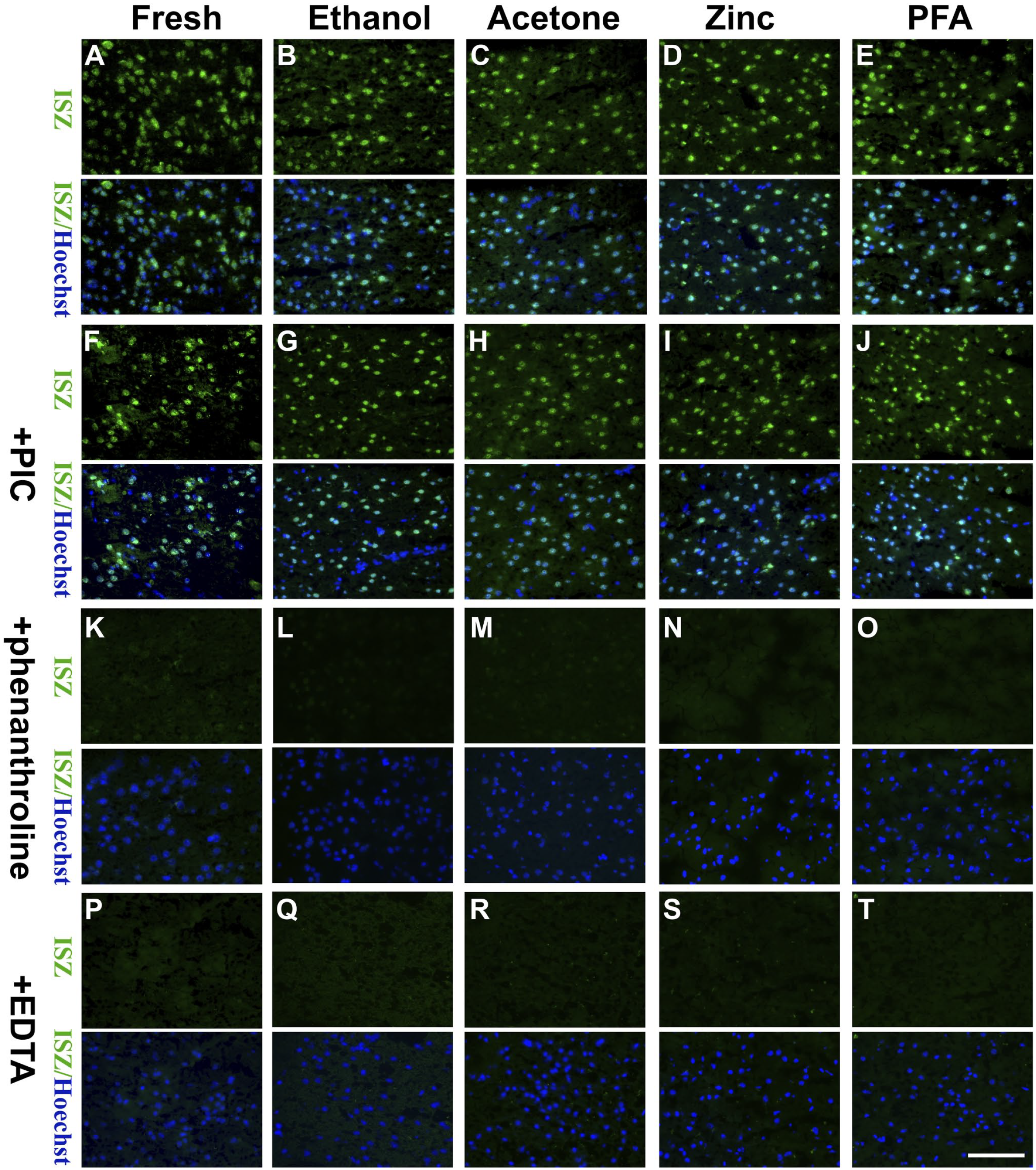
ISZ with the MMP fluorogenic substrate DQ-gelatin in the peri-contusional cortex of cryosections. 10 min of ethanol, acetone, zinc or PFA fixation didn’ t inhibit gelatinolytic activity (A, B, C, D, E); The broad-spectrum MMP inhibitors 1,10-phenanthroline (K, L, M, N, O) and EDTA (metal ions chelating agent) (P, Q, R, S, T), but not PIC (a protease inhibitor cocktail) (F, G, H, I, J), abrogated gelatinolytic activity. Representative figures from three independent experiments. Scale bar=100 µm. Abbreviations:DQ, dye-quenched; ISZ, In situ zymography; PFA, paraformaldehyde; MMP, matrix metalloproteinases.
ISZ on Tissue Sections Fixed With PFA for Various Periods
To investigate the effect of PFA fixation duration on gelatinolytic activity, TBI brain cryosections were fixed with 4% PFA for 30 min, 1 hr, 2 hr, 6 hr, 1 day, 3 days, or 7 days, followed by ISZ. As shown in Fig. 2, the gelatinolytic activity gradually diminished as the fixation time was extended, and the gelatinolytic activity was almost completely abrogated after PFA fixation for more than 24 hr, indicating that PFA inhibits gelatinolytic activity in a time-dependent manner.
