Abstract
Although perineurium has an important role in maintenance of the blood–nerve barrier, understanding of perineurial cell–cell junctions is insufficient. The aim of this study was to analyze the expression of junctional cadherin 5 associated (JCAD) and epidermal growth factor receptor (EGFR) in the perineurium of the human inferior alveolar nerve (IAN) and investigate their roles in perineurial cell–cell junctions using cultured human perineurial cells (HPNCs). In human IAN, JCAD was strongly expressed in endoneurial microvessels. JCAD and EGFR were expressed at various intensities in the perineurium. In HPNCs, JCAD was clearly expressed at cell–cell junctions. EGFR inhibitor AG1478 treatment changed cell morphology and the ratio of JCAD-positive cell–cell contacts of HPNCs. Therefore, JCAD and EGFR may have a role in the regulation of perineurial cell–cell junctions.
Introduction
Neurosensory disturbance is a serious adverse event in medical treatments. Peripheral neuropathy induced by chemotherapy affects daily activities and quality of life long term. 1 Neurosensory complications following oral and maxillofacial surgeries are rare, but patients suffer from various symptoms. The occurrence of neurosensory complications following deeply impacted mandibular third-molar extraction under general anesthesia was 8.4%, and the rate of persistent neurosensory complications was 4.3% in a previous report. 2 Neuropathic pain in the maxillofacial region is very intense. 3 For example, patients with osteonecrosis of the jaw often suffer from severe pain.4,5 An understanding of the underlying mechanisms of nerve damage and recovery is important to develop effective therapies for patients who suffer from neurosensory disturbance in oral and maxillofacial regions. One of the reasons why neurosensory disturbance is difficult to treat is the blood–nerve barrier which restricts entry of neurotherapeutic agents. The blood–nerve barrier consists of the endoneurial microvessels within the nerve fascicle and the investing perineurium and tightly regulates the permeability. 6 The restricted permeability of the endoneurial microvessels and the perineurium protects the endoneurial microenvironment from drastic concentration changes in the vascular and other extracellular spaces and limits the entry of nonspecific molecules. 6
Perineurium maintains constant intrafascicular pressure and guarantees a selective barrier effect for axons and Schwann cells. 6 It can be divided into three zones: the inner zone, in which cell junctions are more prominent, and the intermediate and external zones, which are transiting areas between the perineurium and epineurium. 7 Cell junctions are more prominent at inner perineurial layers and are composed of cell–cell junctional proteins such as transmembrane protein claudins and intracellular scaffold protein zonula occludens-1. 8 Although perineurial cell–cell junctions regulate permeability depending on pathophysiological conditions, such as nerve crush injury and regeneration,9,10 our understanding of perineurial cell–cell junctions is insufficient.
There is an interesting report that epidermal growth factor receptor (EGFR) is expressed in the perineurium. 11 Moreover, a fascinating case report showed that the inhibition of EGFR improved neuropathic pain. 12 Although a recent randomized proof-of-concept trial of an EGFR inhibitor for treatment of neuropathic pain did not provide statistical evidence, a substantial reduction in pain was evident. 13 In rats, both activated and total EGFR contribute to neuropathic pain induced by chronic compression of unilateral lumbar dorsal root ganglions. 14 EGFR signaling has been implicated in neuropathic pain and may be a potential therapeutic target. 15 Therefore, we hypothesized that EGFR signaling may be involved in the barrier functions of perineurium, resulting in a desirable effect on neuropathic pain.
Junctional cadherin 5 associated (JCAD, previously known as KIAA 1462) is an intracellular component of endothelial cell–cell junctions. 16 Some studies have focused on the role of JCAD in cardiovascular diseases17–19 because a previous genome-wide association study reported that a single nucleotide polymorphism of JCAD was associated with coronary artery disease. 20 Although JCAD has been reported to express both endothelial and perineurial cell–cell junctions, 16 no attention has been focused on the role of JCAD in the perineurium. In this study, we confirmed the expression of JCAD and EGFR in cultured human perineurial cells (HPNCs) and the perineurium of the human inferior alveolar nerve (IAN). Next, we evaluated the effect of EGFR inhibition on cell morphology and the ratio of JCAD-positive cell–cell contacts of HPNCs. Finally, we discuss the unknown function of JCAD and EGFR in perineurial cell–cell junctions and potential targets for neurosensory disturbance.
