Abstract
Cutaneous neurofibromas consist of axonal processes, Schwann cells, fibroblasts, perineurial cells, mast cells, and abundant extracellular matrix. The distribution and role of perineurial cells in neurofibromas has been uncertain, partly because there has not been a specific immunohistochemical marker for perineurial cells. In this study, tight junctions (TJs) of 16 neurofibromas from 12 patients with neurofibromatosis type 1 (NF1) were analyzed using electron microscopy, immunohistochemistry, and Western transfer analysis. Cell-cell contacts with typical ultrastructural morphology of TJs were seen between adjacent perineurial cells surrounding the small nerves and between contacting perineurial cell processes embedded in tumor stroma. Immunohistochemistry showed expression of claudin-1, claudin-3, and ZO-1 in the intercellular junctions of a subpopulation of tumor cells. Occludin was present mainly in perineurium and claudin-5 localized to the blood vessels. Double immunolabelings were used to identify the cell types expressing claudin-1. The results showed that claudin-1 positive cells were also positive for type IV collagen and epithelial membrane antigen but not for S-100 protein. This labeling pattern is consistent with perineurial cell phenotype. Using claudin-1 as a marker, our results showed that clusters of perineurial cells are distributed around the rudimentary nerves within cutaneous neurofibromas and at the periphery of some neurofibromas.
Keywords
N
Cutaneous neurofibromas are hamartomatous tumors arising from the connective tissue of peripheral nerves. Histologically they are composed of Schwann cells, fibroblasts, perineurial cells, axonal processes, and mast cells that are embedded in abundant collagenous extracellular matrix (Lassmann et al. 1976; Peltonen et al. 1988). Experiments with transgenic mice have suggested that the formation of neurofibromas requires loss of both NF1 alleles in Schwann cells in NF1+/- background (Zhu et al. 2002). It has been speculated that mast cells would create a cytokine-rich microenvironment that could be permissive for neurofibroma growth (Zhu et al. 2002).
Perineurial cells have been recognized in neurofibromas using morphologic criteria such as long slender cell processes, fragmented basement membrane, and numerous intracellular vesicles (Lassmann et al. 1976; Erlandson 1985). Overlapping processes of adjacent cells are occasionally joined by tight junction (TJ)-like cell contacts (Smith and Bhawan 1980; Erlandson and Woodruff 1982).
Several protein markers such as epithelial membrane antigen (EMA), Glut-1, and type IV collagen have also been used to identify perineurial cells in neurofibromas (Perentes et al. 1987; Ariza et al. 1988; Peltonen et al. 1988; Hirose et al. 2003; Ide et al. 2004). The lack of a single characteristic marker protein has, however, made the recognition of perineurial cells challenging. Consequently, the role of perineurial cells in the development and growth of neurofibromas has been difficult to demonstrate. TJ protein claudin-1 has been suggested to be a perineurial cell marker in perineuriomas (Folpe et al. 2002; Rankine et al. 2004) and neurofibromas (Folpe et al. 2002). Folpe et al. (2002) showed that six out of nine cases of neurofibromas contained claudin-1-positive cells. These cells were generally located at the periphery of the neurofibromas, particularly in plexiform neurofibromas where the claudin-1-positive cells had long, delicate cytoplasmic processes and thus were considered to be perineurial in origin.
TJs form continuous, circumferential seals around cells in epithelia. In transmission electron microscopy, outer plasma membrane leaflets of the adjacent cells appear to fuse to form TJs (Farquhar and Palade 1963). The molecular composition and functions of tight junctions have recently been extensively investigated (Tsukita et al. 2001; Gonzalez-Mariscal et al. 2003; Van Itallie and Anderson 2004). The results emphasize the dynamic regulatory role of TJs in cell proliferation, cell polarity, and tumor suppression (Matter and Balda 2003; Sasaki et al. 2003; Schneeberger and Lynch 2004). The growing number of characterized TJ components can be divided into three categories: (1) integral TJ proteins such as occludin and claudin family that contribute to the permeability barrier; (2) TJ plaque proteins, for example, ZO-1, which is a peripherally associated scaffolding protein that appears to organize the transmembrane proteins and couple them to other cytoplasmic proteins and to actin filaments; (3) miscellaneous group of cytoplasmic and nuclear proteins including tumor suppressors and transcriptional and posttranscriptional factors (Schneeberger and Lynch 2004).
We have recently characterized the expression of TJ components on developing and adult human peripheral nerve (Pummi et al. 2004). The results showed that claudin-1, claudin-3, ZO-1, and occludin localized to the mature perineurium. The purpose of the present study was to elucidate the distribution of TJ proteins in NF1-related cutaneous neurofibromas. Furthermore, we characterized neoplastic perineurial cells by using double immunohistochemistry for claudin-1 with EMA and S-100.