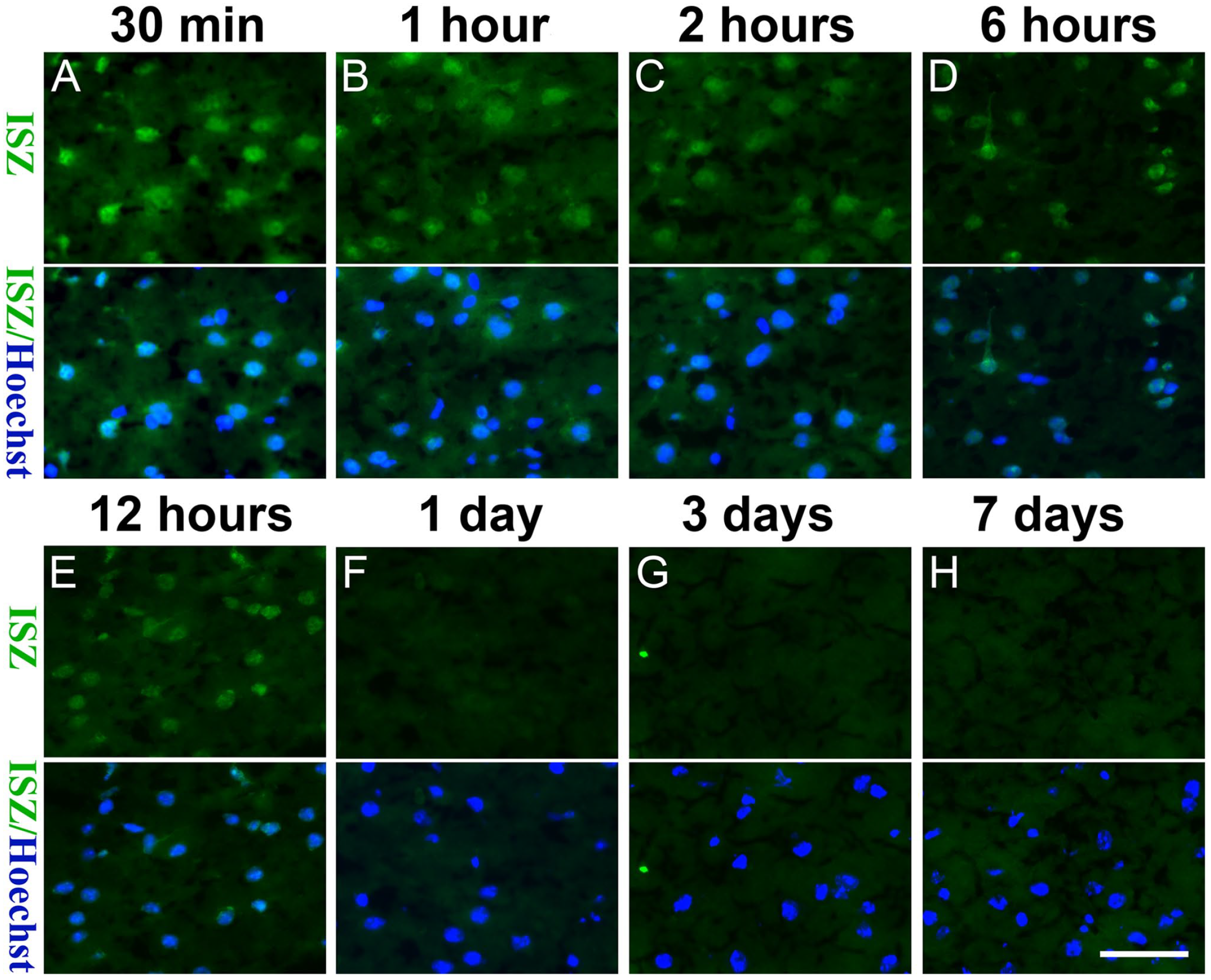
With the extension of the PFA fixation time (A: 30 min, B: 1hour, C: 2 hours; D: 6 hours, E: 12 hours, F: 1 day, G: 3 days, H: 7 days), the gelatinolytic activity gradually weakened. Representative figures of 3 independent experiments. Scale bar = 50 µm. Abbreviations: ISZ, In situ zymography; PFA, paraformaldehyde.
Protein Extraction Efficiency
We evaluated the efficiency of protein extraction from fresh tissues as well as tissues fixed with ethanol, acetone, ZBF, and PFA using various solvents: 1% Triton and 1 M NaCl, 10% DMSO and 1 M NaCl, 8 M urea and 4% CHAPS, and 2% SDS. As shown in Fig. 3A, Triton and NaCl were ineffective at extracting proteins from ethanol-, acetone-, ZBF-, and PFA-fixed brain tissues but were effective at extracting proteins from fresh tissues. Similar patterns were observed for DMSO and NaCl (Fig. 3B). The concentration of protein extracted by urea and CHAPS was low in ethanol- or acetone-fixed tissues and even lower in ZBF-, and PFA-fixed tissues than in fresh tissues (Fig. 3C). SDS could extract proteins from unfixed tissues and ethanol-, acetone-, and ZBF-fixed tissues but not from tissues fixed with PFA for 24 hr (Fig. 3D). In contrast, SDS with heating could extract proteins from all the unfixed tissues and ethanol-, acetone-, ZBF-, and PFA-fixed tissues (Fig. 3E). The protein extraction efficiency of SDS was gradually reduced with the extension of the PFA-fixed time (Fig. 3F).