Materials and Methods
Patients
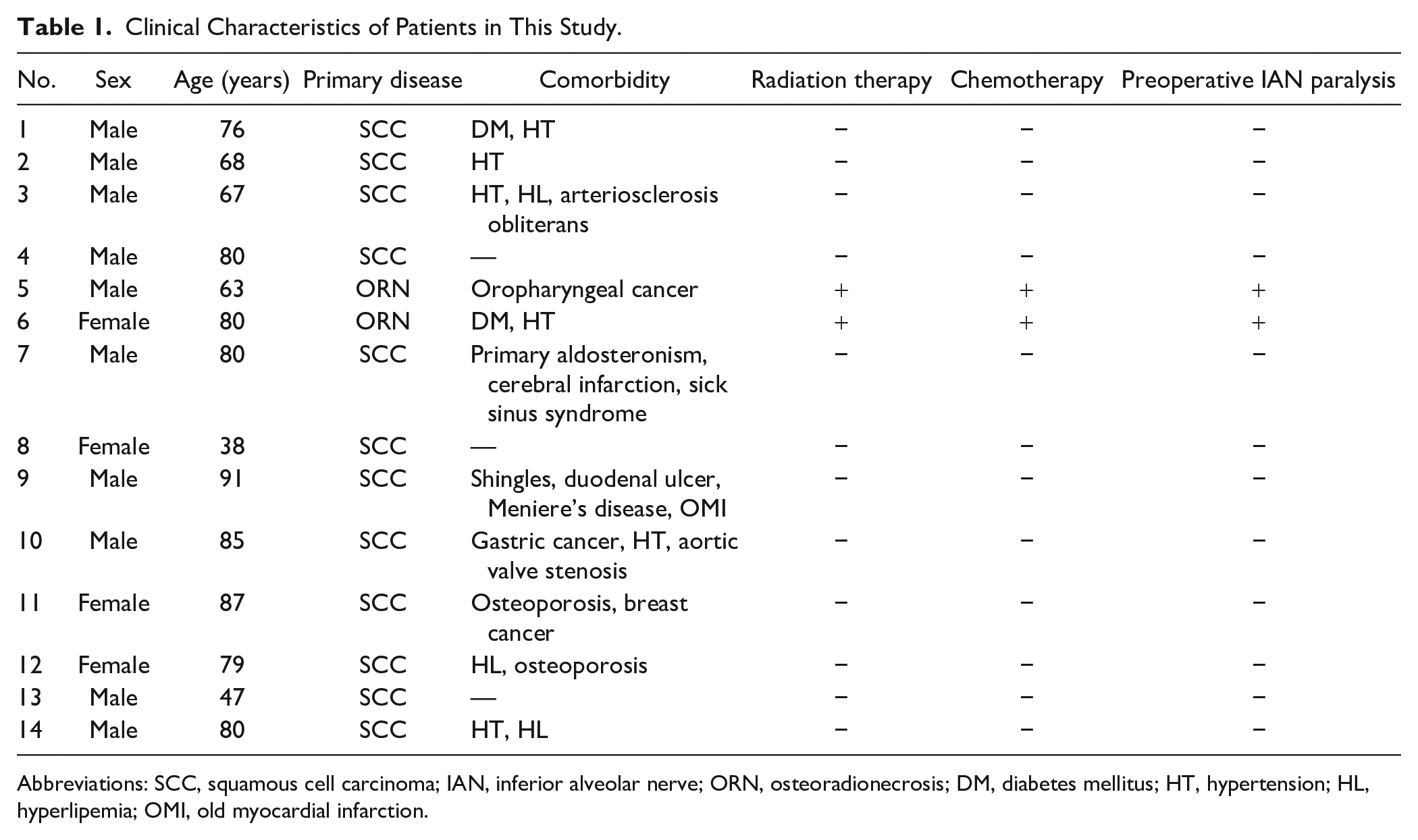
Fourteen patients who underwent mandibulectomy for various diseases at our department between June 2015 and May 2022 were randomly selected. Carcinomas with perineural infiltration of the IAN were excluded. The following epidemiologic data were gathered retrospectively from medical charts: age, sex, primary diseases, comorbidity, radiation therapy, chemotherapy, and paralysis of areas innervated by IAN, such as the lip and chin, before surgery. The Medical Ethics Committee of Kobe University Hospital approved this study (B210289). All patients provided written informed consent to release clinical information and bone samples.
Histopathological Analysis of Human IAN
Bone specimens were decalcified after surgery and fixed in formalin without freezing. Thin sections were prepared from paraffin-embedded blocks and then stained with hematoxylin and eosin for microscopy evaluation. Analysis of IAN has been previously described in detail. 5 Briefly, the number of nerve fascicles within the epineurium was counted (×4 magnification). Subsequently, the cross-sectional area of IAN was calculated using the ImageJ software (National Institutes of Health, Bethesda, MD). The cross-sectional area of epineurium was regarded to be the cross-sectional area of IAN. Image acquisition of bone specimens was performed under a BZ-X 700 (Keyence, Osaka, Japan).
Reagents and Antibodies
A rabbit monoclonal anti-EGFR antibody (clone: SP84, 1:100; Sigma-Aldrich, St. Louis, MO) was used for double immunofluorescence staining. A rabbit monoclonal anti-EGFR antibody (clone D38B1, 1:500; Cell Signaling, Danvers, MA) was used for western blotting. A rabbit polyclonal antihuman claudin-1 antibody (#51-9000, 1:50; Invitrogen, Carlsbad, CA) was used for immunostaining. A rabbit polyclonal antihuman JCAD antibody (HPA019756, 1:500; Sigma-Aldrich, St. Louis, MO) was used for immunostaining and western blotting. Actin filaments were labeled with Alexa 568-Phalloidin (1:200; Invitrogen Molecular Probes, Karlsruhe, Germany). Cell nuclei were labeled with 4′,6-Diamidino-2-phenylindole (DAPI, 1:200; Thermo Fisher Scientific, Inc., Waltham, MA). A secondary antibody conjugated with Alexa 488 (1:500) was purchased from Invitrogen Molecular Probes. The EGFR inhibitor AG1478 was purchased from Merck KGaA (Darmstadt, Germany). For double immunofluorescence staining of human IAN, a secondary antibody Opal Fluorophore Working Solution (Opal570 and 520, NEL810001KT; Akoya Biosciences, Marlborough, MA) was used. Image acquisition was performed under a BZ-X 700 (Keyence).
Cell Culture and EGFR Inhibition
HPNCs (1710) were obtained from ScienCell Research Laboratories (Carlsbad, CA). HPNCs were routinely maintained on poly-L-lysine-coated cell cultureware in Fibroblast Medium (ScienCell Research Laboratories) supplemented with 1% fibroblast growth supplement, 5% fetal bovine serum, and 1% of penicillin/streptomycin. Cells in most experiments were between passage 2 and 10. The concentrations of AG1478 treatment were 2, 5, and 10 μM for 24 hr.
Immunostaining, Immunofluorescence Staining, and Microscopy
Serial sections of tissues at a thickness of 3–4 μm were prepared. Intensity grading of each protein (claudin-1, EGFR, and JCAD) in the perineurium was performed as follows: strong, +++; moderate, ++; weak, +; none, - (×40 magnification). Two authors (Y.H. and M.A.) independently reviewed each sample. Claudin-1 was assessed as a positive control. JCAD expression was confirmed in endoneurial microvessels as a positive control because JCAD is expressed in blood endothelial cell–cell junctions. EGFR expression was confirmed in areas of carcinoma or gingival epithelium as a positive control. The expression of each protein was evaluated in areas with the strongest intensity.
HPNCs were cultured on glass coverslips coated with a fibronectin stock solution (Sigma-Aldrich, St Louis, MO) in 24-well culture plates. Cells were fixed with 4% paraformaldehyde in phosphate-buffered saline (PBS) for 10 min at room temperature, treated with 0.2% Triton X-100 (A16046, Alfa Aesar, Ward Hill, MA) in PBS for 5 min, and washed with PBS. Cells were blocked with 1% bovine serum albumin in PBS overnight and then incubated with primary antibodies overnight. Cells were then rinsed three times with PBS and incubated with appropriate secondary antibodies for 30 min. After rinsing with PBS, the specimens were embedded in FluorSave (Calbiochem, Darmstadt, Germany). Image acquisition was performed under a BZ-X 700 (Keyence).