Materials and Methods
Tissue Samples
Formalin-fixed, paraffin-embedded cutaneous neurofibromas from eight NF1 patients were retrieved from the Department of Pathology, Turku University Hospital, Turku, Finland and were used for avidin-biotin labelings. Neurofibromas from six NF1 patients were obtained from the Department of Dermatology, Turku University Hospital and were used for indirect immunofluorescence labeling (IIF), Western transfer analysis, and transmission electron microscopy. Samples of great auricular nerve were obtained for Western transfer analysis from three tumor patients who underwent parotidectomy. The great auricular nerve had to be sacrificed due to surgical reasons in the Department of Otorhinolaryngology-Head and Neck Surgery, Turku University Hospital. Control skin sample for Western transfer analysis was obtained from plastic surgical operation in the Department of Surgery, Turku University Hospital. All tissue samples were obtained with the appropriate informed consent and approval of the Joint Ethical Committee of the Turku University Hospital and the University of Turku, Finland.
Primary Antibodies
The following primary antibodies were used: affinity-purified rabbit polyclonal antibodies to human claudin-1 (51–9000), claudin-3 (34–1700), occludin (71–1500), and to cow S–100 (18–0046) from Zymed Laboratories Inc. (South San Francisco, CA) mouse monoclonal antibodies to human claudin-5 (35–2500), and ZO-1 (33–9100; Zymed) mouse monoclonal antibodies to human type IV collagen (M785), EMA (M0613), and neurofilament (M762) from DAKO A/S (Glostrup, Denmark).
Western Transfer Analysis
Neurofibroma and great auricular nerve samples were homogenized in RIPA buffer (1% Nonidet-40, 0.5% sodium deoxycholate, and 0.1% SDS in PBS) supplemented with protease inhibitors (Complete, Mini Protease Inhibitors Cocktail Tablets; Roche Diagnostics GmbH, Mannheim, Germany). The epidermis was separated from the dermis and homogenized as described earlier (Peltonen et al. 1999). Protein concentrations of soluble fraction were measured with Bio-Rad DC Protein Assay (Bio-Rad Laboratories; Hercules, CA). Twenty μg of each protein sample was run on a 12.5% SDS polyacrylamide gel. After electrophoresis, proteins were transferred to Hybond ECL nitrocellulose membrane (Amersham Pharmacia Biotech; Buckinghamshire, UK) and immunolabeled with claudin-1 antibody in 3% BSA-PBS-0.5% Tween 20 overnight at 4C. Peroxidase-linked donkey-anti-rabbit (NA 934) (Amersham International) was used as the secondary antibody. Proteins were detected with ECL (Amersham Biosciences), and the filter was exposed to autoradiographic film (Hyperfilm ECL; Amersham Pharmacia Biotech).
Indirect Immunofluorescence Labeling
Neurofibroma samples were snap frozen in liquid nitrogen. Seven-μm-thick cryosections were cut on silanated glass slides and fixed in 100% methyl alcohol at −20C for 10 min. To prevent nonspecific binding, the samples were preincubated in 1% BSA-PBS for 15 min. Primary antibodies were diluted in 1% BSA-PBS and incubated on the samples at 4C for 20 hr. Following six 5-min washes in PBS, the samples were incubated with secondary antibodies at 20C for 1 hr. The samples were washed in PBS and mounted with Glycergel (DAKO). Secondary antibodies for IIF were tetramethyl-rhodamine isothiocyanate (TRITC)-conjugated swine anti-rabbit (R0156) from DAKO and Alexa Fluor 488-conjugated goat anti-mouse IgG (A-11029) from Molecular Probes Inc. (Eugene, OR). Skin covering the neurofibromas served as a positive control in IIF and avidin-biotin immunolabelings for TJ proteins. ZO-1, occludin, and claudin-1 were localized to epidermis covering the neurofibromas as previously described for normal human epidermis (data not shown) (Pummi et al. 2001; Brandner et al. 2002). In negative control reactions, primary antibodies were replaced with 1% BSA-PBS, and only faint background fluorescence was observed.