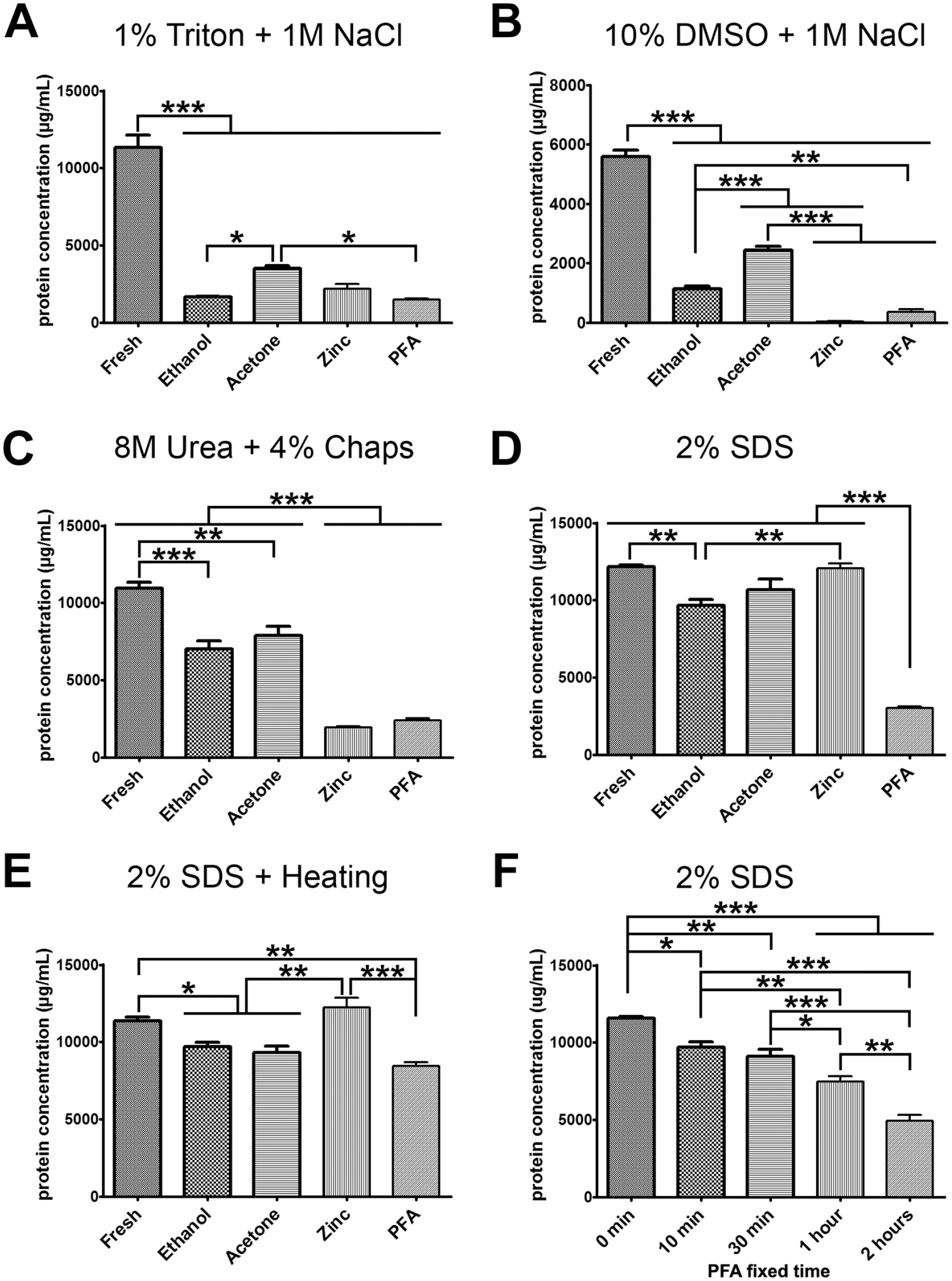
Protein extraction efficiency: (A, B) Triton and NaCl as well as DMSO and NaCl were ineffective at extracting proteins from ethanol-, acetone-, ZBF-, and PFA-fixed brain tissues. (C) the concentration of protein extracted by urea and CHAPS was low in ethanol- or acetone-fixed tissues, and even lower in ZBF-, and PFA-fixed tissues than in fresh tissues. (D) SDS could extract proteins from unfixed tissues and ethanol-, acetone-, and ZBF-fixed tissues but not from tissues fixed with PFA for 24 hr. (E) SDS with heating could extract proteins from all the unfixed tissues and ethanol-, acetone-, ZBF-, and PFA-fixed tissues. (F) the protein extraction efficiency of SDS was gradually reduced with the extension of the PFA-fixed time. All n=6. Abbreviations: CHAPS, 3-([3-cholamidopropyl] dimethylammonio)-1-propanesulfonate; DMSO, dimethyl sulfoxide; PFA, paraformaldehyde; SDS, sodium dodecyl sulfate; ZBF, zinc-based fixative. *p<0.05, **p<0.01, ***p<0.001.
Gelatinase Extraction Using 8 M Urea and 4% CHAPS
The protein distribution in the extractions was determined using gel silver staining. As shown in Fig. 4A, the stripe distribution of extracted proteins from ethanol- and acetone-fixed tissues was similar to that of fresh tissues. However, the distribution of extracted proteins from ZBF-fixed tissues was completely different from that of fresh tissues and ethanol- and acetone-fixed tissues and the extracted proteins from 24-hr PFA-fixed tissues were primarily low-molecular-weight (less than 20 kDa) proteins. WB was used to determine whether the gelatinase protein could be extracted using 8 M urea and 4% CHAPS, and it was found that MMP-9 and MMP-2 can be effectively extracted from fresh tissues and ethanol-, acetone-, and ZBF-fixed tissues but not PFA-fixed tissues (Fig. 4B). Interestingly, proteins extracted from ZBF-fixed tissues had lower levels of β-actin (42–43 kDa). IGZ was used to evaluate the gelatinolytic activity of the extractions and revealed that gelatinolytic activity could be found in the fresh tissue extraction and ethanol-, acetone-, and ZBF-fixed tissue extractions but not PFA-fixed tissue extraction (Fig. 4C).