Western Blot Analysis
HPNCs were lysed in a lysis buffer containing 0.5-M Tris-HCl (pH 6.8), 10% sodium dodecyl sulfate, and glycerol. Prior to loading, 1-M dithiothreitol was added to cell lysates. Samples were loaded on 10% gels for sodium dodecyl sulfate-polyacrylamide gel electrophoresis. Protein mobility was assessed using Precision Plus Protein Dual Color Standards (Bio-Rad Laboratories, Inc., Hercules, CA). Proteins were transferred to a polyvinylidene fluoride membrane, blocked with 5% non-fat dry milk for 1 hr, and incubated with the abovementioned antibodies and a mouse anti-glyceraldehyde 3-phosphate dehydrogenase (GAPDH) monoclonal antibody (5A12, 1:500; Fujifilm Wako Pure Chemical, Osaka, Japan) for 24 hr at 4C, followed by incubation with a horseradish peroxidase–conjugated donkey antirabbit immunoglobulin G (IgG) and sheep antimouse IgG secondary antibody (NA934 and NA931, 1:500 for EGFR and JCAD, 1:1000 for GAPDH; Cytiva, Tokyo, Japan) for 1 hr at room temperature. Immunostained bands were developed using the chemiluminescence agent ImmunoStar LD (296-69901; Fujifilm Wako Pure Chemical). Blots were scanned with an Amersham Imager (GE Healthcare Bioscience, Piscataway, NJ). GAPDH was used as an internal loading control. The experiment was repeated three times.
Quantification of the Cell Number and Changes Induced by EGFR Inhibition
The cell number was calculated by counting DAPI-positive nuclei in five randomly selected, independent microscopic images (×20 magnification). To analyze the effects of AG1478 on cell morphology, the cell surface area and the degree of cell elongation were quantified with reference to a previous report. 21 Briefly, the long axis, short axis, and cell outline were manually traced in immunofluorescence images using the ImageJ software. The long axis was defined as the longest length of the cell in images, and the short axis was defined as the length across the nucleus in a direction perpendicular to the long axis. 21 The ratio of the two axes (i.e., the length of the longest axis divided by the length of the short axis across the nucleus) was defined as the cell-elongation degree.
To analyze JCAD-positive cell–cell contacts, the entire circumference of the five cells not in contact with each other in randomly selected images (×40 magnification) was traced and measured using ImageJ. Next, the length of cell–cell contacts with JCAD expression was measured. Then, the length of JCAD-positive cell–cell contacts divided by the full length of the cell was defined as the ratio JCAD-positive cell–cell contacts. Results are representative of five randomly selected images from four independent experiments.
Statistical Analysis
Data are expressed as the mean ± standard deviation of four independent experiments. R software (R Development Core Team, 2011) was used to analyze data by one-way analysis of variance and Tukey’s multiple comparisons test as a post-hoc test. A p value of less than 0.05 was regarded statistically significant.
Results
JCAD and EGFR Expression in the Perineurium of Human IAN
First, we observed JCAD and EGFR expression in human tissues. Clinical characteristics of the patients are shown in Table 1. Ten male and four female patients (median age: 79.5 years; range: 38–91 years) were randomly selected. Primary diseases of 12 patients were squamous cell carcinoma, and two had mandibular osteoradionecrosis (ORN). No patients received steroids or immunosuppressants. Two patients with mandibular ORN suffered from IAN paralysis before surgery.
Clinical Characteristics of Patients in This Study.
Abbreviations: SCC, squamous cell carcinoma; IAN, inferior alveolar nerve; ORN, osteoradionecrosis; DM, diabetes mellitus; HT, hypertension; HL, hyperlipemia; OMI, old myocardial infarction.
Table 2 shows histopathological analysis of IAN in each patient. We previously reported pathological alterations of IAN caused by irradiation in patients with mandibular ORN. 5 The median cross-sectional area of IAN was 2.52 mm2 (range: 1.79–4.39 mm2). The median number of nerve fascicles was 12 (range: 3–23). The signal intensity of claudin-1 in the perineurium was strong or moderate in all patients. The signal intensity of EGFR varied in the perineurium. Although JCAD expression was clearly detectable in endoneurial microvessels of all patients, the signal intensity of JCAD in the perineurium also varied. Figure 1 shows representative cases of apparent expression of JCAD in endoneurial microvessels (cases 9 and 14) and the strong or moderate signal intensity of JCAD and EGFR in the perineurium. Figure 2 shows the results of the double immunofluorescence staining in the representative cases (cases 11 and 14). The expressions of JCAD and EGFR as well as claudin-1 were confirmed. Although the colocalization was observed between claudin-1 and EGFR and between claudin-1 and JCAD (Fig. 2A and B, white arrowheads), the colocalization of claudin-1 and JCAD was not observed in some areas of the perineurium (Fig. 2B, asterisks).
Results of Histological Analysis of IAN and Intensity of Claudin-1, EGFR, and JCAD Expression in the Perineurium.