Avidin-Biotin Labeling
Formalin-fixed and paraffin-embedded neurofibroma specimens were immunolabeled with avidin-biotin method. The sections were cut on SuperFrost Plus microscope slides (Menzel-Gläser; Braunschweig, Germany), deparaffinized, and hydrated in a descending ethanol series. To retrieve TJ antigens, the samples were boiled for 15 min in a microwave oven in 10 mM Tris, 1 mM EDTA, pH 9, and subsequently cooled in the same solution at room temperature for 15 min. Endogenous peroxidase activity was quenched by treating the sections in 0.3% H2O2 for 30 min. To prevent nonspecific binding, the sections were incubated in horse serum diluted in PBS. Primary antibodies were diluted in 1% BSA-PBS and incubated on the samples overnight at 4C. The bound antibodies were visualized using appropriate avidinbiotin peroxidase kit (Vectastain; Vector Laboratories, Burlingame, CA) with 3.3'-diaminobenzidine tetrahydrochloride (DAB) as the chromogen (DAB peroxidase substrate kit; Vector Laboratories). In double labeling, the sections were first incubated with one primary antibody that was detected using avidin-biotin peroxidase kit with DAB as the chromogen. Thereafter, the sections were treated for 1 hr with 0.1 M Na-citrate buffer, pH 3.2, and subsequently washed three times with 0.1 M Millonig's phosphate buffer, pH 3.2, and three times with 0.1 M Millonig's phosphate buffer, pH 7.4. After washing with PBS and blocking with diluted horse serum, second primary antibody was incubated on the samples overnight at 4C. Immunoreaction was visualized using avidin-biotin alkaline phosphatase kit (Vectastain; Vector Laboratories) and Vector red alkaline phosphatase substrate kit I (Vector Laboratories). Endogenous phosphatase activity was blocked with levamisole (Vector Laboratories). Sections were counterstained with Mayer's hematoxylin (Oy Reagena LTD; Kuopio, Finland). In negative control reactions primary antibody was replaced with 1% BSA-PBS (Figure 3F).
Microscopy
Avidin-biotin labelings were examined and photographed using a Leica DMRB microscope, equipped with a Leica DC 300F camera and Leica IM50 (version 1.20) software (Leica Microsystems AG; Heerbrugg, Switzerland). Resolutions of images were 1950 × 1545 pixels and images were saved in tif format.
Confocal laser scanning microscopy was carried out using a Zeiss LSM 510 META confocal microscope equipped with argon-ion and helium-neon lasers (Zeiss; Jena, Germany) and LSM 3.0 software. The objective magnification used was ×40 (oil immersion, numeric aperture 1.3), ×63 (oil immersion, numeric aperture 1.4) or ×100 (oil immersion, numeric aperture 1.4). For excitation of Alexa Fluor 488, the 488-nm line was used and for TRITC, the 543-nm line was used. The beampath for Alexa Fluor 488 contained a 488-nm main dichroic mirror and a 500-530IR-nm bandpass filter. The beampath for TRITC contained a 543-nm main dichroic mirror and a 560-nm long-pass filter for detection of the emitted fluorescences. The resolutions of original images were 1024 × 1024 pixels, and images were saved in tif format.
Transmission Electron Microscopy
For ultrastructural studies, 1 × 1 mm pieces of neurofibroma tissue were fixed in 5% glutaraldehyde in s-collidine buffer and postfixed in 1% OsO4 and 1.5% potassium ferrocyanide. Samples were then dehydrated and embedded in Epon 812. Thin sections were cut on coated copper grids and stained with uranyl acetate and lead citrate. Samples were examined and photographed using JEOL 1200EX electron microscope (JEOL; Tokyo, Japan).
Results
To evaluate the distribution of the TJ proteins in neurofibromas, avidin-biotin labelings for ZO-1, occludin, claudin-1, −3, and −5 were carried out. In all neurofibromas investigated, immunohistochemistry revealed claudin-1-positive zones in tumor tissue otherwise mostly negative for claudin-1. In addition to claudin-1-positive zones within the tumor, one third of the tumors expressed claudin-1 also at the periphery of tumor tissue (Figures 1A and 1B). High magnification showed claudin-1 at the cell–cell borders of adjacent tumor cells. The labeling pattern was most intense near the nerve fascicles and consisted of closely located spots in rows (Figures 2B and 2C). Labeling changed to a more punctate pattern when distanced from nerves, suggesting that there were fewer claudin-1-containing intercellular contacts. Specificity of the immunoreactions was assessed by Western transfer analysis (Figure 1C) using epidermis and peripheral nerve as references.
The small, seemingly normal nerve fascicles that traversed the neurofibromas expressed claudin-1, claudin-3, ZO-1, and occludin in perineurium (Figures 2A, 2D, 2G, and 2J), which is in agreement with that reported for normal sciatic nerve (Pummi et al. 2004). In the tumor tissue, the distribution of claudin-3 and ZO-1 was apparently the same as that of claudin-1 (Figures 2D–2I). In addition, ZO-1 was prominently expressed in blood vessels. Double immunofluores-cence labeling for claudin-1 and ZO-1 was used to further investigate the presence of intercellular junctions between neighboring tumor cells. Confocal microscopy revealed partial colocalization of claudin-1 and ZO-1 (Figure 3A). Of other TJ proteins, occludin was seen mainly in perineurium and blood vessels but was not detectable between tumor cells (Figures 2J–2L). Claudin-5 preferably labeled the blood vessels (Figure 2N).