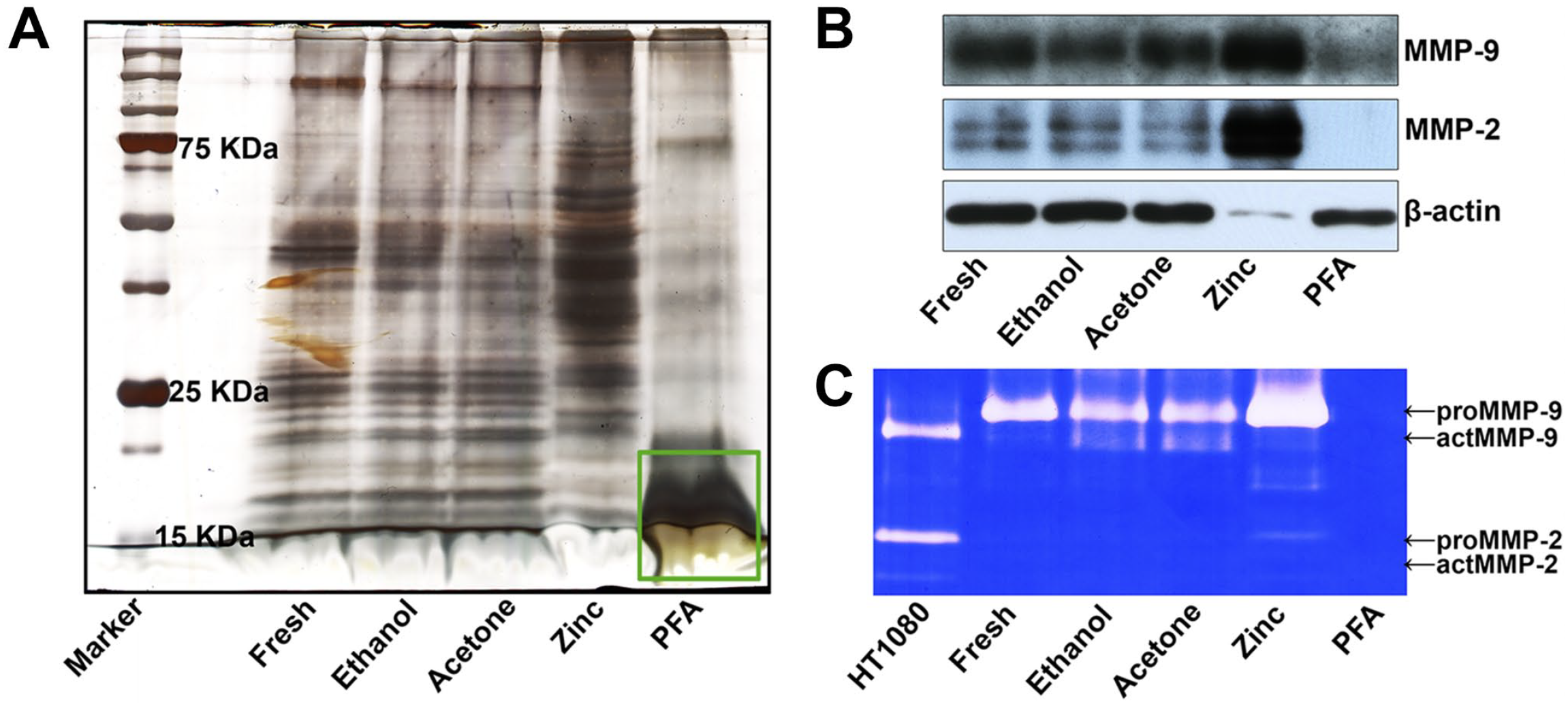
Gelatinase extraction using 8 M urea and 4% CHAPS: (A) gel silver staining analysis revealed that the molecular weight distribution of extracted proteins from ZBF-fixed tissues was completely different from that of fresh tissues and ethanol-, and acetone-fixed tissues, and the extracted proteins from PFA-fixed tissue were mostly low-molecular-weight (less than 20 KDa) proteins (green box). (B) WB revealed that MMP-9 and MMP-2 can be effectively extracted from fresh tissues and ethanol-, acetone-, and ZBF-fixed tissues but not PFA-fixed tissue. (C) IGZ revealed that gelatinolytic activity was preserved in the fresh tissue extraction as well as ethanol-, acetone-, and ZBF-fixed tissue extractions, but not PFA-fixed tissue extraction. Conditioned media of HT1080 cells served as a positive control. Representative figures from three independent experiments. Abbreviations: CHAPS, 3-([3-cholamidopropyl] dimethylammonio)-1-propanesulfonate; IGZ, in-gel zymography; MMP, matrix metalloproteinases; PFA, paraformaldehyde; WB, western blotting; ZBF, zinc-based fixative.
Gelatinase Extraction Using 2% SDS
Gel silver staining analysis revealed that proteins of various molecular weights could be efficiently extracted by 2% SDS from all the fresh tissues and ethanol-, acetone-, and ZBF-fixed tissues but not from 24-hr PFA-fixed tissues, which primarily contained those low-molecular-weight proteins (Fig. 5A). WB also revealed that MMP-9 and MMP-2 can be extracted from all the fresh and ethanol-, acetone-, and ZBF-fixed tissues but not from PFA-fixed tissues. Moreover, it was found that the active form of MMP-9 was better preserved in ethanol- and acetone-fixed tissue extractions (Fig. 5B). IGZ confirmed that gelatinolytic activity was preserved in fresh and ethanol-, acetone-, and ZBF-fixed tissue extractions but not in PFA-fixed tissue extractions (Fig. 5C).