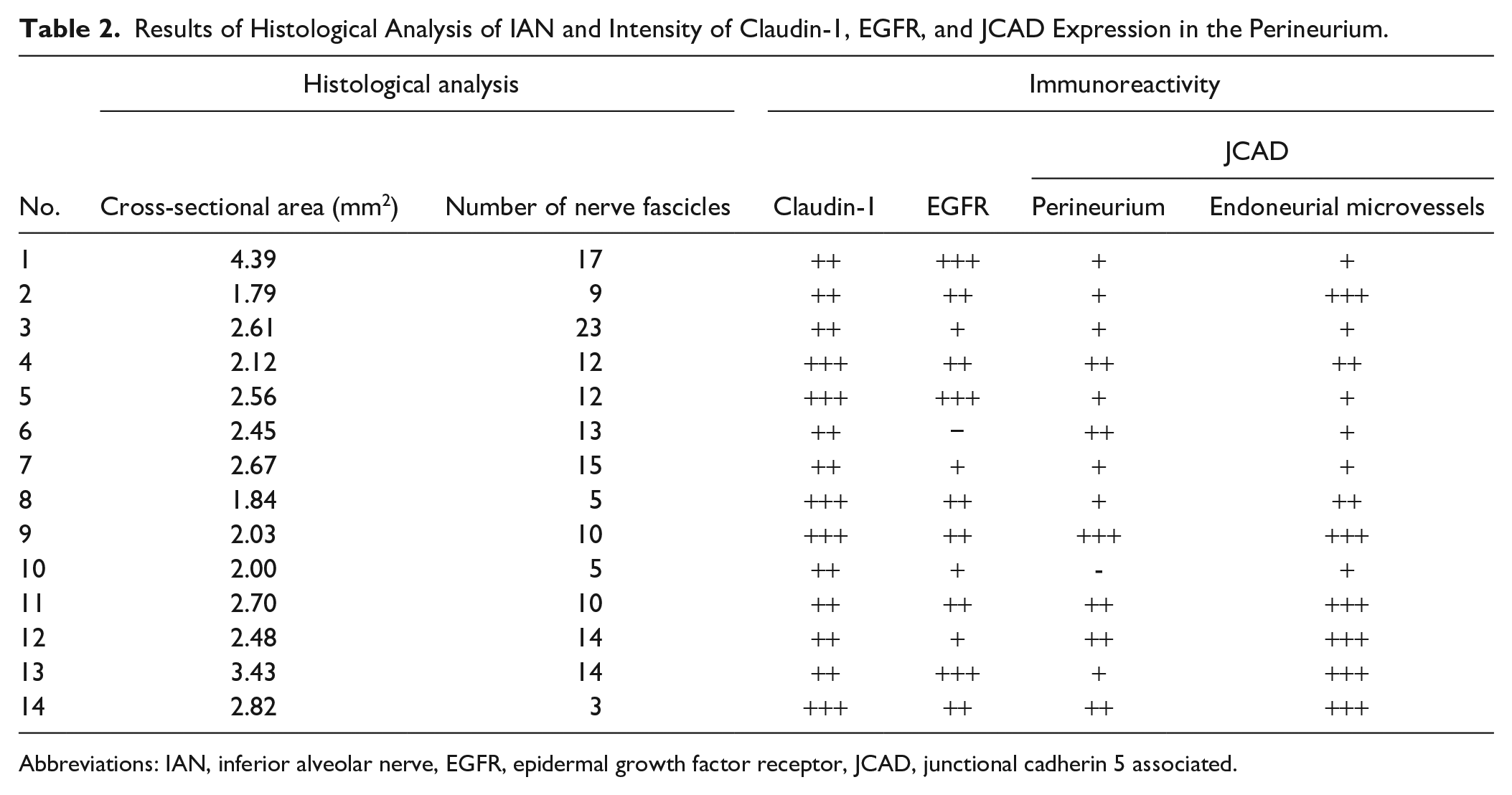
Abbreviations: IAN, inferior alveolar nerve, EGFR, epidermal growth factor receptor, JCAD, junctional cadherin 5 associated.
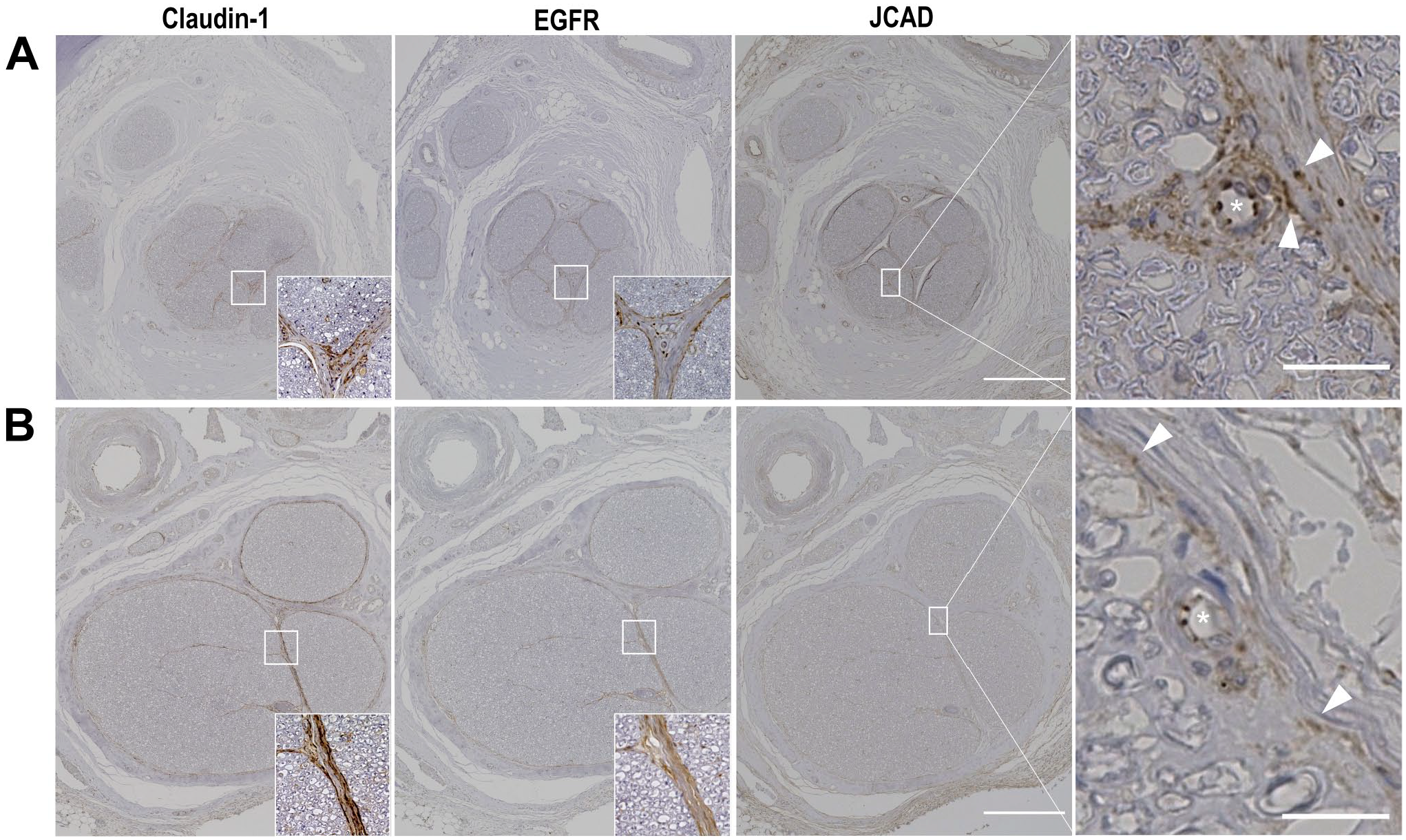
Representative image of the human inferior alveolar nerve. (A) No. 9. The signal intensities of claudin-1 and EGFR were strong and moderate, respectively. The signal intensity of JCAD was strong in endoneurial microvessels (asterisk) and perineurium (white arrowhead) in an enlarged view. (B) No. 14. The signal intensities of claudin-1 and EGFR were strong and moderate, respectively. The signal intensity of JCAD was strong in endoneurial microvessels (asterisk) and moderate in perineurium (white arrowheads) in an enlarged view. Scale bar sizes = 500 μm (20 μm in enlarged views). Abbreviations: EGFR, epidermal growth factor receptor, JCAD, junctional cadherin 5 associated.