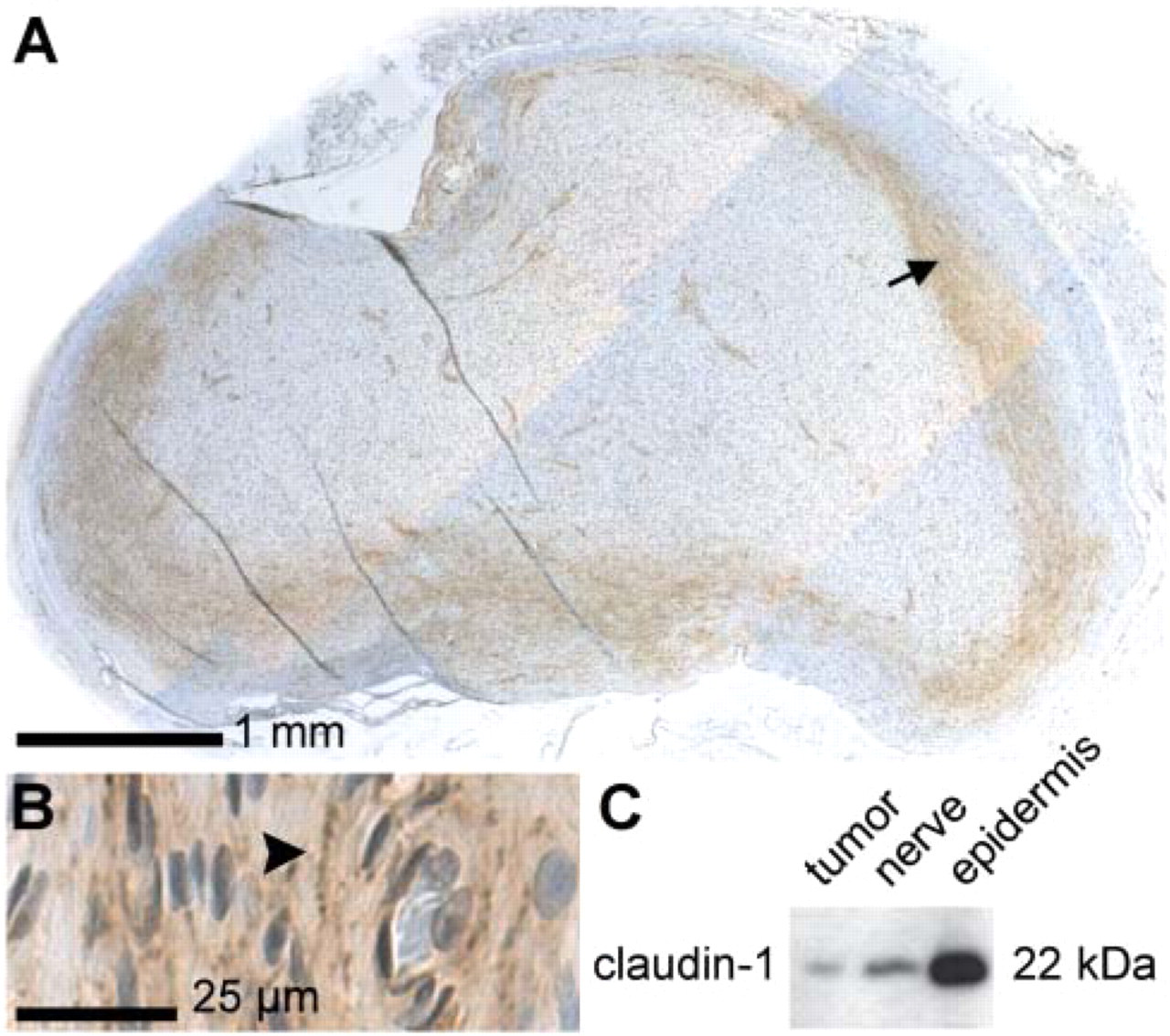
In some neurofibromas claudin-1 labeling was located at the periphery of tumor tissue, thus giving an appearance of a capsule
Double immunohistochemistry was performed to further investigate the cell types expressing TJ proteins. In particular, the identity of densely packed claudin-1-positive cells was studied in more detail. Double immunolabeling with claudin-1 and neurofilament antibodies showed that clusters of claudin-1-positive cells were located in the vicinity of small nerve fascicles, rudimentary nerves, or even single axons traversing the tumor (Figure 3B). Double immunolabeling with claudin-1 and type IV collagen antibodies revealed that claudin-1 was expressed in closely apposed cells positive for type IV collagen (Figure 3C). Earlier studies have shown that both Schwann cells and perineurial cells express basement membrane collagen, type IV, in neurofibroma (Peltonen et al. 1988). Thus, the claudin-1-expressing cells could be either Schwann cells or perineurial cells. To distinguish Schwann cells from perineurial cells in neurofibroma, S–100 labeling was utilized. Double immunolabeling for claudin-1 and S–100 revealed that the clusters of claudin-1-positive tumor cells were mainly negative for S–100 (Figure 3D). Earlier studies have suggested that EMA is specific for perineurial cells and has been used to identify perineurial cells in peripheral nerve sheath tumors (Perentes et al. 1987; Ariza et al. 1988; Hirose et al. 2003; Ide et al. 2004; Rankine et al. 2004). Confocal microscopy of double immunofluorescence labeling between claudin-1 and EMA showed that cells expressing claudin-1 were positive for EMA (Figure 3E) and were concluded to represent perineurial cells.