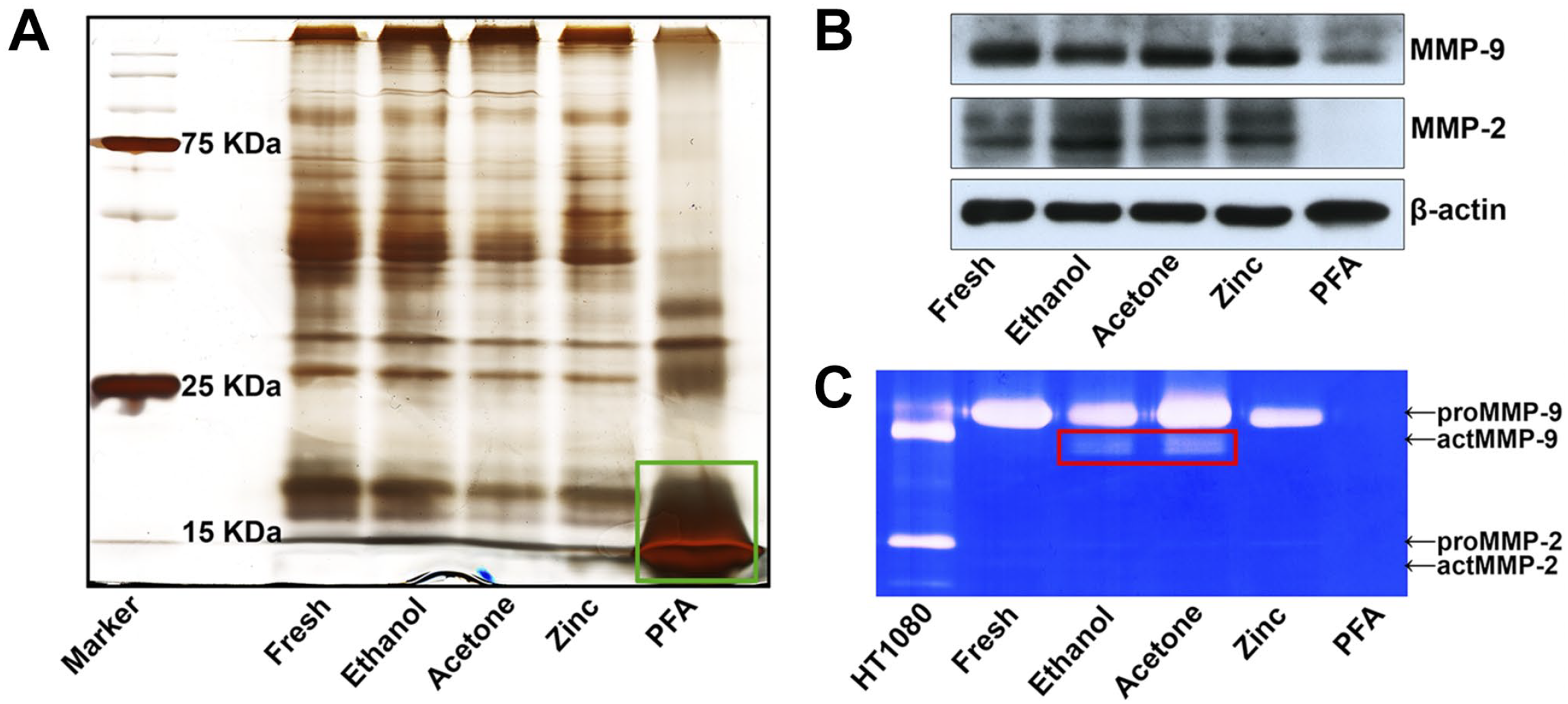
Gelatinase extraction using 2% SDS: (A) gel silver staining analysis revealed that the extractions from fresh tissues and ethanol-, acetone-, and ZBF-fixed tissues contained proteins of various molecular weights, except for those from PFA-fixed tissue, which contained mostly those low-molecular-weight proteins (green box). (B) WB showed that proteins of MMP-9 and MMP-2 can be extracted from all the fresh as well as ethanol-, acetone- or ZBF-fixed tissues but not from PFA-fixed tissues. (C) IGZ revealed that the gelatinolytic activity was preserved in fresh and ethanol-, acetone-, and ZBF-fixed tissue extractions, but not in PFA-fixed tissue extraction. Active MMP-9 was better preserved in ethanol- and acetone-fixed tissue extractions (red box). Conditioned media of HT1080 cells served as a positive control. Representative figures from 3 independent experiments. Abbreviations: IGZ, in-gel zymography; MMP, matrix metalloproteinases; PFA, paraformaldehyde; SDS, sodium dodecyl sulfate; WB, western blotting; ZBF, zinc-based fixative.
Gelatinase Extraction Using SDS With Heating
Gel silver staining analysis revealed that proteins of various molecular weights were efficiently extracted using 2% SDS with heating from all the fresh and ethanol-, acetone-, ZBF-, and even 24-hr PFA-fixed tissues (Fig. 6A). WB confirmed that proteins of MMP-9 and MMP-2, as well as β-actin, could be extracted from all the fresh and ethanol-, acetone-, ZBF-, and PFA-fixed tissues (Fig. 6B). However, IGZ revealed that gelatinolytic activity was completely abrogated in fresh and ethanol-, acetone-, ZBF-, and PFA-fixed tissue extractions (Fig. 6C).