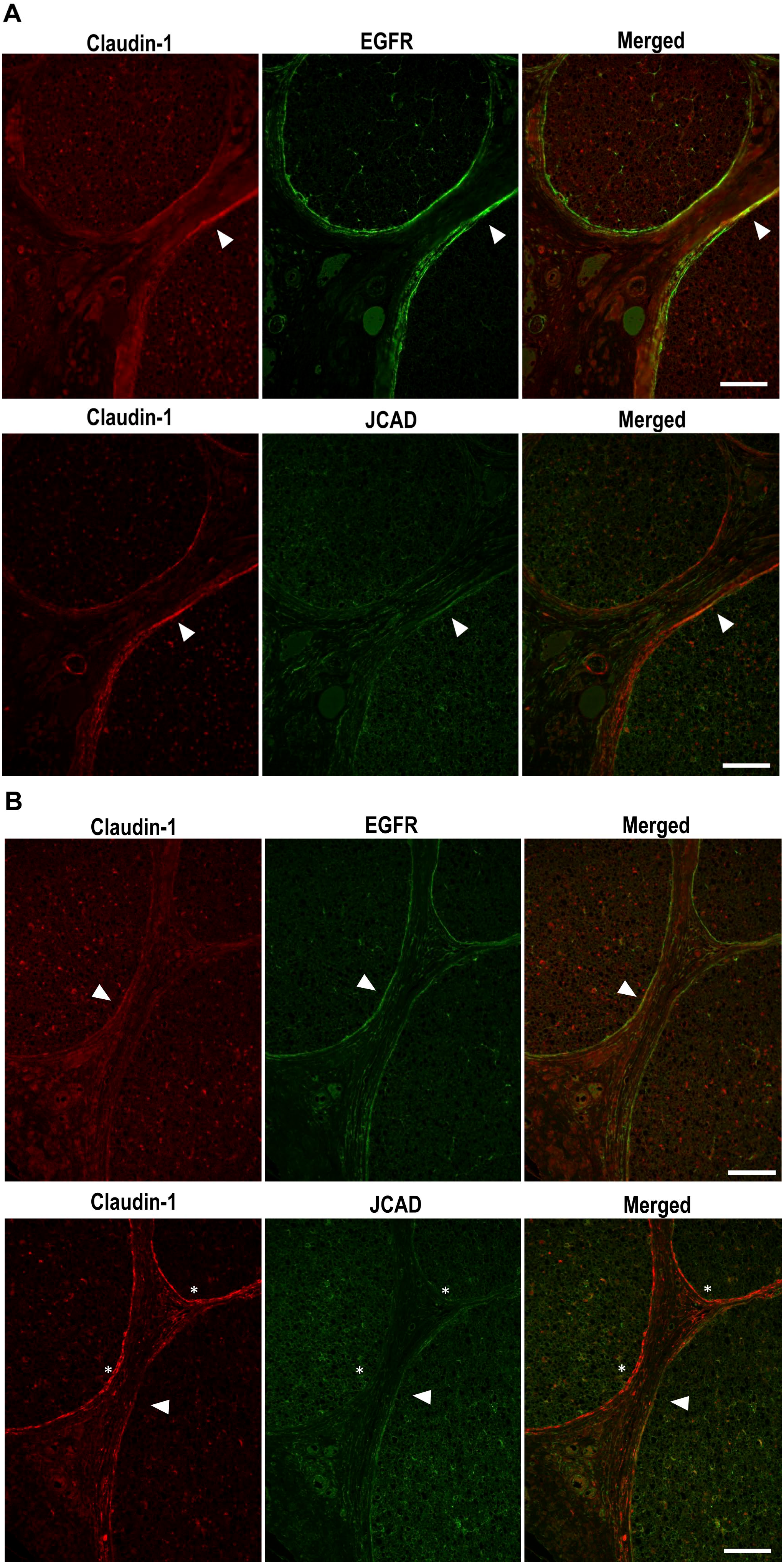
Representative double immunofluorescence image of the human inferior alveolar nerve. (A) No. 11. Colocalization (white arrowheads) was observed between claudin-1 (red) and EGFR (green) and between claudin-1 (red) and JCAD (green). (B) No. 14. Colocalization was observed between claudin-1 (red) and EGFR (green). Although colocalization (white arrowheads) was observed between claudin-1 (red) and JCAD (green), colocalization of claudin-1 and JCAD was not in some areas of perineurium (asterisks). Scale bar sizes = 100 μm. Abbreviations: EGFR, epidermal growth factor receptor, JCAD, junctional cadherin 5 associated.
Expression of JCAD in HPNCs
Next, we confirmed endogenous expression of JCAD in HPNCs. Perineurial cells are derived from cells morphologically resembling fibroblasts. 22 As shown in Fig. 3A, the shape of HPNCs was elongated, and dot-like cell–cell contacts with JCAD expression were prominent. Actin appeared to form stress fibers rather than cortical actin rings as commonly observed in epithelial cells. Endogenous expression of JCAD and EGFR was confirmed by western blotting (Fig. 3B).