Transmission electron microscopy was performed to reveal the morphology of intercellular contacts between perineurial cells. At the ultrastructural level, perineurial cells were identified by their long, slender cell processes, discontinuous basement membrane, and numerous pinocytotic vesicles (Lassmann et al. 1976) or their tendency to encircle groups of Schwann cells. Cell-cell contacts with typical morphology of TJs were seen between adjacent perineurial cells surrounding the small nerves and between contacting perineurial cell processes embedded in tumor stroma (Figure 4). In addition, adherens-like junctions were demonstrated between tumor cells (Figures 4D and 4E).
Discussion
The expression of TJ proteins was investigated to assess the molecular composition of TJs in neurofibroma and to evaluate the potential use of TJ proteins for recognition of perineurial cells in neurofibromas. The results of the present study demonstrated cell-cell contacts with ultrastructural characteristics of TJs between a subpopulation of neurofibroma cells. Morphological and immunocytochemical characteristics suggested that the cells connected by TJs were neoplastic perineurial cells. Immunolabelings showed that claudin-1 was more abundantly expressed in neurofibromas compared with claudin-3, claudin-5, ZO-1, and occludin, as estimated by the tissue distribution and the intensity of labeling. Double labeling demonstrated that claudin-1 and ZO-1 displayed a partial colocalization. It is feasible to suggest that these spots of colocalization represent TJs observed at the electron microscopic level in perineurial cells, and that perineurial cell TJs in neurofibromas contain predominantly claudin-1, claudin-3, and ZO-1. Occludin was not detectable between tumor cells and may be lacking from the TJs observed in neurofibromas. This is in contrast to normal human peripheral nerve where occludin is readily detected between perineurial cells (Pummi et al. 2004). These findings suggest that neoplastic perineurial cells retain their capacity to form TJ-like junctions, which may not contain all of the same components as typical TJs. The expression of TJ proteins can be seen as a sign of differentiation capacity of tumor perineurial cells and may be associated with benign phenotype and often slow growth of cutaneous neurofibromas. Up- or downregulation of TJ protein expression has been reported in several tumor types, and the expression pattern seems to be tumor cell-type specific (Swisshelm et al. 2005). For example, claudin-3 and −4 are expressed at high levels in ovarian cancer, and claudin-1 is expressed at low levels in glioblastomas (Hough et al. 2000; Liebner et al. 2000).
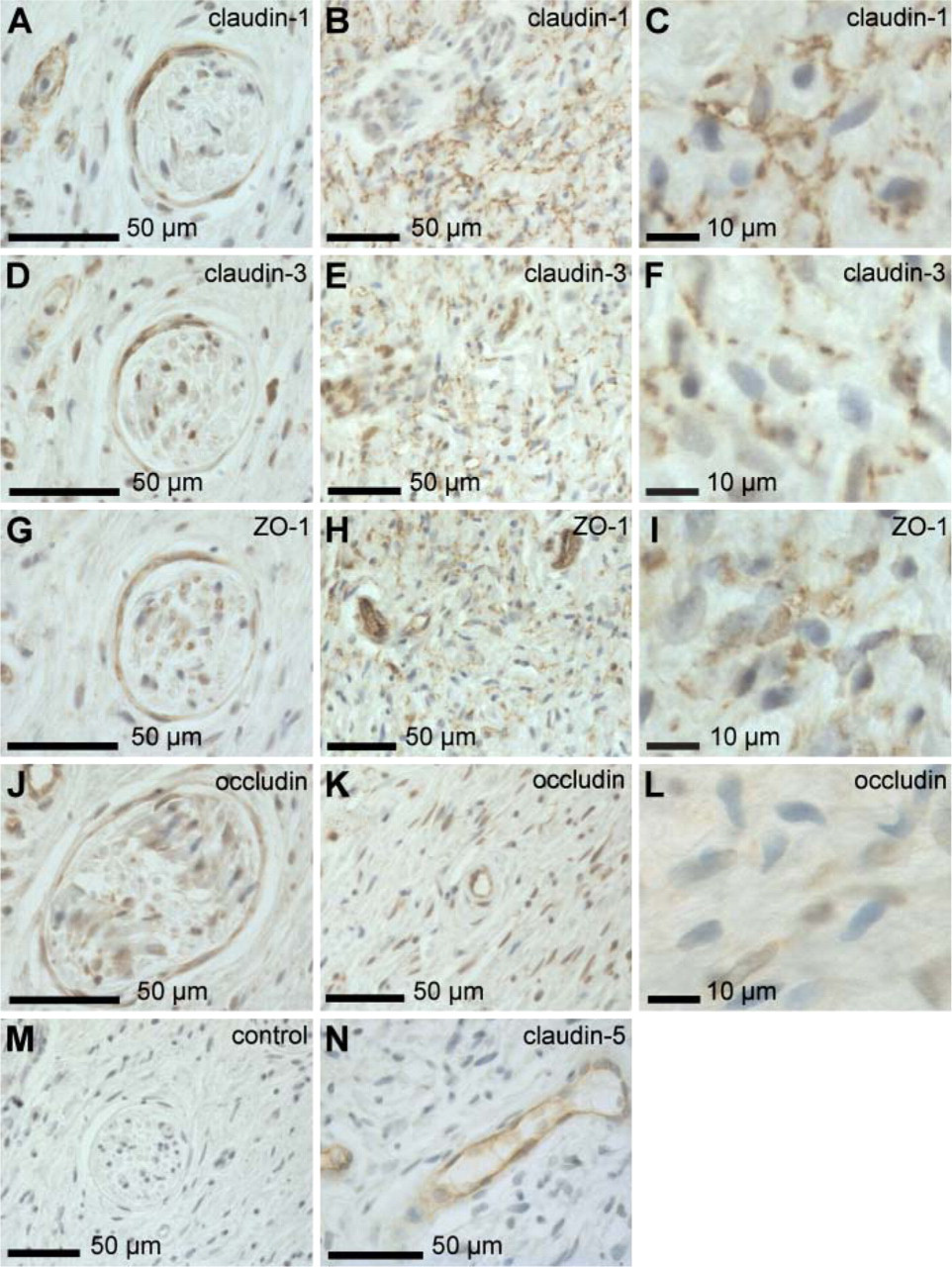