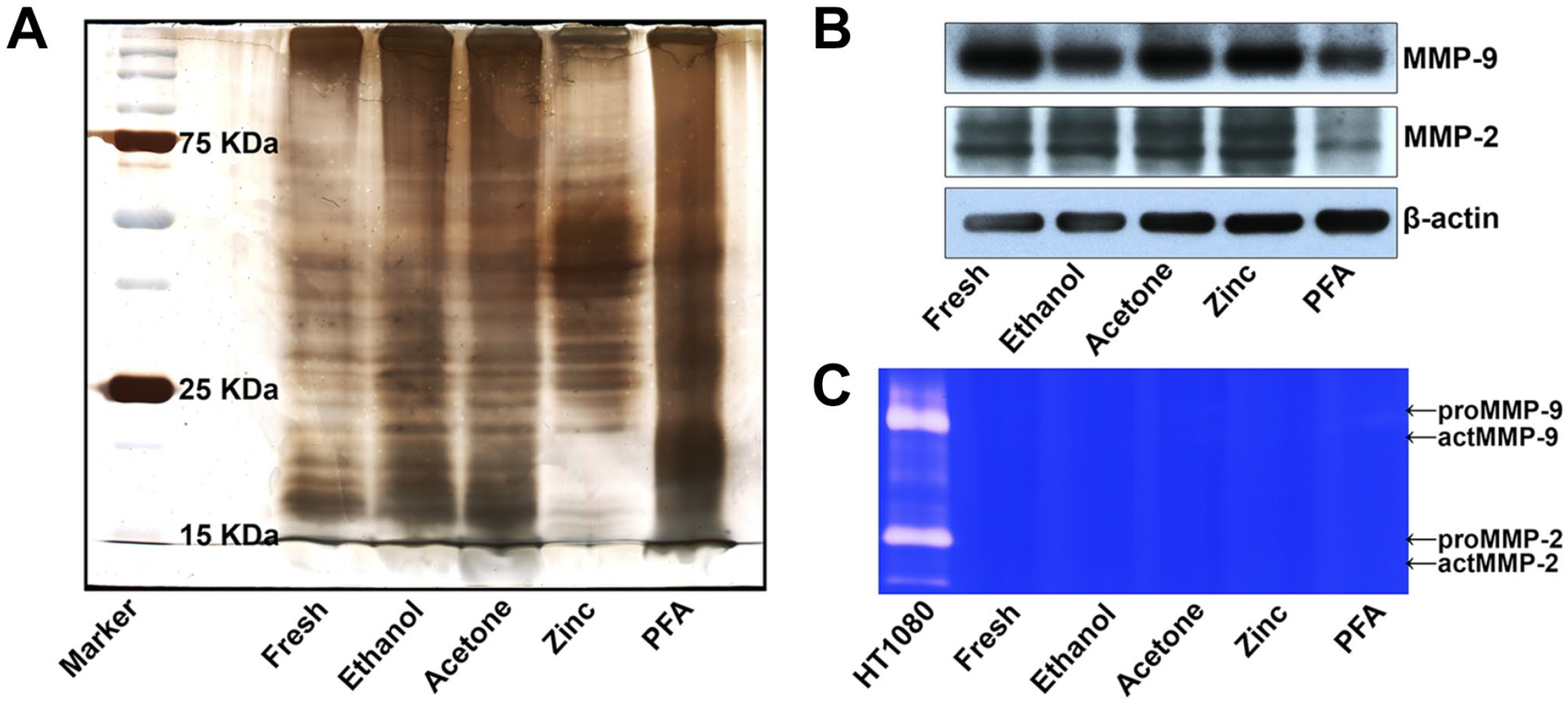
Gelatinase extraction using 2% SDS with heating: (A) gel silver staining analysis revealed that proteins were extracted using 2% SDS with heating from all the fresh as well as ethanol-, acetone-, ZBF- and PFA-fixed tissues. (B) WB revealed that proteins of MMP-9 and MMP-2, as well as β-actin, were extracted from all the fresh as well as ethanol-, acetone-, ZBF, and PFA-fixed tissues. (C) IGZ revealed that gelatinolytic activity was completely abrogated in fresh and ethanol-, acetone-, ZBF-, and PFA-fixed tissue extractions. Conditioned media of HT1080 cells served as a positive control. Representative figures from three independent experiments. Abbreviations: IGZ, in-gel zymography; MMP, matrix metalloproteinases; PFA, paraformaldehyde; SDS, sodium dodecyl sulfate; WB, western blotting; ZBF, zinc-based fixative.
Effect of PFA Fixation Time on Gelatinase Extracted Using 2% SDS
We finally evaluated the efficiency of gelatinase extracted using 2% SDS from tissues fixed with 4% PFA for various periods, that is, 0 min, 10 min, 30 min, 1 hr, and 2 hr. As shown in Fig. 7A, gel silver staining analysis revealed that as the fixation time was increased, macromolecular proteins became more difficult to extract. Furthermore, WB revealed that the extraction efficiency of MMP-9 and MMP-2, but not β-actin, decreased as the PFA fixation time increased (Fig. 7B). IGZ confirmed that the extracted MMP-9 and MMP-2 still had enzyme activity in short-term PFA-fixed samples. However, the gelatinolytic activity in the extractions gradually decreased as the PFA fixation time was increased (Fig. 7C).