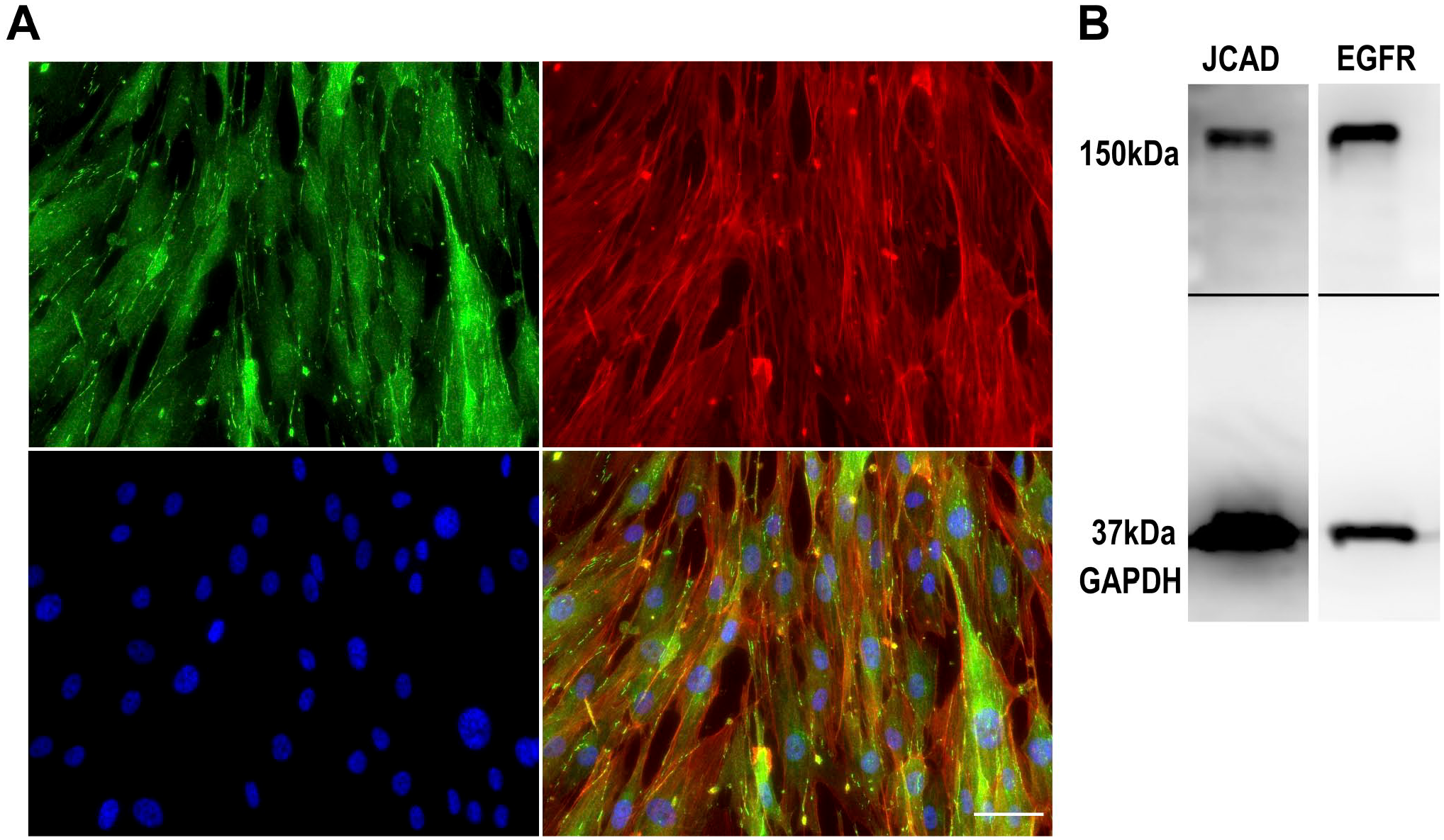
Expression of JCAD in cultured human perineurial cells. (A) Triple immunofluorescence staining of JCAD (green), actin (red), and DAPI (blue). Scale bar = 50 μm. (B) Immunoblot analyses of JCAD and EGFR in cultured human perineurial cells showed detectable bands of JCAD and EGFR. Abbreviations: JCAD, junctional cadherin 5 associated; DAPI, 4′,6-Diamidino-2-phenylindole; EGFR, epidermal growth factor receptor.
Effect of EGFR Inhibition on Cell–Cell Junctions of HPNCs
We analyzed the effect of EGFR inhibition on HPNCs to investigate the role of EGFR in the regulation of perineurial cell–cell junctions. Inhibition of EGFR phosphorylation by AG1478 changed the spindle-shaped fibroblastic morphology in control cells to a flattened morphology (Fig. 4A). Although EGFR inhibition did not change the total cell number or cell surface area, the degree of cell elongation was significantly changed in a dose-dependent manner (Fig. 4B) Notably, although scattered dot-like expression of JCAD at cell–cell contacts was prominent in control cells, the cells with long and linear expression of JCAD at cell–cell contacts appeared among AG1478-treated cells. To confirm this change, we calculated the ratio of JCAD-positive regions occupying cell–cell contacts per single cell (Fig. 5A). EGFR inhibition significantly increased the ratio of JCAD-positive cell–cell contacts of AG1478-treated cells compared with control cells (average ratio was 12.9% in control cells and 29.2% in 10-μM AG1478-treated cells).