Immunolabeling of neurofibroma tissue using antibodies to claudin-1, −3, −5, ZO-1, and occludin. Formalin-fixed, paraffin-embedded cutaneous neurofibromas were immunolabeled with antibodies to tight junction (TJ) proteins using avidin-biotin method. Small nerve fascicles traversing the neurofibroma expressed claudin-1, claudin-3, ZO-1, and occludin in perineurium
The relative number, distribution, and role of perineurial cells in neurofibromas have been difficult issues to assess, partly due to a lack of a specific marker molecule for perineurial cells. The findings of the present and of a previous study (Folpe et al. 2002) suggest that claudin-1 may be highly useful for the identification of perineurial cells in neurofibromas. Double immunolabelings of our study showed that the claudin-1-positive cells were also positive for type IV collagen and EMA but not for S–100 protein. This labeling pattern is consistent with perineurial cell phenotype. Claudin-1 has been shown to be expressed in myelinating Schwann cells between membrane lamellae of the same cell (autotypic junctions) (Poliak et al. 2002). The finding that claudin-1-positive areas in neurofibromas were negative for S–100 suggests that neoplastic Schwann cells do not express claudin-1 in these tumors.
Using claudin-1 as a marker, we showed that (a) perineurial cells are present outside structured nerves mixed with other tumor cells, (b) neoplastic perineurial cells are clustered in densely packed groups around the rudimentary nerves of the cutaneous neurofibromas, and (c) perineurial cells are located at the periphery of some neurofibromas, surrounding the tumor like a capsule. Development of cutaneous neurofibromas is associated with disintegration of nerve structure. The pathogenesis of neurofibromas includes the escape of Schwann cells from the endoneurial space and subsequent proliferation outside the blood nerve barrier (Peltonen et al. 1984). The prerequisite for the Schwann cell proliferation outside the endoneurial space is the disruption of perineurium. In addition, this cascade of events may include dysfunction of multiple cell types including mast cells. This is in part evidenced by results obtained using NF1 gene-mutated mice: development of neurofibromas requires mutations in both NF1 alleles (NF1−/-) of Schwann cells and heterozygosity (NF1+/-) of the other cell types (Zhu et al. 2002). We speculate that the NF1+/- genotype in human neurofibromatosis may result in altered stability of cell-cell junctions including TJs in perineurial cells and in subsequent loss of cohesion of the perineurium. Mast cells may have an additional role in the disruption of perineurium because a mast cell protease has been shown to affect the permeability of TJs by changing the localization of occludin (Scudamore et al. 1998).