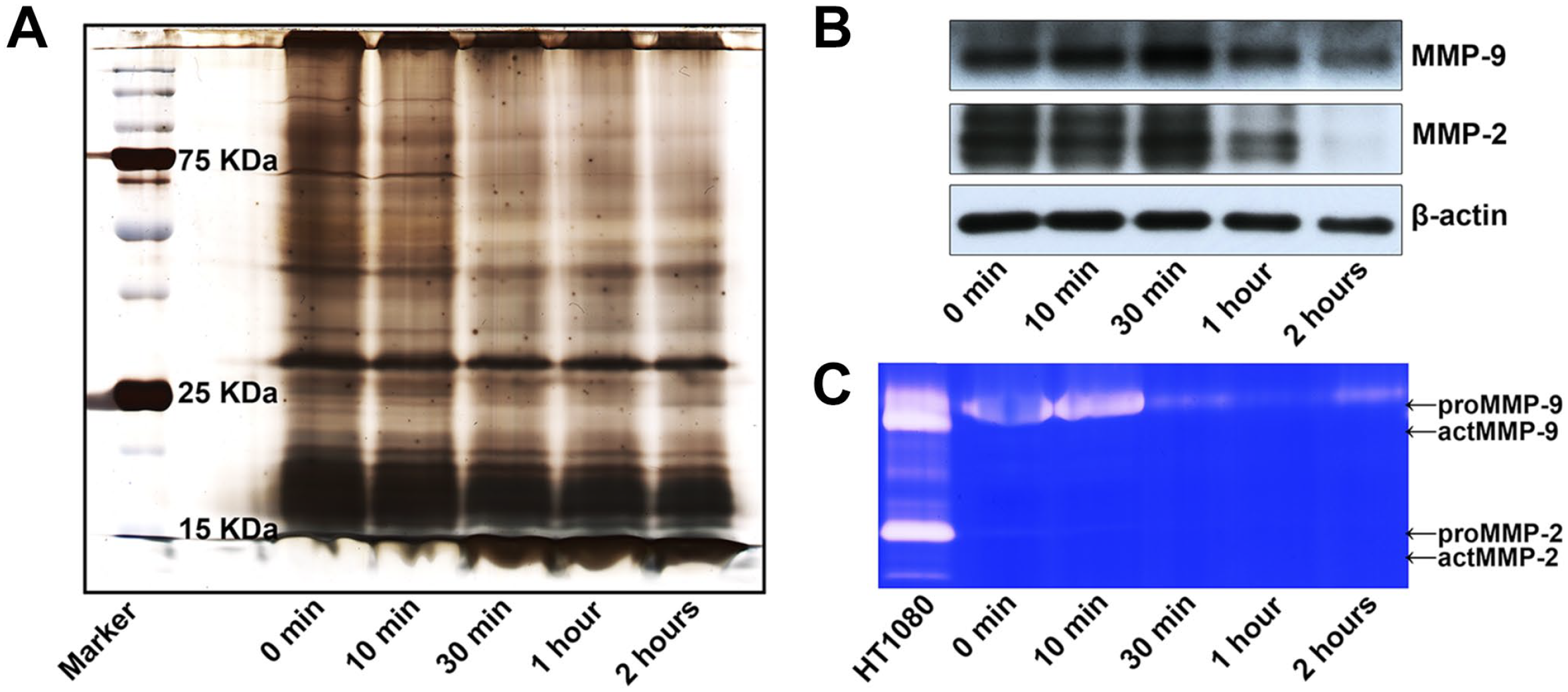
Effect of PFA fixation time on gelatinase extracted using 2% SDS. (A) gel silver staining analysis revealed that as the fixation time was increased, macromolecular proteins became more difficult to extract. (B) WB revealed that the extraction efficiency of MMP-9 and MMP-2 proteins decreased as the PFA fixation time increased. (C) IGZ confirmed that gelatinolytic activity in the extractions gradually decreased as the PFA fixation time increased. Conditioned media of HT1080 cells served as a positive control. Representative figures from three independent experiments. Abbreviations: IGZ, in-gel zymography; MMP, matrix metalloproteinases; PFA, paraformaldehyde; SDS, sodium dodecyl sulfate; WB, western blotting; ZBF, zinc-based fixative.
Discussion
Gelatin zymography is a simple, inexpensive, yet powerful method for detecting proteolytic enzymes capable of degrading gelatin in various biological samples. The technique was first described by Heussen and Dowdle in the 1980s, but it is still a valuable and convenient method for detecting the activity of the various gelatinases in research and diagnostics.12,13 Therefore, any improvements to this method will be useful in laboratory medicine.
Given the advantages of fixed samples over fresh samples in terms of protein and tissue structure preservation, this study aimed to investigate the feasibility of ISZ and IGZ on fixed specimens. Four commonly used representative coagulating or crosslinking fixatives, namely, ethanol, acetone, ZBF, and PFA, were evaluated. Ethanol denatures proteins by breaking the hydrophobic bonds that make up the tertiary structure of protein molecules while preserving the secondary structure for immunohistochemistry. Cold acetone has strong tissue penetration and dehydration properties. It dissolves membrane phospholipids but does not affect proteins or polysaccharides. 14 ZBF, first described in 1994, 15 is a nontoxic and temperature-insensitive fixative that has been shown to be superior for DNA and protein expression analysis in various tissues with no need for additional antigen retrieval. 16 PFA has good tissue penetration, little tissue shrinkage, and good antigenic substance preservation, particularly for fat and various enzymes. However, because PFA fixation involves crosslinking of proteins and nucleic acids by forming methylene bridges between reactive groups, antigen retrieval is required for immunochemistry following PFA fixation. 17 The feasibility of performing ISZ and IGZ on tissues fixed with these fixatives has not been systematically investigated.