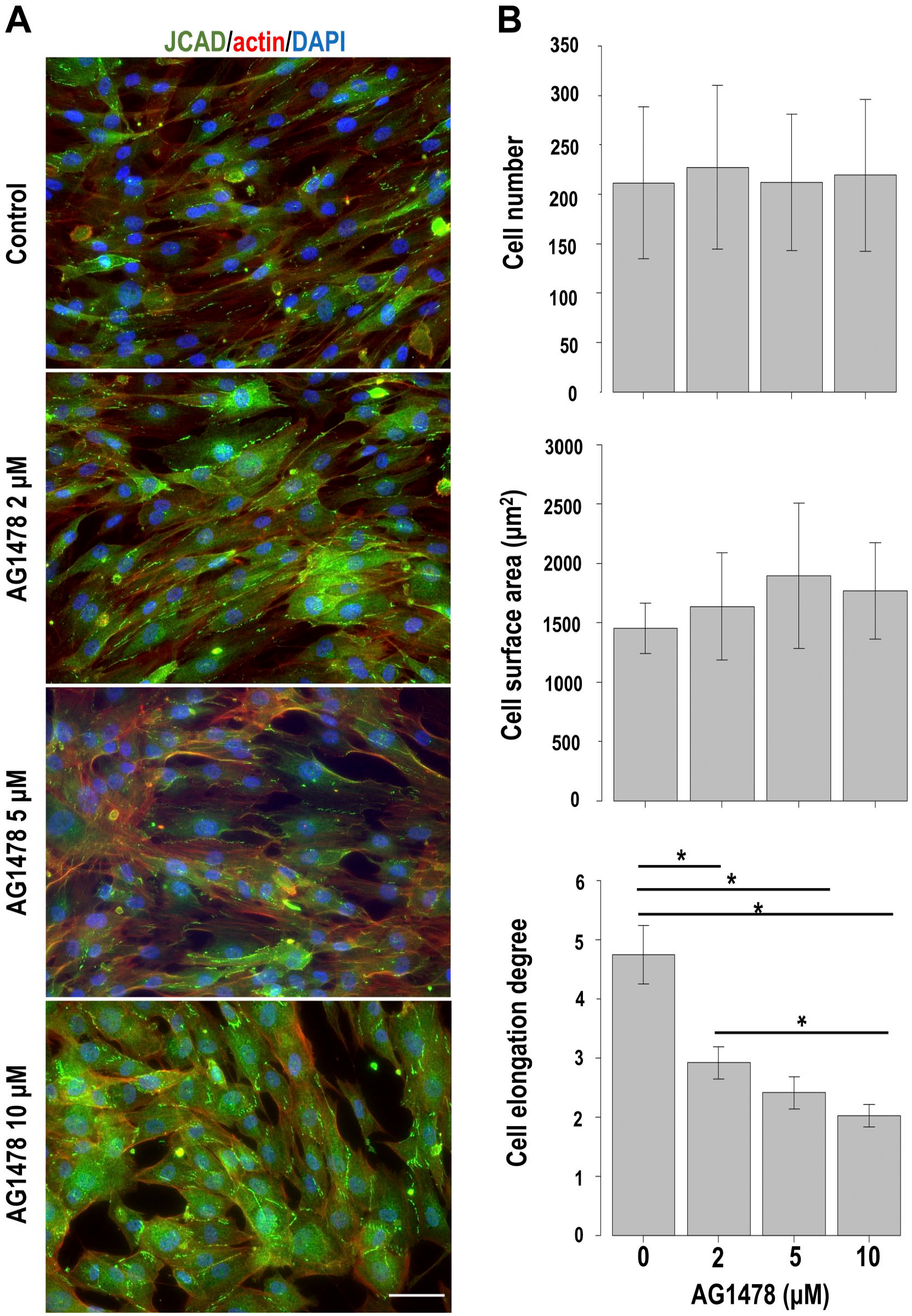
Changes induced by EGFR inhibition in cultured human perineurial cells. (A) Triple immunofluorescence staining of JCAD (green), actin (red), and DAPI (blue) revealed that EGFR inhibitor AG1478 changed the fibroblastic spindle cell morphology in the control to a squamous flattened cell morphology. Scale bar = 50 μm. (B) AG1478 treatment did not change the total cell number or cell surface area but significantly decreased the cell elongation degree (length of the long axis/length the short axis) in a dose-dependent manner. *Statistically significant difference among each concentration. Abbreviations: EGFR, epidermal growth factor receptor; JCAD, junctional cadherin 5 associated; DAPI, 4′,6-Diamidino-2-phenylindole.
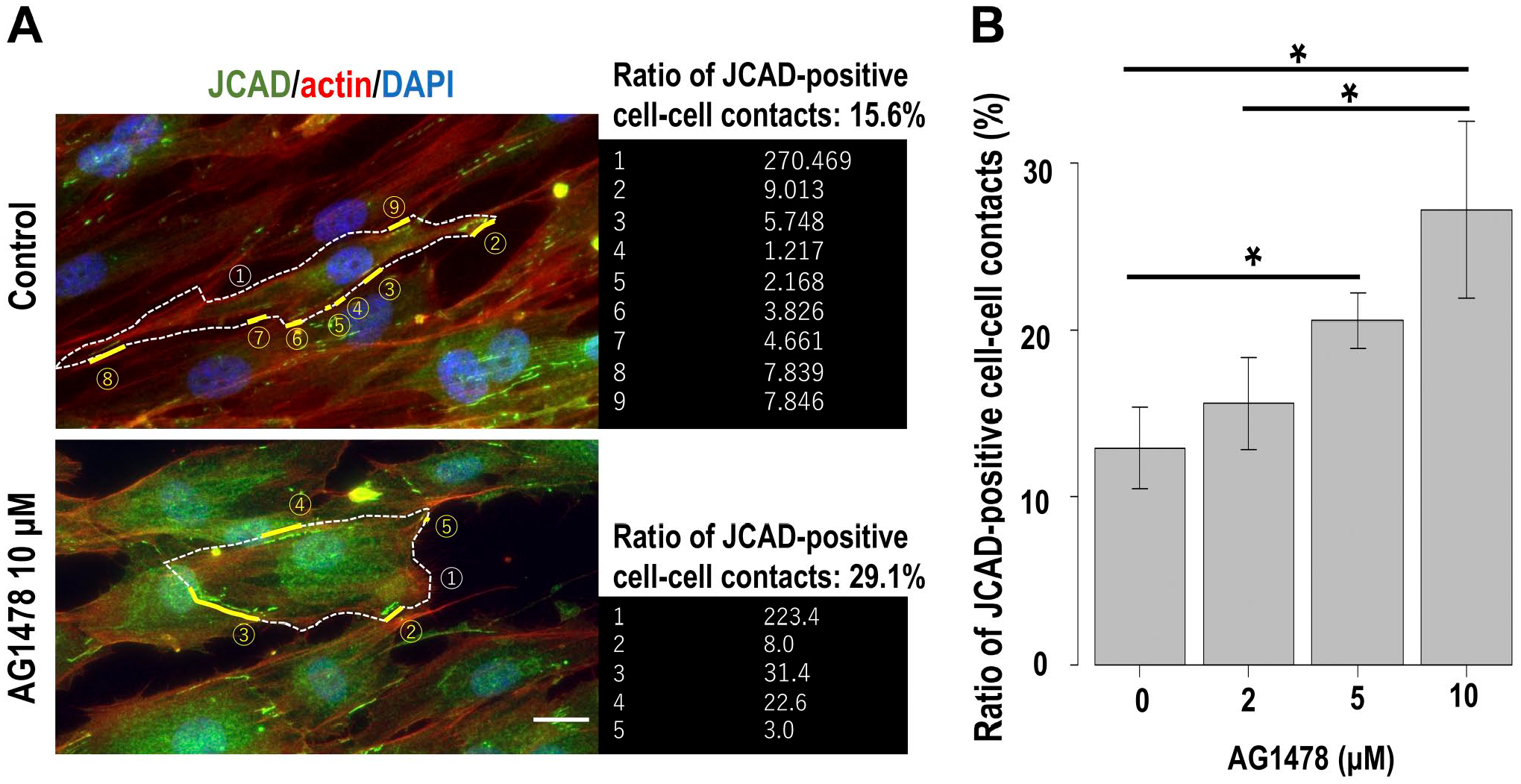
Effect of EGFR inhibition on cultured human perineurial cells. (A) Details of the calculation method of the ratio of JCAD (green)-positive cell–cell contacts. The ratio was defined by the length of JCAD-positive cell–cell contacts/the length of the entire circumference of the cell. Red: actin; blue: DAPI. Scale bar = 20 μm. (B) AG1478 treatment significantly increased the ratio of JCAD-positive cell–cell contacts. *Statistically significant difference among each concentration. Abbreviations: EGFR, epidermal growth factor receptor; JCAD, junctional cadherin 5 associated; DAPI, 4′,6-Diamidino-2-phenylindole.