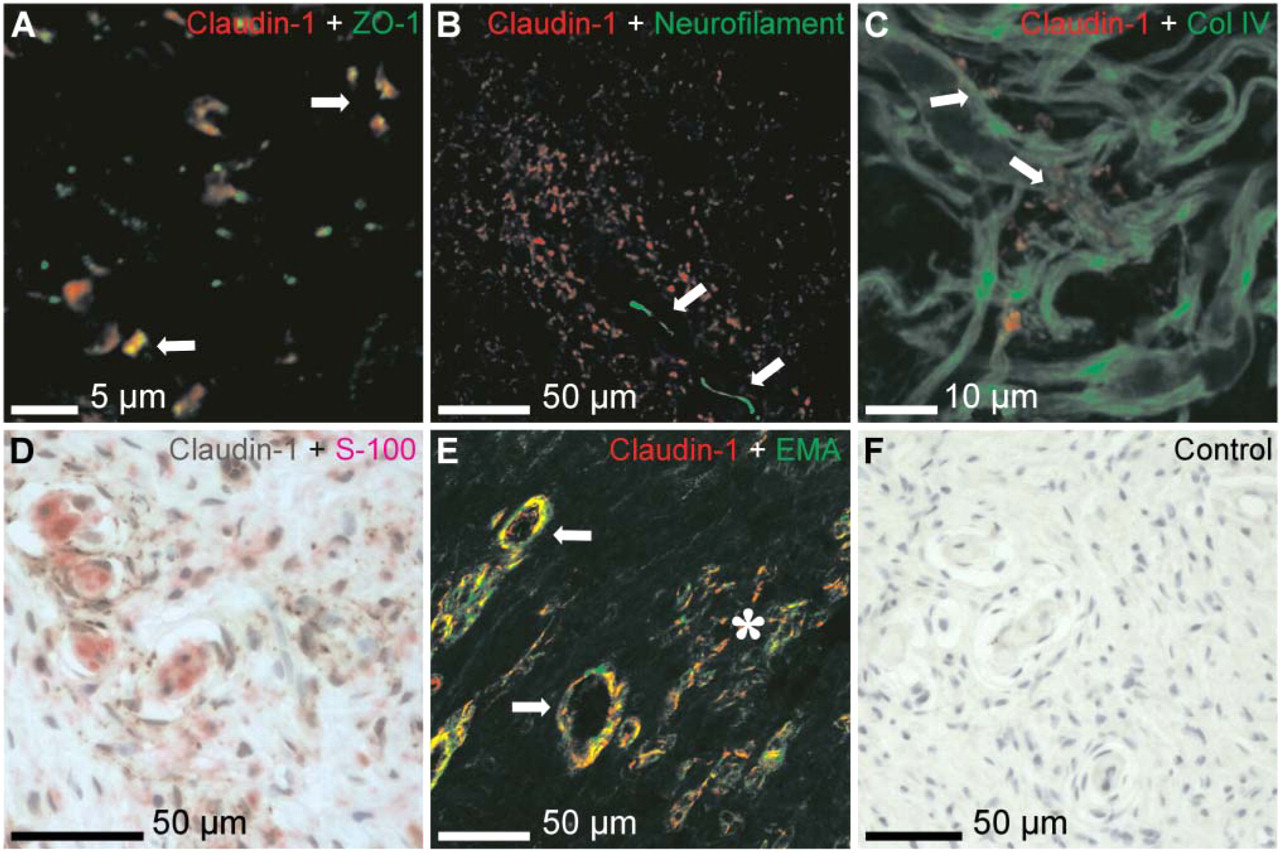
Identification of cell types expressing claudin-1 in neurofibroma. Cryosection of neurofibroma, double immunofluorescence labeling with claudin-1 (red) and ZO-1 (green) showed partial colocalization (arrows)
The role of TJs in neurofibroma formation can only be speculated. Perineurial cells are able to produce several ECM molecules, thus contributing to the accumulation of collagenous extracellular matrix in neurofibromas (Jaakkola et al. 1989a,b). In cancer, the loss of cell polarity and breakdown of TJ barrier functions may enable the growth factors and cytokines to freely penetrate from the surrounding tissue, thus promoting cell proliferation and tumor growth (Mullin 2004; Swisshelm et al. 2005). It can be speculated that disintegration of perineurium might also allow growth factors to stimulate perineurial cell proliferation, ECM production, and neurofibroma growth.