ISZ is a unique technique that allows for the localization of MMPs in tissue sections. ISZ was long considered to be performed on frozen sections of unfixed tissue samples.13,18,19 However, our results demonstrated that 10 min of ethanol-, acetone-, ZBF-, or PFA fixation did not inhibit gelatinolytic activity in TBI sections. All such activities were inhibited by the broad-spectrum MMP inhibitors 1,10-phenanthroline and EDTA but not by the non-MMP protease inhibitor cocktail (PIC), indicating that they were MMP gelatinolytic activities. 20 We also found that the gelatinolytic activity decreased gradually as the PFA fixation time increased. In contrast, prolonged ethanol, acetone, or ZBF fixation did not result in gelatinase inactivation (data not shown). Moreover, compared with the unfixed, snap-frozen sections, the fixed sections displayed significantly more intense and sharper signals, indicating that fixation is beneficial for preventing enzyme degradation. Our findings are comparable to those of Haeckel et al., 21 who demonstrated that the combination of ISZ and immunohistochemistry with subsequent Prussian blue iron staining on ZBF-fixed paraffin-embedded tissue sections produced excellent histologic images of enzyme activity, protease distribution, and iron oxide particle accumulation. Together, the pre-fixation of brain sections with ethanol, acetone, ZBF, or short-term PFA refines the ISZ method, allowing for more precise detection of gelatinolytic activity.
IGZ allows for the simultaneous determination of both active and latent forms of MMP-2 and MMP-9 based on their molecular weight and gelatin degradation ability. The proteins are separated by electrophoresis under denaturing SDS, at this time, the gelatinases are inactive. The gels are then incubated at 37 °C in a nonionic detergent (Triton-X-100), which facilitates the SDS exchange. Because gelatinases are Ca2+-dependent enzymes, the gels are further incubated in an appropriate “refolding” buffer containing Ca2+ ions. The enzymes partially recover their structure and activity during the incubations, and the latent gelatinases are auto-activated without cleavage.13,22,23 Although IGZ is the most simple, sensitive, quantifiable, and least expensive functional assay for gelatinase analysis, there is no report on its successful performance on fixed tissues.
Protein extraction is the most important step in revealing a proteome by electrophoresis. We first evaluated the protein extraction efficiency of various extraction methods for fixed brain tissues. Our data revealed that 1% Triton and 1 M NaCl, as well as 10% DMSO and 1 M NaCl, were unable to effectively extract proteins from brain tissues fixed with ethanol, acetone, ZBF, and PFA for 24 hr, explaining why traditional IGZ methods do not work on fixed tissues.
We then evaluated 8 M urea and 4% CHAPS, which were combined to improve protein resolution and solubilization of various protein classes 24 and have been routinely used to enhance protein solubilization in oil palm proteomic studies in recent years. 25 We found that they successfully extracted proteins from ethanol- and acetone-fixed tissues but not from ZBF- or PFA-fixed tissues, though the extraction efficiency was lower than that of fresh tissues. Gel silver staining revealed that the molecular weight distribution of extracted proteins from ZBF-fixed tissue was completely different from that of fresh as well as ethanol- and acetone-fixed tissues and that extracted proteins from PFA-fixed tissue were mostly low-molecular-weight proteins. WB revealed that MMP-9 and MMP-2 proteins can be extracted from fresh as well as ethanol-, acetone-, and ZBF-fixed tissues but not from PFA-fixed tissues. IGZ further revealed that the extracted gelatinase could be renatured.
We found that 2% SDS, which had previously been used to extract proteins from formalin-fixed tissues for proteomic analysis,26,27 is effective in extracting proteins from fresh as well as ethanol-, acetone-, and ZBF-fixed tissues. WB and IGZ confirmed that MMP-9 and MMP-2 could be effectively extracted while retaining their gelatinolytic activity. However, when it comes to PFA-fixed tissue, our findings reveal that the protein extraction efficiency, extracted gelatinase, and gelatinolytic activity all decrease as the PFA fixation time increases. Only low-molecular-weight proteins and almost no gelatinase could be extracted from 24-hr PFA-fixed tissues.
Because PFA fixation results in the cross-linking of proteins, and thus intact proteins cannot be extracted efficiently, violent conditions, such as heating, should be tried to increase protein recovery. 28 Therefore, we tested SDS with heating and found proteins could be efficiently extracted from all fresh as well as ethanol-, acetone-, ZBF-, and PFA-fixed tissues. WB confirmed that MMP-9 and MMP-2 proteins were extracted. However, IGZ revealed that all gelatinolytic activity had been destroyed. Indeed, a small increase in heat will increase the catalysis potential, but a large increase in heat causes the protein to lose its 3D configuration. It will untwist and unwind, losing its specificity. 29 Our results demonstrated that such heating-induced enzyme denaturation cannot be renatured in the present conditions.
Conclusion
Our findings indicate that ethanol, acetone, ZBF, and short-term PFA fixation do not abrogate gelatinase, but rather protect it from degradation in mouse brain tissue, making it suitable for ISZ analysis. 8 M urea + 4% CHAPS is effective for gelatinase extraction from ethanol- and acetone-fixed brain tissues for WB and IGZ analysis. 2% SDS is effective for gelatinase extraction from ethanol-, acetone-, ZBF-, and short-term PFA-fixed brain tissues for WB and IGZ analysis. Although 2% SDS + heating extracted gelatinase from ethanol-, acetone-, ZBF-, and even long-term PFA-fixed tissues, it failed to preserve the gelatinolytic activity and thus is only suitable for WB but not IGZ analysis. Our findings are anticipated to be an improvement over the conventionally used gelatin zymography methods.
Footnotes
Appendix
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
ZC and ZG conceived and designed the trial. ZC, DS, SC, and BT performed the experiments. ZC and ZG wrote the manuscript with significant input from QH and JC. All authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded, in part, by grants from the University of Missouri School of Medicine Department of Pathology and Anatomical Sciences Research Fund, the NFL Charities Foundation, and the Indiana Department of Health Spinal Cord and Brain Injury Research Program to ZG; as well as the National Natural Science Foundation of China (No. 31870972) and the Special Fund Project of Science and Technology in Guangdong Province (No. 2018YJ029) to ZC. The funders played no role in the study design, data collection and analysis, the decision to publish, or the preparation of the manuscript.