Discussion
Although perineurium has an important role as a physical barrier of nerves, the detailed investigations in human tissues are rare. To our knowledge, this is the first study that histologically assessed the perineurium of IAN in human mandibles. Although previous studies have reported expression of JCAD 16 and EGFR 11 in the perineurium, no studies have focused on their expression in the perineurium of human IAN. JCAD, an intracellular component of endothelial cell–cell junctions, was strongly expressed in endoneurial microvessels, and its expression was also detected in perineurium of human IAN. Unlike claudin-1, JCAD and EGFR expression varied in each patient. JCAD and EGFR expression was confirmed in HPNCs. EGFR inhibition altered cell morphology and JCAD-positive cell–cell junctions of HPNCs.
A previous study has shown immunoreactivity of EGFR in the perineurium of human cutaneous nerves. 11 Studies have recently reported that EGFR inhibition improves neuropathic pain,12,13 and EGFR is expressed in the rat dorsal root ganglion. 14 We previously reported that EGFR inhibition (2 μM AG1478) changes the spindle shape of cultured human oral squamous cell carcinoma cells to a flattened epithelial cell-like squamous morphology and increases the number of E-cadherin-positive cell–cell junctions. 23 EGFR is involved in E-cadherin-mediated force transduction signaling and actin remodeling. 24 EGFR may also function in mechanosensitive signaling of perineurium cell–cell junctions. This study showed that the signal intensity of EGFR in perineurium varied in each patient compared with claudin-1, indicating that EGFR expression in perineurium of human IAN may be affected by unknown factors. Further investigations to identify unknown factors that affect EGFR expression in perineurium are necessary.
JCAD is an endothelial cell–cell component, and its function has been gradually elucidated. In vitro analysis of cultured human umbilical vein endothelial cells showed that JCAD gene expression increases in response to disturbed flow compared with unidirectional laminar flow. 25 JCAD gene expression in mice that undergo partial ligation of the left carotid artery increases in the left carotid artery compared with that in the right carotid artery, indicating that JCAD expression in blood endothelial cells also increases under disturbed laminar flow in vivo. 25 Recently, JCAD has been reported to possibly promote arterial thrombosis. 17 The JCAD level in plasma samples from patients with ST-elevation myocardial infarction was higher than that in controls. 17 Although these current studies suggest that JCAD promotes endothelial dysfunction and triggers expression of proinflammatory genes in blood endothelial cells, 25 the function of JCAD in perineurium is unknown. Endoneurial microvessels are in direct communication with circulating blood and are considered to be blood–nerve barriers, while the perineurium restricts passive diffusion of interstitial fluid from the epineurium into the endoneurium and vice versa. 26 Importantly, this study revealed that the JCAD expression level in endoneurial microvessels was mostly higher than that in the perineurium. This difference in JCAD expression in endoneurial microvessels and perineurium may reflect the function of JCAD in each tissue.
Similar to EGFR, the JCAD expression level varied in each patient. We did not find an apparent factor such as age that affected JCAD expression. However, a tendency of an opposing expression pattern (i.e., a higher expression level of EGFR in cases with a weaker level of JCAD and vice vasa) was found in some patients. In vitro, EGFR inhibition changed the fibroblastic spindle shape to a squamous flattened shape of HPNCs, which was similar to our previous study of oral cancer cells. 23 In this study, EGFR inhibition increased JCAD-positive cell–cell contacts. These effects may be a reason for the reduction of neuropathic pain because changes in blood–nerve barriers, composed of endoneurial microvessels and perineurium, contribute to neuropathic pain by permitting extravasation of endogenous blood-borne (hematogenous) nociceptive molecules and immune cells, such as macrophages and cytokines, through leaky blood–nerve barriers. 27 Therefore, further investigations of JCAD as a potential target for neuropathic pain are needed.
Finally, this study had some limitations. Our histological analysis included a small number of patients. The clinical impact of the expression of JCAD and EGFR on prevention, treatment, or outcome of diseases could not be shown. Furthermore, although we confirmed JCAD expression in endothelial microvessels in all cases as a positive control, the JCAD expression level in perineurium may be affected by the processes of immunohistological staining, such as decalcification. Another limitation is that most study samples were predominantly taken from patients with squamous cell carcinoma. It is unclear if these tumors affect the perineurium, even at a distance from the IAN. The same is true for EGFR expression. In addition, we did not elucidate the direct or indirect link between JCAD and EGFR. In pulmonary microvascular endothelial cells, phosphorylation of intercellular proteins, such as vascular endothelial cadherin and catenin, was induced in part through EGFR activation. 28 JCAD expression at cell–cell contacts depends on vascular endothelial cadherin (VE-cadherin). 16 Importantly, VE-cadherin is expressed in human perineurium. 29 Taken together, JCAD may be involved in the interaction between VE-cadherin and EGFR. Further investigations to identify the link between JCAD, VE-cadherin, and EGFR in perineurial cell–cell junctions are necessary.
In conclusion, the perineurium of IAN in human mandibles expressed JCAD and EGFR. JCAD was also strongly expressed in endoneurial microvessels in human IAN. JCAD expression was confirmed in HPNCs. EGFR inhibition changed cell morphology and cell–cell contacts with JCAD expression.
Footnotes
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
YH managed the execution of the study; collected human mandibular bone samples; carried out imaging experiments of the samples; and performed in vitro experiments and statistical analysis. MM supervised and supported in vitro experiments. YK, DT, AK, and TH supported the collection of human samples and supervised data analysis. MS supervised pathological analysis of human samples. MA conceived the idea of the study, was involved in the design and coordination of the study, planned the methods, and drafted the manuscript. All authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by JSPS KAKENHI Grant Number 22K17155.