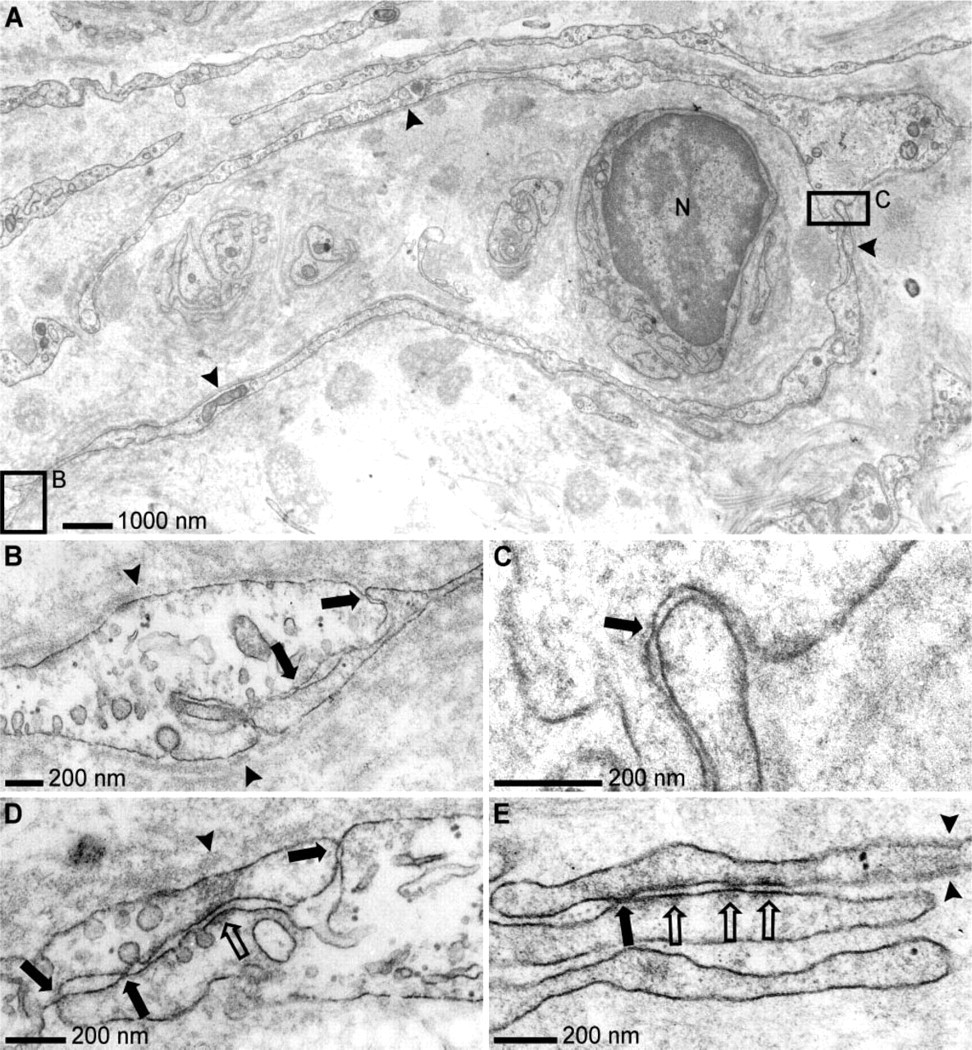
Transmission electron microscopic analysis of neurofibroma, uranyl acetate, and lead citrate counterstain
Ras has been shown to downregulate TJ functions by regulating phosphorylation of occludin and ZO-1 but not claudin-1 (Chen et al. 2000). As NF1 gene encodes neurofibromin, a downregulator of Ras, inactivation of NF1 genein NF1-related neurofibromas might result in elevated Ras activity and altered TJ function. This might participate in the disruption of perineurium.
Our results using claudin-1 as a marker showed that the distribution of perineurial cells in neurofibromas is not random. Instead, clusters of perineurial cells were found around the rudimentary nerves within cutaneous neurofibromas and at the periphery of some but not all neurofibromas. In conclusion, claudin-1 is a useful marker for identifying perineurial cells in neurofibromas.
Footnotes
Acknowledgements
This study was supported by the Academy of Finland, Finnish Cancer Societies, the Finnish Society of Dermatology, the Paijat-Hame Fund of the Finnish Cultural Foundation, the Turku University Foundation, Turku University Central Hospital (Turku, Finland), and University Hospital of Oulu (Oulu, Finland).
We thank Professor Reidar Grénman, MD, PhD, Department of Otorhinolaryngology-Head and Neck Surgery, Turku University Hospital, for providing the peripheral nerve samples. The expert technical assistance of Mrs. Heidi Pakarinen is greatly acknowledged.
